Summary1
Perfluoroalkyl and polyfluoroalkyl substances (PFAS) are a class of chemicals that includes more than 12,0002 different compounds with various chemical properties. PFAS are commonly used in thousands of products, from nonstick cookware to firefighting foams and protective gear, because they have desirable chemical properties that impart oil and water repellency, friction reduction, and temperature resistance. PFAS as a class have a wide variety of distinct chemical properties and toxicities; for example, some PFAS can bioaccumulate and persist in the human body and the environment, while others transform relatively quickly. The PFAS that do transform, however, will become one or more other PFAS because the carbon–fluorine bond they contain does not break naturally. It is for this reason that PFAS are termed “forever chemicals.”
STUDY CONTEXT
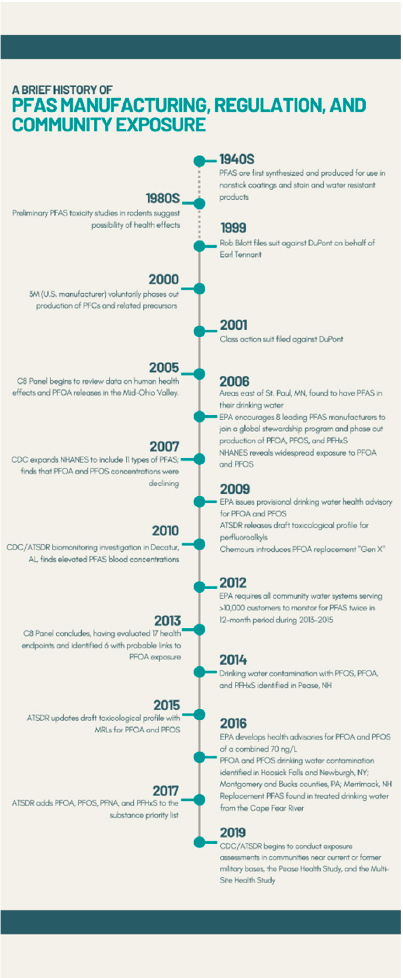
Public concern about the impact of PFAS contamination on human health and the environment began in the late 1990s when perfluorooctanoic acid (PFOA) water contamination was identified in Parkersburg, West Virginia. As a result, 3M, a primary PFAS manufacturer initiated a voluntary phaseout of some PFAS (PFOA, perfluorooctanesulfonic acid [PFOS], and perfluorohexanesulfonic acid [PFHxS]). The contamination in Parkersburg also led to a class action lawsuit that identified several health effects related to PFAS exposure and led to the establishment of a medical monitoring program in 2013. Shortly thereafter, the U.S. Environmental Protection Agency (EPA) began requiring all community water systems serving more than 10,000 people to test for certain PFAS, which led to more communities learning that their water was contaminated. In 2016, researchers found that the drinking water supply for Wilmington, North Carolina, was contaminated with a chemical called “GenX,” a PFOA replacement. This finding led to public concern about the potential health effects of replacement PFAS (see Figure S-1).
Organizations such as the International Agency for Research on Cancer (IARC), the Agency for Toxic Substances and Disease Registry (ATSDR), and the EPA have linked exposure to PFAS (particularly PFOA and PFOS) to multiple cancers, thyroid dysfunction, small changes in birthweight, and high cholesterol. Gaining a complete picture of the threat can be difficult, however, because of the chemical and toxicological differences among individual PFAS and uncertainty about the exposure level at which their adverse effects may occur. In addition, many of the chronic diseases associated with PFAS exposure have myriad causes.
An estimated 2,854 U.S. locations (in all 50 states and two territories) have some level of PFAS contamination (see Figure S-2). Although not all of the contamination represents exceedances of health advisories, the pervasiveness of the contamination is alarming. Furthermore, almost 100 percent of the U.S. population is exposed to at least one PFAS. Although exposures to the phased-out PFAS have been decreasing (see Figure S-3), people are still exposed to those PFAS from site contamination, occupational uses of stored products, and breakdown of PFAS polymer products that are found in homes. Carpeting, for example, is often treated with fluorotelomer-based polymers that can biodegrade to form phased-out PFAS, such as PFOA. Exposures also occur to the PFAS chemicals used to replace those that have been phased out. Although the harms of the replacement PFAS are less well understood, they may have
___________________
1 No references are included in this Summary. References to the content herein are provided in the respective chapters of the main text.
2 EPA Comptox Dashboard (https://comptox.epa.gov/dashboard/chemical-lists/pfasmaster [accessed May 25, 2022]).
comparable or more serious toxicity than the PFAS they have replaced. The state of New Jersey, for example, recently set a groundwater standard for a replacement PFAS that is an order of magnitude lower than drinking water standards for other PFAS. The New Jersey maximum contaminant levels of perfluorononanoic acid (PFNA), PFOA, and PFOS in drinking water are 13, 14, and 13 nanograms per liter (ng/L), respectively,3 while the groundwater standard for the replacement PFAS, chloroperfluoropolyether carboxylates, is 2 ng/L.4
NOTE: ATSDR = Agency for Toxic Substances and Disease Registry; CDC = Centers for Disease Control and Prevention; EPA = U.S. Environmental Protection Agency; MRL = minimal risk limit; ng/L = nanograms per liter; NHANES = National Health and Nutrition Examination Survey; PFAS = per- and polyfluoroalkyl substances; PFC = perfluorochemicals; PFHxS = perfluorohexane sulfonic acid; PFNA = perfluorononanoic acid; PFOA = perfluorooctanoic acid; PFOS = perfluorooctane sulfonic acid.
