7
Respiratory Protection Oversight and Guidance for the Public
Chapter 4 broadly reviews the state of the evidence and practice around providing respiratory protection for the public. That chapter draws on a fragmentary body of evidence, much of it related to wildfire smoke and ambient air pollution, along with the more recent experience with the COVID-19 pandemic. The eight conclusions in that chapter describe gaps that relate to one or more functions of the general framework described in Chapter 5. In this chapter, which is organized around the functions of that general framework, the committee addresses how to fill these gaps to create a cohesive framework specifically targeting respiratory protection for the public. The chapter begins with consideration of different options for a coordinating entity in this framework that would be charged with integrating the efforts of all stakeholders to ensure that the various functions are carried out seamlessly to meet the public’s respiratory protection needs broadly—those of both the general population and members of population groups that are vulnerable to specific hazards. This discussion builds on the examples of coordination models discussed in Chapter 5. The chapter then goes on to discuss, for each of the framework functions, the responsibilities and authorities necessary to close the gaps identified in Chapters 2 and 4, as well as considerations for the different stakeholders that could play key roles in carrying out the activities encompassed by each function.
ENSURING ADEQUATE COORDINATION AND AUTHORITIES TO PROTECT THE PUBLIC FROM INHALATION HAZARDS
While largely centralized oversight authorities for occupational inhalation hazards exist in the National Institute for Occupational Safety and Health (NIOSH) and in the Occupational Safety and Health Administration (OSHA) (as discussed in Chapters 2 and 6), there is no counterpart structure in the context of public protection. As a result, the current landscape of respiratory protection for the public is substantially more complicated to address relative to that of occupational respiratory protection, with considerable fragmentation across multiple federal and state authorities and gaps in critical authorities, particularly as relates to conformity assessment and approval of respiratory protective devices. Stakeholders and authorities may vary depending on whether the hazard is an infectious agent, such as SARS-CoV-2, or an environmental hazard, such as wildfire smoke, as well as on where the exposure occurs (e.g., in a transportation or business environment, indoors or outdoors). This complexity is increased by the lack of a centralized repository of information capturing which authorities are responsible for which hazards under which circumstances. Additionally, there are overlapping federal responsibilities and authorities; state and local public health agencies have specific authorities and act under local control; and manufacturers have a key role, as does the health care sector (see Table 7-1). Given this complex landscape, there is a critical need for a coordinating entity with the leadership, resources, and oversight necessary to integrate the efforts of all stakeholders in order to enhance the nation’s preparedness and provide an actionable response when the public is exposed to inhalation hazards, including ensuring that the right respiratory protection is available to the public at the right time, together with guidance on its effective use.
Organizational Considerations for a Coordinating Entity
Chapter 5 presents a range of options for such a coordinating entity, including several example models. Three different architectures can be envisioned for an organizational structure designed to address the respiratory protection needs of the public. The first is a fully centralized model characterized by a single entity that carries out all functions. On the opposite end of the organizational spectrum is a distributed model with a collection of entities operating autonomously, with ad hoc coordination on specific initiatives through such mechanisms as an interagency task force or working group. A hub-and-spoke model is a third option that lies between the two. In this model, functions may be distributed across multiple stakeholder entities (the spokes), but the central hub provides a formalized, ongoing
| STAKEHOLDER | RESPONSIBILITIES AND AUTHORITIES |
|---|---|
| Local governments | Local government officials are responsible for the public safety and welfare of the people in their jurisdiction, and response to emergency incidents that may require the public to use respiratory protection generally begins at the local level. Decision making and public communication by local officials regarding inhalation hazards may be guided by such local governmental authorities as emergency management, public health, and air quality agencies, which may also communicate directly and indirectly with the public through established channels. In some states, requirements for use of respiratory protective devices for purposes of source control may be set at the local level. During the COVID-19 pandemic, mask requirements have been set by some public school systems. |
| State, territorial, and tribal governments | State, territorial, and tribal governments have significant authority over matters of public safety. These authorities enable state governments to issue so called “mask mandates” requiring the use of respiratory protective devices by the public for purposes of source control. At the request of local governments or when incidents span more than one local jurisdiction, states may supplement local government response efforts, including through coordination of warnings and public information and distribution of stockpiled supplies. States also serve as a critical intermediary in the dissemination of information from the federal to the local level. For example, state public health agencies interface directly with the Centers for Disease Control and Prevention (CDC) and translate and disseminate CDC guidance for local public health agencies. |
| Federal government | Federal capabilities and oversight functions regarding public use of respiratory protection are distributed across multiple federal agencies, with critical functions that include support for product development when needed to address gaps; oversight of conformity assessment for respiratory protective devices; surveillance and hazard assessment; oversight and management of the supply chain in anticipation of and during emergencies; and the development and dissemination of information and guidance related to public use of respiratory protection. Information may be communicated directly to the public or disseminated through existing channels mediated by state and local governments. The federal government has limited authority to issue requirements for public use of respiratory protective devices, except in those areas in which it has jurisdiction, such as interstate travel, federal property, and transportation hubs. |
| Private/Nongovernmental organizations | The private sector has the lead role in the United States for the development (design and manufacturing) of respiratory protective devices, although the government may support such efforts through public–private partnerships. Researchers, including those at academic institutions, may also play a role in device design, as well as contribute to efforts to understand the public’s respiratory protection needs. Nongovernmental organizations may be involved in conformity assessment, including standards development and device testing. Regarding the use of respiratory protective devices, private businesses (e.g., grocery stores) may require and enforce their use by members of the public using their facilities. Nongovernmental and community organizations, as well as health care professionals, play critical roles in the “last-mile” reach of information, and in some cases, supplies, to community members and are often leveraged by local public health and other local government agencies. |
mechanism to ensure coordination of their activities; that is, it serves as the coordinating entity. Different configurations for the hub are potentially suitable, as described below.
