4
Current and Future Directions of Low Dose Radiation Research
The symposium organizing committee invited a number of experts to provide overviews of current and future directions of specific research areas in radiation epidemiology and radiation biology. The organizing committee’s goal was that these experts would provide a synthesis of representative sources and discuss how the experts’ own work fits with better understanding risks at low radiation doses.
4.1 RADIATION EPIDEMIOLOGY
Current risk estimates for cancer and non-cancer diseases due to radiation rely extensively on findings from radiation epidemiology studies. These studies have been the most informative for estimating health risks in moderate and high doses. However, many have questioned whether epidemiology has reached its limit in characterizing disease risks at low doses of radiation because often the background levels of the same diseases are high and because risks at low levels of radiation are expected to be low. A new era of large-scale radiation epidemiology studies of medically, environmentally, and occupationally exposed populations has improved our understanding of risks at low doses of radiation (Kitahara et al., 2015). Some of these studies were highlighted by the symposium presenters and are summarized in the following sections.
4.1.1 Research Activities at the Radiation Effects Research Foundation (RERF)
Studies of the atomic bombing survivors of Hiroshima and Nagasaki began in 1947 with the founding of the Atomic Bomb Casualty Commission (ABCC) by the National Academy of Sciences. In 1975 the ABCC became RERF, a joint U.S.-Japan research organization. The Department of Energy (DOE) and the Japanese Ministry of Health, Labor and Welfare co-fund RERF, and the National Academies support RERF activities through a cooperative agreement with DOE.
The Life Span Study (LSS) is the major epidemiological study conducted at RERF that follows approximately 120,000 men and women of all ages who were either atomic bombing survivors and residents of Hiroshima and Nagasaki or were not in either city at the time of the bombings. The Adult Health Study is a clinical examination program of a subsample of about 20,000 members of the LSS who were invited to participate in biennial health examinations. RERF also conducts in utero studies and follow-up studies of the children of the survivors (F1 studies) conceived after the bombings. For all of these cohort members there is information on estimated individual radiation doses from the atomic bombings as well as information on the risk of various health outcomes following exposure. Findings from the epidemiological studies conducted at RERF have been an important source of scientific information on health effects of exposure to ionizing radiation in humans and for the establishment of international radiation protection standards.
The LSS was constructed to include as many survivors proximally exposed to the atomic bombings as possible. These proximal survivors received high doses of radiation. Despite the large size of the cohort, radiation risks at low doses are difficult to estimate due to weak statistical power. Dr. Bob Ullrich provided additional explanations of why low dose radiation risk studies using the RERF cohorts are difficult.
First, individual dose estimates are less certain for survivors who were farther away from the epicenter and therefore received low doses (distal survivors), because detailed information on their shielding conditions is not available. Second, differences in urbanicity between proximal and distal survivors could confound the association between radiation dose and adverse outcomes such as cancer. Third, among distal survivors, medical radiation exposures and residual radiation exposures may affect dose–response analyses because exposures become larger compared to direct exposure from the bombs.
The most recent analysis of solid cancer incidence among the LSS cohort (Grant et al., 2017) added 11 years of follow-up to the previously reported work (Preston et al., 2007) and reconfirmed that a single acute
exposure increases solid cancer risks for life. It also showed a dose response for all solid cancers that RERF investigators reported as significant down to the range of 0–100 milligray (mGy). The sex-averaged excess relative risk (ERR)/Gy was equal to 0.47 and consistent with ERRs reported at previous solid cancer incidence analyses (Preston et al., 2007; Thompson et al., 1994).
Dr. Preston, who serves as a consultant to the Department of Statistics at RERF, added that reports of cancer incidence and mortality among the LSS cohorts include a series of analyses to investigate the low dose linear slope and evidence of curvature in data restricted over various dose ranges. The radiation dose response for all solid cancers as a group was previously observed to be linear with no evidence of a threshold. However, findings from the most recent analysis were not fully consistent with those previously reported. Specifically, for females, the dose response remained consistent with linearity, but for males, significant upward curvature over the full dose range was observed.
Dr. Ullrich noted that continuing follow-up of the atomic bombing survivors offers additional opportunities to refine site-specific cancer risk estimates and to understand the joint effects of radiation and other factors on cancer risk, but also to produce new knowledge as the time of followup and statistical power increase. He presented highlights of several recent analyses that helped improve current understanding of site-specific cancer risks following single acute exposure including for lung (Cahoon et al., 2017), colorectal (Sugiyama et al., 2019), breast (Brenner et al., 2018), uterine (Utada et al., 2019), and liver (Sadakane et al., 2019) cancers. A linear dose response was observed for all of these cancer sites. In addition, Dr. Ullrich highlighted the influence of age at exposure on the risks for breast (Brenner et al., 2018) and uterine cancers (Utada et al., 2019).
Dr. Preston commented on the need for an update on previous findings on in utero exposure among the atomic bombing survivors. A previous study showed that in utero exposure to radiation increases risks of adult-onset cancers but suggested that lifetime risks following in utero exposure may be considerably lower than for early childhood exposure (Preston et al., 2008). Dr. Preston said that a study of in utero exposure following the atomic bombings to non-cancer endpoints is also warranted.
A number of symposium participants were interested in the longitudinally collected biosamples stored at RERF because they are a valuable resource that can allow for the study of underlying mechanisms of radiation-induced carcinogenesis. Dr. Ullrich noted that as of May 2019, RERF has stored about 1.8 million tubes of biosamples (750,000 tubes of serum, 370,000 of plasma, 500,000 of blood cells, and 210,000 of urine) donated by more than 30,000 study participants. The biosample collection includes 1,000 parent–child trios in which at least one parent was exposed
to radiation from the atomic bombings. Analysis of the trio samples can provide the opportunity to investigate heritable and de novo mutations and generate new data on the heritable effects of parental radiation exposure.
