7
Measuring and Improving System Performance
“The very first step is finding a hospital that will accept you and add you to its waiting list. My coverage determines which doctor or program I can even consult with.”
—Fanny Vlahos, double lung transplant recipient, Cystic Fibrosis Foundation, testimony to the committee during July 15, 2021 public listening session
Over the past 20 years, the U.S. health care system has undergone a significant transformation in defining, measuring, and reporting health care quality aimed at improving patient experiences and outcomes and decreasing health care costs. Measurement is a tool that can lead to better care by identifying opportunities for quality improvement, establishing transparency through public reporting, creating accountability for performance, and facilitating the identification and elimination of health disparities. Despite the exponential growth of health care quality measures over the past 2 decades, many parts of health care still face significant measurement challenges, thus limiting their ability to provide high-value and high-quality care.
In the case of the organ transplantation system, there is an impressive amount of data collected resulting in a body of measurement and public reporting. Compared to many other parts of American health care, the levels of measurement in the organ transplantation system are impressive. As one example, the Scientific Registry of Transplant Recipients (SRTR) website is an extraordinary national resource with extensive, publicly available data collected from transplant centers, organ procurement organizations (OPOs), and immunology laboratories on current and past organ donors, transplant candidates, transplant recipients, and transplant outcomes.1
___________________
1 Scientific Registry of Transplant Recipients website: www.srtr.org (accessed November 2, 2021).
OPTN MEASUREMENT CHALLENGES2
Despite its successes, the Organ Procurement and Transplantation Network (OPTN) still faces significant challenges regarding poor patient experience, overall access to available organs, long waiting list times, high rates of nonuse of procured organs, and high costs. Current data show racial, ethnic, gender, and geographical disparities in access, premature transplant failure and mortality, and reduced quality of life for living donors and recipients (Kulkarni et al., 2019; Puoti et al., 2016; Wesselman et al., 2021; Zhou et al., 2018). Adding a layer of complexity, the OPTN is a diverse ecosystem composed of various stakeholders (e.g., patients, physicians, policy makers, data intermediaries, and health systems) with varying objectives, levels of resources, and data infrastructure, all of which function across vastly different geographical areas. The differences in geography and resources across OPOs can result in varied quality of care and outcomes.
One systematic review found that out of the more than 300 transplant quality metrics reported in the literature, many were poorly defined, had inconsistent definitions, and focused primarily on safety and effectiveness with very few focusing on quality domains such as equity and patient-reported experience measures (Brett et al., 2018). As of 2018, existing transplant quality measures focus primarily on patient survival, transplant center volume, length of stay, and rehospitalization or unplanned return to the operating room. Other metrics include waiting list mortality, patient satisfaction, wait time for initial evaluation, in-hospital mortality, and infections (Brett et al., 2018). Out of all the organs, kidney transplantation has the most measures, and many of the metrics focus specifically on single organ transplant.
Needed Improvements in OPTN Measurement
To address the numerous challenges facing the OPTN and its stakeholders, the committee has identified six key measurement challenges needing action:
- Limited collection and use of disaggregated demographic data necessary to accurately track, understand, and eliminate disparities in procurement and transplantation;
- Limited to no standardization of a short list of key process and outcome measures for assessing basic performance, especially for OPOs;
- Gaps in standardized and publicly reported measurement related to patient referral, evaluation, and wait-listing at transplant centers;
- Overemphasis by regulators, payers, and others on using only one or two measures to assess performance of OPOs (donation and transplant rates) and transplant centers (1-year graft survival);
- Absence of established, consensus-based measurement development and endorsement processes such as those administered by the National Quality Forum; and
- Limited use of patient-centric measures that address what matters most to patients, such as the odds of getting transplanted before death at one center compared to others.
Addressing these six key areas will help to improve systemwide measurement in ways that will inform quality improvement at the organizational and system levels, assist federal regulators with more thoughtful and comprehensive oversight of OPOs and transplant cen-
___________________
2 Much of this section is excerpted or slightly modified from a paper commissioned by the committee for this study (Lantigua et al., 2021). Commissioned papers are in the study’s public access file and are available upon request from the National Academies’ Public Access Records Office (paro@nas.edu).
ters, better inform patient decision making and choices, and enable more robust understanding and interventions to address disparities, especially disparities in access to transplant.
Conclusion 7-1: Performance measures for donor hospitals, OPOs, and transplant centers need to be standardized and aligned to maximize donor referrals, evaluations, and organ procurements; optimize organ allocation; minimize organ nonuse rates and costs; and improve patient experiences and outcomes.
DASHBOARD OF STANDARDIZED, CONSENSUS-BASED PERFORMANCE MEASURES
Standardized performance measurement serves as a valuable method for identifying unwarranted variation in care, improving the quality and outcomes of health care services provided, and promoting interoperability and collaboration across organizations and systems. Additionally, standardized measures serve as a foundation for promoting transparency through public reporting and establishing accountability through pay-for-performance models. Standardized measures can also help to identify and eliminate health disparities by detecting differences in quality and access for specific populations or social groups.
Other areas of health care have successfully deployed integrated measurement dashboards to track progress, assess national performance, and guide improvement. One example is the Measure Applications Partnership (MAP) created in 2011 to fulfill a statutory requirement in which multistakeholder feedback is collected for the performance measures that the U.S. Department of Health and Human Services (HHS) is considering for public reporting and performance-based payment programs. The National Quality Forum created MAP as a pre-rulemaking process that allows room for dialogue, dissent, and consensus building to provide strategic input for federal quality programs, and specifically on the statutory measures under consideration by HHS.
Another integrated and comprehensive approach to national measurement is the Agency for Healthcare Research and Quality’s (AHRQ’s) National Scorecard on Hospital-Acquired Conditions. The AHRQ scorecard synthesizes information from multiple sources within the Centers for Medicare & Medicaid Services (CMS), the Centers for Disease Control and Prevention, and AHRQ to assess overall national progress in hospital patient safety. The scorecard focuses primarily on the 10 major forms of patient harm in hospitals (AHRQ, 2020). Other suites of measures used to track and assess performance include the CMS 5-Star Quality Rating System for Nursing Homes and the 5-Star Quality Rating System for Hospitals.
Dashboard of Standardized Performance Metrics for the U.S. Organ Transplantation System
As described earlier in this report, there is an opportunity to move away from the current focus on a single metric, such as 1-year-graft survival for transplant centers or donation rates for OPOs, and move toward an interrelated and integrated approach to measurement and performance. Emphasizing the standardization of metrics as a central element can help make the transplantation system become more equitable, transparent, and effective. Established, consensus-based processes facilitated by organizations such as the National Quality Forum can be used to develop measure frameworks that identify the largest needs; define policy gaps through the identification of disparities in care; support accreditation, regulation, and patient choice by identifying areas that are most meaningful to care; and reduce opportuni-
ties for manipulation of the system (Obadan-Udoh et al., 2019). For example, some physicians may escalate care to transplant candidates on the waiting list in order to increase their priority in the formalized allocation system (OPTN, 2018).
To help mitigate these measurement challenges and address the six areas for improvement outlined above, the committee recommends the creation of an integrated dashboard of standardized metrics built upon consensus-based measures to holistically track, assess, and guide the full spectrum of the organ transplantation system (see Recommendation 12). The committee believes a dashboard of standardized metrics will help move the organ transplantation system toward an integrated systems approach to measurement, improvement, operations, and results, thereby improving the quality and outcomes of health care services provided in the organ transplantation system.
The suggested standard dashboard does not need to be created from scratch and should be built upon existing extensive OPTN and SRTR measures to the maximum extent possible. Accurate and robust data are needed to have valid and reliable quality measures. Incomplete information and inadequate information exchange can create considerable challenges for care delivery, quality measurement, and health care payment. The committee’s recommendation calls for the creation of standardized performance measures that should be included in the dashboard, but the ultimate quality measures chosen need to be based on a consensus-driven process and not impose undue data collection or reporting burden on health professionals or patients. Data registries support efficient measurement with limited data collection burden. Studies have shown that the use of data registries, electronic health records, and health information exchanges help to regulate data collection, facilitate data exchange across multiple institutions, and allow for national benchmarking (Blumenthal, 2018; D’Amore et al., 2021). Data intermediaries—organizations that import data from disparate sources—calculate and report back quality measures that can also help lower the burden of measurement (Ahmad and Tsang, 2012).