The committee eliminated from further consideration the two models at the extremes of the spectrum. The distributed model is in fact a version of the status quo. Multiple federal agencies, including the Centers for Disease Control and Prevention (CDC) and the Environmental Protection Agency (EPA), currently act independently with regard to different kinds of inhalation hazards. While there have been laudable examples of effective coordination, as with the development of Wildfire Smoke: A Guide for Public Health Officials, such coordination is dependent on relationships rather than being built into an ongoing and established system. Without structure and designated responsibilities, gaps might not be addressed, particularly in the face of changing circumstances and new scientific evidence, and efforts would likely be focused on single threats at particular moments (e.g., SARSCoV-2). Experience with the COVID-19 pandemic has highlighted the many ways in which this model is inadequate. While a stopgap interagency task force could be established (either by executive action or self-organized) to increase coordination among stakeholders with minimal requirements for resources, this approach would not respond to the committee’s charge to provide a framework that constitutes a unified and authoritative source of information and effective oversight in the development, approval, and use of respiratory protective devices across all of the functions involved.
The fully centralized model, whereby a centralized entity would replace the existing fragmented authority of multiple agencies, provides a useful contrast. This model could support a unified and authoritative source of information and effective oversight, but as discussed in Chapter 5, restructuring several federal agencies and reassigning responsibilities and authorities now held by current stakeholders would present enormous organizational, operational, and legislative challenges. Such a model might be more feasible if the required authorities were missing and needed to be created and given a federal home. In the present circumstances, however, many of the required authorities exist within agencies currently responsible for some of the functions laid out in the committee’s framework. Moreover, such a model could isolate respiratory protective devices from more upstream primary prevention efforts and create an additional barrier to exchange, integration, and communication.
The committee therefore views the hub-and-spoke model as the preferable path forward to address the need for a coordinating entity within a broader structure designed to ensure adequate and effective respiratory protection for the public. This model would allow the domain-specific expertise and infrastructure of existing stakeholders to be leveraged while providing mechanisms for coordinating efforts toward a shared objective.
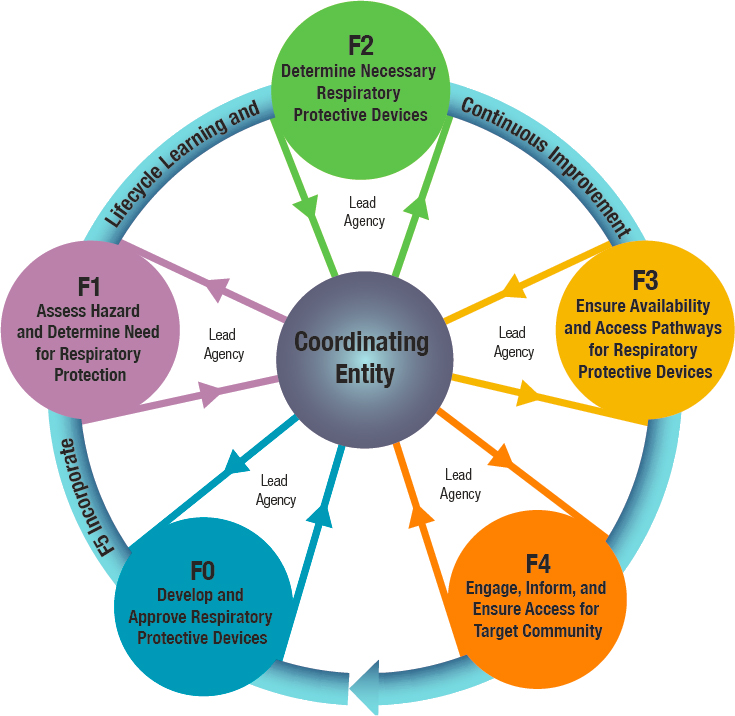
Thus in this model, the hub represents a coordinating entity serving as the unified and authoritative source of information and effective oversight in the development, approval, and use of respiratory protective devices, while the spokes represent stakeholder agencies with domain-specific expertise and infrastructure related to one or more framework functions (see Figure 7-1). Designating a coordinating entity to serve as a central hub also has the advantage of creating a single locus of accountability for congressional and executive oversight. This model differs from the existing situation wherein an empowered and accountable hub is lacking.
This is not to say that the hub-and-spoke model is without challenges. In particular, there are missing authorities that need to be addressed,
particularly for conformity assessment for respiratory protective devices for the public. Additionally, issues could arise in which the authority of the hub could potentially overlap with an authority already granted to an individual spoke. The potential for such overlap would necessitate ongoing cooperation between the coordinating entity and the agencies that already provide hazard-specific or other expertise, and it would remain important for the coordinating entity to be guided by the expertise of the spokes, with feedback being sought from all levels, including those responsible for on-the-ground implementation. In addition, overlapping authority can impede decision making in the absence of consensus and create tensions among existing stakeholders over questions of authority. In the absence of a hierarchical authority structure, requirements for participation and coordination might need to come from the White House or Congress (as codified in legislation). There is no simple solution to the effective management of cross-cutting issues that require the engagement of multiple federal agencies.
The coordinating entity (the hub) could be designated from among existing stakeholders—those that could potentially serve as spokes—or a new agency could be created to fill this role. To the extent that a coordinating entity displaced authority currently allocated to another agency, implementation of the hub might require some restructuring of government agencies (organization and authorities), thereby entailing some of the disadvantages of the fully centralized model, although to a lesser extent. As noted above, the creation of a new federal agency would carry the risk of limiting integration of its function into a larger system of controls aimed at primary prevention. Alternatively, housing the hub in an existing agency could leverage the infrastructure and expertise of that agency.
The committee’s analysis of existing authorities and responsibilities presented in Chapter 2 points to two federal agencies with public health missions that could potentially house the coordinating entity—the Department of Health and Human Services (HHS) and EPA. Within HHS, the coordinating entity might best reside at the level of the Office of the Secretary, but alternatively, it could be housed within an operating division, such as CDC or the Food and Drug Administration (FDA), with implications related to oversight as discussed below. Other potential agencies that might be considered include the Federal Emergency Management Agency (FEMA) within the Department of Homeland Security (DHS) and the Consumer Product Safety Commission (CPSC), but both have lesser responsibilities and expertise related to inhalation hazards and respiratory protection relative to HHS and EPA.
