1
Introduction
More than a decade ago, the United Nations General Assembly passed Resolution 64/292, recognizing that safe and clean drinking water is a human right and is essential to realizing all human rights. Yet today, drinking water in thousands of communities across the United States is contaminated with chemicals known as per- and polyfluoroalkyl substances (PFAS) (Hu et al., 2016) (see Box 1-1). PFAS do not occur in nature and are manufactured for a wide range of purposes, from nonstick cookware, to stain-resistant fabrics and carpets, to firefighting foams and protective gear. PFAS constitute a large class of up to 12,000 different chemicals (EPA, 2020). They are commonly used in thousands of products because they have desirable chemical properties that impart oil and water repellency, friction reduction, and temperature resistance (ITRC, 2017). PFAS as a class have a wide variety of distinct chemical properties and toxicities; for example, some PFAS can accumulate and persist in the human body and the environment, while others transform relatively quickly. The PFAS that do transform, however, will become one or more other PFAS, because the carbon–fluorine bond they contain does not break naturally. It is for this reason that PFAS are termed “forever chemicals.”
In the most rudimentary sense, PFAS can be thought of as either polymer or nonpolymer. Nonpolymer PFAS, such as perfluroalkyl acids (PFAAs), are more commonly detected in the environment and are often considered highly persistent and mobile (Blum et al., 2015; ITRC, 2021). By contrast, many polymer PFAS, such as polytetrafluoroethylene (PTFE), are insoluble, less bioavailable, and often considered less of a direct concern, with respect to human or ecological health (Lohmann et al., 2020; ITRC, 2021). Nonetheless, the production of fluoropolymers requires other PFAS chemicals, such as PFAAs. For example, the nonpolymer PFAS perfluorooctanoic acid (PFOA) was used as a surfactant in the emulsion polymerization of fluoropolymers.1
___________________
1 See https://www.acs.org/content/acs/en/molecule-of-the-week/archive/p/perfluorooctanoic-acid.html (accessed June 22, 2022).
SOCIOHISTORICAL TIMELINE OF PFAS
Public concern about the impact of PFAS contamination on human health and the environment began in the late 1990s when PFOA water contamination was identified in Parkersburg, West Virginia.2 In response, 3M, a primary PFAS manufacturer, initiated a voluntary phase-out of some PFAS (PFOA, perfluorooctanesulfonic acid [PFOS], and perfluorohexanesulfonic acid [PFHxS)]) (ITRC, 2017). The contamination in Parkersburg also led to a class action lawsuit that identified several health effects related to PFAS exposure and subsequently to the establishment of a medical monitoring program (see Box 1-2). Shortly thereafter, the U.S. Environmental Protection Agency (EPA) began requiring all community water systems serving more than 10,000 people to test for certain PFAS, which led to more communities learning that their water was contaminated.3 In 2016, researchers found that the drinking water supply for Wilmington, North Carolina, was contaminated with a chemical called “GenX,” a PFOA replacement.4 This finding led to public concern about the potential health effects of replacement PFAS (see Figure 1-1).
An estimated 2,854 U.S. locations (in all 50 states and two territories) have some level of PFAS contamination (see Figure 1-2). Although not all of the contamination represents exceedances of health advisories, the pervasiveness of the contamination is alarming. Furthermore, almost 100 percent of the U.S. population is exposed to at least one PFAS. Although exposures to the phased-out PFAS have been decreasing (see Figure 1-3), people are still exposed to those PFAS from site contamination, occupational uses of stored products, and breakdown of PFAS polymer products that are found in homes. Carpeting, for example, is often treated with fluorotelomer-based polymers that can biodegrade to form phased-out PFAS, such as PFOA (Washington and Jenkins, 2015). Exposures also occur to the PFAS chemicals used to replace those that have been phased out. Although the harms of the replacement PFAS are less well understood, they may have comparable or more serious toxicity than the PFAS they have replaced (Kwiatkowski et al., 2020). The state of New Jersey, for example, recently set a groundwater standard for
___________________
2 See https://pfasproject.com/parkersburg-west-virginia (accessed June 16, 2022).
3 See https://www.epa.gov/sites/default/files/2016-05/documents/ucmr3-factsheet-list1.pdf (accessed June 16, 2022).
4 See https://news.ncsu.edu/2018/04/finding-genx (accessed June 16, 2022).
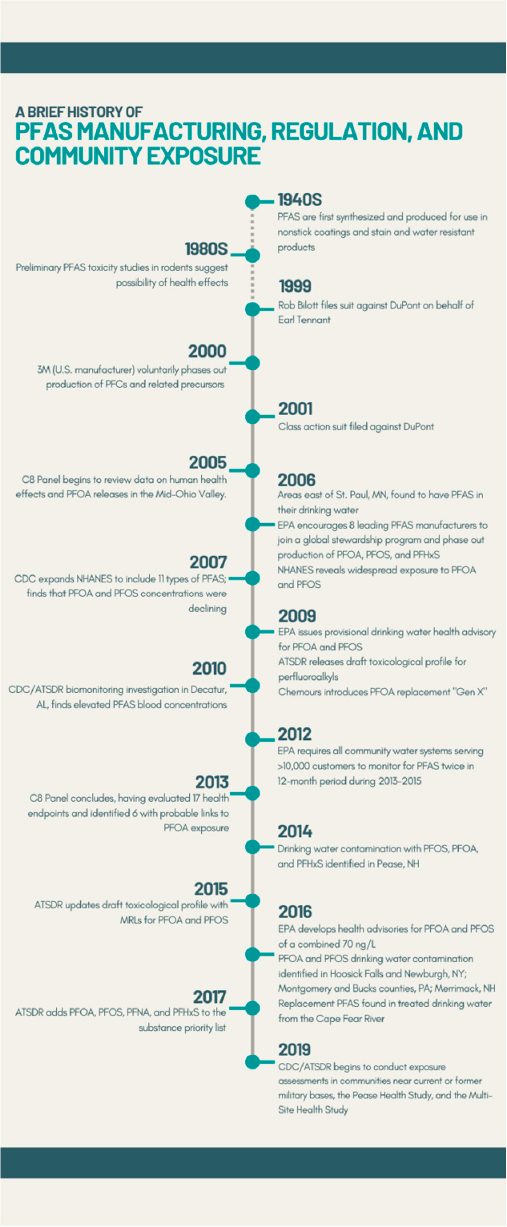
NOTE: ATSDR = Agency for Toxic Substances and Disease Registry; CDC = Centers for Disease Control and Prevention; EPA = U.S. Environmental Protection Agency; MRL = minimal risk limit; ng/L = nanograms per liter; NHANES = National Health and Nutrition Examination Survey; PFAS = per- and polyfluoroalkyl substances; PFC = perfluorochemicals; PFHxS = perfluorohexane sulfonic acid; PFNA = perfluorononanoic acid; PFOA = perfluorooctanoic acid; PFOS = perfluorooctane sulfonic acid.
SOURCE: Committee generated based on slides included by Patrick N. Breysse in a presentation to the committee on February 4, 2021.
a replacement PFAS (chloroperfluoropolyether carboxylates) that is an order of magnitude lower than drinking water standards for other PFAS. The New Jersey maximum contaminant levels of perfluorononanoic acid (PFNA), PFOA, and PFOS in drinking water are 13, 14, and 13 nanograms per liter (ng/L), respectively,5 while the groundwater standard for the replacement PFAS, chloroperfluoropolyether carboxylates, is 2 ng/L.6
A reason PFAS contamination became well known only recently, after decades of their use, is that they can be difficult to detect. The physical and chemical properties that make PFAS persistent and mobile in the environment also make them particularly challenging to analyze (Guelfo et al., 2021). Analytical methods sensitive enough to detect environmentally relevant concentrations became widely available in the early 2010s. Although analyte lists continue to expand, currently available methods still allow identification of only a small fraction of the thousands of PFAS that have reportedly been created and used since the 1950s. As existing analytical methods improve, additional PFAS and new release sites will likely be identified (De Silva et al., 2021; Guelfo et al., 2021).
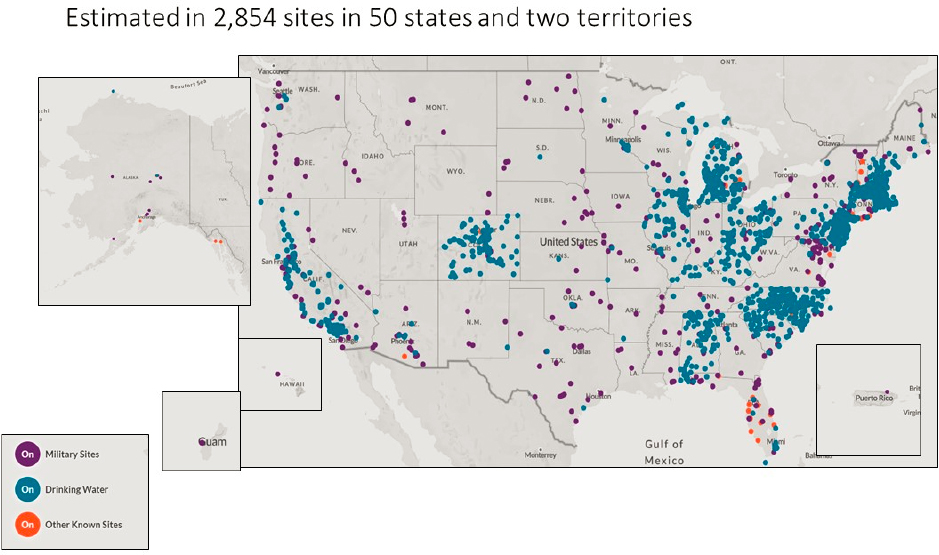
NOTES: According to the Environmental Working Group (EWG), “locations represented on the map are approximate and intended to portray the general area of a contamination site or a community water system. Locations were mapped using the best data available from official records, including data provided by tests of public drinking water systems, the Safe Drinking Water Information System and the Department of Defense report Addressing Perfluorooctane Sulfonate (PFOS) and Perfluorooctanoic Acid (PFOA), and Department of Defense public records, among others. Data on contaminated industrial and military sites was current as of October 2021.” Furthermore, “EWG has worked to ensure the accuracy of the information provided in this map. The map is dynamic. This contaminant site, results, suspected sources and other information in the database may change based on evolving science, new information or other factors. Please be advised that this information frequently relies on data obtained from many sources, and accordingly, EWG cannot guarantee the accuracy of the information provided or any analysis based thereon.”
