5
Modeling the Population Consequences of Exposure to Multiple Stressors
INTRODUCTION
A conceptual model of the Population Consequences of Acoustic Disturbance (PCAD) was first developed by the National Research Council (NRC) (2005). A working group established by the U.S. Office of Naval Research in 2009 has formalized this model structure and extended it to cover all forms of disturbance. This Population Consequences of Disturbance (PCoD) model is described by New et al. (2014). It consists of a series of transfer functions that describe how
- exposure to stressors (such as noise) affects individual behavior,
- the resulting changes in behavior can affect health (defined as all internal factors that affect fitness or homeostasis),
- variations in health may affect individual vital rates (the probability of survival, giving birth, or growth/attaining sexual maturity for an individual), and
- data on the variation in the level of exposure to the stressor experienced by different individuals can be used to scale up the anticipated changes in vital rates so that they can be used to predict population-level effects.
As noted in Chapter 4, these transfer functions and their associated causal flows correspond to the first five levels of biological organization in the hierarchy of responses to a stressor illustrated in Figure 4.1. Approaches for assessing the effects of stressors on the two higher levels of biological organization (communities and ecosystems) are described in Chapter 6.
Full PCoD models have been developed for a number of marine mammal populations (Lusseau et al., 2012; Nabe-Nielsen et al., 2014; New et al., 2014; King et al., 2015). Ideally, the predictions of these models should be fitted to appropriate time series of empirical data obtained over a range of levels of disturbance, and the results of the fitting process used to improve the parameter estimates and quantify the uncertainty associated with the model predictions. Approaches such as Bayesian hidden-process modeling (Newman et al., 2006) may be appropriate for this purpose. However, in no case has this been possible, and such models should be considered “exploratory.” Exploratory models are most useful for comparing the possible consequences of different scenarios and for identifying priority areas for research. It is particularly important that the uncertainties associated with their underlying parameter values are documented, and that the effects of these uncertainties on their predictions are quantified.
New et al. (2014) used the PCoD model structure to investigate the potential effects of lost foraging dives on the health (measured by total lipid mass; see Schick et al., 2013) of adult female southern elephant seals, and the implications of variation in health for pup survival and population dynamics. They used information obtained from data loggers that were attached to animals immediately before they embarked on their ~240-day post-molt foraging trips. The data loggers allowed a reconstruction of their surface transit time and their foraging dive time. During portions of some foraging dives, elephant seals drift, and the rate of vertical movement during the drift is related to the ratio of lipid to lean body mass. The data logger information was calibrated against actual lipid gain during the foraging trip using measurements of body composition collected before and after the foraging trip. The results of other studies were used to link maternal mass to pup mass at weaning (Arnbom et al., 1993) and pup mass at weaning to pup survival (McMahon et al., 2000, 2003). The model was then used to determine the effect of foraging dive disturbance on pup survival. It was assumed that there
were no foraging dives for the duration of the disturbance, and surface transit time was set to the observed maximum for that individual. If animals were disturbed for 50% of their time at sea in 1 year, the predicted decline in population size was small (<1%). However, if this level of disturbance persisted for an extended period (for example, as a result of variations in the extent of the Antarctic ice sheet caused by climate change), the predicted effects were much greater (a 10% decline in abundance over 30 years). This analysis was only possible because detailed longitudinal data on the movements, health, and reproductive success of a large number of adult female seals were available. Such extensive data sets require decades of intensive research and are only available for a few marine mammal populations.
Researchers have adopted a range of techniques to build PCoD models in situations where empirical data are more limited. Nabe-Nielsen et al. (2014) used an individual-based model of the movements of harbor porpoises to estimate the potential effects of responses to the noise associated with wind turbine operation and shipping on their energy reserves. They then used a hypothetical relationship between energy reserves and survival to calculate population-level consequences. Villegas-Amtman et al. (2015) used a similar approach to predict the potential effects of reduced energy intake on reproductive success and survival for gray whales.
If empirical data are sufficient to estimate a relation between behavioral change and health, but not between health and vital rates, it may be possible to use a surrogate measure for the relevant vital rate. Christiansen and Lusseau (2015) used a bioenergetic model and empirical information on the behavioral response of adult female minke whales (Balaenoptera acutorostrata) to whale-watching boats on their summer feeding grounds in Iceland to estimate the effects of these responses on the whales’ health (as measured by their blubber volume). They calculated how different rates of encounter with whale-watching boats would affect an individual whale’s health at the end of the summer, and then used an empirically derived relation between female blubber volume and fetal length (Christiansen et al., 2014) as a surrogate for the relationship between health and the probability of giving birth. Although interactions with whale-watching boats resulted in a 40% reduction in feeding activity, the predicted reduction in a female’s body condition over the course of the summer was very small (0.049%), because encounters with boats were rare. This reduction in body condition was not predicted to affect fetal survival. However, even if Christiansen and Lusseau (2015) had detected a significant effect on fetal survival, they would have been unable to forecast the population-level effects of exposure to whale-watching boats because the proportion of the North Atlantic minke whale population that feeds in Icelandic waters and the percentage that has actually encountered boats is not known.
