2
Factors in Emergence
Emerging infectious diseases are clinically distinct conditions whose incidence in humans has increased. For the purposes of this study, the committee has focused on diseases that have emerged in the United States within the past two decades. Emergence may be due to the introduction of a new agent, to the recognition of an existing disease that has gone undetected, or to a change in the environment that provides an epidemiologic "bridge." (For an example of an emerging disease, see Box 2-1.) Emergence, or, more specifically, reemergence, may also be used to describe the reappearance of a known disease after a decline in incidence. Although an infectious agent plays a role in any emerging infectious disease, other causative factors may be important as well.
|
BOX 2-1 A Deadly Form of Strep It was a shock to many when renowned puppeteer Jim Henson died suddenly in May 1990. How could a healthy man in his early 50s be so easily felled by a case of pneumonia? Since his death, attention has focused on a deadly "new" form of streptococcal bacteria. This new bacterium belongs to a category of strep bacteria called "Group A," a subset of organisms familiar to many as the cause of acute pharyngitis (strep throat). The new strep A has been killing otherwise healthy people (like Henson), and doing so in a frighteningly rapid fashion. This was true for a 30-year-old Canadian man who got a splinter in his finger, which later became infected. Within six days he had become so ill that he was admitted to an intensive care unit and placed on a respirator. He died six weeks later of sepsis (disseminated infection) (Goldman, 1991). |
|
The new strep A bacteria, like all streptococcal organisms, are typically inhaled, but they can also enter the body through a cut or scrape. The infection they provoke once inside the body is especially insidious: its early symptoms are easily mistaken for signs of the flu. In several cases, the bacteria have overwhelmed their host with pneumonia, and in others, with kidney and liver damage before the infected person has sought treatment. So advanced, the infection is extremely difficult to treat. Even if massive doses of penicillin succeed in killing the bacteria, there are no means available to counter the effects of the deadly toxin they produce—which actually causes the pneumonia and tissue damage. Although reports of the first cases of fatal infection with the new strep A appeared in the medical literature in 1989 (Stevens et al., 1989), health problems associated with the streptococcus family of bacteria are not new. In the days before antibiotics, they were responsible for widespread outbreaks of scarlet fever and rheumatic fever. Nor are these bacteria rare. Strep throat is so common an ailment among children that it could almost be considered a rite of passage. Much about the new strep A remains a mystery. Some scientists—noting the similarity between the toxin secreted by the new strep A and the toxin once seen with scarlet fever—believe that this bacterium is an old microbe making a comeback. Others consider this highly virulent form of strep the result of a recent bacterial mutation. Whatever its origin, the new strep A deserves attention. Experts strongly encourage people to seek immediate medical care if they become very ill (high fever, sore throat) in a sudden fashion, especially if they have recently suffered a cut or burn. Although cases of infection with this new, deadly microbe remain quite rare, their increasing incidence in the past two years is cause for concern. A vaccine for streptococcal infections is in development, but researchers estimate that it will not be ready for public use for at least another three years. In the meantime, the Centers for Disease Control is working to track the new strep A more closely, with the hope of learning more about the bacterium and how to stop it. |
Table 2-1 is a list of emerging infectious agents, categorized by type of organism. Appendix B provides more detailed information on each of these agents. The committee recognizes that this list is continually expanding, mainly as a result of the growing numbers of immunocompromised individuals. Therefore, it may not contain all organisms that fit the definition above.
Once a new pathogen has been introduced into a human population, its ability to spread becomes a critical factor in emergence. The same is true for agents that are already present in a limited or isolated human population:
TABLE 2-1 Part 1: Examples of Emergent Bacteria, Rickettsiae, and Chlamydiae
|
Agent |
Related Diseases/Symptoms |
Mode of Transmission |
Cause(s) of Emergence |
|
Aeromonas species |
Aeromonad gastroenteritis, cellulitis, wound infection, septicemia |
Ingestion of contaminated water or food; entry of organism through a break in the skin |
Immunosuppression; improved technology for detection and differentiation |
|
Borrelia burgdorferi |
Lyme disease: rash, fever, neurologic and cardiac abnormalities, arthritis |
Bite of infective Ixodes tick |
Increase in deer and human populations in wooded areas |
|
Campylobacter jejuni |
Campylobacter enteritis: abdominal pain, diarrhea, fever |
Ingestion of contaminated food, water, or milk; fecal-oral spread from infected person or animal |
Increased recognition; consumption of uncooked poultry |
|
Chlamydia pneumoniae (TWAR strain) |
TWAR infection: fever, myalgias, cough, sore throat, pneumonia |
Inhalation of infective organisms; possibly by direct contact with secretions of an infected person |
Increased recognition |
|
Chlamydia trachomatis |
Trachoma, genital infections, conjunctivitis; infection during pregnancy can result in infant pneumonia |
Sexual intercourse |
Increased sexual activity; changes in sanitation |
|
Clostridium difficile |
Colitis: abdominal pain, watery diarrhea, bloody diarrhea |
Fecal-oral transmission; contact with the organism in the environment |
Increased recognition; immunosuppression |
|
Ehrlichia chaffeensis |
Ehrlichiosis: febrile illness (fever, headache, nausea, vomiting, myalgia) |
Unknown; tick is suspected vector |
Increased recognition; possibly increase in host and vector populations |
|
Escherichia coli O157:H7 |
Hemorrhagic colitis; thrombocytopenia; hemolytic uremic syndrome |
Ingestion of contaminated food, esp. undercooked beef and raw milk |
Likely due to the development of a new pathogen |
|
Haemophilus influenzae biogroup aegyptius |
Brazilian purpuric fever: purulent conjunctivitis, high fever, vomiting, and purpura |
Contact with discharges of infected persons; eye flies are suspected vectors |
Possibly an increase in virulence due to mutation |
|
Agent |
Related Diseases/Symptoms |
Mode of Transmission |
Cause(s) of Emergence |
|
Helicobacter pylori |
Gastritis, peptic ulcer, possibly stomach cancer |
Ingestion of contaminated food or water, esp. unpasteurized milk; contact with infected pets |
Increased recognition |
|
Legionella pneumophila |
Legionnaires' disease: malaise, myalgia, fever, headache, respiratory illness |
Air-cooling systems, water supplies |
Recognition in an epidemic situation |
|
Listeria monocytogenes |
Listeriosis: meningoencephalitis and/or septicemia |
Ingestion of contaminated foods; contact with soil contaminated with infected animal feces; inhalation of organism |
Probably increased awareness, recognition, and reporting |
|
Mycobacterium tuberculosis |
Tuberculosis: cough, weight loss, lung lesions; infection can spread to other organ systems |
Exposure to sputum droplets (exhaled through a cough or sneeze) of a person with active disease |
Immunosuppression |
|
Staphylococcus aureus |
Abscesses, pneumonia, endocarditis, toxic shock |
Contact with the organism in a purulent lesion or on the hands |
Recognition in an epidemic situation; possibly mutation |
|
Streptococcus pyogenes (Group A) |
Scarlet fever, rheumatic fever, toxic shock |
Direct contact with infected persons or carriers; sometimes ingestion of contaminated foods |
Change in virulence of the bacteria; possibly mutation |
|
Vibrio cholerae |
Cholera: severe diarrhea, rapid dehydration |
Ingestion of water contaminated with the feces of infected persons; ingestion of food exposed to contaminated water |
Poor sanitation/hygiene; possibly introduced via bilge-water from cargo ships |
|
Vibrio vulnificus |
Cellulitis; fatal bacteremia; diarrheal illness (occasionally) |
Contact of superficial wounds with seawater or with contaminated (raw or undercooked) seafood; ingestion (occasionally) |
Increased recognition |
TABLE 2-1 Part 2: Examples of Emergent Viruses
|
Agent |
Related Diseases/Symptoms |
Mode of Transmission |
Cause(s) of Emergence |
|
Bovine spongiform encephalopathy (BSE) agent |
Bovine spongiform encephalopathy in cows |
Ingestion of feed containing infected sheep tissue |
Changes in the rendering process |
|
Chikungunya |
Fever, arthritis, hemorrhagic fever |
Bite of infected mosquito |
Unknown |
|
Crimean-Congo hemorrhagic fever |
Hemorrhagic fever |
Bite of an infected adult tick |
Ecological changes favoring increased human exposure to ticks on sheep and small wild animals |
|
Dengue |
Hemorrhagic fever |
Bite of an infected mosquito (primarily Aedes aegypti) |
Poor mosquito control; increased urbanization in tropics; increased air travel |
|
Filoviruses (Marburg, Ebola) |
Fulminant, high-mortality hemorrhagic fever |
Direct contact with infected blood, organs, secretions, and semen |
Unknown; in Europe and the United States, virus-infected monkeys shipped from developing countries via air |
|
Hantaviruses |
Abdominal pain, vomiting, hemorrhagic fever |
Inhalation of aerosolized rodent urine and feces |
Human invasion of virus ecologic niche |
|
Hepatitis B |
Nausea, vomiting, jaundice; chronic infection leads to hepatocellular carcinoma and cirrhosis |
Contact with saliva, semen, blood, or vaginal fluids of an infected person; mode of transmission to children not known |
Probably increased sexual activity and intravenous drug abuse; transfusion (before 1978) |
|
Hepatitis C |
Nausea, vomiting, jaundice; chronic infection leads to hepatocellular carcinoma and cirrhosis |
Exposure (percutaneous) to contaminated blood or plasma; sexual transmission |
Recognition through molecular virology applications; blood transfusion practices following World War II (esp. in Japan) |
|
Hepatitis E |
Fever, abdominal pain, jaundice |
Contaminated water |
Newly recognized |
|
Human herpesvirus 6 (HHV-6) |
Roseola in children, syndrome resembling mononucleosis |
Unknown; possibly respiratory spread |
Newly recognized |
|
Agent |
Related Diseases/Symptoms |
Mode of Transmission |
Cause(s) of Emergence |
|
Human immunodeficiency viruses |
|||
|
HIV-1 |
HIV disease, including AIDS: severe immune system dysfunction, opportunistic infections |
Sexual contact with or exposure to blood or tissues of an infected person; vertical transmission |
Urbanization; changes in lifestyles/mores; increased intravenous drug use; international travel; medical technology (transfusions/transplants) |
|
HIV-2 |
Similar to above |
Same as above |
Same as above, esp. international travel |
|
Human papillomavirus |
Skin and mucous membrane lesions (often, warts); strongly linked to cancer of the cervix and penis |
Direct contact (sexual contact/contact with contaminated surfaces) |
Newly recognized; perhaps changes in sexual lifestyle |
|
Human parvovirus B19 |
Erythema infectiosum: erythema on face, rash on trunk; aplastic anemia |
Contact with respiratory secretions of an infected person; vertical transmission |
Newly recognized |
|
Human T-cell lymphotropic viruses (HTLV-I and HTLV-II) |
Leukemias and lymphomas |
Vertical transmission through blood/breast milk; exposure to contaminated blood products; sexual transmission |
Increased intravenous drug abuse; medical technology (transfusion) |
|
Influenza |
|||
|
Pandemic |
Fever, headache, cough, pneumonia |
Airborne (esp. in crowded, enclosed spaces) |
Animal-human virus reassortment; antigenic shift |
|
Drift |
Same as above |
Same as above |
Antigenic drift |
|
Japanese encephalitis |
Encephalitis |
Bite of an infective mosquito |
Changing agricultural practices |
|
La Crosse and California Group viruses |
Encephalitis |
Bite of an infective mosquito |
Increasing interface between human activity and endemic areas; discarded tires as mosquito breeding sites |
|
Agent |
Related Diseases/Symptoms |
Mode of Transmission |
Cause(s) of Emergence |
|
Lassa |
Fever, headache, sore throat, nausea |
Contact with urine or feces of infected rodents |
Urbanization/conditions favoring infestation by rodents |
|
Measles |
Fever, conjunctivitis, cough, red blotchy rash |
Airborne; direct contact with respiratory secretions of infected persons |
Deterioration of public health infrastructure supporting immunization |
|
Norwalk and Norwalk-like agents |
Gastroenteritis; epidemic diarrhea |
Most likely fecal-oral; alleged vehicles of transmission include drinking and swimming water, and uncooked foods |
Increased recognition |
|
Rabies |
Acute viral encephalomyelitis |
Bite of a rabid animal |
Introduction of infected reservoir host to new areas |
|
Rift Valley |
Febrile illness |
Bite of an infective mosquito |
Importation of infected mosquitoes and/or animals; development (dams, irrigation) |
|
Ross River |
Arthritis, rash |
Bite of an infective mosquito |
Movement of infected mosquitoes or people |
|
Rotavirus |
Enteritis; diarrhea, vomiting, dehydration, and low-grade fever |
Primarily fecal-oral; fecal-respiratory transmission can also occur |
Increased recognition |
|
Venezuelan equine encephalitis |
Encephalitis |
Bite of an infective mosquito |
Movement of mosquitoes and amplification hosts (horses) |
|
Yellow fever |
Fever, headache, muscle pain, nausea, vomiting |
Bite of an infective (Aedes aegypti) mosquito |
Lack of effective mosquito control and widespread vaccination; urbanization in tropics; increased air travel |
TABLE 2-1 Part 3: Examples of Emergent Protozoans, Helminths, and Fungi
|
Agent |
Related Diseases/Symptoms |
Mode of Transmission |
Cause(s) of Emergence |
|
Anisakis |
Anisakiasis: abdominal pain, vomiting |
Ingestion of larvae-infected fish (undercooked) |
Changes in dietary habits (eating of raw fish) |
|
Agent |
Related Diseases/Symptoms |
Mode of Transmission |
Cause(s) of Emergence |
|
Babesia |
Babesiosis: fever, fatigue, hemolytic anemia |
Bite of an Ixodes tick (carried by mice in the presence of deer) |
Reforestation; increase in deer population; changes in outdoor recreational activity |
|
Candida |
Candidiasis: fungal infections of the gastrointestinal tract, vagina, and oral cavity |
Endogenous flora; contact with secretions or excretions from infected persons |
Immunosuppression; medical management (catheters); antibiotic use |
|
Cryptococcus |
Meningitis; sometimes infections of the lungs, kidneys, prostate, liver |
Inhalation |
Immunosuppression |
|
Cryptosporidium |
Cryptosporidiosis: infection of epithelial cells in the gastrointestinal and respiratory tracts |
Fecal-oral, person-to-person, waterborne |
Development near watershed areas; immunosuppression |
|
Giardia lamblia |
Giardiasis: infection of the upper small intestine, diarrhea, bloating |
Ingestion of fecally contaminated food or water |
Inadequate control in some water supply systems; immunosuppression; international travel |
|
Microsporidia |
Gastrointestinal illness, diarrhea; wasting in immunosuppressed persons |
Unknown; probably ingestion of fecally contaminated food or water |
Immunosuppression; recognition |
|
Plasmodium |
Malaria |
Bite of an infective Anopheles mosquito |
Urbanization; changing parasite biology; environmental changes; drug resistance; air travel |
|
Pneumocystis carinii |
Acute pneumonia |
Unknown; possibly reactivation of latent infection |
Immunosuppression |
|
Strongyloides stercoralis |
Strongyloidiasis: rash and cough followed by diarrhea; wasting, pulmonary involvement, and death in immunosuppressed persons |
Penetration of skin or mucous membrane by larvae (usually from fecally-contaminated soil); oral-anal sexual activities |
Immunosuppression; international travel |
|
Toxoplasma gondii |
Toxoplasmosis: fever, lymphadenopathy, lymphocytosis |
Exposure to feces of cats carrying the protozoan; sometimes foodborne |
Immunosuppression; increase in cats as pets |
those agents best adapted to human transmission are likely to be those that will emerge. Introduction of a disease-causing agent into a new host population and dissemination of the agent within the new host species can occur almost simultaneously, but they are more commonly separated by considerable periods of time. Changes in the environment and in human behavior, as well as other factors, may increase the chances that dissemination will occur.
For familiar, "old" agents, whose spread has been successfully controlled, reemergence is often the result of lapses in public health measures owing to complacency, changes in human behavior that increase person-to-person transmission of an infectious agent, or changes in the ways humans interact with their environment. The return of dengue fever into areas of South and Central America where previously Ae. aegypti had been eradicated and the resurgence of yellow fever in Nigeria, where more than 400 persons were estimated to have died between April 1 and July 14, 1991 (Centers for Disease Control, unpublished data, 1992), reflect the operation of these mechanisms.
THE CONCEPT OF EMERGENCE
Although specific agents are usually associated with individual diseases, historically it is the diseases that usually have been recognized first. With improved techniques for the identification of microbes, however, this situation is changing. The causative agents for many newly emergent diseases are often discovered virtually simultaneously with (or in some cases before) their associated disease syndromes. For this reason, the term emerging microbial threat as used in this report includes both the agent and the disease.
It is important to understand the difference between infection and disease. Infection implies that an agent, such as a virus, has taken up residence in a host and is multiplying within that host—perhaps with no outward signs of disease. Thus, it is possible to be infected with an agent but not have the disease commonly associated with that agent (although disease may develop at a later time).
In discussions about the emergence of "new" diseases, considerable debate has centered on the relative importance of de novo evolution of agents versus the transfer of existing agents to new host populations (so-called microbial traffic). It is sometimes presumed that the appearance of a novel, disease-causing microorganism results from a change in its genetic properties. This is sometimes the case, but there are many instances in which emergence is due to changes in the environment or in human ecology. In fact, environmental changes probably account for most emerging diseases.
For example, despite the fact that many viruses have naturally high rates of mutation, the significance of new variants as a source of new viral
diseases has been hard to demonstrate, and there appear to be relatively few documented examples in nature. Influenza is probably the best example of a virus for which the importance of new variants (i.e., antigenic drift) can clearly be shown. Variants of the hepatitis B virus also have been shown recently to cause disease. However, cases like these are greatly outnumbered by instances of new diseases or outbreaks resulting from microbial traffic between species. Cross-species transfer of infectious agents is often the result of human activities.
The evolution of viruses is constrained by their requirement for being maintained in a host. It would therefore seem that new variants of nonviral pathogens, such as bacteria, would be more common than new forms of viral pathogens since nonviral organisms are less constrained by host requirements. However, most nonviral pathogens usually show a clonal origin (Selander and Musser, 1990; Musser et al., 1991; Tibayrenc et al., 1991a,b). That is, they appear to be derived from a single ancestor, suggesting that the evolution of a successful new pathogen is a relatively rare event. When it does occur, the new microbe probably originates in a single geographic area and is disseminated through channels of microbial traffic. One implication of this model is that the control of ''new" diseases may be more likely if the new variant is identified early (e.g., by worldwide infectious disease surveillance) and steps are taken to prevent its further dissemination.
It is likely that emerging pathogens generally are not newly evolved. Rather, it appears that they already exist in nature. Some may have existed in isolated human populations for some time; others, including many of the most novel, are well established in animals. Infections in animals that are transmissable to humans are termed zoonoses. As discussed in Chapter 1, throughout history rodents have been particularly important natural reservoirs of many infectious diseases.
The significance of zoonoses in the emergence of human infections cannot be overstated. The introduction of viruses into human populations, for example, is often the result of human activities, such as agriculture, that cause changes in natural environments. These changes may place humans in contact with infected animals or with arthropod vectors of animal diseases, thereby increasing the chances of human infection. Argentine hemorrhagic fever, a natural infection of rodents, emerged as a result of an agricultural practice placing humans in close proximity to the rodents. Marburg, Machupo, Hantaan, and Rift Valley fever viruses are also of zoonotic origin, as, arguably, is human immunodeficiency virus (HIV). Yellow fever, whose natural cycle of infection takes place in a jungle habitat and involves monkeys and mosquitoes in tropical areas of Africa and South America, is probably an ancient zoonosis. Jungle yellow fever occurs when humans interpose themselves in the natural cycle and are bitten by infected mosquitoes. Yet there is also urban yellow fever, in which the same virus is transmitted among
humans by other mosquitoes (e.g., Aedes aegypti) that have adapted to living in cities. It is generally believed that the movement of people through the slave trade and maritime commerce disseminated yellow fever, dengue, and chikungunya viruses, as well as Ae. aegypti , from Africa to other tropical areas. Ae. aegypti is still widespread in many urban areas of the southeastern United States, although the last yellow fever epidemic in a major U.S. city was in New Orleans in 1905.
Although the odds are low that a randomly chosen organism will become a successful human pathogen, the great variety of microorganisms in nature increases those odds. For example, field sampling and disease surveillance efforts have now identified more than 520 arthropod-borne viruses, or arboviruses (Karabatsos, 1985). The disease potential of most of these viruses is unknown, but nearly 100 have been shown to cause human disease (Benenson, 1990). In spite of the demise of the Rockefeller Foundation arbovirus program in 1971, and although only a few laboratories are actively searching for new pathogens in animals and arthropods, new viruses are being discovered every year (see Box 2-2).
