3
Energy Sources, Conversion Devices, and Storage
ENERGY SOURCES, CONVERSION DEVICES, AND STORAGE
Power and energy (P&E) technology in its most basic form centers on energy sources, energy storage, conversion, and management functions. The overall goal is to use energy to provide the maximum operational advantage. How much energy can be stored, the source of that energy, and how efficiently it can be converted into power to perform work are key in the assessment of a particular P&E technology. Military operations stress each of these criteria far beyond commercial demands—military vehicles demand far higher power levels while sources and storage create critical logistical concerns. For these reasons, the committee reviewed and investigated several technology areas from military staples, such as jet propellant 8 (JP8), to future concepts, such as nuclear batteries and small reactors, and assessed their viability against the likely demands of the future operating environment.
Energy Density Is Critically Important
Figure 3.1 provides a useful comparison of gravimetric energy (function of weight) and volumetric energy density (function of volume) of the liquid and gaseous fuel sources that could be considered for battlefield deployment. Using a high energy-density fuel is critically important for the Army, because it determines the amount of fuel that must be logistically brought to the field and stored.
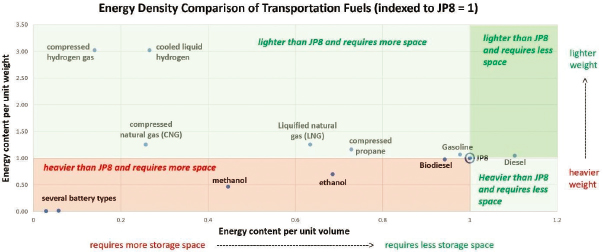
Other criteria that will be considered in evaluating alternative energy sources are safety, availability, ease of handling, and fuel conversion efficiencies.
Liquid Energy Sources
Liquid petroleum-derived fuels have more energy per unit volume (which determines the number of supply trucks) than any other transportation fuel. This high energy density ensures widespread use of petroleum-derived fuels throughout the military. In comparison, the energy density of batteries (roughly 0.7 MJ/kg) is significantly less than JP8 (44 MJ/kg). In addition, as previously discussed in the executive summary, refueling times using liquid fuels are significantly less than recharging times for batteries.1
JP8 versus Diesel
The energy density (per unit volume) of JP8 and diesel exceeds that of all other commonly used transportation fuels, such as gasoline, biodiesel, and compressed natural gas (Figure 3.1). This superiority has a direct impact on the number of trucks per supply convoy (or number of convoys) that deliver energy to the battlefield. Minimizing that fuel transport
___________________
1 U.S. Energy Information Administration (EIA), 2013, “Few Transportation Fuels Surpass the Energy Densities of Gasoline and Diesel,” https://www.eia.gov/todayinenergy/detail.php?id=9991.
also minimizes the number of soldiers and supporting personnel at risk during transport of that fuel.
Diesel has roughly 2 percent more energy per unit weight than JP8 and 9 percent more energy per unit volume. The higher energy-per-unit-volume of diesel is due to its higher density (i.e., 0.832 kg/L for diesel and 0.804 kg/L for JP8). As shown in Table 3.1, there are also some other important differences between JP8 and diesel, particularly in terms of cetane ratings, viscosity, and sulfur content.
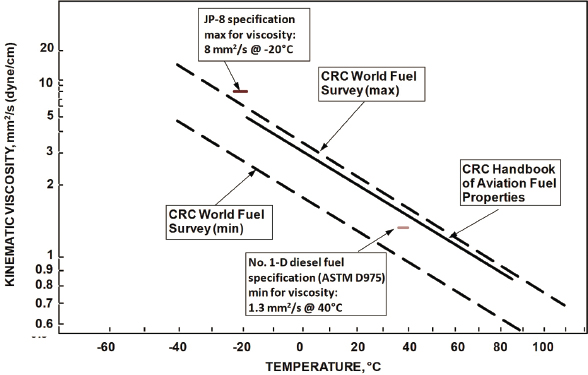
Viscosity
The maximum viscosity for JP8 is specified at −20°C, whereas the maximum viscosity for diesel fuel is specified at 40°C (see Figure 3.2). Under extremely cold environmental conditions, both diesel and JP8 can gel, with diesel being more susceptible to cold weather failure than JP8. Waxing refers to this situation, in which the paraffin hydrocarbons in the fuel congeal, forming wax-like particles that can either coat the surfaces they contact or plug fuel filters. For this reason, the diesel fuel available at service stations is typically a blend of DF1 and DF2, seasonally adjusted based on local ambient temperatures. DF1 is also known as winter diesel fuel because it performs better in cold temperatures. DF2 is typically used during summer conditions.
Sulfur Content
Because military vehicles are not required to meet the same emission standards as passenger and commercial vehicles, they have much simpler exhaust aftertreatment systems. Whereas passenger and commercial vehicles with diesel engines must use ultra-low sulfur fuel (i.e., 15 ppm) to prevent damage to their aftertreatment pollution control devices, the JP8 used in military vehicles can have a sulfur content of up to 3000 ppm.2
Cetane Rating
The biggest complaint about JP8 is the high degree of variability in its cetane rating, particularly at the lower end. Cetane is a measure of a fuel’s tendency to auto-ignite, with higher cetane being easier to auto-ignite than lower cetane. As shown in Figure 3.3, cetane ratings for JP8 vary widely with the source, whereas DF1 and DF2 diesel fuel require
___________________
2 P.A. Muzzell, 2011, “Alternative Fuels for Use in DoD/Army Tactical Ground Systems,” ARC Collaborative Research Seminar Series, U.S. Army Research, Development, and Engineering Command (RDECOM), https://apps.dtic.mil/dtic/tr/fulltext/u2/a537892.pdf.
TABLE 3.1 Diesel versus Jet Fuel
| Diesel Fuel Specification ASTM D975 | Jet Fuel Specifications | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Def Stan 91-91 / ASTM D1655 | MIL-DTL-83133G | MIL-DTL-5624U | ||||||||
| Fuel Grade | DF-1 | DF-2 | Jet A-1 | JP-8 | JP-5 | |||||
| Property (Unit) | Min | Max | Min | Max | Min | Max | Min | Max | Min | Max |
| Cetane Number | 40 | … | 40 | … | … | … | Report (Cetane Index) | Report (Cetane Index) | ||
| Viscosity @ 40°C (mm2/s) | 1.3 | 2.4 | 1.9 | 4.1 | … | … | … | … | … | … |
| Viscosity @ −20°C (mm2/s) | … | … | … | … | … | 8.0 | … | 8.0 | … | 8.5 |
| Density @ 15°C (kg/L) | … | … | … | … | 0.775 | 0.840 | .0775 | 0.840 | 0.788 | 0.845 |
| Sulfur Content (ppm) | … | 15 | … | 15 | … | 3000 | … | 3000 | … | 3000 |
| Flash Point (°C) | 38 | … | 52 | … | 38 | … | 38 | … | 60 | … |
| Lubricity HFRR @ 60°C (μm) | … | 520 | … | 520 | … | 0.85 BOCLE (mm) | … | 0.65* BOCLE (mm) | … | 0.65* BOCLE (mm) |
*As provided by minimum effective treat rate of mandatory lubricity improver additive per QPL-25017 and MIL-PRF-25017
SOURCE: P.A. Muzzell, 2011, “Alternative Fuels for Use in DoD/Army Tactical Ground Systems,” ARC Collaborative Research Seminar Series, U.S. Army Research, Development, and Engineering Command (RDECOM), https://apps.dtic.mil/dtic/tr/fulltext/u2/a537892.pdf.
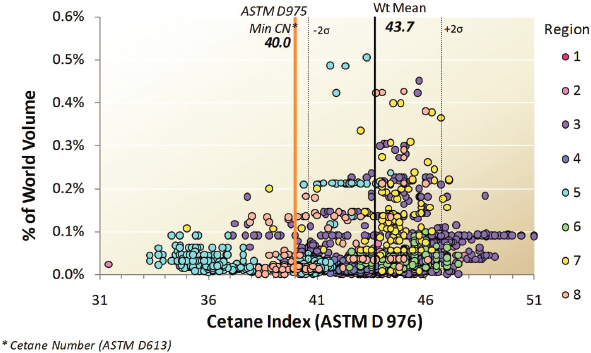
a minimum 40 cetane rating. Although the rating variability is not a problem with turbine-operated aircraft (or the turbine-operated Abrams tank), it can pose a problem for internal combustion engines, particularly in cold weather.
