This chapter discusses the fuel production and use associated with striving to meet the overall study goals of a 50 percent reduction in petroleum use by 2030 and an 80 percent reduction in petroleum use and in greenhouse gas (GHG) emissions from the light-duty vehicle (LDV) fleet by 2050 compared to the corresponding values in 2005. It addresses the primary sources of energy for making alternative fuels, the costs of alternative fuels, and the investment needs and the net GHG emissions of the fuels delivered to the LDV fleet over time. Alternative fuels are transportation fuels that are not derived from petroleum, and they include ethanol, electricity (used in plug-in electric vehicles [PEVs] such as plug-in hybrid electric vehicles [PHEVs] or battery electric vehicles [BEVs]), hydrogen, compressed or liquid natural gas, and gasoline and diesel derived from coal, natural gas, or biomass. Petroleum-based fuels are liquid fuels derived from crude oil or unconventional oils.
The chapter opens with a summary discussion of the study goals, fuel pathways, trends in the fuels market, fuel costs, investment costs, and GHG emissions for an LDV in 2030 using each fuel, and it includes a summary table for each of the last three categories, as well as some cross-cutting findings. More detailed discussions of each fuel follow the summary discussion, with a section devoted to each fuel. Also discussed are carbon capture and storage, and resource needs and limitations.
3.1.1 The Scope of Change Required
The study goals are aggressive and require significant improvements to the vehicle and the fuel system to meet the desired goals. The number of LDVs and the vehicle miles traveled (VMT) are expected to nearly double from 2005 to 2050, adding challenges to meeting the goals.1 To reach the goals with twice as many LDVs on the road in 2050 means that each LDV would consume on average only 10 percent of the petroleum consumed compared to 2005 and emit only 10 percent of the net GHG emissions. Gasoline and diesel made from petroleum would be nearly eliminated from the fuel mix to reach the petroleum reduction goal. The 80 percent net GHG emissions reduction goals can be met by various combinations of lower fuel consumption rate (inverse of fuel economy) and lower fuel net GHG emission (Table 3.1). The higher the reductions in LDV fuel consumption rate, the lower the reductions in fuel net GHG emissions would need to be to reach the GHG reduction goal. As discussed in Chapter 2, LDV fleet economy improvements of 3 to 5 times may be technically feasible by 2050, meaning that the average net GHG emissions of the fuel used in the entire LDV
TABLE 3.1 LDV Fuel Economy Improvement and Fuel GHG Impact Combinations Needed to Reach an 80 Percent Reduction in Net GHG Emissions Compared to 2005 Assuming a Doubling in Vehicle Miles Traveled (VMT)
| LDV Fuel Economy Increase versus 2005 | LDV Fuel Consumption Rate Relative to 2005 (percent)a | Requisite Reduction in Net Fuel System GHG Impact versus 2005 (percent)b |
| 2× | 50 | 80 |
| 3× | 33 | 70 |
| 4× | 25 | 60 |
| 5× | 20 | 50 |
| 6× | 17 | 40 |
aThe vehicle fuel consumption rate (e.g., gal/100 mi) corresponding to a given increase in fuel economy (e.g., miles per gallon) relative to the base year level. For example, a quadrupling (4×) of fuel economy simply means that the fuel consumption rate is 25 percent of the base level.
bThe net reduction of system-wide GHG emissions from fuel supply sectors needed to meet an LDV sector-wide 80 percent GHG reduction goal for a given fuel economy gain when assuming a fixed doubling of VMT, that is, without accounting for induced effects such as VMT rebound due to higher fuel economy.
_______________________
1The EIA Annual Energy Outlook 2011 (EIA, 2011a) is the basis for these projections.
fleet would have to be reduced by 50 to 70 percent per gallon of gasoline equivalent (gge) by that time.
Finding: Meeting the study goals requires a massive restructuring of the fuel mix used for transportation. Petroleum-based fuels must be largely eliminated from the fuel mix. Other alternative fuels must be introduced such that the average GHG emissions from a gallon equivalent of fuel are only about 40 percent of today’s level.
Many different alternative fuel pathways have been proposed, and this study selected seven different fuel pathways to analyze: conventional petroleum-based gasoline, biofuels (including ethanol and “drop-in”2 biofuels), electricity, hydrogen, compressed natural gas (CNG), gas to liquids (GTL), and coal to liquids (CTL). These were selected because of their potential to reduce petroleum use, to be produced in large quantities from domestic resources, and to be technically and commercially ready for deployment within the study period. Most fuels selected have lower net GHG emissions than petroleum-based fuels. Other alternative-fuel pathways were discussed but not included for detailed analysis because they did not meet the first three criteria. For example, methanol is discussed in Appendix G.8 but was not included for detailed analysis because of environmental and health concerns that inhibit fuel distribution and retail companies from broadly offering methanol as a fuel.
The fuel costs, net GHG emissions, investment needs, and resource requirements were analyzed on a consistent basis for the different fuels to facilitate comparisons among fuels. Future technology and cost improvements for the selected fuels are considered and compared on a consistent basis, even though the extent of improvement for different fuels is likely to vary.
3.1.3 Developing Trends in the Fuels Market
Several developments in the energy markets over the past few years will have large impacts on long-term LDV fuel-use patterns. First, the fuel economy of the LDV fleet will increase rapidly over the next decade because of higher Corporate Average Fuel Economy (CAFE) standards effective through 2016 and proposed through 2025. The CAFE standards increase requirements from 23.5 mpg in 2010 to 34.1 mpg in 2016 to 49.7 mpg in 2025. Alternative fuels and new LDV technologies would compete with future gasoline or diesel LDVs that use much less petroleum and have lower net GHG emissions. From a consumer viewpoint, the decreasing volume of gasoline needed to travel a mile reduces the economic motivation to switch from gasoline to an alternative fuel.
Second, biofuel production is expected to increase as a result of the Renewable Fuel Standard 2 (RFS2) passed as part of the 2007 Energy Independence and Security Act (EISA). This legislation mandated the consumption of 35 billion gallons of ethanol-equivalent3 biofuel and 1 billion gallons of biodiesel (about 24.3 billion gge/yr based on energy content) by 2022. The detailed requirements of RFS2 are discussed in Appendix G.1. Based on the 2010 gasoline use of 136 billion gge/yr (8.88 million bbl/d), this mandate increases biofuel use from 9.9 percent (0.87 million bbl/d) to 18 percent (1.59 million bbl/d) of the gasoline mix by volume (EIA, 2011b). Although the mandated volume for cellulosic biofuel is not expected to be met by 2022, any additional biofuel volume in the conventional gasoline mix reduces the need for gasoline from petroleum and the volume of other alternative fuels needed to reach the study goals. See Section 3.2, “Biofuels,” in this chapter for a detailed discussion.
Third, the volume of economic natural gas from shale deposits within the United States has been increasing rapidly. In its June 18, 2009, report the Potential Gas Committee upgraded by 39 percent the estimated U.S. potential natural gas reserves (defined as being potentially economically extractable by the use of available technology at current economic conditions) compared with its previous biannual estimate (Potential Gas Committee, 2009). Based on the new estimates, the probable natural gas reserves would provide about 86 years of consumption if the consumption rate stays at the current level. In 2011, the Potential Gas Committee increased its estimates such that 90 years of probable reserves exist based on 2010 consumption. Many previous studies on alternative fuels did not include natural gas as a possible source for LDV fuel because of limited domestic supply, and the likely price increase in electricity and residential heating costs associated with high natural gas use in the transportation market. With increasing domestic production, natural gas now is a viable option for providing transportation fuels through multiple pathways including electricity, hydrogen, GTL, and CNG. See Section 3.5, “Natural Gas,” in this chapter and Appendix G.7 for a detailed discussion.
3.1.4 Study Methods Used in the Analysis
This study considers conventional and alternative fuels for the 2010-2050 period, and this committee undertook a number of tasks to generate possible fuel scenarios and data for use in the modeling efforts described in Chapter 5. The primary sources for the data are different for each fuel and are explained in the sections that provide details on each fuel below in this chapter. The committee made efforts to standardize input data and definitions between the primary
_______________________
2Drop-in fuel refers to nonpetroleum fuel that is compatible with existing infrastructure for petroleum-based fuels and with LDV ICEs.
3A gallon of ethanol has about 77,000 Btu, compared with 116,000 Btu in 1 gallon of gasoline equivalent.
information sources. The tasks the committee performed include:
- Assessed the current state of the technology readiness for each fuel using information gathered from presentations made to this committee and published literature.
- Estimated future improvements to these technologies that could be broadly deployed in the study period.4
- Estimated the range of costs based on future technology for each fuel delivered to the LDV at a fueling station in a similar way for each fuel. The reference price basis in the Energy Information Administration’s (EIA’s) Annual Energy Outlook 2011 (EIA, 2011a) is used for all primary fuel prices. Investment costs are expressed in 2009 dollars.
- Estimated the initial investment costs needed to build the infrastructure for each fuel pathway.5
- Estimated the net GHG emissions per gallon of gasoline-equivalent for each fuel based on the methods selected for producing the fuel. An upstream GHG component, a conversion component, and a combustion component were included in the estimate of net GHG emissions.
3.1.5 Costs of Alternative Fuels
The costs of alternative fuels through 2035 are estimated based on the energy raw material prices in the reference case of the Annual Energy Outlook 2011 (AEO; EIA, 2011a), and the basis and assumptions for the estimates are explained in the individual fuel sections. Fuel prices beyond 2035 were estimated by the committee. Table 3.2 summarizes the expected alternative fuel costs for 2030 on a $/gge or $/kWh basis for some of the fuel pathways and shows the consumer’s annual fuel costs for a new vehicle of that type based on 2030 estimated vehicle mileage.
While the values in Table 3.2 are useful guideposts for this analysis, there are a few factors to keep in mind. First, the fuel costs shown in Table 3.2 are untaxed—current or future taxes are not included and could alter the actual annual cost that consumers pay. Second, the per-gallon of gasoline-equivalent fuel cost estimates in 2030 are a snapshot in time and will likely change as technology develops and world energy prices change. Third, the untaxed fuel-purchase costs to consumers each year appear similar for most fuels except for CNG and the BEV, which are significantly lower than others. Given the small separation for the other options in 2030, untaxed fuel costs are not expected to be a significant driving force for consumers to switch from gasoline to alternate vehicle technologies in this timeframe. Untaxed fuel cost differences of only several hundred dollars per year will not cover the additional vehicle costs described in Chapter 2.6
TABLE 3.2 2030 Annual Fuel Cost per LDV, Untaxed Unless Noted
| Fuel | Fuel Cost ($/gge or kWh) | Annual Consumer Use (gge or kWh) | Annual Consumer Fuel Cost ($/yr) | ||||||
| Gasoline (taxed) | 3.64/gge | 325 gge% | 1,183 | ||||||
| Biofuel (drop in) | 3.39/gge | 325 gge% | 1,102 | ||||||
| Gasoline (untaxed) | 3.16/gge | 325 gge% | 1,027 | ||||||
| PHEV10a | 3.16/gge | 260 gge% | 913 | ||||||
| 0.141/kWh | 650 kWh | ||||||||
| CTL with CCS | 2.75/gge | 325 gge% | 894 | ||||||
| GTL | 2.75/gge | 325 gge% | 894 | ||||||
| PHEV40b | 3.16/gge | 130 gge% | 752 | ||||||
| 0.175/kWh | 1,950 kWh | ||||||||
| Hydrogen—CCS case | 4.10/gge | 165 gge% | 676 | ||||||
| Natural gas—CNG | 1.80/gge | 325 gge% | 585 | ||||||
| BEV | 0.143/kWh | 3,250 kWh | 465 | ||||||
NOTE: All fuel costs are based on the 2011 AEO (EIA, 2011a) for 2030. The assumed fuel economies are representative of on-road LDV averages for 2030 described in the scenarios in Chapter 5. The following assumptions were made: 13,000 mi/yr traveled and 40 mpgge for liquid and CNG vehicles, 80 mpgge for hydrogen and 4.0 mi/kWh for electric vehicles. PHEV10 gets 20 percent of miles on electric, PHEV40 gets 60 percent. All costs are untaxed unless noted. Electricity cost includes the retail price plus amortization of the cost of a home charger.
aPHEV10 is a plug-in hybrid vehicle designed to travel about 10 miles primarily on battery power only before switching to charge-sustaining operation.
bPHEV 40 is a plug-in hybrid vehicle designed to travel about 40 miles primarily on battery power only before switching to charge-sustaining operation.
Finding: As the LDV fleet fuel economy improves over time, the annual fuel cost for an LDV owner decreases. With high fleet fuel economy, the differences in annual fuel cost between alternative fuels and petroleum-based gasoline decreases and the annual costs become similar to one another. Therefore, over time fuel-cost savings will become less important in driving the switch from petroleum-based fuels to other fuels.
3.1.6 Investment Costs for Alternative Fuels
The investment costs to build the fuel infrastructure are sizable for all of the alternative fuel and vehicle pathways. In fact, these costs remain among the most important barriers
_______________________
4Some future technologies that might be developed during the study period are not included for detailed analysis because future efficiencies and costs are not well understood. Examples of this include photoelectrochemical hydrogen production and biofuels from algae.
5Investment costs are explained in Appendix G.2, “Infrastructure Initial Investment Cost.”
6As pointed out in Chapters 4 and 5, consumers tend to value about 3 years worth of fuel savings when making decisions on initial vehicle purchases. Using the numbers in Table 3.2, 3 years of untaxed hydrogen saves only $1,501 compared with taxed gasoline during 2030. The cost saved is not enough to cover the higher cost of a fuel cell electric vehicle (FCEV).
to rapid and widespread adoption of alternatives. Table 3.3 shows the investment costs on a $/gge per day basis and on a $/LDV basis. This calculation includes only the investment in building a new form of infrastructure needed to make and deliver the fuel to the customer. It does not include investment to expand an already large and functioning infrastructure associated with producing more of the basic resource. For instance, for hydrogen made from natural gas, the investment cost includes the cost of converting natural gas to hydrogen, pipelines to deliver the hydrogen, and the full cost of a hydrogen station, but it does not include investments to produce natural gas or deliver it to a plant. A complete list of which costs are included or excluded is shown in Appendix G.2 “Infrastructure Initial Investment Cost.” Details for these investment costs are found in the individual fuel sections below in this chapter.
The investment cost for a new petroleum refinery is included in Table 3.3 for perspective. However, with increasing fuel economy for the LDV fleet, no new refinery capacity will be needed during the study period. So in effect the initial investment cost for gasoline is near zero. The alternative-fuel-producing industry, in 2030, must make a $1,000 to $3,000 investment for each new alternative-fuel LDV, whereas almost none is needed for new petroleum gasoline LDVs. This cost differential is a major barrier to large-scale deployment of alternative fuels.
The scale, pace, and modularity of the infrastructure investments vary for the different vehicles and fuels. These differences are noted in the right-most column of Table 3.3. Two basic categories are used to describe the infrastructure requirements: centralized and distributed. Centralized infrastructure investments are those that are borne by a select number of decision makers. For example, the infrastructure for CTL, GTL, or gasoline requires large-scale plants (which cost billions of dollars each) that individual companies would pay for. Biofuels require large-scale investments for biorefineries. Hydrogen requires hydrogen production plants plus smaller-scale distributed investments by retailers to install new storage tanks and fuel pumps. The investment costs for BEVs and PHEVs in Table 3.3 include only the costs for home, workplace, and public chargers. The centralized infrastructure for CNG has already been built, and so the incremental CNG infrastructure costs include home fueling systems (paid for by car owners), or new filling stations (paid for by retailers). Thus, the infrastructure requirements vary from a few very large, multibillion-dollar investments (e.g., for biorefineries) made by a few decision makers in industry, to millions of small multithousand-dollar investments made by millions of decision makers such as consumers, ratepayers, and retailers.
Finding: The investment cost for a new fuel infrastructure using electricity, biofuels, or hydrogen is in the range of $2,000 to $3,000 per LDV. This is a significant barrier to large-scale deployment when compared with an infrastructure cost for using petroleum of only about $530 per LDV.
3.1.7 GHG Emissions from the Production and Use of Alternative Fuels
Operational and infrastructure costs (as noted in Tables 3.2 and 3.3) are critical factors to consider for deployment. However, the net GHG emissions for the different vehicle and fuel options need to be examined to determine how the goal of 80 percent GHG reduction could be met. The estimates of annual GHG emissions in 2030 for different vehicle and fuel options are shown in Table 3.4.
Each vehicle and fuel option has a range of net annual GHG emissions because GHG emissions depend
TABLE 3.3 2030 Fuel Infrastructure Initial Investment Costs per Vehicle
| Alternative Fuel | 2030 Investment Cost | LDV Fuel Use per Day | Infrastructure Investment Cost ($/vehicle) | Cost Burden | |||||
| Electricity BEV | $330/kWh per day | 8.9 kWh | 2,930 | Distributed (car owners, ratepayers) | |||||
| Electricity (PHEV40) | $530/kWh per day | 5.4 kWh | 2,880 | Distributed (car owners, ratepayers) | |||||
| Biofuel (thermochemical) | $3,100/gge per day | 0.89 gge | 2,760 | Centralized (industry) | |||||
| CTL (with CCS) | $2,500/gge per day | 0.89 gge | 2,220 | Centralized (industry) | |||||
| Hydrogen (with CCS) | $3,890/gge per day | 0.45 gge | 1,750 | Centralized (industry) and distributed (retailers) | |||||
| GTL | $1,900/gge per day | 0.89 gge | 1,690 | Centralized (industry) | |||||
| Natural gas—CNG | $910/gge per day | 0.89 gge | 810 | Distributed (retailers and car owners) | |||||
| Electricity (PHEV10) | $370/kWh per day | 1.75 kWh | 650 | Distributed (car owners, ratepayers) | |||||
| Gasoline (new refinery—if needed) | $595/gge per day | 0.89 gge | 530 | Centralized (industry) | |||||
NOTE: Basis: 13,000 mi/yr and 40 mpgge for liquid and natural gas vehicles, 80 mpgge for hydrogen, and 4.0 mi/kWh for electric vehicles. PHEV10 gets 20 percent of miles on electric; PHEV40 gets 60 percent. Investment costs are explained in the individual fuel sections.
TABLE 3.4 Estimates of 2030 Annual Net GHG Emissions per Light-Duty Vehicle Used in the Modeling in Later Chapters
| Fuel | Net GHG Emissions (kg CO2e) | Annual Use | Annual GHGs Emissions per LDV (kg CO2e) | ||||||
| CTL with CCS | 12.29/gge | 325 gge | 4,000 | ||||||
| GTL | 11.47/gge | 325 gge | 3,730 | ||||||
| Gasoline | 11.17/gge | 325 gge | 3,630 | ||||||
| PHEV10 | 0.590/kWh | 650 kWh | 380 3,290 | ||||||
| 11.17/gge | 260 gge | 2,910 | |||||||
| Natural gas | 9.20/gge | 325 gge | 2,990 | ||||||
| PHEV40 | 0.590/kWh | 1,950 kWh | 1,146 2,600 | ||||||
| 11.1/gge | 130 gge | 1,454 | |||||||
| Hydrogen—low cost | 12.2/gge | 165 gge | 2,010 | ||||||
| BEV—reference grid | 0.590/kWh | 3,250 kWh | 1,920 | ||||||
| Biofuel—with ILUCa | 5.0/gge | 325 gge | 1,620 | ||||||
| BEV—low-GHG grid | 0.317/kWh | 3,250 kWh | 1,030 | ||||||
| Biofuel—without ILUC | 3.2/gge | 325 gge | 1,040 | ||||||
| Hydrogen—with CCS | 5.1/gge | 165 gge | 840 | ||||||
| Hydrogen—low-GHG case | 2.6/gge | 165 gge | 430 | ||||||
| Biofuel—with ILUC,CCS | –9.0/gge | 325 gge | –2925 | ||||||
aIndirect land-use changes (ILUC) can have large impacts on net GHG emissions but can vary considerably.
Basis: 13,000 mi/yr and 40 mpgge for liquid and NGVs, 80 mpgge for hydrogen and 4.0 miles/kWh for electric vehicles. PHEV10 gets 20 percent of miles on electric; PHEV40 gets 60 percent. GHG estimates are explained in the individual fuel sections.
on how the fuels are produced. The range of net GHG emissions for biofuels is large because the net GHG emissions depend on many factors, including the type of feedstock used,7 the management practices used to grow biomass (e.g., overuse of nitrogen fertilizer could increase N2O flux), any land-use changes associated with feedstock production,8 and the use of carbon capture and storage (CCS) with biofuel production. The range of differences for a BEV is determined by the average GHG emissions of the grid and over time may be quite different than shown in Table 3.4. Hydrogen has a large range of possible GHGs determined by the several different choices of production method.
