2
Salmon Life History and Ecology
INTRODUCTION
Atlantic salmon (Salmo salar) once spawned in northern hemisphere rivers from Long Island Sound, New York, to arctic regions in the western and eastern North Atlantic, the Barents Sea, the Baltic Sea, and south to Spain and Portugal. The historical range has contracted northward in the past century (for the U.S. population, see Baum 1997; for current distribution, see also Collette and Klein-McPhee 2002). Climate warming may be a large factor in range contraction due to temperature and other weather-related effects on lacustrine and coastal marine conditions (see discussion by Dickson and Turrell 2000), but overexploitation of the salmon fishery and loss of habitat due to human activities (for example, dam construction, pollution, stream siltation, and introduction of nonnative species) must also be considered factors (Baum 1997). Natural runs of Atlantic salmon currently occur from Maine to northern Spain and Portugal (Figure 2-1), but spawning runs are at low or even endangered levels in most of those areas (Hutchinson and Mills 2000, O’Neil et al. 2000). The widespread hemispheric decline in salmon, even in streams with high-quality habitat where exploitation has been restricted or prohibited, points to a strong climatic impact in either the riverine or the oceanic portions of the salmon life history or both (Cairns 2001, Hutchinson and Mills 2000, Reddin et al. 2000). The strong coherence of declines for stocks from many varying areas implicates the marine part of the life cycle as a major factor (Reddin et al. 2000), although the spatial patterns of decline are complicated and
suggest that a number of factors and adaptations (such as migration paths) may be at work.
Maine has the last of the wild Atlantic salmon populations in the United States. At one time, 300,000 to 500,000 adults probably entered U.S. rivers each year (Beland 1984, Stolte 1981). The Biological Review Team (1999) used zoogeographic information to construct ecological provinces, including aquatic ecological units (Bailey 1995, Maxwell et al. 1995), to analyze the distribution of Atlantic salmon in the United States. The results suggest that Atlantic salmon populations were divided into at least three distinct groups of populations: (1) those in Long Island Sound, in eight major rivers, including the Connecticut River; (2) those in Central New England, including the Merrimack River in the southern Gulf of Maine; and (3) those in the rest of the Gulf of Maine including the eight DPS rivers, where salmon are listed as endangered under the Endangered Species Act (ESA) (see Chapter 1). A map of Down East Maine showing Atlantic salmon habitat along these rivers is shown in Figure 2-2.
The Long Island Sound populations were gone by the early 1800s (Meyers 1994), followed by the central New England populations in the mid-1800s (Stolte 1981, 1994). The remaining U.S. populations might once have produced 100,000 adults per year, but those numbers have not been seen since the late 1800s. Fewer than 3,000 adults returned per year in the 1960s and 1970s (Figure 2-3). Large stocking efforts in the Penobscot River, especially of smolts, led to a brief period of annual returns numbering 3,000–5,000 fish, but returns to the Penobscot and the other DPS rivers have declined precipitously since the early 1990s (Figure 2-4 [returns versus time]). The decline has occurred despite sustained efforts at stocking and remediating anthropogenic impacts on the Penobscot, strict conservation measures on the DPS rivers, and general improvements in the way that riparian zones are managed. The total return of Penobscot fish for the cohort of smolts released in 1999 (now virtually all accounted for as 1, 2, or 3 sea-winter [SW] fish by 2002) was fewer than 700 adults; for the Gulf of Maine DPS rivers, a minimum estimate of 33 adults returned in 2002 (MASC 2002, USASAC 2003). (Spidle et al. [2003] provide estimates of returns modeled on redd and adult counts from a trap. These estimates include means and 95% confidence limits.) The population decline has been associated with lower return rates, which are now about 1% in the Narraguagus and about 0.2% in the Penobscot. These are below the 2–4% return rate published for many populations (e.g., Reddin et al. 2000). Recent electronic tagging studies in the Narraguagus indicate about half (range = 32–67%) of the total post-riverine mortality is experienced before smolts leave the coastal bay where the Narraguagus enters the sea, but it is not known whether this distribution of the total marine mortality is normal (J. Kocik, NMFS, unpublished data, 2001).
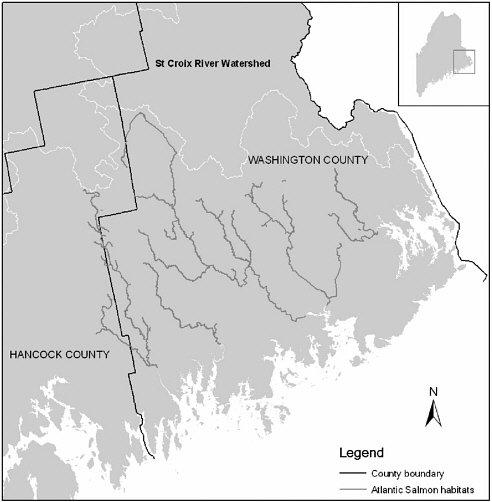
FIGURE 2-2 Atlantic salmon habitat in Down East Maine. SOURCE: Data from Maine Office of Geographic Information Systems. Drawing by Yanli Zhang, University of Massachusetts-Amherst.