SOURCE: See https://www.ewg.org/interactive-maps/pfas_contamination/map (accessed May 11, 2022). Copyright © Environmental Working Group, www.ewg.org. Reproduced with permission.
___________________
5 See https://www.nj.gov/health/ceohs/documents/pfas_drinking%20water.pdf (accessed June 8, 2022).
6 See https://www.nj.gov/dep/standards/ClPFPECA_Standard.pdf (accessed June 8, 2022).
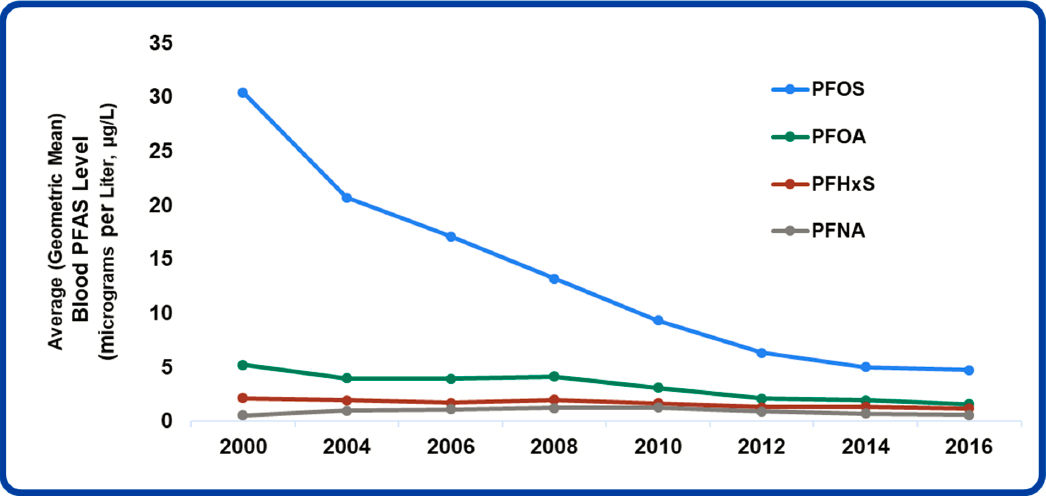
NOTE: Average = geometric mean.
SOURCE: Patrick N. Breysse’s presentation to the committee on February 4, 2021. DATA SOURCE: Centers for Disease Control and Prevention (2019, January). Fourth Report on Human Exposure to Environmental Chemicals, updated tables. Atlanta, GA: U.S. Department of Health and Human Services.
The people who live, work, and play in environments where PFAS contamination exceeds standards often do not know how to protect themselves from the health risks of exposure. Many have been exposed to PFAS for decades and may continue to be exposed in their homes or workplaces. Some members of communities with documented exposure report feeling violated and concerned about the health and well-being of their families, friends, and loved ones (Raponi et al., 2021; Rizzuto, 2021). Many in these exposed communities who have discovered that their exposure exceeds health advisory levels are calling for a medical program to prevent, lead to early detection of, or treat any diseases related to the health risks they may face (Raponi et al., 2021; Rizzuto, 2021). Developing such a program is challenging, however, because of the uncertainties about the health effects of PFAS and potential harms from additional medical treatments; moreover, many of these diseases are not preventable or even treatable once exposures have occurred (Wones et al., 2009). Accordingly, clinicians generally do not know how to respond to patients’ concerns about PFAS.
ENVIRONMENTAL JUSTICE
Compounding the challenge of responding clinically to PFAS contamination are issues of environmental justice and systemic racism in medical care. It is well established that environmental risks are not distributed uniformly across populations. Race, ethnicity, poverty, age, life stage, and other social factors can place people at disproportionately high risk for diseases with environmental causes as a result of hazardous exposures at increased levels compared to the general population (Gochfeld and Burger, 2011). While environmental justice research specific to PFAS contaminants has been limited, place-based factors that may put individuals at greater risk of exposure (siting of chemical companies, refineries, and industrial sites), coupled with insufficient access to environmental screening, information, and adequate health care, have disproportionate impacts on Black, Hispanic, and Indigenous communities, as well as low-income populations. A special report from Scientific American called this a “triple whammy of race, poverty, and environment converging nationwide to create communities near pollution sources where nobody else wants to live” (Kay and Katz, 2012, para. 12).
According to a 2014 paper by the Environmental Justice Health Alliance for Chemical Policy Reform, more than 134 million Americans live within the “vulnerability zones” of industrial facilities that store or use highly hazardous chemicals (Orum et al., 2014). The companies themselves define these vulnerability zones, determined by guidance from the EPA,7 as areas that could be affected by the release of toxic chemicals. The scope of the research on which this paper was based was broad, but included industries with known PFAS contamination, such as waste management and chemical manufacturing. The authors found that, relative to the U.S. average, communities within the vulnerability zones are disproportionately African American or Latinx; are more likely to live in poverty; and have lower housing values, incomes, and education levels. As the extent of contamination becomes known, housing values have been shown to decrease, and those with the means to move are more likely to do so, leaving behind those with the fewest resources and options (Harclerode et al., 2021).
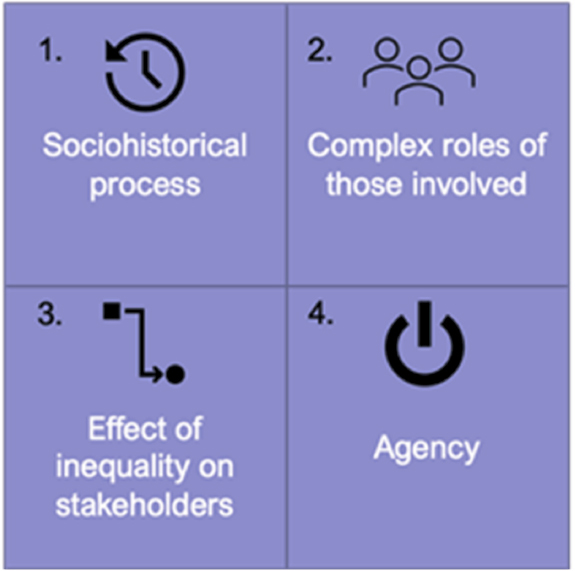
When considering environmental justice, it is important to think beyond exposure disparities (Mohai et al., 2009); core environmental justice issues relevant to PFAS also include rural health, industrial siting, and access to environmental exposure reduction (Bullard, 1996) and clinical care. A framework is needed that allows consideration of structural factors beyond race, ethnicity, and socioeconomic status and accounts for how individuals and communities are impacted by decisions made at the local, state, and policy levels by government, industry, and health care professionals. Pellow’s Environmental Justice Framework accounts for the complexity of relationships and decisions that impact PFAS exposure and associated health outcomes (Pellow, 2004) (see Figure 1-4). Sze and London (2008) advance this argument by incorporating negotiation at the stakeholder level, as well as problem identification at the level of sociohistorical and structural factors, and then solution-oriented approaches reflecting considerations of sustainability and safety.
___________________
7 See https://www.epa.gov/rmp/forms/vulnerable-zone-indicator-system (accessed June 16, 2022).
A first consideration in addressing environmental justice with respect to PFAS is understanding the sociohistorical context rather than a particular discrete event that has contributed to the presence of PFAS in certain communities. For example, PFAS contamination did not just randomly occur in rural communities serviced by well water that happened to be near industrial sites. Rather, the locating of certain industrial sites and decisions to dispose of PFAS with limited regard for the surrounding community’s access to safe water are rooted in the relationship and history of these industries and communities. The disposal of these chemicals did not occur as single events lacking context, but reflected a pattern of decisions made over time. Understanding the historical and social context influencing how and where PFAS are distributed is an essential part of identifying effective mitigation strategies.
Second, Pellow’s Environmental Justice Framework emphasizes the complex roles of the stakeholders involved. In the case of PFAS, environmental inequality can affect many different stakeholders, ranging from industry, to workers, to community members and organizations, to government entities such as local departments of public health. The complexity of these roles means there can exist synergistic and contradictory allegiances that impact what is valued, how resources are accessed and distributed, who has power, and what is considered profitable. With PFAS, industry’s power and profit motive may trump a community’s access to information and ability to test for and reduce exposure. Likewise, key health information for clinicians is governed by boards that decide about the content of medical education and training. Clinicians’ lack of knowledge about environmental exposures such as PFAS, particularly in highly contaminated communities, can be particularly deleterious for the least-advantaged stakeholder groups.