One example of a recently discovered zoonotic virus is Guanarito, the cause of Venezuelan hemorrhagic fever. In the fall of 1989, an outbreak of an unusually severe and sometimes fatal disease was detected in the state of Portuguesa in central Venezuela. Patients presented for treatment with prolonged fever, headache, arthralgia, diarrhea, cough, sore throat, prostration, leucopenia, thrombocytopenia, and hemorrhagic manifestations. Physicians in the region initially diagnosed the disease as dengue hemorrhagic fever (DHF). During one period, from early May 1990 through late March 1991, 104 cases of the disease were recorded. Slightly more than a quarter of these patients, most of them adults, died (Salas et al., 1991).
All of the cases of the DHF-like illness occurred in the Municipio of Guanarito in Portuguesa State, or in adjoining areas in Barinas State. The Municipio of Guanarito, population 20,000, is located in the central plains of Venezuela, a major food-producing region. The outbreak was confined to the municipio's roughly 12,000 rural inhabitants, who either farm or raise cattle (Salas et al., 1991).
In the fall of 1990, a virologist from the Venezuelan Ministry of Health sent serum samples from several patients who were suspected to have DHF to the Yale Arbovirus Research Unit (YARU) at Yale University School of Medicine. No virus could be isolated after routine culture of the sera in mosquito cells (the standard method for recovery of dengue viruses).
In early 1991, a member of the YARU staff visited Venezuela while on a trip to South America collecting dengue virus isolates for an ongoing research project. In Caracas, the YARU staff member was given spleen cultures from two fatal cases of suspected DHF from the Guanarito area. Upon inoculation into newborn mice and Vero (monkey kidney) cell cultures at
|
BOX 2-2 Arboviruses Worldwide, in 1930, only six viruses were known to be maintained in cycles between animal hosts and arthropod vectors like mosquitoes, gnats, and ticks (Karabatsos, 1985). Only one of the recognized arboviruses (arthropod-borne viruses), yellow fever virus, caused disease in humans. The other five viruses were responsible for epizootics and major economic losses in domestic animals: bluetongue in sheep and cattle, Nairobi sheep disease, Louping ill in sheep, vesicular stomatitis in cattle, and African swine fever. Later in the same decade, there was an explosion of newly emerged arthropod-borne diseases in North America. Western and eastern equine encephalomyelitis viruses caused major outbreaks with high case fatality rates in both equines and humans. St. Louis encephalitis virus was associated with more than 1,000 cases and 201 deaths in residents of Missouri. Subsequent research demonstrated that each of these viruses was maintained in a cycle dependent on mosquitoes and birds. When any of the viruses invaded the human population, an epidemic often ensued. Since the 1930s, 86 additional arboviruses have been found in North America. Fortunately, only a few, such as the California encephalitis complex, Colorado tick fever, and the dengue fever viruses, have been consistently associated with human disease. However, all 86 viruses are distributed widely, and many have thus far been shown to cause only inapparent infections in humans. A shift in the virulence of the viruses or in human susceptibility could potentially alter the present equilibrium. Some experts warn that the arboviruses are "viruses looking for a human disease." The threat of arboviral disease is not limited to North America. The 1985 International Catalogue of Arboviruses (Karabatsos, 1985) identified 504 arboviruses worldwide, 124 of which have been associated with a disease. It is of continuing concern that nonindigenous viruses might be introduced into the United States through travel and trade. The rate of discovery of arboviruses reflects the intensity of the worldwide search by the Rockefeller Foundation, government agencies, and universities from 1950 to 1980. There has been a significant decrease in activity for such programs in recent years, as seen in the table below. Yet all the while, new arboviruses continue to be found whenever and wherever a search is made.
|
||||||||||||||||||||||||
YARU, these samples subsequently yielded two isolates of a previously unknown arenavirus, a family of viruses generally thought to be rodent-borne. The organism was distinct from Lassa, Junin, and Machupo viruses, the other arenaviruses that are known to cause severe hemorrhagic illnesses in humans. The new agent has since been designated the Guanarito virus, and its associated disease has been labeled Venezuelan hemorrhagic fever (Salas et al., 1991).
Venezuelan health officials are now attempting to determine the risk factors, geographic distribution, and clinical spectrum of Guanarito virus infection and to update the incidence data on it. Studies are currently in progress at the U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID) to develop an animal model for the disease and to evaluate possible therapeutic agents. In October 1991, scientists from YARU visited Venezuela to initiate a study to identify positively the rodent reservoir(s) of Guanarito virus.
Rodents have been implicated in a number of zoonotic infections, but the zoonotic pool also includes marine animals, such as seals, porpoises, and dolphins, which like humans are susceptible to outbreaks of infectious disease. Most such occurrences pass unnoticed, either because they occur far from shore or because the number of animals affected is too small to draw attention to the possibility of infectious disease. Occasionally, however, marine epidemics do attract attention, usually when large numbers of dead carcasses suddenly appear on a popular beach.
The most recent major epizootic, reported initially in harbor seals living in the waters off Europe and the United Kingdom, began in April 1988. Thousands of the animals died. The hardest-hit area was along Britain's East Anglian coast, where more than half of the native seal population is estimated to have died. The outbreak peaked in August and tapered off through late 1989. Few dead seals have since been reported in this area.
It now appears that the same or a similar disease was present in Siberian seals somewhat earlier than the European epizootic (Grachev et al., 1989). The disease was also found in porpoises (Kennedy et al., 1988) and in dolphins (M. Domingo et al., 1990). Extensive study of the European outbreak resulted in the isolation of the causative agent, a virus, which is similar to measles, canine distemper, and rinderpest viruses.
Occasionally, marine viruses cause disease in terrestrial mammals or humans. For example, a strain of influenza A virus (H7N7) led to epidemic outbreaks in seals in 1980 and caused conjunctivitis in humans who handled the affected seals (Webster et al., 1981). It has been suggested that vesicular exanthema of swine, a serious viral disease caused by a calicivirus, was introduced into pigs through feed that contained material from sea lions. Many caliciviruses of terrestrial mammals may have been introduced from marine sources (Smith and Boyt, 1990). Among human viruses, hepatitis E
virus (the enterically transmitted non-A, non-B hepatitis that is usually waterborne and is widespread in tropical areas including parts of South America) has tentatively been classified as a calicivirus (Reyes et al., 1990).
There are also a number of established diseases whose link to an infectious agent has only recently been discovered. In addition to peptic ulcer, mentioned in Chapter 1, other diseases with a newfound link to a microbe include cervical cancer (associated with human papillomaviruses) and human T cell lymphotropic virus (HTLV)-I-associated myelopathy or tropical spastic paraparesis (resulting from infection with HTLV). Diseases for which possible links to infectious agents are under investigation include rheumatoid arthritis (parvovirus B19, HTLV-I); atherosclerosis (cytomegalovirus [CMV], herpes simplex virus [HSV]-1 and HSV-2, or Chlamydia pneumoniae); and insulin-dependent diabetes mellitus (coxsackievirus B5). Several of these examples are discussed later in this chapter.
Rather than categorize emerging microbial threats by type of agent—viral, bacterial, protozoal, helminthic, or fungal—this report classifies emerging threats according to the factors related to their emergence:
-
Human demographics and behavior
-
Technology and industry
-
Economic development and land use
-
International travel and commerce
-
Microbial adaptation and change
-
Breakdown of public health measures
The classification draws attention to the specific forces that shape infectious disease emergence (see Figure 2-1). These forces (i.e., factors in emergence) operate on different elements in the process of emergence. Some of the factors influence the acquisition of an emerging microbe by humans and other animals; others primarily affect the microbe's spread among populations. Although it is a difficult, if not impossible, task to predict the emergence of "new" infectious diseases/agents, it is helpful to understand the factors that facilitate the emergence and spread of infectious diseases in general. We must focus on what we do know: the infectious disease that will emerge or reemerge is likely to do so through one or more of the "facilitative pathways" diagrammed in Figure 2-1. An awareness of this system of pathways constitutes the first step to reasoned prevention and control of infectious diseases.
Many of the diseases addressed in this report have emerged because of a combination of factors. This is not surprising, given the often complex interactions of microbes, their human and animal hosts, and the environment. As much as possible, however, the committee has attempted to illustrate specific causes of emergence with diseases or agents whose emergence is primarily due to that one factor.
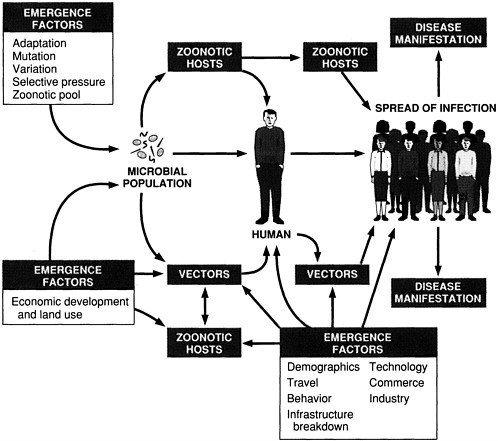
FIGURE 2-1 Schematic of infectious disease emergence.
HUMAN DEMOGRAPHICS AND BEHAVIOR
In the complex set of interactions that result in disease emergence, the human element—population growth, density, and distribution; immunosuppression; and behavior—plays a critical role. Increases in the size, density, and distribution of human populations can facilitate the spread of infectious agents; changes in the distribution of populations can bring people into contact with new pathogenic organisms or with vectors that transmit those organisms. Immunosuppression, a by-product of aging, the use of certain medications, diseases, or other factors, often permits infection by microorganisms that are not normally pathogenic in humans. Individual behavior, particularly sexual activity and the use of illegal drugs, contributes to the transmission of a number of diseases that have a major health impact on this and other countries.
Population Growth, Density, and Distribution
Until recently, most of the world's population lived in rural areas. In 1800, for example, less than 1.7 percent of people lived in urban communities. By 1970, however, more than a third of the world's people lived in urban settings. By the year 2000, that fraction is expected to rise to one-half (Dentler, 1977).
Not only are more people choosing to live in urban areas, but the size and density of many cities are also increasing, in part because of the overall population growth rate—each year the population of the world grows by approximately 70 million. High birth rates in many cities contribute to urbanization. By the end of the century, there will be 425 cities with a million or more inhabitants, an increase of 200 cities since 1985 (United Nations, 1985; World Resources Institute, 1986). Twenty-five cities are expected to have populations that exceed 11 million (Last and Wallace, 1992).
In many parts of the world, urban population growth has been accompanied by overcrowding, poor hygiene, inadequate sanitation (including wastewater disposal), and insufficient supplies of clean water. Urban development, with its attendant construction, emergence of slum areas and shanty towns, and infrastructure needs (e.g., water treatment and waste disposal facilities), has also caused ecological damage. These factors have created conditions under which certain disease-causing organisms and the vectors that carry them have thrived. The dengue viruses and their primary mosquito vector, Ae. aegypti, are one such example.
DENGUE
There are four distinct serotypes of dengue virus, each of which can cause a spectrum of illnesses ranging from mild fever and general malaise (dengue fever) to shock and fatal hemorrhagic disease (dengue hemorrhagic fever/dengue shock syndrome [DHF/DSS]). Dengue typically is a disease of young children, although older children and adults can be affected. Dengue viruses are transmitted to humans by Ae. aegypti mosquitoes.
Although dengue fever has plagued tropical populations for hundreds of years, the more severe form of the disease, DHF/DSS, is relatively new. The first recognized epidemic of DHF/DSS occurred in Manila in 1953 (Hammon et al., 1960). Dengue fever is usually the result of primary infection with one of the four dengue virus serotypes. DHF/DSS occurs in people who have been infected with two or more serotypes. The global spread and mixing of dengue serotypes have been made possible by the movement of infected individuals from one area to another.
Over the past 15 years, outbreaks of dengue fever have become increasingly numerous and severe, especially in urban centers in the tropics. At the
same time, epidemics of DHF/DSS have spread from Asia to the Americas. In the early 1980s, the disease arrived in Cuba, where it killed 158 Cubans in a major outbreak in 1981. The most recent epidemic took place in Venezuela in 1990; more than 3,100 cases of severe hemorrhagic disease were recorded, as well as 73 deaths (Gubler, 1991). There are currently endemic foci of DHF/DSS in the Caribbean and on the Yucatan Peninsula of Mexico.
Although the disease is concentrated in a small number of areas, worldwide incidence rates for DHF/DSS have skyrocketed. Since its appearance in 1956, there have been an average of 29,803 cases of DHF/DSS reported per year. Between 1986 and 1990, the average number of reported cases per year was more than 267,692 (Gubler, 1991). In Southeast Asia, DHF/DSS ranks as one of the leading causes of hospitalization and death among children.
Although the reasons for the increase in dengue activity and the changing disease pattern are not fully understood, the consequences of increased urbanization, densely populated areas, and poor sanitation play a significant role. These conditions favor the growth of mosquito populations. Dengue virus, which is short-lived in the human host, is best maintained in densely populated areas in which Ae. aegypti is abundant and susceptible individuals are concentrated. The lack of effective mosquito control in many tropical urban centers—a by-product of economic and political problems as well as indifference—has undoubtedly contributed to the dramatic rise in dengue infection worldwide.
The United States experienced dengue fever outbreaks in 1922 and 1945 (Langone, 1990). No cases of DHF/DSS have been reported to date, but imported cases of dengue fever occur annually in U.S. citizens who have returned from travel abroad. In 1990, for instance, 24 confirmed cases of imported dengue were reported to the Centers for Disease Control (CDC) (Centers for Disease Control, 1991c). Although Ae. aegypti and Ae. albopictus (a secondary vector) have become firmly established in much of the southeastern United States, epidemics of DHF/DSS, such as those seen in Cuba and Venezuela, are unlikely. The United States is less vulnerable because its standard of living is higher, houses are more likely to be screened, and discarded tires (see Box 2-3) and other containers that can breed Aedes are much less common than in many cities in the tropics. At present, the only effective way to limit the spread of dengue is to attack its principal vector, Ae. aegypti. Government-supported pesticide application programs, and efforts of private citizens to eliminate mosquito breeding sites (i.e., source reduction) near their homes, have been shown to work. The success of such source reduction efforts rests on public education programs.
Like the yellow fever vaccine, a reasonably priced dengue vaccine will be an important adjunct to vector control in stemming the spread of urban epidemics. Dengue vaccine development, however, has been complicated
|
BOX 2-3 Environmental Eyesore or Mosquito Nursery? Both! Discarded tires are an eyesore to most, but to some mosquitoes they offer an ideal location to deposit their eggs. Aedes aegypti and Ae. albopictus, both vectors of diseases such as dengue fever, viral encephalitis, and yellow fever, prefer to lay their eggs in water that collects in containers. Discarded tires, which hold water no matter in what position they land and which do not typically harbor predators like fish or frogs, are perfect incubators for the eggs of these mosquitoes. And each year, the United States throws away a quarter of a billion tires and imports several million (mostly from Japan) to be retreaded and resold. Not only do the mosquitoes find homes in discarded tires, but they also find transportation. When old tires are transported around the country by truck, mosquito eggs often go with them. Eggs then hatch hundreds of miles from where they were laid, and populations of adult mosquitoes can establish themselves in areas they might never have reached. Ae. albopictus actually "hitchhiked" to the United States in 1985 from Japan in a shipment of used tires. Already, this species has established itself as a resident. Tires are not the only human-made accommodations favored by mosquitoes. Any container that holds water—an empty beer or soda can, a bucket, or flowerpot—that is left outside during the warm spring and summer months is an attractive egg-laying site for a female mosquito. Some mosquitoes will even breed indoors in a moist container in a basement, garage, or shed if given the opportunity. This is in part why aerial spraying of pesticides is not an effective way to control mosquitoes; the insects usually lurk (and lay their eggs) in damp hiding places that the chemicals cannot reach. Thus, as innocuous as they may seem to many, discarded items like old tires and empty aluminum cans may play a role in the initiation and spread of mosquito-borne disease. Eliminating human-made breeding sites is a simple, logical way to reduce the chances of such disease. |
by the dengue virus's four serotypes. Scientists do not expect that a vaccine will be available in the next 5 to 10 years.
Immunosuppression
Immunosuppression, a weakening of the immune system, can be caused by a number of factors, including the following:
-
Inherited diseases
-
Aging
-
Prematurity (neonates)
-
HIV infection
-
Radiation treatment
-
Immunosuppressive medications for transplantation, therapy of malignancy (chemotherapy), or treatment of autoimmune disease
-
Malnutrition
-
Pregnancy
-
Severe trauma and burns
-
Other concurrent infections
-
Malignancy
Immunosuppression can result in disease in an individual who otherwise would have been able to fend off illness. Infections caused by typically nonthreatening organisms that take advantage of a person's weakened state are called opportunistic infections.
Although opportunistic infections have received a great deal of attention over the past decade with the onset of the HIV disease pandemic, they are not new. During the pandemics of influenza in the early part of this century, it was well understood that both the very young (who have immature immune systems) and the elderly (who have waning immune defenses and, often, concurrent disease) were in the greatest danger of succumbing to this viral disease. New medical treatments and technologies—for example, therapy for collagen-vascular diseases like rheumatoid arthritis and vasculitis, cancer chemotherapy, and organ transplantation—have created additional openings for opportunistic pathogens.
There is good reason to believe that opportunistic infections will continue to threaten human health. The mean age of the U.S. population continues to rise. More and more people are surviving into their eighties and nineties, when previously non-life-threatening infections become common killers. Suboptimal prenatal care for women of lower socioeconomic status, which often results in premature and disease-prone infants, will likely continue to be the norm. The number of people with AIDS will continue to grow as those who became infected years ago develop full-blown disease. The HIV-infected population serves as a particularly important point of surveillance for emerging opportunistic infections because of its size and because the immunosuppression that characterizes the disease is comprehensive.
In many cases, knowledge of the type and extent of a person's immune dysfunction makes it possible to predict the kinds of infections that person is likely to acquire. When immune deficiency is acute and general in nature, however, any number of infections are apt to result, often simultaneously and often with astounding intensity. Such is the case for people with HIV disease. It is not surprising that opportunistic infections account for 90 percent of all HIV disease-related deaths (Double Helix, 1990).
Opportunistic, infections are often caused by naturally occurring organisms that reside in most individuals. These organisms typically are kept in check by a healthy immune system, and many of them—for example, certain types of digestion-aiding bacteria in the intestine—are actually beneficial to normal body function. Disturbances in the integrity of the gastrointestinal tract, often the result of chemotherapy or radiation therapy, can introduce intestinal bacteria into the bloodstream, which can ultimately lead to life-threatening infection. Prolonged therapy with antibiotics can suppress the normal, resident bacteria that tend to keep fungal organisms like Candida in check, and the fungi can initiate a potentially dangerous infection.
The definition of an opportunistic infection should also include those infections caused by organisms that are normally pathogenic in healthy hosts but that are more common or induce more severe infections in the immune-impaired host. For example, although a nonimmune, healthy person who comes into contact with varicella virus might develop, and recover from, chickenpox, a person with an impaired immune system has a good chance of dying from the infection.
''Reactivated" infections, another type of opportunistic infection, occur in people who were previously infected with an organism that the body was able to suppress but not eliminate completely. When the immune systems of these individuals weaken, the circulating organism has a chance to cause disease again, or, in many cases, for the first time. For example, an estimated 80 percent of Americans are infected with cytomegalovirus (CMV), a herpesvirus (National Institute of Allergy and Infectious Diseases, 1991b). The virus typically does not produce serious illness in healthy adults, but for transplant recipients, who receive immunosuppressive drugs to keep them from rejecting foreign tissue, CMV can be a life-threatening complication.
Tuberculosis (TB) is another example of an infection that can be reactivated during immunosuppression. The causative agent of TB, Mycobacterium tuberculosis, usually persists in the body long after primary infection. Although infection with this bacterium in a previously unexposed person is usually self-limiting, reactivated TB, which can occur years later, can cause life-threatening lung disease. In recent years, TB has stricken HIV-infected individuals with alarming severity, causing a rapidly disseminated disease involving organs throughout the body.
After declining steadily since the 1950s, the incidence of TB in the United States has recently begun to climb. Since 1986, reported cases have increased 16 percent (see Figure 2-2) (Snider and Roper, 1992). This trend is largely attributable to cases of TB among those infected with HIV. TB is also occurring with greater frequency among immigrants and refugees, substance abusers, the homeless, the medically underserved, and the elderly. The majority of the increase has been among racial and ethnic minorities (especially blacks and Hispanics), children and young adults, and immigrants and refugees.

FIGURE 2-2 Incidence of tuberculosis, 1980 to 1990.
SOURCE: Center for Prevention Services, Centers for Disease Control.
The TB incidence rate among people infected with HIV is nearly 500 times the rate for the general population. In contrast to some fungal and other bacterial infections that occur only in the late stages of HIV disease, TB is a sentinel disease for HIV infection and tends to occur prior to other opportunistic infections, often before individuals realize they are HIV seropositive. In healthy individuals, pulmonary tuberculosis can be diagnosed and treated with relative ease (the cure rate is approximately 95 percent). In immunocom-promised persons, however, the disease is often disseminated throughout the body, making it much more difficult to diagnose and treat.
