Two Private-Sector Approaches to Benefit Coverage and Design
To further inform its understanding of how insurers reach decisions on covering benefits, the Institute of Medicine (IOM) committee held a second public workshop, starting off with presentations by a health insurer, WellPoint, and an integrated delivery system with both insurance and delivery components, Kaiser Permanente. Samuel Nussbaum, Executive Vice President, Clinical Health Policy and Chief Medical Officer at WellPoint, started off by describing that company’s approach to building health care improvement into its choice of evidence-based benefits, clinical policies, modes of delivery, and incentives. He stressed that deciding the covered benefits is just the first step, followed by benefit design choices and benefit administration. Sharon Levine, the Associate Executive Medical Director of The Permanente Medical Group, reviewed its framework for deciding benefits, how market factors influence those choices, Kaiser’s environment of examined practice, and variations across plans (e.g., choices in contractual exclusions, requirements of state mandates). These private-sector approaches are illustrative of many steps in defining a benefit package and its implementation.
PRESENTATION BY DR. SAMUEL NUSSBAUM, WELLPOINT, INC.
Dr. Nussbaum began by describing the complementary factors that plans must consider when defining benefits. First, as 5 percent of WellPoint’s 34 million members drive 54 percent of its costs, WellPoint aims to design benefits in a way that promotes health and wellness (Nussbaum, 2011). “Baking health improvement into the benefit package,” he said, will improve health and lower cost. Second, because unsustainable health care costs “actually threaten what we can achieve in terms of technology advances and scientific innovation,” WellPoint’s benefit design aims to balance affordability with value, quality, and effectiveness. WellPoint invests in treatments that have been scientifically proven, and the company works to reduce the use of inappropriate and ineffective treatments. The company monitors improvement in 40 quality measures across domains of screening and prevention, care management, clinical outcomes, and patient safety, and also compares health status achievement among its members.
Dr. Nussbaum cited WellPoint’s process for assessing medical technology as an example of how WellPoint seeks to improve health care and lower costs while building on a foundation of proven clinical science and outcomes. The health plan makes evidence-based decisions about whether and when benefits should be covered after considering emerging science, expert clinical opinions, and reviews of the medical literature. As described in Figure 10-1, committees of clinical experts on specific subjects such as hematology, oncology, and behavioral health guide these decisions. In response to a question from committee member Dr. Robert Galvin requesting more
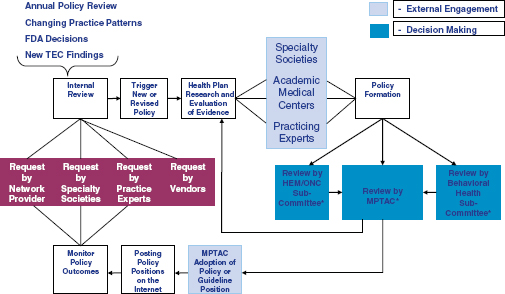
FIGURE 10-1 WellPoint, Inc. has various paths for reviewing benefit coverage to make medical policy decisions.
*Depending on the subject, different review committees will be engaged. This illustration includes the Hematology/ Oncology Committee (HEM/ONC), the Medical Policy & Technology Assessment Committee (MPTAC) and the Behavioral Health subcommittee; each of these includes external physician representation.
SOURCE: Nussbaum, 2011.
specific details about who makes these evidence-based decisions, Dr. Nussbaum clarified that “for clinical decisions, we largely use external experts.” For example, if a new therapy was available for cancer, WellPoint would rely on scientists and clinicians to rigorously assess the evidence for treatment.
This exchange prompted committee member Dr. Santa to explore whether the process depicted in Figure 10-1 allows providers to be “in the position of making medical necessity determinations.” Dr. Nussbaum explained that WellPoint’s benefit decisions and medical policies guide the medical determination. Although WellPoint has well-developed processes that allow physician-to-physician dialogue and decisions based on unique needs of the individual patient, physicians cannot make decisions irrespective of medical policy and coverage documents. Physician groups, for example, cannot independently determine that a new cancer chemotherapy is covered, but can and do contribute their viewpoints to the medical policy decision process. All evidence-based medical policies are available on the company’s website.
