2
Evaluating Exposure to Secondhand Smoke
Important considerations in evaluating the effects of secondhand smoke include the magnitude of exposure to it,1 how exposure can be measured, and how exposure changes with the implementation of smoking bans. This chapter discusses the constituents of secondhand smoke and the measurement of exposure to secondhand smoke, beginning with measurement of airborne tracers of secondhand smoke and of its main biologic markers (or biomarkers)—the nicotine metabolite cotinine and metabolites of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK). It then summarizes the information available on secondhand-smoke concentrations and exposures before and after the implementation of smoking bans.
CONSTITUENTS OF SECONDHAND SMOKE
Cigarette smoke is a complex aerosol2 consisting of thousands of chemicals (Cal EPA, 2005b). It consists of gases and volatile chemicals in which particulate matter (PM) is suspended. The gas phase consists of air, carbon dioxide, carbon monoxide, and many other chemicals, including nicotine, carbonyls (such as acetaldehyde, formaldehyde, and acrolein), hydrocarbons (such as benzene, toluene, and some polycyclic aromatic hydrocarbons
[PAHs]), nitrogen oxides, pyridine, ammonia, nitrosamines, and hydrogen cyanide (Cal EPA, 2005b). The particulate phase, “tar,” consists of thousands more chemicals, including alkaloids, larger PAHs, tobacco-specific nitrosamines, polonium-210, nickel, cadmium, arsenic, and lead. Some compounds, such as cresols and PAHs, are partitioned between vapor and particulate phases.
About 85% of secondhand smoke is composed of sidestream smoke emerging from the burning tip of the cigarette and the remainder is exhaled in mainstream smoke (the smoke inhaled by a smoker when puffing on a cigarette) (Kritz et al., 1995). The measured sidestream emissions of chemicals are quite similar among a wide range of cigarette brands and styles, including regular, unfiltered, filtered, and “low tar, low nicotine” cigarettes.3 Although the composition of sidestream and mainstream smoke are qualitatively similar, there are substantial quantitative differences in composition between mainstream and sidestream smoke because the chemicals emitted in tobacco smoke change with temperature, oxygen concentration, pH, and the extent of combustion.4 Those factors are different in mainstream and sidestream smoke (Jenkins et al., 2000). As summarized elsewhere, most compounds from cigarettes are emitted in sidestream smoke in much higher amounts than in mainstream smoke (Cal EPA, 2005a; Jenkins et al., 2000; NRC, 1986). For instance, the ratio of the mass of benzene emitted into sidestream smoke compared to that emitted into mainstream smoke is approximately 10, while the corresponding ratio for the 4-aminobiphenyl is 30, and that, for nicotine is approximately 2. More recently, Lodovici et al. (2004) reported that the amount of total PAH in sidestream smoke “was about tenfold higher compared with mainstream smoke.” Nicotine is primarily in the particulate phase of mainstream smoke but predominantly in the vapor phase in secondhand smoke (Cal EPA, 2005a). This variable ratio from compound to compound between sidestream and mainstream smoke makes it impossible to characterize a passive smoking exposure as a simple fraction of the dose a smoker receives; such a comparison must be chemical specific (Hammond et al., 1993). Thus, while on average nonsmokers exposed to secondhand smoke have about 1% the cotinine (a metabolite of nicotine) as smokers, they have 14% as much 4-aminobiphenyl (a potent human carcinogen) adducted to their hemoglobin (Hammond et al., 1993).
Animal experiments by Philip Morris laboratories have demonstrated that sidestream smoke is three to four times more toxic than mainstream smoke (Schick and Glantz, 2005).
This complex picture becomes even more complicated over time. The ambient emissions from cigarettes can undergo further chemical reactions and deposit at varying rates on surfaces (Jenkins et al., 2000). For example, chemical analyses of aging sidestream smoke have shown that the carcinogenic nitrosamine NNK can form from nicotine and increase over time (Schick and Glantz, 2007). The chemical and physical properties of PM in secondhand tobacco smoke also change rapidly due, for example, to diffusion and coagulation, particle setting and impaction, and chemical reactions (Benner et al., 1989; Eatough et al., 1989); however, measurements of concentrations in smoking environments averaged over a day to a week have demonstrated similar ratios of PM to nicotine (Daisey, 1999; Leaderer and Hammond, 1991).
The toxicity of sidestream smoke appears to increase over time. Schick and Glantz (2006), using data from a series of inhalation experiments in rats conducted at Philip Morris, compared freshly generated sidestream smoke to sidestream smoke that had been aged for 30–90 minutes in a 30 m3 chamber. When the smoke doses were equalized on the basis of particulate material concentration, aged sidestream smoke was four times more toxic in 21-day exposures and two times more toxic in 90-day exposures than the freshly generated sidestream smoke. Moreover, current methodologic limitations prevent estimation of concentrations of highly reactive compounds; this is particularly important for the more reactive constituents of tobacco smoke and for estimating their concentrations in secondhand smoke dispersed in an unspecified space. A partial list of cigarette smoke constituents in mainstream and sidestream smoke in amounts exceeding 10 μg/per cigarette is presented in Table 2-1.
MEASUREMENT OF SECONDHAND SMOKE
Tobacco smoke is a complex mixture of thousands of compounds. The composition of secondhand smoke changes over time; substances emitted from cigarettes can undergo chemical reactions and deposit on surfaces at various rates (Singer et al., 2002). Several approaches to evaluating and comparing human exposures to secondhand smoke, including measurement of airborne tracers or biomarkers of exposure (see Table 2-2), are useful.
In a 1986 report (NRC, 1986) on secondhand smoke (or environmental tobacco smoke, ETS), the National Research Council stated that “a marker or tracer for quantifying ETS concentrations should be:
-
unique or nearly unique to the tobacco smoke so that other sources are minor in comparison,
-
a constituent of the tobacco smoke present in sufficient quantity such that concentrations of it can be easily detected in air, even at low smoking rates,
-
similar in emission rates for a variety of tobacco products, and
-
in a fairly consistent ratio to the individual contaminant of interest or category of contaminants of interest (e.g., suspended particulates) under a range of environmental conditions encountered and for a variety of tobacco products.”
