2
Overview of the Congressionally
Directed Medical Research Programs
The Department of Defense’s (DoD’s) Congressionally Directed Medical Research Programs (CDMRP) has a well-established process for managing the review and selection of funding applications that it receives for its medical research programs. This chapter presents an overview of that process and provides a brief overview of CDMRP’s organization and structure. The current functions of the program office, how it is funded, and its place within the health care hierarchy of DoD are described. The chapter also includes a brief summary of CDMPR’s program processes from program initiation and goal setting through application solicitation and review to award negotiation. This chapter is intended to provide general information about CDMRP in order to set the stage for the subsequent chapters that detail the specific steps in the review process.
THE CURRENT CDMRP
CDMRP views its role in the medical research community as a leader in advancing medical and scientific research and filling research gaps “by funding high impact, high risk and high gain projects that other agencies may not venture to fund” (CDMRP, 2016a). The vision and mission statements for CDMRP can be found in Box 2-1. CDMRP currently manages 29 research programs (see Box 2-2), including several programs on behalf of other DoD offices, in particular, the Defense Health Agency (DHA). Each research program has its own specific vision and mission statement.
CDMRP has emphasized that its research programs and funded applications need to be relevant to the health of service members, veterans, and their families. The committee is aware that the military health care system (TRICARE) is one of the largest health care systems in the world and cares not only for active duty and retired service members, but also
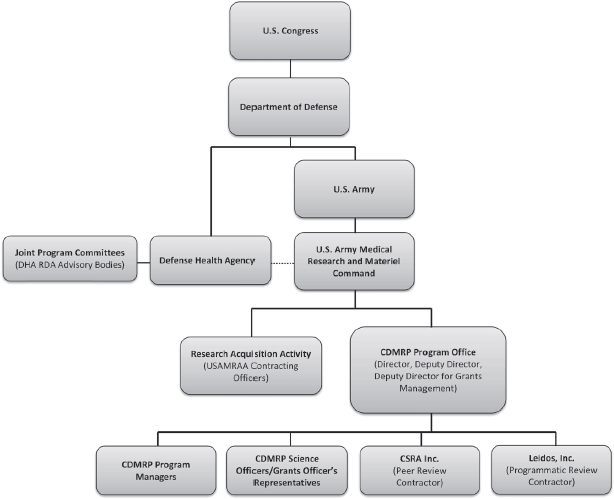
NOTE: DHA = Defense Health Agency; RDA = Research, Development, and Acquisition; USAMRAA = U.S. Army Medical Research Acquisition Activity.
SOURCE: Adapted from Salzer, 2016d.
for their families (TRICARE, 2016). Thus, health-related issues that affect service members or their dependents would fall within the purview of DoD medical research. The scope of research performed by DoD has been a topic of debate in the U.S. Senate. In June 2016, the Senate voted that it would not put restrictions on the DoD money used for medical research so that it may continue to benefit military members and their families and also the general public (Tritten, 2016).
CDMRP is located within the U.S. Army Medical Research and Materiel Command (USAMRMC), headquartered at Fort Detrick, Maryland (see Figure 2-1). The USAMRMC established the CDMRP program office in response to congressional funding for research initiatives; it decides how CDMRP is staffed and how it functions. Although CDMRP is based within the Department of the Army, the program office also has involvement from both the Department of the Navy and the Department of
the Air Force. Over the years, CDMRP leadership has come from all three branches, with representatives from each service having served as CDMRP director.
As noted in Box 2-2, CDMRP provides management support for several research programs on behalf of the DHA’s Research, Development, and Acquisition Directorate. DHA receives core funds1 from Congress for these research programs, which have a strong military focus. Each of those programs is aligned with one or more joint program committees (JPCs), but the level of involvement in a program is at the discretion of the JPC. JPC membership consists of both DoD and non-DoD medical and military technical experts and representatives from the Department of Veterans Affairs and the Department of Health and Human Services. The largest DHA research program for which CDMRP provides management support is the Defense Medical Research and Development Program (DMRDP), which is overseen by the following six2 JPCs:
- medical training and health information services (JPC-1),
- military infectious diseases (JPC-2),
- military operational medicine (JPC-5),
- combat casualty care (JPC-6),
- radiation health effects (JPC-7), and
- clinical and rehabilitative medicine (JPC-8).