The purpose of this chapter is to provide background on the biology and environment of Atlantic salmon specific to the task of understanding why the numbers of fish returning to Maine rivers are declining and recommending steps that would help ensure the survival of these populations. Topics covered in this section include salmon life history, historical and recent changes in abundance, and distribution and migrations. Then, the characteristics of environments that comprise salmon habitat are described: (1) geology and hydrology of soils and forests (including impacts

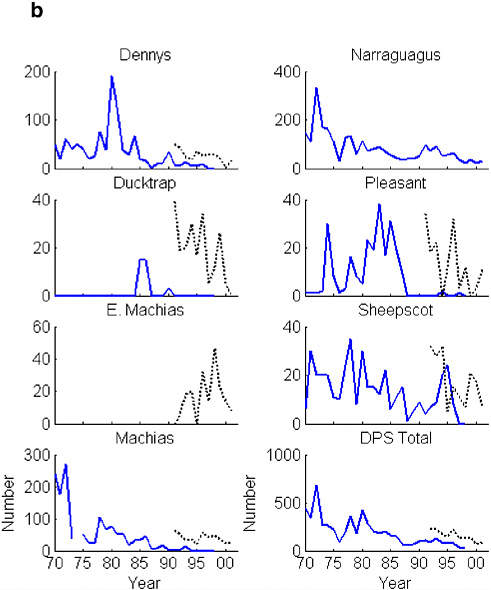
FIGURE 2-3 Documented adult Atlantic salmon returns to all Maine streams. These are rod and trap catches combined for the Penobscot River and primarily rod catches for other rivers. These numbers represent minimum estimates of adult returns. SOURCE: E. Baum, Atlantic Salmon Unlimited, unpublished material, 2001. Printed with permission of the author.
by human activities); (2) aquatic environments; (3) biological communities in the streams and estuaries and along the ocean migration routes; and (4) climate variability. Baum (1997) provides a readable and comprehensive history of salmon in Maine, including maps of individual salmon-producing rivers, detailed histories of stocking efforts, a map of historical fishing weirs, and tables of catch statistics. Those details are not repeated in this report. Bigg (2000) and Dickson and Turrell (2000) provide overviews of climate change and salmon, primarily from the perspective of European stocks. Drinkwater (2000) provides evidence of northern hemisphere climate impacts on North American fisheries. While there are strong suggestions of impact, the exact causal relationships remain unknown. Cairns (2001) provides a lengthy discussion of the many factors that affect salmon abundance and attempts to prioritize them on the basis of their likely and/or potential role in the recent declines. This is an assessment based on the experience and professional judgment of over 60 scientists throughout the range of Atlantic salmon. Finally, a combined report from U.S. National Marine Fisheries Service (NMFS) and U.S. Fish and Wildlife Service (Anadromous Atlantic Salmon Biological Review Team
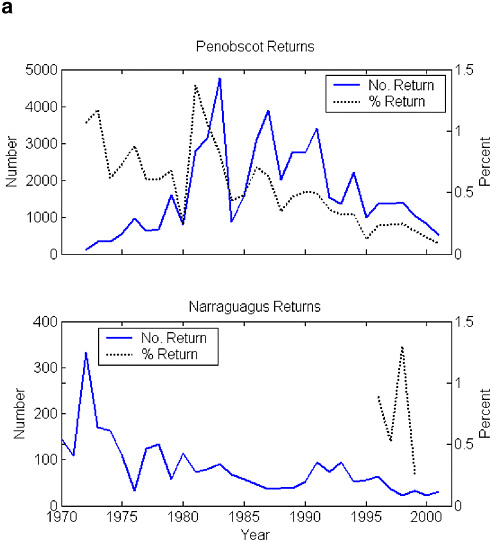
FIGURE 2-4 (a) Minimum estimates of number of fish returning to the Penobscot and Narraguagus rivers and the return rate (%) for that cohort. Penobscot return rate has been adjusted for all multi-sea-winter (SW) returns; the Narraguagus assumes all 2SW fish. SOURCES: Data from K.F. Beland, Maine Atlantic Salmon Commission, unpublished data, 2003; USASAC 1996–2004. (b) Minimum estimates of number of fish returning to seven of the DPS rivers. Solid lines are from fish counts based on rod catches. Broken lines are estimates based on redd counts. (The regression of returns to redds was done on the Narraguagus.) These are underestimates. SOURCES: Data from USASAC 1996–2004.
1999) provides an excellent summary of salmon biology and conservation issues.
SALMON LIFE HISTORY
This subsection provides basic information about the natural history of the Atlantic salmon necessary to understand problems facing their continued existence. The first topic is a description of the sequence of developmental stages and their timing in an individual Atlantic salmon. The second topic concerns key characteristics of the life history of Atlantic salmon as a species. These characteristics include alternative reproductive strategies and anadromy. Finally, recent and historical changes in the distribution and abundance of Atlantic salmon are explained.
Complex Life Cycle
Atlantic salmon are anadromous: they begin their lives in freshwater where the young grow to several inches in length, then migrate to the sea, where they grow more rapidly and become sexually mature after 1, 2, or 3 years1 (Baum 1997). The complex life cycle of the Atlantic salmon consists of a series of morphologically, behaviorally, and physiologically distinct stages as the fish migrate from freshwater to the sea and back to freshwater again. This cycle differs from the simple life cycle of many fish species that complete the transition from juvenile to adult without migrating between different environments. The terms used to describe the Atlantic salmon’s developmental stages are given in Table 2-1.