SOURCE: Committee generated based on slides included by Patrick N. Breysse in a presentation to the committee on February 4, 2021.
___________________
3 See https://www.nj.gov/health/ceohs/documents/pfas_drinking%20water.pdf (accessed June 8, 2022).
4 See https://www.nj.gov/dep/standards/ClPFPECA_Standard.pdf (accessed June 8, 2022).
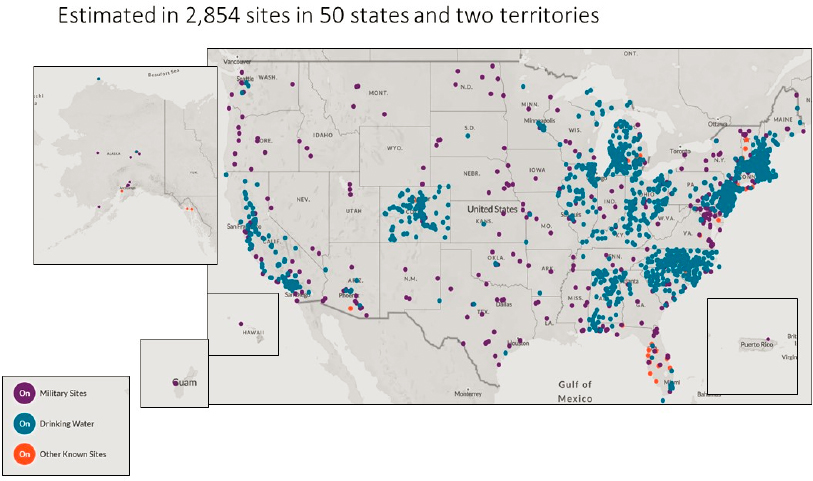
NOTES: According to the Environmental Working Group (EWG), “locations represented on the map are approximate and intended to portray the general area of a contamination site or a community water system. Locations were mapped using the best data available from official records, including data provided by tests of public drinking water systems, the Safe Drinking Water Information System and the Department of Defense report Addressing Perfluorooctane Sulfonate (PFOS) and Perfluorooctanoic Acid (PFOA), and Department of Defense public records, among others. Data on contaminated industrial and military sites was current as of October 2021.” Furthermore, “EWG has worked to ensure the accuracy of the information provided in this map. The map is dynamic. This contaminant site, results, suspected sources and other information in the database may change based on evolving science, new information or other factors. Please be advised that this information frequently relies on data obtained from many sources, and accordingly, EWG cannot guarantee the accuracy of the information provided or any analysis based thereon.”
SOURCE: See https://www.ewg.org/interactive-maps/pfas_contamination/map (accessed May 11, 2022). Copyright © Environmental Working Group, www.ewg.org. Reproduced with permission.
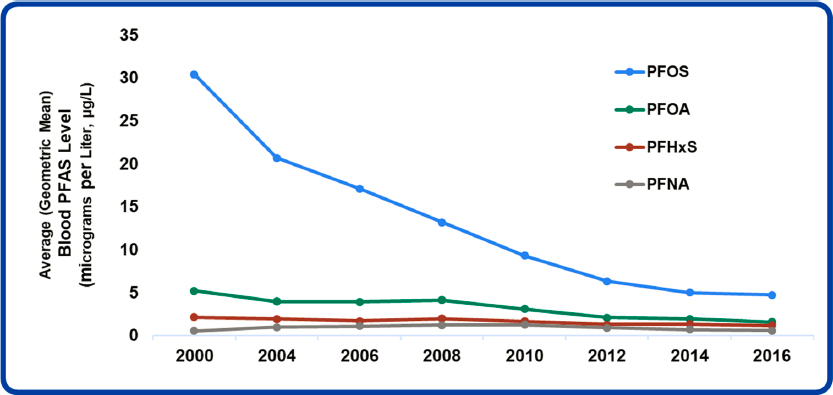
NOTE: Average = geometric mean.
SOURCE: Patrick N. Breysse’s presentation to the committee on February 4, 2021. DATA SOURCE: Centers for Disease Control and Prevention (2019, January). Fourth Report on Human Exposure to Environmental Chemicals, updated tables. Atlanta, GA: U.S. Department of Health and Human Services.
The people who live, work, and play in environments where PFAS contamination exceeds standards do not know how to protect themselves from the health risks of exposure. Many have been exposed to PFAS for decades and may continue to be exposed in their homes or workplaces. Some members of communities that have discovered that their exposures exceed health advisory levels are calling for a medical program to prevent, lead to early detection of, or treat diseases related to the health risks they may face. Developing such a program is challenging, however, because of the uncertainties about the health effects of PFAS and potential harms from additional medical treatments; moreover, many of these diseases are not preventable or even treatable, and many clinicians lack information about what they can and should do for these patients.
To help clinicians5 respond to patient concerns about PFAS exposure, ATSDR published PFAS: An Overview of the Science and Guidance for Clinicians on Per- and Polyfluoroalkyl Substances.6 This guidance summarizes general information about PFAS and PFAS health studies and suggests answers to example patient questions. However, the ATSDR guidance does not provide specific recommendations on when to test for PFAS, how to order the tests, how to interpret the results, or what clinical follow-up based on PFAS exposure might look like. Interpretation of PFAS blood or urine results is challenging because the specific level of exposure at which harms may occur is unknown, and the science on the potential health effects of exposure to PFAS is advancing quickly, making it difficult to provide advice to clinicians about what follow-up tests might be helpful.