JPCs support DMRDP and other CDMRP programs with a strong active-duty military focus by providing guidance, through programmatic review of applications and by making funding recommendations (Resnik et al., 2013). CDMRP states that JPCs also engage in strategic planning activities that may feed into the CDMRP vision setting process (Salzer, 2016a). CDMRP staff and contractors work closely with the JPCs to provide program and award management support for JPC research initiatives (CDMRP, 2016b).
Although all CDMRP research programs are aligned with at least one JPC, programs that do not have a strong military health focus such as the Breast Cancer Research Program, the Autism Research Program, and the Parkinson’s Research Program do not necessarily have substantial input from the affiliated JPC. The strongly JPC-aligned programs are an integral part of CDMRP, but they require different chain-of-command procedures
___________________
1 Core funds are part of the annual DoD budget set by Congress and the president (known as the President’s Budget). The CDMRP appropriations are not considered to be DoD core funds.
2 There is no JPC-3 or JPC-4 committee.
and approvals from those that CDMRP generally uses to manage its other research programs (Salzer, 2016a; Santullo, 2016).
CDMRP FUNDING
Funding for CDMRP research programs is appropriated on an annual basis. Congress, in response to advocacy groups and other interested parties, selects which programs will be funded and at what level each year. Language in the appropriations bills can range from very specific instructions on how to spend the money for a program to quite vague statements or just the line item; this can result in considerable variation in program funding and focus. Figure 2-2 provides an example of line items for CDMRP research programs as well as an example of accompanying congressional language from the Department of Defense Appropriations Act, 2016.
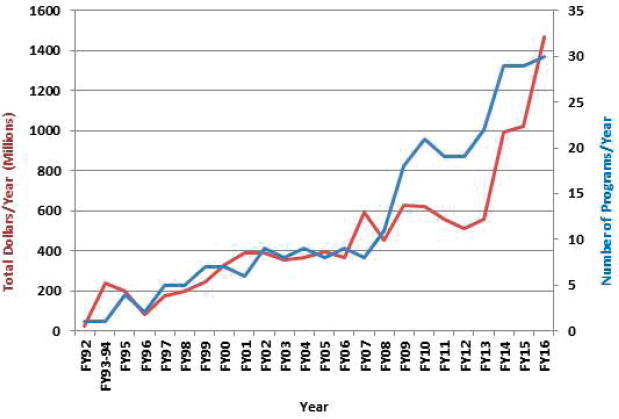
Although appropriations for individual research programs (and thus CDMRP) in general can (and occasionally do) vary from year-to-year, in most cases funding for the individual programs has stayed relatively consistent since their inception; however, a few programs have been discontinued because of a lack of congressional funding (e.g., defense women’s health, osteoporosis, genetic studies of food allergies). Table 2-1 shows the funding history for each program and the number of programs funded by year, and Figure 2-3 summarizes the growth of CDMRP since 1992. Funding for CDMRP as a whole increased by $433.3 million in 2014 from $557.7 million to $991.0 million, and increased again in 2015 by $33.4 million to a total of $1.024 billion. The initial program, the Breast Cancer Research Program, has been funded since 1992 for a total of more than $3 billion, and the Prostate Cancer Research Program (begun in 1997) and the Peer Reviewed Medical Research Program (begun in 1999) have both received more than $1 billion. The Psychological Health/Traumatic Brain Injury Research Program, a program that the CDRMP administers on behalf of three JPCs, is the fourth largest program at just over $800 million, and has been funded for the past 8 years.