RERF is exploring mechanisms to expand research opportunities using the biosamples to the broader scientific community through collaborations. Use of the biosamples stored at RERF is currently being discussed with the Stakeholder Committee on Usage of A-bomb Survivors’ Biosamples. These discussions will inform the development of policies regarding biosample use at RERF.
4.1.2 Occupational Studies
Dr. David Richardson (University of North Carolina) provided an overview of occupational studies of radiation workers and their impact on understanding risks at low radiation doses. He noted that the launching of some of the major occupational studies of radiation workers occurred with the anticipation that potential risks, if any, could not be revealed using these study designs. That did not end up being the case and, instead, occupational studies proved useful in quantifying the risks associated with protracted radiation exposures and can help strengthen the foundation for radiation protection standards for millions of workers worldwide who are externally exposed to ionizing radiation on the job. These workers include those who conduct medical radiological procedures and those employed in the nuclear fuel cycle, in industrial radiography, and in defense activities.
Recognition of external ionizing radiation as an occupational carcinogen followed a classical path taken for the identification of many environmental and occupational carcinogens. Case reports began to appear shortly after the discovery of X-rays and described malignant skin tumors caused by occupational external ionizing radiation exposures (Frieben, 1902). These case studies were followed by reports that described the relative mortality of radiologists in the United Kingdom and the United States compared to physicians not exposed to radiation due to their profession (Court-Brown and Doll, 1957; Seltser and Sartwell, 1965). However, it was the Manhattan Project that led to large industrial cohorts of radiation-exposed workers and facilitated quantitative analytical studies of health effects associated with occupational external radiation exposures. By the 1970s and 1980s cohorts had been enumerated at several nuclear facilities, and national cohorts had begun to be assembled based on registries of radiation workers (Gilbert and Marks, 1979; Wing et al., 1991).
Although these cohorts directly estimated the effects of low dose chronic exposure to external radiation, the sample size has limited the precision of these estimates. Pooled analyses of nuclear worker cohorts, such as those from North America and Europe (Cardis et al., 2005; Laurier et al., 2016),
have suggested positive associations between cumulative external ionizing radiation dose and cancer across relatively low ranges of cumulative doses (see Table 4.1).
Another study of nuclear workers that was not described by Dr. Richardson but was mentioned by a number of other symposium participants is the Million Worker Study, an epidemiological study of 1 million U.S. radiation workers and veterans aiming to examine the lifetime risk of cancer following relatively low dose exposures received gradually over time. The study was conceptualized and directed by former National Council on Radiation Protection and Measurements president Dr. John Boice and is currently funded by DOE. The study is expected to be completed in 4 years, provided that funding is available.
Dr. Richardson said that, looking forward, epidemiological studies of radiation-exposed workers will broaden to encompass increasing numbers of female workers in the nuclear industry and will reflect other changes in demographics and exposures among radiation workers that will contribute to a fuller understanding of radiation risks. New computational approaches to handling measurement error and missing data, and new statistical approaches to strengthen inferences in longitudinal analyses, will accompany and strengthen large-scale collaborative studies on this topic. Dr. Richardson added that large cohort studies are likely to be complemented by efficient nested studies that permit collection of more refined exposure and outcome information.
4.1.3 Environmental Exposure Studies
Dr. Preston stated that in his view, studies of populations with environmental radiation exposures provide the most relevant estimates for radiation protection. These studies generally involve unselected populations that include men and women of all ages who typically received low and often chronic exposures.
There are several studies of populations with environmental radiation exposures for which individual dose estimates are available. These include studies of
- Weapons production and testing. Examples are the Techa River Cohort of individuals residing in riverside villages in the Southern Urals region of Russia between 1950 and 1961 when releases from the Mayak plutonium production complex contaminated the river, the Utah studies of groups exposed to fallout from atmospheric nuclear weapons testing, and the Hanford study of thyroid disease among people who were exposed to radioactive iodine from the Hanford Nuclear Site in Washington State.
TABLE 4.1 Summary of Some Recent Occupational Cohort Studies
| Study | Outcome | Sample Size | Cases | Mean Dose | ERR per 100 mGy | 95% CI | Reference |
|---|---|---|---|---|---|---|---|
| Korean workers | Cancer mortality | 79,679 | 134 | 6 mSv | 0.72 | –0.5 to 2.1 | Ahn et al., 2008 |
| Korean nuclear workers | Cancer incidence | 16,236 | 203 | 20 mSv | 0.21 | –0.19 to 0.9 | Jeong et al., 2010 |
| Rocketdyne employees | Cancer mortality | 46,970 | 647 | 14 mSv | 0.02 | –0.18 to 0.17 | Boice et al., 2011 |
| Japanese workers | Cancer mortality | 200,583 | 2,636 | 12 mSv | 0.13 | –0.03 to 0.30 | Akiba and Mizuno, 2012 |
| Canadian workers | Cancer mortality | 45,316 | 437 | 22 mSv | 0.18 | –0.04 to 0.53 | Zablotska et al., 2014 |
| German nuclear workers | Cancer mortality | 8,972 | 115 | 20 mSv | –0.1 | –0.4 to 0.1 | Merzenich et al., 2014 |
| U.S. radiologic technologists | Breast cancer | 66,915 | 1,922 | 37 mGy | 0.07 | –0.005 to 0.19 | Preston et al., 2016 |
| U.S. nuclear workers | Cancer mortality | 119,195 | 10,877 | 20 mSv | 0.01 | –0.02 to 0.05 | Schubauer-Berigan et al., 2015 |
| UK national registry of radiation workers | Cancer incidence | 174,451 | 11,133 | 25 mSv | 0.03 | 0.004 to 0.05 | Muirhead et al., 2009 |
| French nuclear workers | Cancer mortality | 59,004 | 2,536 | 26 mSv | 0.04 | –0.04 to 0.13* | Fournier et al., 2016 |
| INWORKS | Cancer mortality | 308,297 | 17,957 | 21 mGy | 0.05 | 0.018 to 0.079* | Richardson et al., 2015 |
NOTES: CI = confidence interval; ERR = excess relative risk; INWORKS = International Nuclear Workers Study of nuclear industry workers in France, the United Kingdom, and the United States. *90% CI.