In situations where even surrogate measures are unavailable, expert elicitation (Sutherland and Burgman, 2015) can be used to parameterize some of the transfer functions of the PCoD model. Expert elicitation is a formal process in which a number of experts on a particular topic are asked to predict what may happen in a particular situation. The process is used in conservation science when data are lacking but there is an urgent need for management decisions (Runge et al., 2011; Martin et al., 2012). It is designed to mitigate the well-documented problems that arise when expert judgments are canvassed in an unstructured way. These include anchoring, availability bias, confirmation bias, and overconfidence (Cooke, 1991). These predictions are combined into calibrated, quantitative statements, with associated uncertainty, which can be incorporated into mathematical models (Martin et al., 2012). King et al. (2015) used this approach to parameterize relationships between the number of days on which harbor porpoises were disturbed by noise associated with the construction of offshore wind farms and their survival and reproductive success. These relationships were then used to predict the potential population consequences of different scenarios for the construction of multiple wind farms. Lusseau et al. (2012) used a similar approach to predict the potential aggregate effect of noise associated with wind farm construction, tour boat operation, and harbor expansion on the bottlenose dolphin population in the Moray Firth, Scotland.
In the remainder of this chapter, how the PCoD framework can be expanded to assess the potential population-level effects of exposure to multiple stressors is considered.
DEFINING INDIVIDUAL HEALTH
Evaluation of the potential demographic impacts on marine mammal populations of cumulative exposure to multiple stressors requires the biological upscaling (Cooke et al., 2014) of many levels of organization, including the behavioral responses of individuals, and the effects of these responses on population dynamics, biogeography, and community ecology (see Figure 4.1 in Chapter 4). In this chapter, we consider upscaling to the level of population dynamics. One important factor that links individual behavioral and physiological responses to population dynamics is the “health” of individuals. In 1948, the World Health Organization (WHO) defined health as “a state of complete physical, mental and social well-being and not merely the absence of disease or infirmity.” Similarly, definitions of “disease” in wildlife are broader than just infection by pathogens. They include the potential for cumulative impacts on health from nutrition, exposure to toxic chemicals, and climate (Wobeser, 1981). The WHO definition has been debated and criticized over the years (Jadad and O’Grady, 2008; Huber et al., 2011; Stephen, 2014), and recently it has been proposed that health be considered as “the ability to adapt and self-manage” (Huber et al., 2011), implying that a healthy organism is capable of maintaining physiological homeostasis under changing conditions. For wildlife, such definitions are effec-
tively proxies for fitness, emphasizing the potential effects of health on lifetime reproductive success. The committee therefore adopts “the ability to adapt and self-manage” as the definition of health.
Given this background, an assessment of an individual’s health provides a useful integration of the way physiological and behavioral responses to multiple stressors may affect that individual’s fitness. Potential health indices include body condition, hematological and serum biochemical parameters, steroid hormone levels, and markers of immune function and oxidative stress. This approach offers some potential advantages over empirical attempts to correlate variations in demographic rates with exposure to different stressors, because it can provide an assessment of the potential for reduced survival and reproductive output prior to an actual alteration in these rates. In addition, the application of health-based approaches to modeling the cumulative effects of exposure to multiple stressors may increase understanding of the mechanisms by which these stressors affect fitness.
A CONCEPTUAL MODEL FOR THE CUMULATIVE EFFECTS OF MULTIPLE STRESSORS
In this section, an expanded version of the PCoD model shown in Figure 6 of New et al. (2014) is described that can also be used to understand how specific stressors affect individual animals, how these effects can accumulate as a result of exposure to multiple stressors, and how these cumulative effects may translate into population-level consequences. This model, identified as Population Consequences of Multiple Stressors (PCoMS), provides a framework around which quantitative, predictive models for particular situations can be constructed. Figure 5.1 shows the structure of this framework for a single individual exposed to one stressor. It differs from the original PCoD model in the following ways:
- It can be used to describe the effects of any dosage scenario for any stressor, not just those that cause disturbance.
- The individual-based nature of the model is made explicit.
- It includes the direct, acute effects of predation and anthropogenic causes of mortality, such as bycatch, collisions, and deliberate killing.
- Following the model outlined by McEwan (1998, Figure 1), the initial effect of any stressor is assumed to be on an individual’s physiology. The resulting physiological changes may or may not be translated into behavioral responses, depending on the context (Killen et al., 2013).
- The direct link between the behavioral change and health compartments in the PCoD model has been removed because, in practice, behavior can only affect health indirectly through its effects on physiology.