OVERVIEW OF THE CDMRP REVIEW PROCESS
All CDMRP research programs follow the same general multi-step process for soliciting, reviewing, and making funding decisions for applications (see Figure 2-4). These steps are conducted by two panels: the programmatic panel and the peer review panel. The application review cycle (the left side of Figure 2-4 in blue) spans approximately 12 months, beginning with the annual congressional appropriation of funds and ending with a list of funding recommendations being submitted to USAM-
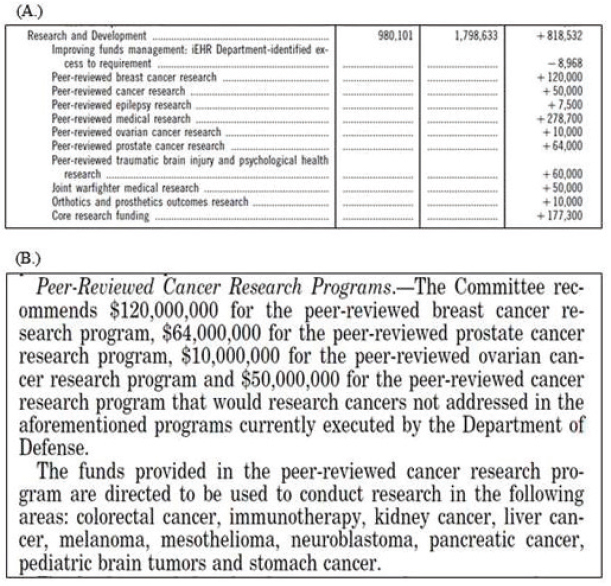
SOURCE: Senate Report to accompany S. 1558 (June 11, 2015).
RMC for approval. Award negotiation and implementation may take an additional year.
After CDMRP receives its appropriations, it has 2 years by law to obligate the money; thus, each CDMRP award is fully funded up front. However, even though each award is fully funded, principal investigators do not necessarily receive all their funding at once; rather, milestones are established and must be met for the release of further funds. Program announcements (see Chapter 4) specify the maximum length of the award over which money may be allocated; the length of the award may not exceed 5 years (IOM, 2004).
The second part of the process is the award management cycle (the right side of Figure 2-4 in red), in which recommended awards are selected and approved for funding, negotiations are undertaken to finalize the awards, and the progress of the awards is then monitored from initiation through to closeout. In this report, the committee evaluates the CDMRP review process only up to the point of the funding recommendations (Step 5).
The five major steps in the application review process considered by the committee (in yellow in Figure 2-4) are as follows (CDMRP, 2015a):
-
Vision Setting (Chapter 4): When the congressional appropriation process is complete, CDMRP receives funds for each research program.
- For existing programs, the first step in the process is the vision setting meeting held by the programmatic panel. Vision setting occurs in months 1–4 of the program cycle to identify research gaps and to define an investment strategy for that year (that is, to choose award mechanisms and topics) to address those gaps. The annual investment strategy identifies the award mechanisms to be used for each research program; the award mechanisms result in program announcements that drive the application process.
- If the appropriation is for a new research program, a one-time stakeholders meeting is held before the first vision setting meeting to help scope out the Congressional intent for the program as well as research needs.
-
Program Announcement Release (Chapter 4): Following the vision setting meeting, a program announcement for each award mechanism is developed by the CDMRP program manager and released by the CDMRP contracting office within 2–5 months of the meeting. In some cases, a pre-announcement will be released to alert interested parties to an upcoming program announcement. Interested researchers can sign up to be electronically notified when new program announcements are released or can find the program announcements online. Program announcements notify the research community that new funding opportunities are available for that program and delineate the information that must be submitted to CDMRP for each application.
- The electronic biomedical research application portal, eBRAP, is used for a variety of program activities, including application submission, notification of availability of program announcements, and communications with applicants. Although government owned, it is maintained by the peer review contractor.