STUDY CHARGE AND APPROACH
ATSDR and the National Institute of Environmental Health Sciences (NIEHS) asked the National Academies of Sciences, Engineering, and Medicine to form an ad hoc committee to advise on PFAS testing and clinical care for patients exposed to PFAS. The Statement of Task asked the committee to
- develop principles for biological testing and clinical evaluation, given substantial scientific uncertainty about the health effects or the value of such measures in informing care,
- review the human health literature for the health effects of PFAS, and
- characterize human exposure pathways and develop principles for exposure reduction.
The Statement of Task asked the committee to recommend
- options and considerations to guide decision making for PFAS testing in a patient’s blood or urine,
- PFAS concentrations that could inform clinical care of exposed patients, and
- appropriate patient follow-up and care specific to PFAS-associated health endpoints for those patients known or suspected to be exposed to PFAS.
The committee also was asked to provide advice on changes to ATSDR’s clinical guidance. The committee was not asked for community prevention guidance or advice on policies that would reduce exposure to PFAS.
Figure S-4 summarizes the committee’s approach to the Statement of Task. A critical component of the approach was community engagement because people who live with potentially harmful exposures have knowledge from experiential learning that precedes scientific findings. The study’s community engagement consisted of a panel of community liaisons, three town halls (summarized in Appendix B),
___________________
5 The committee uses “clinician” throughout this report to refer to “a healthcare professional qualified in the clinical practice of medicine. Clinicians may be physicians, nurses, pharmacists, or other allied health professionals” as defined by the Centers for Medicare & Medicaid Services (https://www.cms.gov/Medicare/Quality-InitiativesPatient-Assessment-Instruments/MMS/QMY-Clinicians [accessed June 14, 2022]).
6 See https://www.atsdr.cdc.gov/pfas/docs/clinical-guidance-12-20-2019.pdf (accessed June 14, 2022).
community speakers at every public committee meeting, open sign-up for public testimony at every public meeting, and encouragement for written testimony throughout the data collection phase of the study. The following sections provide descriptions of the remaining components of the study approach and the committee’s associated recommendations.
COMMITTEE’S PRINCIPLES FOR DECISION MAKING UNDER UNCERTAINTY
Providing clear advice to clinicians on clinical follow-up for PFAS-exposed patients is challenging. The questions in the Statement of Task all have some degree of uncertainty. First, clinicians need to know what the health consequences of PFAS exposure are. If everyone is exposed, what exposure level warrants follow-up? What actions can be taken to reduce exposure or prevent disease? What are the harms of those actions? Answering all of these questions requires making ethical judgments. Building on the work of other experts and evidence-to-decision frameworks, the committee developed five principles to guide decision making under uncertainty for use throughout this report and by ATSDR when updating the guidance for clinicians: proportionality, justice, autonomy, feasibility, and adaptability (see Box S-1). In the clinical setting, these principles converge under the principle of autonomy; thus, shared, informed decision making between clinician and patient is the practical way to incorporate the principles into a clinical encounter.
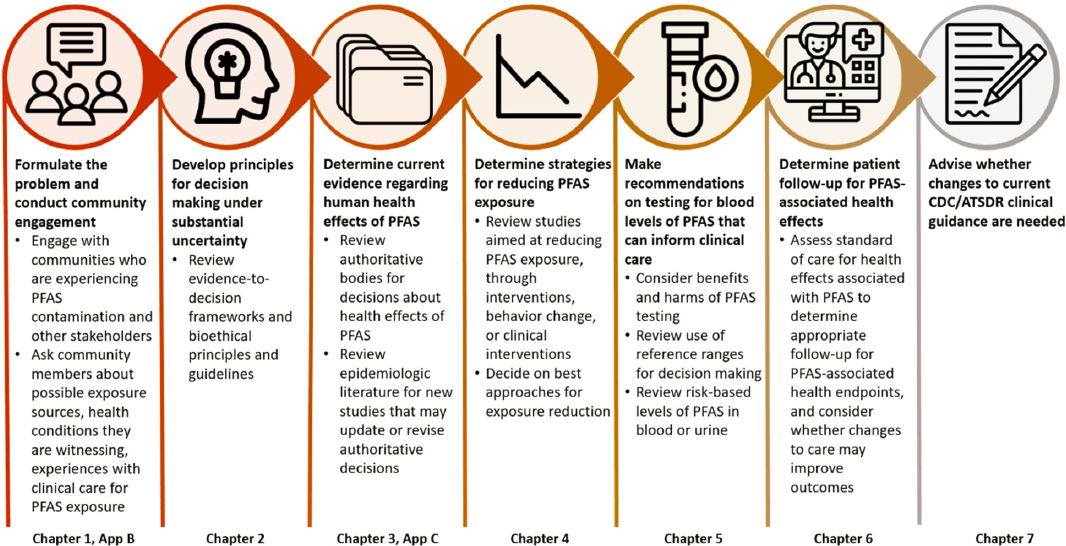
NOTE: ATSDR = Agency of Toxic Substances and Disease Registry; CDC = Centers for Disease Control and Prevention; PFAS = per- and polyfluoroalkyl substances.