The model assumes that an individual’s response to any stressor is always mediated, at least initially, by a physiological response because the initial interaction with that stressor will always be through the nervous system. This reflects one of the fundamental aspects of the allostatic
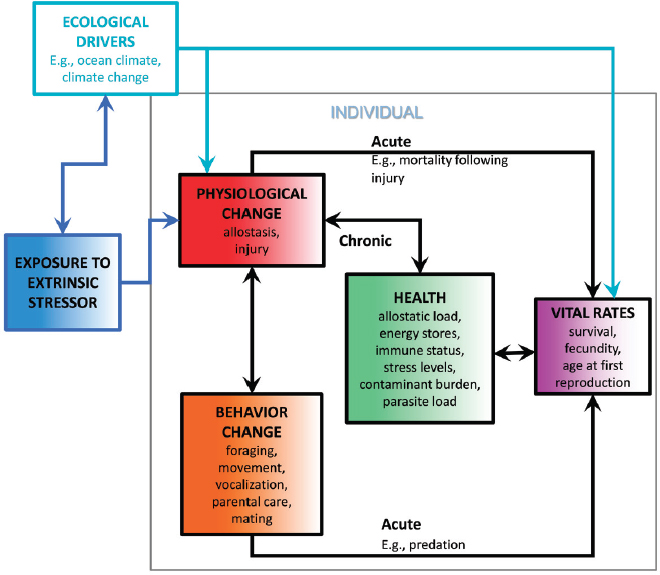
load concept (McEwan, 1998): whether or not an animal exhibits a behavioral response to a stressor will depend on its internal state and a suite of intrinsic stressors. Consider a foraging individual’s response to an approaching vessel. If it perceives the vessel, and its allostatic load is tolerable, it will probably take evasive action (a behavioral response mediated by a physiological response). However, if its body condition is poor, it may choose to keep feeding and may fail to evade the vessel.
Changes in behavior or physiology in response to a stressor may have a direct, acute effect on the vital rates of an individual. For example, an individual may move into an area with a high risk of predation as a result of avoidance behavior, or it may be at increased risk of mortality due to decompression sickness if it changes its diving behavior. For many marine mammal populations, the direct effects of acute stressors, such as bycatch and predation, may be more important than indirect effects. Because these acute effects operate on a short time scale, their cumulative effects are likely to be additive, as discussed in Chapter 4, so they can be modeled in a relatively straightforward way within the PCoMS framework. In this chapter, the focus is on the chronic effects of multiple stressors on health, primarily modeled using the concept of potential allostatic load (McEwen and Wingfield, 2003) that involves the adverse outcome pathways along which nonadditive effects are most likely to occur.
Allostatic load represents the consequences of the individual’s efforts to maintain homeostasis. Examples include reduced immune status, increased long-term levels of stress hormones, and reduced body condition relative to normal levels. The allostatic load associated with exposure to a particular stressor is only “potential” because that exposure will not necessarily have an immediate effect. However, it may have an effect on allostatic load at some later date, possibly because of the interaction with other stressors. A high allostatic load will have implications for all of an individual’s vital rates. For example, an adult female may choose to forgo breeding in order to reduce her potential for allostatic overload.
In some cases it may be sensible to combine compartments in the PCoMS model (i.e., hypothesize a transfer function that “jumps over” an intermediate compartment) if there is insufficient information to treat them separately. For example, explicitly modeling the physiological processes that occur between exposure to a stressor and a behavioral response is unlikely to be necessary.
The framework can readily be expanded to illustrate the effects of multiple stressors on a single individual (see Figure 5.2). Exposure to a particular dosage scenario for each stressor results in a unique set of physiological and behavioral responses (represented by the stack of responses in Figure 5.2, each layer corresponding to the responses to a different stressor), which may interact with the responses to other stressors. The consequences of the responses to many of these stressors are integrated through their combined effect on an individual’s potential allostatic load. Although it is currently impossible to measure allostatic load directly, it may be possible to use proxy measures of health (as described below) as appropriate response variable in studies of cumulative impacts.
The effects of multiple stressors may interact internally to affect allostatic load. For example, contaminants sequestered in the blubber layer may be mobilized during lactation or as a result of elevated stress levels or reduced energy
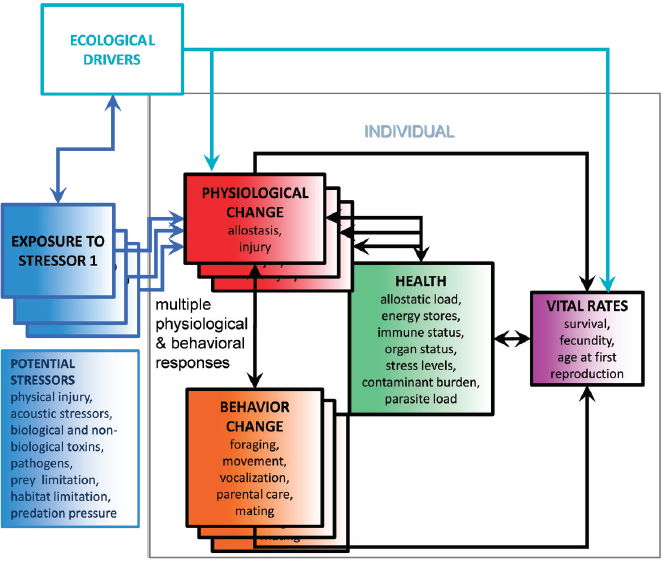
intake that are caused by other stressors. They may then interact with an individual’s immune function and affect its response if it is challenged with a novel pathogen. Similarly, the effect of macroparasite burden and dormant pathogens on health may be amplified if immune status is compromised.