Health Care Cost Drivers
Benefit design, Dr. Nussbaum cautioned, is not the only driver of the use of health care services. Even within the same benefits package, there is “profound variation in the use of services.” This variation is not necessarily driven by what the benefit package covers. As committee member Dr. McGlynn’s research has shown that patients inconsistently receive recommended care—for example, only 40.7 percent of children who saw a health care provider received recommended pediatric preventive care (Mangione-Smith et al., 2007). Dr. Nussbaum reasoned that health care utilization is also impacted by external factors such as a reimbursement system that rewards volume
over quality or outcomes, expanding capacity that triggers increased demand, patients’ preferences that are not based on sufficient knowledge of the effectiveness of alternate treatments, and lack of integration and coordination among clinicians across the delivery system. Clinicians, at times, do not make decisions that are “based on the best scientific knowledge.”
Complexities in Defining the EHB
Dr. Nussbaum outlined four complexities WellPoint faces in providing for the essential health benefits (EHB) defined in the Patient Protection and Affordable Care Act (ACA). First, how can WellPoint ensure that services are provided in high value centers? Second, how can WellPoint continue to focus on value-based insurance design (VBID)? Third, how can WellPoint develop benefits that meet the specific needs of diverse populations? And fourth, how can WellPoint address the variation in benefits that exists across states and employers? Dr. Nussbaum proceeded to describe how WellPoint is approaching each of these complex questions.
High Value Centers
WellPoint’s network design helps to ensure the provision of high-quality care. For example, by only covering coronary artery bypass graft surgery and percutaneous cardiac interventions when performed in facilities qualified as “Blue Distinction Centers of Excellence,” WellPoint’s enrollees benefit from technological advances in cardiac care while WellPoint maximizes the best clinical outcomes. In addition, treatment in these Centers of Excellence costs between $4,000 and $9,000 less per event because patients have fewer complications and reduced readmis-sions (Nussbaum, 2011). This example shows why WellPoint has developed Centers of Excellence, particularly for transplants, cardiac and bariatric surgery, select orthopedic procedures, and rare and complex cancer treatment.
Similarly, to decrease costs and improve care, WellPoint’s ambulatory services benefit design encourages cost-effective treatment by separating coverage for urgent medical needs that require emergency room (ER) services and those medical needs that do not require ER services. WellPoint spends approximately $600 every time a member visits the ER for treatment for a medical need not requiring ER services (e.g., sinusitis, pharyngitis, otitis media), whereas treatment for these common conditions in a primary care office or retail clinic costs WellPoint less than $100 (Nussbaum, 2011).
Innovation in Medical Management and Value-Based Insurance Design (VBID)
WellPoint has used innovative programs to improve health and lower long-term costs through medical management and VBID. For instance, WellPoint created a diabetes management program for a state employer in the Northeast. Among other components, the insurance product waives all co-pays for diabetes medications, steers patients to higher quality hospitals and physicians, removes deductibles for preventive care (before the ACA mandated this), and provides free telephonic diabetes education and support. While the program increased short-term costs to all parties due to increased physician visits and higher prescription drug use, Dr. Nussbaum said that his expectation is that the program is likely to demonstrate long-term savings from higher medication compliance and improved control of blood pressure and glucose, which have been shown to reduce complications of diabetes. If WellPoint had only looked at immediate affordability and prescription costs, the plan (and the employer) would have missed the opportunity to improve outcomes and reduce long-term costs.
Pharmaceutical benefit design provides another example of VBID. WellPoint’s two-step design process first considers quality and outcomes and then considers cost. A clinical review committee categorizes pharmaceuticals based on research, the U.S. Food and Drug Administration (FDA) and pharmaceutical company information, and external physician input before a value assessment committee determines tier and formulary position. A WellPoint product called GenericPremium exemplifies how formulary decisions can ensure availability of all drug classes while encouraging affordability. This insurance product includes all generic drugs as well as “one or two” of the most popular branded drugs per class. Dr. Nussbaum stated that this formulary can be delivered at a 20 percent cost reduction over a more open formulary.
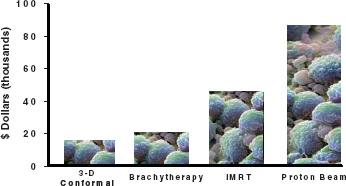
FIGURE 10-2 Prostate cancer treatments vary in cost but not necessarily in outcomes. SOURCE: Nussbaum, 2011.