The cetane index of a fuel affects the engine’s ignition delay—that is, the time between the introduction of fuel and the first indications of heat release. Selecting the optimal injection timing has a major impact on fuel efficiency. Although this optimization is difficult to do on diesel engines with pump/line/nozzle fuel injection systems, optimal injection timing can be achieved with modern diesels employing direct fuel injection with in-cylinder pressure sensors. Auto-ignition and the impact of cetane rating are also important considerations for some advanced combustion technologies, such as homogeneous charge compression ignition and free piston engines.
Biodiesel
Biodiesel, a renewable, carbon-neutral fuel, is used commercially today as an alternative fuel to diesel. It is typically produced from rapeseed (predominant in Europe), soybeans (predominant in the United States), animal fats, and waste cooking oil. Biodiesel cetane ratings typically are around 55, while commercially available pump diesel cetane ratings typically run between 48 and 50.3
Unfortunately, pure biodiesel (i.e., not blended as a low percentage of DF2 diesel) can pose operational concerns, such as the fuel filter plugging or waxing experienced on selected vehicles under specific use profiles and ambient conditions. Hence, some sort of acceptability certification requirement for the various biodiesel sources would be required to assure reliable use in vehicles. There also might be expiration time limits on the fuel.4
Given the increasing urgency to address climate change, biodiesel (a renewable, carbon-neutral fuel) may serve as a preferred fuel source during peacetime as a reduction in greenhouse gases may be a more pressing concern than battlefield supply. The same technology that enables seamless transitions from JP8 to diesel could also enable JP8 to biodiesel transitions.
When the United States is engaged in a war, either JP8 or diesel are preferred fuel choices because both have higher energy density than biodiesel. Diesel has a 9 percent and 15 percent higher volumetric energy density than JP8 and biodiesel, respectively. The use of diesel or JP8 would require proportionately fewer supply trucks to carry the same
___________________
3 SeQuential, 2018, “Comparing Engine Wear: Petroleum and Biodiesel,” https://choosesq.com/blog/comparing-engine-wear-petroleum-and-biodiesel/.
4 J. Van Gerpen, 2005, “The Basics of Diesel Engines and Diesel Fuels,” Chapter 3 in The Biodiesel Handbook, Champaign, IL: AOCS Press.
amount of energy to the battlefield than biodiesel, thereby reducing lives potentially lost in supply convoys. During a military conflict, saving warfighter lives becomes a more important immediate concern than reducing greenhouse gases.5
Finding: Biodiesel may be a preferred fuel source during peacetime, given the growing need to address climate change. Certification for acceptability of the various sources would be needed to ensure any reliability concerns are addressed. (Tier 1, Lead)6
Gasoline
Gasoline has roughly similar energy content to JP8 on both a weight and volume basis. Gasoline is less desirable than JP8 or diesel as a fuel for military vehicles due to its lower flash point.7 Fuels with higher flash points are less flammable, contributing to a less hazardous situation and therefore improve safety and combat survivability. For comparison, the flash point for gasoline is roughly −45°F, whereas the flash point for JP8 is around 100°F.8 For instance, a match dropped into a pool of gasoline generally will ignite its vapors and continue to burn. A match dropped into a pool of diesel will extinguish itself. To create a diesel flame, a hot source is required, such as when a diesel fuel line leaks with the diesel falling on a hot exhaust manifold.
Within the combustion chamber of an internal combustion engine, gasoline is more difficult to auto-ignite than diesel. The standard measure of a gasoline sample’s difficulty in autoignition is its octane rating. From a fuel efficiency standpoint, higher octane ratings are preferred in gasoline engines because they are harder to auto-ignite, thereby allowing spark timing to be advanced providing the combustion energy released by the fuel to be exercised for a greater percentage of the expansion stroke. This desirability of higher octane rating for gasoline fuels is comparable to the desirability of higher cetane ratings for diesel fuels, which is a measure of a diesel sample’s ease of autoignition. Higher cetane ratings are preferred in diesels to ensure reliable and consistent ignition and cold weather starting in the absence of a spark-actuated combustion event.
___________________
5 EIA, 2013, “Few Transportation Fuels Surpass the Energy Densities of Gasoline and Diesel,” https://www.eia.gov/todayinenergy/detail.php?id=9991.
6 The committee’s findings, conclusions, and recommendations are categorized using its three-tiered view with respect to technology readiness levels (Tiers 1 to 3) and its methodology for assessing each technology area (lead, watch, follow), discussed in Chapter 2.
7 A liquid fuel’s flash point indicates the temperature at which existing vapors can combust and ignite.
8 B. Hagerty and S. Peranteau, 2005, “Vehicle Fluid Flammability Tests,” Fire and Arson Investigation, https://garrett-engineers.com/cases-of-the-month/what-auto-fluids-burn/.
Alcohols
Ethanol is typically produced from corn, grains, or agriculture waste (cellulose). Methanol is typically produced from natural gas, coal, or woody biomass. Ethanol and methanol have 69 percent and 45 percent of the energy content per unit volume of JP8, respectively, making them impractical as a sole source of fuel for a military ground vehicle.9
NATO Single Fuel Forward Policy
To date, the Army has relied heavily on JP8 as part of its “single fuel forward” policy—one military fuel on the battlefield across all ground vehicle platforms. In addition to being an Army fuel for ground vehicles, JP8 is a fuel for turbine-powered aircraft and is specified by MIL-DTL-83133 and British Defense Standard 91-87. It is similar to commercial aviation’s Jet A-1 fuel, but with the addition of a corrosion inhibitor/lubricity improver, icing inhibitor, and an antistatic agent. Optionally, a metal deactivation additive and antioxidant may be included in the formulation. In addition to being used as a fuel for ground combat vehicles and generators, JP8 is used as a fuel for heaters and stoves by the U.S. military and its NATO allies.10
This fuel was introduced in 1978 within NATO (with an F-34 fuel designation) in order to simplify the logistics supply chain for petroleum products. The primary goal of the single fuel policy (SFP) is to achieve equipment interoperability through using a single fuel and ensuring that the specification of the fuel is standardized with its commercial equivalent in common use. The physical and chemical characteristics of the fuel are such that it can be introduced, stored, transported, and distributed by the fuel logistic systems.
Finding: JP8, diesel, and/or biodiesel are all potential fuels to be supplied to the battlefield, particularly for high power–use applications such as armored ground combat vehicles. The complexity impact of using multiple fuels on the logistics chain needs to be compared to the benefits discussed. (Tier 1, Lead)
Alternatively Sourced Liquid Hydrocarbon Fuels
The Army is also studying a number of alternative fuels derived from biomass feedstock and fossil energy (shale, coal, petcoke). This initiative
___________________
9 EIA, 2013, “Few Transportation Fuels Surpass the Energy Densities of Gasoline and Diesel,” https://www.eia.gov/todayinenergy/detail.php?id=9991.
10 H. Aydogan and E. Altinok, 2019, Effects of using JP8-diesel fuel mixtures in a pump injector engine on engine performance, Bilge International Journal of Science and Technology Research 3(0):106–111, https://doi.org/10.30516/bilgesci.652473.
is intended to provide further military operation flexibility through the ability to use multiple, reliable fuel sources. In all such cases, the fuel procured must (1) meet JP8 fuel performance specifications; (2) require no changes in the vehicle, equipment, or supply infrastructure; and (3) be capable of being mixed and/or blended with petroleum-derived fuel.11
Biomass-derived jet (biojet) fuel, also known as alcohol-to-jet (ATJ) fuel, is another example of an alternatively sourced hydrocarbon fuel. It has become a key element in the aviation industry’s strategy to reduce operating costs and environmental impacts. As expected, the focus here has been on its acceptability within gas turbine applications with less emphasis on its use in internal combustion engines. ATJ fuel is mixed 50/50 with JP8 to increase its aromatics content, which is essential to ensure that the seals with fuel systems swell to prevent leakage.