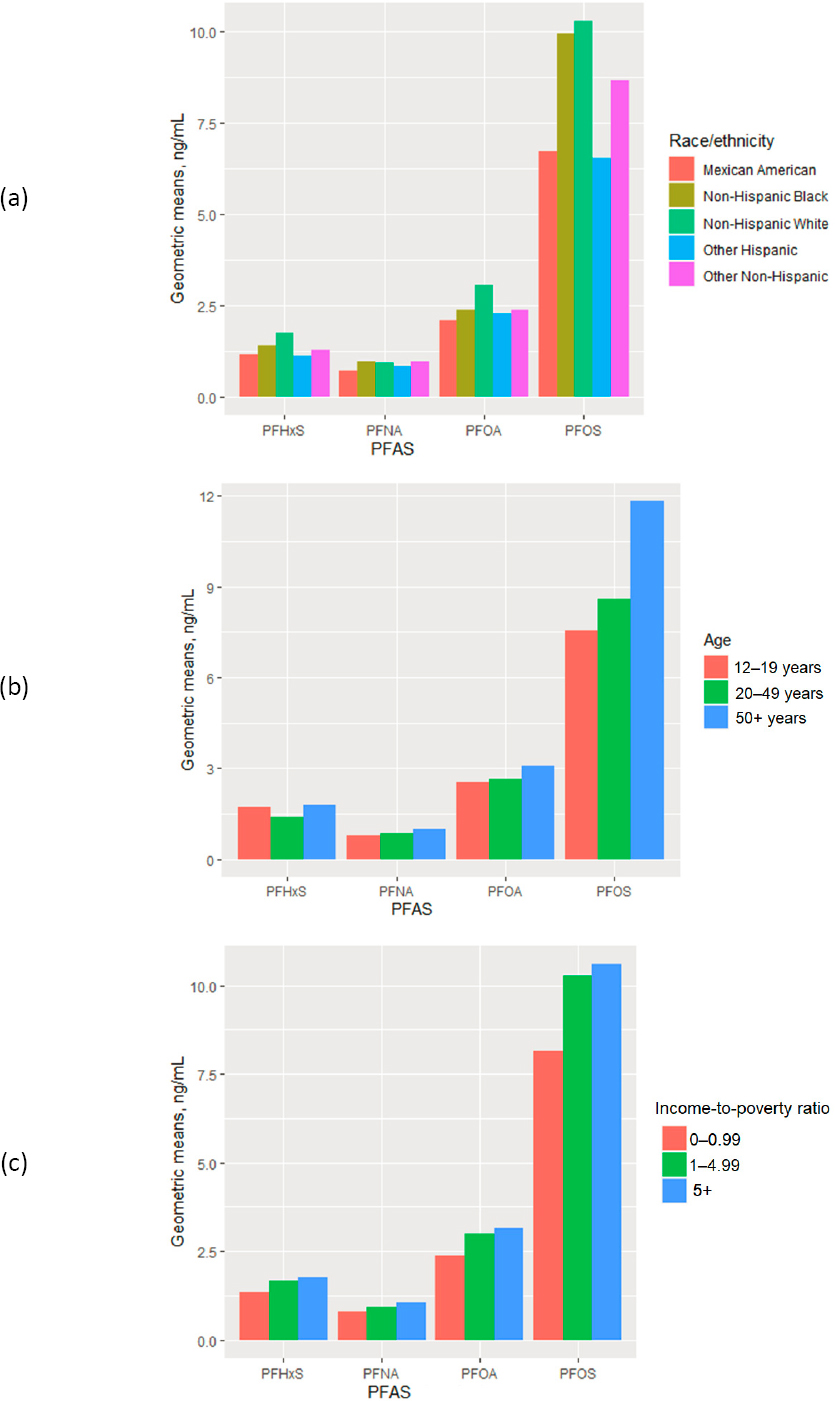
A third consideration is the effects of social inequalities on exposure to PFAS, as relevant social stratification that affects exposure is not always clear or easy to study. Rural and urban disparities, for example, can be analyzed using National Health and Nutrition Examination Study (NHANES) data (with appropriate clearance), and living in a rural versus an urban area can definitely affect PFAS exposure, often through exposure to well water that is common in rural areas. Other, less obviously measurable influences can also drive exposures, however. Food insecurity, for example, even if only temporary, increases subsistence fishing (Quimby et al., 2020), which may cause people to fish for food in contaminated lakes or rivers. These structural and social factors put people at risk of exposure and can contribute to inequality. In short, even though relatively affluent areas can be heavily contaminated with PFAS, and non-Hispanic Whites are exposed to some of the highest PFAS concentrations (see below), solving the larger problem of PFAS contamination will still require attention to persistent social inequities. Otherwise, action to mitigate PFAS exposure could aggravate existing (or create new) environmental health disparities.
Fourth is the need to consider the role of agency—specifically, the power a community has to advocate for itself in conflicts. PFAS provide a unique example. PFAS contamination rose to national prominence partly because of exposures among well-educated, high-income, and mainly White communities. With PFAS, as with many social problems, power matters. At the same time, PFAS exposure varies regionally. In some parts of the country, the highest exposures are borne by less advantaged communities, making their relative ability to advocate for testing and responsive health care or to demand effective mitigation from their government somewhat tenuous.
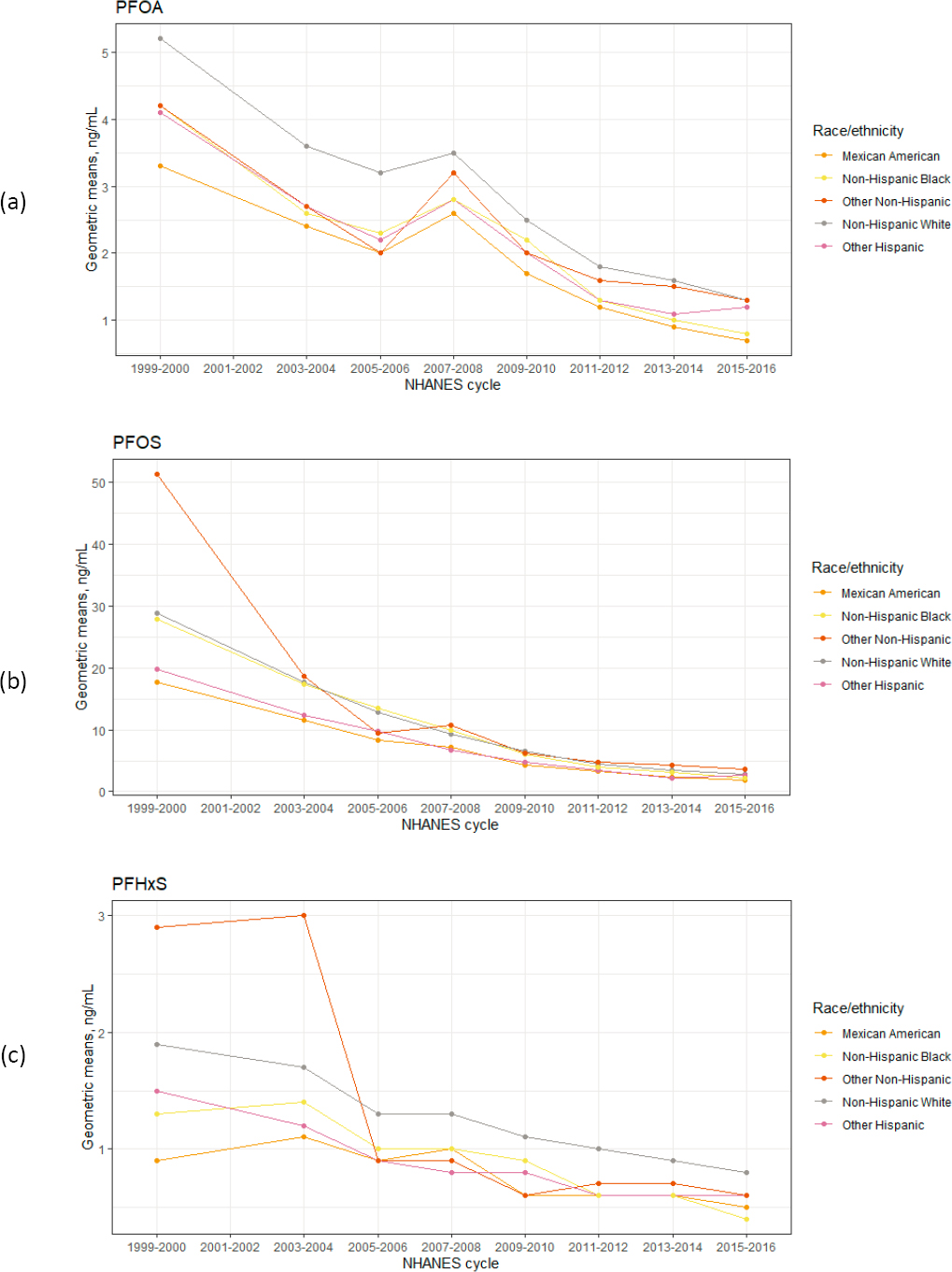
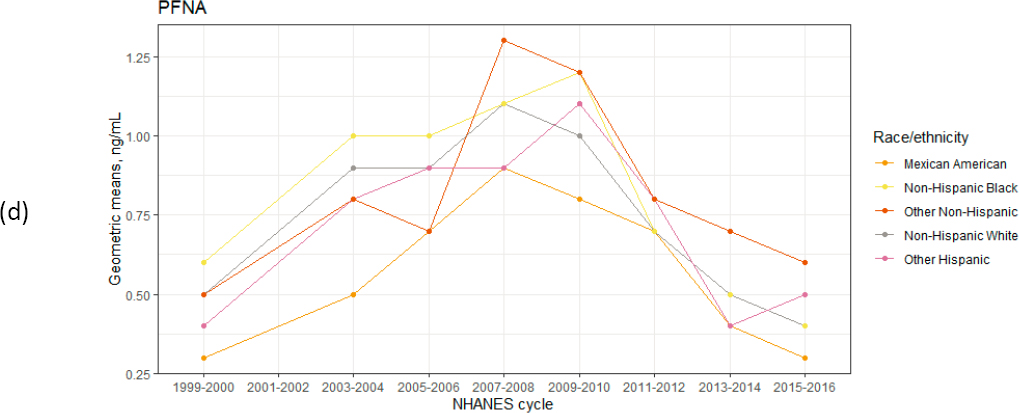
While research on disparities in PFAS exposure is limited, several studies suggest that no differences exist by race, ethnicity, or socioeconomic status (Buekers et al., 2018; Nguyen et al., 2020; Sagiv et al., 2015). If one looks at time-aggregated estimates of PFAS exposure for the U.S. population, non-Hispanic Whites, older adults, and people of higher socioeconomic status have higher concentrations of these chemicals (see Figure 1-5). However, time-aggregated exposures present an incomplete picture. Looking at PFAS exposure by year stratified by race orethnicity suggests that in 2000, before the voluntary phase-out of PFOS andrelated chemicals, those in the racial group other non-Hispanic had the highest exposure to PFOS and PFHxS (see Figure 1-6). The implication of this finding is that widespread policy changes—such as the phase out of certain types of PFAS—greatly reduced exposure disparities. Because there are no national estimates of biomonitoring data for replacement PFAS, however, it is unknown whether the disparities observed with PFOS, PFHxS, and PFNA exist today for other PFAS.