Key aspects of the stages and of transitions between them are as follows. The egg and alevin stages rely on the yolk deposited in the egg by the mother for nutrition. Hence the nutritional state of the adult spawning female affects the welfare of the offspring. Furthermore, any lipophilic pollutants (e.g., polychlorinated biphenyls) consumed by the adult female tend to be deposited into the lipid-rich yolk of the eggs. Embryonic development is especially susceptible to disruption by chemical agents. Successful transition to the feeding fry stage requires functional organ systems and appropriate behavioral responses. Timing of fry emergence from the gravel is important because of seasonal changes in prey availability. An important aspect of the parr stage is the rate of growth. Growth and size determine the timing of the parr-smolt transformation. Parr are young salmon with 8–11 vertical dark bands on their sides. Transforma-
TABLE 2-1 Stages in the Life Cycle of the Atlantic Salmona
tion to the smolt stage occurs in the winter and spring. Smolts are silvery, without parr marks and with a more streamlined body. As indicated in Table 2-1, most Atlantic salmon in Maine grow fast enough to transform to smolt in their second spring (they are called 2 parr), whereas slower-growing parr transform in their third spring (called 3 parr). The parrsmolt transformation is of key importance because the smolt faces the energetic challenge of seaward migration and the osmoregulatory challenge of the transition from freshwater to seawater. For the remainder of
the calendar year in which smolts enter seawater they are called post-smolts. Beginning January 1, they are thereafter called salmon. The term salmon was used in English by the thirteenth century to describe the silvery salmon in the sea. It was not recognized then that the small parr in the streams were members of the same species.
Understanding the life cycle of Atlantic salmon is complicated by their alternative life-history strategies. For example, before reproductive maturity, these alternatives include variable durations in the stages before their seaward migration and variable numbers of years growing in the ocean (see Table 2-2). Reproductive alternatives include variable age and size at maturity, variable timing of homeward migrations to spawn, variable number of years of spawning, and variable fecundity between years. Maine’s Atlantic salmon exhibit two run timings that are in part influenced by genetic factors. “Early run” adults enter freshwater between
TABLE 2-2 Life-History Strategies and Alternatives of the Atlantic Salmon in Rivers in Maine
|
Life History Factor |
Description of Primary and Alternative Strategies |
|
Development |
|
|
Duration of parr stage |
Primary: 2 years (80%) Alternative: 3 years (20%) or 4 years (small %) |
|
Anadromy |
Primary: migrates to the sea for a growth period Alternative: landlocked populations Alternative: male parr become mature (precocious parr) |
|
Time-at-sea |
2 sea-winters (2SW, estimates of 84–94%) Alternative: 1SW, occurs in males, termed grilse (<0.3%); also estimates of <10% 1SW with >95% males Alternative: multiple sea winters (MSW, such 2SW, 3SW) |
|
Reproduction |
|
|
Age at maturity |
Primary: fifth fall of life Alternative: genetics and environment lead to alternatives |
|
Timing of migration to natal streams |
River dependent Early runs from May to mid-July Late runs from mid-July through September |
|
Spawning frequency |
Primary: Semelparity—spawn only once, then die Alternative: Iteroparity or repeated spawns Alternative: Precocious male parr constitute a large percent, which varies widely among rivers and years |
May and mid-July, and “late-run” adults enter freshwater later in the summer. Most Atlantic salmon are semelparous, meaning they spawn once and die. However, 1–6% of anadromous spawning adults survive, return to the sea, and migrate home later to spawn again. Thus, a small percentage of anadromous fish is iteroparous. However, an unknown percentage of mature male parr survive to breed again, either as a parr or as an anadromous adult. The terms used to describe salmon with different reproductive alternatives are given in Table 2-2.
In addition to anadromous Atlantic salmon, Maine has populations of Atlantic salmon that complete their entire life cycle in freshwater. They are called landlocked salmon or ouananiche. They are the same species as the anadromous form. In Maine, they were originally found only in four drainages, but they have been widely stocked elsewhere in Maine. Although there is some small degree of genetic difference between land-locked and anadromous populations, it is not necessarily greater than the differences among anadromous populations, and it is not clear whether the difference in life history has a genetic basis (Tessier and Bernatchez 2000). Landlocked salmon are not endangered, but because they strongly resemble anadromous salmon and in some cases compete with them, they can complicate efforts to rehabilitate wild anadromous populations.
The salmon life-history pattern has major implications for the species’ evolution and survival in different regions. Because the fish migrate upstream to spawn, they are particularly vulnerable to fishing. Because salmon migrate between ocean and freshwater environments, they are subjected to the vagaries of two ecosystems during different parts of their life history. This anadromous life history greatly increases the number of factors that could affect population size.
Salmon are known for their ability to return to the streams where they were hatched. Salmon return to their natal streams to spawn, a trait that segregates populations and leads to a variety of local adaptations, including the timing of spawning runs, growth rates, and other life-history features (e.g., Allendorf and Ryman 1987, Gharrett and Smoker 1993, Heggberget et al. 1986, Hutchings and Jones 1998, Kendall 1935, Kincaid et al. 1994, Nielsen 1998, for Atlantic salmon; NRC 1996a, Saunders 1981, Smoker et al. 1998 for Pacific salmon species; Taylor 1991 both Atlantic and Pacific salmon; Verspoor et al. 1991, Webb and McLay 1996). Straying to another stream occurs at low frequency. For example, Penobscot River salmon show over 98% fidelity to the home stream (Baum 1997).
The low frequency with which salmon stray to neighboring streams results in the development of a metapopulation structure—a set of local breeding populations connected by exchange of some individuals. This network of local populations provides a balance between local adaptation and the evolutionary flexibility that results from exchange of genetic
material among local populations (NRC 1996a). That NRC report concluded that “maintaining a metapopulation structure with good geographic distribution should be a top management priority to sustain salmon populations over the long term.” That conclusion was drawn for Pacific salmon, but it applies to Atlantic salmon as well.