The net GHG emissions from the three typical alternative fuels—biofuels, hydrogen, and electricity—can be either high or low depending on technology choices, carbon costs, regulations, and other factors. Choices driven by technology, economics, and policy determine the GHG emissions for future alternative fuels.
Finding: The GHG emissions from producing biofuels, electricity, and hydrogen can vary depending on the basic resource type and conversion methods used. Making these fuels with methods involving very low GHG emissions increases the technical and cost hurdles, especially during the introductory period. Actions to encourage the use of these more challenging methods should be timed to coincide with large-scale deployment and not be a burden during the introductory period for the fuel. Needed policy actions for each fuel pathway are listed in Appendix G.3.
Biofuel is a generic term that refers to any liquid fuel produced from a biomass source. A number of different biofuel products (e.g., biobutanol and drop-in biofuels9) derived from different feedstocks (e.g., lignocellulosic10 biomass and algae) have been proposed, but only corn-grain ethanol and biodiesel were produced in commercially relevant quantities in the United States as of the drafting of this report. Ethanol and biodiesel have been of interest because they can be easily synthesized using well-known processes from commercially available agricultural products (such as corn and soybeans in the United States, sugar cane in Brazil, and other oil seeds elsewhere). However, neither ethanol nor biodiesel is fully fungible with the current infrastructure and LDV fleet designed for petroleum-based fuels.
Ethanol and biodiesel are usually shipped separately and blended into the fuel at the final distribution point. Ethanol can be blended into gasoline in various proportions but has only about two-thirds of the volumetric energy content of petroleum-based gasoline. As of 2011, ethanol supplied almost 10 percent by volume of the U.S. gasoline demand (Figure 3.1). Biodiesel, produced via the transesterification of various vegetable oils or animal fats, supplied less than 1 percent of U.S. transportation fuel demand in 2011 (see Figure 3.1). U.S. biodiesel production capacity was about 2.7 billion gal/yr in 2010 (NBB, 2010), but actual production is significantly lower. Biomass can also be used to synthesize drop-in fuels, that is, synthetic hydrocarbons that would be fully fungible with existing infrastructure and vehicles.
The EISA included an amendment to the Renewable Fuel Standard in the Energy Policy Act (EPAct) of 2005. RFS2 mandated an increase of over 200 percent in the use of biofuels between 2009 and 2022. (See Box 1.1 in Chapter 1.) Biofuels, including corn-grain ethanol and biodiesel, currently require government subsidies or mandates to compete economically with petroleum-based fuels. Increases in ethanol consumption can also be limited by the “blend wall”
_______________________
7Corn-grain ethanol is likely to have different net GHG emissions than cellulosic biofuel.
8Uncertainties in GHG emissions from land-use changes are a key contributor to the wide range of estimates for net GHG emissions from biofuels. Some biofuel feedstock such as corn stover would not contribute much to GHG emissions from land-use changes.
9Biofuels that are compatible with existing infrastructure and internal combustion engine vehicles (ICEVs) for petroleum-based fuels.
10Plant biomass composed primarily of cellulose, hemicellulose, and lignin.
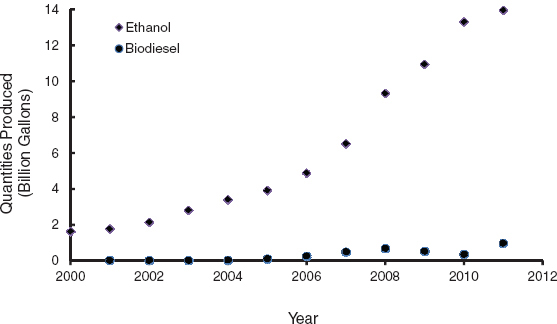
FIGURE 3.1 Amount of fuel ethanol produced in the United States.
SOURCE: Data from EIA (2012b,c).
(NRC, 2011). In 2010, the U.S. Environmental Protection Agency (EPA) approved the use of E15 in internal combustion engine vehicles (ICEVs) of model year 2001 or newer in response to a waiver request by Growth Energy and 54 ethanol manufacturers. Although EPA approved the use of E15 in 2010, its sale just began in July 2012 (Wald, 2012). In April 2012, EPA approved 20 companies for the manufacture of E15 (EPA, 2012a).11 Without an approved method for eliminating misfueling of older cars,12 increased ethanol use is likely to be constrained in the near term. In addition, auto manufacturers do not recommend using E15 in any vehicles that were initially designed to use E10 because of concerns that E15 might damage older engines (McAllister, 2012).
Flex-fuel vehicles (FFVs) can use higher concentrations of ethanol (up to 85 percent), and many auto manufacturers produce flex-fuel vehicles because of the CAFE credit13 they receive (DOE-EERE, 2012c). However, the number of E85 fueling stations is limited (about 2,500 stations across the United States) and varies by state (DOE-EERE, 2012a). The price of E85 has always been higher than petroleum-based gasoline on an equivalent energy content basis.
Although the use of corn-grain ethanol can reduce petroleum imports, its effects on GHG emissions are ambiguous. Life-cycle assessments by various authors have estimated a 0 to 20 percent reduction in GHG emissions from corn-grain ethanol, relative to gasoline (Farrell et al., 2006; Hill et al., 2006; Hertel et al., 2010; Mullins et al., 2010).
The EISA requires the use of additional advanced and cellulosic biofuels that will reduce petroleum imports, lower CO2e emissions, and be produced predominantly from lignocellulosic biomass. (See Appendix G.1 for definitions of biofuels in EISA.) To qualify as an advanced biofuel, a biofuel would have to reduce life-cycle GHG emissions by at least 50 percent compared with petroleum-based fuels.14 To qualify as a cellulosic biofuel, a biofuel would have to be produced from cellulose, hemicellulose, or lignin and reduce life-cycle GHG emissions by at least 60 percent compared with petroleum-based fuels. Although RFS2 specified lifecycle GHG reduction thresholds for each type of fuel and EPA makes regulatory determinations accordingly, the actual life-cycle GHG emissions of biofuels could span a wide range (NRC, 2011). Biofuels facilities that began construction after 2007 would have to be individually certified for both biomass source and production pathway to qualify for renewable identification numbers (RINs).15
The U.S. government and private investors have invested billions of dollars to develop cellulosic biofuels (see Tables
_______________________
11When the U.S. Environmental Protection Agency (EPA) approves a new fuel or fuel component, EPA only evaluates the fuel’s impact on the emission control system and its ability to meet the evaporative and tailpipe emission standards. EPA does not evaluate the impact of the new fuel on any other aspect of vehicle performance, including degradation of vehicle components and performance that are not associated with the emission control system.
12The Renewable Fuels Association submitted a Model E15 Misfueling Mitigation Plan to EPA for review and approval on March 2012. The plan includes fuel labeling to inform customers, a product transfer documentation requirement, and outreach to public and stakeholders. However, those measures will not eliminate the possibility of accidental misfueling.
13CAFE credits were used to incentivize vehicle manufacturers to sell large numbers of vehicles that run on natural gas or alcohol fuels. See Chapter 6 for details.
14In its Renewable Fuel Standard Program (RFS2) Regulatory Impact Analysis (EPA, 2010b), EPA determined the life-cycle GHG emissions to be 19,200 g CO2e/million Btu for petroleum-based gasoline and 17,998 g CO2e/million Btu for petroleum-based diesel.
15The Renewable Identification Number (RIN) system was created by EPA to facilitate tracking of compliance with RFS. A RIN is a 38-character numeric code that corresponds to a volume of renewable fuel produced in or imported into the United States.
2.3 and 2.4 in NRC, 2011); however, no commercially viable processes are operational as of the drafting of this report. Initial research focused on cellulosic ethanol; however, the difficulties associated with integrating ethanol into the existing fuel distribution system and the inability to increase ethanol yields to the desired levels have resulted in a shift in research emphasis away from the biochemical conversion processes to the thermochemical or hybrid conversion processes. Conversion processes of lignocellulosic biomass to fuels are discussed below in this chapter.
The production potential of cellulosic biofuels is determined by the ability to grow and harvest biomass and the conversion efficiency of the processes for converting the biomass into a liquid fuel. Many studies have been published, and they show that the currently demonstrated conversion potential is about 46-64 gge/ton of dry biomass feedstock (as summarized in NRC, 2011). This represents an energy-conversion efficiency to liquid fuel of 25 to 50 percent based on the ratio of the lower heating value of the fuel product to that of the biomass feedstock. Much of the balance of the biomass-energy content is used to produce electricity and to power the conversion processes.
Multiple potential sources of lignocellulosic biomass can be used to produce biofuels. They include crop residues such as corn stover and wheat straw, fast-growing perennial grasses such as switchgrass and Miscanthus, whole trees and wood waste, municipal solid waste, and algae. Each potential source has a production limit. The consumptive water use and other environmental effects of producing biomass for fuels are discussed in detail in Renewable Fuel Standard: Potential Economic and Environmental Effects of U.S. Biofuel Policy (NRC, 2011).
Several studies have been published on the estimated amount of biomass that can be sustainably produced in the United States (NAS-NAE-NRC, 2009b; DOE, 2011; NRC, 2011, and references cited therein). All of the studies focused on meeting particular production goals and none of them projected biomass availability beyond 2030; they are discussed in Appendix G.4. The studies had different target production dates ranging from 2020 to 2030. The most recent study (DOE, 2011) projected that 767 million tons of additional biomass (above that currently consumed) could be available in 2030 at a farm gate price of less than $60/ton. This estimate was based on an annual yield growth of 1 percent and would require a shift of 22 million acres of cropland (or 5 percent of 2011 cropland) and 41 million acres of pastureland (or 7 percent of 2011 pastureland) into energy crop production. That amount was assumed to be available in 2050 in this report.
Finding: Sufficient biomass could be produced in 2050, when converted with current biofuel technology and consumed in vehicles with improved efficiencies consistent with those developed by the committee in Chapter 2 (about a factor-of-four reduction in fuel consumption per mile by 2050), that the goal of an 80 percent reduction in annual petroleum use could be met.16
Several technologies can be used to process biomass into liquid transportation fuels for the existing LDV fleet. Converting corn starch to ethanol and converting vegetable and animal fats to biodiesel or renewable (green) diesel are well-established commercial technologies. As of 2012, the collective capacity of corn-grain ethanol and biodiesel refineries in the United States is sufficient to essentially meet the 2022 RFS2 consumption mandates for conventional biofuels and biomass-based diesel.
There are a number of potential processes for converting cellulosic biomass into liquid transportation fuels. Demonstration facilities have been built for some of the various technologies. Much of the focus on cellulosic biofuel has switched away from ethanol to producing a biofuel that is a drop-in fuel.
Three main pathways are being developed to produce cellulosic biofuels: biochemical, thermochemical, and a hybrid of thermochemical and biochemical pathways. The pathways are discussed in detail in the report Liquid Transportation Fuels from Coal and Biomass: Technological Status, Costs, and Environmental Impacts (NAS-NAE-NRC, 2009b). Briefly, biochemical processes use biological agents at relatively low temperatures and pressures to convert the cellulosic material to biofuels—primarily ethanol and higher alcohols.
Thermochemical conversion uses heat, pressure, and chemicals to break the chemical bonds of the biomass and transform the biomass into many different products. Three main pathways are being considered for thermochemical conversion: gasification followed by Fischer-Tropsch (FT) catalytic processing to make naphtha and diesel, gasification followed by conversion of the syngas into methanol and subsequent conversion into gasoline via the methanol-to-gasoline (MTG) process, and pyrolysis (either high-temperature or lower-temperature hydropyrolysis) followed by hydroprocessing of the pyrolysis oil to produce gasoline and diesel. Other thermochemical pathways are also under development. Thermochemical and biochemical processes can be combined—for example, gasification of the biomass followed by fermentation of the syngas to produce ethanol or other alcohols.
_______________________
16See Chapter 5 modeling results for further detail.
The economics of biofuel production have been discussed in a number of studies. Both NAS-NAE-NRC (2009b) and NRC (2011) compared recent information to develop comparative economics. The report Renewable Fuel Standard: Potential Economic and Environmental Effects of U.S. Biofuel Policy (NRC, 2011) and the references cited therein form the bases for the discussion of economics in this chapter.
Conversion of cellulosic biomass to drop-in biofuels is a relatively new and evolving suite of technologies. Predicting the future developments that can lower the cost of biofuel production is difficult. The cost of production is primarily a function of the cost of biomass, the yield of biofuels, and the capital investment required to build the biofuel conversion facility. Current conversion efficiencies are 46-64 gge/ton of dry biomass (which gives an average value of 55 gge per dry ton with a range of ±9 gge per dry ton).
Current capital costs to build a cellulosic biorefinery vary between 10 and 15 $/gge per year for all of the technologies discussed above. Thus, a biorefinery that would produce 36 million gge/yr consumes about 2,000 dry tons of biomass per day. The biorefinery would cost between $360 million and $540 million to build. An average capital cost would be 12.5 ± 2.5 $/gge per year. Because biorefining is a developing and evolving technology, it is reasonable to assume that yields will increase and that the capital costs will decrease as the technology matures. Yields will increase because of improvements in the catalysts used and in the process configurations. The capital costs are expected to decline primarily because of economies of scale and improvements in the process configurations. Biorefineries that are bigger and more efficient than the first-mover facilities will be built as engineering and construction techniques are refined over time. The analysis is this chapter assumes that yields will increase from a baseline of 55 gge per dry ton in 2012 at a rate of 0.5 percent per year to a yield of 64 gge per dry ton by 2028. The capital costs are assumed to decrease by 1 percent per year through 2050 for an overall reduction in capital cost of 31 percent compared to the present cost. The capital costs given in this report are for fully engineered facilities for a relatively new technology. Others (Wright et al., 2010) have estimated a 60 percent decrease in capital costs as the technology evolves. Figure 3.2 shows the current and future costs to produce cellulosic biofuels based on these assumptions and the assumption that bioenergy feedstock is $75 or $133 per dry ton. Current estimates are for a biomass cost of $75 per ton, but a sensitivity to a higher cost is also included (see Figure 3.2).
Table 3.5 is a summary of projections of cellulosic biofuels that could be available, in addition to the 2012 ethanol and biodiesel production of 14 to 15 billion gal/yr, using different investment rates for new plant capacity. This committee estimated that about 45 billion gge of biofuel would be required to meet the target of 80 percent reduction in petroleum use for the LDV fleet in 2050 and would require about 703 million dry tons per year of biomass feedstock. A uniform annual construction rate of about $10 billion per year can easily produce the projected biofuel needs in 2050. The fuel availabilities are based on the projections discussed
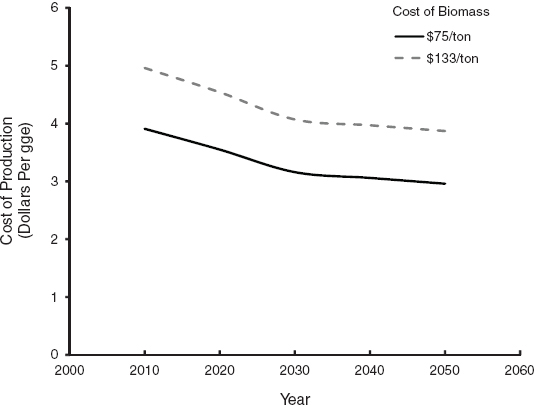
FIGURE 3.2 Sensitivity of biofuel cost to biomass cost.
TABLE 3.5 Estimates of Future Biofuel Availability
| Annual Plant Investment Rate (billion dollars per year) | |||||||||
| 1 | 4 | 7.2 | 10.4 | ||||||
| Biofuel production (billion gge per year) by | |||||||||
| 2022 | 0.9 | 3.7 | 6.7 | 9.7 | |||||
| 2030 | 1.8 | 7.4 | 13.3 | 19.2 | |||||
| 2050 | 4.3 | 17.3 | 31.2 | 45.0 | |||||
| Biomass required in 2050 (million dry tons per year) | 68 | 270 | 488 | 703 | |||||
| Estimated land-use change (million acres) | 5.5 | 22.2 | 40.1 | 57.8 | |||||
| Total investment to 2050 (billion dollars) | 38 | 152 | 275 | 396 | |||||
| Average number of biorefineries built per year | 2.7 | 10.8 | 19.5 | 28.2 | |||||
above. Land requirements are scaled from the U.S. Billion-Ton Update previously discussed (DOE, 2011).
Worldwide expenditures on exploration and production of petroleum are high (Milhench and Kurahone, 2011). For example, ExxonMobil alone invested over $32 billion globally in capital and exploration projects in 2010. The November 7, 2011, issue of the Oil and Gas Journal (2011) reported that the National Oil Companies of the Middle East and North Africa planned to invest a total of $140 billion in oil and natural gas projects in 2012, with even more investments to follow in coming years.
If the biofuels industry grows as projected, many U.S. petroleum refineries will close or be converted to biorefineries. Conversion of a petroleum refinery to a biorefinery will be significantly less costly and labor-intensive than the construction of a “grass-roots” biorefinery.
In all future years, the amount of biofuels that can be produced will most likely be limited not by biomass availability, but rather by the availability of capital to build the biorefineries. However, a potential investor will not start construction without secure contracts for biomass supply and a guaranteed market for the product.17
A large number of biomass conversion facilities would have to be built along with specialized harvesting equipment and a truck fleet to transport the biomass from the fields to the conversion facilities. Economic studies have shown that the conversion facilities need to be near where the crops are grown. Therefore, additional product pipelines would be needed to transport the biofuels from the conversion facilities to the existing petroleum product distribution system. Although drop-in biofuels can use the existing petroleum-product distribution system, feeder lines will most likely be required between the biorefineries and the major petroleum pipelines. However, adding feeder lines will require a relatively small incremental investment.
3.2.7 Regional or Local Effects
Biomass can be grown only in certain parts of the country, and so the conversion facilities will also be located nearby. If drop-in fuels are produced, then the fuels can be shipped via the existing system of petroleum-product pipelines. This system efficiently transports large volumes of petroleum products. Initially, the biofuel refineries will be sited near the locations where the lowest-cost biomass is grown or harvested. Many of these locations are in the Southeast and Midwest United States. The major petroleum pipelines between the Gulf Coast and the Northeast and North Central United States bisect these regions. Tie-ins to these pipeline systems would be relatively short.
The chemical properties of drop-in cellulosic biofuels will be similar to those of existing, petroleum-based LDV fuels, with no additional fuel-related safety hazards. Truck traffic in rural areas is expected to increase, which could increase traffic accidents in these areas.
The primary barrier to displacing petroleum with biofuels is economic. At present, biofuels are more expensive to produce than petroleum-based fuels. The corn-grain ethanol industry had many years of government subsidies and is currently supported by the RFS2 consumption mandate. Subsidies or mandates are projected to be required to support cellulosic biofuel unless the price of oil is close to $190/bbl or conversion costs decline as projected.
As discussed above and in detail in other reports (NAS-NAE-NRC, 2009b; NRC, 2011), ethanol involves definite infrastructure issues. Pure ethanol cannot be used in conventional ICEs because of cold-start problems. It has to be blended with petroleum-based gasoline. The highest content allowed in the United States is 85 percent ethanol by volume (E85). Although E85 could contain up to 85 percent ethanol, its ethanol content typically averages only 75 percent or even less in the winter.
As of 2012, the fuel industry was close to reaching the maximum amount of ethanol that can be consumed by blending into E10. Total U.S. gasoline consumption in 2010 was just over 138 billion gallons. Blending all of this as E10 would consume only 13.8 billion gallons of ethanol, which is
_______________________
17Factors that can affect actual supply of biomass for fuels are discussed in the report Renewable Fuel Standard: Potential Economic and Environmental Effects of U.S. Biofuel Policy (NRC, 2011).
less than the 15 billion gallons of conventional ethanol mandated by RFS2. Fewer than 0.1 billion gallons of E85 were sold in 2009. As the fuel economy of vehicles improves and gasoline sales decline, even less gasoline will be available to be blended with the volume of ethanol mandated. Drop-in biofuels do not have this limitation.