- Nuclear power plant accidents such as the Three Mile Island accident in 1979 and the Chernobyl disaster in 1986.
- Natural and manmade background radiation. Examples are studies of populations living in high-background radiation areas in Kerala, India, and Yangjiang, China, and those of health effects following contamination of 1,700 apartments in Taiwan with cobalt-60.
Findings from main studies in the above categories are presented in Table 4.2. Notably, Dr. Preston did not discuss studies of populations exposed to the Fukushima nuclear power plant accident, because as he explained, such a study with dose information has not been designed. He thought that, despite the anticipated large costs of such a study, this was a missed opportunity for post-accident research, especially on the psychological impacts of the accident. In his view the approach followed by the Fukushima Prefectural Government to conduct a health management survey for all Fukushima residents (about 2 million people) is impractical because it relies on long self-administered questionnaire surveys and places the burden of data collection on the participants. As a result, participation rates have been low1 and therefore the confidence in research findings from the survey is also low.
Dr. Preston recognized that environmental exposure studies present a number of challenges associated with their design, including identification, tracing, and case finding; potential biases; dose reconstruction and dose uncertainty; and a likely lack of statistical power.
4.1.4 Medically Exposed Populations
Dr. Amy Berrington de González (National Cancer Institute) summarized studies on medically exposed cohorts and their contributions toward clarifying long-term health risks of protracted radiation exposures from diagnostic medical imaging procedures. She noted that historical cohort studies from the 1910s to the 1960s such as the Massachusetts and Canadian fluoroscopy cohorts showed increases in breast cancer risk among women exposed repeatedly over many years to medical radiation for tuberculosis (Boice et al., 1991; Howe, 1995; Howe and McLaughlin, 1996). The average tuberculosis patient received 88 chest fluoroscopic procedures over several years, equating to a mean estimated total dose of 0.8 Gy. A later study with lower mean estimated total doses examined a population of women whose spinal disorder was diagnosed and monitored by frequent X-ray examinations during childhood and adolescence and also showed
___________________
1 As described in a National Academies Proceedings of a Workshop (NASEM, 2019), participation rates for the basic survey of all individuals in Fukushima was 27 percent.
TABLE 4.2 Summary of Some Environmental Exposure Studies
| Study | Type | Cancer | Cases | Cohort Size/Controls | Mean/Median Dose (mGy) | ERR at 100 mGy | 95% CI | References |
|---|---|---|---|---|---|---|---|---|
| Weapons Production and Testing | ||||||||
| Techa River | Cohort | Solid Incidence | 1,933 | 17,435 | 52/15 | 0.08 | (0.01; 0.15) | Davis et al., 2015 |
| Techa River | Cohort | Solid Mortality | 2,303 | 29,730 | 35 | 0.06 | (0.004; 0.13) | Schonfeld et al., 2013 |
| Techa River | Cohort | Leukemia | 72 | 28,233 | 250/410 | 0.22 | (0.08; 0.54) | Krestinina et al., 2013 |
| Hanford I-131 | Cohort (prevalence) | Thyroid | 19 | 3,191 | 174/97 | 0.07 | (–0.03; 0.6) | Davis et al., 2004 |
| Utah Fallout | Case-Control | Leukemia | 939 | 4,302 | 2.9 cases 2.7 controls | 4.5 | (–0.4; 14) | Stevens et al., 1990 |
| Nuclear Power Plant Accidents | ||||||||
| Three Mile Island | Cohort | Solid incidence | 1,643 | 21,494 | 0 | –1 | (–1; 30) | Han et al., 2011 |
| Chernobyl-Ukraine | Cohort | Thyroid | 65 | 12,415 | 650/200 | 0.19 | (0.04; 0.63) | Brenner et al., 2011 |
| Chernobyl-Ukraine | Case-Control | Thyroid | 66 | 835 | cases 44 controls 16 | 4.9 | (0.5; 12) | Kopecky et al., 2006 |
| Chornobyl-Belarus | Cohort | Thyroid | 87 | 11,970 | 560/230 | 0.21 | (0.08; 0.54) | Zablotska et al., 2010 |
| Chernobyl-Belarus, Ukraine, Russia | Case-Control | Leukemia | 421 | 824 | 11/0.9 (cases) 4/0.7 (controls) | 3.2 | (0.9; 8.4) | Davis et al., 2004 |
| Natural and Manmade Background Radiation | ||||||||
| United Kingdom | Case-Control | Leukemia | 27,447 | 36,793 | 12 | (3; 22) | Kendall et al., 2012 | |
| Yiangjing, China | Cohort | Solid | 941 | 31,604 | 85 high 22 low | –0.1 | (–0.25; 0.3) | Tao et al., 2012 |
| Switzerland | Cohort | Cancer | 1,782 | 2,093,660 | 9 | 3 | (1; 5) | Spycher et al., 2015 |
| Cohort | Leukemia | 530 | 2,093,660 | 9 | 4 | (0; 8) | ||
| Cohort | Lymphoma | 328 | 2,093,660 | 9 | 1 | (–4; 5) | ||
| Finland | Case-Control | Leukemia | 1,093 | 3,027 | 1.96 cases 1.90 controls | –1 | (–1; 6) | Nikkilä et al., 2016 |
| Kerala, India | Cohort | Solid cancer | 1,349 | 69,958 | 110 | –0.01 | (–0.06; 0.05) | Nair et al., 2009 |
| Taiwan Buildings | Cohort | Cancer | 117 | 6,242 | 48 | 0.19 | (0.01; 0.31) | Hwang et al., 2008 |
NOTE: CI = confidence interval; ERR = excess relative risk.