According to Dr. Nussbaum, evidence is an important component of VBID. For some indications (e.g., back pain, premature elective caesarean-section), evidence is more readily available than others. For prostate cancer, evidence does not clearly specify whether radiation therapy, medical therapy, surgery, or doing nothing is the best treatment. Furthermore, within radiation, treatments include proton beam therapy and intensity modulated radiation therapy among others. Despite the fact that proton beam therapy is nearly double the price of other therapies (see Figure 10-2), Dr. Nussbaum indicated that WellPoint would “encourage” its use if the therapy was known to be “a safer, more effective, treatment with better outcomes.” In the absence of this information, however, there is little justification for the expenditure except in individualized patient circumstances.
When clinical evidence is available, as it is for the treatment and management of back pain, WellPoint uses the evidence to develop payment models that encourage evidence-based care. A study of 172,000 Anthem members revealed that care for back pain was not dependent on clinical need, but rather, on the preferences of the initial treating physician (Nussbaum, 2011). To encourage evidence-based care, WellPoint instituted bundled payments and physician education. Similarly, WellPoint’s data revealed that 6 percent of neonatal intensive care unit (NICU) admissions (each of which cost over $60,000) were related to premature elective caesarean sections (Nussbaum, 2011). In an effort to prevent potentially harmful elective care, WellPoint supports patient safety initiatives that reduce premature caesarian sections. Despite American College of Obstetricians and Gynecologists (ACOG) recommendation to the contrary, approximately 36 percent of elective repeat caesarean sections are performed at less than 39 weeks of gestation (Tita et al., 2009). A future consideration for EHB could involve not covering elective caesarean sections that run counter to ACOG recommendations (ACOG, 2010).
Designing Benefits for Diverse Populations
Dr. Nussbaum explained the importance of retaining flexibility in benefits by describing the differing outcomes of African American and Caucasian women with respect to breast cancer. A study of women with breast cancer conducted by WellPoint and the American Cancer Society (ACS) revealed that insured African American women were 50 percent more likely than insured Caucasian women to have been diagnosed after Stage 2 (Short et al., 2010). These results indicate that for African American women, health insurance coverage and access were not sufficient to guarantee early diagnosis and use of specific hormonal therapies. WellPoint is deploying strategies to provide innovative education to racial and ethnic minority groups regarding the importance of breast cancer screening.
WellPoint Framework
Dr. Nussbaum concluded with WellPoint’s framework for designing EHB. The pillars of this framework— delineation of health benefits, affordability, value, and flexibility for coverage of better care models as they emerge—are supported by a foundation of “proven clinical knowledge and outcomes.” Flexibility, for example, allowed WellPoint to offer a product, described as “slimmed down,” after the plan determined that individuals and certain employers wanted an affordable plan or they were not going to be able to afford insurance coverage. This basic plan offered only generic pharmaceuticals, preventive care, and basic hospital and physician coverage, but kept people in the insurance market.
PRESENTATION BY DR. SHARON LEVINE, THE PERMANENTE MEDICAL GROUP
Dr. Levine opened by describing some of the differences between Kaiser Permanente (KP), an integrated delivery system, and WellPoint, a health insurer: the structure of KP creates what she described as a “mutually exclusive relationship” between the delivery system—physicians, ancillary providers, and hospitals—and the health plan. She then outlined values the committee might consider in making recommendations about a process for designing benefits. First, establishing a floor for benefits across all qualifying health insurance products will promote competition based on value, quality, and cost rather than on risk selection. Second, the elimination of annual and lifetime benefit caps will protect the most vulnerable individuals. Third, the elimination of co-pays and cost sharing for preventive services will facilitate access to high-value services for prevention and early detection of diseases. And fourth, there is a need to be conscious of the “insurance effect,” which has the potential to increase utilization and costs.
Mandates to provide first-dollar coverage for goods or services previously paid for by consumers or available over-the-counter (and thus not covered by an insurance benefit) eliminate any economic barrier to accessing the service or acquiring the products; therefore, first-dollar coverage is beneficial in terms of facilitating access (e.g., to desirable preventive services), but can induce price-insensitive demand. Mandates to cover, for example, “all drugs in a class,” eliminate the need for manufacturers to compete for inclusion in a formulary or preferred drug list based on price, and provide the manufacturer significant and relatively unrestrained pricing power because patients, physicians, and hospitals do not feel the cost impact directly.