In many cases, the standardized measures would replace existing measures, rather than increase the existing burden. For example, OPOs currently track important information like authorization rates. However, the current challenge is that there is no standardized approach to this tracking, which makes it difficult to compare performance, identify low and high performers, appropriately apply incentives and disincentives, guide improvement, or perform other work that requires standardized measurement. The committee’s recommended dashboard should also minimize measurement burden by drawing on existing work, such as the OPTN metrics dashboard and access to transplant dashboard.3
The OPTN Membership and Professional Standards Committee has proposed four new metrics for evaluating transplant center performance. Two pretransplant measures for transplant centers are the rate of pretransplant deaths and the ratio of organ offers made to and accepted for candidates, as well as two posttransplant metrics of 90-day graft survival and 1-year graft survival conditional to the 90-day period. Implementation of these new measures is expected to be staged, with conditional posttransplant graft survival effective June 2022, followed by offer acceptance by July 2023, and pretransplant (waiting list) mortality by July 2024. As discussed previously, the committee supports the idea of using a more holistic approach—more than a singular metric to evaluate transplant program performance—and applauds the rationale behind these proposed measures. At the same time, the committee believes a more robust set of performance measures, including measures related to access prior to wait-listing (i.e., number of patients referred to the center for evaluation and number
___________________
3 The OPTN metrics dashboard and Equity in Access dashboard are available at https://optn.transplant.hrsa.gov/data (accessed November 12, 2021).
of referred patients who were evaluated), need to be included in the dashboard of metrics in order to move the transplantation system toward a more equitable and transparent system. Figure 7-1 contains the committee’s suggestions for the elements of the proposed standardized performance metrics for transplant centers to be included in the dashboard.

Measurement Elements of the Recommended Dashboard of Standardized Performance Metrics
The dashboard of standardized metrics would include existing measures (i.e., number of listed patients who died waiting and number of total deaths), as well as several novel measures, including those that can close gaps related to disparities and equity—particularly related to access to the waiting list. Additionally, the standardized dashboard would permit the consideration and potential use of patient-centric measures, such as waiting time for transplant, chances of dying on the waiting list, and the number of organs declined on behalf of waiting patients.
The committee suggests measurements to be included in the dashboard of standardized metrics for referring organizations and transplant centers, and donor hospitals and OPOs (Figure 7-1).
Using the Dashboard of Standardized Metrics
The data included in the standard dashboard should be publicly available and up to date to remain transparent, accountable, and helpful to relevant organizations. The committee envisioned that the standardized performance metrics could be used first as quality improvement tools and, if successful, in quality improvement efforts, then the metrics can be used as standards of performance with regulatory implications.
Audiences and users of the dashboard include individual organizations (e.g., donor hospitals, OPOs, referring organizations, and transplant centers) to gauge their own overall performance, to identify variation within their own organization, and to target areas of improvement in quality and outcomes. Patients and families seeking to make informed choices about transplant access and care would also use the dashboard. Researchers as well as federal regulators who seek consensus measures that have been endorsed and affirmed by organizations like the National Quality Forum and the regulated community of practice would also use the dashboard. For patients, having standardized process and outcome metrics for the organ transplantation system would contribute mightily as a source of information that likely would be applied in ways other than as a dashboard, such as via applications that package key elements of the data and make it available either proactively, via nudging and other means, or reactively.
Importance of the Patient Voice in Measure Development and Use
In the past decade, the health care enterprise has shifted from patients being passive care recipients to empowered active participants in their care, with quality measurement following this critical shift. Collaboration between patients, caregivers, and health care providers can improve cost and health outcomes. To ensure that standardized measures matter to patients, patients and families need to be engaged in identifying where measures should be used, which measures are most meaningful to them, and where new measures are needed to assess important aspects of care. Patients can be engaged in the development and use of standardized performance measures through patient and family advisory councils, quality improvement teams, and on governing and leadership boards. Creating avenues for patients to inform the use of standardized performance measures can help ensure measures are meaningful and useful.
A Path Forward
Standardized performance measures, derived through a consensus-based process, can serve as a powerful tool in ensuring that the U.S. transplantation system is efficient, fair, equitable, and of high value. This can be accomplished by identifying how to best maximize donor referrals, evaluations, and the procurement and allocation of organs while minimizing nonuse rates and overall costs. The U.S. transplantation system currently faces challenges similar to other sectors of health care regarding disparities in access and outcomes, inefficiencies, and high costs. However, unlike many other areas of health care, the OPTN is well positioned to develop, select, and implement standardized performance measures to drive high-quality, high-value care. Specifically, the OPTN has the existing infrastructure to support standardized measurement, including robust data collection, clear identified partners, and available evidence-based guidelines. Existing partners already formulate and evaluate data for policy and organ allocation system performance metrics and can be engaged alongside other relevant stakeholders to identify process gaps and targets for measurement.
Standardized performance measures would facilitate a better understanding of the geographical- and patient-level challenges facing the organ transplantation system and enable the implementation of data-driven quality improvement to create lasting change. The development of a consensus-based measurement framework for the organ transplantation system can help create a transparent process that incorporates broad stakeholder input, maximizes trust in the associated processes, and encourages buy-in from the field. Additionally, a consensus-based framework can help to advance patient-centered care by centering the patient voice, identifying existing quality gaps and disparities, harmonizing existing and future measurement, and ultimately helping to improve the effectiveness and equity of the organ transplantation system.
Conclusion 7-2: The performance of the U.S. organ transplantation system, and its component parts, is highly variable and often inexplicable, with little understanding or justification for areas of variation that are acceptable or unacceptable. There is a need to reduce variations in the performance of donor hospitals, OPOs, and transplant centers in order to increase equity, efficiency, usefulness, reliability, predictability, and trustworthiness of the transplantation system. Creating standardized, consensus-based metrics to compare performance of donor hospitals, OPOs, and transplant centers needs to be a priority for HHS and the OPTN. Federal agencies overseeing the transplantation system will need to collaborate on data collection, even for the parts of the system they do not oversee, to ensure relevant, accurate, and timely data are available about the transplantation system.
ment, and once they are deemed valid and reliable, they should be used for regulatory purposes. Specific actions HHS should take include the following:
- Establish standardized data collection requirements, with an emphasis on timeliness of reporting, for donor hospitals, OPOs, and transplant centers. All data points collected should reflect demographics—that is, the most updated way of capturing race, ethnicity, and language, as well as socioeconomic factors, disability status, a social deprivation index based on geography, and other factors to better document, understand, reduce, and eventually eliminate disparities.
- Require collaboration among the federal agencies with oversight of the transplantation system on data collection to ensure relevant, accurate, and timely data are available about the transplantation system.
- Collaborate with an organization like the National Quality Forum to develop consensus measures and measure specifications to evaluate and improve the performance of the organ transplantation system in a standardized way. Recommended data points needed from donor hospitals, OPOs, referring organizations, and transplant centers are detailed in Figure 7-1.
- Create a publicly available dashboard of standardized metrics to provide a complete human-centered picture of the patient experience—from patient referral for transplant evaluation, time on the waiting list, to posttransplant quality of life—managed by the SRTR or a similar entity.
QUALITY IMPROVEMENT IN ORGAN DONATION AND TRANSPLANTATION
Over the last 30 years principles of quality improvement (QI) pioneered in manufacturing have been applied in health care to improve patient care and outcomes. While the application of the methods has been uneven across systems and regions, a great deal has been learned about principles of quality improvement and their value. Coupled with the QI movement in health care has been the study of how to take learning and improvement from one location, such as a hospital or clinic, to another setting. The success in improvements in patient safety in the United States in the 1990s and 2000s generated an interest in their application across the health system and social sector in subsequent years. Lessons from this application of improvement methods at a large scale, built on the foundation of improvement science and methods, has great potential to further improve the organ transplantation system in the United States.
In 2003, the organ transplantation system began work to employ these methods that were used to great success, initially to increase the rate of organ donation across the country (Shafer et al., 2008), and subsequently to improve organ recovery after circulatory death and to increase kidney transplants (OPTN, 2020; Tosoc-Haskell et al., 2019). While demonstrating a promising foundation upon which to build, tremendous variation and continued potential for improvement persist among donor hospitals, OPOs, referring organizations, and transplant centers.
Challenges and Opportunities in Quality Improvement
Typically, where systems struggle is not in the generation of ideas and successful practices but in effectively spreading those practices to all who could benefit from a broader
execution. QI methods can successfully facilitate the broad adaptation and adoption of successful practices through an emphasis on execution.