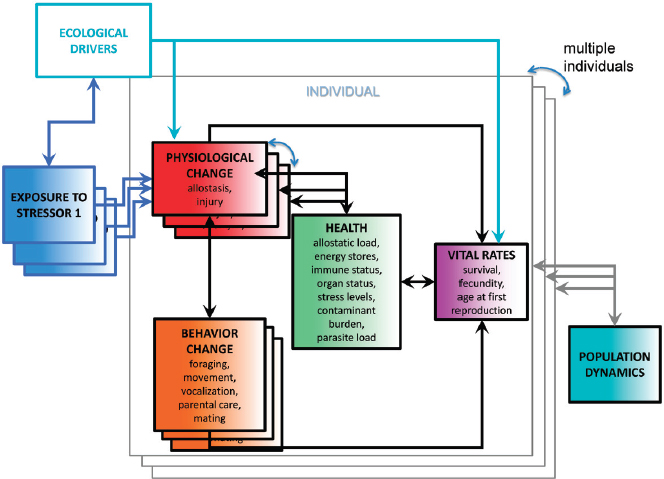
The framework can be expanded to the population level if estimates of the potential exposure of each individual in the population to the suite of stressors under consideration, and the effects of this exposure on physiology and behavior, are available (see Figure 5.3). This will require information on the distribution in time and space of the marine mammal species and the stressors, which can be assembled using approaches similar to those used by Maxwell et al. (2013). However, this will also require definition of appropriate dose–response relationships for each stressor, as well as a functional representation of interactions. The committee does not underestimate the difficulties that will be involved in obtaining the information needed to parameterize these functions for even a small number of stressors. The final step is to integrate the effect of these exposures on individual vital rates across the entire population in order to estimate their population-level consequences.
As noted above, the PCoMS framework treats mortality from predation and anthropogenic activities (such as bycatch, deliberate killing, and fatal ship strikes) as acute effects of exposure to the agents of this mortality (predators, fishing gear, hunting pressure, and vessel traffic). It can also be used to model the effects of natural and anthropogenic ecological drivers. For example, as noted in Chapter 3, changes in ocean climate can have profound effects on some marine mammal populations as a result of the redistribution of prey species. In the PCoMS framework this would be modeled as a change in exposure to a prey limitation stressor. Similarly, the effects of climate change are likely to lead to shifts in the distribution of vessel traffic, which can be modeled as changes in exposure to the risk of physical injury, toxic compounds, pathogens, and acoustic stressors. The effects of ice reduction on pagophilic species can be modeled as a habitat limitation stressor. Exposure to this stressor will result in behavioral changes, which could have acute effects (if seal species that normally breed on ice switch to breeding on land, and are therefore at greater risk of predation) or chronic effects (via the health compartment) as a result of the increased travel costs.
The PCoMS framework is similar to the framework developed by Rider et al. (2012) for assessing the role of nonchemical stressors in modulating the human risk factors associated with chemical exposure. However, Rider et al. (2012) place greater emphasis on how to predict the distribution of stressor doses across a population, and they do not consider the consequences of those doses for population dynamics.
The committee stresses that the PCoMS framework, like the original PCAD framework developed in NRC (2005), is only conceptual: it serves primarily to identify what the committee believes are the most important components of any comprehensive model of cumulative effects. The framework needs to be fleshed out with mathematical functions that describe the relationships between the different compartments, and integrated across all the individuals in the population that are exposed to the stressors under consideration. Determining appropriate forms for these functions and then parameterizing these functions will be extremely challenging. In many cases, it may be possible to ignore some of these relationships because they are not relevant to the population under consideration, but such decisions need to be carefully evaluated and fully justified. In situations where one stressor is considered to be dominant (i.e., its effects are so large that the effects of all other stressors to which the population is
exposed are negligible by comparison), use of a simplified version of the framework that considers only the dominant stressor is appropriate.
Recommendation 5.1: Future research initiatives should include efforts to develop case studies that apply the PCoMS framework to actual marine mammal populations. These studies will need to estimate exposure to multiple stressors, predict changes in behavior and physiology from those stressors, assess health, and measure vital rates in order to parameterize the functional relationships between these components of the framework. Where possible, the data on changes in demography, population size, and the health of individuals collected in these studies should be used to improve estimates of the parameters of the PCoMS model and reduce uncertainty.
APPLYING THE PCOMS FRAMEWORK TO NORTH ATLANTIC RIGHT WHALES
North Atlantic right whales have been protected since the 1930s and intensively studied since the early 1980s (Kraus and Rolland, 2007), yet their population numbers remain perilously low (Kraus et al., 2005). They are exposed to a wide range of stressors on their summer feeding grounds and over their lengthy migration pathways. These include physical injury as a result of entanglement in fishing gear, collisions with shipping, strong interannual variation in prey availability, and exposure to shipping noise (Clark et al., 2009). The North Atlantic Right Whale Catalogue,1 curated by the New England Aquarium, contain records of the life histories of many right whale individuals, as well as more than 700,000 photos and drawings. These records can be used to provide information on variations in the health (Pettis et al., 2004) and location of these individuals over time. Values for a set of visual health parameters are added to the catalog each time a whale is photographed. Schick et al. (2013) used these data to estimate the movements and overall health status of these individuals over time and to relate survival to health status. Rolland et al. (2016) used the same health information and model structure to link the health status of females in one year to their calving success in the subsequent year. Successful females were, on average, significantly healthier than unsuccessful ones. There was a dramatic decline in health status and calving success from 1998 to 2000 that coincided with reduced prey availability.