increases in breast cancer risk (Ronckers et al., 2008). The average scoliosis patient in this study received 27 spinal X-rays over several years, equating to a mean estimated total dose of 120 mGy.
More recent cohort studies have suggested that pediatric computed tomography (CT) scans are associated with increased cancer risks even when the mean estimated total doses are within the low dose range. The UK Pediatric CT scans cohort was the first to report risks of leukemia and brain tumors among children who were examined with CT between 1985 and 2002 (Pearce et al., 2012).
Dr. Berrington de González explained that this new era of epidemiological studies of low dose radiation facilitated by electronic record linkage and pooling of cohorts is promising in addressing critical gaps in knowledge of the long-term health risks of low dose radiation. This is because these studies maximize power by examining radiosensitive populations, that is, children, and radiosensitive cancers, that is, leukemia. Also, because they are based on electronic record linkages, they improve efficiency and avoid recall bias.
The EPI-CT study (Bernier et al., 2018) is a European pooled epidemiological study designed to quantify the risk of radiation-induced cancer from pediatric CTs. The study includes detailed dosimetry and additional data collection to further evaluate factors that can affect the interpretation of results from CT studies, such as reverse causation, confounding by predisposing factors and other causes, and possible effect modification.2 The study uses a common protocol to recruit more than 1 million patients in nine countries (including an expanded UK cohort) and uses electronic record linkage and linkage to population-based cancer and mortality registries to collect dosimetry and health information. Publications from this study are expected in 2020.
Dr. Berrington de González said that conducting large-scale studies on the health effects of CT scans in the United States is impeded by the lack of a centralized health care system and fragmentation of patient services across multiple health care providers. The USA Kaiser Pediatric Imaging Case-Control Study is enabled by an integrated health care system and aims to evaluate patterns of all medical imaging, not just CT scans, in childhood and following in utero exposures. Data collection for this study is still in progress and results are expected to be released within the next 3 to 5 years.
Dr. Berrington de González concluded her presentation by discussing new opportunities for studying the benefits and risks of medical diagnostic and therapeutic radiation, including (1) studies of noncancer effects
___________________
2 Studies by Pearce et al. (2012) and Mathews et al. (2013) have received criticism for not sufficiently addressing factors such as confounding by indication and reverse causation. For more information see Bosch de Basea et al. (2015).
following low dose exposures due to medical procedures, and (2) studies on the health effects of low dose radiation exposure of healthy tissue to neutrons following proton beam radiation therapy.
4.2 RADIATION BIOLOGY
Traditionally, radiation biology studies have been considered the most promising approach to understanding the effects of low dose radiation by examining interactions between radiation exposure and genetic material, cellular structures, and whole organisms. However, to date, there is no good approach for incorporating new knowledge from radiation biology in health risk estimates from low dose radiation.
DOE’s low dose radiation research program supported almost exclusively radiation biology research. Dr. Brooks noted that, in the first few years of the program, resources were primarily allocated to support molecular and cellular radiation biology studies. However, with time and in recognition of the complex biological behavior of cells, complex biological functions in tissues, and integrative levels of organization, resource allocation shifted to support animal studies.
Animal studies have the potential to improve understanding of risks at low doses by elucidating the mechanistic effects of radiation-induced carcinogenesis and other disease generation. The advantage of animal studies is that they allow for the investigation of the effect of a specific agent such as radiation in isolation because everything else, including genetic composition and diet, can be controlled. However, symposium participants noted that the appropriateness of extrapolating findings from animal studies to the human situation can be questionable. In addition, radiation exposure experiments on animals are expensive and time consuming and are only performed in a few irradiation facilities around the world.
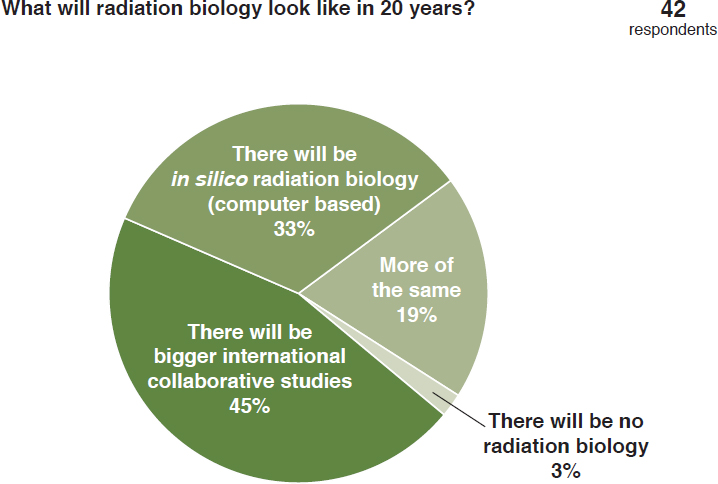
Symposium participants were asked to offer their opinions on the future of radiation biology research (see Figure 4.1). Most respondents thought that in the future there will be a change in how radiation biology research is conducted and that there will be bigger collaborative studies or that there will be in silico (computer-based) radiation biology experimentation.