For people to change they must have the desire to change or demonstrate a will to change, as commonly referenced in QI literature (Nolan, 2007). How to encourage change and generate the will for change is therefore an important undertaking. The psychologist Jonathan Haidt uses the metaphor of a “divided mind” in which human behavior can be understood as a rider on top of an elephant. The rider, representing logical thought, acts as an advisor to the elephant it is riding. The elephant can be understood as human emotion, which in contrast to the rider is larger and seemingly has a disproportionate share of the say in which direction the rider will go (Haidt, 2006). While humans often know what they ought to do, this metaphor illustrates why rational thought and emotions do not always align to produce the desired or expected results. Quality improvement principles described by Deming underscore the importance of psychology and recognizing how and why humans are motivated to change. This understanding is critical to the success or failure of improvement initiatives.
In successful quality improvement, changes must be adopted over time across a desired population. Therefore, it is useful to understand demographically the different “personalities” as they pertain to receptiveness to change. In considering the diffusion of innovation, one must consider the types of people involved and their receptivity to an innovation (Dearing and Cox, 2018). These categories are helpful for change leaders to understand as they consider strategies to build the will for change within their organizations and across entire networks and systems. Different strategies will work for different people, and change leaders need to be prepared to understand the heterogeneity among their audiences and what will compel changes among different categories and individuals.
Improvements in the health care system will almost always require complex behavior change, and successful QI efforts must employ an intentional strategy for spreading changes. As appealing as the concept of natural diffusion sounds, complex behavior change requires active efforts to facilitate the adaptation or adoption of new ideas and practices. McCannon et al. (2007) elaborate that
Good ideas, even when their value is thoroughly demonstrated in one place, will not reliably spread into action through normal communication channels at a pace truly responsive to the enormous health care challenges.
This is also applicable to the U.S. organ transplantation system. Fortunately, strategies to support active dissemination and use of complex changes have been well documented in health care settings. The strategies combine principles from quality improvement, network management, social psychology, and logistics, and apply them to large-scale change efforts. These disciplines come together to form a method, or multitude of methods, by which change is facilitated.
Systematic Quality Improvement Options
Effective systems will have the infrastructure in place to identify what is working on an ongoing basis (“bright spotting”) and to couple that identification with the ability to test, adapt, and spread what works across the entire network (Sutton and Rao, 2014). This process requires support with the tools and resources to make change and track whether the change is leading to improvement. In executing successful changes at scale, three large-scale-change networked learning models may apply to the organ donation system: (1) collaboratives; (2) behavior change campaigns; and (3) mandates, policy, and regulation.
Collaboratives
A collaborative, also called a breakthrough series collaborative, is a results-oriented model designed to spread a change when there is a gap between current practice and desired results with an evidence-based intervention (or set of interventions) that are known to be effective in closing the gap. This method is frequently used when piloting or spreading practices and can be used with groups ranging from the tens to thousands. Collaboratives were developed by the Institute for Healthcare Improvement (IHI) and have been used nationally and internationally. Hallmarks of collaboratives are shared, time-bound aims across the network of participants, a core group of participants who take part in group activities and are responsible for taking on the role of change agents within their home organization, a shared set of standardized measures, and a shared set of interventions being adapted and spread across the network. There is a rhythm to the work wherein participating teams attend meetings that provide just-in-time subject matter and QI information, then apply the newly gained knowledge at their home organization(s) while measuring progress, and then attend a meeting that provides additional knowledge and support. This cycle continues multiple times within a year. Peer-to-peer sharing and transparency are emphasized so results can be known and understood, successes and failures can lead to learning, and progress can be spread and celebrated.
The Health Resources and Services Administration (HRSA) within the U.S. Department of Health and Human Services led the U.S. Organ Donation Breakthrough Collaborative from 2003 to 2007 which was not only successful in increasing the number of eligible organ donors across the country, but also spurred subsequent collaboratives for the next decade that together have yielded positive changes among donor services. The collaborative set a shared aim of increasing the organ donation rate in the country from 46 percent of all eligible donors to 75 percent among the nation’s 500 largest trauma centers. Using a shared set of changes and measures and the collaborative model described above, the organ donation rate increased by 10.8 percent in 2004 with continuing increases in subsequent years (IHI, 2003, 2021a). The collaborative helped to redesign the organ donation system (Shafer et al., 2006), and the effect of the collaborative on the number of monthly U.S. organ donors can be seen in Figure 7-2. In the month following the first learning session of the collaborative in September 2003, the number of U.S. organ donors increased compared to the same month of the prior year for 35 of the next 39 months. The collaborative also had a positive return on investment with an estimated 41,000 life-years gained and $2.5 billion saved in the years since the initiation of the collaborative, while the cost of running the collaborative was just $2.5 million (Schnitzler, 2021).
Subsequent collaboratives in the organ donation community have continued a steady trajectory of improvement within parts of the system. Numerous OPOs have become skilled systems improvers through the use of QI methods that form the foundation of collaborative improvement. Transplant centers have been involved in QI efforts to a lesser degree. However, building on the success of OPO improvement initiatives, the participation of transplant centers in similar initiatives could lead to comparable improvement in the U.S. organ transplantation system.
Notably, in 2020 CMS and HRSA launched a transplant learning collaborative, aimed at reducing the national nonuse rate of procured kidneys from 19 to 15 percent and increasing annual growth in the number of deceased donor kidneys transplanted from 5 to 15 percent (HRSA, 2020). The project aims to build on the best practices of high performers within the U.S. transplantation system and is informed by the work of the OPTN’s Collaborative Innovation and Improvement Network (Wey et al., 2020).
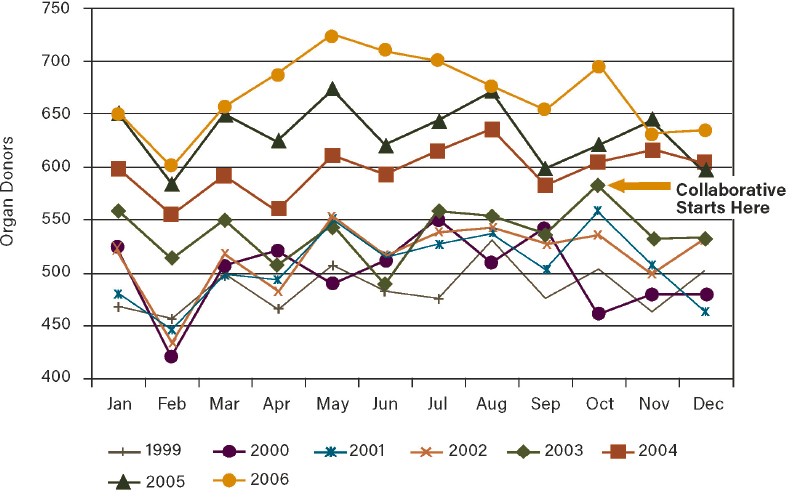
SOURCE: Reproduced with permission from Shafer et al., 2008. https://journals.lww.com/ccnq/Abstract/2008/07000/US_Organ_Donation_Breakthrough_Collaborative.2.aspx (accessed January 27, 2022).
Behavior Change Campaigns
Behavior change campaigns borrow principles of how to reach large audiences from political campaigns and apply them to behavior change. The approach was developed through the 100,000 Lives Campaign and the 5 Million Lives Campaign operated by IHI, and has now been applied successfully both within health care and the social sector (IHI, 2021b; McCannon et al., 2006). Additional successful examples include the 100,000 Homes Campaign, which led to 100,000 homeless people across the country finding permanent housing, and the Partnership for Patients, led by CMS and HHS (Bornstein, 2014; CMS, 2021c). Campaigns are appropriate when trying to reach large audiences with one or a set of simple, highly vetted interventions with well-understood application. For instance, the 100,000 Lives Campaign sought to prevent instances of harm in U.S. hospitals by spreading six evidence-based interventions such as one to prevent surgical site infections, in order to save 100,000 lives (McCannon et al., 2006). Campaigns are built around compelling aims and rely on local infrastructure such as existing networks or “field offices” to support application at a local level.
A campaign could be used effectively in the organ transplantation system if, for instance, high leverage and well-understood interventions from the Organ Donation Breakthrough Collaborative can be distilled with an aim to spread their use further or increase uptake of their use. Similarly, a campaign could take straightforward practices that result in successful organ transplant waiting list management in some regions and spread them to regions that
could benefit from improvement while using local infrastructure for the support and management of the change efforts. Because many OPOs have some QI skills already, they could be prime candidates to help with the local management of such efforts.