When used in internal combustion engines, the cetane ratings of ATJ-blended fuels can present some problems. As shown earlier, JP8 cetane ratings can be as low as 30 depending on the region from which it is obtained. The cetane number of ATJ is even lower, roughly at 18. As a result, the ATJ/JP8 mixtures can create internal combustion engines problems while being fully acceptable for aviation turbines.
One possible approach to address low cetane ratings if ATJ/JP8 blends are used in internal combustion engines would be utilization of cetane additives. To minimize the impact on soldier tasking, one solution would be to use inline fuel filters that meter the addition. If sized properly, these filters could be part of the scheduled maintenance, just as diesel-exhaust fluid containers are replaced on today’s automotive diesels during oil changes.
Conclusion: Alternative liquid hydrocarbon fuels are compositionally variable and may introduce new durability concerns and, in the case of ATJ fuels, may not provide the cetane ratings needed to run properly in internal combustion engines. Although alternative fuels may be suitable for use on an ad hoc basis during combat operations, their suitability as a more permanent staple of the fuel supply system will require a careful cost benefit analysis on a case-by-case basis over a variety of environmental conditions. (Tier 1, Follow)
GASEOUS ENERGY SOURCES
Compressed Propane
Compressed propane has roughly 73 percent of the energy content per unit volume of JP8 and roughly 14 percent better energy content per
___________________
11 Congressional Research Service, 2012, DOD Alternative Fuels: Policy, Initiatives and Legislative Activity, https://fas.org/sgp/crs/natsec/R42859.pdf.
unit weight. Much or all of this energy content per unit weight advantage is offset, however, by the heavier storage tank required versus a JP8 fuel tank.12 To put this in perspective, 250 gallons of compressed propane weigh roughly 1,050 pounds, whereas the tank required to contain it weighs roughly 480 pounds. Due to the volumetric energy density shortfall of compressed propane versus JP8, as well as safety concerns in its transportation, it is considered a less desirable fuel for the battlefield than JP8.
Natural Gas
Compressed (CNG) and liquefied natural gas (LNG) are produced from underground reserves or renewable biogas. The natural gas produced from renewable biogas, such as from landfills, is of a much lower quality with significantly more variability than that recovered from underground.
In the automotive and truck markets, usage of CNG, which consists mostly of methane, is growing because of environmental concerns. Because CNG burns more cleanly than either gasoline or diesel, it provides a significant advantage in greenhouse gas emissions versus both diesel and gasoline. CNG has only 26 percent of the energy content per unit volume of JP8, making it impractical as a fuel source for military combat vehicles, where space is greatly constrained to provide room for ammunition, propulsion, cooling systems, and operators.13 Like compressed propane, a much heavier storage tank would be required and safety concerns abound. Lastly, the number of supply trucks required to transport an equivalent amount of energy to the battlefield would have to grow, putting more lives at risk.
LNG is natural gas that has been cooled to a liquid state, at about −162°C (−260°F). The volume of natural gas in its liquid state is about 600 times smaller than its volume in its gaseous state at atmospheric pressure. It has roughly 63 percent of the energy content per unit volume of JP8. An insulated, cryogenic storage tank is required, with some degassing as it absorbs heat from the environment. Given its storage, transportation, and safety concerns, LNG is not considered a viable alternative to JP8 for military vehicles.14
The opportunity to create dual mode (diesel and gaseous fuel) power sources is mentioned in Chapter 7, “Forward Operating Base Power.”
___________________
12 EIA, 2013, “Few Transportation Fuels Surpass the Energy Densities of Gasoline and Diesel,” https://www.eia.gov/todayinenergy/detail.php?id=9991.
13 Ibid.
14 Ibid.
In select situations, this could enable using local CNG sources when available.
Hydrogen Transported to the Battlefield
Hydrogen is typically produced from natural gas, methanol, or electrolysis of water. It is widely used in manufacturing and chemical processing, including refining. It can be used as a fuel for a fuel cell, an internal combustion engine, or a gas turbine. The byproduct of hydrogen combustion is water (H2O), making this a very “clean” fuel. In particular, no carbon monoxide (CO), carbon dioxide (CO2), sulfur dioxide (SO2), hydrocarbon, or particulate matter are generated except as byproducts of any fuel contamination.
Hydrogen exhibits the highest gravimetric energy density (142 MJ/kg) of any commonly considered chemical fuel, an advantage offset in part by the heavy containers used to store it. On a volumetric basis, compressed and liquefied hydrogen have 14 and 27 percent of the volumetric energy content of JP8, respectively. Since supply trucks “cube out” before they “weigh out,” this results in four to seven times as many supply trucks to deliver an equivalent amount of energy to the battlefield.
Hydrogen is growing as a commercially available transportation fuel15 primarily for use in proton exchange membrane (PEM) fuel cells, with refueling stations planned across the United States and allied nations primarily for use in fuel cell–equipped vehicles.16 Hydrogen poses even greater transportation and storage challenges than natural gas, in that achieving practical handling densities requires that the gas be cooled (down to −253°C) or compressed (to 3,000 to 10,000 psi).17 These conditions translate to heavy containers, inefficiencies, and, ultimately, latent hazards. To the degree that commercial operations can be designed to mitigate these issues, tactical operations generally demand greater mobility while also imposing more severe and varied conditions. Thus, in addition to its inconsistency with the SFP, transporting hydrogen to the battlefield presents some logistics and handling challenges.
Hydrogen can also be used as a source of energy in internal combustion engines. With hydrogen, preignition (autoignition on cylinder head
___________________
15 While adoption of hydrogen as a fuel source has historically been slow, recent years have seen steady growth in supply and demand as a recent IEA report highlights: https://www.iea.org/reports/the-future-of-hydrogen.
16 I. Penn and C. Krauss, 2020, “California Is Trying to Jump-Start the Hydrogen Economy,” The New York Times, November 11, https://www.nytimes.com/2020/11/11/business/hydrogen-fuel-california.html.
17 EIA, 2013, “Few Transportation Fuels Surpass the Energy Densities of Gasoline and Diesel,” https://www.eia.gov/todayinenergy/detail.php?id=9991.
or piston hot spots before spark initiation of the combustion event) is a particular challenge, given hydrogen’s low ignition energy and wide flammability air/fuel ratios. Injection-system durability represents another challenge due to hydrogen’s low lubricity. Despite these challenges, there is renewed interest in hydrogen-powered internal combustion engines as a result of growing climate change concerns.18,19,20
A number of hydrogen storage initiatives under way seek to improve storage capacity and rate of release. Adsorption of hydrogen onto the surface of various metal powders has been investigated as a lower-pressure, room-temperature alternative for transportation use. Recent work with LaNi5 indicates the potential to store as much hydrogen at 30 psi as liquid hydrogen or compressed gas at 30,000 psi. Still, the overall density of adsorbent and hydrogen is too high for practical transportation targets (2 mass% hydrogen versus the Department of Energy [DOE] target of 6.5 mass%). Carbon nanotubes also show promise as a hydrogen sorbent, but significant work remains to relate nanomaterial characteristics to storage performance.21 Similarly, another nanomaterial category known as metal–organic frameworks (MOFs) has been investigated for hydrogen storage (and a range of other adsorption applications). MOFs comprise a metal ion or cluster of metal ions and an organic molecule acting as a linking element, allowing design flexibility to provide adsorption sites with a particular affinity for certain fluid molecules. Some laboratory results (e.g., MOF-65022) indicate storage capacities above DOE transportation targets, but these studies involve milligram quantities because MOF materials are very expensive, can suffer stability issues, exhibit lower capacity in the presence of water vapor, and production capacity is quite limited.
The most active consideration of hydrogen for tactical use involves usage with fuel cells. These energy-conversion devices will be discussed in further depth later in this chapter.
___________________
18 Florida Solar Energy Center, “Hydrogen Basics—Internal Combustion Engine,” http://www.fsec.ucf.edu/en/consumer/hydrogen/basics/utilization-ice.htm, accessed January 2021.
19 FEV Group, 2020, “FEV Is Driving Forward Hydrogen Internal Combustion Engine Development,” October 8, https://www.fev.com/en/coming-up/press/pressreleases/news-article/article/fev-is-driving-forward-hydrogen-internal-combustion-enginedevelopment.html.