Although strays probably have lower reproductive success than fish that are returning to their native streams, they provide a source of new genetic combinations—important for the salmon’s evolutionary potential in the face of changing environments—and they may recolonize streams that have lost their own native runs. For Atlantic salmon populations to have colonized and survived for extensive periods near the southern limit of the species’ range (currently Maine), they probably acquired adaptations to the distinct physical and environmental challenges of local waters. Local adaptations, established by strong homing and selection pressures, are a known property of salmon populations throughout the world (Allendorf and Ryman 1987, Taylor 1991).
The complex transition to saltwater at the smolt stage requires suites of behavioral adaptations for navigating, avoiding predators (including seals, cormorants, and striped bass), and finding marine invertebrate and fish prey. During the oceanic phase, juveniles from most river systems migrate to subpolar seas to feed for 2 or more years before returning to their native streams. A small number of fish, referred to as grilse, return after only 1SW. A known exception to this pattern occurs in rivers draining into the Bay of Fundy, Canada. Fish from these rivers remain within the Gulf of Maine and most return to their natal streams after only one winter at sea. For Maine salmon, maintenance of a stable population would require about 2% survival of smolt to 2SW stage (based on Baum’s [1997] estimate of 90 smolts produced per female). A decrease in either freshwater or oceanic survival would cause a decline of Maine’s wild salmon populations.
Adult salmon return to their natal streams from spring until fall. The peak migration time is a characteristic of individual populations and environments. Spawning occurs in autumn, and the eggs develop in gravel nests (redds) that are dug by the female. Because Maine’s females are mostly large 2SW fish, they deposit more eggs, about 7,000 each. The fry emerge in mid-May and grow into parr during the summer. Vertical bars on parr provide camouflage protection from predators. Most parr remain in freshwater for 2 years before becoming smolts and migrating to the ocean. Some male parr mature in the stream and have some success in fertilizing eggs. Survival from the egg to the smolt stage is estimated to be 1.25% (Baum 1997, Bley and Moring 1988), and thus a rough calculation from Baum’s data suggests that an average of 90 smolts are produced by a wild Maine 2SW female.
The anadromous pattern, with some repeat spawning, means that counting the fish returning to a stream gives information only on part of the population. The rest of the population is either in the river as fry, parr, or smolts or still at sea growing and maturing. In addition, salmon have overlapping rather than discrete generations as a result of precocious development and repeat spawners. The presence of early maturing males (precocious parr) tends to buffer the population somewhat against random variation in the return rate of anadromous (adult) male spawners (Garcia-Vazquez et al. 2001, Martinez et al. 2000). Repeat spawners are important because of the increased egg production of older females and their proven success in the face of natural selection. However, 3SW salmon and repeat spawners make up less than 1% of spawning adults (Baum 1997).
The use of freshwater habitats for reproduction and juvenile rearing improves the survival of early life stages because they are inaccessible to marine predators, although they are still susceptible to freshwater predators. Predation depends on the density of predators (Mills 1989), but it was recognized in early studies of juvenile salmon (Huntsman 1938) that precipitation, and thus stream flow and water depth, could affect predation rates and thus juvenile survival (Ghent and Hanna 1999).
When Atlantic salmon smolts enter the sea, they are entering that portion of their life that seems to have the largest variation in survival rate (Cairns 2001, Reddin 1988). At this point, they range in size from 13 to 23 cm fork length—most often 16 to 20 cm—and are 2 or 3 years old. Parrsmolt transformation is influenced by the size of the fish. Approximately 80% (range 70–90%) of the smolts are 2-year-olds that leave the river in spring (late April to mid-June). Most of the remaining fish leave the river as 3-year-old smolts in an outmigration the following spring. Despite the additional growing season, these smolts average only 1.1 cm longer than the 2-year-olds. A very small fraction of fish has been known to leave as 4-and 5-year-olds (Baum 1997). Initial feeding in the marine environment (estuaries) is on insects (at the surface), euphausiids, amphipods, and decapod crustaceans. (These groups may be found in the upper layer of the ocean, although deeper feeding cannot be ruled out.) Smolts soon begin feeding on herring, sand lance, capelin, and shrimp (Baum 1997). Smolts appear to spend most of their time in the upper part of the water column. Electronic tagging data near the mouth of the Bay of Fundy indicate most smolts are in the upper 10 m (G. Lacroix, unpublished data, 2001; see methods in Lacroix and McCurdy 1996). Norwegian studies show a rapid reduction in smolt catch rates when the upper portions of sampling trawls drop below the surface.
Salmon mortality is high during the rapid passage from river to Gulf. Studies conducted in the Narraguagus River from 1996 to 2000 (J. Kocik,
NMFS, unpublished data, 2001) indicate a loss of 38–63% of outmigrating smolts in this small bay (mean = 50%). This is nearly half of the total losses averaged over 1SW and 2SW fish from this river. The average survival of grilse plus 2SW fish from the Narraguagus is 1.1% (annual averages ranged from 0.87% to1.4% in this study), whereas the true “at-sea” survival over this period was >2% when corrected for the initial losses in the bays (J. Kocik, NMFS, unpublished data, 2001). While the near-shore loss is a large proportion of the total marine losses reported here, it must be remembered that the average return rate for this river and for all rivers in the Gulf of Maine and south for the period of record is low.
Salmon pass through the estuarine environment quickly. Electronic tagging reveals that smolts exiting the Narraguagus River pass out of Narraguagus Bay within a few days (J. Kocik, NMFS, unpublished data, 2001). In Penobscot Bay, where electronic tagging and detection are less practical, special trawling methods were used to follow the passage of elastomer-marked fish in 2001 (R. Brown, NMFS, unpublished data, 2001). Smolts passed through counting traps in the main stem of the Penobscot (Veazie Dam, north of Bangor) beginning in late April. By middle to late May, they were widely distributed throughout the bay (80% of tows were positive for smolts) and some had entered the shelf environment (more than 50% of tows outside the bay caught smolts).