Those criteria remain important today. In a recent report (2006), the National Research Council presented similar criteria that should be considered in selecting a biomarker, regardless of its intended use. The criteria include the sensitivity of the assay for the biomarker, the specificity of the
TABLE 2-1 Amount of Cigarette Smoke Constituents in Tobacco Smoke and Smoking Environments. Partial List of the Cigarette Smoke Constituents Generated in Mainstream and Secondhand Smoke in Amounts Exceeding 10 μg per Cigarette or That Have Been Shown to Be Cardiotoxic
|
Compound |
Average Amount (μg per cigarette except where noted) |
Present in Secondhand Smoke (>10 μg per cigarette) |
Mean Concentration in Smoking Environments |
|
Carbon dioxide |
30,000 |
|
|
|
Carbon monoxide |
20,000 |
Yes |
0.2–33 ppma |
|
Nicotine |
1,650 |
Yes |
0.6–106 μg/m3a |
|
Acetaldehyde |
700 |
|
370–462 μg/m3a |
|
Acetic acid |
570 |
|
|
|
Hydrogen cyanide |
450 |
|
|
|
Formic acid |
340 |
|
|
|
Nitrogen oxides |
300 |
|
3–350 ppba |
|
Formaldehyde |
300 |
Yes |
5–1,100 μg/m3a |
|
Methyl chloride |
300 |
|
|
|
Benzeneb |
30 |
Yes |
2–100 μg/m3a |
|
Acetone |
250 |
|
|
|
Catechol |
195 |
|
|
|
1,3-butadienec |
150 |
Yes |
0.2–19 μg/m3a |
|
Toluene |
150 |
|
|
|
Methanol |
135 |
|
|
|
Hydroquinone |
120 |
|
|
|
Lactic acid |
120 |
|
|
|
Compound |
Average Amount (μg per cigarette except where noted) |
Present in Secondhand Smoke (>10 μg per cigarette) |
Mean Concentration in Smoking Environments |
|
Succinic acid |
120 |
|
|
|
Phenol |
100 |
|
|
|
Ammonia |
100 |
|
|
|
Glycolic acid |
100 |
|
|
|
4-vinylcatechol |
84 |
|
|
|
Acroleinc |
80 |
Yes |
14–100 μg/m3a |
|
Methylethylketone |
70 |
|
|
|
3-cresol |
60 |
|
|
|
4-cresol |
60 |
|
|
|
Propionaldehyde |
45 |
|
25–110 μg/m3a |
|
Resorcinol |
44 |
|
|
|
3-methylfluoranthene |
40 |
|
|
|
4-methylcatechol |
38 |
|
|
|
3-methylcatechol |
38 |
|
|
|
4-vinylphenol |
30 |
|
|
|
2-methylfluranthene |
30 |
|
|
|
Pyridine |
30 |
|
1.34–6.5 μg/m3a |
|
Carbon disulfide |
30 |
Yes |
|
|
4-ethylcatechol |
28 |
|
|
|
3-picoline |
24 |
|
|
|
4-picoline |
24 |
|
|
|
2-cresol |
22 |
|
|
|
3-vinylpyridine |
22 |
Yes |
|
|
Cholesterol |
22 |
|
|
|
Benzoic acid |
20 |
|
|
|
3-ethylphenol |
18 |
|
|
|
4-ethylphenol |
18 |
|
|
|
Crotonaldehyde |
15 |
Yes |
|
|
2-methoxyphenol |
13 |
|
|
|
2-picoline |
12 |
|
|
|
Butyraldehyde |
12 |
Yes |
|
|
4-vinylguaiacol |
11 |
|
|
|
Cadmiumc |
0.5 |
Yes |
|
|
Leadc |
0.4 |
Yes |
|
|
Benzo[a]pyrene |
0.075 |
Yes |
0.4–22 ng/m3a |
|
Chromiumc |
0.07 |
|
1.2–8.9 ng/m3a |
|
Nickelc |
0.05 |
|
2.5–7.2 ng/m3a |
|
Particulate matter |
50f |
Yes |
27–2,000 μg/m3a |
|
a Data are from Jenkins et al., 1999. b Benzene concentration present in sidestream smoke is 163–353 μg per cigarette. c Amount is for sidestream smoke. d Data are from Bolte et al., 2008. e Data are from Brauer and Mannetje, 1998. fThe concentration of particulate matter is in μm/m3. |
|||
TABLE 2-2 Biomarkers and Airborne Tracers
|
|
Advantages |
Disadvantages |
|
Biomarkers |
Dose; integrates exposures from all sources |
Does not distinguish source location |
|
Nicotine in body fluids |
Specific to tobacco |
Very short half-life in fluids (therefore only measures exposure that occurred in previous few hours) |
|
Nicotine in hair, nails |
Specific to tobacco Easy, noninvasive to collect Reflects longer period of exposure |
Does not indicate recent exposures or patterns of exposure |
|
Cotinine in body fluids |
Specific to tobacco Easy, noninvasive to collect in saliva, urine Sensitive (present in high levels so easy to detect low-level exposure) |
Short half-life in fluids, so measures recent exposure (only the previous few days) Blood samples are more invasive to collect |
|
NNK metabolites |
Specific to tobacco Can detect in urine Longer half-life in fluids relative to nicotine (therefore can measure exposure over several weeks) |
Expensive (greater analytical costs for assay) |
|
Airborne Tracers |
Measures and compares exposures from different sources (for example, in different venues such as homes, workplaces, and public places) |
Requires measurement of all sources to determine exposures from all sources Does not reflect individual respiratory rates |
biomarker for the chemical or metabolite of interest, the relevance of the biomarker to the exposure and disease outcome of interest, the practicality of the biomarker (both in the ability to collect a biologic sample and in the analytic method), and the pharmacokinetics of the biomarkers, especially in terms of its half-life of the compound measured. Although few, if any, biomarkers have been shown to meet all the criteria, a number of biomarkers of secondhand-smoke exposure that meet many of the criteria are available.
Measures of exposure in the air and of biomarkers of exposure are complementary. Assuming equally accurate and sensitive methods, bio-
|
|
Advantages |
Disadvantages |
|
Airborne NNK |
Specific to tobacco smoke Of intrinsic health interest (known carcinogen) |
Expensive (greater analytical costs for assay) Less sensitive than nicotine because present in lower concentration (therefore can not measure as low secondhand smoke concentrations) |
|
Particulate matter |
Present at high levels in secondhand smoke so can measure a wide range of concentrations relatively easily Can measure with continuous sampler and get information directly, without laboratory |
Not specific to tobacco smoke and many other sources present at all times, therefore not distinguishable from other sources of PM at lower secondhand-smoke concentrations |
|
|
|
Initial investment in equipment expensive, but little operating cost |
|
Airborne Nicotine |
Specific to tobacco smoke Of intrinsic health interest (known cardiovascular agent) Present at high levels in secondhand smoke facilitating easy measurement of a wide range of concentrations, including very low concentrations |
Different decay rate than other secondhand smoke constituents, so complicates estimation of exposure to those other constituents Requires laboratory analyses |
|
Abbreviations: NNK, 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone. |
||
markers afford better measurement of the dose that a person receives because they integrate all sources of exposure and reflect inhalation rates, which might vary from person to person and for a given person over time. Interpretation of the level of a biomarker, however, must consider its half-life: if its half-life is short, only recent exposure is measured. Airborne tracers of exposure are able to show the relative contributions of different sources or venues of an exposure (for example, home exposures compared with workplace exposures). In contrast, biomarkers do not differentiate between sources of exposure but rather integrate all exposures and reflect true dose.
Airborne Tracers of Secondhand Smoke
Nicotine and its metabolite cotinine have been widely used as tracers of secondhand smoke. Ambient nicotine can be measured accurately and sensitively, and cotinine can be measured in saliva, blood, and urine. One major characteristic that contributes to the widespread use of airborne nicotine and cotinine is that tobacco is virtually the only source of both compounds, so they meet the criterion noted earlier. Furthermore, tobacco smoke contains large amounts of nicotine, so tobacco smoke can be detected even at low concentrations. Sensitive, specific, and accurate methods to measure nicotine in ambient air and cotinine in body fluids are now well established and have been used in dozens of investigations around the world.
Another commonly used tracer for secondhand smoke is particulate matter (PM). In heavy-smoking environments—such as bars, pubs, and many restaurants—the concentration of PM can be extremely high, and direct-reading instruments provide immediate data without the need for a laboratory. However, there are many other sources of PM, which is ubiquitous, so that even if no smoking occurs, PM is present at levels that might affect health, as is known from air-pollution studies. This background level of PM complicates measurement of PM from secondhand smoke at low secondhand-smoke levels. Because virtually all secondhand-smoke particles are less than 2.5 micrometers in diameter, all secondhand-smoke particles are contained in PM2.5, and eliminating particles larger than 2.5 micrometers, for example, by the use of an impactor or other size selector, reduces the contribution of non-secondhand-smoke PM (Cal EPA, 2005a). That does not, however, eliminate the PM from traffic or other combustion sources.