20 M. Brezonick, 2021, “Westport, Scania Cooperate on Hydrogen Engine Research,” Diesel Progress, https://www.dieselprogress.com/news/Westport-Scania-cooperate-onhydrogen-engine-research/8009850.article.
21 L. Schlapbach and A. Zuttel, 2001, Hydrogen-storage materials for mobile applications, Nature 414:353–358.
22 S. Yu, G. Jing, S. Li, Z. Li, and X. Ju, 2020, Tuning the hydrogen storage properties of MOF-650: A combined DFT and GCMC simulations study, International Journal of Hydrogen Energy 45(11):6757-6764, https://doi.org/10.1016/j.ijhydene.2019.12.114.
Conclusion: A logistics distribution network for propane, natural gas, or hydrogen is unlikely to effectively replace hydrocarbon fuels on the battlefield because of their lower volumetric energy density (requiring more fuel transport trucks or convoys) and increased storage complexity versus JP8.
Hydrogen Produced Near the Point of Use
In the event that hydrogen-powered technologies develop with significant military operational benefits, it may be more practical to produce hydrogen near the point of use instead of developing an entire new wholesale field-distribution network. Two approaches are discussed below, both of which require water as the hydrogen carrier (i.e., source), either obtained locally or by transporting it to the site.
The first is the possible use of commercial electrolyzers that produce hydrogen from water, breaking it down into its elemental components. Their commercial use is growing rapidly because they provide a means to address one of the largest dilemmas in the renewable energy industry, which is how to store the energy when it is not in demand. Electrolyzers are available in a variety of sizes, up to and including the system shown in Figure 3.4, which can produce 3,000 tons of hydrogen annually using clean hydropower.
Because electrical energy is required for electrolysis, using JP8 to power an internal combustion engine to power a generator to power an electrolyzer to generate hydrogen to power a fuel cell has some inherent inefficiencies. In addition, as discussed above, using renewable energy sources (solar, wind, hydro, waste) will likely have a limited role in generating energy on the battlefield. It is certainly more efficient to power a ground combat vehicle or unmanned aerial vehicle (UAV) directly with JP8. Nevertheless, in situations where silent operation over an extended range is desired, electrolyzers may provide an acceptable path to hydrogen production.
As an alternative to electrolyzers, powdered aluminum alloys containing gallium have been known for decades to spontaneously generate hydrogen when in contact with water.23 This process can produce high pressures, which can significantly reduce the energy required to compress hydrogen for storage. Theoretically, the aluminum powder and reactant water represent a lower effective energy density than logistic petroleum fuel.
___________________
23 J.M. Woodall, J.T. Ziebarth, C.R. Allen, J. Jeon, G. Choi, and R. Kramer, 2008, “Generating Hydrogen On Demand by Splitting Water with Al Rich Alloys,” pp. 313–315 in Clean Technology 2008: Bio Energy, Renewables, Green Building, Smart Grid, Storage, and Water (M. Laudon, B. Romanowicz, and D.L. Laird, eds.), https://phys.org/news/2007-05-hydrogenaluminum-alloy-fuel-cells.html.

However, if water (potable or nonpotable) is locally available, then solid aluminum could afford a logistic and handling advantage.
The Massachusetts Institute of Technology (MIT) Lincoln Laboratory has developed a method to produce activated aluminum beads that react in a similar manner, producing aluminum hydroxide, hydrogen, steam, and residual contaminants.24
As shown in Figure 3.5, a preliminary prototype design by the MIT Lincoln Laboratory has demonstrated the ability to generate 10 kW on an automotive application using a reaction chamber, conditioning system, and PEM fuel cell. However, some key questions remain to be answered. These include how much aluminum and water would be required to achieve a reasonable vehicle range. How would the aluminum, water,
___________________
24 E. Limpaecher, Massachusetts Institute of Technology Lincoln Laboratory, 2020, “Activated Aluminum for Operational Energy,” presentation to the committee on September 10.
and waste aluminum hydroxide be handled? Could the overall size and weight of the system be competitive with other alternative power and energy systems?
More directly related to a military application, the Army awarded General Atomics, Inc., a 2-year contract in November 2019 to design, fabricate, and test a prototype mobile platform for on-demand generation of high-pressure hydrogen suitable for refueling PEM fuel cell–equipped vehicles in the field. This technology is claimed to use the company’s proprietary aluminum alloy hydrogen-producing technology.25 The committee did not have access to a progress report from General Atomics at the time this report was written.
Despite the technology immaturity issues listed above, enough potential benefits remain to justify further investigation of this opportunity. Among the possible benefits, the hydrogen generated might enable some additional fuel-cell use with its low acoustic signature. A complete
___________________
25 General Atomics, 2019, “General Atomics Awarded Army Contract for Hydrogen Generation System Prototype,” https://www.ga.com/general-atomics-awarded-army-contractfor-hydrogen-generation-system-prototype.
description of these opportunities—at the dismounted soldier and forward operating base level—is contained in Appendix G, “Aluminum Fuel.”
Conclusion: Generating hydrogen from water using aluminum near the point of use offers potential advantages vis-à-vis transporting hydrogen in a supply convoy. However, a number of critical questions remain, including definition of the complete process to be used for each application.
Recommendation: The Army should continue to explore the potential use of aluminum for onsite generation of hydrogen for use in proton exchange membrane fuel cells, not only for use in vehicles, but also for potential use in dismounted and base-camp applications. The latter may leverage ongoing Navy efforts. (Tier 2, Watch [U.S. Marine Corps and Office of Naval Research-led effort])
Nuclear Energy Sources
Nuclear energy comprises the most energy-dense medium currently available for useful application. Various nuclear reactions provide the opportunity to extract more energy from a given form factor compared to the common technologies of thermal, electrochemical, kinetic, or even chemical energy storage. When nuclear fission was developed as an energy source in the past century, it offered a logical progression from petrochemical fuels, leveraging 5 orders of magnitude increase in energy density, abundant supply, and zero environmental emissions.
To put this in perspective, reactor-grade enriched uranium has an energy density of 3,456,000 MJ/kg, which is partially offset by the fact that current fission reactors tap only 5 percent of the latent specific energy in the fuel rods. Despite the inefficiencies, its value (172,800 MJ/kg) dwarfs the 44 MJ/kg of JP8. For all practical purposes, the energy density of the fissile fuel can be considered unlimited, with the challenge being constraints on the size and weight of the equipment required to provide the needed power for specific applications and the life-cycle costs of handling and disposing of highly radioactive spent fuel rods.26
Nuclear energy includes a family of processes, some of which potentially could be useful for current, and especially future, military operations. Miniature long-lived power sources could address challenges to power large numbers of persistent sensors. Larger portable devices could integrate electrochemical storage with radioactive sources to extend device life for dismounted operations. Today, the Army is reconsidering
___________________
26 A. Greig, 2020, “Fundamentals of Nuclear-Powered Engines,” p. 29 in Nuclear Engine Air Power, https://airpower.airforce.gov.au/sites/default/files/2021-03/BPAF02_NuclearEngine-Air-Power.pdf.
nuclear reactors as an alternative to the fueled generators that power large forward bases. If the Army further pursues any such alternatives, implementation may imply a number of related development needs related to such aspects as utilization, transportation, safety, and security.
Additional detail about the various forms of nuclear energy is contained in Appendix M. Miniature and portable devices employing radioisotope decay will be discussed in further depth in Chapter 5, “Dismounted Soldier Power and Light UAVs/UGVs.” Nuclear reactor studies were advocated by the 2016 Defense Science Board report Task Force on Energy Systems for Forward/Remote Operating Bases and will be discussed further in Chapter 7, “Forward Operating Base Power.”27 Approaches to address safety and regulatory concerns are covered in Appendix M. Box 3.1 describes the challenges and opportunities of nuclear isomer energy storage.