Migration and Distribution
Oceanic migration affects growth, maturity schedules, availability to fisheries, and eventually recruitment of salmon populations (Friedland 1998, Narayanan et al. 1995). Migration routes in the Gulf of Maine are unknown. The migration patterns of European post-smolts appear to take advantage of prevailing strong residual currents, such as the Norwegian Coastal Current or the Slope Current along the margin of the shelf (Hansen and Quinn 1998, Holst et al. 2000). If post-smolts leaving the Down East rivers and Penobscot Bay exhibit similar behavior, a likely pathway would involve passage westward along the shelf to the central coast (Penobscot Bay region) and then across the Gulf following the prevailing circulation patterns around Jordan Basin, Georges Basin, and the northern edge of Georges Bank. Passive drift alone could cover this distance in a few weeks. The opposite choice for leaving the Gulf would involve migration eastward across the mouth of the Bay of Fundy and across the southern Scotian Shelf against a residual current that may average about 15–20 kilometers per day at this time of year. Some tagged post-smolts from Maine rivers have been recovered in the Bay of Fundy. Intermediary routes across the open Gulf are also possible and would result in intermediate advantages or disadvantages with respect to the influence of the
residual circulation. A clockwise migration would keep the fish in colder water, and perhaps seawater temperature dominates the migratory behavior. Whichever route is taken, tag returns indicate that post-smolts arrive off northern Nova Scotia (Cape Breton Island) 45–50 days after leaving coastal Maine bays, and in southern Newfoundland shortly thereafter (mid-August, 60–65 days after leaving the Maine coast). In 100–110 days, many salmon have made it to the southern coast of Labrador (see review by Baum 1997; also see data from Friedland et al. 1998). Another factor that might influence post-smolt migration paths is the feeding environment, but this has not been studied in sufficient detail to resolve the relative advantages of the various routes. Bley and Moring (1988) and Friedland (1994a) compared return rates for rivers at different latitudes on both sides of the Atlantic Ocean and suggested that low rates might be characteristic of populations whose natal rivers are located near the limit of the species’ range. The hypothesis was that mortality was higher due to the longer distances traveled between the natal rivers, winter feeding areas, and back. Despite the relatively large numbers of returning fish, the return rate for the Penobscot is less than half that for the Narraguagus, so other factors remain important. The highest return rates in the Gulf of Maine and south occur in the St. John River of New Brunswick and Maine. These higher rates might be due in part to the greater percentage of 1SW fish in the St. John (more than 90% vs. less than 10% in the Maine rivers).
Perhaps more important with respect to interpretation of mortality and return data, most fish from the inner Bay of Fundy do not leave the Gulf of Maine and therefore do not undertake the long migrations of Maine salmon (Ritter 1989). While the return rate for these fish is higher than that for salmon from Maine rivers, these rates also have been declining through the 1990s. Maine’s salmon take part in extensive marine migrations, including movements to the waters off western Greenland (Friedland 1994a), where they become a small portion of a large mixed-stock complex of salmon from both European and North American sources. Unlike Atlantic salmon populations across the Canadian border from Maine, where 1SW fish are common among spawning adults, about 94% of adults returning to Maine are 2SW fish (USASAC 1999). Thus, the average body size of Maine adults is larger than Canadian adults. Because spawning populations of Maine salmon include several age groups (especially 2SW and 3SW adults but also precocious parr2), there is considerable exchange of genetic material across age classes (cohorts).
|
2 |
Parr are young salmon actively feeding in freshwater. Even younger fish, with egg sacs, are called fry. Fish about to migrate to sea are called smolts. See Table 2-1. |
The winter feeding grounds of Atlantic salmon are in the Labrador Sea (primarily North American stocks) and in the North Atlantic east of Greenland (mostly European and Icelandic stocks). These locations are associated with an apparent thermal preference of 4–8 ºC (Reddin et al. 2000). There is a small amount of mixing of stocks from the two continents at this time of the life cycle, but straying of spawning fish from Europe to North America, or the reverse, is very unusual (Reddin et al. 1984).
PHYSIOLOGY
Physiology is the functioning of the individual, and it ties together genetics and ecology. There are three key concepts of particular significance to the discussion of Atlantic salmon. They concern homeostasis, temperature effects on rates, and the neuroendocrine transduction of environmental information (Figure 2-5). They are briefly explained, and their impacts on the timing of parr-smolt transformation and outmigration in the Atlantic salmon are discussed.
Homeostasis is the maintenance of a constant internal environment, which is necessary for life. The internal stability reflects a dynamic equilibrium and requires work. The internal environment differs from the external environment, whether the salmon is in a stream or the sea. The difference is created and maintained at the interfaces between the animal and its environment. These interfaces in the Atlantic salmon are the gills, gut, kidneys, and skin, and they are important for two reasons: First, it is these interfaces that are most susceptible to infection and insult; and, second, the roles of these sites change to meet the challenge imposed by the transitions between freshwater and seawater. At no site is this more obvious than in the gills. Gills regulate internal salts, gases, and nitrogenous wastes. The proxy used by salmon physiologists for indicating seawater readiness in Atlantic salmon during the parr-smolt transformation is an increased level of activity of the enzyme Na+/K+-ATPase in the gill. Gills are damaged by the environmental hazard of steam acidity, as discussed elsewhere.