Mandates, Policy, and Regulation
Mandates, policies, and regulations are often the logical output of the approaches described above. Rather than being the starting point, they can be used effectively to leverage change once an intervention has been well characterized, spread to many locations, and the intricacies of its effect in numerous settings are well understood. All too often these approaches are used as a first line to stimulate change rather than using awareness-raising and will-building strategies. When this is done, there is a risk of unintended consequences because the intervention’s effect is not yet fully understood in certain situations and the nuance of spreading change is overlooked. When planned and timed well, mandates, policies, and regulations can have a significant effect.
Conclusion 7-3: The committee concludes widespread variation in practices and performance among donor hospitals, OPOs, referring organizations, and transplant centers shows there is a need to support and expand the use of proven systemwide quality improvement methods used in other areas of health care and that have successfully been used in the past to improve organ donation.
- increasing donation authorization rates among diverse populations, pursuit of all possible organ donors, how to have culturally sensitive conversations with all families about organ donation, intensive waiting list management, successful use of medically complex organs, and how best to communicate with patients about organ offers.
- Urge hospitals to smooth surgical scheduling to both enable organ donation surgical procedures, and to ensure the hospital’s capability to accept and use organ offers, regardless of which day of the week the gift of donation occurs.
- Explore additional tools and approaches for promoting innovation in the organ transplantation system, including the following:
- Launch a nationwide learning process improvement collaborative to address deceased organ donors, waiting list management, the acceptance of offered organs, transplant rate, and automated organ referrals.
- Encourage preapproved controlled experiments by OPOs and transplant centers to allow experimentation with innovation and the development of evidence to support widespread adoption of best practices.
- Incentivize transplant centers, donor hospitals and OPOs to actively participate in the kidney transplantation collaborative sponsored by CMS and HRSA.
- Require the OPTN to implement an organized system of proactive communication or nudges in the form of special messages or brief reports aimed at calling attention to outlier performance by OPOs and transplant centers, based on SRTR data. Nudges should be sent to both high and low performers. For example, OPOs with a low percentage of donation after circulatory determination of death (DCDD) donors in their deceased donor organ pool could receive a special message or brief report calling attention to their current performance in comparison to other OPOs.
ALIGNING INCENTIVES TO MAXIMIZE ORGAN USE
The committee was tasked with making recommendations to better align the performance metrics or incentives of various stakeholders within the OPTN, specifically donor service areas, OPOs, and transplant centers, to maximize donor referrals, evaluations, procurement, and organ placement and allocation while minimizing organ nonuse rates (see Chapter 1 for the full Statement of Task).
Building on the previous report chapters and the committee’s focus areas of equity, system performance, and maximizing the use of procured organs, the remainder of Chapter 7 includes the committee’s analysis of the evidence around financial incentives and the identification of opportunities to improve alignment of incentives in the organ transplantation system. In addition to the stakeholders referenced in the study task, the committee considers dialysis centers, donor hospitals, and physicians (primary care and specialists such as nephrologists) responsible for referring transplant candidates for evaluation as core participants in the transplantation system. Therefore, the committee considered financial incentives related to these stakeholders as well. As described earlier in the report, the organ transplantation system is complex and the component parts of the system—including payers such as CMS, the Veterans Health Administration, and private payers—do not currently operate as a fully integrated system accountable for achieving predictable, consistent, and equitable results.
Conclusion 7-4: The authority of federal payers and the power of private insurers to set eligibility requirements and payment rates in the organ transplantation system
greatly influences who obtains a transplant and when that occurs. Donors, families, hospitals, OPOs, transplant centers, and patients awaiting a transplant depend on these public and private payers to act in ways that will promote rather than retard steps, such as those recommended in this report, aimed at increasing the number and the equitable distribution of organ transplants.
Select CMS Initiatives in Organ Transplantation Reimbursements
CMS provides federal leadership on reimbursement policies for Medicare-eligible patients in need of transplantation, and in particular individuals needing kidney transplants. Since 1972, Medicare benefits have been provided to individuals with end-stage kidney (renal) disease regardless of age and without a waiting period to enroll in Medicare. The benefits support dialysis or kidney transplant. As discussed in previous chapters, CMS also sets conditions for coverage for OPOs and conditions of participation for transplant programs and donor hospitals—setting requirements that must be met for OPOs, transplant programs, and donor hospitals to receive Medicare and Medicaid payment.
CMS requests that dialysis centers inform new patients of their transplant options, which can potentially lead to referral for transplant evaluation before dialysis even begins (Waterman et al., 2015). Strategies are needed to reduce existing disparities in referral for transplant evaluation, however. In the United States, black patients are less likely to be put on a waiting list for transplant before beginning dialysis than white patients, but early, predialysis discussions about transplantation can improve waiting list placement among black patients (Kutner et al., 2012). Transplant education and engagement activities for dialysis facility staff and patients have also been associated with increased kidney transplant evaluation referrals and improved equity in the referral process in a randomized controlled trial among patients receiving dialysis at facilities in Georgia (Patzer et al., 2017).
The CMS Innovation Center has had an active role in testing different funding models for treatment options for end-stage kidney disease. For example, three relevant activities in the past 5 years include the following:
- The End-Stage Renal Disease (ESRD) Treatment Choices Model began in January 2021 and encourages dialysis facilities and health care providers, through payment adjustments, to decrease disparities in the rates of home dialysis and kidney transplantation among end-stage kidney disease patients with lower socioeconomic status. The test model is mandatory for the dialysis facilities randomly selected by CMS to participate and accounts for 30 percent of all end-stage kidney disease facilities and managing clinicians (Medicare-enrolled physicians and nonphysicians that care for one or more end-stage kidney disease beneficiaries) in the United States (CMS, 2021a).
- The Kidney Care Choices model began in January 2022 and focuses on financial incentives for health care providers—dialysis facilities, nephrologists, and other health care providers—to manage the care for Medicare beneficiaries with chronic kidney disease (stages 4 and 5) and end-stage kidney disease (CMS, 2021b). The goal is to delay the onset of dialysis and incentivize kidney transplantation. One of the payment incentives in the model is a $15,000 bonus payment for Medicare patients that receive a kidney transplant and do not return to dialysis. The payment is made to nephrologists who partner with transplant providers, and possibly dialysis facilities and other providers, to incentivize supporting Medicare patients through the transplant process, including continued care management posttransplant. The $15,000
- payment is disbursed in smaller amounts for each year posttransplant, and the payments must be shared with partners (e.g., transplant teams, OPOs) (CMS, 2019a).
- A learning collaborative through the CMS ESRD Treatment Choices Model was established in 2020 to operate as a voluntary learning system focused on reducing the kidney nonuse rate, and increasing the availability of deceased donor kidneys for transplantation. The learning system includes model participants and other stakeholders such as transplant centers, OPOs, and large donor hospitals, and uses learning and QI techniques to systematically spread the best practices of the highest performers (CMS, 2019b).
Donor Hospital Conditions of Participation
The cooperation and involvement of donor hospitals and medical personnel is an essential function of the organ transplantation system. Shortly after the enactment of the National Organ Transplant Act, Prottas (1998) described the relationship thus,
the primary factor affecting the supply of transplantable organs is the cooperation of health professionals. OPOs must obtain timely access to potential donors who are mainly identified and contacted by the nurses and neurophysicians in ICUs [intensive care units]. The discretion of medical personnel is limited to their judgment about medical suitability for donation while required request laws ensure that the family is offered the option of organ donation.
Over the past 30 years, there have been numerous iterations of state and federal statutes and regulations that have addressed this fundamental relationship between donor hospitals and OPOs. Two effective regulatory and statutory requirements for increasing organ donation have been (1) the timely referral of all individuals whose death is imminent or who died in the hospital, and (2) first person authorization.
The committee received public comments regarding the outdated nature of CMS Interpretive Guidelines (IGs) for hospital conditions of participation, which have been in place since 2008. There is an opportunity to improve alignment in hospital regulations related to organ donation. Current IGs lack sufficient detail and direction in addressing hospital practices related to the timely referral of donors, the ability to pronounce death timely, authorization by first person or legal next of kin, DCDD as a pathway to donation, or physiologically maintaining donors until the OPO can test and allocate organs. The IGs are also silent in requiring the measurement of hospital performance against the conditions of participation.