PFAS CONTAMINATION AND ROUTES OF EXPOSURE
PFAS contamination is global. PFAS have been detected in regions with little human activity, including the atmosphere of remote locations (Shoeib et al., 2010), the Arctic and Antarctic seas (Armitage et al., 2006), and remote soils of every continent (Rankin et al., 2016). Environmental contamination with PFAS occurs in countless ways. Fluorochemical manufacturing sites release PFAS into water as well as into the air, from which they can settle into both soil and water. At military sites and commercial airports PFAS-containing foams are used in training exercises for firefighting. PFAS also can leak from landfills where PFAS-containing wastes are disposed, and can be released into the environment by PFAS-containing wastewater from wastewater treatment plants (Evich et al., 2022; Gomez et al., 2021; Sunderland et al., 2019) (see Figure 1-7). PFAS-treated consumer products have been implicated as sources of exposure in indoor settings (Harrad et al., 2010), and fluorotelomer products used to treat a wide range of consumer household and occupational products have been shown to degrade to form PFAS (Washington and Jenkins, 2015).
PFAS in the environment can contaminate drinking water when the chemicals reach public drinking water systems and private wells (Hu et al., 2016). PFAS can bioaccumulate in fish, shellfish, livestock, dairy, and game animals that contact them through contaminated food or water (De Silva et al., 2021; Death et al., 2021; Domingo and Nadal, 2017). Produce also can be contaminated if it is grown with contaminated drinking water or PFAS-contaminated compost or biosolids (Blaine et al., 2013; Scher et al., 2018).
Certain occupations may lead to increased PFAS exposures. They include, for example, jobs in facilities used to manufacture fluorochemicals or to produce PFAS-containing products, such as textiles or food contact materials. Other jobs with a known increased risk of exposure to PFAS include electroplating, painting, carpet installation and treatment, and jobs that require prolonged work with ski wax; increased exposures also occur among military and civilian firefighters who use PFAS-containing foams in training exercises and wear PFAS-impregnated gear (ATSDR, 2021). In addition, food workers and others in the hospitality industry may have elevated exposure if they handle PFAS-containing food packaging as part of their job (Carnero et al., 2021; Curtzwiler et al., 2021; Schaider et al., 2017).
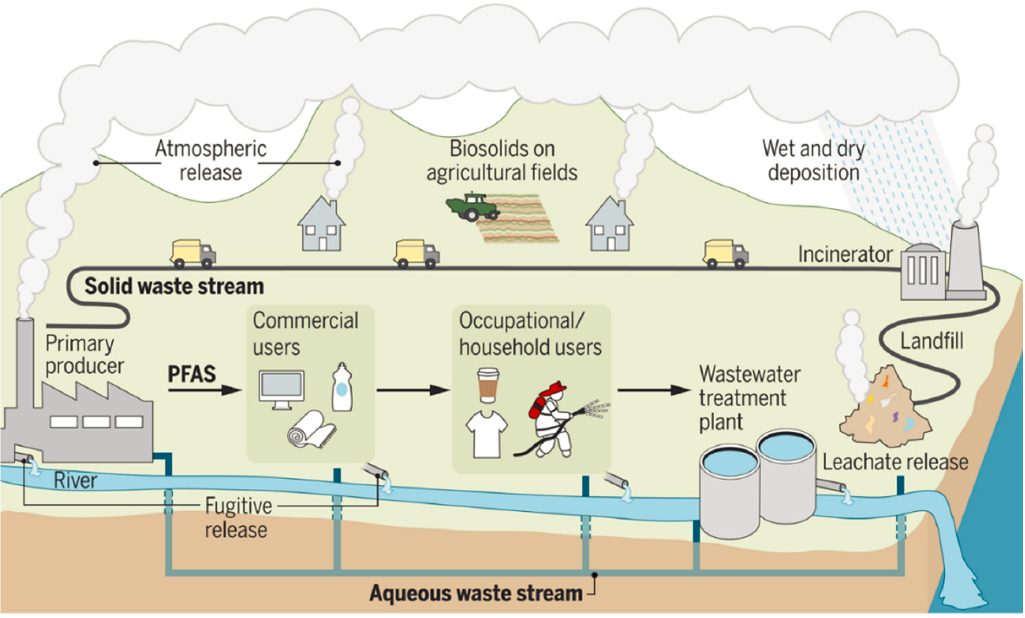
SOURCE: Evich et al., 2022.
Ingestion is the most well-studied route of exposure to PFAS in nonoccupational settings (Trudel et al., 2008). PFAS can be ingested by drinking contaminated water or by eating contaminated seafood or other contaminated foods, such as vegetables, game, or dairy products (Bao et al., 2019, 2020; Death et al., 2021; Domingo and Nadal, 2017; Herzke et al., 2013; Li et al., 2019). PFAS are often used in cookware and food contact materials, such as microwave popcorn bags and packaging of fast foods and processed foods (Carnero et al., 2021; Curtzwiler et al., 2021; Schaider et al., 2017). Exposure can also occur through accidental ingestion of PFAS-containing dusts (ATSDR, 20218). PFAS cross the placenta, and PFAS from the mother’s body burden can be passed on to her developing fetus (Gao et al., 2019; Manzano-Salgado et al., 2015); PFAS can also pass from mother to child through breast milk (Serrano et al., 2021; Zheng et al., 2021).
Inhalation and transdermal absorption are less well studied. Inhalation is well documented in occupational settings that use aerosolized PFAS (Gilliland, 1992). Volatile PFAS have been detected indoors (Fromme et al., 2015; Morales-McDevitt et al., 2021) and have been associated with persistent PFAS in serum (Fraser et al., 2013). Inhalation near factory emissions and incinerators contributes to exposures in nearby communities (ATSDR, 20219). Inhalation from showering in contaminated water is an active area of research, but there are as yet no data formally evaluating this route.
PFAS are used in thousands of products (e.g., water- and stain-resistant clothing) and personal care products (e.g., sunscreen, makeup, dental floss). They also are used in such products as paint, textiles, firefighting foam, electroplating materials, ammunition, climbing ropes, guitar strings, artificial turf, and soil remediation substances (Glüge et al., 2020). The extent to which the use of such products contributes to human exposures remains unclear, however, because the relative contribution of PFAS exposures from sources other than food or water is not well characterized (DeLuca et al., 2021).
___________________
8 This text was altered to correct the references after release of a pre-publication version of the report.
9 This text was altered to correct the references after release of a pre-publication version of the report.
POLICIES THAT COULD REDUCE EXPOSURE TO PFAS
The EPA’s major policy levers for reducing harmful exposures to PFAS are the Toxic Substances Control Act (TSCA), which limits chemicals in commerce; the Safe Drinking Water Act; and the Comprehensive Environmental Response, Compensation, and Liability Act (CERCLA, commonly known as the “Superfund”), which designates hazardous sites for cleanup.
In 2000, the 3M Company (Maplewood, Minnesota), the primary U.S. manufacturer of PFOS, began a voluntary phase-out of perfluorooctanyl chemicals and related precursors, including PFOS, PFHxS, and PFOA8 (ITRC, 2017). Between 2002 and 2013, the EPA issued four Significant New Use Rules under the TSCA that prevented others from manufacturing the chemicals 3M had voluntarily phased out (Richter et al., 2021) and required notifying the agency prior to any manufacture, use, or import of 271 other PFAS.9 In 2006, the EPA encouraged eight leading PFAS manufacturers to join a global stewardship program focused on phasing out and ending the production of PFOA. At the same time, the TSCA allows some industries (including photographic imaging, semiconductors, etching, metal plating, and aviation) to continue using banned PFAS at low levels, and these industries are exempted from the provision requiring that the EPA be notified of any new use of banned PFAS (ITRC, 2017). Furthermore, these bans apply only to a handful of chemicals, most of which could easily be substituted for by other PFAS. And other PFAS not yet banned remain in use by industry while undergoing further biologic profiling and toxicity evaluation (Houck et al., 2021).
If PFAS were designated as hazardous substances, CERCLA could be used to lower exposures from contaminated sites, giving the EPA more authority to investigate and remediate those waste sites. The agency has not yet designated PFOS or PFAS as hazardous, however. And the Safe Drinking Water Act would be more protective if the standards were enforceable.10
Policy changes that could reduce PFAS exposures may be forthcoming. Recently, the EPA established an EPA Council on PFAS charged with building on the agency’s ongoing work to understand better and ultimately reduce the potential risks posed by these chemicals. A PFAS Strategic Roadmap also was released, committing the agency to action. In addition, the EPA released for review by a science advisory board draft documents that will inform an enforceable National Primary Drinking Water Regulation for PFOA and PFOS.11
The EPA is not alone in its lack of action. The U.S. Food and Drug Administration (FDA) has delayed limiting PFAS in bottled water,12 but has revoked regulations that allowed for long-chain PFAS (such as PFOA and PFOS) in food packaging in 2016,13 and reached an agreement to phase out PFAS substances containing 6:2 fluorotelomer alcohol (Hahn, 2020). The Occupational Safety and Health Administration (OSHA) has set no occupational exposure limits.