TABLE 2-1 CDMRP Funding History (in Millions) for Each Program by Year
| Research Program | FY92 | FY93-94 | FY95 | FY96 | FY97 | FY98 | FY99 | FY00 | FY01 | FY02 | FY03 |
|---|---|---|---|---|---|---|---|---|---|---|---|
|
Breast Cancer |
25.0 | 240.0 | 150.0 | 75.0 | 108.3 | 135.0 | 136.8 | 176.3 | 177.4 | 151.5 | 152.2 |
|
Prostate Cancer |
45.0 | 40.0 | 50.0 | 75.0 | 100.0 | 85.0 | 85.0 | ||||
|
Peer Reviewed Medical |
19.5 | 25.0 | 50.0 | 50.0 | 50.0 | ||||||
|
Psychological Health/Traumatic Brain Injury |
|||||||||||
|
Defense Medical Research and and Development |
|||||||||||
|
Institutionally Based Programs |
5.0 | 6.5 | 3.5 | 14.0 | 25.8 | 31.0 | 25.5 | 30.2 | |||
|
Peer Reviewed Orthopaedic |
|||||||||||
|
Neurofibromatosis |
8.0 | 8.0 | 9.8 | 11.5 | 15.0 | 17.0 | 21.0 | 20.0 | |||
|
Ovarian Cancer |
7.5 | 10.0 | 10.0 | 12.0 | 12.0 | 10.2 | 10.0 | ||||
|
Restore Core Research Funding Reduction |
|||||||||||
|
Joint Warfighter Medical |
|||||||||||
|
Peer Reviewed Cancer |
|||||||||||
|
Spinal Cord Injury |
|||||||||||
|
Gulf War Illness |
|||||||||||
|
Deployment Related Medical |
|||||||||||
|
Lung Cancer |
|||||||||||
|
Autism |
|||||||||||
|
Tuberous Sclerosis Complex |
1.0 | 2.0 | |||||||||
|
Amyotrophic Lateral Sclerosis |
|||||||||||
|
Parkinson’s Research |
|||||||||||
|
National Prion |
42.5 | ||||||||||
|
Defense Women’s Health |
40.0 | ||||||||||
|
Multiple Sclerosis |
|||||||||||
|
Peer Reviewed Alzheimer |
|||||||||||
|
Vision |
|||||||||||
|
Army Rapid Innovation Fund |
|||||||||||
|
Orthotics and Prosthetics Outcomes |
|||||||||||
|
Bone Marrow Failure |
|||||||||||
|
Reconstructive Transplant |
|||||||||||
|
Military Burn |
|||||||||||
|
Chronic Myelogenous Leukemia |
5.0 | 4.3 | |||||||||
|
Duchenne Muscular Dystrophy |
|||||||||||
|
Epilepsy |
|||||||||||
|
Trauma Clinical Research Repository |
|||||||||||
|
Small Business Innovation Research/Technology Transfer |
|||||||||||
|
Alcohol and Substance Abuse Disorders |
|||||||||||
|
PH/TBI (Cognitive Rehabilitation Clinical Trials) |
|||||||||||
|
Chiropractic Clinical Trial |
|||||||||||
|
Cooperative DOD/VA Medical |
4.0 | 2.8 | |||||||||
|
Osteoporosis |
5.0 | ||||||||||
|
Peer Reviewed Tick-Borne Disease |
|||||||||||
|
Clinical Research Intramural Initiative |
|||||||||||
|
Genetic Studies of Food Allergies |
|||||||||||
|
Myeloproliferative Disorders |
|||||||||||
| Total Dollars/Year | 25.0 | 240.0 | 200.0 | 83.0 | 175.3 | 198.3 | 245.8 | 331.9 | 387.4 | 391.7 | 353.6 |
| Total Number of Programs/Year | 1 | 1 | 4 | 2 | 5 | 5 | 7 | 7 | 6 | 9 | 8 |
* Year does not include all funding to be executed on behalf of others, which was not available at the time of this report.