EPA has substantial regulatory authorities related to environmental hazards, but a far less prominent role when inhalation hazards are infectious agents, excepting the guidance it develops for indoor exposure to
mold or its approval of disinfectants for airborne agents such as tuberculosis or SARS-CoV-2. HHS has lesser regulatory authority regarding environmental hazards relative to EPA, but its component agencies would bring a depth of expertise in both environmental hazards and infectious agents, as well as conformity assessment (i.e., FDA and NIOSH’s National Personal Protective Technology Laboratory [NPPTL]). Moreover, the secretary of HHS has oversight over CDC, FDA, the National Institutes of Health (NIH), and the Office of the Assistant Secretary for Preparedness and Response (ASPR), four key federal stakeholders with regard to respiratory protection. Therefore, housing the coordinating entity within HHS would enable a more hierarchical authority structure than would be possible were the coordinating entity to be housed within EPA.
As the nation’s preeminent public health agency, CDC is also reasonable to consider as the locus of the coordinating entity. It would certainly be expected to have a prominent role in a framework of responsibilities and authorities related to oversight and guidance for respiratory protection for the public, and indeed has had such a role with SARS-CoV-2. Multiple centers within CDC provide expertise related to hazards both environmental (e.g., the National Center for Environmental Health) and infectious (e.g., the National Center for Immunization and Respiratory Diseases and the National Center for Emerging and Zoonotic Infectious Diseases). Additionally, CDC has established channels for coordination with state, local, tribal, and territorial public health agencies, which are necessary partners in providing respiratory protection for the public. However, coordination within HHS and with stakeholders outside of HHS (e.g., EPA, DHS) could be impeded if the coordinating entity were placed at the level of an operating division. Moreover, the coordinating entity would have to be able to respond to hazards at a pace that reflects the urgency of the need for respiratory protection. During the COVID-19 pandemic, CDC has been slow to respond to rapid advances in scientific evidence (Tanne, 2020), including the evidence clearly documenting airborne transmission of SARS-CoV-2 by aerosols (NASEM, 2020; Samet et al., 2021). These factors are important considerations in determining whether the coordinating entity should reside within the Office of the Secretary of HHS or alternative locations within the department.
Regardless of the location in which it resided, to ensure the successful functioning of the hub-and-spoke model, the coordinating entity would need to have
- sufficient authority to coordinate across multiple agencies effectively and enforceably;
- sufficient resources to generate new research and data where needed, to respond to emerging circumstances with meaningful action, and to enforce requirements placed on stakeholders; and
- systems for accountability to ensure that the coordinating entity would act expeditiously and effectively in addressing the respiratory protection needs of the public.
No stakeholder agency is currently empowered to take on this broad scope of responsibility. A long-term unified solution to fill this gap will require a clear congressional mandate, codified in legislation, and supported by additional resources via congressional action. In the interim, however, urgently needed coordination could be provided through mandates arising from executive action. Ensuring that the agency designated as the coordinating entity was empowered to carry out this coordination function might necessitate one or more mechanisms for facilitating interagency collaboration, such as memorandums of understanding and joint budgeting, and these mechanisms would need to be considered by Congress. Requirements for annual reports to Congress could be considered as one mechanism for accountability.
Of note, while this coordinating entity should be focused on the respiratory protection needs of the public, coordination with OSHA on respiratory protection issues that overlap with the needs of workers is critical to reducing the potential for conflicting messages and confusion and ensuring a seamless system for the nation. Given the inherent overlap in the activities of the groups tasked with carrying out these functions, the science guiding
those activities, and the populations themselves—outside of the work setting, workers make up a substantial proportion of the general population—harmonization between the two frameworks is vital. Issues requiring collaboration with OSHA may include carrying out communication and messaging, assuring the supply chain, and conducting coordinated research on respiratory protection. The committee recognizes that this collaboration may present some challenges; in the recent past, there have been strong disagreements between HHS agencies and external occupational health professionals on the controls needed to protect workers against airborne infectious diseases (Tanne, 2020). Successful collaboration will require that these coordinating agencies overseeing respiratory protection for the public and for workers view each other as partners, each acknowledging the other’s expertise in the realm over which it has responsibility. Including ongoing evaluation of harmonization of the two frameworks among the requirements for regular assessment and reporting, as noted in Recommendation 7-9, will serve to ensure that objectives related to collaboration and coordination are met.
In this section, the committee has laid out options for a coordinating entity structure. It should be emphasized, however, that this proposal represents a starting point for a new system, one that would need to evolve over time through a process of ongoing evaluation. The specific coordination role for this entity for each of the functions in the committee’s framework is described in the sections that follow and compiled at the end of the chapter.
DEFINING RESPONSIBILITIES AND ADDRESSING GAPS IN AUTHORITIES RELATED TO THE FRAMEWORK FUNCTIONS
As discussed in Chapter 2, the regulatory and legal landscape with respect to oversight of nonoccupational use of respiratory protection is extraordinarily complex, with both gaps and overlaps in the authorities related to the key functions of the committee’s framework (described in Chapter 5). The review and assessment presented in Chapters 2 and 4, respectively, document a patchwork of entities currently involved with
the functions and abundant gaps that must be filled if the framework is to be realized. While a coordinating entity is necessary to harness the authorities, resources, and domain-specific expertise of all stakeholders to that end, each function may require one or more leads with the necessary domain-specific expertise, resources, and authority to serve in an oversight role for that function. One critical task as the framework for the public is implemented will be identifying the various entities and stakeholders that are active in relation to these functions and those that could be engaged to serve as leads for each. Consequently, the committee offers the following recommendation.
For each framework function, it will be necessary to consider the role of the coordinating entity and where gaps in responsibilities may need to be filled by a lead agency that has been granted the necessary authorities. In some cases, the lead agency for a function might be the coordinating entity. The committee’s assessment of the necessary capabilities and oversight authorities of stakeholders for each function and its consideration of potential stakeholders that might fill current gaps are discussed in the following sections.
Function F0: Develop and Approve Respiratory Protective Devices
Massive wildland fires in the western United States and the COVID-19 pandemic have underscored the public’s need for access to appropriate respiratory protection. Current devices used by the public with the intent of reducing exposure to inhalation hazards are a mix of NIOSH-approved industrial respirators, medical and nuisance dust masks, and a variety of other commercial and homemade face coverings (see Chapter 4). The current knowledge and technical base for respiratory protection is derived largely from OSHA regulation (i.e., from requirements for workplace respiratory protection programs) and NIOSH research on worker needs. No
comprehensive study has characterized the public as users of respiratory protection or assessed their equipment needs, although as discussed in Chapter 4, such research is now under way as a result of COVID-19.