Nicotine and some other components of secondhand smoke deposit readily onto surfaces, with very small amounts of re-emission. Highly volatile gases in secondhand smoke (such as benzene and butadiene) tend not to deposit on surfaces. A few hours after smoking has ceased, most of the airborne nicotine will have deposited on surfaces, but nearly all the benzene and butadiene will remain in the air (Singer et al., 2002). If nicotine is used as the only tracer for those other gases, and the ratio of nicotine to benzene in fresh smoke is used to estimate the benzene concentration, one may underestimate the exposure of room occupants to benzene. That is true for many other toxic chemicals in secondhand smoke, and this drawback applies to the use of cotinine as a biomarker as well as to nicotine as a tracer in the air. Despite the limitation, airborne nicotine and cotinine remain extremely useful in evaluating exposures in many settings.
Biomarkers of Exposure to Secondhand Smoke
Although most of the toxicants in tobacco smoke are not specific to tobacco-smoke exposure, because they are generic products of combustion of organic materials, two toxicants—nicotine and NNK—are peculiar to tobacco smoke and are known to have adverse health effects. Those compounds or their metabolites can be measured with high sensitivity in various biologic matrices in people exposed to secondhand smoke. Although a number of other tobacco-smoke constituents—such as carbon monoxide, acrolein, benzene, and PAHs and their metabolites—have been used as biomarkers of exposure for active smokers, they are not good biomarkers of exposure to secondhand smoke because they are not unique to secondhand smoke and are present at low levels compared to other sources. Their concentrations in active smokers exceed concentrations seen in most non-smokers, but secondhand smoke contributes only small amounts of them relative to background amounts (for example, from exposures in food and air pollution).
Nicotine and Its Metabolites
Nicotine is present in substantial concentrations in all tobacco products. It is also present in some foods, but the concentrations are much lower, and the contribution of food to the body burden of nicotine and its metabolites is insignificant (Benowitz, 1999). Once nicotine is in the body, hepatic enzymes metabolize it extensively (see Figure 2-1). Nicotine is converted to cotinine, which is converted to trans-3'-hydroxycotinine (3-HC) by the hepatic enzyme cytochrome P450 2A6 (CYP450 2A6) (Hukkanen et al., 2005). Nicotine, cotinine, and 3-HC are converted to their glucuronide metabolites by various uridine diphosphate-glucuronosyl transferase (UGT) enzymes. Cotinine is the major proximate metabolite of nicotine. On average, about 70–80% of nicotine is converted to cotinine, primarily by the liver enzyme CYP450 2A6 (Hukkanen et al., 2005).
Cotinine can be measured in blood, saliva, urine, hair, toenails, and other biologic fluids. The average half-life of cotinine (16 h) in plasma is longer than that of nicotine (2 h). Therefore, cotinine concentrations are more stable throughout the day, and this makes it the preferred biomarker of smoke exposure in blood, saliva, and urine. Both nicotine and cotinine are persistent in hair and toenails. Concentrations of cotinine in blood (including plasma and serum) and saliva are highly correlated and similar. Urinary cotinine concentrations, however, are on average 4–5 times higher than those in blood or saliva, so urine is a more sensitive matrix for detection of low exposure (Benowitz et al., 2009).
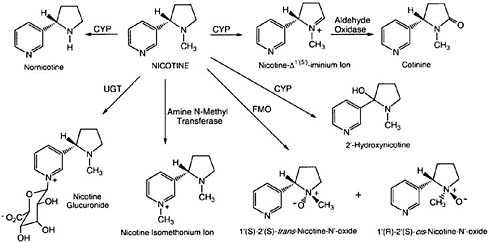
FIGURE 2-1 Primary routes of nicotine metabolism. The figure shows the major routes of nicotine metabolism, with the majority of nicotine being metabolized to cotinine via CYP and aldehyde oxidase. Abbreviations: CYP, cytochrome P450; FMO, flavin-containing monooxygenase; UGT, uridine diphosphate-glucuronosyltransferase.
SOURCE: Hukkanen et al., 2005.
Nicotine is excreted in urine as various metabolites (see Figure 2-1). Excreted nicotine, cotinine, and 3-HC and their glucuronide conjugates account for about 85–90% of a nicotine dose (Hukkanen et al., 2005). Measuring the sum of the metabolites provides a reasonably precise estimate of daily nicotine dose and is the gold standard for biomarker assessment of nicotine exposure.
Interindividual variability in the rate and pattern of nicotine and cotinine metabolism affects the concentration of cotinine that results from a given exposure to nicotine. Factors that may influence nicotine metabolism include genetic variation, race, sex, use of oral contraceptives or other estrogen-containing hormones, renal failure, and use of various medications, such as anticonvulsants and rifampin (Hukkanen et al., 2005). Despite that, cotinine levels are useful to differentiate smokers from non-smokers, to categorize nonsmokers into groups with varying levels of exposure to secondhand smoke, and to track changes in population exposure to secondhand smoke.
NNK Metabolites
NNK is a nicotine-derived nitrosamine that is a potent carcinogen. It is formed primarily in the tobacco-curing process, during which nicotine or pseudo-oxynicotine reacts with nitrite in tobacco (Hecht, 2004). NNK is
metabolized in the body to 4-(N-nitrosomethylamino)-1-(3-pyridyl)-1-butanol (NNAL) and NNAL-glucuronides, which are excreted in urine. NNAL and NNAL-glucuronides are commonly measured together and termed total NNAL. NNAL remains in the body much longer than cotinine, with a terminal half-life of about 3 weeks, so it might be usable for assessing secondhand-smoke exposure over a longer period than cotinine. Although urinary NNAL is sensitive and specific as a biomarker of secondhand-smoke exposure, no studies have evaluated the relationship between urinary NNAL concentration and cardiovascular disease.
EXPOSURES TO SECONDHAND SMOKE
General Trends in Exposure to Secondhand Smoke
Nicotine concentrations measured in diverse environments that allow smoking range over 4 orders of magnitude, from less than 0.1 μg/m3 to several hundred μg/m3. The weekly average concentrations measured in the homes of smokers is typically 0.5–5 μg/m3, with a median of 1 μg/m3 and a mean of 2.2 μg/m3 (Leaderer and Hammond, 1991). The 1 week average nicotine concentrations found in 279 low-income homes with smokers was 3.3 μg/m3 (Emmons et al., 2001). A similar average weekly value, 3.7 μg/m3, was found in the homes of 103 low-income children in Colorado where there were smokers but no strict smoking bans (Wamboldt et al., 2008). One-week sampling of 49 low-income, multi-family homes (including smoking and nonsmoking homes) in the Greater Boston Area found nicotine concentrations ranging from below the limit of detection to 26.92 μg/m3 (Kraev et al., 2009). Clearly secondhand-smoke exposure in the homes of smokers remains high in some cases. The mean and median concentrations were 2.2 and 0.13 μg/m3, respectively, and the concentration was associated with the number of smokers residing in the unit and the number of cigarettes smoked in the home as reported on a questionnaire. Workplace and restaurant concentrations can be over 10 μg/m3, bars over 20 μg/m3, and discotheques over 100 μg/m3 (Hammond, 1999). In a recent study of nine homes with smoking and three smoke-free homes in the United States, PM2.5 measured in real time over a 3-day period averaged 84 μg/m3 in the primary smoking area of the smoking houses, 63 μg/m3 in a distal area from the primary smoking area, and 9 μg/m3 in the nonsmoking homes (Van Deusen et al., 2009).