Sexual Activity and Substance Abuse
The HIV disease pandemic is the most devastating outbreak of a sexually transmitted disease since the emergence of syphilis almost 500 years ago in Western Europe. Syphilis, a bacterial disease, spread rapidly during the late fifteenth and early sixteenth centuries, quickly reaching a prevalence of 20 percent in many urban areas (Hirsch, 1885). Scholars believe that the disease was disseminated by the sexual activities of soldiers, and heterosexual promiscuity was, and continues to be, the primary mechanism by which the infection is spread. The incidence of syphilis in the United States fell dramatically earlier in this century with the introduction of penicillin. Between 1985 and 1990, however, incidence almost doubled, most notably among
heterosexuals who use crack cocaine (Centers for Disease Control, 1992f). A number of factors are associated with this rise, including multiple-partner sex to procure drugs, especially crack cocaine; inadequate health care among groups at risk; and declining educational levels among lower socioeconomic populations (Centers for Disease Control, 1991g).
The origin of HIV, the lentivirus (a member of the retrovirus family) that causes HIV disease and acquired immunodeficiency syndrome (AIDS), is not known. Viruses closely related to HIV have been present for hundreds, if not hundreds of thousands, of years in African nonhuman primates. Similar comparisons of human and nonhuman lentivirus isolates strongly suggest that HIV-2, the variant of HIV found primarily in persons from West Africa, may have infected humans following cross-species transmission from nonhuman primates (Gao et al., in press). This transmission could have occurred through bites of infected monkeys that were kept as pets, captured by trappers, or transported to other countries. Nucleotide sequence analyses show that HIV-2 and some isolates of simian immunodeficiency virus (SIV), an HIV-like virus, belong in the same subgroup; there are no data at present placing an SIV isolate from monkeys in the same subgroup as HIV-1. However, an SIV virus belonging to the same subgroup as HIV-1 has been isolated from chimpanzees. Yet if HIV-1-like viruses are present in nonhuman primates, it is possible that both HIV-1 and HIV-2 were occasionally, but infrequently, transmitted to humans and persisted in remote areas or in isolated individuals or populations for centuries. Although the earliest documented case of HIV infection was obtained from a serum sample collected in central Africa in 1959 (Garry, 1990), the country or continent in which the HIV disease epidemic began is not known. What is clear is that HIV and SIV arose through natural evolutionary processes.
The HIV disease pandemic initially escaped detection because of the disease's long incubation period (the time from infection to onset of life-threatening disease). After reservoirs of infection had been established in African and Western countries, HIV spread to all parts of the globe. In Africa, it is believed that HIV was transported by the movement of infected individuals from isolated rural communities to rapidly expanding cities. This urbanization was accompanied by changes in sexual behavior, which played a major role in the transmission of HIV among (primarily) heterosexual populations in Africa (Quinn et al., 1986). Preexisting infection with other sexually transmitted microorganisms, especially those that cause genital ulcers and local genital tract inflammation, probably facilitated heterosexual transmission of HIV (Hillman et al., 1989).
The early spread of HIV, particularly in the United States and Europe, was largely the result of high-risk sexual practices of some male homosexuals, and it was in this population that most of the early cases were seen. Within a short period, however, another major group of HIV-infected individuals
emerged: intravenous substance abusers. The introduction of crack cocaine in the United States in the mid-1980s added another component to the complex epidemiology of HIV infection. Many persons who abuse crack cocaine use sex as a currency to support their habit. The result has been a striking rise in heterosexually transmitted syphilis, chancroid, and HIV infection. Although in the United States, HIV infection occurs predominately in male homosexuals and intravenous substance abusers, the rate of infection among non-substance-abusing heterosexuals is increasing.
The fact that HIV first established itself in the United States mainly among gay men has both negative and positive repercussions. On the negative side, rapid emergence was facilitated among those individuals who engaged in anal intercourse with multiple partners. On the positive side, unusual diseases related to HIV infection initially occurred in a specific subpopulation, and that specificity probably hastened recognition of the syndrome and its infectious nature. Had the first cases of AIDS occurred in a more diverse population, it is likely that discovery of the exact nature of the problem would have been slowed. Once the disease was recognized as a new entity with the potential for epidemic spread, the biomedical research community began a concerted effort to identify the etiologic agent. Collaborations were established between health care workers, who provided blood samples from patients, and researchers, who in turn isolated and defined properties of the virus so that blood tests could be generated and the development of drugs and vaccines could begin.
Unfortunately, the U.S. political sector was not as responsive to the crisis and by its slow response may have contributed to the explosive growth of the epidemic. A major reason for this hesitancy appeared to be the antipathy of some federal officials to the behaviors of those persons initially affected by HIV disease: gay men and substance abusers. In some instances, federal officials thwarted efforts to curtail the epidemic. For example, former Surgeon General C. Everett Koop has stated, "Even though the Centers for Disease Control commissioned the first AIDS task force as early as June 1981, I, as Surgeon General, was not allowed to speak about AIDS publicly until the second Reagan term. Whenever I spoke on a health issue at a press conference or on a network morning TV show, the government public affairs people told the media in advance that I would not answer questions on AIDS, and I was not to be asked any questions on the subject. I have never understood why these peculiar restraints were placed on me. And although I have sought the explanation, I still don't know the answer" (Koop, 1991).
More detrimental, however, was the government's continued resistance to proposed sex education programs designed to interrupt transmission of HIV (Koop, 1991). The federal government's recent revocation of funding for an approved five-year study of teenage sexual behavior (Marshall,
1991) indicates the continuing controversy and ambivalence that surrounds many aspects of the nation's response to HIV disease.
To date, no drug has been developed that can prevent or cure HIV infection, and it is not likely that a vaccine will be available soon. In many areas of the world, particularly Eastern Europe, India, and Southeast Asia, the numbers of cases of the disease are escalating rapidly. In Africa, the demographics of the pandemic are changing, with HIV-infected individuals moving away from cities and back to rural areas (R. M. Anderson et al., 1991). In Africa, Latin America, the Caribbean, and North America, HIV is infecting increasingly larger numbers of heterosexuals, intravenous substance abusers, and children (Centers for Disease Control, 1991a). In 1989, HIV disease passed heart disease to become the second leading cause of death in U.S. males aged 25 to 44, behind accidental and unintended injury (Centers for Disease Control, 1991e). Estimates now place the total number of adults worldwide who have developed HIV disease at more than 1 million and those who are infected with HIV at 10 million. The World Health Organization (WHO) estimates that as many as 40 million people could be infected with HIV by the year 2000 (World Health Organization, 1991).
TECHNOLOGY AND INDUSTRY
Notwithstanding all of their benefits, technology and industry may directly or indirectly cause the emergence of infectious diseases. Modern medicine has created situations that are ideally suited for the emergence of infectious agents. The food and agriculture industries work continually to prevent the introduction of pathogenic organisms into our food supply, but they are not always successful. Waterborne pathogens are controlled by the careful treatment and disinfection of drinking water, but breakdowns do occur and sometimes result in the spread of infectious disease.
Modern Medicine
Generally, people who enter the hospital expect that their health will be improved by the treatment they receive. For at least 1 out of 20 patients, however, this is not the case. Each year, an estimated 2 million individuals in the United States (about 5 percent of the total number hospitalized) suffer nosocomial infections—viral, bacterial, protozoan, and fungal infections that were not present or incubating at the time of hospital admission (Fuchs, 1979; Wenzel, 1988; Martone, 1990). The rates of nosocomial infections in developing countries may be 5 to 10 times higher (Wenzel, 1987).
The health and financial impacts of nosocomial infections in this country are staggering. More than 20,000 deaths annually are attributed to hospital-acquired
infections, and patients who recover from these infections typically require 10 extra days of hospital care (Fuchs, 1979; Wenzel, 1988). Every year, hospital-acquired infections account for between $5 billion and $10 billion in additional medical-related expenses, most of which are due to excess hospital stays (Wenzel, 1987; Schaechter et al., 1989; Martone, 1990).
Although hospital sanitation has improved markedly since the late nineteenth century, when carbolic acid was first used as an antiseptic during surgery, nosocomial infections continue to challenge efforts to control them (Fuchs, 1979). Medical advances and antimicrobial resistance are at the heart of the struggle.
In February 1991, the CDC's Hospital Infections Division looked at 10-year trends in nosocomial infections using data collected through the National Nosocomial Infections Surveillance System (NNISS). The 1980s saw a tripling of the incidence of bacteremias (Ross, 1990) and a shift in the organisms that are most prevalent as the causes of nosocomial infections, from those that are generally susceptible to antimicrobials (e.g., Proteus mirabilis, Escherichia coli, and Klebsiella pneumoniae) to those that tend to be more refractory to treatment (e.g., Enterobacter, Pseudomonas, Enterococcus, and Candida species) (Schaberg et al., 1991). In addition, there appears to have been a significant increase in both the prevalence and variety of viral and fungal pathogens found to be causes of nosocomial infections (Ross, 1990). All of these observations implicate the hospital setting as a prime site for the emergence of microbial threats to health.
Many of the factors that increase the risk of infection in a hospital are inherent to any health care setting. Not only are persons with serious infections frequently admitted to hospitals, thus providing an intrahospital source of pathogenic organisms, but the proportion of people with increased susceptibility to infections is also greater in a hospital than in the general population. In addition, because health care institutions are not completely isolated from the community (employees, visitors, food, and supplies enter daily), patients are exposed to the same pathogens that circulate in the surrounding locale. Thus, nosocomial infections can be transmitted from staff to patients, from visitors to patients, and from patients to other patients. Infections can also be acquired from contaminated surfaces, such as floors, examining tables, or improperly sterilized instruments, and from the patient's own normal microbial flora, especially during invasive procedures.
Antimicrobial resistance, a problem in the treatment of many bacterial diseases, has particular relevance in the hospital setting. By their very nature, hospitals are filled with people who have increased susceptibility to infection. Also by nature, hospitals tend to use large quantities of antibiotics. (About a third of hospitalized patients receive such agents [Shapiro et al., 1979].) The combination of an immunologically vulnerable population and the widespread use of antibiotics is potentially risky, since the selective
pressure exerted on microbes by the constant challenge of antimicrobial compounds favors the survival of organisms that are resistant to these drugs (Holmberg et al., 1987).
An organism's development of drug resistance through selective pressure usually begins with exposure to an antimicrobial drug. Antimicrobial drugs and other compounds designed to combat human pathogens work by killing or inhibiting the growth of susceptible microorganisms. Because of genetic variability, however, not every bacterium, virus, protozoan, helminth, or fungus is naturally susceptible to these drugs. The result is that the drugs leave untouched a small number of resistant microbes, effectively "selecting for" those organisms that can survive attack by the drugs. These resistant organisms pose a potentially serious threat to health. Although the role of selective pressure in antimicrobial resistance is clear, additional studies (using appropriate epidemiological and molecular biological methodologies) are needed to identify and investigate the risk factors that promote transmission of resistant pathogens in the hospital setting.
Many standard hospital procedures facilitate patient acquisition of nosocomial infections. The use of conventional medical devices is responsible for the greatest share of such infections: several hundred thousand cases of device-related disease occur each year. The most common of these (and the most frequent of all nosocomial infections) is urinary tract infection (UTI) (Harding et al., 1991). The great majority of hospital-acquired UTIs are the result of catheterization. In some cases, infection results from nonsterile insertion of the catheter; more frequently, however, it is the entry with the catheter of normal body bacteria (e.g., E. coli and Staphylococcus species, which are usually kept out of the bladder by the mucosal barriers of the urinary tract) that cause infection. Other devices, such as endotracheal tubes and mechanical ventilators, can cause infection in a similar fashion.
Pneumonia is the second most common hospital-acquired illness and the leading cause of death from nosocomial infection. Infections related to surgical wounds are the third most common type. Skin provides one of the body's natural defenses against microbial invasion, and it is also home to usually harmless staphylococcal bacteria. When the skin is broken, however, as happens during surgery or intravenous catheterization, staphylococci (including antibiotic-resistant hospital strains) can gain access to deeper tissues and cause infection. Bloodstream infections, the fourth most common type of nosocomial condition, occur when microbes make their way deep into the body—typically with the help of medical devices or the use of invasive procedures—and enter the bloodstream. On rare occasions, bloodstream infections, including transfusion-induced yersiniosis and HIV infection, can also result from the use of contaminated blood products (see Box 2-4) (Cover and Aber, 1989; Martone, 1990).
|
BOX 2-4 How Safe Is the Blood Supply? Many of the 4 million people who receive a blood transfusion in the United States each year have concerns about contracting a communicable disease in the process. Like organs and other tissues that are transplanted, blood is a biological product that can host disease-causing microorganisms. Fortunately, however, blood that is donated today goes through a battery of tests designed to ensure that it is free of contamination by infectious agents. The American Red Cross (ARC) now tests donor blood for syphilis, hepatitis B and C, human T-lymphotrophic virus types 1 and 2 (HTLV-I and HTLV-II), HIV-1, and, recently, HIV-2 as well. HIV-2 currently ranks as the primary cause of HIV disease only in West Africa; yet as of September 1991, 31 people in the United States had been diagnosed with HIV-2 infection, making the virus a potential threat to the safety of the blood supply in this country (Johnston, 1991). The Department of Defense (DOD) and the American Association of Blood Banks (AABB) recently took steps to protect the blood supply from contamination with another microorganism, the leishmania parasite. Found primarily in Africa and Asia, the parasite was found late last year in the blood of more than two dozen soldiers returning from the Persian Gulf War. Both the DOD and the AABB, as well as the ARC, plan to refuse donations until at least 1993 from all individuals—mainly members of the U.S. armed services—who have traveled to the Middle East since August 1990. Many of the efforts made by blood banks to improve the safety of the blood supply have been tremendously successful. Thirty years ago, nearly one in three people who received a blood transfusion contracted some form of hepatitis; today, that risk has dropped to less than 1 in 100 (Russell, 1991). The chances of contracting HIV from a blood transfusion are considerably less than in the early 1980s, when AIDS was first identified. Prior to 1985, when testing for HIV in donor blood became widespread, more than 4,300 persons were infected by the virus through blood transfusions. From 1985 through December 1991, only 20 people have acquired HIV through transfusions (Centers for Disease Control, 1992e). The risk of HIV infection from a blood transfusion has been estimated at from 1 in 40,000 to 1 in 150,000 per unit of blood transfused, depending on the region from which the blood originated (Russell, 1991). Safeguards against microorganism-contaminated blood unfortunately are not foolproof. As a result, doctors have become much more conservative about using transfusions. Most encourage patients to contribute their own blood prior to surgery whenever possible, and many doctors have sought new alternatives to transfusions altogether. Automated cell salvage techniques that can be used either during or after surgery to recover, cleanse, and return lost blood are one such alternative. Until the search for an effective blood substitute is successful (several companies appear to be close to developing a safe product), protecting the blood supply and its users from infectious disease remains a top priority. |
Complex invasive procedures, such as tissue or organ transplantation, can also lead to nosocomial infection. The immunosuppressive drugs used to prevent the rejection of the foreign tissue or organ have the undesirable side effect of weakening the body's immune system. Often, these infections do not involve hospital microbes but pathogens from the donor tissue or pathogens that are already present in the recipient. Extensive testing of foreign tissue prior to transplantation guards against transmission of most such microbes. Latent agents, however, like the "slow" virus that causes Creutzfeld-Jacob disease, are extremely difficult to detect and may be inadvertently transferred to the transplant recipient in the seemingly normal tissue of the donor. Cases of HIV infection, hepatitis C, and CMV infection resulting from organ transplantation have all been documented, as have cases of Creutzfeld-Jacob, a degenerative brain disease, in recipients of transplanted corneas and human growth hormone (Lorber, 1988; Pereira et al., 1991).
HEALTH CARE DELIVERY
Changes in health care delivery over the past 20 years undoubtedly have had an impact on nosocomial infection rates. Rising health care costs play a key role. One cost-conscious health care strategy that appears to be contributing to the rise in cases of nosocomial infection is so-called industrial management in hospitals. Industrial management is intended both to maximize the ratio of patients to nurses and to maintain pools of health care workers—particularly nurses—who can rotate frequently between two or more units of an institution. From the hospital's perspective, maximizing the ratio of patients to nurses is desirable because it decreases health care costs. At the same time, the practice can increase disease transmission by reducing the time available for proper sanitation and increasing the number of infected patients to whom a nurse is exposed.
Exacerbating the potential disease-producing quality of these problems is the increasing bidirectional transfer of patients between acute care and chronic care hospitals. The mixing of patients from acute care facilities (who tend to be severely ill) with residents of chronic care hospitals (who tend to have decreased immune function owing to aging or chronic illness, or both) is potentially risky. Compared with hospital-based programs, infection control programs in many long-term care facilities are rudimentary, at best. Unlike hospital-based programs, there are no standardized criteria for defining nosocomial infections in long-term care facilities; in addition, adequate studies designed to assess the efficacy of their surveillance and control measures have not been conducted. This state of affairs contrasts sharply with such efforts in acute care hospitals, which have received for more attention and federal funding.
The problem is likely to grow even more serious with time, given the Agency for Health Care Policy and Research's estimates that 43 percent of all those who turned 65 years old in 1990 will enter a nursing home at some point in their lives (Agency for Health Care Policy and Research, 1990).
In sum, hospitals and long-term care facilities can no longer be viewed as isolated epidemiological units but must be seen as part of a network of patient care facilities. This network makes it possible for nosocomial and community-acquired infections to be rapidly and widely spread.
PREVENTION OF NOSOCOMIAL INFECTIONS
Studies show, surprisingly, that even under the most sanitary of conditions, only about a third to a half of all hospital-acquired infections are preventable (Schaechter et al., 1989; Martone, 1990). Several factors "stack the deck" against infection control efforts. Little can be done to eliminate most of these risk factors, which include age (newborns and the elderly have limited immunity), severity of illness (related to length of stay, also a risk factor), and underlying diseases (latent infections or immune deficiencies) (Freeman and McGowan, 1978). Increased attempts at prevention for high-risk patients may be the only weapon against infection in these circumstances.
Two recent approaches to controlling hospital-acquired infections have been remarkably successful: CDC's 1987 "Universal Blood and Body Fluid Precautions" and hepatitis B vaccination. Under the universal precautions, blood and certain body fluids of all patients are considered potential sources of HIV, hepatitis B virus (HBV), and other blood-borne pathogens. The guidelines are a revision of a 1983 document that recommended special precautions (use of gloves and other protective barriers, and careful handling and disposal of needles and other sharp instruments) for blood and body fluids of patients known or suspected to be infected with blood-borne pathogens. The hepatitis B vaccine was licensed in 1982.
Statistics demonstrate the impact of these two infection control measures. A recent study by the Hepatitis Branch at CDC documented a 75 percent decrease in cases of hepatitis B among health care workers in four sentinel counties between 1982 and 1988 (Alter et al., 1990). The study's authors surmised that the decrease in cases was "probably a direct result of immunization with hepatitis B vaccine and of wider implementation of universal blood precautions" (Alter et al., 1990).
Although the hepatitis B story clearly can be counted as a victory for hospital infection control, new microbial threats are likely to surface in the future. Health care institutions are prime breeding grounds for new and more virulent strains of organisms and may well represent one of the most
important sites of surveillance for new pathogens that will emerge to jeopardize health in the future.
Food Processing and Handling
The potential for foods to be involved in the emergence or reemergence of microbial threats to humans is great, in large part because there are many points in the food chain at which food safety can be compromised. This chain of events begins wherever crops or animals are raised; it proceeds through a complex system of manufacturing, distribution, and retailing and ends with the use of a food product by the consumer. Changes in any of a number of aspects of the farm-to-consumer chain, or inattention to food safety in general, can result in outbreaks of food-borne illness.
Although food containing viruses or parasites can cause illness (as can chemical contamination), the majority of individual cases of food-borne disease of known etiology in the United States are caused by bacteria. However, in more than half of the outbreaks of food-borne illness, the exact cause is unknown (Bean and Griffin, 1990). Although in many cases the lack of an exact cause reflects an incomplete investigation, at least some proportion of those outbreaks are likely to be the result of as yet unidentified food-borne pathogens.
There has been a substantial increase in our knowledge of food-borne diseases during the past 20 years, as reflected in an approximate tripling of the list of known food-borne pathogens. An important component of this increase in understanding is a better scientific grasp of the factors that allow microorganisms, and bacteria in particular, to cause human disease. Because of better methods of identifying food-borne pathogens, it has become clear that only certain strains of a bacterial species may cause food-borne illness.