3.2.10 GHG Reduction Potential
There is ongoing debate regarding the GHG emissions from the production of biofuels, including the time profile of the emissions. The uncertainties and variability associated with the GHG reduction potential of biofuels are discussed in detail in NRC (2011). The values for GHG emissions used in this study were a modified version of those developed by EPA for the RFS2 final regulations. The difference was the treatment of emissions attributable to indirect land-use change (ILUC). The EPA analysis distributes the GHG emissions from ILUC over a 30-year period. For the analysis in this report, all emissions contributed by ILUC were attributed to the first year’s operation of the biofuel conversion facility rather than spread over 30 years. This alternate ILUC treatment and its impact on annual biofuel GHG emissions are discussed in detail in Appendix G.5. These predicted GHG emissions do not include the use of CCS in the production facility to reduce overall well-to-wheels GHG emissions. Applying CCS to a biofuel production facility can potentially provide slightly negative well-to-wheels GHG emissions (NAS-NAE-NRC, 2009a).
3.3 ELECTRICITY AS A FUEL FOR LIGHT-DUTY VEHICLES
In the United States, electricity is widely available, plentiful, and relatively inexpensive. It already is used as fuel for some LDVs available on the general market, including PHEVs (e.g., the Chevrolet Volt) and BEVs (e.g., the Nissan Leaf). Further, electric-power vehicles are in wide use in commercial applications such as in warehouses and factories.
Table 3.6 shows the 2010 capability of the U.S. electricity system (EIA, 2011a). The capacity factor measures the ability of a power source to produce power and reflects both availability to produce power and whether or not the plant is dispatched. Capacity factor is estimated as the annual electricity production for each source divided by the power production it would have achieved when operating at its net summer capacity 24 hours per day for the entire year. Power dispatch is affected by the price of the source relative to other competing sources because lower-priced sources are dispatched preferentially.
TABLE 3.6 Capability of the U.S. Electricity System in 2010
| Source | Net Summer Capacity (GW) | Electricity Production (thousand GWh) | Capacity Factor | ||||||
| Coal | 318.1 | 1,879.9 | 0.67 | ||||||
| Oil and natural gas steam | 113.5 | 123.9 | 0.13 | ||||||
| Natural gas combined cycle | 198.2 | 733.8 | 0.42 | ||||||
| Diesel/conventional combustion turbine | 138.6 | 51.0. | 0.11 | ||||||
| Nuclear | 101.1 | 802.9 | 0.90 | ||||||
| Pumped storage | 21.8. | –0.2 | –0.001 | ||||||
| Renewables | 123.0 | 371.6 | 0.35 | ||||||
| Total | 1,014.4 | 3,962.8 | 0.45 | ||||||
The average U.S. retail price for electricity is about $0.10/kWh with substantial variation across the country because of the time of use, local generation mix, and various incentives or taxes. In general, electricity produced by hydro power costs the least, followed closely by coal, nuclear, and natural gas. Electricity generation from natural gas is expanding rapidly for the following reasons:
- The cost of natural gas generation strongly depends on the cost of fuel. Currently the cost of natural gas is low ($2.5 to $3.5/million Btu) and could remain low for a decade or more.
- CO2 emissions per unit of power generated by natural gas are about half of the CO2 emissions per unit of power generated by coal.
- Emissions of sulfur oxides (SOx), nitrogen oxides (NOx) and other toxic air pollutants from natural gas are much lower than the emissions from coal.
Gas turbines are well suited to provide backup power for intermittent renewable energy generation sources, such as wind and solar, because they can be ramped up relatively quickly. Because of this characteristic, the share of electricity generation from natural gas tends to increase as renewable energy increases. The generation of electricity produces GHG emissions, mainly CO2. In 2010, total GHG emissions from electric power as reported in the AEO 2011 were 2.3 billion metric tons CO2e (EIA, 2011a). There are additional emissions further upstream in the process, for example, in mining coal, producing natural gas, transporting fuels to the power plant, and building solar panels, wind turbines, and power plants. These upstream emissions can be added to the combustion emissions to estimate the total life-cycle emission of any process, including electricity generation. Life-cycle emissions are considered in this report’s analyses of GHG emissions.
The capability (and demand) for electricity generation in the United States is expected to grow slowly from the present to 2050. For the purposes of this study, two cases in the
AEO 2011 (EIA, 2011a) were examined: the 2011 reference case and the GHG price case (hereafter referred to as the low-GHG case). The low-GHG case is based on a steadily escalating carbon tax beginning at $25/metric ton of CO2e in 2013 and escalating at 5 percent per year, reaching $152/metric ton in 2050. The National Energy Modeling System (NEMS) is used by EIA to produce the AEO projections up to 2035. Therefore, the reference and low-GHG cases had to be extrapolated to 2050. For the low-GHG case, the total GHG emissions, power output, and cost data were extrapolated to 2050 using the years 2031 to 2035 to better capture the accelerating effects of the carbon tax increase in shifting the mix of generation sources. For the reference case, data from the period 2020 to 2035 were used because the mix of generation sources does not change much.
The low-GHG case shows that the annual GHG emissions in 2050 are reduced from the reference-case emissions by more than the desired 80 percent; however, this result does not account for the life-cycle emission effects in the electricity-generating sector because in the AEO analyses some of the emissions are attributed to other sectors. To compare fuels used in transportation on a consistent basis, the additional upstream generation of GHG emissions for combusted fuels will have to be included to account for the life-cycle emissions for non-combusted fuels, for example, renewables and nuclear.
For coal and natural gas, the upstream emission factors in the Greenhouse Gases, Regulated Emissions, and Energy Use in Transportation Model (GREET model; Argonne National Laboratory) were used to calculate the total lifecycle emissions.
The AEO 2011 estimated GHG emissions from coal combustion to be 0.9552 kg CO2e/kWh.18 For coal, the upstream emissions embedded in the GREET model are 3.74 kg CO2e/GJ. Using a conversion factor of 1.055 GJ per million Btu and assuming a heat rate of 10,000 Btu/kWh for the conversion of coal to electricity, the upstream emissions are 0.04 kg CO2e/kWh. Accounting for transmission line losses of 7 percent, the correction from both upstream and transmission line losses is an additional 0.042 kg CO2e/kWh, making the total emissions for coal-fired electricity 1.0 kg CO2e/kWh.
The existing value for natural gas combustion emissions in the AEO model is 0.433 kg CO2e/kWh.19 The upstream GHG emissions for natural gas in the GREET model are 13.4 kg CO2e/GJ. The heat rate used in AEO 2011 for converting natural gas to electricity is 8,160 Btu/kWh. Using this as a conversion factor, the upstream emissions of natural gas are 0.115 kg CO2e/kWh. Correcting for transmission line losses of 7 percent makes the total correction 0.123 kg CO2e/kWh, and the total GHG emissions for natural gas are 0.556 kg CO2e/kWh.
There are no GHG emissions assumed in the AEO cases for nuclear and renewable electricity. The life-cycle emissions for nuclear and renewable energy sources were assumed to be 0.02 kg CO2e/kWh, based on the values used in the NRC report America’s Energy Future. Technology and Transformation (NAS-NAE-NRC, 2009a). Table 3.7 summarizes the results for GHG emissions from fuels.
In addition to extending beyond the AEO’s 2035 projections, the current study had to verify that the low-GHG case still gives the desired result of about an 80 percent reduction in GHG emissions by 2050 after all emissions in the life cycle are accounted for. The fraction of electricity generated by each fuel was estimated by extrapolating the 2035 AEO results to 2050. Because the changes in the fuel mix were accelerating in the latter period of the EIA case, 2031-2035, the rate in that period was used as a reasonable basis from which to extrapolate. The result is shown in Table 3.8, which indicates that the GHG emissions are still reduced by more than 80 percent in 2050.
TABLE 3.7 2010 Electricity-Generation GHG Emissions by Source
| Source | Combustion Emissions (kg CO2e/kWh) | Upstream Emissions (kg CO2e/kWh) | Life-Cycle Emissions (kg CO2e/kWh) | ||||||
| Coal | 0.9552 | 0.042 | 1.0 | ||||||
| Natural gas | 0.433 | 0.123 | 0.556 | ||||||
| Nuclear | 0 | 0.02 | 0.02 | ||||||
| Hydro | 0 | ||||||||
| Renewables | 0 | 0.02 | 0.02 | ||||||
SOURCE: EIA (2011a).
TABLE 3.8 Key Parameters of the AEO Base Case and Low-GHG Case
| Parameter | 2010 | 2020 | 2035 | 2050 | |||||
| AEO base-case cost ($/kWh) | 9.6 | 8.8 | 9.2 | 9.4 | |||||
| AEO low-GHG case cost ($/kWh) | 9.6 | 11.2 | 12.7 | 14.8 | |||||
| Carbon tax ($/metric ton CO2e) | 0 | 35 | 73 | 152 | |||||
| AEO base-case output (billions kWh) | 3,963 | 4,158 | 4,633 | 5,140 | |||||
| AEO low-GHG case output (billions kWh) | 3,963 | 3,823 | 3,976 | 4,190 | |||||
| AEO base-case GHG emissions (kg CO2e/kWh) | 0.586 | 0.535 | 0.545 | 0.541 | |||||
| AEO low-case GHG emissions (kg CO2e/kWh) | 0.586 | 0.412 | 0.256 | 0.111 | |||||
_______________________
3.3.3 Grid Impact of Plug-in Electric Vehicles
Neither of the AEO grid models account for the additional load if a large number of electric-powered vehicles are added. To assess the importance of this effect, the energy demand in 2020, 2035, and 2050 was estimated (Table 3.9).
The electricity generation projection in the low-GHG case is the comparison standard because the grid capacity is lower than that in the reference case. The result of this comparison shows that the additional load from PEVs in 2020 and 2035 is a small fraction of the projected total electricity usage and probably well within the uncertainty in the projections. Between 2035 and 2050, the power demand for PEVs is assumed to rise quickly. By 2050, it is assumed to reach 7 percent of the projected power usage and has a growth rate of about 0.5 percent per year. This load increase is well within the historic growth of the grid, which has been as high as 7 percent per year in the mid-1980s, and even the growth rate of 1 to 2 percent per year that has been true over the past 10 years in the United States. However, the low-GHG case projects load growth of less than 0.1 percent a year in the absence of BEV demand. Further, adding plants to the grid is a time-consuming process, and construction of a new plant can take a few years to a decade or more. Therefore, if the low-GHG case is an accurate projection of electricity usage, additional capacity has to be planned, permitted, funded, and constructed at a more rapid pace than projected for the next 20 years as large numbers of PEVs come into service (Table 3.10). If these additional plants cannot be brought online quickly enough, then the growth of PEV use may be restrained or the low GHG emissions may not be achieved as older plants with higher emissions may be required to be kept in service. New plant demand can be reduced to the degree that load shifting to off peak can be used. The amount of this reduction is not well defined.
There are also temporal and local effects on power demand from PEV charging. If owners charge their PEVs during times that the grid is highly used (e.g., during peak load periods), there could be problems with supplying enough electricity. For instance, if most PEVs are returned to their home base late in the afternoon with depleted batteries and are plugged in to charge, this load will be superimposed on the grid at a time when the daily load is already highest. This is especially true in the summer and winter seasons because of air conditioning and heating demands. It also may be desirable to move the load off peak to reduce GHG emissions because when peak loads are high, the oldest and likely dirtiest sources of power will be forced into service. They would not be used when power demands are well below the peak. Based on the estimates above, the peak loading issue until 2035 is unlikely to be a problem overall. But as the LDV charging load on the grid grows, the peak loading becomes of greater concern. However, studies have shown that practical, effective means are available to move the load to alternate charging times (e.g., late at night when other loads are low). One method that utilities are considering using to change consumer behavior is time-of-use (TOU) pricing, which would charge consumers lower rates during off-peak hours (generally between 11 p.m. and 5 a.m.). However, studies show that more comprehensive, integrated, and intrusive load management approaches based on the wide use of smart grid technology can be even more effective than incentives such as TOU pricing in reducing the peak load.
TABLE 3.9 Electric Vehicle Energy Demand Compared to Low-GHG Case
| 2030 | 2035 | 2050 | |||||||
| AEO low-GHG output (billion kWh) | 3,823 | 3,976 | 4,190 | ||||||
| Electric vehicle energy demand (billion kWh) | 3.4 | 72 | 286 | ||||||
| Electric vehicle energy demand (percent of output) | 0.1 | 1.8 | 6.8 | ||||||
NOTE: The demand for electric vehicles was estimated assuming 13,000 miles as the base. The number of miles driven for each vehicle was taken from Elgowainy et al. (2009). The assumed number and mix of vehicles used to estimate the charging load are shown in Table 3.10. The number of vehicles, number of miles, and fraction of the fleet are not predictions by the committee, but were selected to be conservative (high) to illustrate the impact of the charging demand on the grid. For all vehicles the energy consumption is 0.286 kWh/mi.
TABLE 3.10 Assumed Number of Electric Vehicles in Fleet
| 2020 | 2035 | 2050 | |||||||
| Total electric vehicles | 2 million | 30 million | 100 million | ||||||
| Fraction PHEV10 | 0.4 | 0.1 | 0 | ||||||
| Fraction PHEV40 | 0.4 | 0.5 | 0.3 | ||||||
| Fraction BEVs | 0.2 | 0.4 | 0.7 | ||||||
NOTE: BEV, battery electric vehicle; PHEV, plug-in hybrid electric vehicle.
The present power grid has an estimated capability to handle a large fraction of the nationwide LDV fleet simply by taking advantage of the excess capacity in off-peak hours at night (PNNL, 2007). However, that estimate represents a nationwide average, and excess capacity varies throughout the country. For example, while Texas could provide energy for 73 percent of its LDV fleet, the California and Nevada area only could recharge 15 percent of its local fleet with off-peak power. This rate could be problematic given the large number of vehicles present in this region. With larger penetration of PEVs over the coming decade (about 25 percent), it has been suggested that there will be significant strain in regions such as California if the grid does not adapt (Guo et al., 2010).
The local distribution grids of each utility could also be affected by a significant deployment of PEVs (or even by a small number of PEVs if they are concentrated in a small area served by a small number of local transformers). The most likely upgrade required by the addition of PEVs is the replacement of transformers. A study by the Elec-
tric Power Research Institute and the Natural Resources Defense Council (EPRI and NRDC, 2007) and discussions by the committee with Pacific Gas and Electric Company (Takemasa, 2011) and previous discussions with Southern California Edison (Cromie and Graham, 2009) indicate that the local grid effects are manageable and within the utilities’ normal cost of doing business. See Appendix G, Section G.2, for more discussion and an estimate of the investment cost.
There are four potential major sources of investment costs beyond the cost of the electricity itself:
- Charging stations to transfer energy from the electric distribution system to the PEVs;
- Necessary upgrades to the transmission and distribution system uniquely associated with charging PEVs;
- Additional generation capacity needed to provide fuel for large numbers of PEVs; and
- Conversion of the electric power system to realize approximately 80 percent lower annual GHG emissions.
These investment costs are estimated in Appendix G.6. The results are summarized in the following sections.
3.3.4.1 Charging Station Costs
Three types of charging stations are available. Level 1 charging stations use normal 110 V circuits and provide AC power to the vehicle. They are relatively low power and require typical charging times of over 20 hours for a 24 kWh battery. Level 2 charging stations provide AC power via a 240 V circuit (typically used today for electric clothes dryers and electric stoves). Because energy flow goes as the square of the voltage, level 2 charging stations will cut the charging time by a factor of about four. So for today’s batteries, the charging time will decrease to a few hours. Level 3 charging stations convert AC line voltage and provide high-voltage DC to the vehicle. DC stations are not suitable for home use, and DC will likely be provided at charging stations analogous to gas stations. Level 3 charging stations now can charge a typical battery of an electric vehicle to 80 percent of capacity—the recommended maximum level to avoid damage and hence reduction in battery life—in 15 to 30 minutes. Preliminary data available to date suggests there will be very limited use of DC fast chargers and that the price of charging will be significantly higher than charging at home using a level 1 or level 2 charging station.
The bulk of the charging station investment cost will be borne by the electric-vehicle owner. Longer electric-only driving distances require larger batteries and more powerful charging stations, and so the investment cost is a function of the type of electric vehicle. Appendix G.6 estimates these costs per vehicle for a wide range of electric-vehicle types, assumes appropriate charging station mixes for both home and commercial installations, and includes the reference and low-GHG grid cases to 2050. Current costs for charging stations per vehicle range from about $800 for a PHEV10 to about $4,200 for a BEV. By 2050 the investment costs per vehicle will have dropped from about $450 for a PHEV10 to about $1,950 for a BEV. Appendix G.6 also converts these costs to $/gge per day for comparison with other fuels. These costs do not include a cost for a parking space for access to charging. The parking space for access to charging is a significant additional barrier as the EIA Residential Energy Consumption Survey (2009) reported that 52 percent of households cannot park a car within 20 feet of an electrical outlet.
3.3.4.2 Costs of Additions and Changes to the Transmission and Distribution System
The upgrade costs for high-voltage transmission are included in the next two sections. The investment costs for the distribution system are considered to be relatively small and manageable by the local utilities. They likely will be included in the price of the electricity. Therefore, no additional capital costs are included.
3.3.4.3 Cost of Additional Generation Needed for Large Numbers of PEVs
The additional energy demand from 100 million PEVs in 2050 is estimated to be about 286 billion kWh. Meeting that additional demand by new plants will require the addition of the equivalent of about 90 1,000-MWe plants at a cost of about $360 billion for new generating capacity and a total of over $400 billion, including the associated high-voltage transmission system additions.
3.3.4.4 Cost of Conversion of the Power System to 80 Percent Lower Annual GHG Emissions
Beyond the addition of new capacity to provide fuel for PEVs, a large additional investment would be required to reduce the annual GHG emissions from the entire U.S. power system by about 80 percent by 2050. This investment cost is estimated to be about $1 trillion. This cost is required to decarbonize the power sector and is not attributable solely to the LDV sector.
3.3.5 Regional and Local Effects
Regional and local effects for electricity-fueled LDVs influence the method of rolling out the charging infrastructure and changes in distribution system. They also affect the attractiveness of electricity as a fuel because of the
pricing and GHG emissions of the local grid and because of dominant local use of vehicles versus electric-vehicle characteristics.
The rollout of a robust charging infrastructure is coupled to robust sales and use of PEVs, especially BEVs as opposed to PHEVs, because PHEVs can make use of liquid fuel if electricity charging is unavailable. Automobile manufacturers offering BEVs and PHEVs reported to the committee that they have found most sales to date occurring in urban areas with high income levels and a high proportion of people who are more environmentally minded (Diamond, 2010). Thus, the logical basis for expansion of the use of PEVs and the associated charging infrastructure is to proceed in urban areas in which vehicle and charging infrastructure builds rapidly and achieves the needed critical density. As time goes on, these centers are likely to expand and connect along major transportation corridors to provide power to the large number of BEVs needed to substantially reduce petroleum use and GHG emissions. Government support should follow this natural growth pattern and concentrate initial resources in limited areas rather than supporting a broad use of BEVs and expanded charging networks at many locations. Once the process is successful in one “center,” the support there can be phased out and moved to another fertile area (Electrification Coalition, 2009).
Although the U.S. power grid is interconnected, the flow of electricity from all sources to all loads is not perfect. In effect, the country is divided up into a number of regional networks that, while strongly connected internally, have weaker ties to one another. As a result, there are significant regional and even state-to-state differences in pricing and GHG emissions. Electricity as a fuel costs less than gasoline, but customers in areas with higher electricity prices realize smaller fuel-cost savings. Some regional networks with relatively low electricity prices may emit significantly more GHG emissions than others with higher electricity prices (Anair and Mahmassani, 2012). GHG emissions may also be a function of available margin and peak loading on the local grid. Even if the base-load power generation has low GHG emissions, the older and dirtier power sources will be dispatched as the load rises. Thus, the GHG emission characteristics of the local grid might also affect the attractiveness of PEVs to buyers with strong environmental concerns.
The dominant use of the vehicle interacts with the characteristics of the PEVs, and this is likely to vary regionally. BEVs are used primarily as short-commute passenger vehicles and in fleets as vehicles for light hauling, or for relatively short-distance services. Those uses match the BEV’s battery capability and charging time requirements and suggest that BEVs initially, and perhaps permanently, will be concentrated in urban locations. BEVs will not be in wide use in rural areas with longer drives and more widely separated charging locations.