Over the past 25 years, smoking restrictions and bans in the United States in workplaces, restaurants, and other public places have been increasing, both voluntarily and because of regulations. Their efficacy is seen at the national level in the United States in the 70% decrease in serum cotinine concentrations in 14 years. The data in Figure 2-2 are from the
![FIGURE 2-2 Serum cotinine in nonsmokers in the United States exposed to secondhand smoke, 1988–2002. Serum cotinine geometric means and 95% confidence intervals (CIs) in U.S. nonsmokers by study interval. Data are plotted at approximate midpoint for four periods: 1988–1991 (National Health and Nutrition Examination Survey III [NHANES III], phase 1), 1991–1994 (NHANES III, phase 2), 1999–2000, and 2001–2002.](/openbook/12649/xhtml/images/p2001a02fg42001.jpg)
FIGURE 2-2 Serum cotinine in nonsmokers in the United States exposed to secondhand smoke, 1988–2002. Serum cotinine geometric means and 95% confidence intervals (CIs) in U.S. nonsmokers by study interval. Data are plotted at approximate midpoint for four periods: 1988–1991 (National Health and Nutrition Examination Survey III [NHANES III], phase 1), 1991–1994 (NHANES III, phase 2), 1999–2000, and 2001–2002.
SOURCE: Pirkle et al., 2006.
entire country and include regions with and without smoking regulations, so they reflect the national trend but underestimate the reduction in areas with strong smoke-free regulations (Pirkle et al., 2006).
The effect of voluntary and regulatory smoking bans on exposure of workers can be seen in data from the National Health and Nutrition Examination Survey (NHANES), which show an overall reduction from 1988 to 2002 in serum cotinine concentration in nonsmokers working in the service industry (see Figure 2-3a) (Pickett et al., 2006). Arheart et al. (2008) analyzed serum cotinine concentrations from NHANES data in the same period in workers in different sectors (blue-collar, farm, service industry, and white-collar workers). Serum cotinine concentrations showed a declining trend in all sectors and subgroups analyzed from 1988 to 2002 (Figure 2-3b). Farm workers, who often work outdoors, had the lowest cotinine concentrations initially and the smallest change in those concentrations over time, followed in both respects by white-collar workers. Blue-collar and service-industry workers had higher concentrations initially
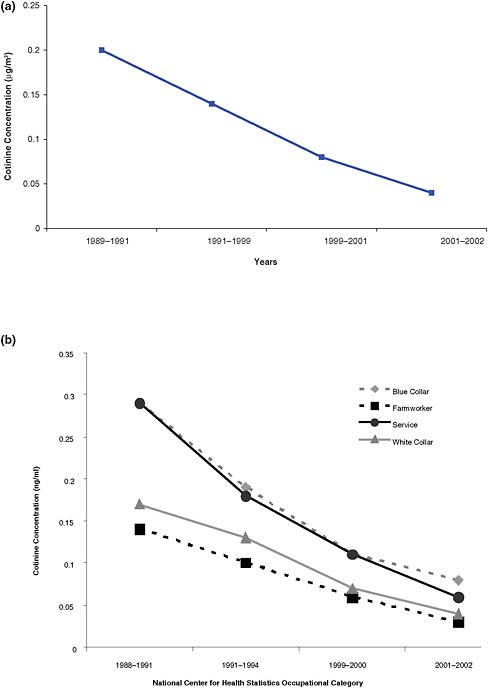
FIGURE 2-3 Serum cotinine in select U.S. populations, 1988–2002. Concentrations were measured in National Health and Nutrition Examination Surveys (NHANES). (a) service workers. Data from Pickett et al., 2006; (b) blue-collar workers, farm-workers, service workers, white-collar workers. Data from Arheart et al., 2008.
and had the greatest declines by 2002. The serum cotinine concentrations in the NHANES data integrate all exposure, including home exposure, and include data from regions where and times when smoking bans were not in place so the reductions in secondhand-smoke exposure when there were smoking bans are much greater than observed here. The reduction in exposure, therefore, could in part reflect voluntary smoking bans in private workplaces, which increased.
Changes in PM, nicotine, and cotinine concentrations after the implementation of smoking bans have been studied. Some of the studies are summarized below.
Airborne Particulate Matter Before and After Smoking Bans
Estimating the contribution of secondhand smoke to airborne PM concentrations requires consideration of background concentrations of respirable particles. Estimates vary widely because of differences in measurement techniques and geographic location. Leaderer and Hammond (1991) have reported PM concentrations in 96 randomly selected residences in New York state: the mean respirable PM in nonsmoking residences was 15 μg/m3, compared with 44 μg/m3 in smoking homes (average of 71 cigarettes smoked in the homes during the week), which could be considered typical of U.S. residences. The extent to which tobacco smoke contributes to a difference from the background concentration varies greatly. A representative study by Spengler et al. (1981) found that in 35 nonsmoking homes the respirable particle concentration averaged 24 μg/m3, while homes with one smoker averaged 36 μg/m3 and homes with two smokers averaged 70 μg/m3. In recent studies, concentrations of PM2.5 in German bars, restaurants, and discotheques where smoking is permitted have been reported to be between 178 and 808 μg/m3 measured over 4 hours (Bolte et al., 2008). The particle number concentration was between 120,000 and 210,000 particles per cm3 and the majority of particles had a size of 0.01–0.5 μm. These results are similar to earlier data obtained from restaurants and bars in Vancouver, British Columbia, which showed a concentration range of 47–253 μg/m3 for PM2.5 and 51–268 μg/m3 for PM10 in restaurants with unrestricted smoking measured over 6 hours (Brauer and Mannetje, 1998). A survey of 32 countries found high levels of PM2.5 (200–300 μg/m3) in countries without a smoking ban (Hyland et al., 2008). Furthermore, significant decreases in PM levels in restaurants have been reported after introduction of smoke-free legislation. In England measurements collected from 49 businesses show a 95% decrease in PM2.5 levels from 217 to 11 μg/m3 (Gotz et al., 2008). Similar levels of reduction in PM2.5 levels of 77% have been reported after implementation of a smoking ban in a North
Carolina correctional facility (Proescholdbell et al., 2008) and of 71–99% in restaurants in Austin, Texas (Waring and Siegel, 2007).
Airborne PM2.5 concentrations in restaurants decreased significantly in Ohio, Massachusetts, New York state, Norway, and Italy after the implementation of smoking bans (Akbar-Khanzadeh et al., 2004; Alpert et al., 2007; CDC, 2004, 2007; Ellingsen et al., 2006; Valente et al., 2007). As can be seen in Figure 2-4a, concentrations decreased from 194 to 67 µg/m3, from 206 to 14 µg/m3, and from 248 to 23.1 µg/m3 in Ohio, Massachusetts, and New York state, respectively. Decreases in Norway and Italy were smaller—from 115 to 77 µg/m3 and from 110 to 61 µg/m3 (CDC, 2004; Ellingsen et al., 2006; Valente et al., 2007)—but initial PM2.5 concentrations in European restaurants were lower pre-ban than in the United States. It is important to note that because of the presence of background PM2.5, PM2.5 concentrations will not reach zero even with 100% compliance with a ban, and most of the concentrations measured after the ban are close to outdoor, or background, concentrations.
Data from New York state, Norway, Scotland, and Italy demonstrated substantial reductions in PM concentrations in other public areas (such as bars and bowling alleys) after implementation of smoking bans (Figure 2-4b). PM2.5 concentrations before smoking bans varied widely among studies, from 61 to 549 µg/m3 in different indoor settings, but decreased greatly after implementation, to 6 to 40 µg/m3 in most venues (background concentrations of PM2.5 are typically about 15–30 µg/m3). PM2.5 decreased, for example, in a study of New York state bars from 549 to about 33 µg/m3 (CDC, 2004), in New York state bowling alleys from 61 to about 20 µg/m35 (CDC, 2004), and in Scottish pubs from 246 to 20 µg/m3 (Semple et al., 2007). Complete smoking bans in the United States, e.g., Massachusetts and New York, led to significant decreases, typically 90%, in PM2.5 concentrations to near background levels, except in bars. In Italy and Scotland, post-ban levels in restaurants were half of pre-ban levels and 10–40% of pre-ban levels in other public places.