These relationships could be used as the transfer functions linking the health and individual vital rates compartments in a PCoMS framework that described the cumulative effects of physical injury (resulting from entanglement and collisions) and variations in prey availability on this population. Additional information in the North Atlantic Right Whale Catalogue could be used to parameterize a transfer function that would describe the changes in health that occur as a result of different levels of exposure to entanglement over the course of an entire year.
QUANTIFYING EXPOSURE-RELATED CHANGES IN PHYSIOLOGY AND ASSOCIATED CHANGES IN BEHAVIOR
Physiology
As noted above, there will be an immediate physiological response to exposure to a stressor mediated by the central nervous system. These kinds of short-term physiological responses to a stressor have evolved to reduce the risk that the animal’s health is compromised. Thus, one of the critical aspects of using physiological measures to assess aggregate and cumulative impacts is the ability to detect physiological changes that actually compromise health. In many cases, the generalized endocrine response to stress can provide relevant information, if there is appropriate contextual information to differentiate between normal adaptive variation and increased allostatic load. Hematological and serum biochemical parameters can be measured from blood to help identify a wide range of disease conditions such as inflammation, liver dysfunction, or anemia. Markers of immune status can provide critical information on the health of an individual, but it may be difficult to differentiate suppression of immune function from absence of exposure to pathogens. The effects of many stressors may be integrated through their impacts on oxidative stress (OS). For example, exposure to organic and inorganic contaminants is associated with dramatic increases in OS and oxidative damage (Ercal et al., 2001; Valavanidis et al., 2006). Exposure to polychlorinated biphenyls is associated with increased OS and oxidative damage to DNA, lipids, and proteins (Stohs, 1990; Oakley et al., 1996). OS also plays an important role in the pathogenesis of viral and bacterial infections (Schwarz, 1996). Chronic activation of the hypothalamic-pituitary-adrenal (HPA) axis and the release of glucocorticoids also enhance OS (Costantini et al., 2008, 2011; Stier et al., 2009; Cote et al., 2010). Such antioxidant responses are energetically expensive and may limit investment in important life-history components (Costantini et al., 2008; Dowling and Simmons, 2009; Monaghan et al., 2009; Metcalfe and Alonso-Alvarez, 2010; Isaksson et al., 2011). Thus, evidence of oxidative damage may provide a valuable marker of the cumulative effect of multiple stressors in marine mammals.
Uses of single physiological markers have yielded strong but inconsistent links to individual and population fitness. For example, a meta-analysis (Bonier et al., 2009) found negative associations between glucocorticoid concentrations and fitness in 51% of published studies. Together, suites of physiological measures that include body condition, hematological and serum biochemical parameters, stress hormones, reproductive hormones, immune markers,
___________________
and OS markers provide the most comprehensive measures of individual health. Changes in global gene expression in tissue samples may allow development of biomarkers that integrate these parameters.
Deep-diving marine mammals are exposed to high hydrostatic pressures and must support the metabolic costs of each dive using the oxygen they bring with them on the dive. If exposure to sound or other stressors changes dive behavior, this could have energetic costs and impose risks from effects of pressure. Marine mammals that dive to 500 m or more are exposed to hydrostatic pressures of 50 atmospheres (atm) or more. This would cause high-pressure nervous syndrome in most mammals tested and it is not known how marine mammals avoid this problem (Kooyman and Ponganis, 1998). More is known about how they avoid problems such as toxicity of oxygen at high pressures. When an air-breathing mammal fills its lungs at 1 atm of pressure and then dives, the volume of air reduces under pressure following Boyle’s law. The parts of the lung where gas is exchanged with the blood are the most compliant, so they contract before stiffer tissues such as the bronchi and trachea (Fahlman et al., 2009). This limits the risk that breath-hold divers are exposed to Po2 high enough to be toxic.
The shallower the depth at which diffusion stops because of alveolar collapse, the lower the Po2 to which breath-hold divers are exposed. Estimating the depth of alveolar collapse is thus an important parameter for determining change in physiology that may be stimulated by exposure to sounds that affect dive behavior. Measurement of arterial Po2 (McDonald and Ponganis, 2012) or arterial Pn2 (Falke et al., 1985) in free-diving pinnipeds has proven a powerful method to estimate depth of lung collapse. The Pn2 measurements were made possible by a portable blood sampling device that could be attached to freely diving seals.
The amount of oxygen available in the lungs is limited so that many marine mammal species store most of the oxygen they take on a dive in blood and muscle. The length of time a mammal can dive is limited by the oxygen available and tolerance of tissue for anaerobic metabolism, which can be detected by the presence of lactate in the blood. Thus, diving behavior represents a complex interaction of physiological adaptation and the requirements of foraging and social behaviors. Alterations in behavior in response to disturbance have the potential to create health impacts when they exceed the constraints imposed by physiology. The aerobic dive limit (ADL) has been defined as the dive duration after which there is an increase of lactate in the blood (Kooyman, 1985). Many studies have estimated the ADL by estimating the O2 store and metabolic rate, but both of these may be modulated by dive behavior, and the estimate is sensitive to assumptions about how low a Po2 an animal can tolerate. Meir et al. (2009) measured arterial and venous Po2 in freely diving elephant seals and found they tolerate unusually low Po2 in their tissues, allowing them to prolong their dives. More measurements of post-dive lactate would improve understanding of ADL, and more measurements of arterial and venous Po2 would help to understand the physiological mechanisms affecting ADL.