Temperature affects all aspects of physiological functioning. The Atlantic salmon is ectothermic, meaning it has the temperature of its environment. Because the environmental temperature fluctuates, physiological functioning fluctuates as well. A general rule is that metabolic rate doubles with every 10 ºC increase. Metabolism underlies development and growth. This means that hatchery, stream, reservoir, estuary, and ocean temperatures strongly affect rates of development and growth.
Neuroendocrine signals are specific chemical signals linking a salmon to its environment (Hoar 1965). A complex array of detectors receives information about the external and internal environments. This informa-

FIGURE 2-5 Multiple interactions between the environment and the organism that lead to smolt development. Growth conditions such as temperature, food, photoperiod, and competition determine growth of Atlantic salmon parr. A critical size (or size-related development stage) is necessary for smolting to proceed, and thus environmental conditions determine the age at which smolting occurs. Once this developmental stage has been reached, photoperiod and to a lesser degree temperature regulate neuroendocrine changes that bring about physiological changes in the spring. Releasing factors such as temperature, flow, and turbidity may have rapid effects (dashed arrows) to initiate downstream migration. Development of the smolt physiological condition (which presumably includes a behavioral readiness or a migration disposition), induced by prior development, photoperiod, and temperature, is necessary for releasing factors to initiate downstream migration (see Baggerman 1960). The possible neuroendocrine or physiological mediators of these rapid effects are not currently known.
SOURCE: McCormick et al. 1998. Reprinted with permission; copyright 1998, NRC Research Press, Ottawa, Ontario.
tion includes daylength, sight, sound, odors, water flow, ambient and internal salinity, pH, and energy stores. Typically the central nervous system integrates the information and governs the effectors that regulate survival, reproduction, and behavior. All behavior requires neuromuscular activity and the expenditure of energy. In this way, behavior is shaped
not only by genes but also by the interaction, mediated by the neuroendocrine system, between the salmon and the environment.
The requirement for homeostasis, the rate-setting role of environmental temperature, and the powerful role of the neuroendocrine system all interact to affect the timing and success of outmigration (see Figure 2-6). The Atlantic salmon prepares in advance for the transition to seawater

FIGURE 2-6 Simple mathematical model of the interaction of migration timing and environmental conditions and their effect on adult survival. Migration timing and survival estimates are typical for Atlantic salmon (e.g., Jonsson and Ruud-Hansen 1985, Hvidsten et al. 1995), but the temporal changes in environmental conditions are largely hypothetical. Values for migration timing are migrants per week. Adult returns are calculated from the weekly number of migrants and weekly survival rates. When migration timing and optimal environmental conditions coincide (solid lines), adult returns are high (total returns, 166). When migration timing and optimal environmental conditions are out of phase by 2 weeks (dashed lines), adult returns are lower (total returns, 94). Results of this simulation indicate that even when the magnitudes of migration and environmental conditions remain the same, alterations in their timing can have significant effects on adult returns. SOURCE: McCormick et al. 1998. Reprinted with permission; copyright 1998, NRC Research Press, Ottawa, Ontario.
rather than adjusting to it (Boeuf 1993); this is called physiological preadaptation. Preadaptation contrasts with acclimatization that occurs in many other species of coastal fishes that adjust to changes in salinity met while moving in and out of estuaries and rivers. The timing of preparedness has been an important issue because of the need to decide when to release hatchery-reared smolts or how to regulate water outflow from dams. It is also an important issue to be considered in our effort to understand the causes of low survival in the transition to the marine environment. The relevance to these issues is explained below.
The environmental cue or zeitgeber for the parr-smolt transformation is photoperiod, specifically the rate of day lengthening (Duston and Saunders 1990). The increase in daylength in the spring is transduced by the neuroendocrine system largely into increased output of pituitary growth hormone, which has the actions of elongating the stocky parr into a sleek and fast smolt and of coordinating preparation for osmoregulation in seawater (Björnsson 1997). Temperature is not a zeitgeber for smolting in Atlantic salmon, but rather temperature affects the rate of change in response to photoperiodic information (Johnston and Saunders 1981, McCormick et al. 2002). There is strong evidence for the significance of photoperiod and temperature in the timing of smolting (Sigholt et al. 1998, Solbakken et al. 1994). Smolting is a developmental phenomenon dependent on reaching a critical size of about 10 cm total length at the end of the previous growing season (Hoar 1988); this is an issue separate from the actions of growth hormone during smolting. This knowledge restricts solutions to the decline in Atlantic salmon to those including springtime seaward migration.
The environmental information used by smolting Atlantic salmon to time outmigration is complex and likely includes temperature, rainfall, and water flow, and the behavior of other smolts as “proximate cues” or “releasing factors” (Jonsson 1991, McCormick et al. 1998). Much of the endocrine system is highly activated for a prolonged period of weeks during smolting (Hoar 1988). The thyroid hormones and cortisol are linked with mobilization of energy stores, change in rheotactic behavior from an upstream to downstream orientation, and outmigration (Iwata 1995, Specker et al. 2000). Thyroid hormones and cortisol are not mediating photoperiodic information; rather they are mediating information about temperature, rainfall, and water flow, and possibly the behavior of other smolts.