SOURCE: Adapted from CDMRP, 2016c.
| FY04 | FY05 | FY06 | FY07 | FY08 | FY09 | FY10 | FY11 | FY12 | FY13 | FY14 | FY15 | FY16* | Total Dollars/Program |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 152.1 | 151.9 | 129.4 | 128.8 | 140.1 | 151.5 | 151.0 | 150.9 | 120.7 | 120.6 | 120.5 | 120.5 | 120.0 | 3,285.5 |
| 85.0 | 85.0 | 80.0 | 80.0 | 80.0 | 80.0 | 80.0 | 80.0 | 80.0 | 80.0 | 80.0 | 80.0 | 80.0 | 1,530.0 |
| 50.0 | 50.0 | 50.0 | 50.0 | 50.0 | 50.0 | 50.0 | 50.0 | 50.0 | 200.0 | 247.5 | 278.7 | 1,370.7 | |
| 301.0 | 57.5 | 50.8 | 61.4 | 91.7 | 77.5 | 86.0 | 101.6 | 125.0 | 952.5 | ||||
| 94.5 | 58.8 | 37.1 | 33.7 | 139.1 | 89.5 | 299.6 | 752.2 | ||||||
| 39.9 | 67.1 | 68.3 | 48.9 | 41.0 | 37.4 | 42.3 | 486.3 | ||||||
| 112.0 | 22.5 | 24.0 | 30.0 | 30.0 | 30.0 | 30.0 | 30.0 | 308.5 | |||||
| 20.0 | 25.0 | 17.0 | 10.0 | 8.0 | 10.0 | 13.8 | 16.0 | 12.8 | 15.0 | 15.0 | 15.0 | 15.0 | 302.9 |
| 10.0 | 10.0 | 10.0 | 10.0 | 10.0 | 20.0 | 18.8 | 20.0 | 16.0 | 20.0 | 20.0 | 20.0 | 20.0 | 276.5 |
| 41.4 | 207.5 | 248.9 | |||||||||||
| 4.0 | 100.0 | 50.0 | 50.0 | 204.0 | |||||||||
| 16.0 | 15.0 | 16.0 | 12.8 | 15.0 | 25.0 | 50.0 | 50.0 | 199.8 | |||||
| 35.0 | 11.3 | 12.0 | 9.6 | 30.0 | 30.0 | 30.0 | 30.0 | 187.9 | |||||
| 5.0 | 10.0 | 8.0 | 8.0 | 8.0 | 10.0 | 20.0 | 20.0 | 20.0 | 20.0 | 129.0 | |||
| 101.9 | 101.9 | ||||||||||||
| 20.0 | 15.0 | 12.8 | 10.2 | 10.5 | 10.5 | 10.5 | 12.0 | 101.5 | |||||
| 7.5 | 6.4 | 8.0 | 8.0 | 6.4 | 5.1 | 6.0 | 6.0 | 6.0 | 7.5 | 66.9 | |||
| 3.0 | 3.2 | 4.3 | 4.0 | 6.0 | 6.0 | 6.4 | 5.1 | 6.0 | 6.0 | 6.0 | 6.0 | 65.0 | |
| 5.0 | 5.0 | 7.5 | 8.0 | 6.4 | 7.5 | 7.5 | 7.5 | 7.5 | 61.9 | ||||
| 16.0 | 16.0 | 16.0 | 48.0 | ||||||||||
| 42.5 | |||||||||||||
| 40.0 | |||||||||||||
| 5.0 | 4.5 | 4.8 | 3.8 | 5.0 | 5.0 | 5.0 | 6.0 | 39.1 | |||||
| 12.0 | 12.0 | 15.0 | 39.0 | ||||||||||
| 8.9 | 10.0 | 10.0 | 10.0 | 38.9 | |||||||||
| 13.6 | 5.9 | 9.4 | 1.9 | 5.0 | 35.7 | ||||||||
| 10.0 | 10.0 | 10.0 | 30.0 | ||||||||||
| 1.0 | 5.0 | 3.8 | 4.0 | 3.2 | 3.2 | 3.2 | 3.2 | 3.0 | 29.6 | ||||
| 15.0 | 12.0 | 27.0 | |||||||||||
| 8.0 | 8.0 | 8.0 | 24.0 | ||||||||||
| 4.3 | 4.3 | 4.3 | 22.1 | ||||||||||
| 4.0 | 3.2 | 3.2 | 3.2 | 3.2 | 3.2 | 20.0 | |||||||
| 7.5 | 7.5 | 15.0 | |||||||||||
| 5.0 | 10.0 | 15.0 | |||||||||||
| 14.8 | 14.8 | ||||||||||||
| 4.0 | 4.0 | 4.0 | 12.0 | ||||||||||
| 11.6 | 11.6 | ||||||||||||
| 8.1 | 8.1 | ||||||||||||
| 6.8 | |||||||||||||
| 5.0 | |||||||||||||
| 5.0 | 5.0 | ||||||||||||
| 2.2 | 2.3 | 4.5 | |||||||||||
| 2.5 | 1.9 | 4.4 | |||||||||||
| 4.3 | 4.3 | ||||||||||||
| 368.5 | 396.5 | 368.3 | 591.2 | 452.4 | 628.9 | 624.2 | 557.1 | 513.5 | 557.7 | 991.0 | 1,024.4 | 1,468.5 | 11,174.0 |
| 9 | 8 | 9 | 8 | 11 | 18 | 21 | 19 | 19 | 22 | 29 | 29 | 30 |