Another important exposure-related change in physiology involves the regulation of N2 and managing risk of decompression. Recent evidence that exposure to sonar can cause decompression sickness (DCS) in deep-diving whales has reinvigorated analysis of risk of DCS in marine mammals (Hooker et al., 2012). When a mammal dives with lungs full, as the hydrostatic pressure increases, N2 diffuses into the blood and tissues, elevating their Pn2. As the lungs collapse under pressure, this diffusion reduces and ceases. However, as the animal ascends, with reducing hydrostatic pressure, there is a decompression, with risk that bubbles may form if tissues or blood are supersaturated with respect to the ambient hydrostatic pressure. There is evidence that chronic exposure to small bubbles may damage the bones of deep-diving sperm whales (Moore and Early, 2004) and explosive DCS has been reported for beaked whales exposed to naval sonar (Fernández et al., 2005). Models of diving physiology have been used to predict risk of gas bubbles based on the dive profiles of tagged deep-diving marine mammals (Fahlman et al., 2014), and these models help us to understand how reactions to anthropogenic noise might disrupt the mechanisms used by these animals to manage gases under hydrostatic pressure, leading to risk of DCS. Marine mammals are breath-hold divers, so rapid ascent from a single dive poses a low risk of DCS. Furthermore, once an animal dives below the depth of alveolar collapse in the lungs, there is no gas exchange. Therefore, one risk factor for DCS is time spent above the depth of alveolar collapse, but deep enough for hydrostatic pressure to increase the nitrogen tension in tissues. Another risk factor for DCS involves long-duration dives at great depth, as these may cause redistribution of dissolved gases from tissues that take up and release gas quickly (e.g., muscle) to tissues that take up and release gas more slowly (e.g., adipose tissue) (Fahlman et al., 2014).
Behavior
The most comprehensive information on quantifying exposure-related changes in marine mammal behavior as a function of measured levels of exposure to a stressor come from studies of the behavioral responses of an increasing number of species to sounds produced by military sonars, or devices that mimic these sounds. Harris and Thomas (2015) have provided a review of these studies. Behavioral response studies are experiments designed to test the causal link between sound exposure and behavioral responses. One challenge for these studies with marine mammals is the difficulties in quantifying sound exposure at the animal and in obtaining continuous unbiased measures of behavioral responses. Johnson and Tyack (2003) describe a sound and movement recording tag that functions as an acoustic
dosimeter and as a sensitive recorder of behavioral responses. These tags have been used in experiments that record baseline behavior, then record exposure and response to controlled playback of sonar and other sounds. Use of a dose escalation design makes it possible to estimate the lowest exposure that elicits each response. Statistical methods for identifying significant changes in behavior are described by Miller et al. (2012a). Miller et al. (2014) used this approach to define the probabilistic dose–response function illustrated in Figure 1a in Box 2.2.
One common response to anthropogenic sound is a marked reduction in marine mammal vocalizations. This may be the result of animals leaving the vicinity of the sound source or ceasing vocalization. Passive acoustic monitoring can be used to derive a relationship between received sound levels and this response. For example, Moretti et al. (2014) used data from an array of hydrophones on a Navy range to derive a relationship between acoustic detections of Blainville’s beaked whales and calculated exposure level of sonar. Thompson et al. (2013b) deployed their own array of acoustic sensors to relate the detection rate of harbor porpoise clicks to distance from a seismic survey.
Controlled experiments and opportunistic monitoring of behavioral responses to anthropogenic noises can often complement one another. Controlled experiments can be critical for demonstrating that a sound causes a response, and for defining how animals respond to the sound. These results, which are often derived from a small sample of short-term experiments, can be used to design a monitoring scheme for the actual activities that produce the sounds. The Moretti et al. (2014) study showed responses to actual sonar exercises that were similar to those predicted from the experiments. Thompson et al. (2013b) were not only able to show the spatial scale of responses to seismic surveys, but were also able to demonstrate how that response reduced over the duration of the survey.
QUANTIFYING EXPOSURE-RELATED CHANGES IN INDIVIDUAL HEALTH
Measures of Body Condition That Are Useful for Assessing Health
Body condition is one of the few proxies for allostatic load that can be measured using conventional methods. Classic methods to measure energy stores involve separating skin, blubber, and other tissues, weighing them, and estimating their caloric values. Noninvasive measures such as ultrasound can also be used to measure blubber layers. The total amount of water in the body (total body water or TBW) can be estimated by diluting a known volume of isotopically labeled water, and total body lipid (TBL) can then be estimated by known relationships between TBW and TBL. Less specific morphometric measurements such as length, weight, and girth are also often used to estimate body condition. These measurements do not require dead animals, but they often require handling live animals. Biuw et al. (2003) used the dilution technique to validate a method for estimating body condition on tagged elephant seals while they were at sea. They used the rate of vertical change in depth of these animals while they were drifting passively through the water column to estimate their buoyancy. The lean tissue of marine mammals is denser than seawater, but lipid stores are less dense, so that the buoyancy of an animal is largely a function of the ratio of lean to lipid tissues (Crocker et al., 1997). Schick et al. (2013) used information of this kind to estimate variations in the health of individual elephant seals over time. These health estimates were then incorporated into the PCoD model developed by New et al. (2014). Monitoring buoyancy appears to be a useful method for quantifying changes in body condition in a number of species. For example, Gordine et al. (2015) describe a filtering method that can reliably detect buoyancy changes in the dive records of drift diving species using the highly summarized data that are normally collected by most of the tags fitted to marine mammals. Aoki et al. (2011) demonstrated that estimates of the body density of elephant seals fitted with tags that could record depth, swim speed, and temperature at 1 second intervals, and three-dimensional accelerations (for detecting pitch and hind flipper movements), were within 1% of the equivalent estimates from isotope dilution from the same individuals. In addition to these detailed studies of buoyancy, information on changes in body condition may be obtained from time series of aerial photogrammetry of the same individual collected using unmanned vehicles (e.g., Durban et al., 2015).