The lack of federal standards has left states and communities responsible for creating policies to reduce exposures to these chemicals in the interest of protecting public health (Brennan et al., 2021). There is no consistency in these policies, and not all states have the authority to set standards more protective than those of the federal government (ECOS, 2020). Furthermore, leaving policy making to
___________________
8 See https://archive.epa.gov/epapages/newsroom_archive/newsreleases/33aa946e6cb11f35852568e1005246b4.html (accessed June 16, 2022).
9 See https://www.epa.gov/assessing-and-managing-chemicals-under-tsca/risk-management-and-polyfluoroalkylsubstances-pfas (accessed June 16, 2022).
10 See https://www.epa.gov/pfas/epa-actions-address-pfas (accessed June 16, 2022).
11 See https://www.epa.gov/pfas/pfas-strategic-roadmap-epas-commitments-action-2021-2024 (accessed June 16, 2022).
12 See https://www.consumerreports.org/bottled-water/fda-delays-setting-limits-on-pfas-in-bottled-water-a8292013869 (accessed June 30, 2022).
13 See https://www.fda.gov/food/chemical-contaminants-food/authorized-uses-pfas-food-contact-applications (accessed June 16, 2022).
state governments encourages a confusing patchwork of rules and advice. Health inequities thrive in such chaotic circumstances.
PROVIDING CLINICAL ADVICE IN COMMUNITIES EXPOSED TO PFAS
To help clinicians14 respond to patient concerns about PFAS exposure, the Agency for Toxic Substances and Disease Registry (ATSDR) published PFAS: An Overview of the Science and Guidance for Clinicians on Per- and Polyfluoroalkyl Substances.15 This guidance summarizes general information about PFAS and PFAS health studies and suggests answers to example patient questions. However, the ATSDR guidance does not provide specific recommendations on when to test for PFAS, how to order the tests, how to interpret the results, or what clinical follow-up based on PFAS exposure might look like. Interpretation of PFAS blood or urine results is challenging because the specific level of exposure at which harms may occur is unknown, and the science on the potential health effects of exposure to PFAS is advancing quickly, making it difficult to advise clinicians about what follow-up tests might be helpful.
ATSDR has the legal authority to issue guidance to clinicians in its “Criteria for Determining the Appropriateness of a Medical Monitoring Program under Comprehensive Environmental Response, Compensation, and Liability Act (CERCLA),”16 promulgated in 1995 (42 USC 9604[i][9]). Under the criteria, the purpose of medical monitoring is to detect individuals with exposures to hazardous substances and refer them to medical care for further evaluation and treatment. The criteria recommends actions that clinicians can take, such as early detection, treatment, or other interventions that interrupt “the progress to symptomatic disease, improve the prognosis of disease, improve the quality of life of the affected individual, or address diseases that are amenable to primary prevention” (42 USC 9604[i][14], p. 38842). Medical monitoring under the criteria is not a research mechanism, and suggests that other epidemiologic studies be carried out to further investigate the cause–effect relationship between exposures and health outcomes. ATSDR’s authority to issue guidance to clinicians comes from a different section in the same law (42 USC 9604[i][14]), which grants the agency the authority to develop education materials on medical surveillance, screening, and methods of diagnosing and treating injury or disease related to exposure to hazardous substances.
The first well-documented case of community water contamination with PFAS resulted in recommendations for medical monitoring (see Box 1-2). Releases of PFOA-contaminated drinking water from the DuPont Washington Works facility near Parkersburg, West Virginia, were detected in the local area and a few nearby communities across the Ohio River. A class action lawsuit was filed—Jack W. Leach et al. v. E.I. du Pont de Nemours & Company (no. 01-C-608 W.Va., Wood County Circuit Court, filed April 10, 2002). The lawsuit resulted in a medical monitoring program that included suggested screenings for high cholesterol, thyroid disease, ulcerative colitis, testicular cancer, kidney cancer, pregnancy-induced hypertension, and blood testing for PFOA (Frisbee et al., 2009). The medical monitoring guidance generated by this high-profile case has led some community members to believe that the ATSDR guidance should provide recommendations for clinical follow-up, and advocates in many PFAS-exposed communities are working to establish medical monitoring programs (Rizzuto, 2021).
The absence of recommendations on testing and follow-up in ATSDR’s PFAS clinical guidance is aligned with standard medical practice, as clinicians are expected to order tests only when how to interpret and act on the results is known. In addition, reporting of levels of PFAS in blood and urine to patients raises ethical questions, such as whether to report results in the absence of established health guidelines.
___________________
14 The committee uses “clinician” throughout this report to refer to “a healthcare professional qualified in the clinical practice of medicine. Clinicians may be physicians, nurses, pharmacists, or other allied health professionals” as defined by the Centers for Medicare & Medicaid Services (https://www.cms.gov/Medicare/Quality-InitiativesPatient-Assessment-Instruments/MMS/QMY-Clinicians [accessed June 16, 2022], para 2).
15 See https://www.atsdr.cdc.gov/pfas/docs/clinical-guidance-12-20-2019.pdf (accessed June 16, 2022).
16 See https://www.govinfo.gov/content/pkg/FR-1995-07-28/pdf/FR-1995-07-28.pdf (accessed June 16, 2022).
At a workshop on PFAS exposure that occurred more than 1 year before the present study began, however, Andrea Amico, a member of a PFAS-impacted community in Pease, New Hampshire, traveled to Washington, DC, to represent her community’s views on PFAS testing. She said that people in her community
were exposed … without their consent, and now they have to fight tooth and nail to get a blood test result to know how much exposure they had? It just seems incredibly wrong…. We don’t have all the answers yet, but not testing them is not the right answer.
Amico also argued for more access to testing despite the knowledge gaps regarding the interpretation of test results. She believes PFAS exposure testing would allow people to compare their levels with those of others in highly exposed communities, which could help them understand their potential health risks (NASEM, 2020). As it stands now, clinicians in communities with known PFAS contamination are left to make their own decisions about whether to test patients for PFAS exposure, which PFAS to test for, how to interpret the results, what health effects are associated with PFAS, and how to determine appropriate follow-up care for exposed patients. Pressed for time in clinic visits, clinicians may dismiss interest in PFAS testing or avoid recommending specific medical follow-up, and they may not even be clear on when or how to order a test.
COMMITTEE’S TASK AND APPROACH
Prompted by the tension between people in PFAS-exposed communities wanting preventive care for their exposure and clinicians not knowing what care should be provided, ATSDR and the National Institute of Environmental Health Sciences (NIEHS) asked the National Academies of Sciences, Engineering, and Medicine to form an ad hoc committee to provide advice on clinical care for patients exposed to PFAS. The committee included experts in epidemiology, toxicology, preventive medicine, pediatrics, nursing, public health, environmental medicine, philosophy, ethics, exposure science, and risk communication (see Appendix A for biographical information on the committee members). The committee’s Statement of Task is provided in Box 1-3.
Figure 1-8 provides an overview of the steps in the committee’s approach to its charge: problem formulation and engagement with communities, development of principles for decision making under substantial scientific uncertainty, determination of strategies for reducing exposure to PFAS, development of advice for PFAS testing and levels of PFAS in blood or urine that could inform clinical care of exposed patients, determination of the health effects of PFAS, appropriate follow-up care of exposed patients, and advice on whether changes are needed with respect to ATSDR’s PFAS clinical guidance.

NOTE: ATSDR = Agency of Toxic Substances and Disease Registry; CDC = Centers for Disease Control and Prevention; PFAS = per- and polyfluoroalkyl substances.
Problem Formulation and Community Engagement
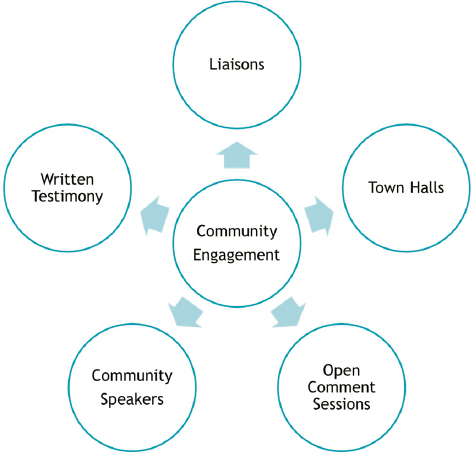
The committee held six public meetings between February 4 and August 12, 2021 (see Appendix C for the agendas of these public meetings). Leading up to its first meeting, the committee began to receive public input on the study. The testimony provided at the first public meeting revealed that many members of the public feel ignored or dismissed by clinicians and the medical community, and have assumed the role of community scientists, becoming experts in their communities’ illnesses and experiences. People who live with potentially harmful exposures can have knowledge that is inaccessible to scientists as their experiential learning provides essential context for scientific findings (Brown, 1992). To incorporate this knowledge into the study, the committee, National Academies staff, and the study sponsors developed a community engagement plan. To aid in this process, the committee considered the Jemez Principles for Democratic Organizing, a set of recommendations developed to serve as a foundation on which diverse coalitions can together make justice-based decisions specifically weighing on inclusivity, as well as letting people speak for themselves (Solís and SPWU, 1997). Ultimately, the committee’s community engagement plan consisted of several key elements (see Figure 1-9):
- a panel of community liaisons;
- three 2021 town halls (April 7, Eastern Town Hall; May 6, Middle Town Hall; May 25, Western Town Hall);
- community speakers from PFAS-impacted communities at every public meeting;
- open sign-up for public testimony to the committee at every public meeting; and
- encouragement for providing written testimony to the committee throughout the data-collection phase of the study.
The community liaisons were solicited through a nomination process similar to that used for National Academies committees. The community liaisons were intended to be an inclusive group; all nominees who appeared to be genuinely interested or had experience working with PFAS or similar environmental contamination issues were asked to join the group. National Academies staff held several meetings with the liaisons throughout the study process. The first call with the liaisons clearly described the mission of the National Academies, the goals of the study, what the study would not address, and the study timeline.