The electrical safety considerations in providing electricity to the vehicle are generally well in hand. For both residential and business charging, the voltages and power levels are well within the state of practice, and safety provisions are well understood and codified. One of the costs associated with charging station installation is that it must meet the requirements of the national and local electrical codes, which means that it will most likely have to be installed by a licensed electrician and inspected and permitted by the appropriate governmental agency. For DC fast chargers used as public chargers, very high power connections between the charger and the vehicle must be made, and additional care is warranted. There are standards in use now for DC charging stations that fall under the formal jurisdiction and requirements of the national, state, and local electrical codes.
There do not appear to be technical barriers in the electrical system upstream of the vehicle. There are, however, several potential financial and societal barriers:
- The investment cost for the charging infrastructure is borne largely by the vehicle owners.
- The capital cost for the full implementation of the needed changes to achieve a low-GHG-emitting electrical power system is large.
- Coordinating the needed investments and infrastructure work will require overcoming the complexity of the power system’s unique ownership, management, and regulatory situation. The electric power system is regulated by a large number of local, state, regional, and federal entities. In most cases, the investors and owners of the transmission and distribution infrastructure are not the same as the investors and owners of the generating sources. Further, in some cases no benefits may accrue to some of those that have to make investments, such as states that have neither the loads nor the generation sources, but must support transmission lines between adjacent states that have loads and sources.
- Permitting and construction of new power system assets are very time consuming. Large power plant projects and large transmission and distribution system projects can take several years to over a decade to complete.
Finding: For electricity as a fuel for LDVs to be effective in reducing net GHG emissions, the entire U.S. electric power system has to shift largely to electricity production from sources that emit low GHG emissions (for example, nuclear, renewables, and natural gas with or without CCS).
3.4.1 The Attraction of Hydrogen
When hydrogen is used as a fuel in fuel cell electric vehicles (FCEVs), the only vehicle emission is water. When hydrogen is used in an internal combustion engine, the emissions are water, some nitrogen oxides, and some trace chemicals mostly as a result of using lubricants. Although CO2 emissions are absent from vehicle emissions when hydrogen is used as an LDV fuel, varying amounts of GHGs are emitted during hydrogen production. The amount depends on the primary fuel source and the technology used for hydrogen production. Most of the hydrogen on Earth is found in either water or hydrocarbons such as coal, oil, natural gas, and biomass. Because of the diverse primary sources for hydrogen, an amount of hydrogen large enough to fuel the entire LDV fleet could be made with only domestic sources. Different process technologies can be used with different primary sources to make a pathway for delivering hydrogen to consumers at different costs and with varying amounts of GHG emissions. The diversity of supply sources and production technologies is an advantage of hydrogen fuel.
For more than 10 years, there have been serious efforts in the United States, Europe, and Japan to develop FCEVs and the needed production and delivery technologies to supply hydrogen. As Chapter 2 indicates, there has been considerable success in developing FCEVs, but some challenges remain. There also has been considerable success in developing production, distribution, and dispensing technologies for making and delivering low-cost hydrogen, but major challenges still exist. The two major challenge areas are the following:
- Making low-cost hydrogen with low GHG emissions. At present, the lowest-cost methods for hydrogen production used by industry are based on fossil fuels and have associated GHG emissions of varying amounts. The low-GHG methods are currently more expensive and need further development to become competitive.
- Building the hydrogen infrastructure will be a large, complex, and expensive undertaking. Hydrogen-fueling stations would have to be available before FCEVs can be sold. Until a large number of FCEVs are in use, the cost of hydrogen as a fuel will be high. Because FCEVs are new and hydrogen as a consumer fuel is new, there are many practical concerns such as safety, codes and standards, permitting, and zoning issues that need to be addressed before growth can flourish.
3.4.3 Current Status of the Market
Hydrogen as an industrial commodity is produced in large quantities in the United States and in many other countries. The amount of hydrogen produced is over 50 million tons per year worldwide (Raman, 2004; IEA, 2007) and over 10 million tons per year in the United States (EIA, 2008b). Most of the hydrogen is used in the chemical processing industry and in refining crude oil, and most of it is produced in large facilities closely associated with the end use. Over 95 percent of U.S. hydrogen is made from natural gas, with other sources including refinery off-gases, coal, and water electrolysis. Several hydrogen pipeline systems (Houston, Los Angeles, and Chicago) exist to move large quantities of gaseous hydrogen between nearby industrial users with over 1,200 miles of hydrogen pipelines. Some established industrial gas companies produce, store, and distribute hydrogen as either a gas or a cryogenic liquid to smaller users by truck. The demand for hydrogen for industrial use has increased consistently for several decades.
Even as the infrastructure for producing, delivering, and using large amounts of hydrogen for this industrial market is well developed, the infrastructure for producing, delivering, and dispensing hydrogen for use as a transportation fuel has yet to be developed. For illustrative purposes, if hydrogen were to be used as a transportation fuel, then the current U.S. production level of 10 million tons per year would be enough to fuel about 45 million cars (at 60 mpgge and 12,000 mi/yr). There is, however, little spare capacity in the existing system for this new market. Therefore, a new hydrogen infrastructure is needed before large numbers of FCEVs are produced. This infrastructure will need to be much different from the existing one because it has to focus on wide distribution of small amounts if distributed through retail outlets, similar to what is done for gasoline today.
Academic, industrial, and government efforts over the past 10 years to define this retail-fuel-oriented infrastructure have mapped out the needed technology improvements, established performance criteria for different parts of the infrastructure, estimated the cost of hydrogen and the infrastructure over time, and suggested possible implementation methods. The NRC report Transitions to Alternative Transportation Technologies—A Focus on Hydrogen (NRC, 2008) contains an analysis of the technical needs, costs, petroleum savings and GHG emission savings possible by moving towards a hydrogen-fuel infrastructure.
3.4.4 Hydrogen Infrastructure Definition
Rather than being built throughout the entire United States before FCEVs are available, a hydrogen infrastructure likely will first be started in a few markets. Then the infrastructure will be built up in conjunction with increasing local FCEV sales. The concentration of demand will result in a decrease in the high initial cost of hydrogen and the infrastructure as
equipment for commercial-scale production is installed and used at commercial rates. This process will then be repeated in additional markets until a critical mass of FCEVs and hydrogen stations is built to a market-sustainable level.
The first hydrogen stations are likely to be supplied by truck delivery from local hydrogen-distribution points. This is a high-cost method that may be largely replaced by hydrogen stations with on-site hydrogen generation capabilities where the hydrogen is made at the retail station rather than supplied from the large plants that now supply the bulk of hydrogen. This approach precludes the need to transport or deliver hydrogen, and the distributed hydrogen generation equipment can be sized for the demand. Several technologies are available for the small hydrogen generators, including natural-gas reforming, water electrolysis, and biofuel reforming.
- Small natural-gas reforming—The process is the same as that used in today’s large natural-gas reforming facilities. However, the reforming apparatus for fuel is small and packaged such that it looks like a large appliance. These reformers have been demonstrated at a number of hydrogen-fueling stations in the United States, Europe, and Japan. CO2 produced in the process is released to the atmosphere because capturing it is difficult.
- Small water electrolysis—Commercial alkaline water electrolysis units are available and have been demonstrated in small hydrogen stations. GHG releases are associated with the source of electricity and can be high or low depending on how the electricity is produced.
- Small biofuel reforming—Ethanol reforming and other biofuel reforming have been demonstrated in laboratories, but research and development (R&D) is still needed to increase hydrogen yields and lower costs to be competitive with small natural-gas reformers and small water-electrolysis methods. GHG releases can be low depending on the source of the biofuel.
As the demand for hydrogen increases in a local market, there will come a point when large centralized facilities similar to today’s will produce hydrogen at lower cost than is possible with small distributed generators. These facilities will also offer the opportunity to make low-GHG hydrogen through the use of other primary fuels and CCS technology. Several primary feedstock and technology choices are possible, including natural-gas reforming, coal gasification, biomass gasification, and large-scale wind or solar electrolysis.
- Natural-gas reforming—This low-cost process is widely used now for generating large amounts of hydrogen. CCS is possible but has not yet been demonstrated with a hydrogen plant.
- Coal gasification—This process has been used commercially for decades, but high CO2 releases require that CCS be available. CCS has not been demonstrated with coal gasification.
- Biomass gasification—This process has been demonstrated in the laboratory, but not yet at large pilot-scale facilities. Further development is needed. If CCS is used, then biomass gasification becomes a CO2 sink with negative releases.
- Large centralized electrolysis with wind or solar power—The process is still being researched to lower costs. This process has low GHG emissions.
Other hydrogen-production methods under research hold long-term promise for making hydrogen at low costs, low GHG emissions, or both, but they are not yet developed enough to understand the availability or the cost implications. Some of these methods include nuclear high-temperature chemical cycles or electrolysis, photoelectrochemical methods, and biological systems.
3.4.5 Hydrogen Dispensing Costs and GHGs
The cost of making, transporting, storing, and dispensing hydrogen at a station has been estimated for all of the primary feedstocks. These estimated costs are highly dependent on many assumptions and can vary considerably depending on future technical advances, feedstock costs, and how quickly the market develops (scale). The estimated costs for some of the different hydrogen pathways based on future technology development are shown in Table 3.11. The estimates are expressed in dollars per gallon of gasoline equivalent ($/gge). A gge of hydrogen contains as much energy (Btu) as a typical gallon of gasoline and is defined as 116,000 Btu/gge in this study. The future price basis and resource requirements used to generate the costs in Table 3.11 are shown in Table 3.12. The hydrogen costs in Table 3.11 are in some cases up to $1.00/gge higher than those determined in prior studies
TABLE 3.11 Hydrogen Costs at the Pump ($/gge), Untaxed
| 2010 | 2020 | 2035 | 2050 | ||||||
| Distributed natural gas reforming | 3.50 | 3.60 | 3.90 | 4.20 | |||||
| Distributed grid electrolysis | 5.80 | 5.40 | 5.50 | 5.69 | |||||
| Coal gasification without CCS | 3.80 | 3.80 | 3.80 | 3.85 | |||||
| Coal gasification with CCS | 4.50 | 4.50 | 4.50 | 4.50 | |||||
| Central natural gas reforming without CCS | 3.30 | 3.40 | 3.70 | 4.10 | |||||
| Central natural gas reforming with CCS | 3.60 | 3.60 | 4.00 | 4.30 | |||||
| Biomass gasification without CCS | 4.10 | 4.10 | 4.10 | 4.10 | |||||
NOTE: Basis: 2008 H2A future cases updated to 2009 dollars using CEPCI and Nelson-Ferrer cost indexes and the AEO 2011 price basis. $2.00/gge included for distribution and station costs for central methods and $1.88/gge included for station costs of distributed methods.
TABLE 3.12 Resource Prices and Requirements Used in Table 3.11
| 2010 | 2020 | 2035 | 2050 | ||||||
| Industrial natural gas, $/million Btu | 4.80 | 5.36 | 7.21 | 9.06 | |||||
| Delivered coal, $/ton | 45.9 | 46.1 | 48.9 | 50.2 | |||||
| Industrial electricity, $/kWh | 0.068 | 0.061 | 0.064 | 0.067 | |||||
| Delivered biomass, $/ton | 75.0 | 75.0 | 75.0 | 75.0 | |||||
| Coal needed, kg/gge H2 | 9.8 | 9.8 | 9.8 | 9.8 | |||||
| Biomass needed, kg/gge H2 | 12.8 | 12.8 | 12.8 | 12.8 | |||||
| Natural gas needed, cubic ft/gge H2 | 170 | 170 | 170 | 170 | |||||
| Electricity needed, kWh/gge H2 | 45 | 45 | 45 | 45 | |||||
NOTE: Basis—AEO2011 (EIA, 2011a) resource prices and 2008 H2A future cases for resource requirements
(NRC, 2008, for example). The increased costs compared to the earlier NRC study result from several factors:
- The costs in the current study are based on the 2008 version of the hydrogen analysis (H2A) production model developed by DOE, whereas the ones in the previous study (NRC, 2008) were from the 2005 version.
- The distribution costs are estimated to be $2/gge, whereas prior ones were $1.00 to $2.00/gge.
- The capital costs are inflated based on actual construction cost inflation to 2009 dollars.
- The costs for biomass and coal are nearly twice what they were in the 2008 study.
The costs in Table 3.11 represent future costs based on using commercial-scale processes and are possible only after about 10 million FCEVs are on the road. Prior to this, the hydrogen cost will be higher because of underutilized or smaller-scale production facilities. Figure 3.3 shows hydrogen costs versus number of FCEVs.
The GHG emissions associated with producing, delivering, and dispensing hydrogen at a station on a life-cycle basis are shown in Table 3.13. This includes an upstream component related to the emissions associated with production and delivery of the base fuel to the hydrogen production plant and, if used, the energy needed to sequester CO2 plus a component for conversion, delivery, and dispensing of GHGs.
3.4.6 Hydrogen Infrastructure Needs and Cost
Building the infrastructure for delivering hydrogen over the vast size of the United States is a significant challenge for the use of hydrogen for transportation. It requires developing some new technologies, establishing codes and standards, overcoming the problem of interdependence of establishing a critical mass of hydrogen-refueling stations and FCEV sales, overcoming the high initial cost of hydrogen, and increasing the use of production methods with low GHG emissions.
The total investment costs used to calculate the hydrogen costs in Table 3.11 for future technologies used at commercial-size plants are shown in Table 3.14. These
FIGURE 3.3 Hydrogen cost versus number of FCEVs.
TABLE 3.13 Total GHG Emissions (kg CO2e per gge of hydrogen)
| H2 Production Method | Upstream CO2e | Plant, Delivery and Dispensing CO2e | Total CO2e | ||||||
| Distributed natural gas reforming | 2.78 | 8.66 | 11.44 | ||||||
| Distributed electrolysis, current grid | 35.44 | 0 | 35.44 | ||||||
| Coal gasification without CCS | 1.13 | 24.67 | 25.81 | ||||||
| Coal gasification with CCS | 2.77 | 2.47 | 5.24 | ||||||
| Central natural gas reforming without CCS | 2.18 | 9.28 | 11.46 | ||||||
| Central natural gas reforming with CCS | 2.71 | 0.93 | 3.64 | ||||||
| Biomass gasification without CCS | –24.37 | 24.57 | 0.20 | ||||||
NOTE: Basis—H2A 2008 future cases modified to use GREET 2011 upstream natural gas figures.
costs are normalized to 2009 dollars per gallon of gasoline equivalent per day of produced hydrogen. The station costs appear to be the largest factor for all but coal technology. The station costs include all costs associated with building grass-roots new stations that include hydrogen storage, compression, and dispensing and are the same for each technology. The actual hydrogen production investment costs are shown separately. Investment costs for CCS are included for the large coal and natural gas facilities.
The NRC report Transitions to Alternative Transportation Technologies—A Focus on Hydrogen (NRC, 2008) outlined one possible hydrogen infrastructure development pathway out to 2050 and estimated the hydrogen cost, GHG emissions, and investment needs over different time periods. The pathway in that report starts with distributed natural gas reforming. As demand increases, coal gasification with CCS and biomass gasification provide the bulk of increased hydrogen production. This is not the only possible pathway to supply the increasing amount of hydrogen, but it relies on some current, low-cost, and mostly commercially developed processes. With future R&D success, other technologies would likely become part of the transition.
TABLE 3.14 Investment Costs ($/gge per day)
| H2 Production Method | Plant + CCS | Distribution | Stations | Total | |||||
| Distributed natural gas reforming | 700 | 0 | 2,345 | 3,045 | |||||
| Distributed electrolysis, current grid | 860 | 0 | 2,345 | 3,205 | |||||
| Coal gasification without CCS | 2,250 | 225 | 2,345 | 4,820 | |||||
| Coal gasification with CCS | 3,020 | 225 | 2,345 | 5,590 | |||||
| Central natural gas reforming without CCS | 400 | 225 | 2,345 | 2,970 | |||||
| Central natural gas reforming with CCS | 740 | 225 | 2,345 | 3,310 | |||||
| Biomass gasification without CCS | 1,040 | 225 | 2,345 | 3,610 | |||||
With the increasing amounts of domestically available natural gas and the lower prices for natural gas compared to crude oil projected in the AEO 2011 (EIA, 2011a) study price basis, several other combinations of basic resources and hydrogen-production processes could be viewed as possible in the future with different hydrogen costs and GHG emissions. Some of the many possible pathways for making large amounts of hydrogen are shown in Table 3.15 with the resulting long-term hydrogen cost and GHG emissions.
- A low-cost case—The emphasis is on low-cost hydrogen from several resources with little to no emphasis on GHG reductions. Hydrogen is produced from: 25 percent distributed natural-gas reforming, 25 percent coal gasification without CCS, 25 percent central natural-gas reforming without CCS, and 25 percent biomass gasification without CCS.
- A partial CCS case—The emphasis is on low-cost hydrogen, but CCS is used for all coal and central natural gas processes. Hydrogen is produced from: 25 percent distributed natural gas reforming, 25 percent coal gasification with CCS, 25 percent central natural gas reforming with CCS, and 25 percent biomass gasification without CCS.
- A low-GHG case—The emphasis is on low GHG emissions with less regard to hydrogen cost. Hydrogen is produced from: 10 percent distributed natural-gas reforming, 40 percent central natural gas reforming with CCS, 30 percent biomass gasification without CCS, and 20 percent low GHG grid electricity for electrolysis.
More than 200 FCEVs have been demonstrated in the United States over the past 10 years. Several of the auto companies developing FCEVs have gone through multiple iterations to improve performance. Five of these companies—General Motors, Daimler, Toyota, Honda, and Hyundai—have reaffirmed near-term (2015) commercializa-
TABLE 3.15 Alternate Scenario Hydrogen Costs and GHG Emissions
| $gge H2 | kg CO2e/gge H2 | |
| Low-cost case | 3.85 | 12.2 |
| Partial CCS case | 4.10 | 5.1 |
| Low-GHG case | 4.80 | 2.6 |
tion plans for FCEVs. Because these are all multinational companies, the commercialization plans certainly will vary in different markets.
In the United States, there have been about 60 hydrogen fueling stations constructed to service the FCEV demonstration efforts (DOE-EERE, 2012a). Given that the number of vehicles is small, none of these stations is of even small commercial size. They demonstrate, however, the importance of distributed technologies to starting the infrastructure. General Motors has joined 10 companies, government agencies, and universities to build 20 to 25 hydrogen-fueling stations in Hawaii by 2015 (DeMorro, 2010). Several countries have formed much larger infrastructure plans and consortiums than the one in the United States to support early FCEV commercialization. In 2010 Japan announced plans for 1000 hydrogen stations and 2 million FCEVs by 2025 (DOE-EERE, 2011a). To support these goals, a consortium of 13 companies was established to focus on the hydrogen infrastructure. Germany has announced plans to build 150 hydrogen stations by 2013 and up to 1000 by 2017.
Although technology is available to provide competitively priced hydrogen from natural gas, technology improvements are needed to provide low-cost hydrogen that is also low in net GHG emissions. Continuous government support for RD&D is required.
The robust performance and the durability of a fueling station with sustained high-volume usage remain to be verified through demonstration.
The high cost of the FCEV is a barrier to wide commercialization for the vehicles and hydrogen. A viable pathway is needed for creating the initial hydrogen infrastructure and for dealing with high initial hydrogen costs. This pathway likely will require government actions.
The lack of an incentive to provide low-GHG fuels in general reduces the benefits for transitioning toward alternative fuels. It also reduces the incentive to make hydrogen from the more costly but lower-GHG methods.
Perceived, real and unknown safety issues with hydrogen production and use especially in a consumer environment could result in delays in acquiring, zoning, and permitting authorizations. There are significant practical challenges of developing sites especially for urban stations within the footprint of existing fueling sites.
Finding: Making hydrogen from fossil fuels, especially natural gas, is a low-cost option to meet future demand from FCEVs; however, these methods result in significant GHG emissions. Making hydrogen with low GHG emissions is more costly (renewable electricity electrolysis) or requires new production methods (e.g., photoelectrochemical, nuclear cycles, biomass gasification, and biological methods) and CCS to manage emissions. Continued R&D is needed on low-GHG hydrogen production methods and CCS to demonstrate that large amounts of low-cost and low-GHG hydrogen can be produced.
3.5 NATURAL GAS AS AN AUTOMOBILE FUEL
Natural gas can be used for transportation via several pathways, each of which has advantages and challenges (see Appendix G.7). None of them is of much commercial significance in the United States as of 2012.