For example, Escherichia coli is part of the natural intestinal flora of humans; its presence in a water sample has been used as evidence of fecal contamination by other pathogenic microorganisms. The majority of isolates of E. coli pose no threat to humans as food-borne pathogens. Researchers, however, have identified five distinct groups of E. coli that cause enteric disease. Based on the mechanism of pathogenesis of each group, they are designated enteroinvasive, enterotoxigenic, enteropathogenic, enteroadherent, and enterohemorrhagic E. coli (Archer and Young, 1988). The ability to detect these pathogenic isolates has been greatly enhanced by diagnostic tests that identify specific virulence-related genes or gene products such as toxins, adhesins, and cell-surface markers.
Improved epidemiologic surveillance has also played an important role in identifying microorganisms that cause food-borne disease. This was the
case for four outbreaks of human listeriosis, a bacterial infection that occurred in the United States, Canada, and Switzerland in the early to mid-1980s. Careful monitoring of disease incidence data by medical facilities allowed these epidemics to be detected, even though the actual number of cases was relatively low. Subsequent epidemiologic investigations implicated cole slaw (Schlech et al., 1983), milk (Fleming et al., 1985), and soft cheeses (Office of Federal Public Health, Switzerland, 1988; Linnan et al., 1988) as the vehicle of infection. Of particular concern is that listeriosis, caused by Listeria monocytogenes, is most often diagnosed in pregnant women or their newborns and in immunosuppressed individuals, in whom it can be fatal. The CDC has recently published recommendations for the prevention of food-borne listeriosis; for those at high risk (immunocompromised individuals, pregnant women, and the elderly) the recommendations cite additional foods to avoid (Centers for Disease Control, 1992h).
AGRICULTURAL CONDITIONS AND PRACTICES
Any change in the conditions or practices associated with the production of agricultural commodities can affect the safety of the food supply. A virtually uncontrollable factor, like the weather, can have a substantial impact. For example, drought can make grains more susceptible to mycotoxin-producing fungi, and toxic fungal metabolites, such as aflatoxin, can threaten the health of both humans and livestock. This particular risk has been substantially lessened by an ongoing U.S. Department of Agriculture (USDA) program that monitors the status of major agricultural commodities. Once identified, contaminated grain is destroyed.
New agricultural procedures can also have unanticipated microbiological effects. For example, the introduction of feedlots and large-scale poultry rearing and processing facilities has been implicated in the increasing incidence of human pathogens, such as Salmonella, in domestic animals over the past 30 years. The use of antibiotics to enhance the growth of and prevent illness in domestic animals has been questioned because of its potential role in the development and dissemination of antibiotic resistance (Cohen and Tauxe, 1986; Institute of Medicine, 1989). Approximately half the tonnage of antibiotics produced in the United States is used in the raising of animals for human consumption. Thus, concerns about the selection of antibiotic-resistant strains of bacteria and their passage into the human population as a result of this excessive use of antibiotics are realistic (Institute of Medicine, 1989). It is conceivable that surveillance of feedlot animals for the development of resistant organisms might be a means of early warning for the emergence of newly drug-resistant pathogens.
Broad-based societal events indirectly related to agriculture may also affect food safety. Recent concerns about bovine spongiform encephalopathy
(BSE) illustrate this point. In 1980, in England, the combination of increasing fuel prices and tighter restrictions on the use of organic solvents for lipid extraction led to changes in the processing of offal, the viscera and trimmings of butchered animals that are a major component of animal feed. The new methods do not appear to inactivate sufficiently the BSE agent, and increased incidence of BSE in domestic animals has been linked to offal. There is considerable controversy, at least in England, about whether the BSE agent may also infect humans (Dealer and Lacey, 1990, 1991; K. C. Taylor, 1991). To date, however, no human infections have been detected.
In addition to modifications of traditional farming methods, the introduction of new types of agriculture can have an impact on the emergence of microbial threats. Aquaculture and mariculture, for example, are rapidly becoming important methods of producing fish and seafood. Yet there has been relatively little effort to understand the potential microbial impact of this new technology. As aquaculture and mariculture farmers attempt to increase their yields of freshwater and marine animals, the stresses of overcrowding and overfeeding create ideal conditions for Aeromonas hydrophila, a common fish pathogen found in fresh and estuarine waters (Plumb, 1975; Hazen et al., 1978). Increasingly, A. hydrophila, A. veronii (biovariant sobria), A. caviae, A. jandaei, A. trota, A. schubertii, and A. veronii (biovariant veronii ) are being implicated as causes of nosocomial, wound, waterborne, and food-borne infections in humans (Daily et al., 1981; Buchanan and Palumbo, 1985; Hickman-Brenner et al., 1987, 1988; Janda and Duffey, 1988; Carnahan et al., 1989; Carnahan and Joseph, 1991; Joseph et al., 1991; Samuel Joseph, Professor, Department of Microbiology, University of Maryland, personal communication, 1992). These bacterial infections are being found in immunocompromised individuals and those in otherwise poor health (W. A. Davis et al., 1978). Although there are a number of potential sources of infection with Aeromonas species, aquaculture and mariculture are probably the most common sources, since the incidence of these organisms in the products of these agricultural methods approaches 100 percent.
The use of human and animal fecal material to enrich pond cultures in parts of China and India raises additional concerns about the safety of some imported aquaculture products (Ward, 1989). Such practices may enhance the spread of pathogens transmitted by an oral-fecal route. In the Calcutta region of India, where this method of enrichment is used to raise prawns, a high incidence of non-O1 Vibrio cholerae contamination has been reported (Nair et al., 1991).
FOOD PROCESSING AND PRESERVATION TECHNOLOGIES
The application of new food processing and preservation technologies can have unexpected effects on the microbial safety of foods. Something
as simple as a change in packaging can be important. For example, plastic overwraps for packages of fresh mushrooms were introduced in 1967 because they enhanced the keeping-quality of this highly perishable food. It was soon discovered, however, that the respiratory rate of mushrooms is so rapid that, even with a semipermeable plastic film, the oxygen in the pack is quickly depleted. This produces an anaerobic environment perfectly suited to Clostridium botulinum, the neurotoxin-producing bacterium that causes botulism (Sugiyama and Yang, 1975). The problem was remedied by punching two holes in the plastic film, which allowed sufficient oxygen transfer to prevent the growth of anaerobes and still permitted enough carbon dioxide accumulation to retard spoilage (Kautter et al., 1978).
Another example comes from China. It appears that the transportation of brined mushrooms in plastic bags in that country provided conditions favorable to the growth of S. aureus (Hardt-English et al., 1990). The resulting presence of staphylococcal enterotoxin brought a halt (which is still in force) in November 1989 to the importation of Chinese mushrooms into the United States.
New food preservation methods, such as modified atmosphere packaging (MAP), are being used with more frequency as U.S. consumers demonstrate a preference for fresh food products that have a minimum of processing and preservatives. MAP uses combinations of gases to suppress aerobic spoilage bacteria that create unpleasant odors and flavors (Seideman and Durland, 1984). Unfortunately, these gases may not discourage, and may even encourage, the growth of other pathogenic microorganisms that are not detectable by smell or taste (Post et al., 1985; Hintlian and Hotchkiss, 1986, 1987; Berrang et al., 1989; Ingham et al., 1990; Wimpfheimer et al., 1990; Hart et al., 1991).
One technology for ridding foods of microbial contaminants is ionizing irradiation. This approach was used for many years to sterilize medical equipment and supplies; at low doses, it can eliminate or control pathogenic bacteria, fungi, protozoa, and helminths in foods (Thayer, 1990). The technology is also highly effective for insect disinfestations. Ionizing irradiation to pasteurize or sterilize foods has been recommended as an effective tool for the control of food-borne diseases (Joint Expert Committee on Food Irradiation, 1980; Council for Agricultural Science and Technology, 1986; Thayer, 1990). It has been approved for various applications in more than 30 countries; this includes, recently, approval for eliminating insects from fruits, Trichinella spiralis from infected pork, and Salmonella from raw poultry. One of the unique characteristics of irradiation is that the appearance of foods processed in this way is not altered. Much of the controversy over the use of irradiation is the result of the misconception that treated foods are radioactive. Extensive research has unequivocally demonstrated
that there is no induction of radiation in foods that are treated with isotope sources of 60Co or 137Cs, x-ray sources of up to 5 million electron volts (MeV), or accelerated electron beam sources of less than 10 MeV (Koch and Eisenhower, 1967; Becker, 1983). It should be noted, however, that irradiation is not a panacea. In dairy products, which contain lipids sensitive to oxidation, for example, taste is affected. Furthermore, the technique is generally considered much less effective for inactivating viruses, compared with its effectiveness with bacteria and other more complex pathogens (e.g., fungi).
DEMOGRAPHICS
There are several important, ongoing demographic changes in the United States that will have direct and indirect effects on the emergence of new food-borne microbial diseases. Foremost is the fact that, at least through the early twenty-first century, the population increasingly will be composed of the elderly, a group particularly susceptible to food-borne pathogens.
Population expansion and the accompanying demands on infrastructure can also affect the safety of the food supply. For example, when estuarine areas are developed for residential or recreational purposes, water treatment capacity often lags behind requirements imposed by the population increase. In some cases, potentially dangerous viral and bacterial pathogens are released into the water from sewage effluent and storm-drain runoff, where they are concentrated by shellfish and subsequently harvested and consumed, often with minimal processing. The closing of shellfish beds because of the presence of these pathogens has become a major public health and economic issue in a number of coastal regions. The problem is compounded by poaching from closed beds. Methods for testing water for human enteric viruses (e.g., hepatitis A and C, Norwalk virus, and caliciviruses) currently are inadequate (Institute of Medicine, 1991b).
CONSUMER ATTITUDES AND BEHAVIOR
In general, particularly in the developed world, the public expects its food supply to be safe. To a great extent this expectation has been met through the safe manufacturing and distribution of prepared foods. The extent of training that individuals receive in proper food handling and preparation is declining. Owing to changes in family structure and the roles of women, home economics courses are being deemphasized in schools, and the use of convenience foods and dining out is increasing. With these changes, assumptions about food safety may lead to complacency. Consumer inattention to appropriate steps for maintaining food safety in the home can easily overwhelm safeguards built into food production and processing.
COMMERCE
Historically, most foods have been produced and consumed locally. The internationalization of the food industry since World War II, made possible by the advent of refrigerated freighters, has changed this simple relationship. Now fresh fruits and vegetables that previously would have been available for only a few weeks can be obtained all year long. Wide-scale importation of food products has greatly increased dietary options for many consumers. At the same time, there is concern that these foods may come from regions in which hygienic practices are not on a par with those in the United States. With this concern in mind, the movement of the South American cholera epidemic into Mexico is being monitored closely by U.S. public health officials, since Mexico supplies the United States with much of its fresh produce during the winter months.
International trade has become so pervasive that it is virtually impossible to screen most of the food entering the country for known microbial hazards, let alone for new microbiological threats. This situation is likely to continue as political agreements remove barriers between trading partners. There is already virtually unrestricted movement of foods among the member nations of the European Economic Community, for example. Similarly, the U.S.-Canada-Mexico free-trade agreement is likely to lead to reduced inspection of imported foods, a subject that is currently being hotly debated. For foods entering the United States, as in Europe, there is likely to be increasing reliance on inspections conducted by the country of origin. This will necessitate the further development and implementation of international standards, such as the Codex Alimentarius of the Joint Food and Agricultural Organization/World Health Organization Food Standards Programme.
International commerce can affect food safety, even when the food itself is not being transported. This was the case in a 1986 outbreak of shellfish-related paralytic poisonings in South Australia and Tasmania. The poisonings were caused by Alexandrium catenella, A. minutum, and Gymnodinium catenatum, three dinoflagellate species not normally found in that part of the world. These marine plankton, which produce potent neurotoxins, are concentrated by shellfish as they filter particulate matter from seawater. The toxins do not affect shellfish, but they can cause serious neurological problems when consumed by warm-blooded animals. The likely source of the organisms was the bilge water of ocean-going freighters. Typically, bilge water is changed while ships are in port, releasing the microscopic stowaways. Studies have indicated that entire miniature ecosystems are transported around the world in this manner (Hallegraeff and Bolch, 1991; Jones, 1991).
DIET AND FOOD PREPARATION
New dietary habits also have had an impact on disease emergence. Immigration to the United States, in particular, the relatively recent heavy influx of peoples from Latin America and Asia, has introduced many U.S. residents to new foods. Although popular, some of these foods, or the ways in which they are prepared, can cause disease as a result of contamination by any number of organisms or their toxic byproducts. Ceviche—raw seafood served in lemon juice—and sushi—raw seafood and rice wrapped in seaweed—are perhaps the most obvious examples of ethnic foods that have been associated with disease transmission, in most cases caused by intestinal worms (helminths).
Trichinosis
Trichinosis, a generally self-limiting but potentially fatal disease caused by an intestinal roundworm that migrates to and encysts in striated muscle tissue, can be contracted from improperly prepared food. Trichinosis incidence in the United States had declined nearly every year since 1947 (Bailey and Schantz, 1990). As a result mainly of a single large outbreak of trichinosis, however, the incidence of the disease increased in 1990. The largest recent outbreak in this country occurred when uncooked, contaminated commercial pork sausage was consumed at a wedding reception. Ninety individuals, most of whom were Southeast Asian immigrants who customarily eat raw sausage, developed trichinosis (Centers for Disease Control, 1991i). Although the majority of cases of trichinosis still result from eating raw or insufficiently cooked commercial pork, consumption of insufficiently frozen or cooked wild game, particularly walrus and bear, has been increasingly associated with the disease, at least since 1975 (McAuley et al., 1991).
Anisakiasis
Cases of human anisakiasis, a food-borne disease, generally are caused by either of two species of nematode worms, Pseudoterranova decipiens and Anisakis simplex, commonly referred to as codworms and herring worms, respectively. Adult worms are found in marine mammals and release eggs that hatch in seawater; they are ingested by krill, which are eaten in turn by squid or fish. Larval-stage worms penetrate the bowel of the fish and encyst in the muscle. Humans contract anisakiasis by eating raw or partially cooked seafood containing nematode larvae.
Anisakiasis occurs most commonly in people living in coastal areas or areas in which the consumption of raw fish is customary. The fish most
often responsible for anisakiasis are cod, pollock, halibut, rockfish, flatfish, mackerel, salmon, and herring. Although only 50 cases were documented in the United States from 1958 to 1988 (McKerrow et al., 1988), these figures probably underestimate the actual incidence of anisakiasis, which is not a reportable disease. In countries in which the consumption of raw seafood is common, anisakiasis is more prevalent. In 1984 in Japan, for example, 3,141 cases were reported (Oshima, 1987). With the current U.S. trend to encourage more people to eat fish for dietary reasons, it is important that the consumer be reminded to thoroughly cook the fish to preclude infection with nematodes, such as those that cause anisakiasis.
Water Treatment
Water that is untreated or that does not receive adequate processing can transmit infectious agents, such as bacteria (Vibrio cholerae, Salmonella typhi, enterotoxigenic Escherichia coli), viruses (hepatitis A virus and other enteroviruses, rotaviruses, Norwalk gastroenteritis viruses), and protozoan parasites (Giardia lamblia, Cryptosporidium, Entamoeba histolytica). A primary source for many of these pathogens is fecal contamination of source water that is subsequently inadequately treated, or similar contamination after treatment. Leakage of wastewater from septic tanks and other sewage disposal facilities into groundwater can also transmit infection. Fortunately, most water used in this country is effectively processed by municipal water treatment facilities.
In some instances, water is not treated. This is often the case with water from private wells or from natural springs. In 1989, for example, an outbreak of some 900 cases of gastroenteritis occurred in a new resort community in north-central Arizona. An investigation revealed that the source of the outbreak was tap water obtained from a deep well on the resort property. A Norwalk-like virus was apparently introduced into the well by a faulty sewage treatment facility nearby, whose untreated sewage passed through fractures in the sandstone and limestone fields surrounding the well (Lawson et al., 1991).
Water used for recreational purposes can also be the source of waterborne infectious disease outbreaks, caused by pathogens such as hepatitis A virus (Bryan et al., 1974) or Shigella (Blostein, 1991). Particularly in the southwestern portion of the United States, water used for recreation increasingly is actually reclaimed wastewater. Wastewater treated to remove undesirable organic and inorganic contaminants is also being used for irrigation, industrial processing, and nonpotable residential (watering lawns and trees, toilet flushing) and commercial (golf courses, for example) applications. Inadvertent ingestion of reclaimed water, which has undergone the same basic treatment as potable water (including disinfection), is not likely
to cause waterborne disease. Reclaimed water used for drinking is more of a concern because of residual contamination.
Not unexpectedly, the higher the quality of source water entering water treatment facilities, the easier it is to produce safe water for drinking. Unfortunately, high-quality sources of potable water are increasingly difficult to locate and maintain. This is especially true for areas that depend on large watersheds, such as the Mississippi and Ohio rivers, which are often polluted with chemicals and biological wastes. To treat such polluted water adequately puts a heavy burden on water treatment plants. In many urban areas, the designed capacities of municipal water treatment facilities are being challenged by the growth in population; in addition, equipment or procedural breakdowns may allow inadequately treated water to reach the consumer.
Outbreaks of waterborne disease in the United States are uncommon because of the nation's extensive public health infrastructure. Public health authorities, however, are especially mindful of potential outbreaks following natural disasters, such as earthquakes and hurricanes, that can lead to contamination of municipal water supplies.
ECONOMIC DEVELOPMENT AND LAND USE
Dam Building and Rift Valley Fever
Until relatively recently, Rift Valley fever, which is caused by a mosquito-borne virus, occurred only in Africa south of the Sahara and was primarily a disease of sheep and cattle. Periodic outbreaks were prevalent in South Africa, Tanzania, Kenya, and, during the mid-1970s, in the Sudan.
The first major outbreak of human disease occurred in Egypt in 1977. An estimated 200,000 people became sick, and 598 died (Meegan and Shope, 1981). Death was usually associated with acute hemorrhagic fever and hepatitis. The outbreak also caused abortions in sheep and cattle, which resulted in a drastic shortage of red meat in the Cairo marketplace.
The Egyptian epidemic has been linked by some to the construction of the Aswan Dam. Completion of the dam, in 1970, required that 800,000 hectares of reclaimed land be flooded. The dam stabilized the water table in the Nile Valley, which caused water to puddle and to serve as breeding sites for mosquitoes. The mosquitoes, in turn, may have offered a conduit for the virus to enter Egypt from Sub-Saharan Africa, although this has never been proven.
Awareness of the possible association of Rift Valley fever with the ecologic change following completion of the Aswan Dam led scientists to examine the potential for outbreaks of the disease in other areas of dam construction. One such effort, started in 1977 by the U.S. Agency for International Development (USAID), focused on the Diama Dam, then under
construction in the Senegal River Basin. Although Rift Valley fever had never been recognized in the region, surprisingly, a third of the local adult inhabitants were found to have antibodies to the virus (Tirrell et al., 1985). The pattern of infection indicated that the disease was endemic. Similar studies at the Institut Pasteur, Dakar, revealed widespread exposure to the virus among people and domestic animals in Mali and Mauritania, two of the countries bordering on the Senegal River. Early in 1987, the institute warned of ''a potentially important risk of amplification of the virus in relation to the migration of domestic animal herds and human populations" in southern Mauritania (Digoutte and Peters, 1989).
Later in 1987, after the Diama Dam was activated, an epidemic of Rift Valley fever occurred near the village of Keur Macene, upstream from the new dam. The epidemic was associated with abortions in sheep and cattle, and severe human disease in 1,264 inhabitants. There were 244 deaths (Jouan et al., 1988). The association of this epidemic with prior construction of a dam paralleled the experience in Egypt 10 years earlier. Whereas in Egypt, Rift Valley fever virus was introduced into an immunologically virgin population, in Mauritania it was almost certainly endemic in people and livestock living near the site of the dam. Prior to 1987, the virus caused either inapparent infection or more serious disease in too few people to be recognized. Once the dam was activated, however, the ecology changed. Nonimmune people and their domestic animals who were already in, or entering, the areas became infected. Under these favorable circumstances, the disease rapidly reached epidemic proportions.
Mosquitoes indigenous to both Europe and North America are potentially capable of transmitting the Rift Valley fever virus. In Egypt, the virus demonstrated its capacity to be transported, and in both Egypt and Mauritania, the virus was able to cause epidemics when ecological changes favored mosquito breeding. In the United States, the virus could enter in the blood of an infected person or by way of an animal imported into a wildlife park or zoo. This is an admittedly unlikely possibility; nevertheless, should Rift Valley fever virus become established in vectors in this country, its control would require expensive, integrated efforts including vaccination of domestic livestock and extensive measures to kill vector mosquitoes.
Reforestation and Lyme Disease
The emergence of Lyme disease, caused by the spirochetal bacterium Borrelia burgdorferi, is intimately tied to changing land use patterns that date back over the past two centuries. Early in the 1800s, the eastern United States was rendered virtually treeless when vast tracts of land were cleared to make way for agriculture (Jorgenson, 1971; Cronon, 1983). Deforestation was enhanced by the prodigious quantities of wood needed for domestic
fuel—as much as 20 cords were required each year for a New England home heated by open fireplaces. Additional wood was converted into charcoal for smelting iron and manufacturing glass. As the forests disappeared, so, too, did the deer population.