The workforce is made up largely of healthy individuals from aged 15–64, although changes in Social Security retirement benefits encourage working to age 70 (Leonesio et al., 2012), and the Americans with Disabilities Act11 promotes employment among persons with disabilities. The public, by comparison, represents a broader age range, from infants to senior citizens, as well as individuals with a variety of health issues and special needs. As a result, the numerous factors that affect the design and appropriate use of respiratory protection (e.g., facial characteristics, fit in the absence of fit testing, acceptable breathing resistance, wearability, the need for an end-of-service-life indicator) are not well understood in the context of public use (Chughtai et al., 2019; Fischer et al., 2020; Regli et al., 2021; van der Sande et al., 2008).
No federal entity comparable to NIOSH/NPPTL is responsible for overseeing the development, approval, marketing, or postmarketing surveillance of all respiratory protective devices for the public, or for generating or overseeing a conformity assessment program for managing these processes. The lack of an appropriate management system and user information could result in multiple failures of a device worn by the public, resulting in inadequate respiratory protection and the potential for unacceptable risks.
To address these major shortfalls, a robust research and approval program is needed to evaluate the needs of users and device requirements and guide the development of comprehensive performance standards and innovative respiratory protective devices specifically designed for public use, including those that would not require fit testing. The needs and requirements should be informed by ongoing scenario planning, a function that itself suffers from lack of coordination within the federal government. As with the development of workplace regulations and consensus standards, private-sector manufacturers and other businesses need to be active participants in these efforts, providing input on technology for future product development and considerations related to manufacturing and supply chain processes. The conformity assessment component of this program will need to ensure the quality, effectiveness when used outside of a respiratory protection program, and appropriate labeling of respiratory protective devices to be used by the public.
___________________
11 Americans with Disabilities Act of 1990 as amended by the ADA Amendments Act of 2008 (Public Law 110-325).
Addressing Gaps in Responsibilities and Authorities
In addressing how best to meet the need for a robust research and approval program focused on nonoccupational use of respiratory protection, the committee considered the respective responsibilities of the coordinating entity and a federal-level organization that would have the authority to oversee standards development for and approval of respiratory protective devices intended for use by the public (i.e., the scheme owner). These respective roles and different potential approaches to addressing current gaps in oversight authorities are described in Box 7-1.
As laid out in Box 7-1, there are two potential approaches for establishing the necessary capability; each has pros and cons, and the committee did not identify a preferred approach. One model is to expand NIOSH/NPPTL’s research portfolio and respirator approval program to encompass respiratory protection for the public, including a wide range of potential public exposure scenarios and user needs. Advantages of this approach include harnessing its unique capabilities in respiratory protection (e.g., conformity assessment and respirator research) built within NPPTL over the past two decades. Doing so might address the public’s needs more quickly relative to establishing a new organization. However, expansion of NPPTL’s mandate beyond workers could pose challenges given its location within NIOSH, as well as have unanticipated impacts on its occupational oversight functions. Another risk is that inadequate additional funds could be provided to NPPTL for this expansion on the assumption that this is work similar to what it currently performs. In actuality, this work related to the public is likely to be quite different from the work done for workers. Furthermore, such expansion would require congressional action, which could delay the implementation of this approach and NPPTL’s ability to address the public’s needs expeditiously.
An alternative would be to develop a parallel system for nonoccupational contexts with a laboratory analagous to NPPTL as the lead for research on and approval of respiratory protective devices for the public. The advantages of establishing a new laboratory include a dedicated focus on meeting the unique needs of the many members of the public for whom currently available respirators are problematic—including infants, young children, and the frail elderly—and the ability to house the new laboratory in an organization with a broader primary prevention mission than that of NIOSH. This approach could require some duplication of infrastructure and would need to be explored in close collaboration with NPPTL to build on the best practices and lessons learned over its two decades of experience. This collaboration would likely help advance the development of the new organization while avoiding duplication of resources.
Considerations that should guide the choice between expanding the capacity and funding for NPPTL and establishing a new laboratory include the differences between the user populations to be covered (i.e., the general population versus workers) and the implications for the necessary capabilities of the laboratory, the comparative efficiencies of the two approaches, and the administrative infrastructure that would have to be created around providing respiratory protection for the public. Assessment of the two approaches would logically be one of the first tasks in the effort to assign and organize the roles and responsibilities described in Recommendation 7-3. Regardless of which approach is chosen, significant additional funding will be necessary to carry out these major new tasks.
Function F1: Assess Hazard and Determine Need for Respiratory Protection
Protecting the public from such diverse inhalation hazards as wildfire smoke, dust storms, infectious aerosols (e.g., SARS-CoV-2), and mold from flooding events, among others, will require a system for identifying and assessing hazards, evaluating exposure and risk, and determining the need for respiratory protection. This core function is complex because of the diversity of inhalation hazards faced by the public and the potential need for a range of scientific expertise to assess them.
CDC has various systems and a formalized process for identifying new infectious disease outbreaks, but its process for evaluating risk for the public and generating specific public health recommendations regarding the need for public use of respiratory protection is fragmented and ad hoc. While experts in infectious diseases are dispersed across multiple centers within CDC, expertise in respiratory protection is limited to those program units focused on occupational safety and health. In the absence of a permanent locus within CDC focused on respiratory protection for the public, relevant experts from NIOSH and CDC’s Division of Healthcare Quality Promotion make decisions about the public’s need for respiratory protection through an emergency management structure (i.e., incident command) that is activated with each new public health emergency (Delaney,
2021). Such structures have been used to negotiate recommendations on respiratory protection for the public during the H1N1 influenza and COVID-19 pandemics, and the unprecedented duration of the latter pandemic has enabled enhanced functionality as the response has matured. However, incident command structures are inherently temporary and hazard specific, and therefore not well suited to the development of forward-thinking and formalized processes for considering inhalation hazards faced by the public or the public’s respiratory protection needs. Moreover, reliance on CDC’s occupational safety and health experts for the substantial task of addressing the public’s respiratory protection needs could distract from the important focus of such groups on the needs of workers. Occupational safety and health experts also may have limited expertise regarding the full range of subpopulations for whom respiratory protection poses unique challenges, including young children, the frail elderly, and people with certain medical conditions.