POTENTIAL HEALTH EFFECTS OF PFAS
To recommend PFAS levels that should inform clinical care, the committee conducted a literature review to determine health effects or conditions associated with PFAS. The Statement of Task limited this review to human studies of those PFAS included in the Centers for Disease Control and Prevention’s (CDC’s) National Report on Human Exposure to Environmental Chemicals (see Table S-1). Other PFAS may also cause harm, though they do not all have the same biological persistence and toxicity. Most people are exposed to mixtures of PFAS such that specific effects are difficult to disentangle. Considering these issues, and recognizing that some PFAS are infrequently measured, the committee provided one strength-of-evidence determination for all PFAS for each health effect, recognizing that providing one conclusion across PFAS may not account for the distinct physical, chemical, and toxicological properties of each type of PFAS.
| Abbreviated Name | Full Name | CAS Registry No. |
|---|---|---|
| MeFOSAA | Methylperfluorooctane sulfonamidoacetic acid | 2355-31-9 |
| PFHxS | Perfluorohexanesulfonic acid perfluorohexane sulfonic acid | 355-46-4 |
| n-PFOA (linear isomer), Sb-PFOA (branched isomers) | Perfluorooctanoic acid | 335-67-1* |
| PFDA | Perfluorodecanoic acid | 335-76-2 |
| PFUnDA | Perfluoroundecanoic acid | 2058-94-8 |
| n-PFOS (linear isomer), Sm-PFOS (branched isomers) | Perfluorooctanesulfonic acid | 1763-23-1* |
| PFNA | Perfluorononanoic acid | 375-95-1 |
NOTES: CAS = Chemical Abstracts Service. * = CAS number refers to linear isomer only. Previous survey years have also included perfluorobutane sulfonic acid (PFBS), perfluoropentanoic acid (PFpA), perfluorododecanoic acid (PFDoDA), perfluorooctane sulfonamide (FOSA), and 2-(N-ethyl-perfluorooctane sulfonamido)acetate (EtFOSAA), according to Patrick N. Breysse’s presentation to the committee on February 4, 2021.
The committee began its review by identifying decisions of other authoritative bodies regarding PFAS exposure and any human health effects. The committee then considered more recent human studies, both systematic reviews and published epidemiologic research articles, that could inform updates to those decisions. This approach improved efficiency while minimizing the risk of excluding scientific findings that could inform the committee’s conclusions.
The committee considered animal studies discussed in ATSDR’s Toxicological Profile for Perfluoroalkyls and in systematic reviews to aid in its interpretation of the human studies. The committee synthesized the available evidence into four categories of association used by other National Academies committees: sufficient evidence of an association, limited or suggestive evidence of an association, inadequate or insufficient evidence of an association, and limited or suggestive evidence of no association (see Figure S-5). All associations between PFAS and a health outcome were considered inadequate or insufficient by default unless available evidence from all aspects of the committee’s review warranted placing the evidence in another category of association.

NOTES: The categories of association only describe how strong the evidence is between PFAS and the health outcome. The risk of developing an outcome from exposure to PFAS for things in the same category can vastly differ and are dependent on whether an individual has other risk factors for developing the outcome.
The committee found sufficient evidence of an association for the following diseases and health outcomes:
- decreased antibody response (in adults and children),
- dyslipidemia (in adults and children),
- decreased infant and fetal growth, and
- increased risk of kidney cancer (in adults).
The committee found limited or suggestive evidence of an association for the following diseases and health outcomes:
- increased risk of breast cancer (in adults),
- liver enzyme alterations (in adults and children),
- increased risk of pregnancy-induced hypertension (gestational hypertension and preeclampsia),
- increased risk of testicular cancer (in adults),
- thyroid disease and dysfunction (in adults), and
- increased risk of ulcerative colitis (in adults).
The committee observed gaps in the evidence, rendering the evidence inadequate or insufficient, for many health effects including the following:
- immune effects other than reduced antibody response, and ulcerative colitis;
- cardiovascular outcomes other than dyslipidemia;
- developmental outcomes other than small reductions in birthweight;
- cancers other than kidney, breast, and testicular;
- reproductive effects other than hypertensive disorders of pregnancy;
- hepatic effects other than liver enzyme levels;
- endocrine disorders other than those involving thyroid hormone levels;
- respiratory effects;
- hematological effects;
- musculoskeletal effects, such as effects on bone mineral density;
- renal effects, such as renal disease; and
- neurological effects.
For some outcome categories, the research spanned many different tests or measures of effect, all of which assessed slightly different health outcomes, making the evidence difficult to synthesize and use to draw strong conclusions. The committee found this issue most apparent when reviewing the literature on neurodevelopmental effects, such as effects on learning and behavior. Another research issue is that most studies reviewed by the committee were not conducted among people known to have high exposures to PFAS, indicating a gap in understanding the effects of PFAS among those highly exposed.
PFAS EXPOSURE REDUCTION
Some people may be interested in reducing their exposure to PFAS. The primary route to PFAS in nonoccupational settings is likely ingestion, which may include drinking contaminated water; eating seafood from contaminated water; or consuming other contaminated foods, such as vegetables, game, or dairy products. PFAS are often used in materials that come in contact with food, such as microwave popcorn bags or packaging used for fast foods or processed foods. Exposure may also occur when dust containing PFAS is ingested. Inhalation is the most common route in occupational settings, and is a route of exposure for people living near fluorochemical plants or incinerators. PFAS transfer to the fetus during pregnancy and in early life during lactation. Dermal exposure is understudied.
If PFAS are in drinking water, switching to consumption of water lower in PFAS will reduce exposure. In general, however, it is difficult to reduce exposure to PFAS through personal behavior modifications.