Dr. Levine elaborated on challenges KP faces with regards to the third issue: co-pays for preventive services. The ACA does not specify whether surveillance is a diagnostic benefit or a preventive benefit. Surveillance occurs once risk factors are associated with a patient (as a result of a procedure or a family history). The frequency of surveillance and the technologies chosen for screening vary and have cost implications if co-pays and co-insurance are waived, and have utilization impacts if they are not waived and not treated as preventive services. Dr. Levine suggested that the U.S. Department of Health and Human Services (HHS) consider whether and under what circumstances surveillance should be treated as a diagnostic benefit or a preventive benefit so that all carriers and health insurance products adopt the same approach. For example, fecal occult blood testing for colorectal cancer screening is covered as a preventive service. A positive test requires a follow-up colonoscopy, which could be treated as an extension of the preventive screening, with no cost sharing, or as a diagnostic service in follow up to a positive preventive screening. The former approach will eliminate economic barriers to completing the investigation, and also eliminate any incentive to replace fecal occult blood testing for preventive screening with colonoscopies in all cases. The latter approach could eliminate interest among physicians and patients to continue to use fecal occult blood testing, but proceed directly to colonoscopy to avoid the potential for patient exposure to cost sharing in the face of a clinical finding. Similarly, a benign polyp found and removed on routine colonoscopy dictates a different interval for repeat testing—a risk factor for colorectal cancer is identified, but no disease is found. Is the repeat colonoscopy at an earlier interval still a screening procedure or, as enhanced surveillance, is it now a diagnostic procedure? A standardized approach across carriers and products will be important for consumers.
Decision Framework for Covered Benefits
Dr. Levine proceeded to describe the decision framework KP uses to determine its covered benefits, update benefits, and change cost sharing. The framework is principally used when adding benefits; benefits are rarely removed though services may no longer be provided or prescribed because of changes in science and evidence. KP considers:
- Whether the benefit is a health care service. If so, would it improve or maintain health or prevent disease or deterioration and would it be provided by a licensed health care provider.
- The strength of the evidence for a health benefit.
- The impact on cost of the benefit package. Dr. Levine stated, however, that KP does not make coverage decisions based on cost.
- The social insurance question: is it is reasonable to ask others in the risk pool to subsidize the cost of providing the benefit?
Committee member Dr. Galvin asked for additional details about who makes these coverage decisions. Dr. Levine responded that “the decision makers in KP are both clinicians and folks from the health plan insurance business side.” For example, when clinicians, dermatologists, and rheumatologists wanted to increase access to UV therapy for psoriasis, they met with the health plan’s contracts and benefits committee and the decision was jointly made to increase access by eliminating co-pays for this therapy. These decisions are internal to the organization, balancing what the providers recommend with what patients desire and what the plan determines is beneficial.
Given the potential risk of adverse selection, KP also considers what else is available in the market when making benefit decisions. For example, she said that KP “fought long and hard to eliminate the option of insurers selling products in the individual market in California that did not have maternity coverage.” But if competitors offer products without maternity coverage (a less expensive product), KP loses the ability to enroll those individuals looking for the less expensive health insurance product and disproportionately enrolls those intending to, or more likely to use the maternity coverage. Dr. Nussbaum supported Dr. Levine’s position, describing a situation in which WellPoint was the only plan offering bariatric surgery in several markets. Due to adverse selection, offering this benefit was “not sustainable.”
Medical Necessity Decisions
KP uses what Dr. Levine described as “a common definition” of medical necessity (for further discussion of medical necessity, see Chapter 5):
medically appropriate and indicated and required to prevent, diagnose a condition or clinical symptom in accord with generally accepted professional standards of practice and consistent with standards of care in the community.
KP’s physicians are “involved on a daily basis in determinations of medical necessity” for otherwise contractually covered benefits and in their practice. In response to a question from committee member Dr. Santa, she clarified the intersection of coverage and medical necessity: “medical necessity determination is only for otherwise covered services,” and physicians do not “make, prescribe, perform, or offer services not covered” under the benefits package. Her response prompted Dr. Santa to ask about the impact of physicians making these medical necessity decisions. The KP system, Dr. Levine said, creates “an environment of examined practice and constant peer interaction. So it is not individual physicians somewhere just determining that this is right or wrong. There is a fair amount of collective engagement in that process.”
That said, if a physician determines that a treatment is not medically necessary and the patient disagrees, an appeals process is in place. Dr. Levine described this process as follows: the patient can appeal, and if the plan upholds the physician’s determination at the medical center level and regional level, then the member’s appeal goes to the regulator (in California, the regulator is the Department of Managed Health Care). If the regulator determines the appeal is related to a coverage issue, the regulator makes the decision. If the regulator determines the appeal is a medical necessity issue, the determination goes to independent external medical review (Chapter 12 further describes California’s review processes for managed care).