Fuel Cells
Fuel cells electrochemically convert the chemical energy of a fuel into electrical power without any combustion. The exhaust from fuel cells is totally carbon-free if hydrogen is used as the fuel. However, if a hydrocarbon fuel is used, the exhaust contains CO2 in direct proportion to the
___________________
27 M. Anastasio, P. Kern, F. Bowman, J. Edmunds, G. Galloway, W. Madia, and W. Schneider, 2016, “Task Force on Energy Systems for Forward/Remote Operating Bases,” Defense Science Board, Under Secretary of Defense for Acquisition, Technology, and Logistics (USD(AT&L)), https://dsb.cto.mil/reports/2010s/Energy_Systems_for_Forward_Remote_Operating_Bases.pdf.
amount of fuel consumed, but there are no NOx or particulate emissions. The two most common types of fuel cells today for power generation are PEM fuel cells and solid oxide fuel cells (SOFCs). Alkaline-exchange membrane (AEM) fuel cells are also undergoing a research renaissance with the recent development of more stable hydroxide-ion conductive polymers, but these cells are not at the same level of commercial adoption.
PEM fuel cells are the predominant technology for the hydrogen-powered passenger cars and trucks being tested today on the road. They operate at 60°C to 120°C and require pure hydrogen as the fuel. If the use of a hydrocarbon fuel is desired, it will first have to be reformed to produce pure hydrogen containing no CO or sulfur, because each easily poisons the platinum-based catalysts that reduce molecular oxygen and oxidize the fuel in PEM fuel cells.28 However, reformation of hydrocarbon fuels such as JP8 or diesel to produce hydrogen with no CO and sulfur is extremely complex and should be further investigated regarding its applicability for onsite or on vehicle reformation. For military use, PEM fuel cells (PEMFCs) may be worth considering if either (1) the studies mentioned earlier of onsite hydrogen production from activated aluminum prove to be attractive or (2) the Army considers silent operation, low thermal infrared signature, or long-endurance UAVs/unmanned ground vehicles (UGVs) (>25 hours) to be so important that convoy transportation of hydrogen to the field is warranted.
An SOFC produces electricity by electrochemically oxidizing a fuel at efficiencies up to about 60 percent; actual efficiency depends on the fuel used and the operating conditions. It consists of a dense oxide electrolyte sandwiched between two electrodes—the anode and the cathode. In an SOFC power system, SOFC cell stacks are combined with the balance of the plant (BOP) consisting of fuel cleanup equipment (mainly for desulfurization) and fuel reformer (if any), blowers/compressors (for fuel/air delivery), heat exchangers/recuperators/combustors (for thermal management), power electronics (for power conditioning), and controllers (for system control). At present, the most common materials for SOFCs are yttria-stabilized zirconia (YSZ) for the electrolyte, nickel-YSZ for the anode, lanthanum strontium cobalt ferrite (LSCF) for the cathode, and stainless steel or a conducting ceramic for the cell interconnects.
SOFCs are fuel flexible. Suitable fuels for SOFCs include hydrogen, natural gas, biogas, alcohols, propane, and other low-sulfur hydrocarbons. SOFCs can either operate directly on natural gas (internal reformation) or on its reformates (predominantly a mixture of CO and H2) from external steam reformation.
___________________
28 R.F. Service, 2010, The case of the poisoned fuel cell, Science, July 16, https://www.sciencemag.org/news/2010/07/case-poisoned-fuel-cell.
Use of heavy hydrocarbon fuels is possible but requires reformation to break down the fuel into CO and H2. Reformation can be accomplished using steam reforming (SR), autothermal reforming (ATR), dry reforming (DR), catalytic partial oxidation (CPOX), or a combination of these processes.29 Each of these processes has certain advantages and disadvantages. Steam reformation is the most efficient reforming process; in addition, the water and heat required for the reformation can be supplied by recirculation of the hot SOFC exhaust gas. ATR is less efficient than steam reformation, but the system is lighter and more compact. CPOX uses ubiquitous air as the oxidant; however, the syngas concentration is low due to the dilution by nitrogen from the air.
Sulfur compounds poison SOFC anode materials, and all fuels need to be desulfurized to about 1 ppm sulfur for use with SOFCs.30 To put this in perspective, JP8 and ultra-low sulfur diesel fuels are allowed to contain as much as 3,000 and 15 ppm sulfur, respectively. Liquid and gas-phase adsorptive desulfurization of JP8 can reduce sulfur to a level that would be acceptable for SOFC operation. However, it may require desulfurization both upstream and downstream of the reformer. In large applications, such as at operating bases, the sulfur-adsorbing beds could be thermally regenerated if needed.
The Army’s Ground Vehicle Systems Center (GVSC) will be integrating a 10 kW JP8-based SOFC power system using a monolith reformer into a Multi-Utility Tactical Transport (MUTT) vehicle in 2021, thereby demonstrating the capability of full-time silent power generation. By designing this as a hybrid, the SOFC only needs to meet the average power demand while the batteries can assist in meeting the peak power demand. In fiscal year 2023 as part of the Next Generation of Combat Vehicle family work, GVSC also is planning to demonstrate a 10 kW JP8 power system on a light robotic combat vehicle (RCV-L). GVSC is also working on a heavier modified RCV platform using General Motors’ (GM’s) commercial hydrogen PEM fuel cell technology. The GM effort using hydrogen will use at least 80 kW fuel cell stacks.31
A major disadvantage of SOFCs is their operation at 700°C to 1,000°C, which mandates either a lengthy start-up time (currently ranging from 30 minutes to a few hours) or ongoing continuous operation. This time lag will need to be factored into any decision to deploy a SOFC application in the field. Alternatively, use of an onboard diesel-fueled SOFC as a
___________________
29 S. Sengodan, R. Lan, J. Humphreys, D. Du, W. Xu, H. Wang, and S. Tao, 2018, Advances in reforming and partial oxidation of hydrocarbons for hydrogen production and fuel cell applications, Renewable and Sustainable Energy Reviews 82, Part 1:761–780.
30 P. Boldrin, E. Ruiz-Trejo, J. Mermelstein, J.M. Menéndez, T. Reina, and N. Brandon, 2016, Strategies for carbon and sulfur tolerant solid oxide fuel cell materials, incorporating lessons from heterogeneous catalysis, Chemical Reviews 116:13633–13684, https://pubs.acs.org/doi/pdf/10.1021/acs.chemrev.6b00284.
31 K. Centeck, U.S. Army CCDC Ground Vehicle Systems Center, 2020, email communications with committee member.
charger to battery propulsion power for ground vehicles might be a good option to extend the range of battery-powered vehicles and overcome the lengthy start-up issue of SOFCs.
Use of small power SOFCs for remote sensors and dismounted soldiers will be discussed in greater detail in Chapter 5, “Dismounted Soldier and Small UAVs/UGVs.” Use of SOFCs for ground combat vehicles will be discussed in greater detail in Chapter 6, “Vehicle Power and Large Weapon Systems.” Use of SOFCs for forward operating bases will be covered in in Chapter 7, “Forward Operating Base Power.”
Conclusion: Given that fuel-cell technology may serve as a key enabling technology for near-silent operation, low thermal signature, and long-endurance UAVs/UGVs, combined with the prevalence of JP8 on the battlefield through 2035, the committee supports continued investment by the U.S. Army to fund the technology and economic analysis of the reformation process with diesel and JP8 fuels for use in SOFC power systems. (Tier 2, Lead)
Other Power and Energy Sources (Solar, Wind, Hydro, Geothermal)
A number of alternative energy sources are in growing use around the world today, most of which are intermittent and diffuse. The committee did not focus on these because of its focus on an Armored Brigade Combat Team use case. Wind and sunlight obviously depend on location, weather, time of day, and other factors beyond the control of users.
Nevertheless, the committee recognizes the importance of these alternatives as contributors to fuel-supply logistics and encourages the Army to continue exploring their use for its domestic and permanent overseas facilities. In addition, the committee recognizes that small, flexible roll-up solar panels and small solar trailers, which are now commercially available, can provide expeditionary personnel with a fallback battery charger or power source for laptop computers and radios.
As part of a 2016 report on energy systems for forward and remote operating bases, the Defense Science Board examined the availability, technical maturity, and operational considerations of alternative energy sources, including solar, wind, hydrokinetic, geothermal, and ocean thermal power (see Table 3.2). The study found that these alternative, renewable “energy sources are advantageous only in a limited set of cases” and noted that this has been the conclusion of several other studies conducted during the previous decade.32
___________________
32 M. Anastasio, P. Kern, F. Bowman, J. Edmunds, G. Galloway, W. Madia, and W. Schneider, 2016, “Task Force on Energy Systems for Forward/Remote Operating Bases,” Defense Science Board, Under Secretary of Defense for Acquisition, Technology, and Logistics (USD(AT&L)), https://dsb.cto.mil/reports/2010s/Energy_Systems_for_Forward_Remote_Operating_Bases.pdf, pp. 26-28.