For clinicians, based on its review of the evidence on PFAS exposure reduction, the committee makes the following recommendations:
Recommendation 4-17: Clinicians advising patients on PFAS exposure reduction should begin with a conversation aimed at first determining how they might be exposed to PFAS
___________________
7 The committee’s recommendations are numbered according to the chapter of the main text in which they appear.
(sometimes called an environmental exposure assessment) and what exposures they are interested in reducing. This exposure assessment should include questions about current occupational exposures to PFAS (such as work with fluorochemicals or firefighting) and exposures to PFAS through the environment. Known environmental exposures to PFAS include living in a community with PFAS-contaminated drinking water, living near industries that use fluorochemicals, serving in the military, and consuming fish and game from areas with known or potential contamination.
Recommendation 4-2: If patients may be exposed occupationally, such as by working with fluorochemicals or as a firefighter, clinicians should consult with occupational health and safety professionals knowledgeable about the workplace practices to determine the most feasible ways to reduce that exposure.
Recommendation 4-3: Clinicians should advise patients with elevated PFAS in their drinking water that they can filter their water to reduce their exposure. Drinking water filters are rated by NSF International, an independent organization that develops public health standards for products. The NSF database can be searched online for PFOA to find filters that reduce the PFAS in drinking water included in the committee’s charge. Individuals who cannot filter their water can use another source of water for drinking.
Recommendation 4-4: In areas with known PFAS contamination, clinicians should advise patients that PFAS can be present in fish, wildlife, meat, and dairy products and direct them to any local consumption advisories.
There are fewer evidence-based, exposure-reduction recommendations for patients without known sources of exposure:
Recommendation 4-5: Clinicians should direct patients interested in learning more about PFAS to authoritative sources of information on how PFAS exposure occurs and what mitigating actions they can take. Authoritative sources include the Pediatric Environmental Health Specialty Units (PEHSUs), the Agency for Toxic Substances and Disease Registry (ATSDR), and the U.S. Environmental Protection Agency (EPA).
Recommendation 4-6: When clinicians are counseling parents of infants on PFAS exposure, they should discuss infant feeding and steps that can be taken to lower sources of PFAS exposure. The benefits of breastfeeding are well known; the American Academy of Pediatrics, the American Academy of Family Physicians, and the American College of Obstetricians and Gynecologists support and recommend breastfeeding for infants, with rare exceptions. Clinicians should explain that PFAS can pass through breast milk from a mother to her baby. PFAS may also be present in other foods, such as the water used to reconstitute formula and infant food, and potentially in packaged formula and baby food. It is not yet clear what types and levels of exposure to PFAS are of concern for child health and development.
Additionally, there is a critical need for more data to understand PFAS exposure among breastfed infants:
Recommendation 4-7: Federal environmental health agencies should conduct research to evaluate PFAS transfer to and concentrations in breast milk and formula to generate data that can help parents and clinicians make shared, informed decisions about breastfeeding.
PFAS TESTING AND CONCENTRATIONS TO INFORM CLINICAL CARE OF EXPOSED PATIENTS
Decisions about PFAS testing require shared informed decision making between patient and clinician. Clinicians should explain that exposure biomonitoring may provide important information about an individual’s exposure levels that might guide clinical follow-up. At the same time, this information cannot indicate or predict the likelihood that an individual will end up with a particular condition. Allowing people the opportunity to determine whether they will undergo PFAS testing shows respect for patient values. Discussions about PFAS testing should always include information about how PFAS exposure occurs, potential health effects of PFAS, limitations of PFAS testing, and the benefits and harms of the testing (see Box S-2).
The German Human Biomonitoring Commission has risk-based guidance levels for two PFAS chemicals—PFOS and PFOA. The European Food Safety Authority has one guidance level for the sum of PFOA, PFOS, PFHxS, and PFNA. No individual values are available for PFHxS and PFNA, and the committee could find no values for methylperfluorooctane sulfonamidoacetic acid (MeFOSAA), PFDA, and perfluoroundecanoic acid (PFUnDA). Based on its review of the evidence for PFAS testing, the committee recommends:
Recommendation 5-1: As communities with PFAS exposure are identified, government entities (e.g., Centers for Disease Control and Prevention [CDC]/Agency for Toxic Substances and Disease Registry [ATSDR], public health departments) should support clinicians with educational materials about PFAS testing so that they can discuss testing with their patients. These educational materials should include the following information:
- How people can be exposed to PFAS: Exposure routes include occupational exposures and work with fluorochemicals or as a firefighter; consumption of contaminated drinking water in communities that obtain their water from sources near commercial airports, military bases, fluorochemical manufacturing plants, wastewater treatment plants, landfills, or incinerators where PFAS-containing waste may have been disposed of or farms where sewage sludge may have been used; and consumption of contaminated fish or game if fishing or hunting occurs in contaminated areas. Individuals living near fluorochemical plants may also be exposed via inhalation of air emissions.
- Potential health effects of PFAS exposure and strategies for reducing exposure.
- Limitations of PFAS blood testing: PFAS blood testing does not identify the sources of exposure or predict future health outcomes; it only assesses body burden at the time of sample collection. For example, a person with low blood levels today may have had higher levels in the past.
- The benefits and harms of PFAS testing.