Less than 3 percent of the natural gas consumed in the United States is for transportation, and most of that is used for powering the transportation pipeline and distribution system for natural gas. Natural gas as an automobile fuel will have to compete with other existing uses of the gas (for electricity generation, and for residential, commercial, and industrial uses). This section addresses the direct use of CNG in internal combustion engines (CNG vehicles, or CNGVs).
The other pathways are considered in other sections of this report. Methanol as a transportation fuel is discussed in Appendix G.8.
3.5.1.1 Net GHG Emissions from CNG Use
Natural gas from production wells is composed mostly of methane (70 to 90 percent), with some ethane, propane, and butane (0 to 20 percent), CO2 (0 to 8 percent), N2 (0 to 5 percent), H2S (0 to 5 percent), traces of O2, and traces of the noble gases Ar, He, Ne, and Xe (NaturalGas.org, 2011). Natural gas holds promise for providing part of the energy requirements of automobile transportation. Displacing a significant portion of petroleum-based fuels would have large societal and economic benefits by reducing the externalities associated with petroleum importation (e.g., supply and price instabilities, security and defense costs, oil import-related trade and export-import imbalances).
Natural gas vehicles, fueled by CNG or liquid natural gas, are among the most immediately attainable alternative-fueled vehicles. Given methane’s molecular structure, natural gas has the highest energy content or hydrogen-to-carbon weight ratio of all fossil fuels. Nevertheless, the use of natural gas, like other forms of primary energy, has associated GHG emissions, including methane emissions, during exploration, well drilling, and the well-to-tank transmission for natural gas. Life-cycle analyses that account for upstream and downstream GHG emissions for natural gas have been published by the DOE’s National Energy Technology Laboratory (DiPietro, 2010). In terms of kg CO2e/million Btu, drilling and extraction generate 19.9 and pipeline transport generates 3.3 (mostly natural gas to power the pumps), for a total upstream (well to tank; WTT) of 23.2. Compression of natural gas into CNG from pipeline pressure to about 3,600
psi adds another 3.5 percent (range 2 to 5 percent), or 0.8 kg CO2e/million Btu to the GHG emissions.
The Argonne National Laboratory’s GREET model uses smaller WTT estimates. For example, the 1.8b version of that model released in September 2008 estimated the upstream emissions to be 9.6 kg CO2/million Btu (ANL, 2011). The model estimated vehicle tank-to-wheel (TTW) CO2 emissions of 53.9 kg CO2/million Btu. Thus, the well-to-wheels CO2 emissions for CNG as a fuel are 9.6 + 53.9 = 63.5 kg CO2/million Btu. In 2011 the GREET model estimates were updated to include higher effects of methane leakage and other changes, yielding an upstream estimate of 14.2 kg CO2/million Btu for shale gas. This estimate is used in this report for all pathways using natural gas as a primary source.20 Another life-cycle analysis by Burnham et al. (2012) indicated that the life-cycle GHG emissions of natural gas are 23 percent lower than those of petroleum-based gasoline and 43 percent lower than those of coal. Jiang et al. (2011) estimated the life-cycle GHG emissions for producing electricity from shale natural gas to be 20 to 50 percent lower than the lifecycle GHG emissions for producing electricity from coal.
Fugitive natural gas emissions from increasing use of natural gas are the subject of current analyses. In 2010, the EPA reissued its methane emissions guidelines during natural gas extraction, with substantially increased figures versus their previous estimates (EPA, 2010a). Howarth et al. (2011) estimated the leak rate of methane as a percentage of total natural gas produced to be in the range of 3.6 to 7.9 percent. Of the methane leaked, 1.6 percent was attributed to methane escaping from flow-back fluids (1.6 percent) and from drill-out (0.33 percent). The remainder was attributed to venting and equipment leaks, and emissions during liquid unloading, gas processing, and transport, storage, and distribution. The methodologies and data used in the estimates of methane leakage by the EPA and by Howarth et al. were strongly critiqued by an IHS CERA report, Mismeasuring Methane: Estimating Greenhouse Gas Emissions from Upstream Natural Gas Development (Barcella et al., 2011). Analysis in that report suggests much lower fugitive methane emissions. Burnham et al. (2012) estimated methane leakage in the range of 0.97 to 5.47 percent for conventional natural gas pathways and 0.71 to 5.23 percent for shale-gas pathways. Methane leakage from the sources mentioned is a concern because of the large global warming potential of methane, but its extent is uncertain (Alvarez et al., 2012). The sources of leakage are amenable to various forms of reduction or control by conventional technologies, representing ongoing considerations in sorting out the environmental aspects of shale gas and conventional natural gas. Several studies are underway to consolidate and define fugitive natural gas emissions from shale-gas operations as of the writing of this report.
Recognizing that some cost-effective measures exist for reducing methane emitted from producing natural gas, in 2011, the EPA proposed amendments to its air regulations for the oil and gas industry that will reduce GHG and other emissions from exploration, drilling, and production (EPA, 2011c). The final regulation was issued in April 2012. In it, the EPA estimates reductions of 1.0 to 1.7 million tons per year of methane emissions associated with drilling and transportation of natural gas (EPA, 2012b).
3.5.2.1 Natural Gas Supply, Demand, and Prices
The United States used about 98 quads (quadrillion, or 1015, Btu) of energy from the nation’s primary energy sources in 2010 (LLNL, 2012). Of the 24 quads of natural gas consumed in the United States in 2010, 98 percent originated from North America and 85 percent was of domestic origin. (In comparison, the United States consumed 37 quads of petroleum, about 50 percent of which was imported.) Transportation used 28 quads of primary energy, 95 percent of which was from petroleum. With a typical 25 percent overall efficiency, a useful energy of about 7 quads is turning the wheels of the U.S. transportation fleet.
Of the 24 quads of natural gas, about 7 quads were used to generate electricity. Natural gas is becoming more attractive for electricity generation than coal, according to recent references quoting numbers from the DOE’s Energy Information Administration (Begos, 2012). Electricity generation from natural gas in the United States increased from about 601 billion kWh in 2000 to 981 billion kWh in 2010. During the same period, electricity generation from coal declined from 1,966 billion kWh to 1,850 kWh (EIA, 2011b). Between 2010 and 2035, 80 percent of all newly added electricity generation capacity is expected to come from natural gas-fired plants (EIA, 2011a; NaturalGas.org, 2012). With recently increased concerns about the future of nuclear energy, some of the contemplated future nuclear electric capacity will likely shift to natural gas-fired power plants as well.
According to the June 18, 2009, report of the Potential Gas Committee on the assessment of the year-end 2008 natural gas reserves (Potential Gas Committee, 2009), the United States has 1,836 tcf (trillion, or 1012, standard cubic feet; 1 tcf is equal to approximately 1 quad) of probable natural gas resources, defined as being potentially economically extractable by the use of available technology at the then-current economic conditions. The above number (1,836 tcf) is the sum of 1,673 tcf in traditional reserves and 163 tcf in coal-bed reservoirs. Of the 1,836 tcf of probable reserves, shale gas accounts for 616 tcf (33 percent). In addition to the above probable reserves, the United States also has 238 tcf of proved natural gas resources, defined as deemed to
_______________________
20The CNG GHG emissions are estimated as follows: 14.2 kg CO2/million Btu upstream plus 59.8 kg CO2/million Btu combustion plus 7 percent of this total for pipelining and compression = 79.2 kg CO2/million Btu or 9.2 kg CO2/gge.
be economically extractable (rather than being potentially extractable) or already being extracted economically. The estimated total natural gas reserves of 2,074 tcf (1,836 + 238) represent an increase of 542 tcf (35 percent) over the estimate in the previous biannual assessment. The natural gas consumption of the United States was about 24.1 tcf in 2010 (EIA, 2011b). Dividing the 2008 estimated total of probable and proved natural gas reserves by the 2010 annual consumption gives an estimate of 86 years’ worth of natural gas. It has been argued that only a fraction of probable reserves can be recovered economically (Brooks, 2010), so that the “probable technically recoverable resources” would be only 441 tcf, of which 147 tcf is the shale-gas component.
The 2009 report upgraded the probable reserves mainly by reclassifying known shale gas reserves from possible to probable, due to the rapid evolution and deployment of new technology. The new shale gas extraction technology combines two technologies from the oil fields, horizontal drilling and hydraulic fracturing. (See Technology Review, 2009, for a video schematic of these processes.)
The newly reclassified shale gas reserves are located in Louisiana, Texas, the Rocky Mountains, West Virginia, Pennsylvania, and New York. There are large shale gas fields outside the United States as well, and these fields also are likely to be accessible via the new technology. The BP Energy Outlook 2030 (BP, 2011) stated that in 2009, the world had 6,621 tcf of proved gas reserves, which would be sufficient for 63 years of production at 2010 production levels. Global reserves of unconventional natural gas could potentially add another 30 years to natural gas use.
Most of the natural gas-based transportation fuels are expected to gain new impetus in light of the dramatically upgraded estimates of global natural gas resources. Future natural gas supply and consumption volumes and prices, broken down to sources and uses, are published yearly by the U.S. Department of Energy’s Energy Information Administration, AEO. The AEO 2011 early release projects to the year 2035 (EIA, 2012a). According to AEO (EIA, 2011a), between the years 2010 and 2035, natural gas consumption will grow by 16.8 percent. The share of shale gas will increase from 23 to 49 percent (Figure 3.4). The share of natural gas in transportation will remain at 3 percent, which roughly accounts for the amount of natural gas used for operating the pipelines. In other words, the 2011 AEO is not counting on any significant increase in the use of natural gas for transportation in the United States. This seems to also hold on the global scale. The BP Energy Outlook 2030 (BP, 2011) projected global use of CNG for transport to be limited to 2 percent of the global demand for transportation fuels.
3.5.2.2 Will There be Enough Natural Gas for LDVs?
In the year 2000, the 110,000 natural gas vehicles in the United States consumed between 8.3 and 12.3 billion standard cubic feet of natural gas, which is between 0.036 and 0.053 percent of the U.S. natural gas consumption (Campbell-Parnell, 2011). According to the 2011 AEO (EIA, 2011a), the U.S. LDV vehicle stock will increase from about 128 million vehicles in 2011 to about 186 million vehicles in 2035. Assuming a 10 percent penetration of CNGVs in 2035 (EIA, 2011a), 45 mpgge, and 14,000 mi/yr, this would translate to a natural gas consumption of 0.73 tcf/yr. Natural gas consumption is forecasted by the AEO 2011 to increase from 24.1 tcf in 2010 to 26.5 tcf in 2035 (with only 6 percent for transportation; mostly natural gas consumed by powering the pipeline system itself). Therefore, a 10 percent CNGV penetration in the 2035 LDV fleet would add only 2.8 percent to the natural gas consumption in that year. Thus, the
FIGURE 3.4 U.S. natural gas production (trillion standard cubic feet) from 1990 to 2035.
SOURCE: EIA (2012a).
natural gas supply is unlikely to limit the early penetration of CNGVs.
Several studies project that LDVs powered by natural gas will remain a niche for a while in the United States. Those studies include The Future of Natural Gas by the Massachusetts Institute of Technology (MIT, 2011), a market analysis of natural gas vehicles by TIAX (Law et al., 2010), an analysis of long-term natural gas demand by Simmons & Company (2011), and an analysis of natural gas demand for transportation by IHS-CERA (IHS, 2010). TIAX (Law et al., 2010) compared the incremental lifetime costs of LDVs using different technologies and found that the direct costs of natural gas vehicles are favorable compared to BEVs, PHEVs, FCEVs, and flex-fuel vehicles. They concluded that CNGVs could become significant with appropriate policy and incentive programs and projected the use of 5.5 billion gge of CNG (still only 0.7 tcf) by 2035.
The reasons for the slow and late development of light-duty CNGVs in the United States transcend the barriers of CNGV and vehicle conversion costs, lack of luggage and tank volume, and the lack of refueling infrastructure. Development of CNGVs also may be significantly hampered by the attractiveness of alternate uses of natural gas, specifically for electricity generation. The AEO 2011 (EIA, 2011a), for example, shows year 2016 levelized costs for electricity generated by 16 different power plant and fuel technologies. Of these, the lowest levelized cost is shown for natural gas-fired combined-cycle power plants (<7 cents/kwh), followed by hydro (8.64), conventional coal (9.48), wind (9.70), biomass (11.25), advanced nuclear (11.39), advanced coal with CCS (carbon capture and storage) (13.62), and photovoltaic solar (21.02). The AEO projections suggest that natural gas will indeed be most attractive for electric power generation because of its low levelized cost.
3.5.3.1 Natural Gas Fuel Costs and Cost Projections
At filling stations CNG and liquid natural gas are metered and sold on a gallon of gasoline-equivalent basis; the conversion factor of 1 gge = 5.66 lb of natural gas was determined by the National Institute of Standards and Technology (NIST). The prices of natural gas on a gallon of gasoline-equivalent basis are published on the Internet, and they vary by state, region, city, and individual filling station. Natural gas at the time of this writing had a price advantage of about $1 to $2/gge, depending on the particular filling station. For example, overall average U.S. fuel prices reported for the last quarter of 2011 were $3.37/gal for gasoline, and $ 2.13/gge natural gas (DOE-EERE, 2012b).21
At a price differential of $1.24/gge in favor of CNG, 30 mpg, 13,000 mi/yr, and 433 gge/yr consumed, the fuel cost savings would be about $540/yr, returning the original investment in a 2012 Honda Civic Natural Gas (versus the LX) in about 13 years (7,500/540 = 14 years). This payback period is not likely to be perceived by the consumer as economically attractive. Various states and the federal government have offered subsidies, which could amount to $4,000 per vehicle. With a $4,000 subsidy, the economic return period would be reduced to 6 years. CNG economics can thus be significantly better in the states that subsidize CNGVs.
Natural gas prices have declined in recent years, whereas oil prices have been rising. With fuel and vehicle subsidies for natural gas, any continued gasoline price increases could eventually make the original equipment manufacturers’ natural gas vehicles economically attractive.
The appeal of natural gas as an automotive fuel depends to a large extent on the ratio of oil prices to natural gas prices (Figure 3.5). Long-term future natural gas prices have been forecasted by the 2011 AEO (EIA, 2011a) (Table 3.16).
The price customers would pay at the CNG filling station for filling a vehicle was calculated by taking the average of commercial and industrial prices for natural gas and adding a margin sufficient to generate a 15 percent return on an investment of $1.3 million in a CNG filling station servicing 1,000 cars per week at 10 gge per fill per week. This margin was calculated to be $7.76/million Btu or $0.90/gge NG. (The operating costs and capital expenses of this filing station, excluding fuel costs, were $273,351/yr.) CNG filing station costs and additional natural gas pipeline needs are discussed in Appendix G.9.
The U.S. Department of Energy’s Alternative Fuels and Advanced Vehicles Data Center lists 975 public CNG refueling stations as of January 9, 2012 (DOE-EERE, 2012a). Unevenly distributed across the country, they are clustered primarily in California (229 stations), New York (106 stations), Utah (81 stations), Oklahoma (67 stations), Texas (35 stations), and Arizona (30 stations).
The distribution of CNG filling stations corresponds somewhat to the clustering of CNGVs. The EIA (2008a) listed a CNGV count of 113,973 as of 2008, with the largest number in California (35,980 vehicles), followed by Texas (11,032 vehicles), Arizona (10,072 vehicles), and New York
TABLE 3.16 Long-term Future Natural Gas Prices ($/million Btu) Forecasted by the 2011 Annual Energy Outlook
| 2010 | 2020 | 2035 | 2050 (extrapolated) | ||||||
| Commercial natural gas | 8.91 | 8.95 | 10.98 | 13.02 | |||||
| Industrial natural gas | 4.80 | 5.36 | 7.2 | 9.06 | |||||
| Vehicle natural gas in $/gge | 13.94 | 14.24 | 16.81 | 18.80 | |||||
| 1.69 | 1.73 | 1.96 | 2.18 | ||||||
SOURCE: Data from EIA (2011a).
_______________________
21In 2011, the quarterly average price ranged from $3.37 to $3.69/gal for gasoline and from $2.06 to $2.13/gge for natural gas (DOE-EERE, 2011b,c,d; 2012b).
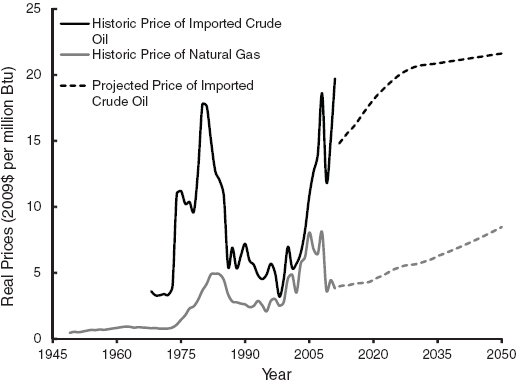
FIGURE 3.5 Historic and projected prices of natural gas and imported crude oil.
NOTE: The prices from 2035 to 2050 were projected by extrapolating the 2030-2035 annual growth rate in EIA (2011a).
SOURCE: Data from EIA (2011a,b).
(10,017 vehicles). The regional clustering of CNG filling stations as a practical model for infrastructure build-up matches the results of models for the clustering of hydrogen filling stations for FCEVs and of public charging stations for BEVs.
CNG prices vary regionally and locally. According to the DOE’s Alternative Fuels and Advanced Vehicles Data Center (DOE-EERE, 2011b), average CNG prices per unit gallon of gasoline-equivalent in April 2011 ranged from $1.39 to $2.41 ($2.41 in the Central Atlantic, $2.38 in New England, $2.32 on the West Coast, $1.87 in the Lower Atlantic, $1.84 in the Gulf Coast region, $1.66 in the Midwest, and $ 1.39 in the Rocky Mountain region).
Environmental standards, construction permits, labor costs, natural gas and gasoline costs, vehicle and population density, purchasing power and customer preferences, proximity to natural gas pipelines, the corresponding industrial and commercial natural gas prices, and a host of other factors vary with individual cities, counties, states, and regions, all of which have some effect on the actual and potential extent and rate of penetration of CNGVs. Because of the recent discovery of the U.S. abundance of natural gas, the subject of regional differences needs to be further examined.
3.5.4 Safety of Natural Gas and Compressed Natural Gas Vehicles
Natural gas has a narrow flammability range, which is between 5 and 15 percent by volume in air. Natural gas is lighter than air, and so a gas leak disperses quickly. Unlike gasoline, natural gas will not cause a combustible liquid spill. Its high autoignition temperature means that natural gas does not easily self-ignite on hot surfaces below 540°C,a property quoted as another safety factor in its favor.
CNGVs meet the same safety standards as gasoline and diesel vehicles, and they also meet the National Fire Protection Association’s Vehicle Fuel System Code. CNG tanks meet DOE and other government safety standards and have been certified for that purpose. The Clean Vehicle Education Foundation has published a Technology Committee Bulletin (Clean Vehicle Education Foundation, 2010) that provides a detailed treatise of safety considerations for CNGVs. The Clean Vehicle Foundation actually stated that CNG-powered vehicles are considered to be safer than gasoline-powered vehicles.
The DOE has detailed safety analysis and operating recommendations for natural gas filling stations. Properly designed, maintained, and operated facilities for CNG refueling appear to represent no undue safety problems to the public.
Public policies at various government levels have not kept up with the increased abundance of natural gas in the United States and are expected to develop rapidly in the coming years.
The CNG infrastructure (filling stations, gas distribution) is in its early stage of development and requires massive expansion. Regional, clustered development will remain the preferred model.
Finding: With increasing economic natural gas reserves and growing domestic natural gas production mostly
from shale gas, there is enough domestic natural gas to use within the transportation sector without significantly affecting the traditional natural gas markets. The opportunities include producing electricity for PHEVs, producing hydrogen for FCEVs, and using as a fuel in CNGVs.
Finding: CNG used as a transportation fuel is an important near-term transition opportunity that could be exploited because of its ability to economically replace petroleum and to reduce GHG emissions from the LDV fleet.