During the mid-1800s, agriculture in the United States began a monumental transition westward to the Great Plains, and the resulting abandonment of farms soon caused vast portions of the East to be retaken by forest. Unlike the relatively open primeval forest, however, this new woodland was choked with undergrowth and contained no predators large enough to regulate deer populations. Indeed, reforestation was so rapid that by 1980, the region was blanketed by four times as much woodland as in 1860, when less than a quarter of the land was forested (Thomson, 1977). Not surprisingly, deer in the eastern United States proliferated as their woodland habitat increased (Spielman, 1988). This rebound commenced during the early 1900s and became explosive during the past decade (Southeastern Cooperative Wildlife Diseases Study, 1922-1988).
At the same time that deer populations were increasing, people began to visit and to live in forested, rural areas, a trend that continues today. The resulting proximity of people, mice, deer, and ticks promotes human infection by the Lyme spirochete. This microbe is transmitted by the bite of certain Ixodes ticks (see Figure 2-3). The definitive host for these vectors is deer; the reservoir for the pathogen is the white-footed mouse.
Lyme disease is now the most common vector-borne disease in the United States. Cases of Lyme disease have been reported in all 50 states; 13 states reported more than 100 cases in 1990 (see Figure 2-4). Since 1982, 40, 108 cases of Lyme disease have been officially reported to the CDC (D. Dennis, Chief, Bacterial Zoonoses Branch, Centers for Disease Control, personal communication, 1992). Diagnosis of Lyme disease is so frequent an occurrence for physicians who practice in areas in which transmission is intense, however, that many fail to take the time to report each case, and reporting efficiency within an endemic site tends to decline with time. For this reason, the official tally of cases of Lyme disease in the United States should be viewed as a highly conservative reflection of the actual state of affairs.
Lyme disease was originally called erythema migrans or erythema chronicum migrans because of the distinctive rash that migrates from the point at which the infecting tick attaches itself. Throughout the first half of the twentieth century, the condition was rarely seen and was diagnosed solely in a few residents of forested parts of northern Europe (Afzelius, 1921).
In the United States, the first case of what would later be called Lyme disease was reported in a resident of Wisconsin in 1969 (Scrimenti, 1970). Retrospective studies have since identified an even earlier apparent case, acquired in 1962 on Cape Cod in Massachusetts (Steere et al., 1986). The first recognized outbreak of Lyme disease occurred in coastal Connecticut
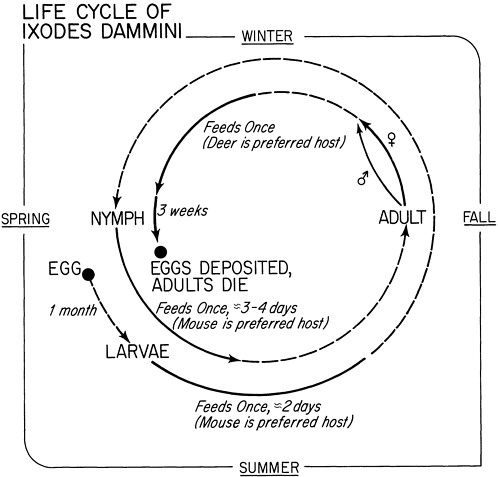
FIGURE 2-3 Life cycle of the Lyme disease tick, Ixodes dammini.
SOURCE: Spielman et al., 1985. Used with permission.
in 1975 (Steere et al., 1977). In 1975 and 1976, 51 people living in and around the town of Old Lyme, Connecticut, suffered from a condition that was tentatively diagnosed as juvenile rheumatoid arthritis and later as Lyme arthritis, in recognition of the focus of the epidemic. The link between Lyme arthritis and erythema migrans was established in the mid-1970s (Steere et al., 1986). In recognition of the enlarging spectrum of the disease, the term Lyme disease soon replaced Lyme arthritis.
Because Lyme disease initially was thought to be endemic only in south-central Connecticut, travel to that part of the country was considered, until the early 1980s, to be an essential criterion for diagnosis. Thus, residents of other states who presented with symptoms consistent with Lyme disease were not diagnosed as such unless a relevant travel history could be documented.
The ironic result was that Lyme disease was not considered to have occurred in Massachusetts until the case criteria were changed in 1982. This confusion resulted in an inordinate number of diagnoses of spider-bite during the 1970s, before these skin lesions were correctly attributed to the bites of spirochete-infected Ixodes ticks.
Once deer and infected ticks become well established in a populated site, the risk of human Lyme disease increases rapidly. Such was the case in the New York vacation communities on Fire Island, which became notorious foci of Lyme disease during the late 1970s, registering an annual incidence of about 1 percent (Hanrahan et al., 1984). A similar rise in cases occurred between 1975 and 1983 on Great Island, Massachusetts, where the annual incidence came to exceed 5 percent (Steere et al., 1986). An epidemic outbreak in Ipswich, Massachusetts, began in 1980, the year after Ixodes ticks were first discovered on the carcasses of deer. By 1986, the annual incidence of Lyme disease in that community came to exceed 10 percent (Lastavica et al., 1989). Similarly explosive growth in the trend of human cases of Lyme disease has been noted elsewhere, particularly in parts of New York, New Jersey, Pennsylvania, Wisconsin, and Minnesota.
FIGURE 2-4 Distribution of Lyme disease in the United States, 1991.
SOURCE: D. Dennis, Division of Vector-Borne Infectious Diseases, Centers for Disease Control.
Epidemic Lyme disease is an increasing problem in Europe, fueled by a post-World War II trend of reforestation and proliferation of deer. Epidemic Lyme disease was first diagnosed in Europe in the mid-1980s, a few years after the epidemic was recognized in the United States (Matuschka and Spielman, 1989). Reports of Lyme disease in Europe rose rapidly, until by 1988, twice as many cases were reported there as in the United States (Matuschka and Spielman, 1989). At least 1,000 new cases occur annually in Sweden (Jaenson, 1991), and similar incidence levels have been reported in Switzerland and Austria. The comparable annual estimate of new cases for Germany is much higher—between 30,000 and 60,000 cases (Matuschka and Spielman, 1989). Indeed, Borrelia -specific antibody is said to be detectable in some 7 percent of German residents. This extraordinarily high frequency of Lyme disease may be related to the large number of Central Europeans who live in forested areas and visit the forests for recreational purposes.
Lyme disease has been reported in many temperate parts of the world, including northeastern China, Japan, South Africa, and Australia (Jaenson, 1991). Unconfirmed, anecdotal reports of Lyme disease have been received from tropical Africa and South America.
A variety of etiologically distinct infectious agents are transmitted by the same ticks and maintained in the same rodents that perpetuate the agent of Lyme disease (Spielman, 1988). In the United States, babesiosis, a malaria-like disease caused by Babesia microti, has been diagnosed in people living in or near the same areas in which Lyme disease is prevalent. In human hosts, both of these infections can be fatal. Simultaneous infection is common.
Global Warming
Although it is still a controversial issue, the potential effects of global warming on disease transmission must be considered. This is particularly true for diseases caused by mosquito-borne viruses, since temperature increases in cooler climates may enlarge areas suitable for mosquito breeding. Unfortunately, it is currently impossible to predict accurately the effect of warming on disease emergence. This does not mean, however, that the issue should not be addressed. It is thus disturbing to note the apparent lack of interest in global warming as a possible contributor to public health crises on the part of funding agencies and environmental groups.
According to the best estimates obtainable from mathematical modeling, the earth's temperature could increase by as much as 5°C by the year 2050 (National Research Council, 1992). Should this occur, the distribution of disease vectors and the organisms they transmit might very well change. Research has shown that replication of virus in the mosquito is temperature dependent (Hardy et al., 1983). A warmer planet would likely enhance the transmission of some viruses, while slowing or halting the transmission of
others. For example, in the western United States, the geographical distribution of St. Louis encephalitis virus might expand north into areas in which transmission typically is blocked by cool temperatures (Hess et al., 1963). At the same time, the range of western equine encephalitis virus might shift from the southern part of the country northward, to cooler, more temperate habitats.
Other possible effects of global warming, such as increased precipitation or a rise in sea level, could affect the distribution of vectors that rely on water to complete their life cycles. For example, some mosquito species might alter their range and thus come into contact with new viruses or hosts; other species might become extinct in certain areas.
A change in climate can also affect the survival of infections agents, particularly viruses, outside their hosts. Humidity can favor or hinder the transmission of pathogenic agents. The seasonality of some human viral diseases (such as influenza A) may be due to climatic factors that exert an influence on the organism, its host, or both; sudden alterations in the climate could dramatically affect disease incidence.
The ability of infectious agents to adapt to changed conditions is considerable. Whether global warming occurring over an extended period of time would have any appreciable effects on these organisms, their distribution, or their ability to cause disease is unknown. Based on current knowledge, however, this committee believes that the impact of population growth (particularly when it leads to high population density) is likely to have a more predictable effect on the emergence of infectious diseases than the projected changes in global temperatures.
INTERNATIONAL TRAVEL AND COMMERCE
Travel
Travel, which involves the movement of people and microbes from one region to another, has always contributed to the emergence of infectious diseases. Whether new diseases emerge depends on the novelty of the microbe being introduced, its transmissibility, and the existence of an environment suitable for maintaining the disease and its agent. It is important to distinguish between transient introductions or acquisitions of novel diseases, which are common, and the establishment and propagation of a new pathogen, which are rare. Two examples of such establishment are syphilis and smallpox. According to the view that is still most widely held, syphilis is believed to have been introduced into Europe by sailors returning from the New World (Ampel, 1991). European explorers are believed to have introduced smallpox to the Americas (Crosby, 1972).
Until its global eradication in 1977, smallpox was frequently spread by travel. In 1940, nine years before the disease was eradicated in the United
States, there were nearly 3,000 cases reported in this country (Fenner et al., 1988), all of which occurred in or were the result of transmission from infected foreign travelers. The same pattern holds for dengue (see the earlier discussion), which is currently hyperendemic in parts of Asia and the Caribbean. In 1990, the U.S. Public Health Service (PHS) reported 102 suspected cases of imported dengue, although only 24 cases could be confirmed (Centers for Disease Control, 1991c).
Many other diseases common in other countries are periodically introduced into the United States by travelers. Lassa fever, an acute viral illness, is endemic to West Africa. It first came to the attention of U.S. health officials because of a series of epidemics in Africa between 1969 and 1974 (Carey et al., 1972; Monath et al., 1973; Fraser et al., 1974). Many of the original outbreaks, including almost all of the secondary cases, involved health care personnel who had cared for infected patients. The best known of these instances was an outbreak at an American mission hospital in Jos, Nigeria (Frame et al., 1974), which is the subject of a popular book, Fever! (Fuller, 1974).
Because Lassa fever is endemic to parts of Africa, sporadic introductions into the United States by travelers returning from that country are likely (see Box 2-5). However, since the virus is maintained in a rodent species not normally found in the United States, it is unlikely that the disease will become established in this country (barring the emergence of a suitable rodent host in the United States or changes in the host range of the virus). Nevertheless, each imported case of the disease has the potential to be followed by a significant number of secondary infections among close contacts.
MALARIA
Malaria, considered one of the greatest contemporary killers among infectious diseases, is no longer endemic to this country but is one of the diseases that is most frequently imported. The CDC reported 1,173 imported malaria cases in 1991 (Centers for Disease Control, 1992b). Figure 2-5 shows malaria incidence data for 1930 through 1990. During this period, there were four major peaks of malaria, including one that began in 1980. During this latest peak, virtually all of the cases were imported.1
|
BOX 2-5 Imported Lassa Fever In early 1989, a man who had visited Nigeria for a funeral became sick after returning to the United States. The patient was a 43-year-old mechanical engineer living in a Chicago suburb. Shortly after his return from Nigeria, he walked into a health clinic complaining of fever and sore throat. The area was in the midst of a winter influenza epidemic, and he was advised to take acetaminophen for the fever but was not otherwise treated. The symptoms worsened, and when he returned three days later, swollen lymph nodes and a phlegm-covered throat were noted, for which he was given penicillin. Five days later, his condition had deteriorated; his symptoms now included bloody diarrhea and facial swelling, and he had elevated liver enzymes. An attentive specialist who saw the patient at a local hospital suspected a viral hemorrhagic fever after reviewing the patient's history. That history revealed that the patient had been in Nigeria for his parents' funerals until a few days before his illness began. His mother's death, of a febrile illness, had occurred two weeks before the patient's symptoms appeared, and was followed by his father's death from a similar illness 10 days later. The travel and disease history made the clinician suspect Lassa fever, a disease known to be endemic to Nigeria, and he called the CDC in Atlanta. The CDC was later able to confirm the diagnosis of Lassa fever by virologic methods. Ribavirin, the only drug presently available for this infection, was ordered for the patient, but he died of cardiac arrest before the drug arrived. A total of 102 people had come into contact with the patient when he was likely to have been infectious. High-risk contacts (in this instance, immediate family who had had intimate contact, washed soiled linens, and shared utensils) were also placed on prophylactic ribavirin. Medium-risk contacts included a laboratory technician, the patient's nurse, and a physician who was not wearing gloves when he inserted an intravenous line into the patient. None of the contacts became infected, a somewhat unusual circumstance in the case of this disease. The patient was undiagnosed for almost two weeks, during which time the virus could have been passed to the patient's care givers, to other patients of these care givers, and to family members. Transmitted primarily through direct contact with the blood or other bodily fluids of an infected person, Lassa has been suspected in some cases to be spread by airborne transmission. Had this been true in the Chicago case, the number of direct and indirect contacts in danger of contracting the disease could have been much greater. The ease with which people can travel around the world today means that "exotic" diseases can move just as quickly. Physicians must be consistently aware of infectious diseases that originate in other parts of the world, and vigilant about obtaining a travel history for patients with undiagnosed illness, especially if it is accompanied by fever. SOURCE: Holmes et al., 1990. |
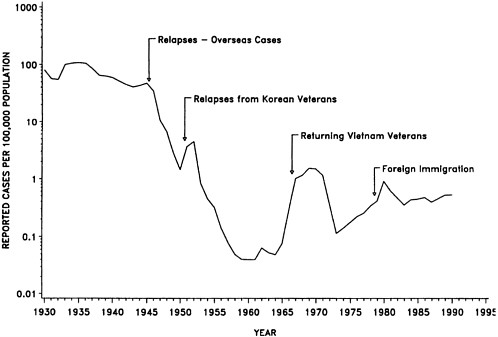
FIGURE 2-5 Incidence of malaria in the United States, by year, 1930-1990.
SOURCE: Centers for Disease Control, 1991h.
Until the mid-1970s, most cases of imported malaria in the United States occurred in war veterans who had acquired the disease while on overseas duty.
Outbreaks of nonimported (indigenous) cases of malaria, which appear to be associated with infected migrant workers, have occurred in southern California (mostly in San Diego County) and Florida (Branati et al., 1954; Centers for Disease Control, 1991f). These outbreaks have been small and so far relatively isolated, but the potential exists for the disease to become reestablished in the United States, since competent mosquito vectors are present in abundance. In fact, secondary cases in local contacts of individuals with imported disease are periodically recognized (Maldonado et al., 1990; Centers for Disease Control, 1991f).
In the areas of California and Florida where malaria outbreaks have occurred, sanitary facilities and housing are often substandard, a fact that can complicate vector-control efforts as well as medical treatment. Finding and treating illegal migrant workers infected with the malaria parasite are especially hard. The demand for cheap migrant labor and the limited availability of housing and health care for these workers may mean that the
scenarios in California and Florida will be repeated in other areas of the country. One recent report documented two cases of malaria in New Jersey in which neither individual had traveled outside of the state (Malaria Branch, Centers for Disease Control, unpublished report, 1991). States in the Southwest and along the Gulf of Mexico are at particular risk for malaria because of their proximity to the border with Mexico, where many of the illegal immigrants gain access to the United States.
Commerce
The international transportation of goods has indirectly led to the emergence of a number of infectious diseases. Most often it is not the goods themselves that pose the problem. Rather, it is infected animals in the cargo hold of a plane or ship, or bilge water contaminated with potentially pathogenic microbes that can hitch a ride from one part of the world to another. As discussed later in this chapter, the current cholera epidemic in the Western Hemisphere appears to be the direct result of international shipping. Transportation also played a significant role in the emergence of plague in the United States in 1900 (see Chapter 1).
Viruses are a particular problem because of their wide distribution, the ecology of their vectors, and their potential to cause human disease. The current International Catalogue of Arboviruses (Karabatsos, 1985) lists more than 500 separate viruses, about a fifth of which are capable of causing human disease. In general, viral diseases are hard to diagnose because of their variety and the difficulties associated with working with them in the laboratory. Viruses are often maintained in nature through enzootic cycles of transmission. Human infection is not essential to their long-term survival; in fact, humans often represent a dead-end host. There are undoubtedly undetected viruses in rural areas of the United States and in remote corners of the world that could cause human disease. With continued movement of people into rural areas, environmental damage caused by development, and the transport of people and products between remote areas and more developed parts of the world, the stage has been set for the emergence of "new" viral diseases.
HANTAAN AND RELATED HANTAVIRUSES
The Hantaan virus and related hantaviruses are some of the most recently recognized causes of an emerging disease. This group of viruses causes hemorrhagic fever with renal syndrome (HFRS), which is known as epidemic hemorrhagic fever in China and Korean hemorrhagic fever in Korea. The prototype virus, Hantaan, was first isolated in 1976 (H. W. Lee et al., 1978) from the lungs of its natural reservoir host, the striped field
mouse, Apodemus agrarius. Subsequent studies have identified antigenically related viruses that are also capable of causing acute, life-threatening human disease. These include Seoul virus, maintained in the domestic rat, Rattus norvegicus (Lee et al., 1982), and Puumala virus, maintained by the bank vole, Clethrionomys glareolus (Brummer-Korvenkontio et al., 1980).
Seoul virus causes a less severe form of HFRS, while Puumala virus is the cause of nephropathia epidemica, a disease commonly seen in Scandinavia, in the western portion of Russia, and, with increasing frequency, in Western Europe. Seoul virus is distributed virtually anywhere in the world where there are large, uncontrolled populations of R. norvegicus (LeDuc et al., 1986). This virus has been isolated and human infections documented in both North and South America (LeDuc et al., 1984, 1985; Childs et al., 1987; Glass et al., 1990). Undoubtedly, the present-day distribution of Seoul virus had its origin in international commerce and its unwanted rodent passenger, R. norvegicus.
The hantaviruses have also found their way into laboratory rodent colonies, in which they cause chronic, asymptomatic infections (LeDuc, 1987). Serious human disease and death have been documented in animal handlers, scientists, and others who unknowingly have been exposed to hantavirus-contaminated rat colonies. These problems continue to exist today, especially in Asia, where quality control of rodent breeding facilities is not as rigorously monitored as in the United States (Umenai et al., 1979; Desmyter et al., 1983; Lloyd et al., 1984).
The viruses have been spread in laboratory animals in several ways. Inbred strains of infected rats have been distributed to investigators around the world. Transplantable tumors, traditionally maintained in laboratory rats, have been the source of additional rodent and some human infections (Yamanishi et al., 1983). Continuous cell lines may also harbor the viruses, although examination of all rat-origin cell lines held by the American Type Culture Collection failed to identify any hantavirus contamination (LeDuc et al., 1985). The risk of contamination to reagents, such as monoclonal antibodies produced in infected rodent hosts, is currently unknown but clearly plausible. Recent studies have demonstrated that the common house mouse, Mus musculus, may harbor hantaviruses. Hantaviruses have been isolated from domestic mice in Texas (Baek et al., 1988) and in Yugoslavia (Diglisic et al., 1991; T. Avsic-Zupanc, Microbiologist, Institute of Microbiology, Medical Faculty of Ljubljana, Slovenia, personal communication, 1991).
Stopping the distribution of hantavirus-contaminated laboratory animals, cell cultures, or reagents is difficult, since there is no readily available commercial test for screening animals for infection. This issue is discussed more fully below.
THE FILOVIRUSES
In the late 1960s, a group of nonhuman primates was shipped from Uganda to Marburg, Germany, for use in vaccine development. The monkeys were infected with what was then an unknown virus, later to be named Marburg virus in recognition of the site of its discovery. Several animals died during shipment or immediately after arrival, and animal handlers and technicians were infected with this new virus. The resulting human disease was especially virulent, secondary cases occurred, and many people died (Martini, 1969).
Several years later, epidemics caused by another virus, later shown to be closely related to Marburg virus, occurred in Zaire and Sudan. These outbreaks claimed nearly 200 lives and almost completely eliminated medical support for the affected areas. The virus was later named Ebola virus, after the Ebola River which passes through the epidemic region. Marburg and Ebola are now recognized as members of the Filoviridae, a distinct family of viruses (Pattyn, 1978).