Recommendation 5-2: Clinicians should offer PFAS testing to patients likely to have a history of elevated exposure. In all discussions of PFAS testing, clinicians should describe the potential benefits and harms of the testing and the potential clinical consequences (such as additional follow-up), related social implications, and limitations of the testing so that patient and clinician can make a shared, informed decision. Patients who are likely to have a history of elevated exposure to PFAS include those who have
- had occupational exposure to PFAS (such as those who have worked with fluorochemicals or served as a firefighter);
- lived in communities where environmental and public health authorities (Centers for Disease Control and Prevention [CDC], Agency for Toxic Substances and Disease Registry [ATSDR], U.S. Environmental Protection Agency [EPA], state and local environmental or health authorities), or academic researchers have documented PFAS contamination; or
- lived in areas where PFAS contamination may have occurred, such as near facilities that use or have used fluorochemicals, commercial airports, military bases, wastewater treatment plants, farms where sewage sludge may have been used, or landfills or incinerators that have received PFAS-containing waste.
Recommendation 5-3: Clinicians should use serum or plasma concentrations of the sum of PFAS* to inform clinical care of exposed patients, using the following guidelines for interpretation:
- Adverse health effects related to PFAS exposure are not expected at less than 2 nanograms per milliliter (ng/mL).
- There is a potential for adverse effects, especially in sensitive populations, between 2 and 20 ng/mL.
- There is an increased risk of adverse effects above 20 ng/mL.
* Simple additive sum of MeFOSAA, PFHxS, PFOA (linear and branched isomers), PFDA, PFUnDA, PFOS (linear and branched isomers), and PFNA in serum or plasma. Caution is warranted when using capillary blood measurements as levels may differ from serum or plasma levels.
The cutoff levels should be updated as additional information becomes available. The committee also noted that children younger than 12 years are not routinely included in the National Report on Human Exposure to Environmental Chemicals, nor are pregnant people included in large numbers. More reference- and risk-based values are needed, for other PFAS and other biological matrices, but given the expansiveness of the class, this gap can best be addressed with relative potency factor approaches rather than the development of risk-based levels for each PFAS.
Recommendation 5-4: The National Health and Nutrition Examination Survey should begin collecting and sharing more data on children younger than 12 years of age and pregnant people to generate reference populations for those groups.
Testing for PFAS, although expensive, offers an opportunity to identify people who may need to reduce their PFAS exposure and are at increased risk of certain health outcomes. It is important to recognize, however, that race, age, and other social and demographic characteristics already have disadvantaged many patients with respect to accessing clinical preventive services, such as PFAS testing. The disadvantage would be compounded as PFAS testing services should be linked to counseling on steps to mitigate exposure and its impacts. Therefore, encouraging testing primarily among people with relatively stable access to care could have the unintended effect of aggravating disparities in exposure to PFAS absent a funded, national PFAS testing program with a counseling component.
APPLICATION OF THE COMMITTEE’S PRINCIPLES TO PATIENT FOLLOW-UP FOR PFAS-ASSOCIATED HEALTH EFFECTS
Many health outcomes or conditions that the committee found to be associated with PFAS exposure are common in the general population. All have multiple known risk factors. The committee categorized the strength of the evidence for an association between PFAS and various health outcomes, and concluded that all conditions with an adequately supported association should be considered for patient follow-up. The committee then used its established cutoff levels to determine appropriate followup based on PFAS exposure level. Risks from PFAS likely increase with exposure, and PFAS levels of 3 ng/mL and 19 ng/mL do not represent the same risk even though they are listed in the same category. Clinical providers should use judgment and shared, informed decision making in making follow-up decisions based on PFAS exposure and other risk factors. Figure S-6 suggests that clinicians engage in shared decision making with their patients regarding follow-up care for PFAS-associated health endpoints.
Recommendation 6-1: Clinicians should treat patients with serum PFAS concentration below 2 nanograms per milliliter (ng/mL) with the usual standard of care.
Recommendation 6-2: For patients with serum PFAS concentration of 2 nanograms per milliliter (2 ng/mL) or higher and less than 20 ng/mL, clinicians should encourage PFAS exposure reduction if a source of exposure is identified, especially for pregnant persons. Within the usual standard of care clinicians should:
- Prioritize screening for dyslipidemia with a lipid panel (once between 9 and 11 years of age, and once every 4 to 6 years over age 20) as recommended by the American Academy of Pediatrics (AAP) and the American Heart Association (AHA).
- Screen for hypertensive disorders of pregnancy at all prenatal visits per the American College of Obstetricians and Gynecologists (ACOG).
- Screen for breast cancer based on clinical practice guidelines based on age and other risk factors such as those recommended by the U.S. Preventive Services Task Force (USPSTF).

NOTE: MeFOSAA = methylperfluorooctane sulfonamidoacetic acid; PFDA = perfluorodecanoic acid; PFHxS = perfluorohexane sulfonic acid, PFNA = perfluorononanoic acid; PFOA = perfluorooctanoic acid; PFOS = perfluorooctanesulfonic acid; PFUnDA = perfluoroundecanoic acid.
Recommendation 6-3: For patients with serum PFAS concentration of 20 nanograms per milliliter (ng/mL) or higher, clinicians should encourage PFAS exposure reduction if a source of exposure is identified, especially for pregnant persons. In addition to the usual standard of care, clinicians should:
- Prioritize screening for dyslipidemia with a lipid panel (for patients over age 2) following American Academy of Pediatrics (AAP) guidelines for high-risk children and American Heart Association (AHA) guidance for high-risk adults.