3.6 LIQUID FUELS FROM NATURAL GAS
The production of liquid fuels—diesel, gasoline, or a combination of both—from natural gas has been practiced commercially since the early 1980s. As in the case of coal, the first step in the GTL process is the conversion of natural gas into a mixture of carbon monoxide and hydrogen (synthesis gas). There are two options for using this synthesis gas to produce liquid fuels. One is the production of methanol followed by the conversion of methanol into gasoline (MTG). The other option is the conversion of the synthesis gas via FT chemistry to a broad range of paraffinic hydrocarbons. The hydrocarbon molecules with more than 20 carbons are then hydrocracked into molecules in the diesel (15-20 carbons) and naphtha (6-12 carbons) range. The quality of the diesel fuel is excellent but the naphtha has a low octane value and has to be further processed to be used as gasoline (NAS-NAE-NRC, 2009b).
For nearly 10 years in the 1980s, Mobil Corporation operated a facility in New Zealand that produced gasoline by the MTG process (ExxonMobil, 2009). Today, the facility makes only methanol for chemical use (Tabak, 2006) because converting the methanol to gasoline is not viewed as economical at current gasoline prices. Shell has produced diesel fuel and lubricants since the late 1980s in a facility in Malaysia via FT chemistry and Shell is building a plant in Qatar, based on the same process chemistry. That facility is expected to eventually produce more than 140,000 barrels of diesel fuel per day (Kingston, 2011). Another facility in Qatar that is smaller (about 34,000 bbl/d) and based on the same FT chemistry is coowned by Sasol, Chevron, and the Government of Qatar. Similar facilities have been proposed for gas-rich locations such as Nigeria (Chevron, 2011).
The conversion of natural gas into synthesis gas is significantly simpler when compared to the production of synthesis gas from coal. At present, the preferred pathway uses what is called an auto-thermal reactor (ATR). In an ATR, a portion of the natural gas (methane) is burned with oxygen into CO2 and water vapor. This reaction is highly exothermic (that is, it releases heat) and results in a mixture of CO2, unreacted methane, and steam at temperatures close to 2,000°C. This mixture is converted into carbon monoxide and hydrogen in a fixed bed containing a nickel-based catalyst. Although ATRs are very efficient and compact, the design and operation of the feedstock and burner system requires careful attention to the mixing of oxygen, steam, and methane (Haldor Topsoe, 2011).
The processing steps are significantly less complicated than in a coal plant. The natural gas, if needed, is cleaned of sulfur compounds before being fed to the ATR. Because methane has four hydrogen atoms for each carbon atom, the synthesis gas from the ATR has the required ratio of two molecules of hydrogen per molecule of carbon monoxide. Thus, the synthesis gas can be used without further processing to produce either methanol or FT hydrocarbons followed by the conversion of these into gasoline or a diesel/naphtha mixture as discussed above.
The data presented in Table 3.17 were derived from a report prepared for the Alaska Natural Resources to Liquids LLC and requested by the Alaska legislature (Peterson and Tijm, 2008). The results of that study were in good agreement with data published by various companies (Shell, Sasol, and ExxonMobil) on GTL technology performance and economics.
As in the case of CTL, this committee assumes that the GTL plants built later will benefit from a learning curve. Therefore, the estimated investment required was $5 billion for a 2020 facility, $4 billion for a 2035 facility, and $3 billion for a 2050 facility. These investment costs do not include CCS. Although CCS could be used in a GTL facility, the amount emitted from a GTL facility is significantly less than that for similar-size CTL facilities. Therefore, CCS was not included in GTL facilities for the purpose of this study.
TABLE 3.17 GTL Outlook Process Data
| GTL/MTG | 2020 | 2035 | 2050 | ||||||
| Gas, million scf/d | 400 | 400 | 400 | ||||||
| Fuel production, bbl/d | 50,000 | 50,000 | 50,000 | ||||||
| Investment, $billion | 5.0 | 4.0 | 3.0 | ||||||
| Product cost, $/bbl | 103.5 | 106.0 | 109.0 | ||||||
| CO2e produced by the process, metric tons/d | 3,840 | 3,840 | 3,840 | ||||||
| CO2 vented, metric tons/d | 2,110 | 2,110 | 2,110 | ||||||
| CO2 stored, metric tons/d | — | — | — | ||||||
NOTE: Product cost basis: (1) 20 percent of capital annual charge (financing, return on capital, maintenance), 90 percent capacity utilization; (2) natural gas prices as per AEO 2011 (EIA, 2011a), $5.36/million cubic feet for 2020, $7.21/million cubic feet for 2035 and $9.06/million cubic feet in 2050; (3) CO2e emissions from gas production are based on GREET estimates for the production and transport of gas.
The cost estimates for GTL are based on the FT process economics (see Table 3.17). There are no published data available for the MTG option. For the purpose of this study, capital cost and overall performance data for the MTG option are expected to be similar to the numbers presented in Table 3.17. The investment required for the GTL processes is lower than the investment estimated for the CTL options. This is expected because CTL requires the greater complexity of coal gasification and the complex cleaning of the synthesis gas, and because of the fact that half of the coal has to be converted to CO2 (to make hydrogen), which in turn has to be captured and stored (CCS).
The cost for the liquid fuel from a GTL plant is about $106/bbl in 2035, which is less than the price of crude oil in 2035 ($125/bbl) forecasted by EIA (2011a). However, the GTL cost estimate is based on a natural gas price of $7.21/million cubic feet in 2035, which is lower than natural gas prices in 2008 and earlier. If the price of natural gas were to reach $10.0/million cubic feet, the liquid product cost would increase to $130/bbl. The cost of the liquid product in 2050 is estimated at $109/bbl based on a natural-gas price of $9.06/million cubic feet. If the natural-gas price were $11.0/million cubic feet, the liquid-product cost escalates to close to $130/bbl.
GTL technology has been commercialized in a number of locations where the price of natural gas is low because those locations are far away from markets where the gas can be used directly for power and heat generation. Moreover, all the GTL facilities are based on producing diesel fuel, naphtha, and in some cases high-value lubricants.
When considering the application of GTL technology in the United States, two factors need to be considered. First, the MTG option might be preferred because gasoline is a more widely used transportation fuel than diesel. Second, the price of natural gas will likely be significantly higher in the United States than in other areas of the world where it is readily available (e.g., in the Middle East and in West Africa) because it can be readily used in heating, power generation, petrochemical production, and other industries. The forecasted production of liquid fuels from natural gas (GTL) assuming an optimistic outlook and a more realistic outlook is summarized in Table 3.18.
The estimates for fuel production from GTL are sensitive to natural gas prices. Using the 2011 AEO (EIA, 2011a), the cost of the fuel in 2035 is about $105/bbl, which is lower than the crude-oil price forecasted for that year. However, a 25 percent increase in the price of natural gas would raise the final-product price well above the crude-oil price.
The GHG emissions for the production of GTL fuel are, as in the case of coal, comparable to the emissions from producing petroleum-based fuels. Thus, GTL without CCS for LDVs reduces the consumption of petroleum-based fuels but does not yield any GHG reduction. Adding CCS to a GTL facility would have a small effect on the life-cycle GHG emissions of the fuel produced because the GHG releases that could be captured at the conversion facility are small compared to the CO2 release from combusting the liquid fuel.
TABLE 3.18 GTL Outlook Production Estimates
| 2020 | 2035 | 2050 | |||||||
| Optimistic outlook | |||||||||
| GTL/MTG plants | 1 | 4 | 12 | ||||||
| GTL/MTG production, bbl/d | 50,000 | 200,000 | 600,000 | ||||||
| Realistic outlook | |||||||||
| GTL/MTG plants | 1 | 3 | 6 | ||||||
| GTL/MTG production, bbl/d | 50,000 | 150,000 | 300,000 | ||||||
Because natural gas is readily available throughout most of the country, there are no major issues with either infrastructure or the location of GTL facilities.
Although the GTL process includes a complex step for generating synthesis gas, there are no unique safety issues. Natural processing, transmission, and use are widely practiced in the United States. The process of converting natural gas to a liquid fuel for LDVs has many similarities to petroleum-refining processes, and well-known safety practices can be applied.
One important barrier to the wide use of natural gas to make liquid fuels is the cost over the life of commercial GTL facilities and the availability of natural gas. Recent technology advances for producing gas from tight shales and other low porosity reservoirs suggest that the natural-gas resources in the United States are significantly greater than previously estimated. The resource availability is a positive factor, but the cost and the environmental impact of producing this tight gas are unclear at present. Moreover, natural gas is used in all sectors of the economy, and the distinct advantage of using natural gas in electricity generation suggests that the demand for gas in this sector could increase dramatically. Use of natural gas directly in LDVs is also being proposed. (See Section 3.5, “Natural Gas as an Automobile Fuel.”) The balance between supply and demand for natural gas in the United States depends on the level of consumption in many sectors and the level of production. Therefore, predicting the future price of natural gas is difficult. Because the cost of the gas feedstock is a major factor in the cost of the GTL fuel made, the estimate for total liquid fuels produced from natural gas in 2050 is less than 600,000 bbl/d in the optimistic
case. That production level requires an annual consumption of 1.6 tcf of natural gas, or about 8 percent of the present production in the United States.
Liquid fuels, both gasoline and diesel, have been produced from coal at a significant scale since the 1930s. At present, the CTL facilities with the largest capacity are in South Africa and produce more than 100,000 bbl/d of liquid products. Moreover, a number of proposed facilities are being considered in China.
There are two technology options for the production of liquid fuels from coal: direct and indirect liquefaction. The direct liquefaction of coal involves reacting coal with hydrogen or a hydrogen-donating solvent. This technology option has been the subject of research, development, and pilot-scale demonstration since the late 1970s. The consensus view is that this technology is still in development and that the complexity of the process scheme and the poor quality of the liquid products are major limitations. However, a demonstration facility was built in China, and that facility may provide a definitive assessment of the coal-to-liquid fuels option (NMA, 2005; NPC, 2007; NAS-NAE-NRC, 2009b).
This section focuses on the indirect liquefaction option that involves the gasification of coal to a mixture of carbon monoxide and hydrogen (synthesis gas) followed by the conversion of this gas into liquid products. There are two schemes to make the synthesis gas into liquid-fuel products. One option is to convert the synthesis gas into methanol followed by MTG (Zhao et al., 2008). The second option is to convert the synthesis gas into a broad range of hydrocarbons via FT chemistry followed by the hydrocracking of the molecules with more than 20 carbons into shorter-chain molecules. The FT option results in a mix of liquid products that includes mostly diesel fuel and a significant amount of naphtha that can be upgraded to gasoline.
The commercial-scale facilities in South Africa are producing diesel and gasoline from coal by the FT option. Although the Mobil Corporation operated a facility that used the MTG option, the feedstock was natural gas rather than coal.
In the report Liquid Transportation Fuels from Coal and Biomass: Technological Status, Costs, and Environmental Impacts (NAS-NAE-NRC, 2009b), a process scheme labeled coal-and-biomass to liquid fuel (CBTL) is proposed. The process uses a separate gasifier for the coal and the biomass feedstocks. The effluents from these gasifiers undergo a number of separation steps to remove solid and gaseous impurities. The biomass gasifier effluent also includes a thermal cracking step to convert the tar produced from the biomass to lighter products. The clean-up streams are then combined and undergo the required processing steps to make liquid products from carbon monoxide and hydrogen and remove and compress the CO2.
A number of cases presented in NAS-NAE-NRC (2009b) include or exclude CCS, and in other cases the proposed facility produces significant amounts of electric power (these are called once-through cases). Although interesting synergies have been identified in these schemes, all process schemes require different gasification reaction systems for the coal and for the biomass. They can be viewed as requiring a separate CTL and BTL gasification plants in a given site. The number of sites in the United States where there are significant amounts of biomass and coal for commercial-scale facilities might be small.
The potential benefits of combining the gas products from the biomass and coal gasification to make liquid fuels and electric power are clear from the studies available. A CBTL facility produces liquid fuels at a higher cost than does a CTL facility but at lower cost than a BTL facility. Moreover, by capturing the CO2 produced in the biomass portion of the facility, the process drastically reduces the life-cycle GHG emissions of the liquid fuels (the emissions during their combustion are counterbalanced by the CO2 taken up during plant growth). The potential benefits of CBTL facilities, while significant, will require commercial-scale demonstrations of BTL technology and combining it with CTL technology.
The CBTL process was not included in the case study model runs explained in Chapter 5 because it is a derivative process of two commercially available processes. Coal conversion and biomass conversion to liquids are individually included in all of the model scenarios.
The United States has ample coal resources that can allow the production of significant amounts of liquid fuels such as gasoline and diesel from coal. Most coal produced in the United States (about 1 billion tons per year) is used to generate electricity. In principle, additional coal could be mined to produce liquid fuels because the coal reserves in the United States are estimated to be in the range of 250 billion tons. However, concerns have been raised about the environmental impact of coal mining and of the disposition of mineral ash present in coal. Those concerns apply to all uses of coal (AAAS, 2009; EPA, 2011a,b).
The process to convert coal into a liquid fuel is complex and expensive. The gasification of the coal is the most challenging process step. The coal has to be fed into a reactor that operates at pressures ranging from 20 to 50 atmospheres along with pure oxygen and water. The average reactor temperature is about 800°C. Because coal is a solid and its quality varies, the feed system is complex and sensitive to the coal quality. Moreover, coal contains a number of impurities including mineral ash, sulfur, nitrogen and mercury. A
number of process steps are needed to remove the byproducts of the gasification reaction to yield a pure stream of carbon monoxide and hydrogen (KBR, 2011).
The second major challenge in making liquid fuels from coal that applies to both the FT and the MTG options is the fact that chemistry dictates that two molecules of hydrogen react with one molecule of carbon monoxide. Because coal, on average, contains only an atom of hydrogen per atom of carbon, half of the carbon monoxide produced in the gasification step has to be used to make additional hydrogen. This is done using the water gas shift reaction where water and carbon monoxide are converted into carbon dioxide and hydrogen. Thus, this reaction step yields the required 2:1 mole ratio of hydrogen to carbon monoxide needed for the subsequent reaction steps and also produces one molecule of carbon dioxide for each molecule of carbon monoxide. In other words, half of the coal is converted to CO2 and the other half into the reactants needed for the next process steps. Therefore, CCS is necessary if coal is to be used to make liquid fuels with life-cycle GHG emissions in the range of those from use of petroleum-based fuels. Although there are a few facilities that use CCS, there is consensus that a large-scale demonstration in a variety of geological formations is required before CCS can be deemed commercially acceptable.
The conversion of carbon monoxide and hydrogen via MTG or FT to diesel or gasoline presents less of a technology challenge and, has been done commercially for many years (ExxonMobil, 2009; NAS-NAE-NRC, 2009b). Most of the commercial facilities have used or are using natural gas rather than coal as the feedstock. The use of natural gas to make liquid fuels is discussed in a separate section above in this chapter.
The data presented in Table 3.19 are derived from Liquid Transportation Fuels from Coal and Biomass: Technological Status, Costs, and Environmental Impacts (NAS-NAE-NRC, 2009b), which describes in detail the process schemes briefly reviewed here. It also described the challenges and potential of the various technology options. It includes estimates of the capital and operating costs for CTL facilities.
Here, the cost of the first CTL facility built by 2035 has been estimated to be 20 percent higher than the facilities built later on. The MTG facility is estimated to be lower in capital cost and to require less coal for the same level of production of 50,000 bbl/d of liquid-fuel product than would the FT process. The MTG process is more selective than the FT process as indicated by the higher energy conversion efficiency. Efficiency is the percent of the energy content of the coal that is contained in the liquid produced. The efficiency in the 50 percent range indicates that close to half of the coal has to be converted into CO2. That amount of CO2 has to be “stored” via CCS in both cases. The capital cost estimated for a facility with a 50,000 bbl/d capacity is high and thus has a major impact on the cost of the liquid-fuel product.
TABLE 3.19 CTL Outlook Process Data
| CTL/FT | 2020 | 2035 | 2050 | ||||||
| Coal, tons/d | 26,700 | 26,700 | 26,700 | ||||||
| Fuel production, bbl/d | 50,000 | 50,000 | 50,000 | ||||||
| Investment, $billion | 6.0 | 6.0 | 5.0 | ||||||
| Product cost, $/bbl | 126.8 | 122.5 | 104.7 | ||||||
| CO2 coal production, metric tons/d | 2,580 | 2,580 | 2,580 | ||||||
| CO2 vented, metric tons/d | 5,011 | 5,011 | 5,011 | ||||||
| CO2 stored, metric tons/d | 29,208 | 29,208 | 29,208 | ||||||
| CTL/MTG | 2020 | 2035 | 2050 | ||||||
| Coal, tons/d | 23,200 | 23,200 | 23,200 | ||||||
| Fuel production, bbl/d | 50,000 | 50,000 | 50,000 | ||||||
| Investment, $billion | 5.0 | 5.0 | 4.0 | ||||||
| Product cost, $/bbl | 105.2 | 102.5 | 86.0 | ||||||
| CO2 coal prod, metric tons/d | 2,243 | 2,243 | 2,243 | ||||||
| CO2 vented, metric tons/d | 5,520 | 5,520 | 5,520 | ||||||
| CO2 stored, metric tons/d | 23,280 | 23,280 | 23,280 | ||||||
NOTE: Product cost basis: (1) 20 percent of capital annual charge (financing, return on capital, maintenance), 90 percent capacity utilization (2) $50/metric ton of CO2 pipelined and stored underground in 2020, $40 in 2035, and $30 in 2050; (3) coal prices as per AEO 2011 (EIA, 2011a), $1.85/million Btu in 2020, $1.98 in 2035, and $2.00 in 2050; (4) CO2 emissions from the coal production are based on GREET estimates for the production/transport of coal.
SOURCE: Data from NAS-NAE-NRC (2009b).
The cost of the liquid-fuel product made in the CTL facilities is within the range of the cost of a barrel of crude oil forecasted for 2035 in the 2011 AEO (EIA, 2011a) and the cost of a barrel of crude oil extrapolated to 2050. However, the CTL estimate is based on a coal price that remains essentially constant from the 2009 price; a doubling of the coal price will yield product costs of over $150/bbl. Conversely, coal prices could decrease as a result of increasing use of natural gas or other resources for electricity generation. The CTL facilities take a long time to build, and thus their payback requires high product prices for a long period of time.
The process cost estimate for CTL is based on the facilities using Illinois #6 coal and the CTL plants being built in the Midwest. Therefore, the mining and transport of the coal to the CTL facilities are assumed to be handled within the present infrastructure. The liquid-fuel products from the facilities will be consumed in the Midwest and will be marketed using the present infrastructure. The CO2 is assumed to be pipelined and stored underground within a 150-mile range because geological studies indicate a significant storage potential in the Illinois Basin (Finley, 2005). Therefore, the main new infrastructure needed will be the pipelines to transport the CO2, the injection wells to store it in underground
formations, and the equipment to monitor CO2 emissions in the pipelines and from the underground storage formations. All of these costs are included by adding $50/metric ton of CO2 stored in 2020, $40/metric ton of CO2 stored in 2035, or $30/metric ton by 2050 to the product cost.
As mentioned above, CTL technology is used in South Africa at present. The main reason for its commercialization was the need to provide liquid fuels in a country rich in coal. Another major consideration was the embargo of crude oil and petroleum products imposed on the country because of its Apartheid Policy. Economic considerations were, therefore, secondary. While a number of feasibility studies on CTL have been announced in the last 10 years, none of the facilities have reached commercialization. China has been operating a CTL demonstration project (China Shenhua Coal to Liquid and Chemical Co. Ltd., 2010; Reuters, 2011).
There are major barriers to the widespread commercialization of CTL technology. First, the process is complex and costly. Second, large amounts of CO2 generated by the facilities need to be captured and stored. The process to capture CO2 is based on the absorption of the gas in a liquid solvent. A number of solvents have been used, and the process is practiced at a commercial scale. It requires a significant amount of energy, thus reducing the efficiency of the overall process. Third, the transportation and storage of CO2 add to the cost. The gas would be compressed to a pressure of about 125 atmospheres and then pipelined to a region where there is a porous underground formation for storage. Wells will be used to transfer the gas to the formation zone, where the gas is expected to either dissolve in the formation water or be converted to a carbonate salt. In 2011, DKRW Advanced Fuels LLC announced that its subsidiary, Medicine Bow Fuel and Power LLC, entered into a contract to produce liquid fuels from coal and to sell the carbon captured for enhanced oil recovery (DKRW Advanced Fuels LLC, 2011).