Variation in Benefits and Benefit Design
Dr. Levine then described the degree of variation in benefits among KP plans and the reasons for this variation. She stated that plans vary in cost sharing, deductibles, co-insurance, and out-of-pocket maximums, with deductibles ranging from $250 to $5,000 for individuals and from $500 to $10,000 for a family insurance product (Levine, 2011). Beyond these benefit design elements, some variation results from contractual exclusions and different state mandates in the actual benefits covered.
Contractual Exclusions
Dr. Levine said that unlike the self-insured market, KP’s fully insured model provides limited ability to use criteria-based coverage (e.g., when a plan only covers spine surgery after meeting certain criteria) and “limited ability to customize exclusions.” Services are either covered under the contract or excluded. Approximately 25 percent of KP’s purchasers accept KP’s exclusions “as-is,” while 50 percent expand the list of exclusions, and 25 percent opt to either “buy up or limit the number of exclusions.” Exclusions tend to fall into one of three categories: they are not a health care service, they do not pass the “social insurance test,” or they are excluded based on the site of care.
Non-health care services, for KP, include teaching Braille or American Sign Language, exercycles, gym memberships, and personal trainers, among others. While exercise options could be beneficial to health, Dr. Levine said KP does not view them as a part of health care delivery nor does it consider certain types of educational interventions—for example, interventions that improve functioning and productivity in persons who are blind or hearing impaired—part of health care delivery. Some educational services such as diabetes or asthma self-management training are covered services, as they are directly related to medical care and clinical outcomes.
Social insurance test exclusions are more subjective. This test asks whether “it is fair to ask others in the risk pool to subsidize or provide coverage for something” even if this service must be provided by a physician. Cosmetic services, reversal of voluntary sterilization, growth hormone as a performance enhancer in adults, and assisted reproductive technologies often fail the “social insurance test” as they are not “generally accepted” for coverage given “today’s societal norms.” In general, these are considered life-enhancing rather than medically required to maintain or improve patient health. Some of these procedures, though, must be covered in particular states because of state mandate requirements. Massachusetts, for example, requires coverage of unlimited cycles of assisted reproductive technology.
When committee member Ms. Ginsburg later asked for additional details on the “social insurance test,” Dr. Levine cited KP’s approved exclusion of the drug Pleconaril for treatment of common cold symptoms as an example of a social insurance test exclusion. Pleconaril, an antiviral agent effective against picornaviruses, was expected to cost $75 for approximately 48 hours of symptom mitigation for viral upper respiratory illness. KP feared the “insurance effect” would raise costs and induce utilization disproportionate to the clinical benefit derived, given that most colds resolve within the 48-hour timeframe without such intervention. Ultimately, the regulator approved exclusion of the drug from coverage based on the social insurance argument.1
Site of care exclusions usually relate to coverage by a non-plan provider that was not authorized as a referral, an emergency, or an out-of-area urgent need.
State Mandates
Dr. Levine concluded her presentation by pointing out that there is wide variation in state mandates; she characterized these as mandates “to cover,” mandates “to offer,” and mandates “to provide.” Mandates to cover as part of the benefit package relate to the service itself, the frequency, or the site of service. Many states, for example, mandate coverage of annual PSA (prostate-specific antigen) testing, and California mandates coverage of diabetes equipment and supplies, reconstructive surgery, and cancer clinical trials, among others. Maryland, in what Dr. Levine called a “singular” requirement, mandates coverage of a wig for patients who have lost hair as a
____________________
1 At the time of this presentation, the drug had not received FDA approval for the treatment of upper respiratory infections, and the exclusion for drugs intended to lessen the symptoms of viral upper respiratory infections remains.
result of cancer chemotherapy. Many states have mandates “to offer” services such as orthotics and prosthetics. A mandate to offer requires the carrier to offer to sell, outside the basic benefit package, a supplemental benefit at an actuarially sound price. Mandates to provide are rarer and often occur within the context of care delivery. An example would be a mandate to provide a specific service (e.g., interpreter services) or a product when a diagnosis is made (e.g., a specific set of information produced by a federal or state agency relating to the condition).
For a closed network, integrated delivery system like KP, any willing provider and any willing pharmacy mandates are particularly problematic. KP’s ability to deliver coordinated care across the continuum, with close linkages among primary care, specialty care, ambulatory, and inpatient settings depends on having a dedicated delivery system in which all parties have the same incentives, access to the same information and the same information platforms, and operate in a linked and coordinated fashion based upon a consistent set of values relating to quality, safety, evidence, and resource stewardship. Dr. Levine said that this becomes very difficult to sustain in an “any willing provider” environment.