-
At all well visits:
- conduct thyroid function testing (for patients over age 18) with serum thyroid stimulating hormone (TSH),
- assess for signs and symptoms of kidney cancer (for patients over 45), including with urinalysis, and
- for patients over 15, assess for signs and symptoms of testicular cancer and ulcerative colitis.
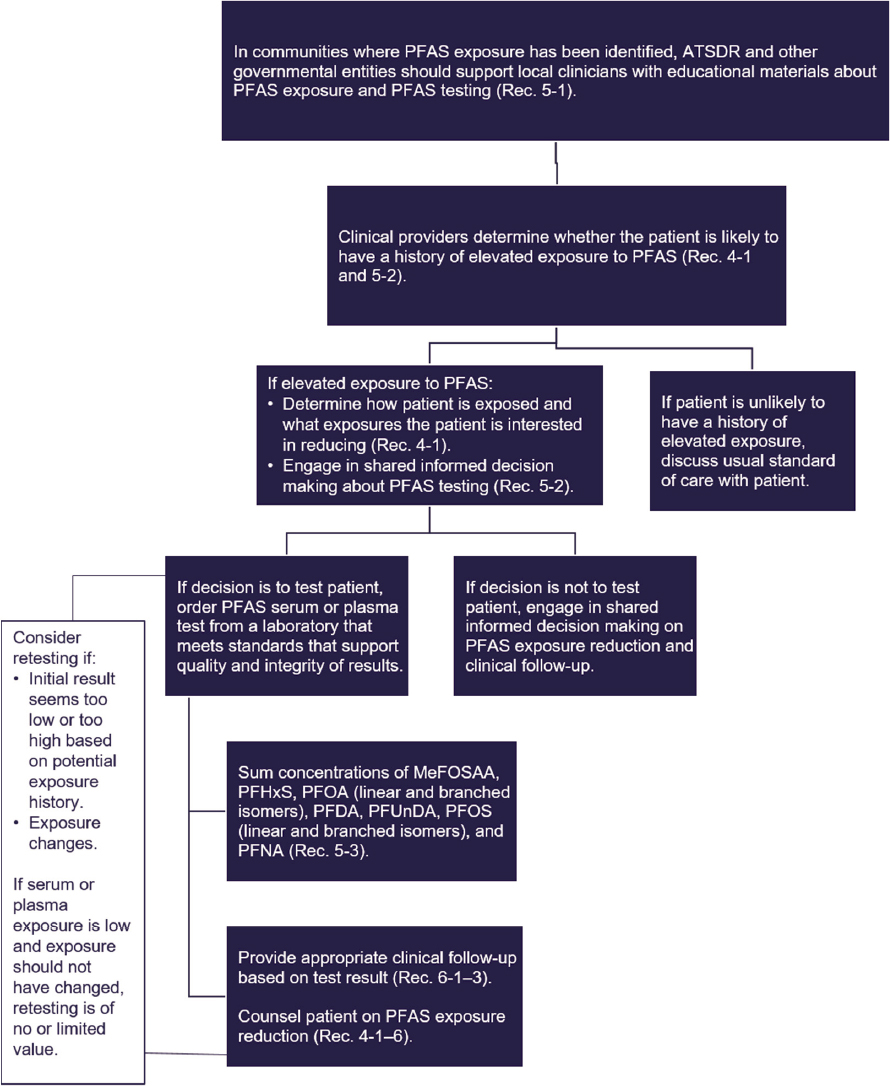
APPLYING THE COMMITTEE’S EXPOSURE, TESTING, AND CLINICAL FOLLOW-UP RECOMMENDATIONS
The committee created a flow chart summarizing PFAS education, exposure assessment, and clinical follow-up (see Figure S-7). In communities where PFAS exposure has been identified, ATSDR and other government entities should support local clinicians with educational materials about PFAS exposure and testing. Clinicians should then determine whether a particular patient is likely to have a history of elevated exposure to PFAS. If so, the clinician should offer PFAS testing and make a shared, informed decision on that testing. If testing is chosen, the labs should be ordered (Test Code 39307 Current Procedural Terminology [CPT] code 82542). Test results should be interpreted by summing the concentrations of MeFOSAA, PFHxS, PFOA (linear and branched isomers), PFDA, PFUnDA, PFOS (linear and branched isomers), and PFNA. The laboratory may not report results for all PFAS considered by the committee or may include different PFAS in its panel. In that case, the sum of PFAS should include only the PFAS in the analyte list that the committee considered. For example, if the lab tests for PFOA, PFOS, PFHxS, PFNA, and perfluorobutane sulfonic acid (PFBS), the summation should include PFOA, PFOS, PFHxS, and PFNA. Differing analytes lists may cause some variation in response. Still, as long as PFOA, PFOS, PFHxS, and PFNA are included in the analyte list, the results may not vary too greatly as these four analytes are most commonly detected in the United States. If any analyte is below the limit of detection, the clinician should calculate the analyte limit of detection divided by the square root of 2 and use this value in the summation. The sum thus derived should be compared against Figure S-6 to determine an appropriate clinical follow-up plan based on shared, informed decision making between patient and clinician.