Airborne Nicotine
Airborne concentrations of nicotine, a tracer that is specific for tobacco smoke, decreased even more dramatically after the implementation of smoking bans. Because there are no other important sources of nicotine, the background airborne concentration of nicotine should be zero.
Data from three countries demonstrated that smoking bans in restaurants led to greater than 90% reductions in airborne nicotine. In Ohio
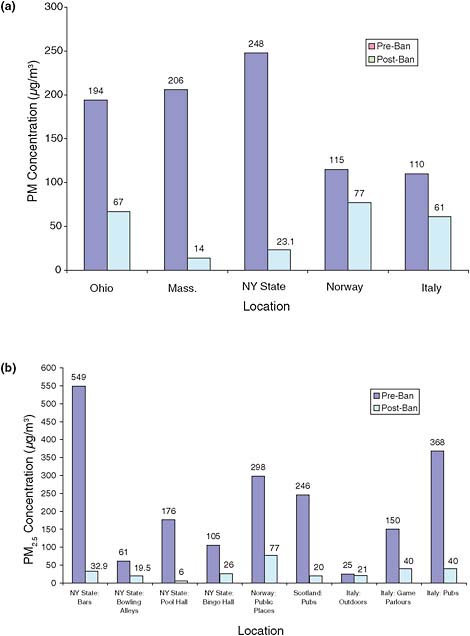
FIGURE 2-4 Airborne PM2.5 concentrations in (a) restaurants and (b) public places before and after implementation of smoking bans. Data from Akbar-Khanzadeh et al. (2004) examined exposure in Ohio, which implemented a clean air ordinance allowing smoking in separate sections in restrauants. Data from Akbar-Khanzadeh et al., 2004; Alpert et al., 2007; CDC, 2004; Elligsen et al., 2006; Semple et al., 2007; and Valente et al., 2007.
(Akbar-Khanzadeh et al., 2004), Norway (Ellingsen et al., 2006), and Florence and Belluno, Italy (Gorini et al., 2008), nicotine concentrations in restaurants decreased from 9.8, 28.3, and 2 μg/m3, respectively, to less than 0.1 μg/m3 after implementation of smoking bans (Figure 2-5a), that is, to less than 5%, and usually <1% pre-ban levels.
Similarly, in other public places (such as pubs and discos), smoking bans resulted in large decreases in airborne nicotine concentrations (Figure 2-5b), from concentrations about 35–165 μg/m3 to less than 6 μg/m3 after bans were implemented in Norway, Italy, and Ireland (Ellingsen et al., 2006; Gorini et al., 2005, 2008; Mulcahy et al., 2005). For example, Mulcahy et al. (2005) measured the effect of the Irish smoking ban on airborne nicotine concentrations in pubs and cotinine concentrations in hospitality workers. In a sample of 20 bars in Galway, Ireland, air nicotine decreased by 83% (from a median of 36 to 6 μg/m3; p < 0.001) between the Friday night preceding the ban and 5 weeks after the ban was implemented (Mulcahy et al., 2005).
In the first multicenter study in Europe, Nebot et al. (2005) measured nicotine vapor concentrations in public places that included transportation, education, and leisure settings (Nebot et al., 2005). The study used passive samplers placed in public places for 4 h to 2 weeks. In cities in seven European countries (in Vienna, Paris, Athens, Florence, Oporto, Barcelona, and Orebro), nicotine concentrations were highest in bars and discos (median 19 μg/m3, values up to 122 μg/m3), followed by restaurants, airports, and train stations (Nebot et al., 2005). Schools and hospitals had the lowest concentration of nicotine.
In the absence of smoke-free policies, nicotine concentrations in offices can be high. Reduction in workplace exposure to secondhand smoke can have a large effect on overall exposure to secondhand smoke because of the exposure duration of what is typically an 8-h workday in the United States.
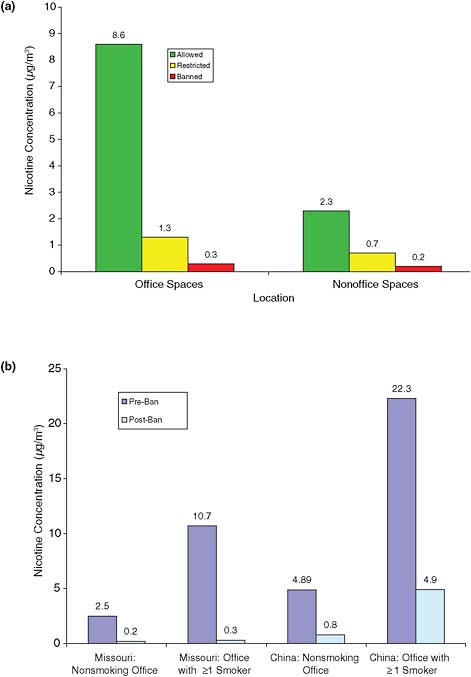
Research has compared concentrations of nicotine in workplaces that allowed smoking to those that have policies restricting smoking to a few designated areas and workplaces that have policies that ban smoking in the workplace (see Figure 2-6) (Hammond, 1999). Nicotine concentrations were lower (generally under 1 μg/m3) in workplaces that banned smoking than in workplaces that allowed smoking (mean concentrations, generally 2–6 μg/m3 in offices, 3–8 μg/m3 in restaurants, and 1–6 μg/m3 in blue-collar workplaces). Hammond (1999) also reported that workplace concentrations are variable but could be more than 10 times higher than average home concentrations and that for 30% of workers the workplace is the principal source of secondhand smoke (Hammond, 1999).
In a cross-sectional study, sampling at nonsmokers’ desks or workstations in 25 Massachusetts office and nonoffice workplaces (such as
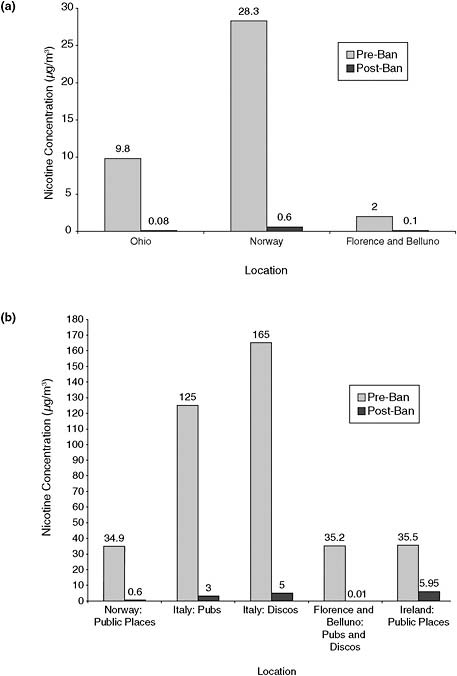
FIGURE 2-5 Airborne nicotine concentrations in (a) restaurants and (b) other public places before and after implementation of smoking bans. Nicotine concentrations represent median not mean amounts in Ireland study. All other data represent mean nicotine concentrations. Data from Akbar-Khanzadeh et al., 2004; Ellingsen et al., 2006; Gorini et al., 2005, 2008; and Mulcahy et al., 2005.
manufacturing, printing workplaces, and fire stations) found that nonoffice workplaces that allowed smoking had nicotine concentrations of 0.1 to over 20 μg/m3 (median, 2.3 μg/m3). Open offices with several workers had even higher concentrations: a median of 8.6 μg/m3 and some values over 40 μg/m3 (Figure 2-6a) (Hammond et al., 1995). Those values were markedly different among the companies that did not allow smoking indoors; for nonsmokers, median nonoffice values dropped from 2.3 to 0.2 μg/m3 and median values in open offices from 8.6 to 0.3 μg/m3 (see Figure 2-6a).