The symposium organizing committee invited four experts to talk about current research in radiation biology and future directions.
4.2.1 Low Dose and Low Dose Rate Responses in Animals
Dr. Gayle Woloschak (Northwestern University) noted that within the period from the 1950s to the 2000s, laboratories in Europe, Japan, and the United States conducted extensive studies on low dose radiation research in a variety of animal model systems. These included mice, rats, beagle dogs,
and non-human primates. These historic studies were large. For example, at Argonne National Laboratory 7,000 beagles and 50,000 mice were used in experiments (Haley et al., 2011). The main goal of most of these experiments was to supplement information coming from human data, and they were focused on changes in life expectancy and cancer incidence following radiation exposure. These lifespan studies involved a wide range of irradiation doses, dose rates, and qualities.
More recent low dose radiation studies (from the 2000s to today) included fewer animals and focused on different genetic makeups, or on introduction of stresses before or after low dose radiation exposures. This research found that, molecularly and cytologically, the same low dose radiation exposures can be beneficial in specific disease contexts (e.g., regenerative neovascularization as found by Ministro et al. [2017]) and detrimental in others (e.g., neovascularization supporting tumor growth in SCID mice3 as found by Sofia Vala et al. [2010]).
The large investments in animal radiation research and the increasing interest in low dose radiation studies have stimulated recent efforts to archive data and tissue samples around the world and make them accessible
___________________
3 SCID mice are immunodeficient mice that are able to accept human hematopoietic stem cells harvested from human bone marrow or thymus.
to the broad scientific community. Zander and colleagues (2019) reviewed major archives and databases that have shaped the field of radiation biology.
Data and tissue samples from the earlier research on irradiated animals funded by the U.S. government have been deposited into the Northwestern University Radiation Tissue Archives (NURA). NURA is thought to be the world’s largest collection of irradiated animal tissue samples. The most recent additions to the archives are biosamples collected from animals that lived within a 20-kilometer radius of the Fukushima Daiichi Nuclear Power Plant and were euthanized to prevent consumption of contaminated meats. A comprehensive dose evaluation project concerning these animals has been set up (Takahashi et al., 2015).
The data and samples archived at NURA are available upon request to investigators from around the world who are looking to explore the effects of radiation. However, Dr. Woloschak noted that NURA’s operations are underfunded and therefore the ability to respond to tissue requests in a timely fashion is compromised. This underfunding is also affecting the timely archiving of available data and samples.
Dr. Woloschak noted that the increasing sensitivity and reliability of techniques in biology and the increasing ability to produce different radiation doses, dose rates, and radiation qualities in experimental designs will help enhance findings in low dose radiation research. In addition, new approaches for meta-analyses (e.g., literature re-evaluation and data reinterpretation) and new techniques for evaluation of archival biospecimens will enhance understanding of low dose radiation health effects by making use of already collected tissues from experiments that are unlikely to be recreated today.
4.2.2 Molecular Injury Responses
Dr. Al Fornace (Georgetown University) discussed studies conducted in his own laboratory related to molecular injury responses triggered by low dose radiation and their long-term implications in normal tissues. These studies have employed human cell lines, peripheral blood lymphocytes from patients irradiated ex vivo and in vivo, as well as mouse models and non-human primates. They have showed that cellular responses to radiation injury and other forms of genotoxic stress involve a complex network of signaling processes that contribute to recovery from injury but can also have adverse effects in vivo. Interestingly, many of these responses have been shown to be induced even by low dose radiation (Amundson et al., 1999). In the case of transcriptional responses, p53 signaling can be induced by doses of 100 mGy or less (Li et al., 2015). Using genetic and metabolomic approaches, the Fornace laboratory has shown that low doses of radiation can affect many tissues in vivo and can impact immune
function and energy metabolism by inhibiting specific signaling pathways (Kumar et al., 2018).
Long-term effects of radiation are known to be associated with immune dysfunction and pro-inflammatory signaling in many tissues and can lead to incompetent metabolic reprogramming in activated T-cells even at low doses (Li et al., 2015). After either low or high linear energy transfer low dose radiation, investigators at the Fornace laboratory have shown triggering of long-term stress signaling, elevated oxidative stress, increased senescent cells, and induction of a pro-inflammatory state characteristic of the senescence-associated secretory phenotype (Kumar et al., 2018).
Dr. Fornace noted that radiation-induced premature senescence is well known in the field. It has been shown that even a very small number of senescent cells can result in physical dysfunction. For example, senescent cells can impact normal somatic stem cell function with a variety of deleterious long-term consequences (Kumar et al., 2018). In his view, understanding the roles of low dose radiation in senescence, metabolism, inflammation, and immune function could help delineate a systematic view of the physiological impacts of low dose radiation. Incorporation of state-of-the-art approaches and models into low dose radiation research offers exciting opportunities to engage scientists in multidisciplinary research and to make breakthroughs in a variety of areas, from basic mechanistic science to practical applications including risk estimation and potential targets for risk reduction.
4.2.3 Epigenetic Alterations from Low Dose Radiation
Dr. Randy Jirtle (North Carolina State University) defined epigenetics as the study of the heritable changes in gene function that occur without a change in the sequence of DNA. He focused his presentation on experiments conducted in his laboratory using the Agouti viable yellow (Avy) isogenic strain of mice (see Figure 4.2). The Avy mouse model, in which coat color variation and disease susceptibility are correlated to epigenetic changes during gestation, has been used to investigate impacts on adult disease formation from nutritionally and environmentally induced influences in the fetal epigenome (Jirtle and Skinner, 2007).