Updated CMS guidance in the Interpretive Guidelines would more effectively integrate hospital and OPO systemness, fairness, equity, and the transparency necessary for improvements in organ donation, and ultimately transplantation. The following elements could be considered in updating CMS guidance and survey procedures to enhance clarity for hospitals and OPO performance:
- Require hospitals to create electronic or automated referral processes with the OPO to ensure timely referrals of potential donors, and provide patient-level information necessary to determine donor potential. Ideally, this information would also be reported electronically to CMS as part of the calculations used to assess the number of eligible donors.
- Require honoring first person authorization, and establish processes for OPO and hospital responsibilities when there is initial opposition to the donor registry.
- Address several processes essential to DCDD donation: require hospitals to participate in DCDD donation; provide guidance addressing DCDD referral triggers, which may be dif
- ferent than for donation after neurological determination of death (DNDD); and require hospital support in the declaration of death, whether in the ICU, emergency department, operating room, or elsewhere in the hospital.
- Strengthen the requirements that hospitals maintain potential donors while necessary testing and placement of organs takes place, including the declaration of DNDD or DCDD.
- Require trained OPO staff to approach donor families, eliminating the provision for hospital-designated requestors. Hospital staff would serve in a supportive capacity in all family conversations related to donation.
- Update areas where ventilator support might be used, and donors supported, beyond the ICU or emergency department.
- Include in the CMS survey procedures the addition of a policy or process map showing integration of the OPO into the hospital quality assurance and performance improvement process, including an interview with OPO staff to verify.
- Survey hospital compliance against top-line measures of 100 percent timely referral of all deaths, and determine compliance with measurable elements of hospital agreements with OPOs.
- Hold hospitals accountable for smoothing surgical scheduling to ensure the capacity to recover and transplant donated organs 7 days a week.
Conclusion 7-5: The CMS Interpretive Guidelines and survey processes for donor hospitals need updating to reflect changes that have taken place in medical practice and processes related to the relationship between donor hospitals and OPOs in order to maximize every donation opportunity.
Opportunities to Align Reimbursement Incentives
In considering opportunities to improve alignment of reimbursement incentives for stakeholders in the organ transplantation system, the committee explored the following areas:
- Referrals for transplant evaluation;
- Reimbursement to transplant centers for transplanting medically complex organs and caring for patients who receive these transplants;
- Intensive waiting list management;
- Enhanced communication and education with patients on the waiting list;
- Medical care and support for waiting list patients, including those with complex needs; and
- Innovative technology to maximize organ use.
The remainder of this chapter discusses these opportunities for improved alignment, reflects on differing viewpoints within the committee regarding the appropriate role of financial incentives in changing behaviors, and ends with the committee’s recommendations for aligning reimbursement and programs with desired outcomes and behaviors (Recommendation 14).
Referrals for Transplant Evaluation
For patients with advanced chronic kidney disease and end-stage kidney disease, kidney transplantation is the treatment of choice. As described in Chapter 2, transplantation
is superior to dialysis in improving both patient survival and quality of life (OPTN, 2015). Additionally, kidney transplantation is also the most cost-effective treatment for end-stage kidney disease, although the extent of cost savings vary by quality of the donor organ. Clinical practice guidelines indicate all patients with advanced chronic kidney disease (stages 4 and 5) and end-stage kidney disease need to be referred for transplant evaluation, as should certain patients with a higher level of kidney function. Among the benefits of early referral are the opportunity to provide more comprehensive patient education about transplant options and potentially intervene earlier in the course of disease. To improve the quality of care for patients with kidney disease, strategies are needed to incentivize nephrologists and dialysis centers to educate and refer patients for transplant evaluation.
Reimbursement for Medically Complex Cases
As stated previously, compared to dialysis, kidney transplantation increases overall survival rates and improves quality of life for patients with end-stage kidney disease, in addition to being cost effective. An economic assessment of kidney transplant practice found that compared to dialysis, all transplantation types were associated with improved survival and were cost effective regardless of donor type, despite the higher costs associated with the use of medically complex organs and living donors. Compared to dialysis, the authors found cost savings with living donor and low–Kidney Donor Profile Index (KDPI) deceased donor transplantations, while transplantations using high-KDPI deceased donors, ABO-incompatible living donors, or human leukocyte antigen-incompatible living donors were found to be cost effective (less than $100,000 per quality-adjusted life year) (Axelrod et al., 2018).
In the inpatient prospective payment system Final Rule for 2021, CMS recognized that increasing the range of donated organs used in transplantation to include more medically complex organs can result in some patients requiring more medical support. For example, the two new Diagnosis Related Group (DRG) codes created for kidney transplant patients who require dialysis posttransplant and for such patients with major complications and comorbidities are, respectively, 15 and 40 percent higher than the DRG payment for uncomplicated kidney transplants. Likewise, the new DRG for simultaneous kidney and pancreas transplants where the patient needs dialysis is 23 percent higher than what is paid when that form of posttransplant care is not required.
The Donor Risk Index (DRI), a measurement of donor organ quality, was developed to quantitatively assess risk of graft failure following liver transplant (Feng et al., 2006). Increasing DRI is correlated with risk of organ failure. DRI is also an independent predictor of the perioperative and postoperative costs of liver transplant. A retrospective database analysis of the interactions between donor characteristics, recipient liver disease severity, and total inpatient costs of liver transplantation found that the donors in the highest DRI quartile added nearly $12,000 to the cost of transplantation and nearly $22,000 to posttransplantation cost compared to the lowest-risk donors (Salvalaggio et al., 2011). The confluence of these factors may be particularly effective on transplant centers in competitive regions where liver transplant patients with higher Model for End-Stage Liver Disease scores receive organs with high DRI (Salvalaggio et al., 2011).
Intensive Waiting List Management
Optimizing transplant waiting list management is critical for maintaining candidacy of kidney transplant recipients and effectively managing the waiting list as organs become available. A novel kidney transplant waiting list management strategy called Transplant Readiness
Assessment Clinic (TRAC), was developed to support active management for patients whose Kidney Allocation Scores exceeded a certain threshold (Cheng et al., 2018). The use of TRAC resulted in a higher proportion of patients being actively managed at 18 months. An aggressive waiting list management protocol was designed and tested in two hospital settings, finding that the costs associated with this aggressive protocol were offset by the economic benefits of increased transplantation and reduced time lost in the allocation of deceased donor organs (Grafals et al., 2014). Improving waiting list management requires more clarity about waiting list inactivity prior to kidney transplantation, which is known to affect clinical outcomes. For instance, a study of adult kidney transplant patients in the United States found that inactive periods on the waiting list were predictive of increased pre- and posttransplantation mortality (Norman et al., 2013).
Enhanced Communication and Education with Patients on the Waiting List
The provision of kidney transplant education is associated with higher rates of transplant (Balhara et al., 2012). Patients who initiate the process of transplant evaluation with greater transplant knowledge and motivation tend to be more likely to receive successful transplants years later (Rodrigue et al., 2014). Educational materials about transplantation—including information about waiting list practices and organ offers and declines—need to be presented to waiting list patients in a broadly accessible manner using clear communication strategies.
Transplant education is of particular importance within the dialysis center context, as 70 percent of ESRD patients are on dialysis (Waterman et al., 2020). Dialysis centers that have higher referral rates and transplant wait-listing rates tend to use multiple patient education strategies (Waterman et al., 2015). Education strategies that have been associated with increased transplant waiting list rates include distribution of print education and use of more than one intensive education practice within dialysis centers (Waterman et al., 2020). According to a survey of nephrologists, most respondents (81 percent) reported that patients at dialysis centers should receive at least 20 minutes of education; however, less than half reported actually spending more than 20 minutes educating individual patients and their families (Balhara et al., 2012).
It is critical to identify and mitigate ways that disparities in the provision of transplant education contribute to disparities in access to transplantation. An evaluation of the association between transplant education practices and access to transplantation analyzed United States Renal Data System data from 2005 to 2007 (Kucirka et al., 2012). Patients who were older, obese, uninsured, Medicaid insured, and patients at for-profit centers were less likely to be assessed at all; women were more likely to be deemed medically unfit; and black patients were more likely to be assessed as psychologically unfit. Moreover, the readability of educational materials can impede access to transplant among patients with lower levels of health literacy if the materials are not easy to understand (Bababekov et al., 2017).