- Pre-Application Screening, Review, and Invitation to Submit Full Application (Chapter 4): Applications must be submitted individually in response to a specific program announcement; no other applications are accepted. Most programs use a pre-application step to reduce the number of full applications to be reviewed. Pre-application submission requirements and screening criteria are specified in the program announcement. Some program announcements for specific award mechanisms, such as the Clinical Consortium Research Site Award, use letters of intent as the pre-application and do not have screening criteria. Pre-applications are typically submitted and screened by the programmatic panel during months 5–8. Applications that meet the pre-application screening criteria, as determined by the programmatic panel, are then invited to submit a full application.
- Peer Review (Chapter 5): Between months 7 and 11, full applications are received and undergo review for scientific and technical merit by the peer review panel; this represents the first tier of CDMRP’s two-tier review process. The results of the peer review (both numeric scores and narrative summaries) are submitted to the programmatic panel.
- Programmatic Review (Chapter 6): Following peer review, applications are reviewed by the same programmatic panel as in steps 1 and 3 to determine programmatic relevance and portfolio balance, in addition to considering the scores and summaries from peer review. Programmatic review (months 9–14) serves as the second tier of the two-tier review process and results in a list of applications that are recommended for funding, along with a list of alternate applications that may be considered for funding if sufficient funds become available.
- The committee notes that CDMRP established an Inquiry Review Panel to address questions and appeals by applicants regarding either peer or programmatic review of their applications. The committee was informed that less than 0.5% of
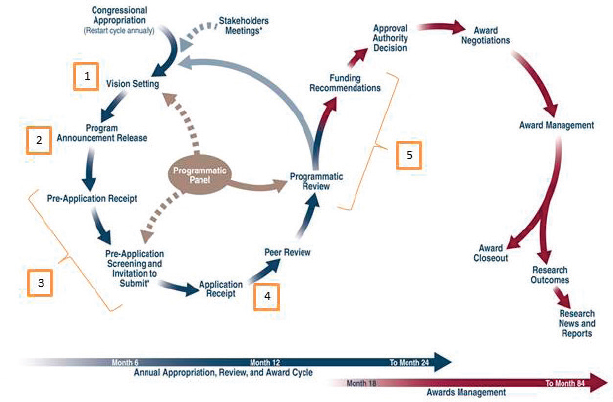
* As needed.
SOURCE: Adapted from Salzer, 2016a.
-
applications have re-review requests (Colonel Wanda Salzer, CDMRP director, personal communication, October 16, 2015).
- Once the list of funding recommendations has been completed, it must be approved or concurred with by the commanding general, USAMRMC, and/or the DHA’s Research, Development and Acquisition Directorate.
Awards that have been approved for funding move into the award-management phase (the right side of Figure 2-4 in red). Award management begins with award negotiations, which can take more than 1 year to complete (months 11–24). Finally, once an award is in place, it is actively managed and monitored by CDMRP throughout its duration. Award management includes, but is not limited to, progress reports from the research institution and financial reporting. When an award’s end date approaches, preparations are made to close the award, and the closure is finalized approximately 6 months after the period of performance has expired.