Dr. Jirtle and colleagues showed that single low dose radiation exposures of embryonic stem cells at the blastocyst stage of development (i.e., 0.7 to 7.6 centigray [cGy]) induce a sex- and dose-dependent positive adaptive response in the offspring that results from alterations in DNA methylation at the Avy metastable epiallele locus (Bernal et al., 2013). Dr. Jirtle noted that these findings provide the first evidence that the phenomenon of radiation hormesis results from modifications in the epigenome. He added that these experiments also provided evidence that maternal dietary antioxidant supplementation mitigates these DNA methylation changes,

indicating that they are mediated in part by oxidative stress. Thus, in his view, the interesting possibility exists that nutritional supplementation may be useful in altering radiation responses at low doses.
Dr. Woloschak and Dr. Fornace expressed concerns about the use of these experiments as a foundation of supporting hormesis and pointed to other studies in the literature that have shown detrimental effects of low dose radiation. Dr. Jirtle responded that such variation in biological responses is expected because phenotypic changes due to epigenetic alterations are not only dose dependent but also can be modulated by environmental factors such as nutrition (Dolinoy et al., 2006).
Dr. Fornace questioned even the relevance of alterations in DNA methylation at the Avy metastable epiallele locus to radiation risk because no evidence exists linking it to cancer induction. At the same time, he acknowledged that radiation-induced epigenetic changes clearly occur and it is an area that warrants intensive investigation. In response to these comments, Dr. Jirtle reiterated that the experiments he presented indicate that further
research is needed to more clearly define the role of the epigenome in human biological responses to low dose radiation. A complete genomic atlas of metastable epialleles has recently been defined for humans (Gunasekara et al., 2019); therefore, such investigations are now possible. Dr. Jirtle proposed that research in the epigenome can be directed to answer the following questions:
- Do other frequencies of electromagnetic radiation and high-energy particles also alter the epigenome at low doses and, if so, can nutritional supplements modify the responses?
- What are the signal transduction pathways that link the formation of reactive oxygen species to altered DNA methylation?
- Does low dose ionizing radiation alter the human epigenome?
- Is the linear no-threshold model potentially inappropriate for estimating human risk from low doses of ionizing radiation?
4.3 NEW DIRECTIONS
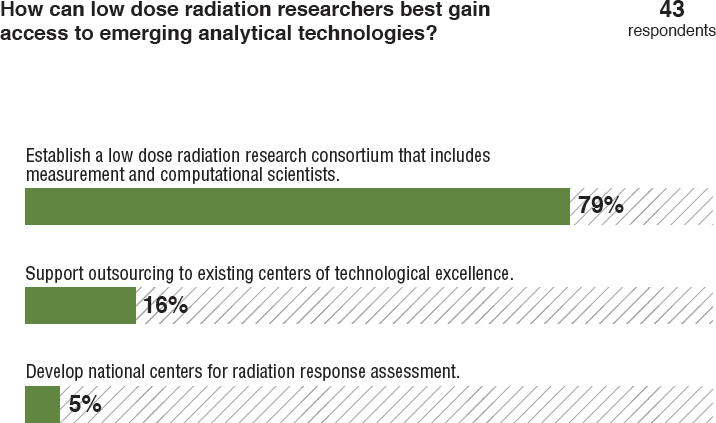
This section summarizes presentations that took place at the symposium on scientific advances and emerging analytical technologies that have the potential to revolutionize radiation research and provide important insights about the causality of health risks associated with low dose and low dose rate radiation exposures. Some of these advances are already being explored by radiation researchers but others are not. Dr. Joe Gray (Oregon Health & Science University) polled the symposium participants on their views on how researchers can best gain access to emerging analytical technologies. Most respondents thought that the best way is by establishing a low dose radiation research consortium that includes measurement and computational scientists (see Figure 4.3).
4.3.1 Biomarkers for Molecular Epidemiological Studies
A number of symposium participants noted that if biomarkers are applied and integrated in epidemiological studies, they would revolutionize radiation epidemiology and provide important insights about the causality of health risks associated with low dose and low dose rate radiation exposures. Dr. Kreuzer, who provided a presentation at the symposium on behalf of Janet Hall (French National Institute of Health and Medical Research), said that an increasing number of potential new biomarkers are being developed. These biomarkers fall within the following four categories (Pernot et al., 2012):
- Exposure biomarkers that become available at some point after exposure and are suitable for estimating the dose received;
- Biomarkers of susceptibility that are available before, during, or after exposure and can predict an increased risk in radiation effects;
- Biomarkers of late effects that can be used to assess the effects that are present in the late stages of a disease that was induced by radiation; and
- Biomarkers of persistent effects that allow the assessment of radiation effects that are present over a long period of time after exposure.
Dr. Kreuzer noted that the increasing number of potential new biomarkers underpins the need for guidelines for biomarker validation and for deciding which biomarkers to carry forward from discovery to implementation. Hall et al. (2017) published a roadmap to aid in this decision-making process. This roadmap took into account factors such as assay reproducibility, sensitivity, and specificity for radiation exposure. It was then used to summarize the current status of proposed biomarkers for epidemiological studies investigating low dose health effects.
Hall and colleagues concluded that most potential biomarkers remain at the discovery stage and that robust validation will be essential before
implementation (Hall et al., 2017). Radiation-specific mRNA transcript profiles were identified as potential biomarkers in the final stages of development and are a priority for further research. Dr. Kreuzer found that the challenge moving forward is validating the use of these biomarkers in prospective studies.