The incident command structure at CDC includes mechanisms and pathways for assembling evidence on infectious inhalation hazards to guide recommendations on respiratory protection for the public (and workers). A chief science officer is responsible for ensuring that those recommendations are guided by the best available evidence; the evidence supporting the recommendations is distilled into science briefs (de Perio, 2021), providing some measure of transparency. Multistakeholder meetings are held to facilitate discussions on how the latest science might inform policy considerations, although there is no formalized process for determining the need for interagency engagement. Notable challenges include the lack of data to inform risk evaluations and reliance on systems built for academic publication of data (Bell, 2021). While preprint servers have increased access to recently generated data that have not yet been peer-reviewed, the timeframe for publication still poses barriers to rapid reviews of quickly evolving evidence. As a component of a response structure, these processes for compiling evidence are hazard-specific and do not represent a long-term solution for the ongoing evaluation of inhalation hazards faced by the public and its respiratory protection needs.
A useful example of an ongoing standardized, rigorous process for monitoring hazards and translating the available science to public health policy is EPA’s process for setting National Ambient Air Quality Standards (NAAQS). The Clean Air Act Amendments of 197722 mandates that the agency repeat this process every 5 years for the major criteria air pollutants. The process begins with a science policy workshop held to gather input from the scientific community, followed by the creation of an Integrated Review Plan designed to move the process forward (EPA, 2020). The review
___________________
22 Clean Air Act Amendments of 1977, Public Law 95-95.
contains three parts in order: an Integrated Science Assessment (ISA), a formal, structured review and synthesis of the current policy-relevant science; (2) a Risk/Exposure Assessment that both characterizes the uncertainty in the estimates described by the ISA and uses those estimates to characterize risk to humans and the environment; and (3) a Policy Assessment that provides the scientific basis for different policy options. Based on this formal review, EPA makes final recommendations for specific air quality standards for such pollutants as PM2.5, ozone, and nitrogen dioxide, which are finalized following a period of public comment and public hearings. These standards are also translated into EPA’s Air Quality Index, a color-coded metric used to communicate relative risk to the public.33 It should be noted, however, that EPA does not currently conduct such ongoing assessments for other environmental inhalation hazards that pose risks to the public, such as wildfire smoke and volcanic ash.
Addressing Gaps in Responsibilities and Authorities
To protect the public health from the diverse inhalation hazards of concern, a coordinating entity is needed to oversee the compilation of existing knowledge on exposure and risk; gather input from experts; and guide the public, including specific subgroups, on the need for respiratory protection, accounting for alternative strategies (e.g., other control methods, such as social distancing and vaccination) and potential health risks of using respiratory protective devices. This core function will require coordination across multiple agencies, based on the relevant scientific and administrative expertise of each agency and the nature of the inhalation hazard in question (Shaffer, 2021). The respective roles of the coordinating entity and the agencies with the relevant expertise for different inhalation hazards faced by the public are considered in Box 7-2.
Decisions about the public’s need to use respiratory protection have broad policy, operational, and societal implications. Such decisions need to be based on the best available science, but also informed by a breadth of perspectives so that the full ramifications of such determinations are thoroughly considered. Consequently, the committee favors Approach 1 in Box 7-2, whereby recommendations on public use of respiratory protective devices are made through a collaborative, interagency effort mediated by the coordinating entity rather than by a single agency.
To ensure that determinations regarding the public’s need to use respiratory protection are science based, they need to be informed by a structured scientific review and risk evaluation process led by the agency with
___________________
33 For more information on the Air Quality Index, see https://www.airnow.gov/aqi/aqi-basics (accessed October 5, 2021).
the relevant expertise for the hazard at hand (e.g., EPA for environmental hazards, CDC for infectious hazards). While EPA’s NAAQS process can serve as a useful example of formalizing the steps of hazard identification, exposure monitoring, and risk evaluation to inform recommendations on the public’s need for respiratory protection, such a process does not necessarily require a congressional mandate. In some cases (such as novel infectious agents), a formal evaluation may require an accelerated timeline for the scientific review of the evidence. Thus, in addition to the creation of a standardized process similar to that used for the NAAQS, it could be helpful to create a supplemental process that uses rapid reviews (e.g., following the Cochrane rapid review guidelines) (Garritty et al., 2021) as a starting point, followed by more comprehensive reviews.
Any system broadly addressing respiratory protection for the public would differ from EPA’s process for establishing the NAAQS in other important ways. Whereas the NAAQS process is limited to specific air pollutants, the coordinating entity for respiratory protection for the public would need to regularly survey existing and emerging hazards that might trigger that need. Thus, a novel system would be needed. Such a surveillance system would have to be transparent to the public and to include opportunities for public comment and stakeholder engagement. One factor that complicates implementing such a system, however, is that population exposure data and quantitative risk assessment are currently lacking for hundreds of potential inhalation hazards. In contrast to EPA’s national monitoring network for criteria air pollutants, for example, the United States currently has a very limited ability to measure or forecast public exposure to such hazards as wildfire smoke (Rice et al., 2021) and volatile chemical compounds from industrial accidents. In the case of infectious agents, information on transmissibility or virulence may not exist, especially in the early stages of disease outbreaks.
Another key distinction is that the NAAQS process began at a time when a large, published literature of cohort studies, mechanistic animal research, and controlled human studies had already demonstrated adverse health effects of exposure to criteria air pollutants, which could be used to estimate risk (i.e., exposure-response relationships). For new hazards, the task of estimating exposure-response, quantifying transmission risks (for infectious agents), or identifying higher-risk/vulnerable populations may begin with a very sparse scientific literature. Thus, coordinating and integrating new scientific knowledge on exposure and risk is crucial. A major advantage of a rigorous scientific review process is that knowledge gaps are identified in a timely fashion, so that research to address such gaps can be prioritized.
Lastly, while the NAAQS process leads to ambient air quality standards, any recommendations regarding the public’s use of respiratory protection
involve an intervention at the level of the individual, with potential health risks that remain poorly described. Therefore, as part of the core function of assessing hazards and making recommendations for the use of respiratory protection, the coordinating entity will need a clear understanding of the safety of respiratory protective devices across different population subgroups. The coordinating entity will then need to compare the anticipated benefits of respiratory protection against any (known) risks of use among members of the public and consider alternative strategies for reducing their exposure.