Medical Care and Support for Waiting List Patients, Including Those with Complex Needs
Many transplant candidates have multiple physical and mental health comorbidities that can affect their clinical outcomes and quality of life while they are wait-listed. For example, conditions such as diabetes, hypertension, and obesity can contribute to the development of ESRD—which itself is a risk factor for comorbidities such as cardiovascular disease—and the number of comorbidities among kidney transplant recipients has increased substantially in recent years (Wu et al., 2020). A prospective cohort study that analyzed the relationship
between donor kidney recipient comorbidity and survival outcomes in the United Kingdom found that heart failure and diabetes were associated with poorer transplant survival among living donor kidney transplant recipients (Wu et al., 2020). Comorbidity assessment is of particular importance among patients with ESRD being evaluated for kidney transplantation, because comorbidities can accelerate the progression of ESRD and reduce the chance of survival (Fernández et al., 2019). A study evaluating the association between comorbidity and waiting list mortality among frail and nonfrail kidney transplantation candidates found that nonfrail candidates with high comorbidity burdens at kidney transplant evaluation have increased risk of waiting list mortality (Fernández et al., 2019). New care modalities such as remote video monitoring may contribute to improved patient safety and satisfaction, as has been demonstrated in lung-transplant candidates with multiple comorbidities (Zubrinic et al., 2019).
Many organ transplant recipients experience mental health conditions both pre- and posttransplant. For example, depression is thought to affect up to 60 percent of solid organ transplant recipients (Corbett et al., 2013). Mental health comorbidities can affect these patients’ clinical outcomes and undermine their quality of life; however, when properly supported, transplant patients with mental health conditions have outcomes similar to the general transplant population (Corbett et al., 2013). Therefore, potential transplant candidates should be assessed for mental health issues, as well as preexisting medical conditions that mimic mental health problems, so that pharmacological treatment and other interventions can be offered to patients while they are on the waiting list (Corbett et al., 2013; Crawford et al., 2013). In solid organ transplant patients, feeling a sense of control has been associated with positive health outcomes (Crawford et al., 2013).
A randomized trial investigated the effects of two health educational models on the psychology and nutrition of patients waiting for deceased donor kidney transplantation (Ye et al., 2011). The observational group had lower self-rated anxiety and depression after the intervention as well as higher triceps skinfold thickness, hemoglobin, and albumin. These findings suggest that comprehensive health education methods can contribute to alleviating mental stress, improving nutrition, and contributing to better quality of life for patients awaiting kidney transplantation.
Pretransplant psychosocial vulnerability factors are associated with negative outcomes for transplant patients, including higher infection rates, reduced treatment adherence, increased rejection episodes, acute late rejection, hospital readmission, increased cost of care, posttransplant malignancies, graft loss, and decreased transplant survival (Maldonado, 2019). A cross-sectional survey on the concerns and anxiety of patients awaiting organ transplant was conducted in Hong Kong, revealing the importance of providing sufficient psychosocial support to those awaiting organ transplantation (Li et al., 2012). To better support patients, a psychosocial pretransplant evaluation could help to identify and provide care for mental health issues the patient may have, as well as helping to strengthen the patient’s support system (Maldonado, 2019).
A patient’s social support is often used as a factor for determining transplant eligibility in the United States, despite limited supporting evidence, the subjectivity and variability of its assessment, vague regulatory guidance, and low provider confidence (Berry et al., 2019; Ladin et al., 2019). According to a survey that measured the use of social support to determine transplant eligibility, respondent transplant providers estimated that almost 10 percent of patients evaluated during the survey year were disqualified based on the inadequacy of their social support (Ladin et al., 2019). This suggests the need to reevaluate social support criteria and examine its potential contribution to inequities and disparities, including biased wait-listing processes (Berry et al., 2019; Ladin et al., 2019).
Innovative Technology to Maximize Organ Use
Recent innovations in methods for preserving organs prior to transplant have the potential to substantially expand the volume of the donor organ pool, mitigate organ injury, improve the quality of donated organs, and contribute to maximizing the use of solid organs (Petrenko et al., 2019; Salehi et al., 2018; Tatum et al., 2020). For instance, advances in organ perfusion technology have led to increases in the transplantation of medically complex donor organs (Chew et al., 2019). In the case of transplants using hearts donated after cardiac death, ex vivo blood-based perfusion technology is increasingly being introduced into clinical practice (Saemann et al., 2020). This technology has allowed cardiac allografts to be procured from greater distances than would be possible using cold static storage; the process also provides nutrients that reduce the risk of ischemic injury for functional preservation (Chew et al., 2019). In the field of liver and kidney transplantation, a clinical trial of ex vivo hypothermic oxygenated perfusion was conducted using organs from medically complex donors after brain death (Ravaioli et al., 2020). The clinical outcomes suggest that machine perfusion is a safe and effective system to reduce ischemic preservation injuries in kidney and liver transplants. A meta-analysis found hypothermic machine perfusion to be superior to static cold storage in deceased donor kidney transplantation in terms of reducing the risk of delayed graft function; it was also cost-saving at 1 year compared to cold storage (Tingle et al., 2020).
Committee Considerations
In considering the alignment of incentives in the organ transplantation system, including the appropriate role of financial incentives, members of the committee held differing views. Some believe that transplant centers face higher costs if they provide wait-listed patients intensive management and take on especially complicated cases (e.g., using more medically complex organs, treating them to improve their usefulness, and transplanting patients with extensive medical and social needs), and they will therefore be disinclined to follow this report’s recommendation that they take on more such cases unless they are reimbursed at an appropriately higher rate. Other members pointed out that increased payment should not be necessary since some high-performing transplant centers are already able to excel in meticulously managing their waiting list and in handling a large number of complicated cases within the current regulatory and payment structures. For example, if one transplant center can accept and successfully transplant a high number of medically complex organs under current rules and payment structures, then other transplant centers should be able to reach a similarly high level of performance without increased payment. Under this reasoning, quality improvement efforts and the sharing of best practices across transplant centers, rather than increased payments, are the right way to increase the number of organs used and the diversity of patients successfully transplanted. In the end, the committee agreed on the following conclusions and recommendation:
Conclusion 7-6: This report presents a number of conclusions and recommendations that aim to provide more transplants more fairly, such as by making better use of donated organs; by increasing the number of organs obtained and the proportion treated to improve their suitability for transplantation; by broadening the pool of patients who are referred, evaluated, and listed for a transplant; and by better management of patients on the waiting list. Implementing the committee’s recom
mendations will require changes in policies and practices, and some—particularly those recommendations that entail using more medically complex organs or treating organs more extensively to improve their usefulness for transplantation—are likely to have financial costs.
Conclusion 7-7: OPOs or transplant centers are less likely to implement recommended changes in policies and practices—such as using more medically complex organs or transplanting patients with more serious comorbidities—when they involve added financial costs that are not specifically covered.
Conclusion 7-8: When considering any reductions in the formulas used to pay transplant centers and OPOs of the sort proposed, but not adopted, in the CMS FY 2021 IPPS rule, CMS could use the Innovation Center’s testing authority to fully explore the potential effect of such changes to avoid unintentionally interfering with the implementation of policies, such as those recommended by the committee, that aim to alleviate problems with the fairness, efficiency, and transparency of the organ transplantation system.
REFERENCES
Ahmad, F., and T. Tsang. 2012. The melody of quality measures: Harmonize and standardize. Health Affairs Blog. https://www.healthaffairs.org/do/10.1377/hblog20120221.017126/full (accessed November 2, 2021).
AHRQ (Agency for Healthcare Research and Quality). 2020. AHRQ National scorecard on hospital-acquired conditions. https://www.ahrq.gov/hai/pfp/index.html (accessed November 2, 2021).
Axelrod, D. A., M. A. Schnitzler, H. Xiao, W. Irish, E. Tuttle-Newhall, S.-H. Chang, B. L. Kasske, T. Alhamad, and K. L. Lentine. 2018. An economic assessment of contemporary kidney transplant practice. American Journal of Transplantation 18(5):1168-1176.
Bababekov, Y. J., B. Cao, F. C. Njoku, Y.-C, Hung, J. T. Adler, J. J. Pomposelli, C. D. Chang, and H. Yeh. 2017. Readability of patient education websites of liver transplant centers—A barrier to the waitlist? Journal of the American College of Surgeons 225(4):e78.
Balhara, K. S., L. M. Kucirka, B. G. Jaar, and D. L. Segev. 2012. Disparities in provision of transplant education by profit status of the dialysis center. American Journal of Transplantation 12(11):3104-3110.