REVISING ATSDR’S PFAS CLINICAL GUIDANCE
The committee recommends several changes to ATSDR’s guidance to ensure consistency with the findings, conclusions, and recommendations presented in this report and improve the guidance’s writing, design, dissemination, and implementation:
Recommendation 7-1: The Agency for Toxic Substances and Disease Registry (ATSDR) should update its PFAS clinical guidance to make it more succinct and accord with the review of PFAS-associated health effects, exposure reduction considerations, PFAS testing recommendations and interpretation, and recommendations for clinical follow-up presented in this report. When describing the health effects of PFAS, ATSDR should avoid using terms typically used to
categorize toxicants, such as “endocrine disrupter” or “neurotoxin,” because they are vague and not necessarily clinically meaningful. When discussing the strength of the association between PFAS and a health outcome, ATSDR should use standard categories of association (such as sufficient evidence of an association, limited or suggestive evidence of an association, inadequate or insufficient evidence of an association, and limited or suggestive evidence of no association).
NOTE: ATSDR = Agency for Toxic Substances and Disease Registry; MeFOSAA = methylperfluorooctane sulfonamidoacetic acid; PFDA = perfluorodecanoic acid; PFHxS = perfluorohexane sulfonic acid, PFNA = perfluorononanoic acid; PFOA = perfluorooctanoic acid; PFOS = perfluorooctanesulfonic acid; PFUnDA = perfluoroundecanoic acid.
Recommendation 7-2: The Agency for Toxic Substances and Disease Registry (ATSDR) should incorporate a reader-centered approach when developing its guidance, with the knowledge that many different audiences will turn to its clinical guidance document to prepare for discussions with their clinicians. ATSDR should also solicit feedback on the guidance from a variety of stakeholders, such as community groups, practicing clinicians, and medical associations. In addition, ATSDR should encourage clinicians to use evidence-based organizational health literacy strategies to support shared, informed decision making; patient-centered care; cultural humility; and accessible language when communicating with patients about potential health risks.
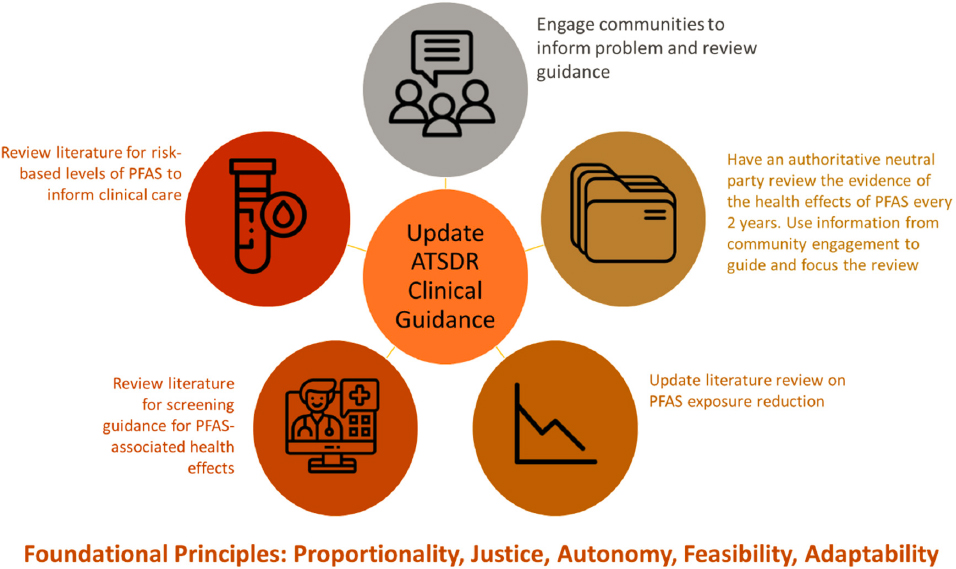
Evidence on the health effects of PFAS should be updated every 2 years, and the clinical guidance should be updated at least every 5 years. In that process, ATSDR should strive to ensure that its clinical guidance follows criteria for making guidelines trustworthy when possible. Figure S-8 provides an overview of how the process used by this committee could be applied by ATSDR to improve its clinical guidance.
Recommendation 7-3: The Agency for Toxic Substances and Disease Registry (ATSDR) should develop a process for updating its PFAS guidance that adheres to criteria for making guidelines trustworthy, such as being based on a thorough, transparent, unbiased review of the evidence and being developed by a knowledgeable panel of experts free from strong biases and conflicts of interest. A review of the evidence on the health effects of PFAS should be completed by an authoritative neutral party every 2 years, and the clinical guidance should be updated every 5 years or sooner if warranted by the evidence on the health effects of PFAS. Clinicians and members of communities with elevated PFAS exposure should be engaged to inform the problem and review updated guidance.
IMPLEMENTING THE COMMITTEE’S RECOMMENDATIONS TO IMPROVE PUBLIC HEALTH
Public health requires the use of multifaceted approaches to emerging health issues. In environmental health—the subset of public health focused on environmental factors—mitigation of potential harms associated with chemical exposures is often complicated because there is no exposure surveillance system for most chemicals. The committee’s recommendations will best protect the public health if they are part of a national effort focused on increased biomonitoring, exposure surveillance, and education of clinicians and public health professionals on environmental health issues:
Recommendation 8-1: Laboratories conducting PFAS testing of serum or plasma should report the results to state public health authorities, following the respective states’ statutes and reporting regulations. This reporting would improve PFAS exposure surveillance; it could be linked with the Centers for Disease Control and Prevention’s (CDC’s) environmental public health tracking network and help build capacity for improvements in the state-based national biomonitoring network.
The people and communities with high exposures to PFAS need to be identified. As the committee looks forward, it sees a pressing need for a robust environmental health infrastructure to continue to respond to PFAS and address other complex emerging and persistent environmental challenges.

