The role of the community liaisons was to provide input to the committee on behalf of PFAS-impacted communities. The liaisons aided the study process by suggesting speakers, topics, and discussion questions for the public meetings, and by answering questions to inform the report or the committee’s work. They also prepared letters to the committee providing consensus opinions related to the Statement of Task and suggested reviewers for the report for consideration by National Academies staff.
Health Effects Associated with PFAS Exposure
The committee carefully considered the purpose of the literature review of putative health effects prescribed in its Statement of Task: to establish a basis for prioritized clinical surveillance or monitoring of PFAS health effects. Accordingly, the committee’s review was focused on determining a set of health effects that may be associated with PFAS, which could then be used for preventive medicine recommendations and decisions. The Statement of Task limited the review to those PFAS included in the Centers for Disease Control and Prevention’s (CDC’s) National Report on Human Exposure to Environmental Chemicals (see Table 1-1).
Although the committee believes that studies on health effects in humans will most likely be limited to those PFAS in Table 1-1 because they are the most commonly studied in humans, other PFAS may cause harm because of similarities in biological persistence and toxicities (Kwiatkowski et al., 2020). Moreover, while different PFAS have distinct physical, chemical, and toxicological properties, people are exposed to more than a single PFAS. As a result, exposures are often to mixtures of PFAS such that specific effects are difficult to disentangle. Considering these issues and recognizing that some PFAS are less frequently measured than others, the committee ultimately decided to provide one strength-of-evidence determination for all PFAS for each health effect. Further description of the methods used for the committee’s review is provided in Appendix D.
| Abbreviated Name | Full Name | CAS Registry No. |
|---|---|---|
| MeFOSAA | Methylperfluorooctane sulfonamidoacetic acid | 2355-31-9 |
| PFHxS | Perfluorohexanesulfonic acid perfluorohexane sulfonic acid | 355-46-4 |
| n-PFOA (linear isomer), Sb-PFOA (branched isomers) | Perfluorooctanoic acid | 335-67-1* |
| PFDA | Perfluorodecanoic acid | 335-76-2 |
| PFUnDA | Perfluoroundecanoic acid | 2058-94-8 |
| n-PFOS (linear isomer), Sm-PFOS (branched isomers) | Perfluorooctanesulfonic acid | 1763-23-1* |
| PFNA | Perfluorononanoic acid | 375-95-1 |
NOTES: CAS = Chemical Abstracts Service. * = CAS number refers to linear isomer only. Previous survey years have also included perfluorobutane sulfonic acid (PFBS), perfluoropentanoic acid (PFpA), perfluorododecanoic acid (PFDoDA), perfluorooctane sulfonamide (FOSA), and 2-(N-ethyl-perfluorooctane sulfonamido)acetate (EtFOSAA), according to Patrick N. Breysse’s presentation to the committee on February 4, 2021.
Reducing Exposure to PFAS
This report does not review EPA policy actions, which are beyond the scope of the committee’s task. A review of individual-level exposure reduction strategies is presented in Appendix E. Chapter 4 provides an overview of that literature and medical interventions to reduce PFAS body burden. The report reviews these individual-level strategies because the task calls for them; however, the committee does not believe that people should be responsible for protecting themselves from harmful chemicals. Well-established exposure prevention frameworks, such as the hierarchy of controls and the health impact pyramid, dictate that individuals should not be responsible for making such decisions (Frieden, 2010). Systems approaches to exposure reduction, such as setting an enforceable water standard, regulating PFAS from nonessential uses, and cleaning up hazardous waste sites, will be far more effective at reducing population-level exposures (see Figure 1-10).
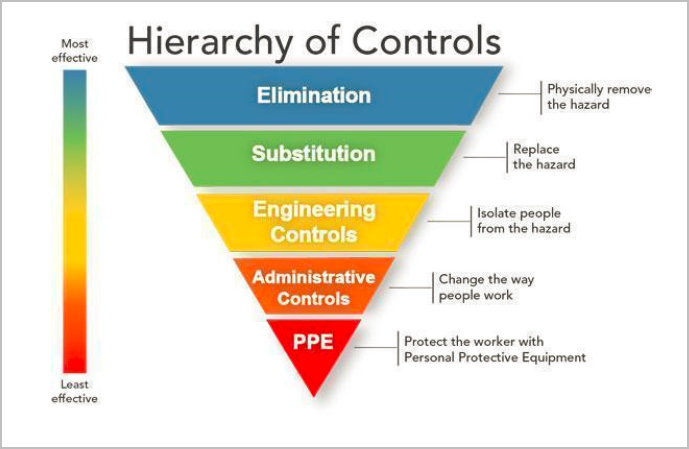
SOURCE: See https://www.cdc.gov/niosh/topics/hierarchy/default.html (accessed June 16, 2022).
Advice on PFAS Testing and Levels That Could Inform Clinical Care
The committee’s approach to providing advice on PFAS testing and clinical care included a review of the harms and benefits of testing. To determine the levels of PFAS exposure that could inform clinical care of exposed patients, the committee did not complete dose-response modeling to determine a tolerable risk level. Instead, the committee reviewed various strategies for interpreting PFAS concentrations, such as use of reference ranges for comparison, and levels of risk determined by other agencies.
Follow-Up Care for Patients Known or Suspected to Be Exposed to PFAS
The committee considered relevant for patient follow-up recommendations for each condition that its review found had suggestive or limited suggestive evidence of an association with PFAS exposure. The committee believed that while it was important to categorize the strength of the evidence, all conditions with an association should be considered for patient follow-up since acknowledging the potential risk may make clinicians and patients more likely to prioritize screenings.
ORGANIZATION OF THE REPORT
This report is organized into eight chapters and five appendixes. Chapter 2 presents the committee’s general principles for decision making under substantial scientific uncertainty. Chapter 3 presents the committee’s assessment of the health effects of PFAS. Chapter 4 reviews strategies for reducing exposure to PFAS. Chapter 5 provides recommendations for PFAS testing and concentrations in blood or urine that could inform clinical care. Chapter 6 presents the committee’s specific recommendations for patient follow-up. Chapter 7 addresses suggested changes to the ATSDR PFAS clinical guidance. Finally, Chapter 8 considers the implementation of the committee’s recommendations. Appendix A provides biographical information on the committee members, National Academies staff, and the community liaisons; Appendix B provides a summary of the town halls; Appendix C contains the agendas of the committee’s public meetings; Appendix D is a summary of the methods used in the committee’s literature review; and Appendix E is a white paper describing a review of the PFAS personal intervention literature.
REFERENCES
Armitage, J., I. T. Cousins, R. C. Buck, K. Prevedouros, M. H. Russell, M. MacLeod, and S. H. Korzeniowski. 2006. Modeling global-scale fate and transport of perfluorooctanoate emitted from direct sources. Environmental Science & Technology 40(22):6969–6975. https://pubs.acs.org/doi/10.1021/es0614870.
ATSDR (Agency for Toxic Substances and Disease Registry). 2021. Toxicological profile for perfluoroalkyls. Atlanta, GA: U.S. Department of Health and Human Services. https://www.atsdr.cdc.gov/toxprofiles/tp200.pdf (accessed July 1, 2022).
Bao, J., W. J. Yu, Y. Liu, X. Wang, Y. H. Jin, and G. H. Dong. 2019. Perfluoroalkyl substances in groundwater and home-produced vegetables and eggs around a fluorochemical industrial park in China. Ecotoxicology and Environmental Safety 171:199. https://doi.org/10.1016/j.ecoenv.2018.12.086.
Bao, J., C. L. Li, Y. Liu, X. Wang, W. J. Yu, Z. Q. Liu, L. X. Shao, and Y. H. Jin. 2020. Bioaccumulation of perfluoroalkyl substances in greenhouse vegetables with long-term groundwater irrigation near fluorochemical plants in Fuxin, China. Environmental Research 188:109751. https://doi.org/10.1016/j.envres.2020.109751.
Blaine, A. C., C. D. Rich, L. S. Hundal, C. Lau, M. A. Mills, K. M. Harris, and C. P. Higgins. 2013. Uptake of perfluoroalkyl acids into edible crops via land applied biosolids: Field and greenhouse studies. Environmental Science and Technology 47:14062. https://doi.org/10.1021/es403094q.
Blum, A., S. Balan, M. Scheringer, X. Trier, G. Goldenman, I. Cousins, M. Diamond, T. Fletcher, C. Higgins, and A. Lindeman. 2015. The Madrid statement on poly- and perfluoroalkyl substances (PFASs). Environmental Health Perspectives 123(5):A107–A111. https://doi.org/10.1289/ehp.1509934.
Brennan, N. M., A. T. Evans, M. K. Fritz, S. A. Peak, and H. E. von Holst. 2021. Trends in the regulation of per- and polyfluoroalkyl substances (PFAS): A scoping review. International Journal of Environmental Research and Public Health 18(20):10900. https://doi.org/10.3390/ijerph182010900.
Brown, P. 1992. Popular epidemiology and toxic waste contamination: Lay and professional ways of knowing. Journal of Health and Social Behavior 33(3):267–281. https://doi.org/10.2307/2137356.
Buck, R. C., J. Franklin, U. Berger, J. M. Conder, I. T. Cousins, P. D. Voogt, A. A. Jensen, K. Kannan, S. A. Mabury, and S. P. J. van Leeuwen. 2011. Perfluoroalkyl and polyfluoroalkyl substances in the environment: Terminology, classification, and origins. Integrated Environmental Assessment and Management 7(4):513–541. https://doi.org/10.1002/ieam.258.