Measures of Organ Status That Are Useful for Assessing Health
Hematology and serum chemistry parameters are routinely used in human health care to assess physiological state and are generally organized into panels that represent specific pathological processes or organ systems. In circumstances where blood samples can be collected from marine mammals these measures can provide information on basic metabolic status, kidney function, inflammation, liver disease, or thyroid disorders.
Measures of Immune Status That Are Useful for Assessing Health
Wild populations and individuals are constantly challenged by pathogens. The immune responses to these pathogens influence the demographic parameters of populations (Daszak et al., 2000; Morens et al., 2004). Immune responses are energetically expensive, and the ability to mount them may be influenced by nutritional state, stress hormones, and toxics exposure (Hammond et al., 2005; Peck et al., 2016). The primary difficulty of assessing immune response is
interpreting variation in markers without information on the exposure of individuals to pathogens. To date, studies on immune function in marine mammals suggest that they share all of the primary immune components identified in biomedical studies. However, it is likely that there are modifications to marine mammal immune function that serve to preserve response under the diverse environmental conditions experienced, including high pressure, cold temperatures, and extreme hypoxemia, conditions that are immunosuppressive in many human studies (Shepard and Shek, 1998; Brenner et al., 1999).
A variety of approaches have been developed to assess immune competency from cross-sectional samples. Functional immune assays have been developed for both pinniped and cetacean species that quantify the proliferative response of lymphocytes (e.g., Levin et al., 2005; Mori et al., 2006; Schwacke et al., 2012). Cytokines regulate the development of humoral and cellular immune responses. For species where blood or tissue sampling is feasible, a suite of markers are available to measure individual innate and adaptive immune responses, including circulating levels of cytokines, acute phase proteins, and immunoglobulins. Microarrays and RNA sequencing allow examination of cytokine expression in tissue. Multiplex cytokine arrays have been optimized for individual marine mammal species (Mancia et al., 2007; Vechhione et al., 2008; Eberle et al., 2013). DNA sequences for cytokines for many species have been published and can be used to develop quantitative assays (King et al., 1996; Inoue et al., 1999). Commercial assay antibodies have also been validated for use in numerous marine mammal species (e.g., Peck et al., 2016). Innate immune function can be assessed with serum from any species through simple complement killing assays, such as hemolytic complement (CH50) and bacteria killing assays. As measures of adaptive immune response, total immunoglobulin levels have been measured using species-specific and commercial antibodies (King et al., 1998; Peck et al., 2016), and pathogen-specific immunoglobulins have been measured to document exposure to a wide variety of diseases using direct agglutination assays, immunohistochemical staining, and commercial enzyme-linked immunosorbent assays. Together these measures represent a formidable arsenal of tools that could, in principle, be used to assess individual and population innate and adaptive immune function. However, collecting the appropriate samples for analysis will be challenging, particularly because large cross-sectional data sets on immune markers in populations are needed to differentiate robust and appropriate immune responses that occur as part of life-history variation from exaggerated or suppressed immune responses in individuals that indicate impaired health. The association between immunosuppression and increased infections is well documented in humans (Luebke et al., 2004), but the form of that relationship varies with life stage and the level of immune suppression. Given the well-documented exposure to pathogens and parasites in wild marine mammals, it is likely that immunosuppression will lead to an increase in rates of infection.
Measures of Stress That Are Useful for Assessing Health
One approach to measuring the cumulative physiological impact of multiple stressors on marine mammals is through the measurement of stress hormones. Physiological stress can be defined as a complex physiological response to aversive environmental stimuli that challenge fluctuating homeostatic set points. The mammalian neuroendocrine stress response is driven largely by activation of the HPA axis, which results in the release of glucocorticoids into circulation (Sapolsky et al., 2000). Glucocorticoids bind to tissue receptors and alter expression of genes affecting a diverse array of physiological processes, including metabolism. Meta-analysis has shown that anthropogenic disturbances are associated with elevation of glucocorticoids in wildlife regardless of the kind of disturbance (Dantzer et al., 2014), although the fitness impacts of these elevations are less clear. While acute stress responses are usually adaptive, and may even increase subsequent fitness through the process of hormesis (Boonstra, 2005), biomedical studies have suggested that chronic activation of stress responses can have negative effects on survival and reproduction, mainly through suppression of immune and gonad function. Thus, chronic activation of the HPA axis may be an important mechanism by which cumulative exposure to diverse stressors leads to physiological and demographic impacts. Chronic stress resulting from persistent or cumulative exposure to stressors may lead to dysregulation of the HPA axis. This dysregulation is thought to result from loss of negative feedback, when chronic elevation of glucocorticoids decreases the number of glucocorticoid receptors in areas of the brain that regulate activation of the response (Dickens et al., 2009).