Berry, K. N., N. Daniels, and K. Ladin. 2019. Should lack of social support prevent access to organ transplantation? American Journal of Bioethics 19(11):13-24.
Blumenthal, S. 2018. Improving interoperability between registries and EHRs. American Medical Informatics Association Joint Summits in Translational Science Proceedings 2018:20-25. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5961768 (accessed February 1, 2022).
Bornstein, D. 2014. The push to end chronic homelessness is working. New York Times. https://opinionator.blogs.nytimes.com/2014/05/28/the-push-to-end-chronic-homelessness-is-working/ (accessed November 2, 2021).
Brett, K. E., L. J. Ritchie, E. Ertel, A. Bennett, and G. A. Knoll. 2018. Quality metrics in solid organ transplantation: A systematic review. Transplantation 102(7):e308.
Cheng, X. S., S. Busque, J. Lee, K. Discipulo, C. Hartley, Z. Tulu, J. D. Scandling, and J. C. Tan. 2018. A new approach to kidney wait-list management in the kidney allocation system era: Pilot implementation and evaluation. Clinical Transplantation 32(11):e13406.
Chew, H. C., P. S. Macdonald, and K. K. Dhital. 2019. The donor heart and organ perfusion technology. Journal for Thoracic Disease 11(6):S938-S945.
CMS (Centers for Medicare & Medicaid Services). 2019a. Kidney Care Choices (KCC) Model: Request for Applications (RFA). https://innovation.cms.gov/files/x/kcc-rfa.pdf (accessed November 10, 2021).
CMS. 2019b. Medicare programs; Specialty care models to improve quality of care and reduce expenditures. https://www.federalregister.gov/documents/2020/09/29/2020-20907/medicare-program-specialty-care-models-to-improve-quality-of-care-and-reduce-expenditures#p-2346 (accessed November 11, 2021).
CMS. 2021a. ESRD Treatment Choices (ETC) Model. https://innovation.cms.gov/innovation-models/esrd-treatment-choices-model (accessed November 10, 2021).
CMS. 2021b. Kidney Care Choices (KCC) Model. https://innovation.cms.gov/innovation-models/kidney-care-choices-kcc-model (accessed November 10, 2021).
CMS. 2021c. Innovation models: Partnership for patients. https://innovation.cms.gov/innovation-models/partnership-for-patients (accessed November 2, 2021).
Corbett, C., M. J. Armstrong, R. Parker, K. Webb, and J. M. Newberger. 2013. Mental health disorders and solid-organ transplant recipients. Transplantation 96(7):593-600.
Crawford, I., T. Hogan, and M. J. Silverman. 2013. Effects of music therapy on perception of stress, relaxation, mood, and side effects in patients on a solid organ transplant unit: A randomized effectiveness study. The Arts in Psychotherapy 40(2):224-229.
D’Amore, J. D., L. K. McCrary, J. Denson, C. Li, C. J. Vitale, P. Tokachichu, D. F. Sittig, A. B. McCoy, and A. Wright. 2021. Clinical data sharing improves quality measurement and patient safety. Journal of the American Medical Informatics Association 28(7):1534-1542.
Dearing, J. W., and J. G. Cox. 2018. Diffusion of innovations theory, principles, and practice. Health Affairs 37(2):183-190.
Feng, S., N. P. Goodrich, J. L. Bragg-Gresham, D. M. Dykstra, J. D. Punch, M. A. Debroy, S. M. Greenstein, and R. M. Merion. 2006. Characteristics associated with liver graft failure: The concept of a donor risk index. American Journal of Transplantation 6(4):783-790.
Fernández, M. P., P. M. Miguel, H. Ying, C. E. Haugen, N. M. Chu, D. M. Rodriguez-Puyol, L. Rodriguez-Manas, S. P. Norman, J. D. Watson, D. L. Segev, and M. A. McAdams-DeMarco. 2019. Comorbidity, frailty, and waitlist mortality among kidney transplant candidates of all ages. American Journal of Nephrology 49(2):103-110.
Grafals, M., M. Rogers, P. Weems, J. Moore, J. Verbesey, A. Gilbert, E. Gonzales, R. Ghasemian. D. Zwerski, T. Fishbein, and M. Cooper. 2014. Kidney transplant wait list management optimization: Abstract A450. Transplantation 98:810.
Haidt, J. 2006. The happiness hypothesis: Finding modern truth in ancient wisdom. New York: Basic Books.
HRSA (Health Resources and Services Administration). 2020. Advisory Committee on Organ Transplantation (ACOT): April 2020 meeting minutes. https://www.hrsa.gov/sites/default/files/hrsa/advisory-committees/organ-transplantation/acot-april-2020-meeting-minutes.pdf (accessed February 1, 2022).
IHI (Institute for Healthcare Improvement). 2003. The breakthrough series: IHI’s collaborative model for achieving breakthrough improvement. Boston, MA: Institute for Healthcare Improvement. http://www.ihi.org/resources/Pages/IHIWhitePapers/TheBreakthroughSeriesIHIsCollaborativeModelforAchievingBreakthroughImprovement.aspx (accessed November 2, 2021).
IHI. 2021a. Improvement stories: Organ donation breakthrough collaborative. http://www.ihi.org/resources/Pages/ImprovementStories/OrganDonationBreakthroughCollaborative.aspx (accessed November 2, 2021).
IHI. 2021b. Overview: 5 million lives campaign. http://www.ihi.org/Engage/Initiatives/Completed/5MillionLivesCampaign/Pages/default.aspx (accessed November 2, 2021).
Kucirka, L. M., M. E. Grams, K. S. Balhara, B. G. Jaar, and D. L. Segev. 2012. Disparities in provision of transplant information affect access to kidney transplantation. American Journal of Transplantation 12(2):351-357.
Kulkarni, S., K. Ladin, D. Haakinson, E. Greene, L. Li, and Yanhong Deng. 2019. Association of racial disparities with access to kidney transplant after the implementation of the new kidney allocation system. JAMA Surgery 154(7):618-625.
Kutner, N. G., R. Zhang, Y. Huang, and K. L. Johansen. 2012. Impact of race on predialysis discussions and kidney transplant preemptive wait-listing. American Journal of Nephrology 35(4):305-311.
Ladin, K., J. Emerson, K. Berry, Z. Butt, E. J. Gordon, N. Daniels, T. A. Lavelle, and D. W. Hanto. 2019. Excluding patients from transplant due to social support: Results from a national survey of transplant providers. American Journal of Transplantation 19(1):193-203.
Lantigua, C., K. Ibarra, A. Herr, K. Giblin, and C. Queram. 2021 (unpublished). An approach to developing a framework for consensus-based standardized performance metrics in the United States transplant system. Paper commissioned by the Committee on a Fairer and More Equitable, Cost-Effective, and Transparent System of Donor Organ Procurement, Allocation, and Distribution, National Academies of Sciences, Engineering, and Medicine, Washington, DC.
Li, P. K., K. H. Chu, K. M. Chow, M. F. Lau, C. B. Leung, B. C. H. Kwan, Y. F. Tong, C. C. Szeto, and M. M. M. Ng. 2012. Cross sectional survey on the concerns and anxiety of patients waiting for organ transplants. Nephrology (Carlton) 17(5):514-518.
Maldonado, J. R. 2019. Why it is important to consider social support when assessing organ transplant candidates? Abingdon-on-Thames, UK: Taylor & Francis.
McCannon, C. J., M. W. Schall, D. R. Calkins, and A. G. Nazem. 2006. Saving 100,000 Lives in US Hospitals. BMJ 332:1328-1330.
McCannon, C. J., D. M. Berwick, and M. R. Massoud. 2007. The science of large-scale change in global health. JAMA 298(16):1937-1939.
Nolan, T. W. 2007. Execution of strategic improvement initiatives to produce system-level results. IHI Innovation Series white paper. Cambridge, MA: Institute for Healthcare Improvement.
Norman, S. P., M. Kommareddi, and F. L. Luan. 2013. Inactivity on the kidney transplant wait-list is associated with inferior pre- and post-transplant outcomes. Clinical Transplantation 27(4):E435-E441.
Obadan-Udoh, E. M., J. M. Calvo, S. Panwar, K. Simmons, J. M. White, M. F. Walji, and E. Kalenderian. 2019. Unintended consequences and challenges of quality measurements in dentistry. BMC Oral Health 19(1).