The physiology of Atlantic salmon indicates that there is a “smolt window” that both opens and closes in the spring. Our current understanding of the impact of temperature on the window is that warmer temperatures accelerate the opening and closing of the window and can shorten the time during which salmon can successfully transition to the
sea. Thus, dams and impoundments and other changes to the riparian environment can interfere with physiology and development. Three kinds of studies taken together illustrate the importance of the rate-setting role of environmental temperature in the timing and success of outmigration in Atlantic salmon. The first are studies providing evidence that increased temperature can accelerate the loss of smolt characteristics in Atlantic salmon in hatcheries (Duston et al. 1991). The second are studies concluding that hatchery-reared smolts released as smolt characteristics were declining had lower recapture rates, indicating reduced survival (Staurnes et al. 1993, Virtanen et al. 1991). The third study showed that migrating Atlantic salmon smolts in the Connecticut and Penobscot rivers lost their high salinity tolerance and gill Na+/K+-ATPase activity as the rivers warmed at the end of spring, whereas Atlantic salmon smolts in the more northern and colder Conne River and Catamaran Brook retained their smolt characteristics longer (McCormick et al. 1999). In the southern rivers, there was also year-to-year variation, supporting the conclusion that warm temperatures caused a more rapid decline of smolt characteristics.
AQUATIC ENVIRONMENTS
Salmon are a cold-water species. They spawn in streams characterized by clear, flowing water with gravel areas for egg deposition and embryonic development and productive, physically heterogeneous sections of river habitat for juvenile growth and survival. Stream size varies and ultimately affects the size of local runs through limitations of habitat space for spawning and the growth of parr.
The Pleasant, Narraguagus, Machias, and East Machias rivers empty into small coastal bays that develop seasonal stratification during warm months of the year. The Dennys River empties into a larger and more complex bay system (Cobscook and Passamaquoddy bays) with stronger tidal flows and less vertical stratification. The entire region is tidally energetic. All the Down East bays open to a coastal shelf (out to a 100-m isobath) that is dominated by the Eastern Maine Coastal Current (EMCC, see below). The Penobscot River empties at the head of Penobscot Bay, which is second in size on the East Coast only to Chesapeake Bay. The Ducktrap River also empties into Penobscot Bay, about halfway down its western shore (Figure 1-1). Penobscot Bay opens to the coastal shelf near the western end of the EMCC. The Kennebec and Androscoggin rivers empty into Merrymeeting Bay near Bath, and then the Kennebec River flows into the ocean just east of Casco Bay. The St. John River, more than twice as large as the Penobscot in drainage area and flow volume, is partly in Maine, partly shared with New Brunswick, and flows into St. John Harbour in New Brunswick.
The Gulf of Maine is characterized as a marginal sea because its connections with the North Atlantic are significantly constrained by large offshore banks (Georges Bank and Browns Bank). The Gulf has a general cyclonic (counterclockwise) residual circulation (the flow that is left after removing the tides). Surface water (upper 75 m) enters the Gulf of Maine across the southern Scotian Shelf, originating from the Labrador Sea and Gulf of Saint Lawrence to the north. Except for the occasional presence of a warm-core ring, the surface water is colder than the rest of the Gulf for most of the year. The temperature and biota associated with this circulation pattern provide a relative continuity of habitat between the Maine DPS rivers and the winter feeding grounds in the Labrador Sea. By contrast, rivers to the west of Penobscot Bay empty into an aquatic regime that is distinctly different during warm months of the year, approximately May to October. Most of the water exiting the Gulf does so via a narrow jet along the northern edge of Georges Bank, while a smaller volume leaves through the shallower Great South Channel.
CLIMATE CHANGE
Atlantic salmon distributions have been influenced by geological changes, including ice ages (MacCrimmon and Gots 1979). Populations in the United States probably date from the end of the last ice age 10,000 years ago. Atlantic salmon were probably present in all watersheds from the Hudson River in New York north to the St. Croix River at the U.S.-Canadian border (Kendall 1935). Atlantic salmon once occupied 34 rivers and streams in Maine (Beland 1984, Rounsefell and Bond 1949). Today, wild Atlantic salmon populations in the United States are found only in Maine,3 from the lower Kennebec River in the southwest to the Canadian border, a range contraction that may in part be due to climate change.
Few fish species in the North Atlantic are as affected by climate variation over as wide a region as Atlantic salmon. The ocean migrations of Atlantic salmon rival those of the large pelagic species such as tuna, with documented returns of North American salmon from the eastern side of the Atlantic and European fish from the western side (Hansen and Jacobsen 2000, Tucker et al. 1999). The migrations themselves vary in response to currents and temperature distributions, among other factors. But the environment’s effect on salmon is not limited to the conditions that adult
salmon experience at sea. Freshwater mortality, hitherto considered less variable than marine mortality (Chadwick 1987), may be amplified by direct and indirect effects of changes in precipitation, seasonal ice formation, temporal patterns of stream flow and other local properties associated with global climate change (Cunjak et al. 1998). During their first year at sea, the migration cues, first feeding opportunities, and ocean nursery conditions affecting juveniles may be strongly affected by climate (Drinkwater 2000, Friedland et al. 1998, Montevecchi et al. 2002). To understand the relationship between salmon and climate variation, it is important to deal with three critical life-history stages of salmon: juveniles in freshwater, juveniles during their first year at sea, and maturing adults.
Climate can affect the dynamics of juvenile salmon populations in freshwater nurseries through modulation of growth rates, principally by the effect of temperature on growth. Habitat is a limiting factor in the production of juvenile salmon, and the factors that affect the pace at which cohorts move through rearing habitat the overall production of pre-recruits to the stocks (Bardonnet and Baglinière 2000). Juvenile rearing in freshwater may last for as long as 7 years in northern streams to as little as 1 year in southern habitats (Power 1981). Since migration from freshwater is growth-mediated, climate conditions that affect growth will determine the pace at which cohorts leave nursery streams. Smolt ages will probably decrease, and precocious maturation will probably become more frequent across much of the rearing habitat in North America if the anticipated increases in temperature associated with global climate change are realized (Juanes et al. 2000, Minns et al. 1995).