Several conceptual models have been developed to represent the physiological impacts of chronic stress, including allostatic overload (McEwan and Wingfield, 2003) and homeostatic overload (Romero et al., 2009). Individuals undergoing chronic stress responses would be expected to exhibit higher baseline levels of circulating glucocorticoids, enhanced glucocorticoid responses to environmental stressors, and increased time for glucocorticoid levels to return to baseline following a stressor (Dickens and Romero, 2013). In biomedical studies, chronic elevation of glucocorticoids directly suppresses immune and gonad function (Sapolsky et al., 2000), although these relationships are less well established in wildlife species than in humans. Because the detrimental physiological effects of chronic stress are thought to result from a larger cumulative exposure to glucocorticoids and because conserved glucocorticoid stress responses can result from a wide variety of stressors, measurement of glucocorticoids represents a potentially important proxy for cumulative stress and health in marine mammals.
Unfortunately, measurement of the magnitude of stress responses and the status of negative feedback regulation is not possible for most marine mammal species, because it requires repetitive blood samples or experimental manipulations (adrenocorticotropic hormone or dexamethasone injection). Baseline (i.e., not altered by sampling) glucocorticoid concentrations can be measured in rapidly acquired blood samples, although this kind of sampling is not feasible for most species of marine mammals. For pinniped species that haul out on land, studies have suggested that chemical immobilization may ameliorate the stress response to handling, allowing measurement of baseline levels in some species (Champagne et al., 2012). Extensive work is under way to develop and validate techniques for measurement of glucocorticoids in other sample matrices that are appropriate for use in free-ranging cetaceans, including fecal samples, blow, blubber, and skin (reviewed by Hunt et al., 2013), sometimes called “integrated measures.” Measures from these matrices may be superior to blood samples in allowing identification of chronic elevation in baseline glucocorticoids. Fecal measures are the least invasive and may be more sensitive to anthropogenic disturbances (Dantzer et al., 2014) but are sometimes difficult to link to targeted individuals. Blubber samples acquired by biopsy dart have perhaps the greatest potential as a matrix for measurement of glucocorticoids in large whales. Highly fat-soluble glucocorticoid hormones dissolve in perfused blubber. Blubber samples can be targeted to specific individuals and taken prior to any alteration in glucocorticoids from sampling. In addition to measurement of glucocorticoids, blubber samples can also be analyzed for reproductive hormones, fatty acids, and contaminants, allowing increased understanding of potential integration among stressors. One key limitation in the current utility of measuring blubber glucocorticoids is understanding how blubber concentrations respond to acute and baseline changes in plasma (i.e., turnover and lag times). This issue can potentially be addressed through controlled experiments in tractable species that allow manipulation of cortisol levels and repetitive sampling. It is also important to understand how blubber cortisol levels may be influenced by important life-history events like fasting or reproduction. This need can be addressed through large sample size, cross-sectional, or longitudinal studies that measure glucocorticoids across multiple matrices. Finally, there is great potential for development of gene expression markers in marine mammal blubber that differentiate between acute and chronic elevation in glucocorticoids (Khudyakov et al., 2015).
Recent developments in the technologies available for long-term time series of stress and reproductive hormones, as well as potential exposure to contaminants, have the potential to provide unique insights into the historical variation in stress responses and reproduction. Earplugs from several species of large cetaceans provide time series of hormone and contaminant data over the lifetime of the individual, as long as 65 years in currently analyzed samples (Trumble et al., 2013). These profiles potentially reveal the timing of pregnancies and lactation, baseline stress hormones, and exposure to several important classes of contaminants. Similarly, baleen samples can provide individual time series of stress and reproductive hormones lasting up to 20-25 years (Hunt et al., 2014).
Interpretation of the potential relationship between glucocorticoid levels and individual fitness requires extensive contextual data. Currently there are few large cross-sectional data sets of stress hormones from marine mammals that can be used to quantify natural variation in glucocorticoids with age, gender, season, and/or reproductive status. However, such data are critical for assessing anthropogenic impacts on stress hormone levels and their potential for health and reproductive effects as well as for determining key periods where sampling is likely to be most informative about health. A primary research need is to collect glucocorticoid measurements across life-history stages in species of interest. These data will not only provide a basis for identifying unusual glucocorticoid levels in individuals or populations but will also enhance understanding of how natural variation in glucocorticoids may regulate the allocation of energy resources between immune response and reproduction, and how intrinsic factors might modify responses to anthropogenic stressors. For example, a large literature in seabirds has focused on the roles that natural variation in glucocorticoids plays in regulating breeding decisions (e.g., Kitaysky et al., 2007), carry-over effects between stress responses at various life-history stages (e.g., Schultner et al., 2014), and the interaction of glucocorticoid stress responses with exposure to toxins (e.g., Nordstad et al., 2012; Tartu et al., 2015). Currently, no parallel literature exists for marine mammals. Understanding the adaptive uses of stress responses in marine mammal systems is critical to assessing how cumulative stress impacts might integrate and when they are most likely to have demographic consequences.