TABLE 3.2 Alternative Energy Sources and Technologies
| Energy Source | Availability | Technical Maturity | Operational Considerations |
|---|---|---|---|
| Solar Power | Available globally; varies with location, season weather, time | Widely deployed on the civil grid and military installations; limited deploy of tactical units | Small rugged panels can be beneficial; possible visible target; glint/glare concerns; require cleaning |
| Wind Power | Available globally; varies with location, season, weather, time | Widely deployed on the civil grid and military installations; small units exist, but are typically not attractive for military use | While potentially beneficial, concerns with small wind turbines include reliability, visbility, and interference with communications |
| Hydrokinetic Power | Common but not everywhere; varies with location, season, weather, time | Utility-scale hydroelectric dams are mature and common; small portable tidal, wave, and micro-hydro power systems are under development | Requires sophisticated technologies and potentially a large material footprint; variable but more predictable than wind and solar |
| Geothermal Power | Exists in limited locations worldwide; where present, heat output is often steady | Very mature for civil applications | Requires considerable time and initial capital cost for construction; likely attractive for some enduring locations |
| Ocean Thermal Power | Exists in the deep sea and near specific islands | Under civil sector development and under evaluation for use on U.S. Kwajalein Army Base | Requires significant initial capital cost and large structures; may be attractive for some enduring locations |
SOURCE: M. Anastasio, P. Kern, F. Bowman, J. Edmunds, G. Galloway, W. Madia, and W. Schneider, 2016, “Task Force on Energy Systems for Forward/Remote Operating Bases,” Defense Science Board, Under Secretary of Defense for Acquisition, Technology, and Logistics (USD(AT&L)), https://dsb.cto.mil/reports/2010s/Energy_Systems_for_Forward_Remote_Operating_Bases.pdf.
Conclusion: Similar to the 2016 Defense Science Board report,33 the committee concludes that solar, wind, and geothermal power sources present significant environmental benefits and are worthy of consideration for domestic and permanent overseas facilities. However, current and near-future iterations provide far less utility for mobile forces in multi-domain operations (MDO) and are unlikely to meet the power needs of a brigade combat team. As demonstrated in recent operations in Southwest Asia and elsewhere, such technologies can help reduce logistical requirements, especially in remote and dismounted operations. (Tier 1, Follow)
Electrochemical Batteries and Capacitors
Batteries are ubiquitous, unseen, and unappreciated—until the device they power stops running. That loss of function leads to ever-increasing requirements for more energy (stored) and more power (delivered on demand) in a lighter, less voluminous package. Because a lack of power can compromise mission accomplishment, the Department of Defense (DoD) continually seeks battery improvements to power a broad spectrum of military-specific platforms and missions. Batteries are a go-to choice for power because they cover an energy spectrum of microwatt-hours (microsensor power) to beyond megawatt-hours (microgrid power) as demarcated by their packaged weight (Watt hours [Wh] per kilogram), volume (Wh per liter), or footprint (Wh per cm2 of cross-sectional area).
As a sealed delivery vehicle of mission-required electrons, the simplicity of the packaged battery—an anode physically isolated from the cathode by a separator—masks the functional physicochemical complexity within. The boundary conditions of the two-terminal energy-storage device (Figure 3.6) are constrained by the thermodynamics of the chemistry within (which dictate cell voltage) and the kinetics at which electrons are released from or returned to the active materials in the two electrodes (walking the line between controlled delivery of electrons versus a bomb).
The classic Ragone plot that maps increasing power on the y-axis and increasing energy on the x-axis (Figure 3.7) captures the frustration of the user: instant gratification (the demand for electrons now [i.e., power] versus waiting for an anticipated reward [the ability to tap electrons over extended time—i.e., energy]). The C rate34 also captures that dichotomy—batteries designed to deliver all stored energy at a 4C rate would drain that capacity in 15 min when application flexibility and endurance may require 15 h (a rate of C/15). What does the
___________________
33 Ibid.
34 A C rate is a measure of the time it takes to charge or discharge the nominal total capacity of a battery; for example, full charge to the rated capacity in 2 h equates to a C rate of 0.5C or C/2.
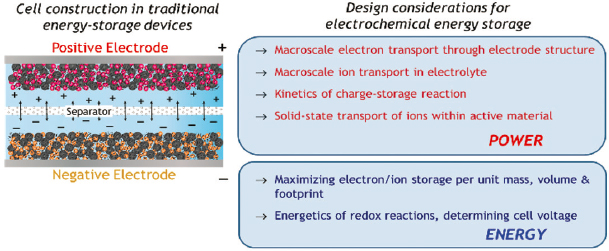
user actually want from an energy-storage device? Both functions, as needed. That demand places the performance metrics of an electrical energy-storage device in unoccupied territory—up and to the right on the power versus energy Ragone plot—where neither present-day electrochemical capacitors (ECs) provide sufficient energy nor batteries provide sufficient power. Note that “sufficient” is in the eye of the
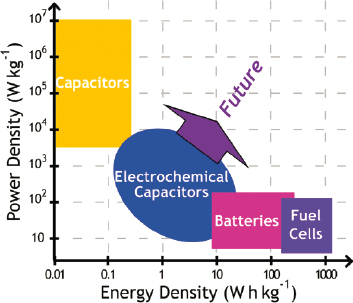
beholding user. ECs using high surface–area carbon-composite electrode structures in which charge is stored at the electrified interface are a mature technology and commercially available.
More than one U.S. program manager supporting battery research has noted they found little had changed when returning to battery science and technology (S&T) reviews after stepping away for 15 years.35 In keeping with that observation, note the first recommendation in the 2004 National Academies report Meeting the Energy Needs of Future Warriors,36 which assessed power and energy needs for the Army using the land warrior as its focal point:
Recommendation 1: The Army should focus on batteries with a specific energy of 300 Wh/kg and higher for insertion into future versions of the Land Warrior (LW) ensemble. It should continue to promote and support innovative approaches to disposable and rechargeable batteries that can be adapted for military use. To select the best candidates for a given application, the Army should explore the trade-off space that exists between lifetime (measured in terms of charge-discharge cycles), specific power, specific energy, safety, and cost (p. 4).
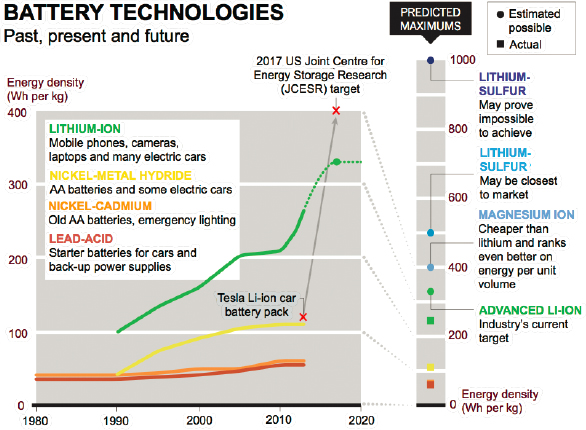
The consumer expects a lithium-ion (Li-ion) battery under the hood of an automobile or laptop or smartphone (Figure 3.8). Older consumers are still grateful for the lightened laptop load from 30 years ago when the energy was stored in nickel-cadmium or nickel-metal hydride batteries. The military requires batteries indifferent to thermal, mechanical, and propulsive forces. Safety issues persist with Li-ion batteries in a battlefield environment. Although containment measurements for a rifle shot have been identified (Figure 3.9), protection against larger projectiles remains a concern. Propagation of thermal runaway in a damaged Li-ion cell risks conflagration of a Li-ion battery pack and requires mitigation that adds weight and volume, which means multiple Li-ion cells become a system.