State mandates are a concern for plans, Dr. Levine said, not only because of state-by-state variation, but also because they tend to be static. The dynamic nature of science and technology means that guidelines evolve and practices change; once state mandates appear, however, they are rarely repealed. Georgia, for example, still mandates that plans offer coverage for autologous bone marrow transplant (ABMT) for breast cancer (NAIC, 2009), despite the fact that this treatment was found to be less effective than conventional therapies and harmful to patients (Stadtmauer et al., 2000). Given the evolving nature of science and technology, Dr. Levine suggested that the committee consider “how granular to get” in mandating specific EHB benefits: “the more granular, the more often they’ll need to be revisited.” She suggested, for example, that cancer screening mandates be broad because if specific technologies for cancer screening are mandated, many of these will be “obsolete long before anyone thinks to look at the regulation.”
Committee member Mr. Koller asked both Drs. Levine and Nussbaum how they would propose addressing this state-by-state variation in mandates if they were developing the EHB design. For mandates in which there is “absolute proof that something is beneficial,” Dr. Nussbaum recommends a “national coverage model.” Conversely, he said, for areas in which benefits are unproven or rapidly evolving, flexibility at the state and federal coverage levels may be necessary. Dr. Levine suggested that criteria regarding the level of evidence needed to mandate a benefit would be beneficial, as would “resistance to granularity.” Broader mandates, such as mandated coverage for cancer screening, would be more beneficial than mandated coverage for PSA testing for prostate cancer.
ACOG (American Congress of Obstetricians and Gynecologists). 2010. ACOG practice bulletin no. 115: Vaginal birth after previous cesarean delivery. Obstetrics and Gynecology 116(2 Pt 1):450-463.
Levine, S. 2011. PowerPoint Presentation to the IOM Committee on the Determination of Essential Health Benefits by Sharon Levine, Associate Executive Medical Director, the Permanente Medical Group, Costa Mesa, CA, March 2.
Mangione-Smith, R., A. H. DeCristofaro, C. M. Setodji, J. Keesey, D. J. Klein, J. L. Adams, M. A. Schuster, and E. A. McGlynn. 2007. The quality of ambulatory care delivered to children in the United States. New England Journal of Medicine 357(15):1515-1523.
NAIC (National Association of Insurance Commissioners). 2009. NAIC’s compendium of state laws on insurance topics: Mandated benefits—other. Kansas City, MO: National Association of Insurance Commissioners.
Nussbaum, S. 2011. Health insurance plan variance in coverage (inclusions, exclusions, networks) and benefit design for quality improvement. PowerPoint Presentation to the IOM Committee on the Determination of Essential Health Benefits by Sam Nussbaum, Executive Vice-President, Clinical Policy and Chief Medical Officer, WellPoint, Inc., Costa Mesa, CA, March 2.
Short, L. J., M. D. Fisher, P. M. Wahl, M. B. Kelly, G. D. Lawless, S. White, N. A. Rodriguez, V. J. Willey, and O. W. Brawley. 2010. Disparities in medical care among commercially insured patients with newly diagnosed breast cancer. Cancer 116(1):193-202.
Stadtmauer, E. A., A. O’Neill, L. J. Goldstein, P. A. Crilley, K. F. Mangan, J. N. Ingle, I. Brodsky, S. Martino, H. M. Lazarus, J. K. Erban, C. Sickles, S. M. Luger, T. R. Klumpp, M. R. Litzow, D. L. Topolsky, J. H. Glick, and Philadelphia Bone Marrow Transplant Group. 2000. Conventional-dose chemotherapy compared with high-dose chemotherapy plus autologous hematopoietic stem-cell transplantation for metastatic breast cancer. New England Journal of Medicine 342(15):1069-1076.
Tita, A. T., M. B. Landon, C. Y. Spong, Y. Lai, K. J. Leveno, M. W. Varner, A. H. Moawad, S. N. Caritis, P. J. Meis, R. J. Wapner, Y. Sorokin, M. Miodovnik, M. Carpenter, A. M. Peaceman, M. J. O’Sullivan, B. M. Sibai, O. Langer, J. M. Thorp, S. M. Ramin, and B. M. Mercer. 2009. Timing of elective repeat cesarean delivery at term and neonatal outcomes. New England Journal of Medicine 360(2):111-120.