Buekers, J., A. Colles, C. Cornelis, B. Morrens, E. Govarts, and G. Schoeters. 2018. Socio-economic status and health: Evaluation of human biomonitored chemical exposure to per- and polyfluorinated substances across status. International Journal of Environmental Research and Public Health 15(12):2818. https://doi.org/10.3390/ijerph15122818.
Bullard, R. D. 1996. Environmental justice: It’s more than waste facility siting. Social Science Quarterly 77(3):493–499. https://www.jstor.org/stable/42863495.
C-8 Medical Panel. 2013. C-8 Medical Panel Report. https://www.hpcbd.com/wp-content/uploads/sites/1601601/2021/01/Medical-Panel-Report-2013-05-24.pdf (accessed March 24, 2021).
Carnero, A. R., A. Lestido-Cardama, P. V. Loureiro, L. Barbosa-Pereira, A. R. B. de Quirós, and R. Sendón. 2021. Presence of perfluoroalkyl and polyfluoroalkyl substances (PFAS) in food contact materials (FCM) and its migration to food. Foods 10(7). https://doi.org/10.3390/foods10071443.
Curtzwiler, G. W., P. Silva, A. Hall, A. Ivey, and K. Vorst. 2021. Significance of perfluoroalkyl substances (PFAS) in food packaging. Integrated Environmental Assessment and Management 17(1):7–12. https://doi.org/10.1002/ieam.4346.
De Silva, A. O., J. M. Armitage, T. A. Bruton, C. Dassuncao, W. Heiger-Bernays, X. C. Hu, A. Kärrman, B. Kelly, C. Ng, A. Robuck, M. Sun, T. F. Webster, and E. M. Sunderland. 2021. PFAS exposure pathways for humans and wildlife: A synthesis of current knowledge and key gaps in understanding. Environmental Toxicology and Chemistry 40(3):631–657. https://doi.org/10.1002/etc.4935.
Death, C., C. Bell, D. Champness, C. Milne, S. Reichman, and T. Hagen. 2021. Per- and polyfluoroalkyl substances (PFAS) in livestock and game species: A review. Science of the Total Environment 774:144795. https://doi.org/10.1016/j.scitotenv.2020.144795.
DeLuca, N. M., M. Angrish, A. Wilkins, K. Thayer, and E. A. Cohen Hubal. 2021. Human exposure pathways to poly- and perfluoroalkyl substances (PFAS) from indoor media: A systematic review protocol. Environment International 146:106308. https://doi.org/10.1016/j.envint.2020.106308.
Domingo, J. L., and M. Nadal. 2017. Per- and polyfluoroalkyl substances (PFASs) in food and human dietary intake: A review of the recent scientific literature. Journal of Agricultural and Food Chemistry 65(3):533–543. https://doi.org/10.1021/acs.jafc.6b04683.
ECOS (Environmental Council of the States). 2020. Processes and considerations for setting state PFAS standards. https://www.ecos.org/wp-content/uploads/2020/02/Standards-White-Paper-FINAL-February-2020.pdf (accessed March 25, 2020).
EPA (U.S. Environmental Protection Agency). 2020. EPA CompTox chemicals dashboard. https://comptox.epa.gov/dashboard/chemical-lists/pfasmaster (accessed June 16, 2022).
Evich, M. G., M. J. B. Davis, J. P. McCord, B. Acrey, J. A. Awkerman, D. R. U. Knappe, A. B. Lindstrom, T. F. Speth, C. Tebes-Stevens, M. J. Strynar, Z. Wang, E. J. Weber, W. M. Henderson, and J. W. Washington. 2022. Per- and polyfluoroalkyl substances in the environment. Science 375(6580):eabg9065. https://doi.org/10.1126/science.abg9065.
Fenton, S. E., A. Ducatman, A. Boobis, J. C. DeWitt, C. Lau, C. Ng, J. S. Smith, and S. M. Roberts. 2021. Per- and polyfluoroalkyl substance toxicity and human health review: Current state of knowledge and strategies for informing future research. Environmental Toxicology and Chemistry 40(3):606–630. https://doi.org/10.1002/etc.4890.
Fraser, A. J., T. F. Webster, D. J. Watkins, M. J. Strynar, K. Kato, A. M. Calafat, V. M. Vieira, and M. D. McClean. 2013. Polyfluorinated compounds in dust from homes, offices, and vehicles as predictors of concentrations in office workers’ serum. Environment International 60:128–136. https://doi.org/10.1016/j.envint.2013.08.012.
Frieden, T. R. 2010. A framework for public health action: The health impact pyramid. American Journal of Public Health 100(4):590–595. https://doi.org/10.2105/AJPH.2009.185652.
Frisbee, S. J., A. P. Brooks, Jr., A. Maher, P. Flensborg, S. Arnold, T. Fletcher, K. Steenland, A. Shankar, S. S. Knox, C. Pollard, J. A. Halverson, V. M. Vieira, C. Jin, K. M. Leyden, and A. M. Ducatman. 2009. The C-8 health project: Design, methods, and participants. Environmental Health Perspectives 117(12):1873–1882. https://doi.org/10.1289/ehp.0800379.
Fromme, H., S. Dietrich, L. Fembacher, T. Lahrz, and W. Völkel. 2015. Neutral polyfluorinated compounds in indoor air in Germany—The LUPE 4 study. Chemosphere 139:572–578. https://doi.org/10.1016/j.chemosphere.2015.07.024.
Gao, K., T. Zhuang, X. Liu, J. Fu, J. Zhang, J. Fu, L. Wang, A. Zhang, Y. Liang, M. Song, and G. Jiang. 2019. Prenatal exposure to per- and polyfluoroalkyl substances (PFASs) and association between the placental transfer efficiencies and dissociation constant of serum proteins-PFAS complexes. Environmental Science & Technology 53(1):6529–6538. https://doi.org/10.1021/acs.est.9b00715.
Gilliland, F. D. 1992. Fluorocarbons and human health: Studies in an occupational cohort. Thesis. University of Minnesota. https://static.ewg.org/reports/2019/pfa-timeline/1992_1-46-Gilliland-Thesis.pdf (accessed July 1, 2022).
Glüge, J., M. Scheringer, I. T. Cousins, J. C. DeWitt, G. Goldenman, D. Herzke, R. Lohmann, C. A. Ng, X. Trier, and Z. Wang. 2020. An overview of the uses of per- and polyfluoroalkyl substances (PFAS). Environmental Science: Processes & Impacts 22(12):2345–2373.
Gochfeld, M., and J. Burger. 2011. Disproportionate exposures in environmental justice and other populations: The importance of outliers. American Journal of Public Health 101(Suppl 1):S53–S63. https://doi.org/10.2105/AJPH.2011.300121.
Gomez, J. A., D. Raynes, T. Doriss, L. Andersen, A. C. Capaccio, C. K. Gilbert, R. P. Johnson, and D. C. Royer. 2021. Man-made chemicals and potential health risks: EPA has completed some regulatory related actions for PFAS. Washington, DC: Government Accountability Office. https://www.gao.gov/assets/gao-21-37.pdf (accessed July 1, 2022).
Guelfo, J. L., S. Korzeniowski, M. A. Mills, J. Anderson, R. H. Anderson, J. A. Arblaster, J. M. Conder, I. T. Cousins, K. Dasu, and B. J. Henry. 2021. Environmental sources, chemistry, fate, and transport of per‐ and polyfluoroalkyl substances: State of the science, key knowledge gaps, and recommendations presented at the August 2019 SETAC Focus Topic Meeting. Environmental Toxicology and Chemistry 40(12):3234–3260.
Hahn, S. M. 2020. FDA announces voluntary agreement with manufacturers to phase out certain short-chain PFAS used in food packaging. Silver Spring, MD: U.S. Food and Drug Administration. https://www.fda.gov/news-events/press-announcements/fda-announces-voluntary-agreement-manufacturers-phase-out-certain-short-chain-pfas-used-food (accessed June 30, 2022).
Harclerode, M., S. Baryluk, H. Lanza, and J. Frangos. 2021. Preparing for effective, adaptive risk communication about per- and polyfluoroalkyl substances in drinking water. American Water Works Association Water Science 3(5):e1236. https://doi.org/10.1002/aws2.1236.
Harrad, S., C. A. de Wit, M. Abou-Elwafa Abdallah, C. Bergh, J. A. Bjorklund, A. Covaci, P. P. O. Darnerud, J. de Boer, M. Diamond, and S. Huber. 2010. Indoor contamination with hexabromocyclododecanes, polybrominated diphenyl ethers, and perfluoroalkyl compounds: An important exposure pathway for people? Environmental Science & Technology 44(9):3221–3231.
Herzke, D., S. Huber, L. Bervoets, W. D’Hollander, J. Hajslova, J. Pulkrabova, G. Brambilla, S. P. De Filippis, S. Klenow, G. Heinemeyer, and P. De Voogt. 2013. Perfluorinated alkylated substances in vegetables collected in four european countries: Occurrence and human exposure estimations. Environmental Science and Pollution Research 20:7930.
Houck, K. A., G. Patlewicz, A. M. Richard, A. J. Williams, M. A. Shobair, M. Smeltz, M. S. Clifton, B. Wetmore, A. Medvedev, and S. Makarov. 2021. Bioactivity profiling of per- and polyfluoroalkyl substances (PFAS) identifies potential toxicity pathways related to molecular structure. Toxicology 457:152789.
Hu, X. C., D. Q. Andrews, A. B. Lindstrom, T. A. Bruton, L. A. Schaider, P. Grandjean, R. Lohmann, C. C. Carignan, A. Blum, S. A. Balan, C. P. Higgins, and E. M. Sunderland. 2016. Detection of poly- and perfluoroalkyl substances (PFASs) in U.S. drinking water linked to industrial sites, military fire training areas, and wastewater treatment plants. Environmental Science and Technology Letters 3(10):344–350. https://doi.org/10.1021/acs.estlett.6b00260.