Two estimates for the eventual production of liquid fuels from coal are presented in Table 3.20. One is an optimistic estimate, and the other one is a realistic outlook. Both estimates assume that no CTL facilities would be operational in 2020. The technology requires demonstration that large amounts of CO2 can be captured, pipelined, and stored safely, and such demonstrations are not expected to be completed until later in this decade. Moreover, the design and construction of CTL facilities are expected to take at least 5-6 years for the first few facilities.
The actual production of liquid fuels from coal presents the typical safety issues encountered in the handling, gasification, and refining of coal. Thus, CTL safety is expected to benefit from many decades of prior experience. However, there is much less experience with the safety of pipelining and storing large quantities of CO2 (at least 9 million metric tons per year from one CTL facility). Although 3,900 miles of national CO2 pipeline infrastructure exist (Dooley et al., 2001) to transport about 65 million metric tons of CO2 each year for enhanced oil recovery (Melzer, 2012), geologic storage of CO2 is only in the demonstration phase (NAS-NAE-NRC, 2009b; see Section 3.8, “Carbon Capture and Storage,” below in this chapter). The key issue with CCS is to ensure that the CO2 does not leak from either the pipeline or the formation itself. At concentrations higher than 2 percent in air, CO2 can asphyxiate humans and animals (Praxair, 2007). Storing CO2 entails health and ecological risks associated with acute or chronic leaks (NAS-NAE-NRC, 2009b). Clearly, the safety of CCS operations will be a major concern. CCS is being practiced for oil well stimulation in the North Sea, Algeria, and Saskatchewan, Canada, but at a scale much smaller than what is envisioned for a single CTL facility. There are also a number of pilot demonstrations of CTL in the United States (NETL, 2011).
TABLE 3.20 CTL Outlook Production Estimates
| Optimistic Outlook | 2020 | 2035 | 2050 | ||||||
| CTL/FT plants | — | 1 | 2 | ||||||
| CTL FT production, bbl/d | — | 50,000 | 100,000 | ||||||
| CTL/MTG plants | — | 2 | 6 | ||||||
| CTL/MTG production, bbl/d | — | 100,000 | 300,000 | ||||||
| Total production, bbl/d | — | 400,000 | 150,000 | ||||||
| Realistic Outlook | 2020 | 2035 | 2050 | ||||||
| CTL/FT plants | — | 1 | 1 | ||||||
| CTL/FT production, bbl/d | — | 50,000 | 50,000 | ||||||
| CTL/MTG plants | — | 2 | 3 | ||||||
| CTL/MTG production, bbl/d | — | 100,000 | 150,000 | ||||||
| Total production, bbl/d | — | 100,000 | 200,000 | ||||||
An important issue to be considered when estimating the potential supply of CTL liquids is the actual production of coal with its inherent environmental and safety challenges. If only 500,000 bbl/d of liquid-fuel products are to be produced from coal, 85 million tons of coal would have to be mined and transported each year. Locating CTL facilities close to mines would reduce transportation costs. The coal consumption is equivalent to about 10 percent of the U.S. coal production in 2012. There also are environmental and safety issues related to the disposal of coal ash from the coal gasification step. Thus, a major increase in coal consumption to make liquid fuels is not likely.
The most important barrier to the large-scale use of coal to make liquid fuels is the GHG emissions from these facilities. The process eventually yields a liquid fuel for LDVs that has chemical properties substantially similar to those of petroleum-based fuels. Thus, the carbon content of the fuel is the same as the carbon content of petroleum-based fuels. Moreover, the production of CTL fuel with CCS is estimated to emit at least as much CO2 as the production, transport and refining of the same fuel from petroleum. For CTL fuels to have life-cycle GHG emissions equivalent to those of petroleum-based fuels, an amount of CO2 equivalent by weight to the weight of the coal used has to be captured and stored. Thus, CTL technology can reduce the amount of petroleum used in LDVs but does not contribute to reducing GHG emissions.
Finding: GTL fuel and CTL fuel with CCS can be used as a direct replacement for petroleum-based fuel. However, the GHG emissions from GTL or CTL fuel are slightly higher than those from petroleum-based fuel. The role of GTL and CTL with CCS in reducing petroleum use will thus be small if the goals of reducing petroleum use and reducing GHG emissions are to be achieved simultaneously.
3.8 CARBON CAPTURE AND STORAGE
In carbon capture and storage, CO2 is captured from various processes, compressed into supercritical conditions to about 125 atmospheres, pipelined, and then injected into a deep (>2,500 ft), porous subsurface geologic formation. Capturing, storing, and transporting CO2 all have commercial challenges, but, in most cases, the technologies have been demonstrated or are in the demonstration phase. With CCS there are two major options for storage: deep saline formations and enhanced oil recovery.
3.8.1.1 Deep Saline Formations
In the case of a non-hydrocarbon-bearing formation, the CO2 in supercritical state will be dissolved partially in the subsurface formation’s water phase, and the rest will remain in a separate phase. In certain formations, the CO2 will react over a very long period of time with the solids and form solid carbonates. These are slow reactions, because it takes decades for a significant amount of CO2 to be converted to a solid carbonate. Experimental work is being conducted to determine the feasibility of extending this concept to storing CO2 in subsea formations. Currently, demonstrations of deep saline formation CCS of more than 1 million metric tons per year of CO2 are in progress in a number of locations (Michael et al., 2010). Additional smaller demonstration projects are planned or underway in the United States and other regions of the world (NETL, 2007, 2011).
3.8.1.2 Enhanced Oil Recovery (EOR)
CO2 can be injected into already-developed oil fields to recover the oil that is not extracted by initial production techniques. Injected CO2 mixes with the oil in reservoirs and changes the oil’s properties, enabling the oil to flow more freely within the reservoirs and be extracted to the surface. The CO2 is then separated from the extracted oil and injected again to extract more oil in a closed-loop system. Once economically recoverable oil has been extracted from one area of a given reservoir, an EOR project operator reallocates CO2 to other productive areas of the same reservoir. Once all economically recoverable oil has been extracted from a given reservoir, the CO2 remains within the reservoir and the project is plugged and abandoned.
The capture of CO2 from a gaseous stream has been practiced commercially for many years—for example, CO2 has been removed from natural gas produced from reservoirs (Statoil, 2010), and the Weyburn project in Saskatchewan, Canada, has used CO2 captured from a North Dakota coal gasification facility for EOR (Preston et al., 2005, 2009). EOR uses injection of CO2 into a oil reservoir to assist in oil production. In the United States, typical EOR uses about 5,000 cubic feet of CO2 per barrel of oil produced (that is, about 160 lb of carbon produce one barrel of oil, which contains about 260 lb of carbon). Oil and gas reservoirs are ideal geological storage sites because they have held hydrocarbons for thousands to millions of years and have conditions that allow for CO2 storage. Furthermore, their architecture and properties are well known as a result of exploration for and production of these hydrocarbons, and infrastructure exists for CO2 transportation and storage.
To calculate the largest amount of CO2 that could be stored by EOR, all the CO2 used is assumed to remain in the ground. The United States produces about 281,000 bbl/d of crude oil using CO2 EOR (Kuuskaraa et al., 2011). Based on the best-case scenario for CO2 use in EOR, this would sequester 0.26 million metric tons per day of CO2. If all U.S. crude oil was produced by EOR, about 2 million metric tons of CO2 could be stored per day.
The typical process for capturing CO2 is by contacting the gaseous stream with a solvent that absorbs the CO2. A number of solvents have been used. The CO2 is then desorbed as a concentrated gas and the solvent reused. This process is widely used for processing natural gas streams but much less used with gaseous streams from coal gasifiers or coal combustion units. The key concern is the degradation of the solvent by coal-derived impurities in the process gas. Other
processes are being considered and developed to reduce the cost and energy consumption required.
CO2 compression to about 125 atmospheres for transport and injection is straight forward but consumes a significant amount of energy. High-pressure compression is desirable because it reduces the volume of gas being pipelined, and the supercritical state facilitates injection and retention of the CO2 (IPCC, 2005).
Pipelining of CO2 is another conventional and proven step. The key concern is leakage of CO2 into the atmosphere. An asphyxiant denser than air, CO2 tends to stay close to the ground and is not easily dispersed. CO2 is fatal at high concentrations and detrimental to humans at lesser concentrations (Praxair, 2007). Thus, properly designed CCS facilities will include a CO2 monitoring system and a leak-prevention system.
Specially designed injection wells are required for CCS. Abandoned oil and gas wells will not be used for CO2 injection into spent oil and gas formations because these wells may not be capable of handling the acidic supercritical CO2, and they may not be properly cemented to ensure that CO2 does not leak into aquifers used for drinking water.
The cost of CO2 capture is $30-$40/metric ton of CO2 for a coal gasifier process stream, about $90/metric ton for a natural gas combined-cycle facility (because of a lower concentration of CO2 compared to coal gasification), and $70-$80/metric ton for coal-fired power facilities (IPCC, 2005). Adding in the cost of compression, pipelining, monitoring and injection into a suitable formation would increase the total cost by $30-40/metric ton (IPCC, 2005). For most CTL facilities, the cost of CO2 capture is included in the facility design and construction cost. However, additional costs are incurred for compression, pipelining, monitoring, injection, and storage. These costs are estimated at $40/metric ton of CO2 in the first-mover facilities (2035 timeframe) and $30/metric ton in facilities built later (2050 timeframe). In cases of CTL where the costs of capture are to be included, $80/metric ton of CO2 for 2035 and $70/metric ton of CO2 for 2050 are used.
CCS requires a large infrastructure—primarily the construction of pipelines to transport the CO2 from where it is captured to injection wells for storage underground. In the United States, potential reservoirs with a capacity for storing more than 100 years’ worth of injected CO2 are available within 100-150 miles of expected sources in most regions of the country (NACAP, 2012).
The cost of CCS is significant but probably not the major implementation barrier. The major barrier is the public acceptance of pipelines, injection wells, and storage of large amounts of carbon dioxide in subsurface formations (Court et al., 2012; de Best-Waldhober et al., 2012; Kraeusel and Moest, 2012), especially if these are near population centers. Leakage of stored CO2 is an issue that is still being investigated through research programs conducted by industry and DOE. Careful design and operation of CCS can likely prevent and mitigate any potential emissions of CO2, but gaining public acceptance is expected to be difficult given the large quantities of CO2 to be transported and stored. A single CTL facility producing 50,000 bbl/d of liquid fuels will require CO2 storage in the range of about 4 million to 9 million metric tons per year.
Finding: CCS is a key technology for meeting the study goals for GHG reductions by 2050. It will be very difficult to make large quantities of low-GHG hydrogen without CCS being widely available. Combining CCS with biofuel production would improve the chances of meeting the study goals.
3.9 RESOURCE NEEDS AND LIMITATIONS
Reducing petroleum consumption and GHG emissions from the LDV fleet will have a significant impact on energy resource use in the United States. Comparing existing resources with the estimated demands on resources for fueling the vehicles in representative scenarios in its analyses, the committee here draws conclusions about whether the projected demands on resources can be met.
Alternative LDV fuels can be produced from natural gas, coal, biomass, or other renewable energy sources, such as wind, solar, and hydro power. The U.S. consumption of natural gas, coal, and biomass in 2010 is shown in Table 3.21. Of the amounts consumed, 976 million tons of coal and 7.378 tcf of natural gas were used for electricity generation (EIA, 2011b). The biomass was used primarily for power in wood-processing plants, with some generated electricity going into the grid.
TABLE 3.21 U.S. Consumption of Natural Gas, Coal, and Biomass in 2010
| Consumption in Quads (higher heating value) | Amount Consumed | |
| Natural gas | 24.1 | 23.4 tcf |
| Coal | 22.1 | 1,050 million tons |
| Biomass | 4.30 | 269 million tons |
TABLE 3.22 Estimated Amount of Natural Gas Required to Fuel the Entire LDV Fleet via Different Fuel and Vehicle Technologies
| Natural Gas Required Annually for Different Vehicle-Fuel Combinations (tcf) | |||||||||
| Year | Vehicle Miles Traveled (trillion mi/yr) | ICE-CNG | ICE-drop-in | ICE-Methanol | HEV-CNG | Electric | FCEV | ||
| 2010 | 2.784 | 15.6 | 23.8 | 22.9 | 15.1 | 7.6 | 11.7 | ||
| 2030 | 3.727 | 10.1 | 15.5 | 14.9 | 8.4 | 7.5 | 7.3 | ||
| 2050 | 5.048 | 10.0 | 15.4 | 14.8 | 7.9 | 7.8 | 7.2 | ||
Biomass, coal, and natural gas can all be converted into “drop-in” liquid fuels by several routes (e.g., direct liquefaction of biomass or coal, and gasification followed by FT or MTG of all sources). These drop-in fuels will use the existing petroleum products distribution system and existing vehicles. The use of any of these alternative fuels would be transparent to the vehicle owner. The remaining alternative fuel and vehicle combinations include electricity in BEVs and PHEVs, hydrogen in FCEVs, and natural gas as a vehicle fuel, either directly as CNG or through conversion to methanol. All of these fuels can be produced from natural gas via mature technologies, and so a meaningful comparison would be to calculate the amount of natural gas that would be required to fuel the entire LDV fleet via the different fuel and vehicle technologies (Table 3.22). The vehicle efficiencies are assumed to be the mid-range efficiencies outlined in Chapter 2.
Direct use of CNG as a vehicle fuel is more resource efficient and less costly than conversion of natural gas to any liquid fuel. The advantages of conversion to a liquid fuel are the use of the current fuel infrastructure, the ease of onboard storage, and the familiarity of the driving population with liquid fuels. Conversion of natural gas to electricity or hydrogen as an energy carrier is currently more resource efficient than direct use of natural gas, but direct-use efficiency converges with that for PEVs and FCEVs by 2050 because of the differences in efficiency improvements with time. Both electricity and hydrogen carry additional socioeconomic burdens and infrastructure costs as discussed in previous sections. Electricity and hydrogen, as well as GTL and methanol, can be produced from other resources such as coal and biomass. Electricity and hydrogen can also be produced from nuclear, solar, and wind power.
There are two distinct goals for the scenarios evaluated by the committee: one goal targets only petroleum reduction, and the second goal targets reduction of GHG emissions. Both cases use the same vehicle and fuel technologies; however, in the low-GHG cases, the technology and fuels used to generate electricity and hydrogen were modified to reduce GHG emissions. The driving force for the low GHG grid case is discussed above in this chapter. Table 3.23 shows the impact of the low-GHG grid case on the mix of generating sources.
TABLE 3.23 Effect of the Low-Greenhouse Gas Grid on the Mix of Generating Sources
| Total Generation (billion kWh/yr) | |||||||||
| 2009 | 2050 Reference Grid | 2050 Low-GHG Grid | |||||||
| Coal without CCS | 1,693 | 2,368 | 238 | ||||||
| Coal with CCS | 0 | 15 | 17 | ||||||
| Petroleum and natural gas without CCS | 871 | 1,290 | 1,225 | ||||||
| Petroleum and natural gas with CCS | 0 | 0 | 489 | ||||||
| Nuclear | 795 | 855 | 1,255 | ||||||
| Hydroelectric | 274 | 314 | 323 | ||||||
| Biomass | 38 | 159 | 179 | ||||||
| Solar | 3 | 21 | 56 | ||||||
| Wind | 71 | 163 | 330 | ||||||
| Other | 34 | 66 | 66 | ||||||
The largest changes between the reference grid and the low-GHG grid are an almost 90 percent decline in coal usage, a doubling of natural gas, and a 50 percent increase in nuclear power. Total renewable electricity increases by over a factor of two and rises from 11 percent of total generation to 23 percent.
Table 3.24 shows the fuel usage and resource demands for 10 scenarios: five different vehicle mix scenarios, compounded with the reference grid and the low-GHG grid case and two different resource mixes for producing hydrogen.22 The implementation of these cases would be driven by various government policies. The reference case scenario is driven by existing and currently proposed policies for LDV CAFE standards and RFS2. The other cases stress increased biofuels, PEVs, FCEVs and CNGVs.
These scenarios have not been optimized to minimize costs, resource use, or GHG emissions. The reference scenario reduces petroleum use by 25 percent, and the others all meet or exceed the goal of an 80 percent reduction in petroleum use. GHG emission reductions are all similar for the reference-grid scenarios. Additional reductions in GHG emissions are possible for the electric and hydrogen cases
_______________________
22These scenarios are described in greater detail in Section 5.3.2.
with the use of a low-GHG grid and a change in the mix of resources used to generate hydrogen. Only the FCEV scenario meets the goal of reducing GHG emissions by 80 percent in 2050. The biofuel case can also meet the GHG emissions target if CCS is added to the biorefineries.
The resource demands can be met but involve some challenges. The largest changes are needed to achieve a low-GHG grid. These include an increased use of almost 7 tcf/yr of natural gas (a doubling of the current consumption for electricity), the construction of about fifty 1,000-MW nuclear power plants and about 100,000 new wind turbines and the capture and storage of more than 200 million metric tons/yr of CO2.
The most challenging related demands concern increased use of biomass and natural gas and public acceptance of the construction of a large number of nuclear power plants. As discussed above in this chapter, the demand for biomass is expected to be achievable and to be less than the biomass availability estimated in other recent analyses. Shipping and handling the mass and volume of biomass involved will be challenging. Natural gas demand doubles over the amount currently used to generate electricity. This increase represents essentially all of the additional natural gas expected to be available for use based on the most recent estimates of future gas availability in the United States.
There are important ancillary impacts from these resource demands on the associated infrastructure:
- Cleaning up the electric grid by 2050, as envisioned in 2011 AEO (EIA, 2011a), the basis for this discussion, will reduce current coal use by 85 percent or about 800 million tons per year, an amount that represents 44 percent of the total annual U.S. railroad freight tonnage. Shipments of biomass could mitigate that impact.
- Most petroleum products are currently shipped long distances by pipeline. Significant increases in hydrogen or electricity as an LDV fuel would idle a large fraction of the petroleum pipeline system.
- The large increase in natural gas consumption would require a significant expansion in natural gas pipelines. Use of hydrogen as an LDV fuel would require construction of an additional hydrogen pipeline system.
- CCS has to be economical and meet stringent performance requirements at large scale. CCS demonstrations at appropriate scale are needed to validate performance, safety, and costs.
Nearly 50 percent of U.S. petroleum refining output is currently used to fuel the LDV fleet. An 80 percent reduction in use of petroleum for LDVs will impact the availability and price of the refining byproducts that are used by other industries.
TABLE 3.24 Fuel Demands for Illustrative Scenarios and Resources Used
| Scenario | 2005 Actual | Reference | Biofuels | Electric | FCEV | CNG | |||
| Petroleum based fuels, billion gge/yr | 124.8 | 93.1 | 17.2 | 13.9 | 3.8 | 4.1 | |||
| GTL and CTL, billion gge/yr | 0 | 7.7 | 7.7 | 7.7 | 0.8 | 0.8 | |||
| Total biofuels, billion gge/yr | 4.9 | 24.1 | 55.9 | 24.1 | 19.2 | 19.1 | |||
| Electricity, billion gge/yr | 0 | 1.3 | 0 | 14.4 | 1.6 | 1.0 | |||
| Hydrogen, billion gge/yr | 0 | 0.5 | 0 | 1.1 | 33.5 | 0.5 | |||
| CNG, billion gge/yr | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 51.0 | |||
| Petroleum reduction, % | 25.4 | 86.2 | 88.9 | 97.0 | 96.7 | ||||
| Ethanol, % of liquid fuels | 5.6 | 11.9 | 17.5 | 30.9 | 30.7 | 33.9 | |||
| Resources Used to Power Vehicles, Reference Electric Grid | |||||||||
| Corn, million tons/yr | 81 | 165 | 165 | 165 | 84 | 99 | |||
| Other biomass, million tons/yr | 0 | 208 | 703 | 220 | 325 | 208 | |||
| Natural gas, billion cubic ft/yr | 18 | 1,021 | 888 | 1,915 | 3,038 | 6,969 | |||
| Coal, million tons/yr | 0 | 50 | 39 | 150 | 108 | 14 | |||
| Net GHG emissions reduction, % | — | 11 | 67 | 55 | 60 | 56 | |||
| Resources Used to Power Vehicles, Low GHG Electric Grid and Hydrogen Production | |||||||||
| Corn, million tons/yr | 81 | 165 | 165 | 165 | 84 | 99 | |||
| Other biomass, million tons/yr | 0 | 209 | 703 | 226 | 358 | 209 | |||
| Natural gas, billion cubic ft/yr | 18 | 1,105 | 890 | 2,613 | 4,664 | 7,039 | |||
| Coal, million tons/yr | 0 | 41 | 39 | 54 | 15 | 6 | |||
| Net GHG emissions reduction, % | — | 13 | 67 | 72 | 85 | 58 | |||
AAAS (American Association for the Advancement of Science). 2009. Coal-to-liquid technology. AAAS Policy Brief. Available at http://www.aaas.org/spp/cstc/briefs/coaltoliquid/. Accessed December 28, 2011.