Some research has shown that those who live in homes with smokers who smoke in the home benefit from nonsmoking workplaces. In a reanalysis of the data from the 16 Cities Study (Jenkins et al., 1996)6 to stratify home smoking status and compare exposures by workplace smoking status, people who were exposed to smoking both at home and at work had over twice the 24-h average exposure compared to those who were exposed in the home but not at work (Barnes et al., 2006). The authors concluded that “if workplaces were smoke-free, the total SHS [secondhand smoke] exposure of those living with smokers could be cut in half, and the total SHS exposure of those living in nonsmoking homes would become negligible, a significant worker safety and public health benefit” (Barnes et al., 2006).
Direct evidence that policies banning smoking in the workplace reduce airborne nicotine can be seen in two studies in which nicotine was measured in offices before and after smoking restrictions were implemented (Vaughan and Hammond, 1990). Vaughan and Hammond (1990) measured nicotine in 30 office locations in one building in Missouri before and after control of secondhand smoke (see Figure 2-6b). Nicotine vapors in the air were measured with passive filters and active pumps. Before the ban, offices with more than one smoker were sometimes shared with nonsmokers. The authors found over a 90% reduction in nicotine concentrations measured at workers’ desks after smoking was restricted to the snack bar. Nicotine vapor concentrations decreased in smoker, nonsmoker, and vacant spaces by 81–98% (Vaughan and Hammond, 1990).
A study of 14 office buildings in China evaluated weekly average nicotine concentrations in buildings according to their smoking policies regardless of extent of enforcement (see Figure 2-6b) (Gan et al., 2008). In addition, one building was sampled before and after a smoke-free policy was implemented. The authors found that
For all 14 buildings, offices in buildings with smoking policies had less than half SHS as offices without smoking policies. In one building where we sampled the air before and after a smoke-free policy was implemented on January 1, 2006, the SHS concentrations decreased significantly after the policy was enacted.
For example, nicotine concentrations in offices with at least one smoker fell 90% from 18.8 to 1.9 μg/m3.
Biomarkers of Secondhand-Smoke Exposure Before and After Smoking Bans
Evidence indicates that the implementation of smoking bans is effective in reducing individual exposures to secondhand smoke but that exposures do not decrease to zero, because there are other sources of exposure (such as homes and vehicles). Most of the data come from workers in public establishments, such as restaurants, bars, and hotels.
Al-Delaimy et al. (2001) measured nicotine concentrations in the hair of bar and restaurant workers in Wellington and Auckland, New Zealand, when partial smoking restrictions were in place that required restaurants to designate 50% of seating as smoke-free and bars were exempt from restrictions. In nonsmokers, hair nicotine varied with the type of smoke-free policy in the workplace, which was categorized as 100% smoke-free, 50% smoke-free, or no restrictions. People working in smoke-free establishments had significantly lower hair nicotine concentrations (0.62 ng/mg; Kruskal-Wallis χ2 = 26.4; p < 0.0001) than people in 50% smoke-free establishments (2.72 ng/mg) or establishments with no restrictions (6.69 ng/mg).
In Norway, Ellingsen et al. (2006) showed decreased exposure to secondhand smoke, as demonstrated by decreased cotinine concentrations, in the urine of nonsmoking employees of restaurants and bars and decreased air concentrations of nicotine and decreased total dust concentrations in the 13 establishments surveyed after the implementation of a ban on smoking in bars and restaurants.
Data on employees of public establishments in New York state (Farrelly et al., 1999), Scotland (Menzies et al., 2006), Ireland (Mulcahy et al., 2005), and Italy (Valente et al., 2007) demonstrate large decreases in exposure after implementation of smoking bans (see Figure 2-7a). In the New York state study, saliva cotinine concentrations decreased from 3.6 to 0.78 ng/mL; in Scotland, serum cotinine concentrations decreased from 5.15 to 2.93 ng/mL; in Ireland, salivary cotinine concentrations decreased from 2.86 to 1.29 ng/mL; and in Italy, urinary cotinine concentrations decreased from 17.8 to 5.5 ng/mL. In Ireland (Mulcahy et al., 2005), data were categorized by type of staff in hotels (Figure 2-7b): waiters had the largest
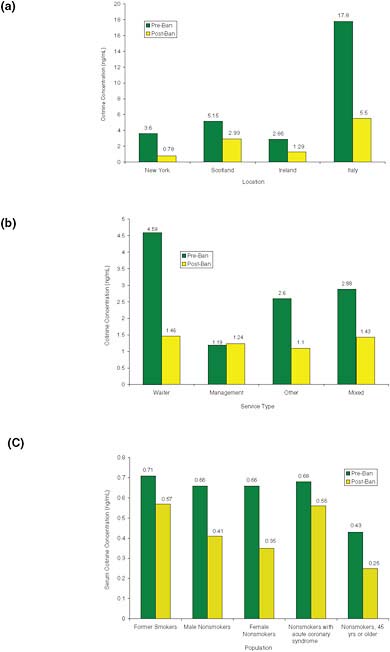
FIGURE 2-7 Exposures to secondhand smoke in (a) workers in public establishments, (b) hotel staff in Ireland, and (c) former smokers and nonsmokers in Scotland. Data from New York state and Ireland are salivary cotinine concentrations. Data from Scotland are serum cotinine concentrations. Data from Italy are urinary cotinine concentrations. Data from Farrelly et al., 2005; Menzies et al., 2006; Mulcahy et al., 2005; Pell et al., 2008; and Valente et al., 2007.
decrease in salivary cotinine, from 4.59 to 1.46 ng/mL, and management had a low cotinine concentration both before and after the ban (1.19 and 1.24 ng/mL, respectively).
Pell et al. (2008) showed reductions in serum cotinine concentrations in a variety of demographic groups after implementation of the Scottish smoking ban, including former smokers, male and female nonsmokers, nonsmokers with acute coronary syndrome, and nonsmokers over 45 years old (Figure 2-7c) (Pell et al., 2008). The largest decreases occurred in nonsmokers.
Pickett et al. (2006) used data from the NHANES surveys to examine the relationship between smoke-free laws and secondhand-smoke exposure of nonsmoking adults in the United States. The authors categorized 57 NHANES locations as to their smoke-free law coverage (“extensive,” “limited,” or “no laws”) and looked at serum cotinine concentrations in nonsmokers, as defined by self-reported smoking status and serum cotinine concentrations (a concentration below 10 ng/mL was considered that of a nonsmoker). Both male and female nonsmokers living in areas with extensive smoke-free laws had significantly lower probabilities of having detectable cotinine (at least 0.05 ng/mL) than those who lived in areas without smoke-free laws. For example, the percentage of nonsmoking men with detectable cotinine dropped from 57% in areas with only limited restriction to only 10% in areas with extensive smoke-free regulations; for women, the decline was from 90% to 19%.
CONCLUSIONS
-
Airborne tracers of secondhand smoke and biomarkers of exposure to secondhand smoke are complementary. Airborne tracers measure concentrations in specific venues while biomarkers integrate all sources of exposure and incorporate inhalation rates. Because of its short half-life, cotinine reflects only recent exposures. NNAL has a longer half-life, but has not been used as widely. Concentrations of cotinine in serum, saliva, and urine are specific indicators of total exposure to secondhand smoke. Airborne measures of exposure can demonstrate the contribution of different sources or venues of exposure but do not reflect total dose unless all venues are measured.