4.3.2 Technological Advancements
Dr. Gray (see Figure 4.4) noted that advancements in multiscale imaging in the past decade have dramatically increased the ability to quantify the components and multiscale architectures of cells and tissues, leading to new insights into disease genesis and behavior. He said that these technological advancements allow for a “spatial systems biology approach” to understanding human disease genesis and behavior. Based on this approach, diseases such as cancer are viewed as a manifestation of aberrant multiscale interactions of biological structures. He illustrated the utility of five technologies for quantifying these interactions and described their applications to assessment of human cancerous tissues:
- Single-cell topographic sequencing to quantify the consequences of interactions. This method has been used to measure genomic copy number profiles of individual tumor cells while preserving their spatial context in tissue sections (Casasent et al., 2018);
- Multiplex immunostaining and image analysis to quantify the cellular composition, molecular states, and organization of complex tissues (Goltsev et al., 2018; Lin et al., 2018; Tsujikawa et al., 2017);
- 3D electron microscopy to visualize nanoscale inter- and intracellular interactions in tissues (López et al., 2017);
- Superresolution fluorescence microscopy to study the dynamics of nanoscale interactions (Guo et al., 2018; Möckl et al., 2014); and
- Single-particle cryoelectron microscopy to visualize molecular conformations with few-angstrom resolution and interactions between structures (Cheng et al., 2017; Earl et al., 2017; Zhao et al., 2019).
Dr. Gray noted that most of the above-mentioned technologies have been applied to human tissues and can be considered for use in the assessment of radiation health effects. He added that researchers interested in applying these technologies can make use of information produced by large consortia and programs such as the Human BioMolecular Atlas Program4
___________________
4 See https://commonfund.nih.gov/hubmap (accessed October 16, 2019).

and the Human Tumor Atlas Network,5 which aim to map healthy and cancer cells, respectively, in the human body and determine how the relationships between cells can affect the health of an individual.
Dr. Sadik Esener (Oregon Health & Science University) focused his presentation on technological advancements in isolating and studying the role of various nano-particulates that circulate in body fluids such as extracellular vesicles (EVs)6 and cell free (cf) proteins, DNA and RNA, and their potential use in elucidating systemic effects of low dose radiation. Until recently, isolation of these nano-particulates has been difficult because of their small size and low density. Researchers at the University of California, San Diego, developed a new technology that allows for their quick isolation
___________________
5 See https://www.cancer.gov/research/key-initiatives/moonshot-cancer-initiative/implementation/ human-tumor-atlas (accessed October 16, 2019).
6 EVs are membrane-coated bodies actively released by various cell types.
from small amounts of blood (50 mL). The technology involves a chip that contains hundreds of small electrodes that generate a rapidly oscillating electric field that selectively pulls the nanoparticles out of a plasma sample. Dr. Esener noted that this new nanoparticulate separation technology has the potential to enable researchers to study the role of EVs and cfDNA in radiation environments.
Changes in the levels of free circulating nucleic acids (Schwarzenbach et al., 2011) have been linked with several types of cancer and therefore are being explored as potential blood biomarkers. A group demonstrated that oxidized cfDNA released from dying cells exposed to radiation could generate adaptive responses and radiation-induced bystander effects (Sergeeva et al., 2017). Another study investigated the role of bone marrow–derived EVs in mediating systemic radiation-induced bystander effects in vivo and found that EVs could transmit biological information from irradiated to unirradiated cells (Szatmári et al., 2017).
4.3.3 Single-Cell Genomics
The consequences of exposures to low doses of mutagens on single human cells and how those changes alter tissue health are largely unknown. This has been mainly due to a lack of availability of technical tools to study global changes in the nucleic acid content of single cells. Dr. Charles Gawad (St. Jude Children’s Research Hospital) provided an overview of recent progress in the development of single-cell sequencing methodologies for DNA and RNA and listed a number of questions researchers can address by studying gene–environment interactions at a cellular level:
- Does exposure to specific environmental agents, including radiation, alter cell states?
- Does it alter tissue homeostasis and increase risk of disease?
- How many mutations are introduced in a cell after exposure to a specific environment?
- Do mutagens have distinct signatures of genomic damage?
- Are there patterns of genetic damage that are enriched in specific tissues, as well in certain cell types within a tissue?
Dr. Gawad said that initial work is needed to see if single-cell genomics is currently sensitive enough to detect changes induced by exposure to low doses of mutagens, such as low dose radiation. However, the technologies are continuously improving, so he thought that if not now, it will likely be possible in the near future. He identified the investigation of changes in germ
cells as important to study due to effects on all of the cells of an offspring. Additional single-cell technologies are being developed that will allow scientists to address more complex questions. These include technologies that allow in situ sequencing of single cells within tissue sections, sequencing the genome and transcriptome from the same cell to simultaneously determine genome changes and cell states, and single-cell epigenomics.
Dr. Gawad closed his presentation by discussing his laboratory’s recent development of primary template-directed single-cell whole genome amplification (PTA), which enables the accurate detection of about 85 percent of genetic variation in each individual cell. The Gawad laboratory has applied PTA to map genome-wide interactions of mutagens with single living human cells at base-pair resolution and is currently using PTA to examine the mutagenesis rates and signatures of environmental compounds.
4.3.4 Immune Responses
Dr. Silvia Formenti (Weill Cornell Medical College) discussed the effects of focal radiation therapy, beyond that of a therapy that merely aims to accomplish local control of tumors (Formenti and Demaria, 2013). She noted that accumulated evidence exists in support of metastatic cancer regression at a distance from the irradiated fields, known as the abscopal effect. The biological characteristics of the abscopal effect are not completely understood, but more recently this phenomenon has been postulated to be a result of the activation of the immune system induced by radiation therapy (Abuodeh et al., 2016). Dr. Formenti noted that the abscopal effect opens the door for research in the ability of radiotherapy to enhance responses to immunotherapy by “turning the tumor into an individualized cancer vaccine.”