The available energy stored in Li-based batteries at the system level is greatly reduced by the weight and volume of added safety measures such that the impressive per-cell energy density plummets. With some measure of propagation resistance to minimize runaway thermal events, commercially available rechargeable Li-ion batteries provide 150 Wh/kg. When rechargeability is mission-warranted, this specific energy makes them Tier 1 candidates—if the risk of damage to the soldier or the platform is deemed acceptable. If not, further mitigation measures will lower the system energy even further. Concerns with large banks of Li-ion batteries
___________________
35 Personal communications to committee member (late 1980s and early 2000s).
36 National Research Council, 2004, Meeting the Energy Needs of Future Warriors, Washington, DC: The National Academies Press, https://www.nap.edu/catalog/11065.

already have designers of consumer grid-storage systems reconsidering heavy, low-energy-density lead-acid batteries as the alternative.37
Efforts worldwide, including the substantial investment by DOE in the battery hub known as JCESR (Joint Center for Energy Storage Research), are working to develop new cathode materials, higher capacity Li-based anodes, and new electrolytes to create rechargeable batteries that achieve >300 Wh/kg (system).38 An even more ambitious consortium out of DOE, known as Battery500, focuses on Li-metal batteries. Stretch goals include a threefold increase in specific energy to 500 Wh/kg at the cell level. A potential concern lies in manufacturing Li-metal cells at a large scale, which will require ultrapure Li metal and glovebox handling.
The extra energy packed per kg into these advanced, Tier 3 Li-based batteries is accompanied by both higher cell voltage (>3.5 V) and higher capacity, but such gains are also accompanied by increased safety issues. As potential late-Tier 2 candidates, the safety issues may override the desirable energy density, particularly for platforms requiring multicell assemblies. The recent increase in dangerous-goods regulation for international air transport of present-day, lower energy density Li-based batteries, which cannot be shipped fully charged, will present additional onerous logistics issues should the proposed >350 Wh/kg Li-based batteries become commercialized and acquired for MDO usage.
Even when spent, Li-based batteries continue to pose a hazard. In 2017,39 a rail car containing Li batteries for recycling caught fire and exploded outside of Houston, Texas, fortunately with no reported injuries. The recycling industry for Li batteries is still nascent and more concerned with the high-value metals in the cathode (Co, Ni, Mn) than the modest amount of Li present in the anode. In 2019, DOE initiated a three-phased Lithium-Ion Battery Recycling Prize worth $5.5 million. Fifteen Phase 1 winners were each awarded $67,000, with their efforts representing five areas—collection; separation and storage; safe storage and transport; reverse logistics; and other innovative ideas. The worldwide effort to diminish or eliminate cobalt in the cathodes of Li-based batteries will make the economic argument less compelling for recycling Li batteries,
___________________
37 P.P. Lopes and V.R. Stamenkovic, 2020, Past, present, and future of lead–acid batteries, Science 369(6506):923–924, doi: 10.1126/science.abd3352.
38 Battery500 goals include development of next-generation Li-metal anode cells delivering a threefold increase in specific energy to 500 Wh/kg. See Department of Energy, 2020, “Battery500: Progress Update,” Office of Energy Efficiency and Renewable Energy, May 19, https://www.energy.gov/eere/articles/battery500-progress-update.
39 M. Dempsey, 2017, “Train Explosion Leads to Chemical Release in Downtown Houston,” Houston Chronicle, April 24, https://www.chron.com/news/houston-texas/houston/article/Train-explosion-leads-to-chemical-release-in-11095738.php.
although the safety and environmental arguments remain—as will the logistics concerns of the U.S. military.
A key safety issue in Li-based batteries is formation of metal dendrites at the anode, especially under forcing conditions such as charging the battery when cold40 or demanding power beyond rated specifications. This concern is amplified when using Li metal anodes, such as proposed in DOE’s Battery 500 initiative. Using solid ceramic electrolytes to minimize growth of Li metal dendrites from extensive charge–discharge cycling is achieved currently by operating at loads of approximately 1 mA cm−2. This limit is likely to be overridden in the field to obtain necessary pulse power, one of the key conditions that favors the growth of dendrites, which then launches the accompanying safety concerns inherent to Li chemistry. These operational conditions are those that degrade the ability to tap the rated energy density.
Efforts to create safer Li batteries by using a water-in-salt electrolyte, which is nonflammable, could marry energy density to higher operational safety, but scalability remains an issue that is not yet solved for this electrolyte, and the dendrite concerns remain. A recent evaluation of aqueous Li-ion batteries using super-concentrated (water-in-salt) electrolyte finds that the growth of the passivating solid electrolyte interphase (SEI) at the anode does not protect against degradation of the electrolyte during cycling or on storage. Of greater concern for military applicability of such batteries is the conclusion that these aqueous water-in-salt Li-ion batteries cannot compete with commercial lead-acid, nickel-cadmium, or nickel-metal-hydride aqueous batteries in terms of price, operating temperature range, lifetime, or their capacity to fade upon storage.41
To reach the Army-desired energy density of 300, 400, or even 500 Wh/kg (system) for post-2025, one returns (back) to primary batteries. Meeting the Energy Needs of Future Warriors noted that primary batteries “now provide the main energy source, but the acquisition, storage, distribution, and disposal of over a hundred different battery types poses an enormous logistical challenge on the battlefield.”42
Primary batteries are energy rich because during discharge an electron-rich metal corrodes. Intensive efforts to tap even a fraction of the theoretical energy density of Li-air batteries (40,104,000 J/kg or 11,140 Wh/kg) have proven elusive and are offset by the safety
___________________
40 C.T. Love, O. Baturina, K.E. Swider-Lyons, 2015, Observation of lithium dendrites at ambient temperature and below, ECS Electrochemistry Letters 4: A24–A27.
41 L. Droguet, A. Grimaud, O. Fontaine, and J.-M. Tarascon, 2020, Water-in-salt electrolyte (WiSE) for aqueous batteries: A long way to practicality, Advanced Energy Materials 10(43):2002440, https://onlinelibrary.wiley.com/doi/10.1002/aenm.202002440.
42 National Research Council, 2004, Meeting the Energy Needs of Future Warriors, Washington, DC: The National Academies Press, https://www.nap.edu/catalog/11065.
downsides from using Li metal and flammable solvents. A workhorse primary battery in the military relies on the Li-SO2 chemistry that stores the energy used by BA-5590 batteries to power radios. But on mission where the battery is reconfigured by the warfighter to operate under high loads, requiring pulse power, more like an electrochemical capacitor, the power draw degrades the ability to tap the rated energy density. Multiple batteries would need to be packed on the mission to meet the required energy, which is then further multiplied by the customary three times redundancy factor.
A collision thus occurs between current manufacturing practice to produce the positive electrode (cathode on discharge) in the primary battery and how the warfighter uses the battery. The cathode structure is typically formulated as a powder composite through an inexpensive process that physically mixes carbon powder as an ad hoc electron wire in the structure with the active material that takes up electrons on discharge (or uses those electrons to catalyze reduction of O2, SO2, or SOCl), plus a polymer binder to hold the mixture together. Under low-to-moderate loads (i.e., over hours or days), the capacity of the active materials and thus the battery can be drained to manufacturer-rated levels of energy density.
Power performance out of the same electrode structure cannot be ensured because electron flow from the current collector to the distributed active material relies on surface contact of the active material with carbon agglomerates. Powder composites establish a junctioned pathway—from carbon particulate to carbon particulate—rather than a direct electron-wired path of the poor-to-moderate electron conductive active material through the volume of the electrode structure. Power demand forces the electron-transfer reactions at the active material to predominate at a high rate at surface, instead of the bulk of capacity, which can, with repetitive pulses, lead to mechanical or chemical changes in the active material that compromise the bulk of capacity in the active material upon returning to low-to-moderate loads.
Standard battery electrode structures are not designed to interchangeably provide high power demand and high energy density. In operation on the battlefield, traditional batteries are forced to perform both functions, and when forced, fail at delivering the rated stored energy. But research over the past 20 years holds out hope for next-generation batteries that provide hybrid function within one device, namely sustaining pulse-power demands while retaining accessibility to the inherent charge-storing capacity of the active materials. A key innovation arose by re-thinking battery construction as integrated in three dimensions rather than built up as layers (Figure 3.10).43
___________________
43 J.W. Long, B. Dunn, D.R. Rolison, and H.S. White, 2004, Three-dimensional battery architectures, Chemical Reviews 104(10):4463–4492, https://pubmed.ncbi.nlm.nih.gov/15669159/.