Function F2: Determine the Necessary Respiratory Protective Devices
Once it has been determined that an inhalation hazard faced by the public warrants the use of respiratory protection, recommendations on specific devices will need to be matched to the nature of the hazard and guidelines developed regarding their use. For example, widely available FFRs (e.g., N95 FFRs) are appropriate for particulate hazards, including infectious microorganisms (OSHA, 2011). Many inhalation threats faced by the public are particulate in nature, including particulate matter components of wildfire smoke, respiratory viruses, and mold spores. For other potential hazards, including such gases as chlorine, appropriate respirators, such as elastomeric half-mask respirators, may not be widely available to the public. Additionally, any device-specific risks to the wearer (Kyung et al., 2020) will need to be considered in developing recommendations for the selection of respiratory protective devices. Risk-benefit considerations for respiratory protection more generally are discussed under Function F1, but they play a crucial role in making those recommendations.
Ideally, recommended devices will be approved for public use following a conformity assessment process (Function F0) and selected from some form of approved equipment list, analogous to the NIOSH Certified Equipment List.44 As yet, however, the government has approved no respiratory protective devices for public use in the United States. During the COVID-19 pandemic, wildfires, and air pollution events, members of the public have found themselves using devices not designed for them or improvising devices that provide an unknown level of protection. The absence of approved devices will remain an impediment for the public until the necessary systems and infrastructure can be established to fill this gap (Recommendation 7-3), and industry can manufacture and scale up the design of appropriate respiratory protective devices. In the interim, emergency use authorization (EUA) could be granted for existing devices, a step taken by FDA during the COVID-19 pandemic (CRS, 2021), with authorized devices being listed in a central database.
Current market-driven approaches for the development, manufacture, and marketing of respiratory protective devices rely heavily on the regulatory model for occupational respiratory protection in that devices are manufactured with the knowledge that their use will be required in certain industries to perform certain tasks. Thus, manufacturers have a guaranteed market. However, respiratory protective devices for the public would likely not carry such requirements, and their manufacture would therefore be a riskier proposition for industry. Indeed, two 3M respirators that had received regulatory approval for use by the public during public health emergencies were made available during the H1N1 pandemic. However, few were ever purchased because the pandemic was over by the time the products were marketed, and they were then discontinued in 2013 (FDA, 2009; McCullough, 2021). Accordingly, both consumer demand (e.g., as a result of public education and preparedness campaigns) and incentives for manufacturers to develop and produce these devices are necessary to ensure an ongoing market for public use.
The provision of such incentives would likely entail government work to develop relationships with manufacturers for the new devices, as well as federal government purchase guarantees (e.g., for inclusion in the Strategic National Stockpile [SNS]) (Robinson, 2021; Watney and Stapp, 2020). Within ASPR, the Biomedical Advanced Research and Development Authority (BARDA) supports advanced development and scale-up of manufacturing capacity for medical countermeasures, such as personal protective equipment (PPE), and promotes partnerships with developers
___________________
44 NIOSH’s CEL is available at https://wwwn.cdc.gov/niosh-cel (accessed September 28, 2021).
and manufacturers. Inclusion in a database of approved devices for use by the public would likely help drive the consumer market to those products.
Addressing Gaps in Responsibilities and Authorities
Current processes for generating recommendations on devices meeting the public’s needs for respiratory protection are ad hoc and often rely on collaborative relationships among multiple federal stakeholders. Needed is a formalized and enduring process and structure with a clear
lead organization responsible for determining which existing respiratory protective devices best meet those needs based on evaluation of the hazard and risks at hand (Function F2) and review of the technical specifications and capabilities of existing devices. This lead organization also will need to develop rubrics for use at the state and local levels to provide recommendations on respiratory protection that accord with local environmental conditions. Stakeholder roles and different potential approaches to addressing current gaps in oversight authorities for Function F2 are described in Box 7-3.
As indicated in Box 7-3, the characteristics of a lead organization for Function F2 overlap with those for Function F0. Thus, the laboratory called for in Recommendation 7-3 is also well suited to lead the process for determining which existing respiratory protective devices best meet the needs of the public and developing guidelines for their use to inform targeted and tailored guidance for specific audiences (Function F4). To ensure that such specific device recommendations are grounded in an understanding of the hazards and risks of concern, the coordinating entity will need to ensure that the laboratory can access the hazard and risk evaluation conducted in the context of Function F1.
Function F3: Ensure Availability and Access Pathways for Respiratory Protective Devices
Supply chain failures during the COVID-19 pandemic disrupted the supply of respiratory protective devices for occupational settings (particularly for health care) (CRS, 2020; Mason and Friese, 2020), a problem exacerbated when those same devices became sought by the public. Countries established export restrictions on various respiratory protective devices and source materials (e.g., meltblown fabric) they deemed necessary for their own nation’s needs (Cohen and Rodgers, 2020; Gruley and Clough, 2020). Given the future likelihood of these types of global supply chain disruptions, differentiating manufacturers’ domestic and global capabilities would provide a more complete picture of availability during a global crisis.
To address the kinds of shortfalls in domestic production capability and capacity for a variety of respiratory protective devices experienced during the COVID-19 pandemic and other past emergency events, the development of such products for the public—particularly those that do not rely heavily on the model of the occupational respiratory protection program, including fit testing, needs to be encouraged. As discussed above, doing so will require building strong relationships with and providing incentives for manufacturers (Patel et al., 2017) to produce and innovate in the development of such
devices. Moreover, the SNS and other stockpiles cannot be static depots of supplies that are used only when needed. Rather, they need to be integrated into the national supply chain such that there is turnover in inventory and ongoing evaluation of the performance of stockpiled devices. This would reduce the likelihood of stockpiled devices not being properly maintained or reaching their expiration dates without having been used, as well as yield greater understanding of which devices were being used in practice. To this end, the SNS requires a sophisticated product tracking system.