In 1989, the scenario of Marburg virus threatened to be repeated again, but this time in the United States. Scientists at the U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID) at Fort Detrick in Frederick, Maryland, were notified of excessive mortality among nonhuman primates recently imported from the Philippines to a facility in nearby Reston, Virginia, just outside Washington, D.C. USAMRIID staff had recently investigated an outbreak of simian hemorrhagic fever, a disease known to cause substantial mortality among infected nonhuman primates, and the Reston officials suspected that the same disease was present in their colony. However, when virus isolates were obtained from sick or dead monkeys and examined by electron microscopy, virus particles of characteristic filovirus morphology were observed (Geisbert and Jahrling, 1990). Public health officials were immediately notified, since preliminary attempts to identify the virus suggested that it was the highly virulent Ebola virus (Jahrling et al., 1990). Only after considerable study was it determined that the virus isolated was a distinct, new filovirus, later named Reston virus.
Several people were infected with Reston virus during the epizootic; however, to date, none has suffered overt clinical symptoms. At present, the potential of the Reston virus to cause human disease is uncertain. Regardless of the characteristics of this particular virus, the episode clearly demonstrates the critical role played by international commerce in introducing an exotic pathogen into the United States.
Perhaps the greatest problem associated with international commerce and its relation to disease emergence is the lack of appropriate, widely available diagnostic tests to allow effective screening of animals (and products made in animals) that are destined for investigational use. In contrast to arthropod
vectors of disease, which can be controlled through the judicious use of pesticides, it is not desirable to kill potential animal reservoirs of disease when the research being pursued requires that the particular animal remain alive. Those who work with imported biological materials need to be aware of the potential for contamination by infectious agents. Although it is generally not done, screening imported laboratory animals, cell lines, and transplantable tumors (including hybridomas used to make monoclonal antibodies) for exotic pathogens probably is the only effective mechanism to reduce the risk of emerging diseases.
MICROBIAL ADAPTATION AND CHANGE
Microbes are exceedingly numerous and diverse, but only a small fraction are capable of causing disease in animals or humans. To survive, most microbial species, whether pathogenic or not, must be well adapted to a particular ecological niche and must compete effectively with other micro-organisms. Their small size and high surface-to-volume ratios facilitate rapid growth and extensive impact on their environment. Microbial pathogens can colonize animals, humans, and arthropods because they have acquired (or evolved) a number of genes and gene products that enable them to do so. These gene products are extremely varied, but they include factors involved in transmission from one host to another, in cell-surface attachment and invasiveness, in countering or suppressing specific and nonspecific host immune responses, in persisting or surviving inside and outside a host organism, and in resistance to antimicrobial drugs. Nonpathogens can become pathogens (a rare event), and low-virulence pathogens can become highly virulent through mutation, recombination, and gene transfer.
Because of the relatively small amount of DNA or RNA, or both, that they carry, their rapid growth rate, and large populations, microbial pathogens can evolve and adapt very quickly. These evolutionary mechanisms (Table 2-2) allow them to adapt to new host cells or host species, produce ''new" toxins, bypass or suppress inflammatory and immune responses, and develop resistance to drugs and antibodies. The ability to adapt is required for the successful competition and evolutionary survival of any microbial form, but it is particularly crucial for pathogens, which must cope with host defenses as well as microbial competition. There are, for example, a number of determinants that can exert an influence on viral evolutionary events (Table 2-3). In fact, although hosts can help to drive the evolution of their parasites, the opposite is probably also true (Hamilton et al., 1990). Co-evolution of pathogens and their animal and human hosts will continue to be a challenge to medical science because change, novelty, or "newness" is built into such relationships.
TABLE 2-2 Examples of Viral Evolutionary Mechanisms
Natural Variation/Mutation
RNA VIRUSES
RNA viruses confront us with a paradox. On the one hand, their mutation rates are extraordinarily high (because unlike DNA viruses, RNA viruses have no mechanisms for correcting errors made during replication). On the other hand, the clinical expressions of the diseases they cause (such as poliomyelitis and measles) have remained constant for centuries. Nevertheless, analysis of RNA virus genomes reveals that each "virus" comprises a heterogenous mixture of mutants in variable proportions. Thus, any given strain or isolate is, in fact, polymorphic or represents a subset of the quasispecies
TABLE 2-3 Evolution of New Viruses: Constraints and Opportunities
|
Constraints |
|
|
|
Extreme viral alterations are lethal |
|
|
There may be requirements for co-evolution of viral cellular proteins |
|
|
Virus survival requires a critical level of virulence |
|
|
Propagation in alien hosts tends to be attenuating |
|
|
Adaptation to ecological niches is exquisitely specific |
|
|
Penetration of human immunologic barrier usually requires major antigenic change |
|
|
Infection with nonhuman (zoonotic) viruses is sometimes but not always contagious |
|
Opportunities |
|
|
|
High viral mutation rates |
|
|
Interviral genetic interaction |
|
|
Ecological change increasing opportunity for contact of man with vectors or viruses |
|
|
Changes in human behavior (e.g., sexual) |
|
|
Altered behavior of viruses in immunocompromised hosts. |
|
Adapted from Kilbourne, 1991. Used with permission. |
|
(E. Domingo et al., 1978). A virus, then, is identified as a consensus that reflects the predominating mutant(s) in a mixed population. Because predominating mutants seldom change, only unusual selective pressure by the host's immune response or other factors, such as host adaptation, will allow new mutants to gain ascendancy, resulting (rarely) in the emergence of distinguishably new viruses.
Influenza A Virus
Although influenza viruses mutate at a rate similar to other RNA viruses, they are unique in that they also evolve (undergo meaningful changes) relatively rapidly in nature. This is due to selective pressure on the virus from the large population of partially immune people, who have antibodies to the virus as a result of previous infections. To survive, the virus must undergo some degree of genetic mutation (or "antigenic drift"). This process is continuous and results in regional epidemics of influenza.
Much less frequently, the surface proteins—called hemagglutinin (H) and neuraminidase (N)—of the influenza virus undergo a radical change (an "antigenic shift") that creates a virus so different that no person possesses immunity to it. A pandemic of life-threatening disease results. Interestingly, the radical changes that have produced pandemic influenza viruses are rooted in the virus's acquisition of genetic material from animal influenza A viruses. Scientists have hypothesized that agricultural practices in Southeast
Asia (where most new strains of influenza arise) that put ducks, pigs, and farmers in close proximity may facilitate genetic shift in the virus (Scholtissek and Naylor, 1988; Langone, 1990; Morse, 1990). This theory is based on the idea that pigs have been infected by both an avian influenza virus and a human influenza virus. These viruses, while inside the pig, exchange genetic material, potentially resulting in a more virulent virus capable of initiating a pandemic if passed to humans.
To date, 14 distinct H and 9 distinct N influenza A antigens have been identified in birds and animals. Only three subtype combinations (representing three H and two N antigens) are known to cause disease in man: H1N1, H2N2, and H3N2. There have been two antigenic shifts in the influenza A virus since 1918: the first, in 1957, produced the H2N2 subtype; the second, in 1968, the H3N2 subtype. Antigenic drift and antigenic shift both challenge the immunity of human populations and require constant changes in the composition of influenza vaccine. Predicting the direction of these changes (based on worldwide surveillance data) is difficult.
Human Immunodeficiency Virus
A second example of RNA variability is HIV. HIV-1 and HIV-2, the two known HIV viruses, exhibit extensive genetic variability and exist in infected individuals as a complex mixture of closely related genomes, or quasispecies. These quasispecies undergo rapid genetic change such that the major viral form present in chronically infected persons differs over time. This continual change can result in alterations of those parts of the virus recognized by the human immune system; the effect of such alterations is to make the immune system less able to eliminate or suppress the virus.
It is likely that the extensive genetic variability of HIV-1 and HIV-2 will present major problems in the development of an effective vaccine. Because the differences between HIV-1 and HIV-2 are significant, a vaccine against one will probably not be effective against the other. More worrisome is the possibility that an HIV-1 or HIV-2 vaccine will not be effective against all quasispecies within that one subgroup. The quasispecies in any one person are closely related to each other but are different from those in someone else, raising the possibility that vaccines of different antigenic composition will be needed to prevent infection or disease in different individuals. Of additional concern is the fact that the majority of HIV isolates from other countries, such as Thailand and countries in Africa, differ substantially from strains found in the United States and Europe. It is highly probable that a widely effective HIV vaccine will need to be a composite of viral antigens from different regions of the globe or that multiple vaccines, targeted for specific regions, will need to be developed.
The mutability of HIV has been mentioned repeatedly during the HIV disease pandemic, raising questions about how broad a biological change the virus might undergo (Vartanian et al., 1991). It is important to make a distinction between point mutations—to which HIV is prone—and changes in pathogenic properties, such as initiation of infection, that would influence host range and mode of transmission. HIV does undergo frequent point mutations, especially in regions of the genome that are likely to be targeted by vaccines (Phillips et al., 1991). Although it is theoretically possible that such a change could alter its infective properties, the stability of other RNA viruses, with regard to host and organ specificity, indicates that this possibility is unlikely. Also reassuring is the general experience that evolutionary adaptation of pathogens tends toward lesser virulence.
DNA VIRUSES
Hepatitis B Virus
Hepatitis B is a DNA virus, but because it uses reverse transcriptase to replicate, it shares with RNA viruses the tendency to undergo significant and rapid genetic change. In recent years, a number of rare viral variants have been identified in patients infected with hepatitis B. These have generally fallen into two categories: variants with truncated protein products ("precore" and "pre-S" variants) and vaccine escape mutants.
The precore and pre-S variants have mutations in regions of DNA that immediately precede the coding sequences for certain viral proteins (core or surface protein, respectively). The mutations cause a truncated product to be manufactured and also appear to alter pathogenesis, especially in the case of precore variants (Neurath and Kent, 1988; Carman et al., 1991; Liang et al., 1991; Omata et al., 1991). The precore region of the hepatitis B virus is not required for the production of viral particles. Precore mutants thus are viable but lack the viral antigen known as HBeAg, which is a key component of some diagnostic tests. This variant was first isolated in patients in Italy and Greece who had an unusual form of severe chronic hepatitis but were negative for HBeAg (Carman et al., 1989; Carman et al., 1991). Since then, the precore variant has also been isolated from patients with fulminant acute hepatitis B infection (Carman et al., 1991; Liang et al., 1991; Omata et al., 1991).
In one study in Japan (Omata et al., 1991), nine patients with either acute fulminant hepatitis B infection (five patients) or severe exacerbation of chronic hepatitis B (four patients) were tested. Hepatitis B virus was successfully recovered from seven of the nine patients, and a precore variant with the identical nonsense (terminating) mutation was isolated. This variant was not found in 10 controls with acute, self-limited hepatitis B. In
another study, the precore variant was found in sera from eight of nine patients who were negative for HBeAg and had fulminant hepatitis B infection, but not in sera from six other patients who had fulminant hepatitis B but were positive for HBeAg. Finally, a nosocomial outbreak of fulminant hepatitis B with five fatalities occurred in Haifa, Israel, in 1989. The outbreak was traced to an intravenous heparin flush bottle that had been contaminated after use in a patient who was a chronic hepatitis B carrier. The same hepatitis B precore variant was detected in each of the patients (Liang et al., 1991). In addition to the single nonsense mutation at nucleotide 1896 (seen in all the precore isolates that have been identified to date), there was an additional mutation at nucleotide 1901, which the authors speculated might be related to the especially high mortality caused by this variant.
A vaccine escape mutant, by contrast, generally does not cause altered pathogenesis but lacks a particular antigenic site, allowing it to infect hosts protected by immunization or passively acquired antibody in much the same way that antigenic drift occurs in influenza A. Hepatitis B vaccine has been used in Italy since 1982 to protect infants born to mothers who are chronic carriers of hepatitis B. The hepatitis B vaccine escape mutant was found during follow-up studies of this population (Carman et al., 1990). Of 1,590 vaccinees (infants and family contacts), 44 became positive for hepatitis B surface antigen, indicating that they had become infected with hepatitis B despite immunization and prophylactic hepatitis B hyperimmune globulin. Another 7 patients were identified later. Viral DNA from a patient (in the original group) chosen for detailed study demonstrated a variant that lacked a major antigenic determinant and was therefore no longer neutralized by hyperimmune globulin or (presumably) by vaccine-induced antibodies. The loss of the antigenic site was due to a point mutation that altered a single amino acid. The other apparent vaccine failures were not as thoroughly investigated. Although it is not known conclusively whether this mechanism accounted for the other cases, there were indications that at least some of the other vaccine failures were due to an escape mutant. The localization of these mutants to a particular geographic region (the Mediterranean) was puzzling. This may have been due to host immunogenetic factors or suboptimal immunization schedules, or to the use of both vaccine and immunoglobulin, which increases selective pressure favoring an escape mutant.
BACTERIA
Bacteria cause disease because they produce so-called virulence factors. Virulence factors have several roles in bacterial pathogenesis: they allow the bacteria to resist nonspecific host clearance mechanisms; they help the bacteria acquire nutrients necessary for growth and survival; they assist the
bacteria to resist specific host immune mechanisms; and they provide the bacteria with a competitive advantage by inhibiting other microorganisms in the host.
The evolution, ab initio, of virulence by mutations would be an extraordinarily complex chain of events and is more often inferred as a natural process than observed in the laboratory. Resistance to antibiotics and to serum bactericidal factors, however, is a unit step often seen in experiments. More often, virulence genes can be characterized by their transmission from one cell to another.
Virulence factors vary from organism to organism and can often be transferred among receptive bacteria by bacteriophages and plasmids. This movement of genetic material is one way that bacteria cope with changes in their surroundings, such as the presence of antibodies or phagocytic cells. Bacteria may possess more than one virulence factor, including toxins (neuro-, entero-, endo-, cyto-, erythrogenic, etc.), enzymes, colonization factors, adhesins, bacteriocins, hemolysins, and cell invasion and drug resistance factors (see Table 2-4).
In addition to the versatility afforded by gene transfer, an unexpected plasticity has been found in bacteria that amplifies particular genes related to rapid growth or virulence (Terzaghi and O'Hara, 1990). Hence, salmonellae growing rapidly in rich media are genotypically different from those starved for specific carbon sources (Sonti and Roth, 1989). The same is true for cholera vibrios in their host environment compared with aqueous reservoirs (Mekalanos, 1983).
This plasticity has introduced new complications into the evaluation of pathogenicity of bacterial strains that have been stored in the laboratory, supporting the intuitions of a prior generation of medical bacteriologists. The underlying genetic mechanisms are still under study; it is not yet known whether they involve more than intense natural selection operating on large populations subject to modest rates of spontaneous mutation. Whatever the mechanism, the emergence of enhanced virulence potential is observed within time intervals measured in days or hours.
Brazilian Purpuric Fever
The most recent example of the emergence of a new disease that is likely to have been the result of a mutation causing enhanced virulence occurred in 1984. In that year, an outbreak of severe disease in 10 children, aged three months to eight years, occurred in a single town in São Paulo State in Brazil. Symptoms included high fever, vomiting, and abdominal pain, followed by the development of purpura and shock owing to vascular collapse. All 10 children died within 48 hours of the onset of the fever.
It was thought at first that the children had contracted meningococcal
TABLE 2-4 Representative Examples of Virulence Factors Encoded by Bacteriophages, Plasmids, and Transposons
meningitis. Microscopic examination and culture of cerebrospinal fluid (CSF), however, were negative for Neisseria meningitidis (Centers for Disease Control, 1985). Within the next two years, additional outbreaks and isolated cases were reported in nine other towns in São Paulo State. It was also determined that a similar outbreak had occurred in May 1984, in the neighboring state of Paraná (Centers for Disease Control, 1985, 1986; Brazilian
Purpuric Fever Study Group, 1987a,b). Subsequent epidemiologic investigation revealed that the majority of the children had developed purulent conjunctivitis (from infection by the bacterium Haemophilus influenzae , biogroup aegyptius), which had resolved prior to the acute onset of the high fever, vomiting, and abdominal pain.
No pathogenic organisms were cultured from blood or CSF in the first reported outbreak, probably because antibiotics had been administered before culture material was obtained (Centers for Disease Control, 1985). In subsequent outbreaks, however, H. influenzae, biogroup aegyptius, was isolated from blood or bloody CSF (Centers for Disease Control, 1986; Brazilian Purpuric Fever Study Group, 1987b; Irino et al., 1987). This was the first time this bacterium had been shown to produce invasive disease.
The disease, which became known as Brazilian purpuric fever, has since been reported in Central Australia. In one of two reported Australian cases, the organism was cultured from blood but not CSF (McIntyre et al., 1987). Other cases may have occurred, but because of the high prevalence of meningococcal disease in that part of the country, they were diagnosed as meningococcaemia. Brazilian purpuric fever may in fact have a wide geographic distribution. Parts of the world in which Haemophilus conjunctivitis is common, such as northern Africa (McIntyre et al., 1987), are potentially at risk for epidemics of Brazilian purpuric fever.
Selective Pressure and the Development of Resistance
The emergence of resistance in a known infectious agent may be a greater threat to public health than the emergence of a new disease. This is especially true if the agent causing the new disease is susceptible to currently available antimicrobial drugs or, if a vector is involved in transmission, the vector is susceptible to control strategies. If an emerging disease agent is resistant to currently available antimicrobials, the problem of control is considerably magnified, as it would be if an insect vector developed pesticide resistance. Thus, resistance to antimicrobials or pesticides is a critical factor in the emergence of infectious diseases.
RESISTANCE TO ANTIBIOTICS
An increasingly important contributor to the emergence of microbial threats to health is drug resistance. Microbes that once were easily controlled by antimicrobial drugs are, more and more often, causing infections that no longer respond to treatment with these drugs. Almost always, this resistance is the result of selective pressure.
There has always been resistance to antibiotics. No drug is universally effective against all bacteria, and as a drug is used, resistant organisms
emerge from the initially susceptible population (Jacoby and Archer, 1991). For example, the fluoroquinolones, when first introduced, were broadly effective and had low toxicity. With increasing use, most methicillin-resistant Staphylococcus aureus (MRSA) have become quinolone resistant, as have increasing numbers of Pseudomonas aeruginosa and other gram-negative nosocomial pathogens. The only available drug effective against MRSA and other methicillin-resistant staphylococci is vancomycin. About the only pathogenic bacterium that has not developed resistance to at least one antibiotic is Treponema pallidum, the spirochete that causes syphilis. Treating resistant infections requires the use of more expensive or more toxic alternative drugs and longer hospital stays; in addition, it frequently means a higher risk of death for the patient harboring a resistant pathogen. Estimates of the cost of antibiotic resistance in the United States annually range as high as $30 billion (Phelps, 1989). Even with the continuing development of new drugs, resistance to antibiotics is an increasingly important problem with certain bacterial pathogens.
Drug discovery programs have been crucial to the development of more effective antibiotics. A recent survey of vice presidents at major U.S. and Japanese pharmaceutical companies, however, found that about half of those companies have decreased or recently halted their antibiotic research programs. These decisions were reportedly based on perceptions that market needs for antibacterial products have been "satisfied" and that the market is "saturated" (Shlaes et al., 1991).
Pneumococci
Streptococcus pneumoniae is the commonest cause of community-acquired bacterial pneumonia and is also implicated in meningitis, otitis, sinusitis, bronchitis, and peritonitis, among other infections. For many years, pneumococcus was consistently susceptible to relatively low doses of penicillin (minimal inhibitory concentrations [MIC] of 0.1 microgram per milliliter or less). In the past 25 years, increasing numbers of penicillin-resistant isolates have been reported. Of the more than 5,000 S. pneumoniae isolates submitted to the CDC between 1979 and 1987 as part of a 35-hospital surveillance system, 5 percent had MICs greater than or equal to 0.1 microgram per milliliter (Spika et al., 1991). In other parts of the world, the frequency of resistance is even higher: more than 20 percent in Chile, South Africa, and parts of Spain (Klugman, 1990) and as high as 58 percent in Hungary (Marton et al., 1991). Patients infected with such strains can fail to respond to penicillin treatment, and some penicillin-resistant strains are also resistant to chloramphenicol, clindamycin, erythromycin, tetracycline, and trimethoprim-sulfamethoxazole, thus leaving few options for therapy. Today, in some areas, pneumococcal susceptibility to antibiotics can no longer
be assumed. Isolates obtained from blood and spinal fluid samples of seriously ill patients need to be tested for drug sensitivity.
Staphylococci and Enterococci
Infection with Staphylococcus aureus can cause a number of serious conditions, including bacteremia, endocarditis, meningitis, osteomyelitis, pneumonia, and urinary tract infection. Beta-lactamase-producing S. aureus emerged soon after penicillin came into clinical use and now constitutes 90 percent of isolates. Beta-lactamase gives the organism the ability to inactivate beta-lactam antibiotics. Beta-lactams that are not inactivated by the staphylococcal enzyme, like methicillin, nafcillin, oxacillin, cloxacillin, and many cephalosporins, provided effective therapy until methicillin-resistant S. aureus appeared. In 1990, 15 percent of all S. aureus isolates in the United States were resistant to methicillin, and in critical care units the frequency was often higher (Wenzel et al., 1991). These resistant organisms are just as virulent as their methicillin-susceptible counterparts and can cause life-threatening infections.