Experiments in syngeneic mouse models7 that mimic the setting of advanced human cancers have demonstrated promising outcomes of combining radiation therapy with immunotherapy (Demaria et al., 2005). Dr. Formenti said that the preclinical data have been confirmed clinically and researchers have reported cases of the abscopal effect in patients with melanoma and lung cancers treated with ipilimumab8 and radiotherapy (Formenti et al., 2018; Postow et al., 2012). Currently, multiple clinical trials are exploring optimal combinations and scheduling of focal
___________________
7 The mouse models appropriate to study the interaction between different types of tumors and the immune system.
8 Ipilimumab is a monoclonal antibody that inhibits an immunologic checkpoint on T-cells, cytotoxic T-lymphocyte–associated antigen 4.
radiotherapy and immunotherapy. A recent report showed that the choice of radiation dose and fractionation is important to convert unresponsive patients into responders to immunotherapy (Vanpouille-Box et al., 2017).
Dr. Formenti noted that the immune effects of low dose total body exposure to radiation therapy remain poorly understood. For instance, it is unknown whether scattered dose to the rest of the body following radiation therapy contributes to systemic effects. Dr. Formenti pointed to one study in mice that showed that low doses of radiation had a different effect on apoptosis of mouse splenocytes isolated from the irradiated mice compared to high doses. Specifically, exposure to 2 Gy increased apoptosis in all splenocyte subpopulations in mice but low doses decreased apoptosis in some subpopulations (Bogdándi et al., 2010). This finding has been interpreted by some as evidence of a potential hormetic effect of low doses of radiation on the immune system (Cui et al., 2017).
4.3.5 Systems Biology
Dr. Francis Cucinotta (University of Nevada, Las Vegas) said that definitions of systems biology range from the description of the behavior of complex biological processes in terms of molecular constituents to the use of functional -omics and pathway modeling in multi-scale descriptions of biological processes in cells and tissues. He added that systems biology relies on computational and mathematical modeling of complex systems that use engineering concepts such as motifs, modularity, robustness, and positive and negative feedback controls. A recent review of the computational methods and tools currently available to systems biologists demonstrates the range of modeling and analysis techniques employed in the field (Bartocci and Lió, 2016). An example of a systems biology approach to examine the broad effects of immunotherapy was conducted by Spitzer and colleagues (2017). The study demonstrated the critical impact of systemic immune responses that drive tumor rejection that would not have been revealed by focusing on local immune responses in the tumor microenvironment.
Dr. Cucinotta defined two additional terms: (1) systems radiation biology, which is the application of systems biology in the study of radiation effects, and (2) translational systems radiation biology, which is risk prediction at low dose based on scientifically sound extrapolation of experiments.
Dr. Cucinotta demonstrated an in silico modeling approach by his laboratory to study radiation damage to neurons following charged particle exposures such as those to patients undergoing radiation therapy and astronauts during space travel (Alp et al., 2015). Using this approach, his group described for the first time the spatial dependence of a particle’s
microscopic dose deposition events on a detailed neuron structure and provided a model that can be used to support the development of biophysical models of the modifications of spine dendritic morphology observed after low dose charged particle irradiation.
In closing, Dr. Cucinotta noted that systems biology research provides excellent opportunities for experimentalists and theorists to interact.
4.3.6 Microbiome
Dr. Eleanor Blakely (Lawrence Berkeley National Laboratory) defined the microbiome as the collection of resident microorganisms in the body. The human microbiome resides in 18 different sites, orifices, and surfaces of the body and contains more than 1014 microbes. The majority of microbes are distributed in the gastrointestinal tract. Each individual has a unique microbiome modified by age, environmental exposures, lifestyle, diet, and other factors.
Studies in mice aimed to investigate the role of radiation in altering the microbiome during deep-space travel showed that whole-body exposure to various levels of high linear energy transfer radiation induced substantial changes in the composition and functional potential of the gut microbiome. The investigators reported that, somewhat unexpectedly, the mouse gut microbiome was more sensitive to lower doses of radiation (0.1 and 0.25 Gy) as compared to the highest dose (1 Gy) (Casero et al., 2017). A second study also showed that combined exposure to sparsely ionizing protons and densely ionizing heavy ions altered the gut microbiome (Raber et al., 2019). In the Blakely laboratory, mice irradiated with a single low dose exposure to 40, 80, 160, or 320 mGy of silicon ions showed significant increases in segmented filamentous bacteria when tested 16 months later. These bacteria are known to be associated with T-cell regulation.
Dr. Blakely and colleagues see several opportunities for studying the effects of low dose radiation on the microbiome:
- To identify radiation biomarkers that assess levels and types of radiation exposure;
- To identify host gene, protein, and microbial targets for development of medical treatments;
- To provide the evidence needed to predict disease risk after radiation exposure and by incorporating individual variability in genes, environment, and lifestyle for each person; and
- To define the mechanism by which radiation exposure affects health risk using multi-scale and multi-omics “systems biology” approaches across different model systems.
Recognizing the complex interplay between host genetics and exposures on the microbial and metabolic composition of the gut, researchers at Lawrence Berkeley National Laboratory have used a population-based mouse model known as the “Collaborative Cross” (Mao et al., 2015; Snijders et al., 2016) to start answering questions about the mediating effect of the microbiome on radiation exposure–associated immune sensitivity.
