ASPR, which leads the Public Health Emergency Medical Countermeasures Enterprise (PHEMCE), oversees the procurement of medical countermeasures, including PPE such as respirators, and their associated inventory management, including stockpiling (CDC, 2014; Gerstein, 2020; NASEM, 2016). Both ASPR and its component agency, BARDA, have lead roles in procuring medical countermeasures for stockpiling. The Department of Defense (DoD) and the Department of Veterans Affairs may also serve as needed in planning or executing stockpiling options (ASPR, 2015). The SNS provides for significant purchasing power on the part of the federal government by establishing and guaranteeing a marketplace for respiratory protective devices (CDC, 2014). This governmental marketplace will be needed until the concept of respiratory protection for the public is embraced, and the public actively seeks out such devices and includes them in disaster supply kits for use during infectious disease, wildfire, and air pollution events. Such purchasing power can also drive innovation in respiratory protective devices through the establishment of requirements for products that will be stockpiled (Robinson, 2021).
During emergencies necessitating respiratory protection for the public, the federal government will likely need to serve as the gatekeeper of purchasing and distribution operations. Carrying out this function will require knowledge of the various distribution mechanisms and understanding of equity issues and relationships between the SNS and state, local, tribal, and territorial governments. In the event of wildfires or other pollution events in one region of the country, for example, the federal government will need to be able to divert more devices to affected areas from areas with either less need or less utilization, by, for example, developing and updating (in real time) a “heat map” of available and needed devices nationwide (NASEM, 2019).
During the COVID-19 pandemic, there were clear disparities in who had access to respiratory protective devices, leading to a widening of the COVID-19 risk differences across some groups. Diverse pathways for distribution of respiratory protective devices are needed in response to the heterogeneity of the population and the accessibility challenges for many of its subgroups. Design of access pathways needs to be prospectively considered. Given the importance of community trust in respiratory protective
devices to people’s willingness to use them, it will be important to consider issues of health equity in the procurement and distribution process (e.g., having minority-owned businesses as part of the procurement chain). Federal agencies overseeing the supply chain have historically not had the infrastructure to prioritize elimination and prevention of disparities among different population groups. Consideration should be given to opportunities to partner with other agencies that have this focus, including those at the subfederal level that are able to solicit feedback directly from community partners to guide the creation of a more equitable system.
The National Strategy for a Resilient Public Health Supply Chain, released in July 2021, calls for a strategy for improving the coordination and distribution of PPE, including respiratory protective devices, in public health emergencies (HHS et al., 2021). In the development of such a strategy, it will be important to attend to, among other issues,
- a process for ensuring that stockpiled products continue to meet performance requirements and for rotating out and replacing stock nearing its shelf life;
- inventory and distribution systems, including processes for coordinating with and leveraging state, local, tribal, and territorial agencies to ensure access for underserved groups; and
- a means of guiding appropriate use of the Defense Production Act (DPA) (see Chapter 2) to address shortages.
Addressing Gaps in Responsibilities and Authorities
The supply chain related to PPE, including respiratory protective devices, has received inadequate attention, with consequences that were apparent during the COVID-19 pandemic. Remedying this gap will require a responsible party with the authority to serve as the interface between manufacturers and existing federal authorities related to supply chain. As described in Box 7-4, this function could be filled by the coordinating entity or another lead agency. Given that this is a clear coordination function, the committee favors the former option.
The coordinating entity would be tasked with monitoring and guiding federal efforts to manage the supply chain so as to ensure adequate supplies of respiratory protective devices to meet public needs (e.g., providing input to those with the authority to invoke the DPA or working with manufacturers to incentivize production). Coordination with OSHA will be important to ensure that such efforts are not siloed. The coordinating entity would need to maintain the expertise to understand and engineer the market forces involved in the manufacture of these devices. The entity would also need to establish and sustain relationships with manufacturers both to ensure
production capability and to trigger innovation within the industry (e.g., devices with improved fitting characteristics, devices suitable for children of a variety of ages), similar to the way DoD and BARDA work with their respective manufacturing bases. These relationships enable the entity to work nimbly with manufacturers to shift production between different types of respiratory protective devices, if possible, to address immediate needs.
Function F4: Engage, Inform, and Ensure Access for the Target Community
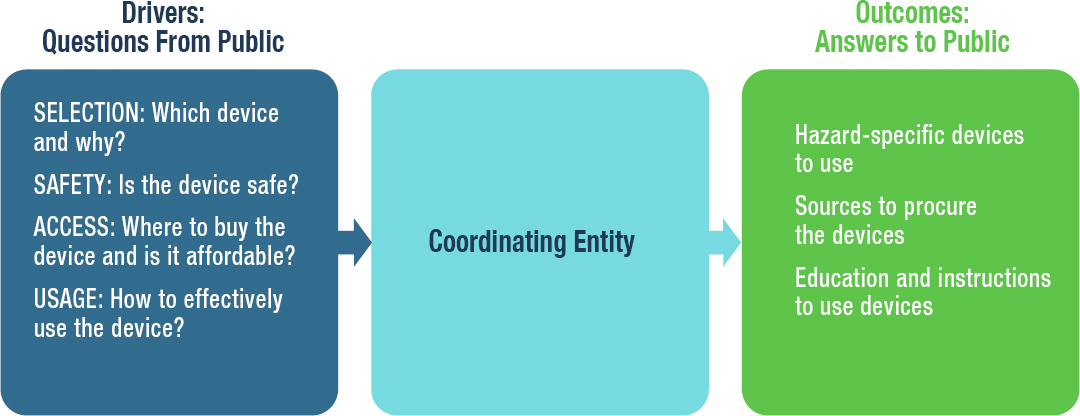
Mitigating inhalation hazards outside of occupational settings is challenging because the public does not know where, which, how, and when respiratory protective devices should be used (see Chapter 4). People need clear, consistent, and timely guidance on the selection, acquisition, and effective use of appropriate respiratory protective devices for specific hazards that addresses their risk perceptions, preferences, and beliefs for that hazard (see Figure 7-2).
Given that respiratory protection is effective only if used correctly, education and training, ideally incorporating some means of feedback, are critical. A core function of public health agencies is health education. As discussed in Chapter 4, health information originating from CDC is typically passed through state health departments to local health departments, which then disseminate the information to health care providers and the public after ensuring that it is understandable and linguistically and culturally appropriate.