Although S. aureus is a frequent cause of infection associated with medical devices (artificial heart valves, joint replacements and other prosthetic devices, and venous catheters), the coagulase-negative staphylococci have become the most common cause of these infections in the past decade (Mandell et al., 1990). Most of these organisms make beta-lactamase, and 40 percent are resistant to methicillin and other beta-lactams, making vancomycin almost the sole effective agent for treatment (Jacoby, 1991).
Although a few vancomycin-resistant, coagulase-negative staphylococcal isolates have been reported in Europe and the United States, vancomycin-resistant enterococci may represent an even greater threat as an emerging nosocomial pathogen. Outbreaks have been reported in several U.S. cities within the past two years. Some strains (also resistant to ampicillin, gentamicin, and teicoplanin) are resistant to all currently licensed antibiotics that are recommended for treatment of serious enterococcal infections. This finding is particularly worrisome, since these organisms are becoming a major cause of nosocomial infections in this country.
Pseudomonas aeruginosa
Pseudomonas aeruginosa is an important cause of nosocomial infections, especially in the immunocompromised patient (Schaberg et al., 1991). This organism has an outer cell membrane that can exclude various antibiotics, and it has many inactivating and modifying enzymes to counter aminoglycosides, beta-lactams, and other antimicrobial agents. Although P. aeruginosa is frequently susceptible to ceftazidime, resistance can arise by
mutations that result in the overproduction of its chromosomal beta-lactamase. Resistance can also emerge to imipenem and other carbapenems through production of a plasmid-mediated beta-lactamase (Watanabe et al., 1991) or by mutational loss of an outer membrane protein that is the major channel for imipenem entry (Trias and Nikaido, 1990). Other mutations affecting drug uptake confer resistance to virtually all aminoglycosides (Maloney et al., 1989), whereas resistance to fluoroquinolones can evolve through mutations that affect drug accumulation or DNA gyrase (Robillard and Scarpa, 1988).
Mycobacterium tuberculosis
After a decline in rates of infection over several decades, the United States is experiencing a disturbing increase in tuberculosis (TB). Compared with the incidence of many other diseases, 28,000 excess U.S. cases of TB in a seven-year period seems a relatively low figure (Fox, 1992). Because TB is highly contagious, however, it poses a profound threat to public health. TB bacteria are easily passed from person to person in airborne droplets formed when a person with active TB sneezes or coughs.
Even more alarming has been the rise of multidrug-resistant tuberculosis (MDRTB). Prior to 1984, about 10 percent of TB bacteria isolated from patients in the United States were resistant to even a single antibacterial drug. In 1984, 52 percent of patients were infected with tubercle bacilli resistant to at least one drug, and 32 percent were resistant to one or more drugs (Marwick, 1992). Outbreaks of MDRTB have been reported in 13 states. Ten percent of the recorded MDRTB cases to date have occurred in previously healthy people whose mortality rate—70 to 90 percent—has been nearly the same as that of immunosuppressed persons with MDRTB (Snider and Roper, 1992).
The CDC has released preliminary results of a joint study with the New York State Health Department showing that cases of drug-resistant TB have more than doubled since 1984 (Goodstein, 1991). CDC data from the first quarter of 1991 show that many of these drug-resistant strains are resistant to both of the frontline TB drugs, rifampin and isoniazid (Centers for Disease Control, unpublished data, 1992). Outbreaks of MDRTB have occurred in hospitals in Miami and New York City, as well as in the New York State prison system. In one hospital in New York City, the median interval between diagnosis of MDRTB and death was only four weeks. Additional clusters of MDRTB were reported to the CDC in 1990 and 1991 from Mississippi, Missouri, and Michigan (Centers for Disease Control, 1992g).
There are five frontline drugs known to be highly effective against Mycobacterium tuberculosis and five second-line drugs that can be used when
resistance to one or more of the frontline drugs is detected. Ironically, in the United States, until April 1992, there were shortages of antituberculosis drugs, some of which are crucially needed when resistance to the frontline drugs rifampin and isoniazid is present (Centers for Disease Control, 1991j; Marwick, 1992). These shortages had occurred because several pharmaceutical companies had ceased production of these drugs.
Because of its persistence in the body, the tubercle bacillus is a notoriously difficult pathogen to control. Although bacille Calmette-Guerin (BCG) vaccine protects against severe tuberculous meningitis and disseminated TB in children (Benenson, 1990), its efficacy against pulmonary TB in adults has varied widely in different parts of the world. Treatment of conventional TB is effective, but expensive, requiring daily treatment with multiple drugs for a minimum of six months. There is a universal tendency among TB patients to stop taking their drugs when the drugs begin to have their beneficial effect or to take the medications only intermittently. When this happens, relapses are frequent and very often are caused by drug-resistant tubercle bacilli that have survived the initial course of treatment. The emergence of drug-resistant M. tuberculosis is in many ways an index of individual compliance with antituberculosis chemotherapy and of the inability of the health care infrastructure to ensure adequate treatment. Many public health agencies that once could play key roles in this process have had their budgets cut drastically in recent years and hence are unable to perform this crucial service.
MDRTB is extraordinarily difficult to treat, and a majority of patients do not respond to therapy. Total treatment costs for an individual with MDRTB can be as much as $150,000, ten times the cost of traditional treatment; the cost of the treatment drugs alone can be as much as 21 times as great (see Table 2-5). In an outbreak of MDRTB in 1990 in Forth Worth, Texas, the cost of treating 10 patients was $950,433. The budget available that year to the Fort Worth/Tarrant County, Texas, Tuberculosis Control Program was less than one-fifth that amount (Centers for Disease Control, 1990b).
The preferred treatment for classical TB consists of isoniazid, rifampin, and pyrazinamide. For patients whose tubercle bacilli are thought to be resistant to isoniazid, a fourth drug, ethambutol, should be added to the regimen until drug susceptibility results are known. Isolates of tubercle bacilli resistant to both isoniazid and rifampin, now representing about 20 percent in some cities, require specialized treatment with additional medications, which may include streptomycin and ciprofloxacin for almost two years.
The tubercle bacillus is a slow-growing organism. Three to six weeks are needed to grow the bacteria in the clinical laboratory, and an additional three to six weeks are needed to screen for antibiotic resistance. Such extended
TABLE 2-5 Drug Costs per Course of Treatment for One Individual with either Susceptible or Varying Levels of Drug-Resistant Tuberculosis
laboratory procedures can result in a delay in diagnosis, which means that patients with unrecognized drug-resistant TB may be treated ineffectively and remain infectious for a longer period. In HIV-positive individuals, MDRTB usually causes death within 4 to 16 weeks after being diagnosed, which is often before laboratory tests on drug susceptibility and resistance can be completed.
There is no evidence that mutation rates in M. tuberculosis organisms have increased or that increased virulence is to blame for the recent deadly outbreaks of TB (Weiss, 1992). It is likely that drug-resistant forms of tuberculosis arose because of patient noncompliance with the 6- to 12-month regimen of antibiotics required to treat TB. Ineffective treatment regimens also play a role in the rising incidence of TB. To address noncompliance, some states with high TB rates are considering approaches to outreach, such as expanding directly observed therapy (DOT;) others may reestablish inpatient facilities similar to the TB sanatoria of the first half of this century. Standard treatment regimens for TB have also been updated. Instead of taking two or three antibiotics, TB patients now take four. Still, as noted earlier, the current shortages of antituberculosis drugs in the United States have made even standard treatment difficult.
RESISTANCE TO ANTIVIRALS
Some viral infections can be successfully controlled with currently available antiviral drugs. Unfortunately, as has been the case for antibiotics, resistance to antiviral drugs has been reported (see Table 2-6). Two examples of resistance are discussed below. Potentially effective new drugs undergoing preclinical and clinical testing may replace some antivirals that have been rendered unusable as a result of excessive patterns of resistance.
Despite the efforts of researchers to discover new, effective antiviral drugs, very few ever reach the point at which they become available to those who need them. Thousands of compounds may be screened before a single candidate with desirable antiviral properties and acceptable tolerance is found. Drugs that are potentially useful against viral infections fit into three categories: those that inactivate viruses (virucides); those that inhibit the replication of viruses within their host cells (antivirals); and those that work indirectly by augmenting or modifying the host's immune response to viral invasion (immunomodulators) (Hayden and Douglas, 1990). There are no clinically practical virucidal drugs at this time, since those currently available are toxic to host cells as well as viruses. Unlike those antibiotics that are bactericidal and can rid the patient of the organism, current antivirals only suppress viral replication. Ultimately, control of the viral infection relies on the individual's immune response.
TABLE 2-6 Antiviral Drugs for Which Resistance Has Been Demonstrated
|
Antiviral Agent |
Antiviral Spectrum |
|
Acyclovir |
Herpes simplex virus |
|
|
Varicella-zoster virus |
|
Amantadine/rimantidine |
Influenza A |
|
Dideoxycytidine |
Human immunodeficiency virus-1 |
|
Dideoxyinosine |
Human immunodeficiency virus-1 |
|
Foscarnet |
Cytomegalovirus |
|
|
Herpes simplex virus |
|
Ganciclovir |
Cytomegalovirus |
|
|
Herpes simplex virus |
|
Idoxuridine (topical) |
Herpes simplex virus |
|
Trifluridine (topical) |
Herpes simplex virusa |
|
Vidarabine |
Herpes simplex virusa |
|
Zidovudine |
Human immunodeficiency virus-1 |
|
a Resistance to these drugs can be demonstrated in the laboratory but has not yet been documented in clinical isolates. SOURCE: F. Hayden, Associate Professor of Internal Medicine and Pathology, Department of Internal Medicine, University of Virginia Hospital. |
|
Antiviral drugs can interfere with viral invasion and replication at several specific points. For example, the drug can prevent the attachment of the virus to the host cell, or it may interfere with the assembly of new viruses within the host cell. Although many compounds that have antiviral activity exist or can be synthesized, most cannot be used because of toxicity, because they adversely affect a host cell function, or because they fail to reach concentrations required for antiviral activity in humans (Hayden and Douglas, 1990).
The emergence of antiviral resistance is not well understood. Primary resistance, which exists even before first exposure to a drug, is uncommon, although virus populations appear to consist of heterogenous mixtures of mutants with varying drug sensitivities. Emergence of resistant subpopulations, or de novo mutations creating drug resistance, occur because of selective drug pressure. The frequency and speed at which this occurs varies among virus-drug combinations and is heavily influenced by the type of infection and nature of the host. The usefulness of drug sensitivity assays in such situations is limited. The clinical significance of in vitro resistance is often unclear, a factor that complicates therapeutic decisions.
Antiviral resistance is linked to critical amino acid changes in viral proteins that are targets of drug action or responsible for drug metabolism. Individuals who are immunocompromised, with chronic or recurrent viral infections (particularly herpesvirus infections), often develop drug-resistant viruses. Because resistance to antiviral drugs appears to occur quite rapidly in such individuals, appropriate use and availability of drugs with alternative mechanisms of action are important. Sufficient data are not yet available, however, to recommend limitations on the use of antiviral drugs.
Acyclovir and Herpes Simplex Virus
Acyclovir, an antiviral that inhibits viral DNA synthesis, is the agent of choice for managing infections with herpes simplex virus (HSV). Resistance to acyclovir was a relatively uncommon event prior to the onset of the HIV disease pandemic. Previously, occasional resistance to acyclovir was seen in cancer patients or transplant recipients who were receiving treatment for HSV infection. Acyclovir-resistant HSV in HIV-infected individuals with advanced disease can cause extensive ulcerative lesions that may lead to progressively more serious HSV disease (Laughlin et al., 1991).
The occurrence of resistance does not appear to be linked to dosage or to treatment schedule. Even initial exposure to acyclovir can induce resistance. Most commonly, resistance is caused by a mutation in the virus that results in an inability to produce thymidine kinase, the enzyme toward which acyclovir is targeted. Foscarnet, an antiviral drug that inhibits viral DNA polymerase, is used in the treatment of acyclovir-resistant HSV (Laughlin et
al., 1991). Currently, there is no evidence of person-to-person spread of this drug-resistant virus.
Zidovudine and Human Immunodeficiency Virus-1
Zidovudine, also known as azidothymidine (AZT), is effective in prolonging the survival and improving the quality of life of those infected with HIV-1, including those with AIDS, those with symptomatic HIV infection, and HIV-infected asymptomatic individuals. Zidovudine works by inhibiting the viral enzyme reverse transcriptase (RNA-dependent DNA polymerase) (Hayden and Douglas, 1990).
Unfortunately, resistance to the drug has been documented and appears to be associated with length of treatment (most isolates demonstrate reduced drug susceptibility after six months of treatment) and CD4-lymphocyte counts of less than 200 per cubic milliliter (Laughlin et al., 1991). Resistance develops gradually as the result of point mutations in the gene for reverse transcriptase; multiple mutations are necessary to confer high-level resistance (Laughlin et al., 1991).
RESISTANCE TO ANTIMALARIALS
Over the past 15 years, there has been a tremendous resurgence of malaria around the world and an increase in the percentage of cases caused by Plasmodium falciparum, the most virulent of the four human malaria parasites (Institute of Medicine, 1991a). Worldwide, as many as 2 million people will die from malaria in 1992, most of them children in Africa.
The seriousness of the worldwide increase in malaria incidence is heightened by the spread of parasites resistant to the available assortment of antimalarial drugs. Malaria parasites, like bacteria and viruses, can develop resistance to the drugs used to prevent or treat infection. And, as is the case with antibiotics, resistance is often the result of overuse or misuse of antimalarial drugs. However, unlike the case for antibiotics, the development of antimalarial drugs is not a high priority for U.S. pharmaceutical companies. Within the United States, this task is almost entirely the domain of the Walter Reed Army Institute of Research. Globally, the Special Programme for Research and Training in Tropical Diseases, a joint project of the United Nations Development Programme, the World Bank, and the World Health Organization, is an important participant in antimalarial drug development.
Resistance to quinine, the first antimalarial drug ever marketed, was initially reported in 1910 in Germans who developed malaria while working in Brazil (Peters, 1987). Since then, a number of other antimalarials have been developed, the most notable of which is chloroquine.
Resistance to chloroquine in P. falciparum was first reported in 1961 in
two Colombian workers (Young and Moore, 1961) and has been partly to blame for the resurgence of malaria. Soon after this account, similar reports were received from Thailand, Vietnam, Cambodia, Malaya, and Brazil. By 1970, nearly 20 percent of all malarious regions had recorded cases of chloroquine-resistant P. falciparum malaria (Lepes, 1981). Today, there are only a few areas in the world where chloroquine is effective against this parasite.
Resistance to the newer antimalarials, such as mefloquine, is also occurring. In Thailand, for example, there is evidence of considerable P. falciparum resistance to the drug (Peters, 1990; Institute of Medicine, 1991a). This is of great concern, both because this drug has not been on the market for long (demonstrating the rapidity with which these parasites can adapt to the presence of the drug) and because the development of new antimalarial drugs to which the parasites are susceptible is not a rapid process; mefloquine was originally synthesized in the late 1960s and underwent 17 years of testing to demonstrate efficacy before being licensed (Institute of Medicine, 1991a).
Although P. falciparum causes the most severe type of malaria and is increasing in prevalence, P. vivax is responsible for the majority of malaria morbidity worldwide. It is of considerable concern, therefore, that P. vivax has also developed resistance to chloroquine, which was first reported in 1989 in Papua New Guinea (Rieckmann et al., 1989). Chloroquine-resistant P. vivax malaria has also been diagnosed in Indonesia (Schwartz et al., 1991). No one knows for certain how quickly and how far this resistance will spread. Because of the problem of resistance to chloroquine, travelers should seek the latest guidance on prophylaxis before going into malariaendemic areas.
Given the distribution of malaria cases throughout the world, drug resistance is of much greater concern outside the United States than within it. Still, the potential for the reemergence of malaria in this country, and the role of drug resistance in such a scenario, cannot be overlooked. In the United States, as recently as the early 1900s, up to 500,000 cases of malaria occurred each year, most of them in the South (Institute of Medicine, 1991a). Currently, some 1,200 total cases of malaria both drug susceptible and drug resistant are reported each year in this country, almost all of which occur in individuals who have been infected in other parts of the world (socalled imported malaria). Small outbreaks of nonimported malaria, the result of mosquito transmission from imported cases, have also been reported (Maldonado et al., 1990; Centers for Disease Control, 1990c, 1991f). Thus far, the outbreaks have been quickly and easily contained. A continued increase in drug-resistant malaria throughout the world, however, may increase the number of cases of imported malaria, thereby improving the chances for malaria to regain a foothold in the United States.
VECTOR RESISTANCE TO PESTICIDES
Agriculture accounts for about 75 percent of the pesticides used in the United States. In 1987, approximately 407,000 tons of pesticides were applied in agricultural settings throughout the nation, of which about 89,500 tons were insecticides. About 10 percent of the pesticides used worldwide are applied for public health purposes, mainly to control malaria, filariasis, schistosomiasis, onchocerciasis, and trypanosomiasis (Moses, 1992).
This high volume of pesticide use for agricultural purposes has contributed to the development of resistance in infectious disease vectors, particularly mosquitoes. Public health use of insecticides has also played a role in the emergence of resistance, although not in the United States, where public health use of insecticides is not sufficiently regular to elicit the development of resistance.
Resistance, in the field, to a number of pesticides belonging to organochlorine, organophosphate, and other insecticide groups has developed in vector arthropods. In addition, recent evidence from laboratory studies points to the emergence of resistance in mosquito larvae to the delta endotoxins of the commercialized microbial control agents Bacillus thuringiensis israeliensis (Georghiou, 1990) and B. sphaericus (Rodcharoen and Mulla, in press). Although resistance to these agents has not yet been demonstrated in a field situation, the laboratory finding illustrates the strong potential for the development of such resistance.
New Understandings: Microbes as Cofactors in Chronic Disease
Although medical science has been able to discern the causes of many diseases, the etiology of some that have a significant impact on the health of the U.S. population is still speculative, even after decades or more of research. The recognition that an ''old" disease, with heretofore unknown causes, is associated with an infectious agent is one of the more interesting ways infectious diseases emerge. A number of diseases are now thought to be caused by microbial infection or to involve microbes as cofactors in pathogenesis. These include, but are not limited to, atherosclerosis, rheumatoid arthritis, insulin-dependent diabetes mellitus, Reye's syndrome, Kawasaki disease, systemic lupus erythematosis, and Alzheimer's disease. The final chapters on these diseases have not been written; it remains to be seen which, if any, will be determined with certainty to involve microbial agents. The examples below illustrate the relationships between some of these diseases and the infectious agents associated with them.
HUMAN T-CELL LEUKEMIA VIRUS TYPES I AND II
Isolation of the first pathogenic human retrovirus was reported in 1980 (Poiesz et al., 1980) and was soon followed by the isolation of a second, closely related but distinct virus (Kalyanaraman et al., 1982). These viruses subsequently became known as human T-cell leukemia virus types I and II (HTLV-I, HTLV-II); they are sometimes referred to as human T-lymphotropic viruses. Multiple isolates of the HTLVs have now been obtained, novel regulatory properties identified, and areas of endemicity throughout the world defined by seroepidemiological studies.
Although HTLV-II has not been linked definitively to a specific disease, HTLV-I has been shown to be etiologically associated with two very different diseases. The first, adult T-cell leukemia/lymphoma (ATLL), for which the virus is named, was first described in southern Japan (Uchiyama et al., 1977), where the virus is endemic. ATLL is a malignancy primarily of CD4+ (T-helper/inducer) lymphocytes, and in the leukemic phase, the HTLV-I provirus is monoclonally integrated into the DNA of neoplastic cells. The onset of disease occurs many years after the initial infection, and the most severe forms of the disease are characterized by generalized lymphadenopathy, visceral and cutaneous involvement, and bone lesions (Kuefler and Bunn, 1986). Effective treatment is not available, and death usually occurs within one year of diagnosis. Fortunately, only 2 to 5 percent of HTLV-I-infected persons develop ATLL (Murphy et al., 1989).
The second HTLV-I-associated disease is a neurological condition called tropical spastic paraparesis (TSP), which was first described in the Caribbean (Gessain et al., 1985). The same syndrome was subsequently described by doctors in Japan, who called it HTLV-I-associated myelopathy (HAM) (Osame et al., 1986). This disease, which is now usually referred to as TSP/HAM, begins with difficulties in walking and weakness and spasticity in the legs; it can also include back pain, sensory disturbances, urinary incontinence, and impotence in men. Disability progresses over several years, and eventually victims may become confined to a wheelchair. Because afflicted individuals have high concentrations of HTLV-I-specific antibodies in serum and spinal fluid (Osame et al., 1990), sometimes in association with an human leukocyte antigen (HLA)-linked high immune responsiveness to HTLV-I (Usuku et al., 1988), some investigators feel that TSP/HAM is an immunological disease triggered by the virus. As with ATLL, TSP/HAM occurs in a small percentage (about 1 percent) of HTLV-I-infected persons (Kaplan et al., 1990).