ITRC (Interstate Technology and Research Council). 2017. History and use of per- and polyfluoroalkyl substances (PFAS). https://pfas-1.itrcweb.org/wp-content/uploads/2017/11/pfas_fact_sheet_history_and_use__11_13_17.pdf (accessed March 25, 2020).
ITRC. 2021. Per- and polyfluoroalkyl substances technical and regulatory guidance. https://pfas1.itrcweb.org/wp-content/uploads/2022/03/PFAS-Full-PDF-December-2021-Update.pdf (accessed May 25, 2022).
Kay, J., and C. Katz. 2012. Pollution, poverty and people of color: Living with industry. Scientific American. http://www.scientificamerican.com/article/pollution-poverty-people-color-living-industry (accessed July 1, 2022).
Kwiatkowski, C. F., D. Q. Andrews, L. S. Birnbaum, T. A. Bruton, J. C. DeWitt, D. R. U. Knappe, M. V. Maffini, M. F. Miller, K. E. Pelch, A. Reade, A. Soehl, X. Trier, M. Venier, C. C. Wagner, Z. Wang, and A. Blum. 2020. Scientific basis for managing PFAS as a chemical class. Environmental Science & Technology Letters 7(8):532–543. https://doi.org/10.1021/acs.estlett.0c00255.
Li, P., X. Oyang, Y. Zhao, T. Tu, X. Tian, L. Li, Y. Zhao, J. Li, and Z. Xiao. 2019. Occurrence of perfluorinated compounds in agricultural environment, vegetables, and fruits in regions influenced by a fluorine-chemical industrial park in China. Chemosphere 225:659.
Lohmann, R., I. T. Cousins, J. C. DeWitt, J. Glüge, G. Goldenman, D. Herzke, A.B. Lindstrom, M. F. Miller, C. A. Ng, S. Patton, M. Scheringer, X. Trier, and Z. Wang. 2020. Are fluoropolymers really of low concern for human and environmental health and separate from other PFAS? Environmental Science and Technology 54(20):12820–12828. https://doi.org/10.1021/acs.est.0c03244.
Manzano-Salgado, C. B., M. Casas, M. J. Lopez-Espinosa, F. Ballester, M. Basterrechea, J. O. Grimalt, A. M. Jiménez, T. Kraus, T. Schettgen, J. Sunyer, and M. Vrijheid. 2015. Transfer of perfluoroalkyl substances from mother to fetus in a Spanish birth cohort. Environmental Research 142:471–478. https://doi.org/10.1016/j.envres.2015.07.020.
Mohai, P., D. Pellow, and J. Timmons Roberts. 2009. Environmental justice. Annual Review of Environment and Resources 34(1):405–430. https://doi.org/10.1146/annurev-environ-082508-094348.
Morales-McDevitt, M. E., J. Becanova, A. Blum, T. A. Bruton, S. Vojta, M. Woodward, and R. Lohmann. 2021. The air that we breathe: Neutral and volatile PFAS in indoor air. Environmental Science & Technology Letters 8(10):897–902. https://doi.org/10.1021/acs.estlett.1c00481.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2020. Understanding, controlling, and preventing exposure to PFAS: Proceedings of a workshop—in brief. Washington, DC: The National Academies Press. https://doi.org/10.17226/25856.
Nguyen, V. K., A. Kahana, J. Heidt, K. Polemi, J. Kvasnicka, O. Jolliet, and J. A. Colacino. 2020. A comprehensive analysis of racial disparities in chemical biomarker concentrations in United States women, 1999–2014. Environment International 137:105496. https://doi.org/10.1016/j.envint.2020.105496.
OECD (Organisation for Economic Co-operation and Development). 2021. Reconciling terminology of the universe of per- and polyfluoroalkyl substances: Recommendations and practical guidance. Series on Risk Management, no. 61. Paris: OECD. https://www.oecd.org/officialdocuments/publicdisplaydocumentpdf/?cote=ENV/CBC/MONO(2021)25&docLanguage=En.
Orum, P., R. Moore, M. Roberts, and J. Sanchez. 2014, May. Who’s in danger? Race, poverty, and chemical disasters. Brattleboro, VT: Environmental Justice and Health Alliance for Chemical Policy Reform. https://comingcleaninc.org/assets/media/images/Reports/Who%27s%20in%20Danger%20Report%20FINAL.pdf (accessed July 1, 2022).
Pellow, D. 2004. The politics of illegal dumping: An environmental justice framework. Qualitative Sociology 27(4):511–525. https://doi.org/10.1023/B:QUAS.0000049245.55208.4b.
Quimby, B., S. E. S. Crook, K. M. Miller, J. Ruiz, and D. Lopez-Carr. 2020. Identifying, defining and exploring angling as urban subsistence: Pier fishing in Santa Barbara, California. Marine Policy 121:104197. https://doi.org/10.1016/j.marpol.2020.104197.
Rankin, K., S. A. Mabury, T. M. Jenkins, and J. W. Washington. 2016. A North American and global survey of perfluoroalkyl substances in surface soils: Distribution patterns and mode of occurrence. Chemosphere 161:333. https://doi.org/10.1016/j.chemosphere.2016.06.109.
Raponi, I., P. Brown, and A. Cordner. 2021. Improved medical screening in PFAS-impacted communities to identify early disease. Environmental Health News. https://www.ehn.org/pfas-testing2653577444.html (accessed November 22, 2021).
Richter, L., A. Cordner, and P. Brown. 2021. Producing ignorance through regulatory structure: The case of per- and polyfluoroalkyl substances (PFAS). Sociological Perspectives 64(4):631–656.
Rizzuto, P. 2021. The doctor will see the PFAS-exposed plaintiff now. Bloomberg Law: Energy and Environment Report. https://news.bloomberglaw.com/environment-and-energy/the-doctor-will-see-the-pfas-exposed-plaintiff-now (accessed November 22, 2021).
Sagiv, S. K., S. L. Rifas-Shiman, T. F. Webster, A. M. Mora, M. H. Harris, A. M. Calafat, X. Ye, M. W. Gillman, and E. Oken. 2015. Sociodemographic and perinatal predictors of early pregnancy per- and polyfluoroalkyl substance (PFAS) concentrations. Environmental Science & Technology 49(19):11849–11858.
Schaider, L. A., S. A. Balan, A. Blum, D. Q. Andrews, M. J. Strynar, M. E. Dickinson, D. M. Lunderberg, J. R. Lang, and G. F. Peaslee. 2017. Fluorinated compounds in U.S. fast food packaging. Environmental Science & Technology Letters 4(3):105–111. https://doi.org/10.1021/acs.estlett.6b00435.
Scher, D. P., J. E. Kelly, C. A. Huset, K. M. Barry, R. W. Hoffbeck, V. L. Yingling, and R. B. Messing. 2018. Occurrence of perfluoroalkyl substances (PFAS) in garden produce at homes with a history of PFAS-contaminated drinking water. Chemosphere 196:548.
Serrano, L., L. M. Iribarne-Durán, B. Suárez, F. Artacho-Cordón, F. Vela-Soria, M. Peña-Caballero, J. A. Hurtado, N. Olea, M. F. Fernández, and C. Freire. 2021. Concentrations of perfluoroalkyl substances in donor breast milk in Southern Spain and their potential determinants. International Journal of Hygiene and Environmental Health 236:113796. https://doi.org/10.1016/j.ijheh.2021.113796.
Shoeib, M., P. Vlahos, T. Harner, A. Peters, M. Graustein, and J. Narayan. 2010. Survey of polyfluorinated chemicals (PFCs) in the atmosphere over the northeast Atlantic Ocean. Atmospheric Environment 44(24):2887–2893.
Solís, R., and SPWU (Southwest Public Workers Union). 1997, April. Jemez principles for democratic organizing. SouthWest Organizing Project. https://nation.mothersoutfront.org/jemez_principles_for_democratic_organizing (accessed July 1, 2022).
Steenland, K., T. Fletcher, C. R. Stein, S. M. Bartell, L. Darrow, M. J. Lopez-Espinosa, P. Barry Ryan, and D. A. Savitz. 2020. Review: Evolution of evidence on PFOA and health following the assessments of the C-8 Science Panel. Environment International 145. https://doi.org/10.1016/j.envint.2020.106125.
Sunderland, E. M., X. C. Hu, C. Dassuncao, A. K. Tokranov, C. C. Wagner, and J. G. Allen. 2019. A review of the pathways of human exposure to poly- and perfluoroalkyl substances (PFASs) and present understanding of health effects. Journal of Exposure Science & Environmental Epidemiology 29:131.
Sze, J., and J. K. London. 2008. Environmental justice at the crossroads. Sociology Compass 2(4):1331–1354.
Trudel, D., L. Horowitz, M. Wormuth, M. Scheringer, I. T. Cousins, and K. Hungerbühler. 2008. Estimating consumer exposure to PFOS and PFOA. Risk Analysis 28(2):251–269. https://doi.org/10.1111/j.1539-6924.2008.01017.x.
Washington, J. W., and T. M. Jenkins. 2015. Abiotic hydrolysis of fluorotelomer-based polymers as a source of perfluorocarboxylates at the global scale. Environmental Science & Technology 49(24):14129–14135. https://doi.org/10.1021/acs.est.5b03686.
Wones, R., S. M. Pinney, J. M. Buckholz, C. Deck-Tebbe, R. Freyberg, and A. Pesce. 2009. Medical monitoring: A beneficial remedy for residents living near an environmental hazard site. Journal of Occupational and Environmental Medicine/American College of Occupational and Environmental Medicine 51(12):1374.
Zheng, G., E. Schreder, J. C. Dempsey, N. Uding, V. Chu, G. Andres, S. Sathyanarayana, and A. Salamova. 2021. Per- and polyfluoroalkyl substances (PFAS) in breast milk: Concerning trends for current-use PFAS. Environmental Science & Technology 55(11):7510–7520.