OPTN (Organ Procurement and Transplantation Network). 2015. Educational guidance on patient referral to kidney transplantation (OPTN minority affairs committee). https://optn.transplant.hrsa.gov/resources/guidance/educational-guidance-on-patient-referral-to-kidney-transplantation (accessed November 10, 2021).
OPTN. 2018. Manipulation of the organ allocation system waitlist priority through the escalation of medical therapies. https://optn.transplant.hrsa.gov/media/2500/ethics_whitepaper_201806.pdf (accessed November 17, 2021).
OPTN. 2020. OPTN to launch a collaborative improvement project to increase recovery of DCD organs. https://optn.transplant.hrsa.gov/news/optn-to-launch-a-collaborative-improvement-project-to-increase-recovery-of-dcd-organs (accessed November 2, 2021).
Patzer, R. E., S. Paul, L. Plainga, J. Gander, L. Sauls, J. Krisher, L. L. Mulloy, E. M. Gibney, T. Browne, C. F. Zayas, W. M. McClellan, K. H. Arriola, and S. O. Pastan. 2017. A randomized trial to reduce disparities in referral for transplant evaluation. Journal of the American Society of Nephrology 28(3):935-942.
Petrenko, A., M. Carnevale, A. Somov, J. Osorio, J. Rodriguez, E. Guiber, B. Fuller, and F. Froghi. 2019. Organ preservation into the 2020s: The era of dynamic intervention. Transfusion Medicine and Hemotherapy 46(3):151-172.
Prottas, J. 1998. Shifting responsibilities in organ procurement: A plan for routine referral. JAMA 260(6):832-833.
Puoti, F., A. Ricci, A. Nanni-Costa, W. Ricciardi, W. Malorni, and E. Orona. 2016. Organ transplantation and gender differences: A paradigmatic example of intertwining between biological and sociocultural determinants. Biology of Sex Differences 7:35.
Ravaioli, M., V. De Pace, A. Angeletti, G. Comai, F. Vasuri, M. Baldassarre, L. Maroni, F. Odaldi, G. Fallani, P. Caraceni, G. Germinario, C. Donadei, D. Malvi, M. Del Gaudio, V. R. Beruzzo, A. Siniscalchi, V. M. Ranieri, A. D’Errico, G. Pasquinelli, M. C. Morelli, A. D. Pinna, M. Cescon, and B. La Manna. 2020. Hypothermic oxygenated new machine perfusion system in liver and kidney transplantation of extended criteria donors: First Italian clinical trial. Scientific Reports 10(1):1-11.
Rodrigue, J. R., M. J. Paek, O. Eguna, A. D. Waterman, J. D. Schold, M. Pavlakis, and D. A. Mandelbrot. 2014. Making house calls increases living donor inquiries and evaluations for blacks on the kidney transplant waiting list. Transplantation 98(9):979-986.
Saemann, L., Y. Guo, Q. Ding, P. Zhou, M. Karck, G. Szabo, and F. Wenzel. 2020. Machine perfusion of circulatory determined death hearts: A scoping review. Transplantation Reviews (Orlando) 34(3):100551.
Salehi, S., K. Tran, and W. L. Grayson. 2018. Focus: Medical technology: Advances in perfusion systems for solid organ preservation. Yale Journal of Biology and Medicine 91(3):301.
Salvalaggio, P. R., N. Dzebisashvili, K. E. MacLeod, K. L. Lentine, A. Gheorghian, M. A. Schnitzler, S Hohmann, D. L. Segev, S. E. Gentry, and D. A. Axelrod. 2011. The interaction among donor characteristics, severity of liver disease, and the cost of liver transplantation. Liver Transplantation 17(3):233-242.
Schnitzler, M. A. 2021. How much could we invest in deceased donor organ procurement? Presentation during February 3–4, 2021 public workshop for the Committee on a Fairer, More Equitable, Cost-Effective, and Transparent System of Donor Organ Procurement, Allocation, and Distribution. https://www.nationalacademies.org/event/02-04-2021/a-fairer-and-more-equitable-cost-effective-and-transparent-system-of-donor-organ-procurement-allocation-and-distribution-a-virtual-workshop (accessed November 11, 2021).
Shafer, T. J., D. Wagner, J. Chessare, F. A. Zampiello, V. McBride, and J. Perdue. 2006. Organ donation breakthrough collaborative: Increasing organ donation through system redesign. Critical Care Nurse 26(2):33-48.
Shafer, T. J., D. Wagner, J. Chessare, M. W. Schall, V. McBride, F. A. Zampiello, J. Perdue, K. O’Connor, M. J-Y. Lin, and J. Burdick. 2008. US organ donation breakthrough collaborative increases organ donation. Critical Care Nursing Quarterly 31(3):190-210.
Sutton, R. I., and H. Rao. 2014. Scaling up excellence: Getting to more without settling for less. New York: Crown Business.
Tatum, R., T. J. O’Malley, A. S. Bodzin, and V. Tchantchaleishvili. 2020. Machine perfusion of donor organs for transplantation. Artificial Organs 45(7):682-695.
Tingle, S. J., R. S. Figueiredo, J. A. Moir, M. Goodfellow, E. R. Thompson, I. K. Ibrahim, L. Bates, D. Talbot, and C. H. Wilson. 2020. Hypothermic machine perfusion is superior to static cold storage in deceased donor kidney transplantation: A meta-analysis. Clinical Transplantation 34(4):e13814.
Tosoc-Haskell, H., K. Sisaithong, and R. Carrico. 2019. The collaborative improvement and innovation network project to drive quality improvement. Current Opinion in Organ Transplantation 24(1):73-81.
Waterman, A. D., J. D. Peipert, C. J. Goalby, K. M. Dinkel, H. Xiao, and K. L. Lentine. 2015. Assessing transplant education practices in dialysis centers: Comparing educator reported and Medicare data. Clinical Journal of the American Society of Nephrology 10(9):1617-1625.
Waterman, A. D., J. D. Peipert, H. Xiao, C. J. Goalby, S. Kawakita, Y. Cui, and K. L. Lentine. 2020. Education strategies in dialysis centers associated with increased transplant wait-listing rates. Transplantation 104(2):335-342.
Wesselman, H., C. G. Ford, Y. Leyva, X. Li, C. C. H. Chang, M. A. Dew, K. Kendall, E. Croswell, J. R. Pleis, Y. H. Ng, M. L. Unruh, R. Shapiro, and L. Myaskovsky. 2021. Social determinants of health and race disparities in kidney transplant. Clinical Journal of the American Society of Nephrology 16(2):262.
Wey, A., J. Foutz, S. K. Gustafson, R. J. Carrico, K. Sisaithong, H. Toscoc-Haskell, M. McBride, D. Klassen, N. Salkowski, B. L. Kasiske, A. K. Israni, and J. J. Snyder. 2020. The Collaborative Innovation and Improvement Network (COIIN): Effect on donor yield, waitlist mortality, transplant rates, and offer acceptance. American Journal of Transplantation 20(4):1076-1086.
Wu, D. A., M. L. Robb, J. L. R. Forsythe, C. Bradly, J. Cairns, H. Draper, C. Dudley, R. J. Johnson, W. Metcalfe, R. Ravanan, P. Roderick, C. R. V. Tomson, C. J. E. Watson, J. A. Bardley, and G. C. Oniscu. 2020. Recipient comorbidity and survival outcomes after kidney transplantation: A UK-wide prospective cohort study. Transplantation 104(6):1246-1255.
Ye, H. J., L.-J. Hu, Y.-Y. Yao, and J.-H. Chen. 2011. The effects of two health education models on psychological and nutritional profile of patients waiting for kidney transplantation. Zhonghua Nei Ke Za Zhi 50(10):845-847.
Zhou, S., A. B. Massie, X. Luo, J. M. Ruck, E. K. H. Chow, M. G. Bowring, S. Bae, D. L. Segev, and S. E. Gentry. 2018. Geographic disparity in kidney transplantation under KAS. American Journal of Transplantation 18(6):1415-1423.
Zubrinic, M., N. Marks, L. Brzozowski, J. Qiu, D. Lin, K. Wang, J. De Romana, L. Singer, and S. Keshavgee. 2019. Prelung transplant waitlist mortality rate reduction using remote video monitoring technology. Journal of Heart and Lung Transplantation 38(4):S199.


