-
The concentration of airborne nicotine is a specific tracer for secondhand smoke. PM can also be used as an indication of secondhand-smoke exposure but, because there are other sources of PM, it is a less specific tracer than nicotine.
-
Both airborne monitoring studies and biomonitoring studies demonstrate that exposure to secondhand smoke is substantially re-
-
duced after implementation of smoking bans. Air concentrations of nicotine and PM decreased by more than 80% in restaurants, bars, and workplaces in most studies after smoking bans were implemented; serum and salivary cotinine concentrations decreased by 50% or more in most studies. The residual concentration reflects continued exposure in unregulated areas, such as homes.
REFERENCES
Akbar-Khanzadeh, F., S. Milz, A. Ames, S. Spino, and C. Tex. 2004. Effectiveness of clean indoor air ordinances in controlling environmental tobacco smoke in restaurants. Archives of Environmental Health 59(12):677-685.
Al-Delaimy, W., T. Fraser, and A. Woodward. 2001. Nicotine in hair of bar and restaurant workers. New Zealand Medical Journal 114(1127):80-83.
Alpert, H. R., C. M. Carpenter, M. J. Travers, and G. N. Connolly. 2007. Environmental and economic evaluation of the Massachusetts smoke-free workplace law. Journal of Community Health 32(4):269-281.
Arheart, K. L., D. J. Lee, N. A. Dietz, J. D. Wilkinson, J. D. Clark, 3rd, W. G. LeBlanc, B. Serdar, and L. E. Fleming. 2008. Declining trends in serum cotinine levels in US worker groups: The power of policy. Journal of Occupational & Environmental Medicine 50(1):57-63.
Barnes, R. L., and S. A. Glantz. 2007. Endotoxins in tobacco smoke: Shifting tobacco industry positions. Nicotine and Tobacco Research 9(10):995-1004.
Barnes, R. L., S. K. Hammond, and S. A. Glantz. 2006. The tobacco industry’s role in the 16 Cities Study of secondhand tobacco smoke: Do the data support the stated conclusions? Environmental Health Perspectives 114(12):1890-1897.
Benner, C., J. M. Bayona, F. M. Caka, H. Tang, L. Lewis, J. Crawford, J. Lamb, M. I. Lee, E. A. Lewis, L. D. Hansen, and D. J. Eatough. 1989. Chemical composition of environmental tobacco smoke. 2. Particulate phase compounds. Environmental Science Technology 23:688-699.
Benowitz, N. L. 1999. The biology of nicotine dependence: From the 1988 surgeon general’s report to the present and into the future. Nicotine & Tobacco Research 1 Suppl 2: S159-S163.
Benowitz, N. L., J. T. Bernert, R. S. Caraballo, D. B. Holiday, and J. Wang. 2009. Optimal serum cotinine levels for distinguishing cigarette smokers and nonsmokers within different racial/ethnic groups in the United States between 1999 and 2004. American Journal of Epidemiology 169(2):236-248.
Bolte, G., D. Heitmann, M. Kiranoglu, R. Schierl, J. Diemer, W. Koerner, and H. Fromme. 2008. Exposure to environmental tobacco smoke in German restaurants, pubs and discotheques. Journal of Exposure Science & Environmental Epidemiology 18(3):262-271.
Brauer, M., and A. Mannetje. 1998. Restaurant smoking restrictions and environmental tobacco smoke exposure. American Journal of Public Health 88(12):1834-1836.
Cal EPA (California Environmental Protection Agency). 2005a. Proposed identification of environmental tobacco smoke as a toxic air contaminant. Part A: Exposure assessment. Sacramento: California Environmental Protection Agency.
———. 2005b. Proposed identification of environmental tobacco smoke as a toxic air contaminant. Part B: Health effects. Sacramento: California Environmental Protection Agency.
CDC (Centers for Disease Control and Prevention). 2004. Indoor air quality in hospitality venues before and after implementation of a clean indoor air law—western New York, 2003. MMWR—Morbidity & Mortality Weekly Report 53(44):1038-1041.
———. 2006. Fact sheet: Secondhand smoke. (Accessed December 2008, from http://www.cdc.gov/tobacco/data_statistics/fact_sheets/secondhand_smoke/secondhandsmoke.htm).
———. 2007. Reduced secondhand smoke exposure after implementation of a comprehensive statewide smoking ban—New York, June 26, 2003-June 30, 2004. MMWR—Morbidity & Mortality Weekly Report 56(28):705-708.
Daisey, J. M. 1999. Tracers for assessing exposure to environmental tobacco smoke: What are they tracing? Environmental Health Perspectives 107 Suppl 2:319-327.
Eatough, D., C. Benner, J. Bayona, G. Richards, J. Lamb, M. Lee, E. Lewis, and L. Hansen. 1989. Chemical composition of environmental tobacco smoke. 1. Gas-phase acids and bases. Environmental Science Technology 23(6):679-687.
Ellingsen, D. G., G. Fladseth, H. L. Daae, M. Gjolstad, K. Kjaerheim, M. Skogstad, R. Olsen, S. Thorud, and P. Molander. 2006. Airborne exposure and biological monitoring of bar and restaurant workers before and after the introduction of a smoking ban. Journal of Environmental Monitoring 8(3):362-368.
Emmons, K. M., S. K. Hammond, J. L. Fava, W. F. Velicer, J. L. Evans, and A. D. Monroe. 2001. A randomized trial to reduce passive smoke exposure in low-income households with young children. Pediatrics 108(1):18-24.
Farrelly, M. C., W. N. Evans, and A. E. Sfekas. 1999. The impact of workplace smoking bans: Results from a national survey. Tobacco Control 8(3):272-277.
Gan, Q., S. K. Hammond, Y. Jiang, Y. Yang, and T. W. Hu. 2008. Effectiveness of a smoke-free policy in lowering secondhand smoke concentrations in offices in China. Journal of Occupational & Environmental Medicine 50(5):570-575.
Gorini, G., A. Gasparrini, M. C. Fondelli, A. S. Costantini, F. Centrich, M. J. Lopez, M. Nebot, and E. Tamang. 2005. Environmental tobacco smoke (ETS) exposure in Florence hospitality venues before and after the smoking ban in Italy. Journal of Occupational & Environmental Medicine 47(12):1208-1210; author reply 1210.
Gorini, G., H. Moshammer, L. Sbrogio, A. Gasparrini, M. Nebot, M. Neuberger, E. Tamang, M. J. Lopez, D. Galeone, and E. Serrahima. 2008. Italy and Austria Before and After study: Second-hand smoke exposure in hospitality premises before and after 2 years from the introduction of the Italian smoking ban. Indoor Air 18(4):328-334.
Gotz, N. K., M. van Tongeren, H. Wareing, L. M. Wallace, S. Semple, and L. Maccalman. 2008. Changes in air quality and second-hand smoke exposure in hospitality sector businesses after introduction of the English smoke-free legislation. Journal of Public Health (Oxford, England) 30(4):421-428.
Hammond, S. K. 1999. Exposure of U.S. workers to environmental tobacco smoke. Environmental Health Perspectives 107 Suppl 2:329-340.
Hammond, S. K., J. Coghlin, P. H. Gann, M. Paul, K. Taghizadeh, P. L. Skipper, and S. R. Tannenbaum. 1993. Relationship between environmental tobacco smoke exposure and carcinogen-hemoglobin adduct levels in nonsmokers. Journal of the National Cancer Institute 85(6):474-478.