Interest in HTLV-I increased with the in vitro observation of several investigators that the efficiency of replication of HIV, the virus that causes
HIV disease and AIDS, increased in cells transformed by HTLV-I. The subsequent identification of persons in endemic areas, many with AIDS, who were infected with both HIV and HTLV-I (Cortes et al., 1989; Hattori et al., 1989) led to the speculation that interaction of these two viruses may potentiate disease progression. The prevalence of infection with both HIV and HTLV-I or HTLV-II is increasing in populations of intravenous drug abusers in the United States (Lee et al., 1991). This increase offers the possibility of a study group, albeit an unwanted one, in which to assess this additional risk for disease.
HTLV-I has also been implicated as a factor in other disease syndromes, including polymyositis, arthritis, infective dermatitis, mycosis fungoides, and multiple sclerosis (although the latter is controversial). In addition, HTLV-II was recently linked to chronic fatigue syndrome (DeFreitas et al., 1991). It is probable that the full spectrum of diseases and immunological abnormalities associated with the human retroviruses has yet to be delineated. It is also probable that additional human retroviruses exist but have not yet been discovered.
ATHEROSCLEROSIS
Atherosclerosis, commonly known as hardening of the arteries, is the result of an uncontrolled proliferation of arterial smooth muscle cells, which eventually can block the flow of blood through the vessel. This disease is the underlying cause of strokes and myocardial infarctions and results in more deaths in the United States (and in other industrialized countries) than any other single disease. The burden on the U.S. health care system is estimated to be in excess of $60 billion per year (Levi and Moskovitz, 1982; Kannel et al., 1984).
Although it is well known that smoking, high cholesterol levels, and elevated blood pressure are major risk factors for atherosclerosis, viruses can generate the pathologic events—cell destruction, metabolic changes within cells, and cell transformation—that precede the appearance of atherosclerotic lesions. This observation has led some researchers to conclude that a virus or viruses may play some role in the disease. Supporting this theory are reports that chickens can develop atherosclerotic lesions as a result of infection with an avian herpesvirus (Fabricant et al., 1978, 1980; Minick et al., 1979). In humans, two similar viruses, herpes simplex virus (HSV-1 and HSV-2) and cytomegalovirus (CMV), infect infants and young children worldwide. During infection, the viruses are often found in the blood vessels, potentially exposing the smooth muscle cells to their effects. In one recent study, CMV infection was demonstrated in patients with atherosclerosis; there was no evidence of either HSV-1 or HSV-2 infection in the same individuals (Melnick et al., 1990). Other studies have implicated
CMV, as well as HSV-1 and HSV-2 in human atherosclerotic disease (Benditt et al., 1983; Yamashiroya et al., 1988; Hendrix et al., 1989, 1990).
Recently, a bacterium, Chlamydia pneumoniae, was reported to play a potential role in the pathogenesis of atherosclerotic disease. The investigators examined the consequences of human infection with C. pneumoniae and found evidence, in the form of persistently elevated levels of anti-C. pneumoniae antibodies and immune complexes containing chlamydial lipopolysaccharide, that chronic infection with this bacterium was associated with increased risk for coronary heart disease. This risk was shown to be independent of those factors—age, smoking, total cholesterol to high-density-lipoprotein-cholesterol ratios, and hypertension—most often associated with atherosclerosis (Saikku et al., 1992). Because C. pneumoniae infection is fairly common and can be treated with antibiotics, a proven association of this organism with atherosclerosis and subsequent myocardial infarction could have a significant public health impact.
HUMAN PAPILLOMAVIRUS
Papillomaviruses were described and associated with disease in the early to mid-1900s. Research on these viruses, however, suffered from the inability to grow them in cells in the laboratory. It was not until the 1980s that techniques to identify and characterize human papillomavirus (HPV) became readily available (deVilliers, 1989). In the past 10 years, more than 67 HPV types have been defined; the molecular biology of the virus has been developed in exquisite detail; and knowledge concerning molecular mechanisms of infection and disease has emerged (Reeves et al., 1989).
Epidemiological studies have shown that HPV infection (with HPV types 16 and 18) is the major risk factor for cervical cancer (Reeves et al., 1989), an important public health problem in the United States and the developing world. The American Cancer Society estimated that 13,000 new cases would occur in the United States in 1991, resulting in about 4,500 deaths (American Cancer Society, 1991). Most of these cases and deaths could have been prevented by appropriate screening and control programs. Cervical cancer is uniquely amenable to secondary prevention by screening and early treatment since it evolves through surgically curable premalignant stages to invasive disease over a 10- to 20-year period (Tabbara et al., 1992).
Other factors besides HPV infection, however, play a role in the development of cervical cancer. For example, a woman's risk of cervical cancer is directly related to the number of sexual partners she has had (the greater the number of partners the greater her risk) and inversely related to age at first intercourse (the younger her age, the greater the risk). It has only recently been recognized that male sexual behavior also influences cervical cancer risk. Spouses of women with cervical cancer are more likely to
exhibit high-risk sexual behavior than spouses of women without the disease (Brinton et al., 1989). These behaviors undoubtedly increase the chance that the men will be exposed to sexually transmitted agents such as HPV. There is growing evidence that other venereal disease agents, such as HSV-2 and HIV, interact with HPV in a multiplicative fashion to boost the risks for cervical cancer (Hildesheim et al., 1991).
Women infected with HIV have at least a 10-fold increased risk for active HPV infection and a 12-fold increase in risk of cervical neoplasia (Laga et al., 1992). HPV infection and cervical disease also progress more rapidly and are more refractory to treatment in women with HIV infection. As the prevalence of heterosexually transmitted HIV increases among women, HPV infection and cervical disease will continue to emerge as major opportunistic complications. The strong association between HIV and HPV infection may involve interactions between the proteins of HPV and HIV, in addition to general immunosuppression.
For all of the apparent links between HPV infection and cervical disease, the relationship is not a simple one of cause and effect. For example, no study has found HPV in all cervical cancers, and a variety of studies have shown that 10 to 50 percent of healthy women are infected with HPV (Burk et al., 1986; Cox et al., 1986; Toorn et al., 1986; and Reeves et al., 1987). HPV infection can only be estimated by detecting viral DNA, and there is no single accepted criterion for judging the results of such testing. In addition, there is no clinically useful serological assay that can independently estimate past HPV infection. Well-standardized commercial kits can be used to diagnose HPV infection, but infection alone is neither sufficient nor necessary for cervical cancer. The condition appears to be brought on by a complex interaction of infections and demographic, behavioral, and hormonal risk factors. The only currently efficacious cervical cancer control strategy is secondary prevention—detecting and ablating preinvasive cervical intraepithelial neoplasia (CIN) lesions. New strategies based on HPV detection and Pap smear screening may be possible.
BREAKDOWN OF PUBLIC HEALTH MEASURES
The control of many infectious illnesses has occurred as societies themselves have become more advanced. Improvements in medicine, science, and public health have come only as a result of the complex growth and maturation of modern civilization. Many of the protections now in place, such as vaccination, proper hygiene, water and sewage treatment, and safe food-handling and distribution practices have vastly improved our ability to control infectious disease outbreaks.
Despite the appearance of security, however, there is only a thin veneer protecting humankind from potentially devastating infectious disease epidemics.
Alone or in combination, economic collapse, war, and natural disasters, among other societal disruptions, have caused (and could again cause) the breakdown of public health measures and the emergence or reemergence of a number of deadly diseases.
Inadequate Sanitation: Cholera
Cholera, a sometimes rapidly fatal diarrheal disease caused by the bacterium Vibrio cholerae, reached epidemic levels in South America in January 1991 for the first time in almost a century. Inadequate sanitation played a role in its reappearance, which occurred initially in several coastal cities in Peru; then the disease spread through much of the continent. A scattering of epidemic-related cases were reported later in the year in Central America and the United States. As of December 1991, there had been 366,056 reported cases of the disease and 3,894 deaths in these three regions (Pan American Health Organization, 1991).
It is believed that V. cholerae was first introduced into the harbor at Lima, Peru, through the dumping of bilge water by a ship arriving from the Far East. Once in the water, the bacteria rapidly contaminated the fish and shellfish, which were then consumed (often in the form of ceviche, a dish made with raw seafood that is popular in that part of the world). Following the initial seafood-related cases in humans, the organisms are thought to have been spread by fecal contamination of the water supply.
Epidemiologic investigations in Peru have implicated such contaminated municipal water supplies as the principal means by which the disease is now being transmitted. Based on a study by the U.S. Environmental Protection Agency (EPA) that showed a possible link between chlorination and cancer, Peruvian officials apparently ceased treating much of the country's drinking water in the early 1980s (C. Anderson, 1991). A paucity of hygienic food preparation practices also appears to have played a role.
The epidemic is traveling northward at a rapid rate; several cases have already been reported in the United States, originating from contaminated foodstuffs. U.S. public health authorities are maintaining a close watch on foods imported from South and Central America (the majority of fresh fruits and vegetables imported into the United States during the winter come from Mexico). In February 1992, at least 31 of 356 passengers and crew aboard a flight from Buenos Aires, Argentina, to Los Angeles, California (with a brief stopover in Lima) were diagnosed with cholera (18 in California, 9 in Nevada, 3 in Japan, and 1 in Argentina—1 person died). At least 54 other passengers reported having diarrhea of unknown etiology. This illustration demonstrates the ease with which this disease can be transported worldwide (Centers for Disease Control, 1992c).
Cholera can almost always be treated successfully with oral rehydration therapy, which replaces fluids and essential salts lost in diarrheal stools. Disease surveillance and maintenance of a clean water supply are the most effective methods of preventing disease spread. Currently available vaccines are of limited effectiveness.
Extensive transmission of cholera and other waterborne infectious diseases in the United States is highly unlikely, owing to the generally high standards required of U.S. municipal water and sewage treatment facilities. Outbreaks could occur, however, in areas with substandard water supplies and inadequate sewage disposal. Occasional cholera outbreaks in the United States are most often caused by fecal contamination of estuarine waters. Inadequately treated sewage can be taken up by the fin fish and shellfish harvested from these waters; their consumption can infect humans if the fish are prepared improperly. As coastal areas become increasingly crowded and water treatment facilities are overwhelmed or poorly maintained, a rise in cases of Vibrio infection is likely.
Complacency
There can be a delicate balance between maintaining control of a disease and the initiation of an epidemic. It is one thing to have this balance disrupted by essentially uncontrollable elements; it is quite another to have it go awry as a result of individual or organizational complacency.
Sometime in the 1950s, the attention given to acute infectious diseases by public health and medical officials, physicians, researchers, and others began to wane, and a shift in focus to chronic, degenerative diseases occurred. Much of the reason for this shift was the notion that infectious disease problems were becoming a thing of the past—science, medicine, public health, and an improved standard of living had brought most of these diseases under control. People were living longer and developing more chronic illnesses as a result. The emergence of HIV disease and AIDS swung the pendulum back to infectious diseases. HIV disease and the host of opportunistic infections that accompany it have severely challenged the scientific, medical, and public health communities, as well as politicians. Infectious diseases clearly are not a problem of the past.
Measles is highlighted here as an example of complacency regarding the control of infectious diseases. Tuberculosis is yet another example that has already been discussed in other sections of this report. There are certainly other diseases, however (including typhoid, diphtheria, whooping cough, tetanus, and louse-borne typhus) that may pose significant threats to health when complacency sets in.
INADEQUATE LEVELS OF IMMUNIZATION: MEASLES
Immunization against infectious diseases is one of the most effective ways of improving overall public health. Medical researchers have developed dozens of vaccines, a number of which have been incorporated into childhood vaccination programs. These programs have greatly reduced the health-related and financial impacts of a number of previously devastating diseases. However, if such programs falter or are carried out incompletely, diseases that are now relatively rare can reemerge as the killers they once were. This phenomenon is a danger to all countries, particularly those with poor or inactive immunization programs (see Table 2-7).
The incidence of measles, a highly communicable viral disease, declined rapidly in the United States after the introduction of an effective vaccine in 1963. There were some 500,000 reported cases each year during the 1950s, a number that had dropped to less than 2,000 by the early 1980s. In 1989, however, the number of measles cases began to climb, and by 1990, more than 26,000 cases were reported, the largest number since 1977 (Atkinson and Markowitz, 1991). What happened?
TABLE 2-7 Percentage of 1-Year-Olds Immunized Against Measles in the Americas in 1990
|
Country |
Percentage |
Country |
Percentage |
|
Panama |
99 |
Belize |
81 |
|
Anguilla |
99 |
Nicaragua |
81 |
|
British Virgin Islands |
99 |
Turks and Caicos |
81 |
|
Montserrat |
99 |
Brazil |
77 |
|
St. Kitts/Nevis |
99 |
Paraguay |
77 |
|
Chile |
98 |
El Salvador |
75 |
|
Dominican Republic |
96 |
Jamaica |
74 |
|
Argentina |
94 |
Suriname |
74 |
|
Cuba |
94 |
United Statesa |
70 |
|
Honduras |
91 |
Guatemala |
68 |
|
Antigua |
89 |
Mexico |
66 |
|
Barbados |
87 |
Peru |
64 |
|
Bahamas |
86 |
Venezuela |
64 |
|
Costa Rica |
85 |
Grenada |
63 |
|
Colombia |
82 |
Ecuador |
62 |
|
St. Lucia |
82 |
Bolivia |
53 |
|
Uruguay |
82 |
Haiti |
31 |
|
a Data are for 2-year-olds. SOURCE: Bernier, 1991. |
|||
Measles vaccine induces immunity in more than 95 percent of individuals over one year of age (R. M. Davis et al., 1987). The requirement that school-age children in the United States present evidence of measles vaccination in order to be admitted to school was largely responsible for the dramatic reduction in the incidence of measles in the 1960s, 1970s, and early 1980s. Vaccination levels in preschool-age children, however, were lower than for those attending school (Orenstein et al., 1990; Atkinson and Markowitz, 1991).
In 1990, there were large outbreaks of measles in Dallas, San Diego, and Los Angeles; each city reported more than 1,000 cases. In New York City, an outbreak that started in March 1990 continued through 1991, with more than 2,000 cases reported in the first five months of 1991. Epidemiologic data from the New York outbreak revealed that a majority of cases were among preschool-age black and Hispanic children who had not been immunized. Vaccination goals in this population were not being met (Centers for Disease Control, 1991d).
There have also been additional outbreaks among vaccinated school-age groups, indicating that in some cases the one-time vaccination was insufficient to prevent disease. In response, the American Academy of Family Physicians, the American Academy of Pediatrics, and the Immunization Practices Advisory Committee (ACIP; a committee of experts convened by the CDC) recommended, in 1989, that a second dose of measles vaccine (as part of the trivalent measles, mumps, and rubella vaccine) be given (Atkinson, 1991). In early 1991, the National Vaccine Advisory Committee issued recommendations to improve the availability of childhood vaccines and urged a two-dose schedule for the measles-mumps-rubella (MMR) immunization (National Vaccine Advisory Committee, 1991).
Available data for 1991 indicate that the trend of rising measles incidence is reversing itself. As of November 1991, there were 60 percent fewer cases than had been reported during the same period in 1990 (Cotton, 1991). Although efforts have been made over the past several years to reduce the number of people, particularly children, who have not been vaccinated against measles, the present decline in cases is too great to be attributable to better vaccine coverage alone (Cotton, 1991). At this time, however, a full explanation is not available.
War
When deployed outside of this country, U.S. military forces are at high risk of being exposed to a variety of infectious disease agents. In past conflicts, infectious diseases have produced higher hospital admission rates among U.S. troops and, until World War II, higher mortality rates, than battle injuries (Gordon, 1958; Reister, N.d.; Washington Headquarters Service
Directorate for Information, Operations, and Reports, 1985). Table 2-8 shows the types of infectious diseases that have accounted for the greatest morbidity among American soldiers.
The relative importance to deployed forces of any particular infectious disease depends on a number of environmental factors and operational circumstances, including, principally, the geographic area of deployment, the time of year, the mission and composition of the force, and the intensity of the conflict. For example, military operations generally result in large numbers of susceptible individuals living in close proximity, circumstances that can facilitate the transmission of respiratory diseases. Under field conditions, food and water sanitation services may be rudimentary and subject to disruptions, opportunities to exercise good personal hygiene are diminished, and there may be exposure to the bites of infected arthropod vectors of both human and zoonotic diseases. In addition, warfare usually creates some degree of social disruption, which can produce refugee populations that are frequently subject to epidemics of infectious disease. Troops may be at added risk of infection to the extent that they become involved in the supervision and care of the refugees. Because troops are frequently living under relatively primitive field conditions, there is also the potential for accidental transmission of previously unknown zoonotic diseases. The return of
TABLE 2-8 Infectious Diseases Causing High Morbidity in U.S. Forces in Past Conflicts: World War II, Korea, Vietnam, and Operation Desert Storm
|
Disease Category |
Conflict |
|
Acute respiratory diseases and influenza |
All |
|
Acute diarrheal diseases |
All |
|
Malaria |
WWII, Korea, Vietnam |
|
Hepatitis |
WWII, Korea, Vietnam |
|
Sexually transmitted diseases |
WWII, Korea, Vietnam |
|
Arthropod-borne diseasesa |
WWII, Vietnam |
|
Rickettsial diseasesb |
WWII, Vietnam |
|
Leptospirosis |
WWII, Vietnam |
|
Leishmaniasis |
WWII, Desert Storm |
|
Schistosomiasis |
WWIIc |
|
NOTE: WWII=World War II. a Especially dengue fever, sandfly fever, hemorrhagic fevers, and encephalitides. b Principally scrub typhus, whose distribution is limited to parts of Asia and northern Australia. c Principally in engineer bridge-building units in Luzon, Philippines. SOURCE: L. J. Legters, Department of Preventive Medicine, Division of Tropical Public Health, Uniformed Services University of the Health Sciences, Bethesda, Maryland. |
|
U.S. troops from foreign soils offers a unique opportunity for the introduction or reintroduction of infectious diseases into the United States.
The following examples describe infectious diseases that have emerged in association with military operations, some of which involved U.S. Troops.
-
Epidemic typhus, caused by Rickettsia prowazekii, has been a frequent accompaniment of warfare in Europe, dating back to the siege of Naples by the French in 1528 (Zinsser, 1935). The disease, which is acquired by contact with the feces of infected body lice, was a major problem in World War II.
-
Trench fever, caused by Rochalimaea quintana (Rickettsia quintana) , which are transmitted by body lice, made its first appearance in troops during World War I (Fuller, 1964).
-
Epidemic hemorrhagic fever, now known to be due to a zoonotic infection caused by Hantaan virus, was reported in Japanese and Soviet troops in Manchuria before the onset of World War II and was later (1951) recognized in United Nations troops in Korea (Benenson, 1990).
-
Scrub typhus, caused by a rickettsia transmitted by the bite of an infected larval mite of the genus Leptotrombidium, was a major medical problem (surpassed only by malaria in some areas) in the Pacific Theater in both World War II (Philip, 1948) and the Vietnam conflict (Berman et al., 1973).
-
A massive outbreak (involving 30,000 to 50,000 cases) of acute schistosomiasis, caused by Schistosoma japonicum, is reputed to have occurred in Chinese troops in 1950. The troops were being taught to swim in infected canals in southern China in preparation for what, because of the outbreak, became an aborted invasion of Taiwan in 1950 (Kiernan, 1959).
-
Leptospirosis, caused by Leptospira interrogans, was first identified as an important military disease in British troops during jungle operations against Communist terrorists in what was then Malaya in the late 1950s (U.S. Army Medical Research Unit, Malaya, 1962).
-
Leishmaniasis, due to Leishmania tropica, was known to be endemic in the Persian Gulf region before U.S. troops were committed in Operation Desert Shield. Known to cause cutaneous lesions, the capacity of L. tropica to ''visceralize" (i.e., invade bone marrow, liver, and spleen) was not well documented before its discovery in returning U.S. troops. The apparently atypical visceral expression of the disease may not be unusual at all but merely a natural consequence of the exposure of more than 500,000 susceptible troops to infected sand fly vectors in an endemic area. A total of 26 cases of leishmaniasis in U.S. troops—17 cutaneous and 9 visceral—had been diagnosed as of April 1992 (M. Grogl, Chief, Leishmania Section, Division of Experimental Therapeutics, Walter Reed Army Institute of Research, Washington, D.C., personal communication, 1992).















































