Concerns about parr mortality during the winter before they leave freshwater have increased recognition of the relationship between climate and the structure of the rearing habitat. Winter mortality is associated with the relationship between premigrant parr and their rearing habitat, which may be marginal in providing refuge during their last winter in freshwater. During their last winter in freshwater, premigrant parr are relatively large for their habitat and they often live beneath winter ice (Cunjak et al. 1998). The mortality of premigrant parr may be quite high for some populations and subject to climate variations that affect the stability of the ice cover (Whalen et al. 1999a,b). The smaller members of the nursery population may be better adapted to surviving these shifting conditions because their smaller size makes more specialized refuges available (Cunjak 1988). Changing climate conditions could destabilize ice cover and cause pre-migrant parr mortality to increase.
The next transition for salmon is the movement of smolts into the ocean, which is affected by climate conditions in many ways. At the outset, smolt migrations are cued by environmental signals, such as temperature in freshwater rearing areas (Jonsson and Ruud-Hansen 1985, Solomon
1978). In theory, smolts have adapted to environmental cues that deliver them to specific “migration windows” in the coastal ocean, where the fish are able to take advantage of prey, avoid predators, and find suitable habitat conditions. The fish are already under physiological stress since they are challenged by the transition of moving from freshwater to saltwater; timing the migration to optimize ecological conditions improves survival (Friedland and Haas 1988). If adaptations to initiate the migration to sea are not robust to climate variability, the consequence for regional stock groups may be profound, especially for stocks at the margins of salmon distribution that may already have low return rates.
Although many sources of mortality affect salmon throughout their marine life, the largest source is thought to be predation during their first few weeks at sea (Fisher and Pearcy 1988, Holtby et al. 1990). The size and variability of this source of mortality make it an important determinant of the return rate (Pearcy 1992, Salminen et al. 1995). Many attempts have been made to establish a link between salmon survival and climate (Friedland 1998, Friedland et al. 2000, and references therein). Although progress has been made with correlations, the causal mechanisms have remained elusive. Recent analyses (Friedland et al. 2003) provide the first indication for North Atlantic salmon that survival is negatively correlated with sea-surface temperature (SST) in June if SST exceeds the preferences of the local stocks of salmon. The importance of temperature during the first few months at sea is supported by data on salmon from Iceland and the Baltic (Salminen et al. 1995, Scarnecchia 1984).
The nursery zone for European post-smolts is located in the open ocean, whereas North American post-smolts appear to use inshore habitats. Holm et al. (2000) described the distribution of European post-smolts from surface-trawling operations in the northeastern Atlantic. The nursery is confined to a region within the Norwegian Sea, the northern extent of which appears to be defined by current transport. The post-smolts co-occur with surface schools of herring and mackerel and occupy a similar ecological niche (Jacobsen and Hansen 2000). In North America, postsmolts can be found in high numbers in the Labrador Sea during the fall of the year (Reddin and Short 1991). However, during the earlier part of the post-smolt period (i.e., through the spring and summer) fish are also found in the Gulf of St. Lawrence, along the coast of Nova Scotia, and elsewhere (Dutil and Coutu 1988, Friedland et al. 1999, Ritter 1989). Furthermore, North American stocks may not mix for many months after entering the ocean, while European stocks appear to be concentrated in a single, albeit large, ocean area (Friedland and Reddin 2000).
Age at maturation has important consequences for the total complement of eggs deposited during spawning. Younger fish do not produce as many eggs as multi-sea-winter salmon. Although the decision to mature
has a strong genetic component (Gjerde 1984), environment also plays a significant role through effects on growth (Saunders et al. 1983a). Growth at various times during the post-smolt year may be important for achieving maturity (Duston and Saunders 1999, Gudjónsson et al. 1995, Scarnecchia et al. 1991). Alternatively, some investigators have suggested that climate variations that extend migrations beyond the normal return distance affect the proportion of grilse in the return (Martin and Mitchell 1985).
The North Atlantic Oscillation (NAO) is the dominant mode of atmospheric variation in the North Atlantic (Dickson et al. 2000) and has been associated with the effects of climate variation on the survival and maturation of Atlantic salmon. The spring thermal habitat areas associated with post-smolt survival of central European salmon stocks are derived from large ocean areas where the distribution of SST is affected by the NAO (Dickson and Turrell 2000). Likewise, the winter thermal habitat associated with the abundance of specific age components in the Northwest Atlantic are also derived from areas where SST distribution is correlated with the NAO (Friedland et al. 1993). However, it would be premature to suggest that the NAO is the only mode of climate forcing that affects salmon. For example, high-frequency fluctuation in currents in the Barents Sea appears to create a lagged linkage between Icelandic and Russian salmon stocks (Antonsson et al. 1996). Other atmospheric indices might be useful in developing hypotheses about transoceanic and global stock synchrony and in explaining salmon population trends (Klyashtorin 1998).
The unprecedented decline in Atlantic salmon abundances over the past few decades raises concerns about the effect that climate change may have on Atlantic salmon. With climate at the core of many of the factors contributing to the decline of stocks, the effect of further shifts, beyond the reactive norms to which salmon populations have adapted, now poses the threat of a range shift for the species. If climate changes are compounded by other anthropogenic factors affecting the health and size of the stock (e.g., through habitat effects on the freshwater part of the life cycle), local populations may be driven to extinction.