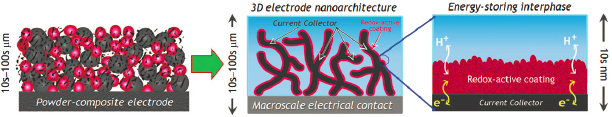
Subsequent elaborations and spin-offs on reconfiguring battery function in three dimensions have demonstrated the way forward. The first is a redesign of electrode structures as architectures in which the paths for electrical charge (electrons and ions) and molecular transport are directly wired within the volume of the electrode. Electrodes in next-generation hybrid battery-capacitors can be designed by modifying the surfaces of a three dimensional (3D), porous current collector (e.g., a carbon nanofoam) with the active material obviating the need to add conductive carbon or by creating monolithic, high conductivity foams such as a carbon nanofoam for Li-SO2 cells or a zinc sponge for Ni-Zn, Ag-Zn, MnO2-Zn alkaline cells. The second is demonstrating that well-wired, nanoscale-textured active materials increase surface-to-volume ratio to innately allow surface-based, capacitive charge/discharge at high load without decrementing the total charge stored or released at low-to-moderate loads. Combining the two redesigns affords power performance commensurate with an electrochemical capacitor while retaining the energy density designed into the battery.
Finding: Battery technology will be a part of Army operations for the foreseeable future. However, traditional Li-ion batteries present certain limitations that will not meet all of the Army’s emerging needs. However, redesigning electrode structures as 3D architectures may permit greater performance with retention of battery-effective energy density and can improve the performance of both primary and rechargeable batteries.
Battery Research and Development Opportunities Now Under Way
Research investments under way on several fronts offer promise for development in time to be deployed as a Tier 2 technology. The issues with respect to even the available generation of Li-based batteries, including the logistics complications surrounding ensuring that transport of Li-batteries occurs at less than full state-of-charge, emphasizes the practicality of optimizing aqueous-based or all solid-state–based energy storage for the Army. The following energy-storage systems offer other means to deliver both power and energy using safer chemistries and advanced electrode designs.
- Increasing the technology readiness level (TRL) of asymmetric electrochemical capacitors in which nanometric coatings of battery materials deposited on 3D porous electrodes provide pulse power in aqueous electrolytes that are more energy dense than electrolytic double-layer capacitors (EDLCs).44,45
- Developing Zn-ion batteries with neutral to mildly acidic aqueous electrolytes;46 when the positive electrode is designed as a 3D architecture and Na ions are added to the Zn2+-based electrolyte, capacitive power can be obtained while traditional powder-composite structures formulated with the same manganese oxide active material cannot deliver pulse power.47
- Scaling 3D, tricontinuous, all solid–state batteries, including those manufactured using 3D printing,48 which already show promise for microbatteries and on-chip power, to sizes relevant for wearables using sponge form factors.
- Developing rechargeable alkaline batteries using dendrite-suppressing Zn sponge and related 3D anodes.49
___________________
44 P. Galek, A. Mackowiak, P. Bujewska, and K. Fic, 2020, Three-dimensional architectures in electrochemical capacitor applications—Insights, opinions, and perspectives, Frontiers in Energy Research 8:139, https://www.frontiersin.org/articles/10.3389/fenrg.2020.00139/full.
45 Y. Shao, M.F. El-Kady, J. Sun, Y. Li, Q.H. Zhang, M.F. Zhu, H.Z. Wang, B. Dunn, and R.B. Kaner, 2018, Design and mechanisms of asymmetric supercapacitors, Chemical Reviews 118(18):9233–9280, https://doi.org/10.1021/acs.chemrev.8b00252.
46 J. Shin, J. Lee, Y. Park, and J.W. Choi, 2020, Aqueous zinc ion batteries: Focus on zinc metal anodes, Chemical Science 11:2028–2044.
47 J.S. Ko, M.B. Sassin, D.R. Rolison, and J.W. Long, 2018, Combining battery-like and pseudocapacitive charge storage in 3D MnOx@carbon electrode architectures for zinc-ion cells, Sustainable Energy and Fuels 2: 626–636, https://doi.org/10.1039/C7SE00540G.
48 M. Cheng, R. Deivanayagam, and R. Shahbazian-Yassar, 2020, 3D printing of electrochemical energy storage devices: A review of printing techniques and electrode/electrolyte architectures, Batteries and Supercaps 3(2):130–146, https://doi.org/10.1002/batt.201900130.
49 J.F. Parker, C.N. Chervin, E.S. Nelson, D.R. Rolison, and J.W. Long, 2014, Wiring zinc in three dimensions re-writes battery performance—Dendrite-free cycling, Energy and Environmental Science 7:1117–1124, https://doi.org/10.1039/C3EE43754J.
TABLE 3.3 New Capabilities for Next-Generation Zinc-Based Batteries in Order of Development Time Line
| Battery type | Function improvement with 3D redesign |
|---|---|
| Primary Zn-air | 30-40% greater run time than conventional Zn-air |
| Air-rechargeable pulse power | |
| Size scalability from microbatteries to large stacks | |
| Retain high specific energy even with challenging duty cycles | |
| Rechargeable Ag-Zn, Ni-Zn, MnO2-Zn | Extended cycle life (relative to conventional Zn batteries) |
| High power (>400 W kg−1) | |
| Balance of plant (less swaddling for safety) | |
| Rechargeable Zn-air | High specific energy (2-3× Li-ion) |
| Air-chargeable pulse power | |
| Balance of plant (less swaddling for safety) | |
| 3D all solid-state Ag-Zn, Ni-Zn, MnO2-Zn | Extended temperature range (cold and hot) |
| High power | |
| Recharge faster than Li-ion | |
| No orientation effects with respect to gravitational field |
SOURCE: D.R Rolison and J.W. Long, 2013, unpublished white paper, U.S. Naval Research Laboratory.
By reformulating Zn into a sponge form-factor, the Naval Research Laboratory (NRL) has pushed Zn utilization to >90 percent in primary Zn-air cells (versus the approximately 50–60 percent customarily obtained) and innately suppressed dendrite formation, even under demanding charge–discharge conditions—all while using aqueous electrolytes rather than the flammable nonaqueous electrolytes used in Li-ion batteries. This 21st-century design breakthrough using 19th-century battery chemistry provides DoD with a transformative opportunity to take the military-validated, aqueous-based Ag-Zn, Ni-Zn, MnO2-Zn, and Zn-air primary batteries and transform them into rechargeable batteries that are safe, cost-effective, domestically sourced, and meet or exceed the performance of Li-ion batteries on the system level (Table 3.3). Near-term payoffs arise by swapping out powder-composite Zn anodes for the Zn sponge and using it in existing Zn battery configurations. To match the innate capabilities of architected Zn electrodes—a two-electron anode versus the one-electron Li-based anode—further research and development will be required to optimize complementary positive electrode compositions and structures, including identifying multi-electron active materials (e.g., Ag/AgxO, potentially MnO2 and NiOOH, and trifunctional air-breathing
cathodes that reduce O2 on discharge, evolve O2 on charge, and provide pulse power).
Conclusion: Zn-based batteries, once moved to a new performance curve, may bypass the safety issues associated with Li-ion and the low-energy limitations of lead-acid while providing the following critical functions: (1) extended mission life for a given battery weight or volume; (2) platform simplification, because less balance-of-plant is required for safe, aqueous-based cell chemistry; and (3) simultaneous energy and power delivery from a single device. (Tier 2, Lead)
As discussed above, many excellent initiatives are already under way in the area of battery research and should continue to be pursued. Although commercial industry developments are encouraging, the Army and other branches of the military have some unique considerations. First and foremost is the need for soldier safety, which includes consideration of attack by high-powered projectiles. Since loss of life is at stake in many situations, cost considerations are less important than in the commercial market.
Recommendation: Since the Army and Navy have many of the same battery safety concerns, close cooperation between the two services is encouraged. For the Army, fast rechargeability is an important objective that enables expeditious tapping into the vast supply of electricity available from generators and microgrids, as well as unmanned and manned combat vehicles. (Tier 1, 2, Lead)
