Alvarez, R.A., S.W. Pacala, J.J. Winebrake, W.L. Chameides, and S.P. Hamburg. 2012. Greater focus needed on methane leakage from natural gas infrastructure. Proceedings of the National Academy of Sciences of the United States of America 109(17):6435-6440.
Anair, D., and A. Mahmassani. 2012. State of CHARGE. Electric Vehicles’ Global Warming Emissions and Fuel-Cost Savings across the United States. Cambridge, Mass.: Union of Concerned Scientists.
ANL (Argonne National Laboratory). 2011. GREET Model. The Greenhouse Gases, Regulated Emissions, and Energy Use in Transportation Model. Available at http://greet.es.anl.gov/. Accessed July 31, 2011.
Barcella, M.L., S. Gross, and S. Rajan. 2011. Mismeasuring Methane: Estimating Greenhouse Gas Emissions from Upstream Natural Gas Development. Englewood, Colo.: IHS CERA.
Begos, K. 2012. Electric Power Plants Shift from Coal to Natural Gas. Available at http://www.huffingtonpost.com/2012/01/16/electric-plants-coal-natural-gas_n_1208875.html. Accessed July 19, 2012.
BP. 2011. BP Energy Outlook 2030. London: BP.
Brooks, A. 2010. Separating wheat from the chaff of unconventionals. Musings from the Oil Patch. November 23.
Burnham, A., J. Han, C.E. Clark, M. Wang, J.B. Dunn, and I. Palou-Rivera. 2012. Life-cycle greenhouse gas emissions of shale gas, natural gas, coal, and petroleum. Environmental Science and Technology 46(2):619-627.
Campbell-Parnell. 2011. Why CNG? April 15, 2011. Available at http://www.usealtfuels.com/education_cng.htm.
Chevron. 2011. Gas-to-Liquids Transforming Natural Gas into Super-clean Fuels. Available at http://www.chevron.com/deliveringenergy/gastoliquids/. Accessed December 28, 2011.
China Shenhua Coal to Liquid and Chemical Co. Ltd. 2010. China Shenhua Direct Coal Liquefaction Demonstration Project. Available at http://www.csclc.com.cn/ens/cpyfw/youpin/2010-12-21/320.shtml. Accessed May 2, 2012.
Clean Vehicle Education Foundation. 2010. How Safe Are Natural Gas Vehicles? January 19, 2012. Acworth, Ga.: Clean Vehicle Education Foundation.
Court, B., T.R. Elliot, J. Dammel, T.A. Buscheck, J. Rohmer, and M.A. Celia. 2012. Promising synergies to address water, sequestration, legal, and public acceptance issues associated with large-scale implementation of CO2 sequestration. Mitigation and Adaptation Strategies for Global Change 17(6):569-599.
Cromie, R., and R. Graham. 2009. Transition to Electricity as the Fuel of Choice. Presentation to the NRC Committee on Assessment of Resource Needs for Fuel Cell and Hydrogen Technologies, May 18.
de Best-Waldhober, M., D. Daamen, A.R. Ramirez, A. Faaij, C. Hendriks, and E. de Visser. 2012. Informed public opinion in the Netherlands: Evaluation of CO2 capture and storage technologies in comparison with other CO2 mitigation options. International Journal of Greenhouse Gas Control 10:169-180.
DeMorro, C. 2010. GM Announces Hydrogen Infrastructure Pilot Project in Hawaii. Available at http://gas2.org/2010/05/12/gm-announces-hydrogen-infrastructure-pilot-project-in-hawaii/. Accessed June 27, 2012.
Diamond, R. 2010. Electric Vehicles: Their Potential and How to Achieve It. Presentation to the NRC Committee on the Potential for Light-Duty Vehicle (LDV) Technologies, 2010-2050: Costs, Impacts, Barriers and Timing for Review. December 14.
DiPietro, D. 2010. Life Cycle Analysis of Coal and Natural Gas-Fired Power Plants. Available at http://www.netl.doe.gov/energy-analyses/pubs/LCA_coal&NG_plants.pdf.
DKRW Advanced Fuels LLC. 2011. DKRW Advanced Fuels Signs Major Gasoline Contract: Moving Forward on Medicine Bow Project in Wyoming. Available at http://www.dkrwadvancedfuels.com/_filelib/FileCabinet/PDFs/Press_Releases/PR_News_Release_Proof_(2).pdf. Accessed June 26, 2012.
DOE (U.S. Department of Energy). 2011. U.S. Billion-Ton Update. Washington, D.C.: U.S. Department of Energy.
DOE-EERE (U.S. Department of Energy-Energy Efficiency and Renewable Energy). 2011a. 2010 Fuel Cell Technologies Market Report. Washington, D.C.: U.S. Department of Energy-Energy Efficiency and Renewable Energy.
———. 2011b. Clean Cities. Alternative Fuel Price Report. April. Washington, D.C.: U.S. Department of Energy.
———. 2011c. Clean Cities. Alternative Fuel Price Report. July. Washington, D.C.: U.S. Department of Energy.
———. 2011d. Clean Cities. Alternative Fuel Price Report. October. Washington, D.C.: U.S. Department of Energy.
———. 2012a. Alternative Fueling Station Total Counts by State and Fuel Type. Available at http://www.afdc.energy.gov/afdc/fuels/stations_counts.html?print. Accessed January 19, 2012.
———. 2012b. Clean Cities. Alternative Fuel Price Report. January. Washington, D.C.: U.S. Department of Energy.
———. 2012c. Flexible Fuel Vehicle Availability. Available at http://www.afdc.energy.gov/afdc/vehicles/flexible_fuel_availability.html. Accessed April 25, 2012.
Dooley, J.J., R.T. Dahowski, and C.L. Davidson. 2001. Comparing existing pipeline networks with the potential scale of future U.S. CO2 pipeline networks. Energy Procedia 1(1):1595-1602.
EIA (Energy Information Administration). 2008a. Estimated Number of Compressed Natural Gas (CNG) Vehicles in Use, by State and User Group. Available at http://www.eia.gov/cneaf/alternate/page/atftables/attf_v9.html. Accessed January 19, 2012.
———. 2008b. The Impact of Increased Use of Hydrogen on Petroleum Consumption and Carbon Dioxide Emissions. SR-OIAF-CNEAF/2008-04. Washington, D.C.: U.S. Department of Energy.
———. 2011a. Annual Energy Outlook 2011 with Projections to 2035. Washington, D.C.: U.S. Department of Energy.
———. 2011b. Annual Energy Review 2010. Washington, D.C.: U.S. Department of Energy.
———. 2012a. AEO 2012 Early Release Overview. Washington, D.C.: U.S. Department of Energy.
———. 2012b. Biodiesel Overview. Available at http://www.eia.gov/totalenergy/data/monthly/pdf/sec10_8.pdf. Accessed July 19, 2012.
———. 2012c. Fuel Ethanol Overview. Available at http://www.eia.gov/totalenergy/data/monthly/pdf/sec10_7.pdf. Accessed July 19, 2012.
———. 2009. Residential Energy Consumption Survey. Available at http://www.eia.gov/consumption/residential/data/2009/. Accessed July 19, 2012.
Electrification Coalition. 2009. Policy Report Fleet Electrification Roadmap. Available at http://www.electrificationcoalition.org/sites/default/files/EC-Fleet-Roadmap-screen.pdf.
Elgowainy, A., A. Burnham, M. Wang, J. Molburg, and A. Rousseau. 2009. Well to Wheels Energy Use and Greenhouse Gas Emissions of Plug In Hybrid Electric Vehicles. ANL/ESD/09-2. Chicago, Ill.: Argonne National Laboratory.
EPA (U.S. Environmental Protection Agency). 2010a. Greenhouse Gas Emissions Reporting from the Petroleum and Natural Gas Industry. Washington, D.C.: U.S. Environmental Protection Agency.
———. 2010b. Renewable Fuel Standard Program (RFS2) Regulatory Impact Analysis. US EPA 420-R-10-006. Washington, D.C.: U.S. Environmental Protection Agency.
———. 2011a. Coal Ash. Available at http://www.epa.gov/radiation/tenorm/coalandcoalash.html. Accessed December 28, 2011.
———. 2011b. Mid-Atlantic Mountaintop Mining. Available at http://www.epa.gov/region3/mtntop/. Accessed December 28, 2011.
———. 2011c. Oil and natural gas sector: New source performance standards and national emission standards for hazardous air pollutants reviews. Proposed rule. Federal Register 76(163):52738-52843.
———. 2012a. Alphabetical List of Registered E15 Ethanols. Available at http://www.epa.gov/otaq/regs/fuels/additive/web-e15.htm. Accessed April 25, 2012.
———. 2012b. Oil and natural gas sector: New source performance standards and national emission standards for hazardous air pollutants reviews. Final rule. Federal Register 77(159):49490-49600.
EPRI and NRDC (Electric Power Research Institute and Natural Resources Defense Council). 2007. Environmental Assessment of Plug-In Hybrid Electric Vehicles. Volume 1: Nationwide Greenhouse Gas Emissions. Available at http://my.epri.com/portal/server.pt?open=514&objID=223132&mode=2. Accessed July 31, 2012.
ExxonMobil. 2009. Methanol to Gasoline (MTG): Production of Clean Gasoline from Coal. Fairfax, Va.: ExxonMobil Research and Engineering.
Farrell, A.E., R.J. Plevin, B.T. Turner, A.D. Jones, M. O’Hare, and D.M. Kammen. 2006. Ethanol can contribute to energy and environmental goals. Science 311(5760):506-508.
Finley, R. 2005. An Assessment of Geological Carbon Sequestration Options in the Illinois Basin. Final Report. Champaign, Ill.: Midwest Geological Sequestration Consortium.
Guo, J.Y., G. Venkataramanan, B. Lesieutre, A. Smick, M. Mallette, and C. Getter. 2010. Consumer Adoption and Grid Impact Models for Plug-in Hybrid Electric Vehicles in Dane County, Wisconsin. Part A: Consumer Adoption Models. Madison, Wisc.: Public Service Commission of Wisconsin.
Haldor Topsoe. 2011. Autothermal Reforming. Available at http://www.topsoe.com/business_areas/methanol/Processes/AutothermalReforming.aspx. Accessed December 28, 2011.
Hertel, T.W., A.A. Golub, A.D. Jones, M. O’Hare, R.J. Plevin, and D.M. Kammen. 2010. Effects of US maize ethanol on global land use and greenhouse gas emissions: Estimating market-mediated responses. BioScience 60(3):223-231.
Hill, J., E. Nelson, D. Tilman, S. Polasky, and D. Tiffany. 2006. Environmental, economic, and energetic costs and benefits of biodiesel and ethanol biofuels. Proceedings of the National Academy of Sciences of the United States 103(30):11206-11210.
Howarth, R.W., R. Santoro, and A. Ingraffea. 2011. Methane and greenhouse-gas footprint of natural gas from shale formations. Climatic Change, doi:10.1007/s10584-011-0061-5.
IEA (International Energy Agency). 2007. Hydrogen Production and Distribution. Paris: International Energy Agency.
IHS. 2010. Natural Gas for Transportation: Niche Market or More? Englewood, Colo.: IHS.
IPCC (Intergovernmental Panel on Climate Change). 2005. Carbon Capture and Storage. Geneva: Intergovernmental Panel on Climate Change.
Jiang, M., W.M. Griffin, C. Hendrickson, P. Jaramillo, J. VanBriesen, and A. Venkatesh. 2011. Life cycle greenhouse gas emissions of Marcellus shale gas. Environmental Research Letters 6(3).
KBR. 2011. Coal Gasification. Available at http://www.kbr.com/Technologies/Coal-Gasification/. Accessed December 28, 2011.
Kingston, J. 2011. Pearl’s Gas-to-Liquids Project Rolls Out, Taking ITS Cues from Many World Markets. Available at http://www.platts.com/weblog/oilblog/2011/06/17/pearls_gas-to-l.html. Accessed December 28, 2011.
Kraeusel, J., and D. Moest. 2012. Carbon capture and storage on its way to large-scale deployment: Social acceptance and willingness to pay in Germany. Energy Policy 49:642-651.
Kuuskaraa, V.A., T. Van Leeuwen, and M. Wallace. 2011. Improving Domestic Energy Security and Lowering CO2 Emissions with Next Generation CO2-Enhanced Oil Recovery (CO2-EOR). Pittsburgh, Pa.: National Energy Technology Laboratory.
Law, K., M. Chan, W. Bockholt, and M.D. Jackson. 2010. US and Canadian Natural Gas Vehicle Market Analysis. Cupertino, Calif.: TIAX, LLC.
LLNL (Lawrence Livermore National Laboratory). 2012. Energy Flow. Available at https://flowcharts.llnl.gov/. Accessed January 19, 2012.
McAllister, E. 2012. AAA Calls for Suspension of E15 Gasoline Sales. Available at http://www.reuters.com/article/2012/11/30/us-usa-gasoline-idUSBRE8AT0JF20121130. Accessed December 3, 2012.
Melzer, L.S. 2012. Carbon Dioxide Enhanced Oil Recovery (CO2 EOR): Factors Involved in Adding Carbon Capture, Utilization and Storage (CCUS) to Enhanced Oil Recovery. Midland, Texas: Melzer Consulting.
Michael, K., A. Golab, V. Shulakova, J. Ennis-King, G. Allinson, S. Sharma, and T. Aiken. 2010. Geological storage of CO2 in saline aquifers—A review of the experience from existing storage operations. International Journal of Greenhouse Gas Control 4(4):659-667.
Milhench, C., and I. Kurahone. 2011. Energy E&P Spending to Reach Record High. Available at http://www.reuters.com/article/2011/12/05/us-barclays-capex-idUSTRE7B428A20111205. Accessed April 25, 2012.
MIT (Massachusetts Institute of Technology). 2011. The Future of Natural Gas. Cambridge, Mass.: Massachusetts Institute of Technology.
Mullins, K.A., W.M. Griffin, and H.S. Matthews. 2010. Policy implications of uncertainty in modeled life-cycle greenhouse gas emissions of biofuels. Environmental Science and Technology 45(1):132-138.
NACAP (North American Carbon Atlas Partnership). 2012. The North American Carbon Storage Atlas. Available at http://www.netl.doe.gov/technologies/carbon_seq/refshelf/NACSA2012.pdf. Accessed July 31, 2012.
NAS-NAE-NRC (National Academy of Sciences, National Academy of Engineering, National Research Council). 2009a. America’s Energy Future: Technology and Transformation. Washington, D.C.: The National Academies Press.
———. 2009b. Liquid Transportation Fuels from Coal and Biomass: Technological Status, Costs, and Environmental Impacts. Washington, D.C.: The National Academies Press.
NaturalGas.org. 2011. Background. Available at http://www.naturalgas.org/overview/background.asp. Accessed July 31, 2011.
———. 2012. Electric Generation Using Natural Gas. Available at http://naturalgas.org/overview/uses_eletrical.asp. Accessed July 19, 2012.
NBB (National Biodiesel Board). 2010. NBB Member Plant Locations. Available at http://www.biodiesel.org/buyingbiodiesel/plants/default.aspx?AspxAutoDetectCookieSupport=1. Accessed November 17, 2010.
NETL (National Energy Technology Laboratory). 2007. Carbon Sequestration Technology and Program Plan. Pittsburgh, Pa.: National Energy Technology Laboratory.
———. 2011. 2010 Carbon Sequestration Atlas of the United States and Canada. Pittsburgh, Pa.: National Energy Technology Laboratory.
NMA (National Mining Association). 2005. Liquid Fuels from U.S. Coal. Washington, D.C.: National Mining Association.
NPC (National Petroleum Council). 2007. Coal to Liquids and Gas. Washington, D.C.: National Petroleum Council.
NRC (National Research Council). 2008. Transitions to Alternative Transportation Technologies: A Focus on Hydrogen. Washington, D.C.: The National Academies Press.
———. 2011. Renewable Fuel Standard: Potential Economic and Environmental Effects of U.S. Biofuel Policy. Washington, D.C.: The National Academies Press.
Oil and Gas Journal. 2011. Deloitte: MENA NOCs to Invest $140 Billion in 2011. Available at http://www.ogj.com/content/ogj/en/articles/print/volume-109/issue-45/general-interest/deloittemena-nocs-to-invest.html. Accessed June 16, 2012.
Peterson, R., and P. Tijm. 2008. An Alaska North Slope GTL Option. Anchorage, Alaska: Alaska Natural Resources to Liquids, LLC.
PNNL (Pacific Northwest National Laboratory). 2007. Impacts Assessment of Plug-In Hybrid Vehicles on Electric Utilities and Regional U.S. Power Grids. Part 1: Technical Analysis. Available at http://www.ferc.gov/about/com-mem/wellinghoff/5-24-07-technical-analy-wellinghoff.pdf. Accessed July 31, 2012.
Potential Gas Committee. 2009. Potential Supply of Natural Gas in the United States (December 31, 2008). Golden, Colo.: Colorado School of Mines.
Praxair. 2007. Product: Carbon Dioxide. Praxair Material Safety Data Sheet. Available at http://www.praxair.com/praxair.nsf/AllContent/D3CBF4EF4186073485256A860080D221/$File/p4574j.pdf. Accessed May 2, 2012.
Preston, C., M. Monea, W. Jazrawi, K. Brown, S. Whittaker, D. White, D. Law, R. Chalaturnyk, and B. Rostron. 2005. IEA GHG Weyburn CO2 monitoring and storage project. Fuel Processing Technology 86(14-15):1547-1568.
Preston, C., S. Whittaker, B. Rostron, R. Chalaturnyk, D. White, C. Hawkes, J.W. Johnson, A. Wilkinson, and N. Sacuta. 2009. IEA GHG Weyburn-Midale CO2 monitoring and storage project—moving forward with the final phase. Energy Procedia 1(1):1743-1750.
Raman, V. 2004. Hydrogen production and supply infrastructure for transportation—Discussion paper. In The 10-50 Solution: Technologies and Policies for a Low-Carbon Future. Washington, D.C.: Pew Center on Global Climate Change and the National Commission on Energy Policy.
Reuters. 2011. China Shenhua Coal-to-Liquids Project Profitable-Exec. Available at http://www.reuters.com/article/2011/09/08/shenhua-oil-coal-idUSL3E7K732020110908. Accessed May 2, 2012.
Simmons & Company. 2011. Contemplating Long-term Natural Gas Demand Opportunities. Houston, Texas: Simmons & Company.
Statoil. 2010. Leading the World in Carbon Capture and Storage. Available at http://www.statoil.com/en/TechnologyInnovation/NewEnergy/CO2MANAGEMENT/pages/carboncapture.aspx. Accessed December 28, 2011.
Tabak, S. 2006. ExxonMobil methanol to gasoline. Commercially proven route for production of synthetic gasoline. In The U.S.-China Oil and Gas Industry Forum. Hangzhou City, China.
Takemasa, R. 2011. Electric Utility Preparations for Electric Vehicles. Presentation to the NRC Committee on the Potential for Light-Duty Vehicle (LDV) Technologies, 2010-2050: Costs, Impacts, Barriers and Timing, June 28.
Technology Review. 2009. Drilling for Shale Gas. Available at http://www.technologyreview.com/video/?vid=439. Accessed January 19, 2012.
Wald, M.L. 2012. In Kansas, Stronger Mix of Ethanol. The New York Times. July 11. Available at http://www.nytimes.com/2012/07/12/business/energy-environment/at-kansas-station-e15-fuel-reaches-the-masses.html?_r=2. Accessed July 31, 2012.
Wright, M.M., J.A. Satrio, R.C. Brown, D.E. Saugaard, and D.D. Hsu. 2010. Techno-Economic Analysis of Biomass Fast Pyrolysis to Transportation Fuels. Golden, Colo.: National Renewable Energy Laboratory.
Zhao, X., R.D. McGihon, and S.A. Tabak. 2008. Coal to clean gasoline. Hydrocarbon Engineering. September.



