Hammond, S. K., G. Sorensen, R. Youngstrom, and J. K. Ockene. 1995. Occupational exposure to environmental tobacco smoke. JAMA 274(12):956-960.
Hecht, S. S. 2004. Carcinogen derived biomarkers: Applications in studies of human exposure to secondhand tobacco smoke. Tobacco Control 13 Suppl 1:i48-i56.
Hinds, W. C. 1999. Aerosol technology: Properties, behavior, and measurement of airborne particles. 2nd ed. New York: John Wiley & Sons, Inc.
Hukkanen, J., P. Jacob, 3rd, and N. L. Benowitz. 2005. Metabolism and disposition kinetics of nicotine. Pharmacological Review 57(1):79-115.
Hyland, A., M. J. Travers, C. Dresler, C. Higbee, and K. M. Cummings. 2008. A 32-country comparison of tobacco smoke derived particle levels in indoor public places. Tobacco Control 17(3):159-165.
Jenkins, R. A., A. Palausky, R. W. Counts, C. K. Bayne, A. B. Dindal, and M. R. Guerin. 1996. Exposure to environmental tobacco smoke in sixteen cities in the United States as determined by personal breathing zone air sampling. Journal of Exposure Analysis and Environmental Epidemiology 6(4):473-502.
Jenkins, R., M. R. Guerin, and B. A. Tomkins. 2000. The chemistry of environmental tobacco smoke: Composition and measurement. Boca Raton, FL: Lewis Publishers.
Kraev, T. A., G. Adamkiewicz, S. K. Hammond, and J. D. Spengler. 2009. Indoor concentrations of nicotine in low-income, multi-family housing: Associations with smoking behaviors and housing characteristics. Tobacco Control 18(6):438-444.
Kritz, H., P. Schmid, and H. Sinzinger. 1995. Passive smoking and cardiovascular risk. Archives of Internal Medicine 155(18):1942-1948.
Leaderer, B. P., and S. K. Hammond. 1991. Evaluation of vapor-phase nicotine and respirable suspended particle mass as markers for environmental tobacco smoke. Environmental Science and Technology 25(4):770-777.
Lodovici, M., V. Akpan, C. Evangelisti, and P. Dolara. 2004. Sidestream tobacco smoke as the main predictor of exposure to polycyclic aromatic hydrocarbons. Journal of Applied Toxicology 24(4):277-281.
Menzies, D., A. Nair, P. A. Williamson, S. Schembri, M. Z. H. Al-Khairalla, M. Barnes, T. C. Fardon, L. McFarlane, G. J. Magee, and B. J. Lipworth. 2006. Respiratory symptoms, pulmonary function, and markers of inflammation among bar workers before and after a legislative ban on smoking in public places. JAMA 296(14):1742-1748.
Mulcahy, M., D. S. Evans, S. K. Hammond, J. L. Repace, and M. Byrne. 2005. Secondhand smoke exposure and risk following the Irish smoking ban: An assessment of salivary cotinine concentrations in hotel workers and air nicotine levels in bars. Tobacco Control 14(6):384-388.
Nebot, M., M. J. Lopez, G. Gorini, M. Neuberger, S. Axelsson, M. Pilali, C. Fonseca, K. Abdennbi, A. Hackshaw, H. Moshammer, A. M. Laurent, J. Salles, M. Georgouli, M. C. Fondelli, E. Serrahima, F. Centrich, and S. K. Hammond. 2005. Environmental tobacco smoke exposure in public places of European cities. Tobacco Control 14(1):60-63.
NRC (National Research Council). 1986. Environmental tobacco smoke: Measuring exposures and assessing health effects. Washington, DC: National Academy Press.
———. 2006. Human biomonitoring for environmental chemicals. Washington, DC: The National Academies Press.
Pell, J. P., S. Haw, S. Cobbe, D. E. Newby, A. C. H. Pell, C. Fischbacher, A. McConnachie, S. Pringle, D. Murdoch, F. Dunn, K. Oldroyd, P. Macintyre, B. O’Rourke, and W. Borland. 2008. Smoke-free legislation and hospitalizations for acute coronary syndrome. New England Journal of Medicine 359(5):482-491.
Pickett, M. S., S. E. Schober, D. J. Brody, L. R. Curtin, and G. A. Giovino. 2006. Smoke-free laws and secondhand smoke exposure in US non-smoking adults, 1999-2002. Tobacco Control 15(4):302-307.
Pirkle, J. L., J. T. Bernert, S. P. Caudill, C. S. Sosnoff, and T. F. Pechacek. 2006. Trends in the exposure of nonsmokers in the U.S. population to secondhand smoke: 1988-2002. Environmental Health Perspectives 114(6):853-858.
Proescholdbell, S. K., K. L. Foley, J. Johnson, and S. H. Malek. 2008. Indoor air quality in prisons before and after implementation of a smoking ban law. Tobacco Control 17(2):123-127.
Schick, S., and S. Glantz. 2005. Philip Morris toxicological experiments with fresh sidestream smoke: More toxic than mainstream smoke. Tobacco Control 14(6):396-404.
Schick, S., and S. A. Glantz. 2006. Sidestream cigarette smoke toxicity increases with aging and exposure duration. Tobacco Control 15(6):424-429.
Schick, S. F., and S. Glantz. 2007. Concentrations of the carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone in sidestream cigarette smoke increase after release into indoor air: Results from unpublished tobacco industry research. Cancer Epidemiology, Biomarkers & Prevention 16(8):1547-1553.
Semple, S., K. S. Creely, A. Naji, B. G. Miller, and J. G. Ayres. 2007. Secondhand smoke levels in Scottish pubs: The effect of smoke-free legislation. Tobacco Control 16(2):127-132.
Singer, B. C., A. T. Hodgson, K. S. Guevarra, E. L. Hawley, and W. W. Nazaroff. 2002. Gas-phase organics in environmental tobacco smoke. 1. Effects of smoking rate, ventilation, and furnishing level on emission factors. Environmental Science Technology 36(5):846-853.
Spengler, J. D., D. W. Dockery, W. A. Turner, J. M. Wolfson, and B. G. Ferris Jr. 1981. Long-term measurements of respirable particulates and implications for air pollution epidemiology. Atmospheric Environment 15:23-30.
Valente, P., F. Forastiere, A. Bacosi, G. Cattani, S. Di Carlo, M. Ferri, I. Figa-Talamanca, A. Marconi, L. Paoletti, C. Perucci, and P. Zuccaro. 2007. Exposure to fine and ultrafine particles from secondhand smoke in public places before and after the smoking ban, Italy 2005. Tobacco Control 16(5):312-317.
Van Deusen, A., A. Hyland, M. J. Travers, C. Wang, C. Higbee, B. A. King, T. Alford, and K. M. Cummings. 2009. Secondhand smoke and particulate matter exposure in the home. Nicotine & Tobacco Research 11(6):635-641.
Vaughan, W. M., and S. K. Hammond. 1990. Impact of “designated smoking area” policy on nicotine vapor and particle concentrations in a modern office building. Journal of the Air & Waste Management Association 40(7):1012-1017.
Wamboldt, F. S., R. C. Balkissoon, A. E. Rankin, S. J. Szefler, S. K. Hammond, R. E. Glasgow, and W. P. Dickinson. 2008. Correlates of household smoking bans in low-income families of children with and without asthma. Fam Process 47(1):81-94.
Waring, M. S., and J. A. Siegel. 2007. An evaluation of the indoor air quality in bars before and after a smoking ban in Austin, Texas. Journal of Exposure Science & Environmental Epidemiology 17(3):260-268.