Education is most successful in reaching all segments of the nation’s increasingly diverse communities when public health agencies engage with other organizations that routinely connect with and communicate health information to broader populations. While guidance can and should be provided directly to the public, its acceptability and use are amplified when it is conveyed through such trusted sources as health care providers and systems, faith leaders, community leaders, and other community-based organizations that serve specific population subgroups (Stout et al., 2019; Udow-Phillips and Lantz, 2020). Individuals who are from the target community, such as community health workers and promotoras, can be particularly effective at facilitating communication and education in local contexts. Effective communication is achieved when repeated and consistent messages in the audience’s preferred language are delivered through multiple channels, through trusted individuals and organizations. Formatting guidance as
talking points that can easily be tailored to specific audiences and communication needs facilitates its dissemination (see Chapter 4). During the COVID-19 pandemic, agencies at the state and local levels have leveraged their community partners and staff in educational efforts designed to promote prevention measures that limit community transmission, including safe isolation and quarantine, increased testing, and vaccination (Buchner et al., 2021). Such community partnerships are critical to equity such that all groups have access to key information and resources. Additionally, these community partners can play a critical role in helping public health stakeholders understand perceived barriers impeding adherence to the guidance provided, such as the reasons behind the hesitancy toward the COVID-19 vaccine.
Health care providers also have traditionally played a critical role in health education because of the high level of trust their patients have in
them when making difficult health-related decisions. They have the added benefit of knowing the medical histories of their patients, enabling them to offer personal guidance that can increase effective use of respiratory protective devices and compliance with public health guidance.
In the absence of a formal respiratory protection program for the public then, public health professionals and health care providers are left with the critical responsibility of providing guidance to the public on the medical indications for and use of respiratory protection (Harber et al., 1996). However, many lack adequate training and the tools and resources needed to perform this function well (Alfano-Sobsey et al., 2006; Barratt et al., 2020; John et al., 2017). To be effective in this role, they need to be better equipped to do so. Thus a critical gap is the lack of formal requirements or mechanisms for public health agencies and health care providers to be trained in respiratory protection so they can be confident in evaluating resources to which they can direct their patients.
In addition to communication pathways, the form and content of health messages influence their effectiveness. As discussed in Chapter 4, however, messages about respiratory protection have not been adequately tested, and the most effective strategies for ensuring appropriate selection and use of these products by the diverse communities that make up the U.S. public are unknown. Instructions on appropriate use of respiratory protective devices need to be clear (Clayman et al., 2010) and to take account of health literacy principles, such as those espoused in the National Standards for Culturally and Linguistically Appropriate Services (CLAS) in Health and Health Care (Think Cultural Health, 2018). The National CLAS Standards are a set of action steps intended to advance health equity, improve quality, and help eliminate health care disparities by providing a blueprint for individuals, public health agencies, and health care organizations to provide culturally and linguistically appropriate services (Think Cultural Health, 2018). It is especially critical that messages be developed with the input of relevant experts to ensure their accuracy and tested with members of the target audiences to ensure that they have the intended effect in different population groups. Reliance on “common sense” strategies will not necessarily lead to effective messaging (Council of Canadian Academies, 2015; NASEM, 2017).
Individually focused strategies for promoting the use of respiratory protective devices during a public health emergency such as COVID-19, when the hazard is invisible, may not be as successful as would be the case with a visible hazard such as smoke during a wildfire (Lakhina and Lebeda, 2021; Santana et al., 2021). Recognizing that risk perception varies, risk should be communicated in a relatable way and described by comparison with commonly known inhalation hazards—the approach taken with air quality
warnings and COVID-19 with seasonal influenza and chickenpox being used as analogies to communicate transmissibility (Buchner et al., 2021).
Messaging to the public needs to address not only what should be done in the midst of an incident or emergency resulting in exposure to inhalation hazards but also what preparedness steps should be taken before such an event occurs. Recognizing that the SNS is often not immediately available to a locality at the outset of a public health emergency, state and local emergency response plans include stockpiling of PPE and other critical supplies, which also may not be readily available to the public for hours. Therefore, local preparedness plans often include community engagement designed to promote individual and family preparedness and convey the importance of having a disaster supply kit. In response to the COVID-19 pandemic, CDC has expanded the list of items that should be in a disaster kit, which include masks for everyone aged 2 and above (Ready.gov, 2021). It will be important for this new guidance to be reflected in local emergency preparedness communications directed at the public to increase their awareness and early use of respiratory protective devices during events related to infectious diseases, wildfires, and air pollution.
Beyond the significant issues related to education and communication, it is equally important to address issues related to access to information and protective products. The coordinating entity needs to ensure that issues of equity and access, including product cost, availability, and acceptability to a broad range of groups, are considered throughout the entire lifecycle process. It is particularly important to consider access issues among the most vulnerable, and to devote resources to ensuring that inequities in access are prevented or eliminated. Depending on cost, it may be critical for subsidies to be provided so that those who are most vulnerable do not end up receiving information and education but not the protective devices they need.
Addressing Gaps in Responsibilities and Authorities
Multiple federal agencies develop and disseminate guidance for the public on the selection and use of respiratory protection based on the nature of the inhalation hazard at hand. For example, EPA, which regulates environmental pollutants and provides air monitoring data, has produced guidance for public health officials on protecting the public from wildfire smoke, which includes the use of respiratory protective devices (EPA et al., 2019), while CDC has published a multitude of guidance on the public’s use of masks and face coverings (and respirators to a lesser degree) during the COVID-19 pandemic (CDC, 2021). This lack of a central, coordinated body to provide guidance on respiratory protective devices for the public has led to a decentralized and fragmented process for public health recommendations and education, which in turn has resulted in considerable
confusion around selection and use of the devices by the public. The coordinating entity could fill this gap by facilitating a collaborative process for the development of such guidance or harmonizing independently developed guidance. The role of the coordinating entity in ensuring consistent guidance and different potential approaches to structuring stakeholder responsibilities in that effort are described in Box 7-5.
Of the approaches presented in Box 7-5, the committee favors proactive collaborative development of guidance and training over post hoc efforts to harmonize independently developed materials. Given the scale of the undertaking, this task is not well suited to the coordinating entity and would distract from the core functions of the laboratory charged with oversight of standards development and approval for respiratory protective devices intended for public use (Recommendation 7-3). A lead agency with the requisite expertise and communication infrastructure is needed. As the nation’s public health agency, CDC has deep expertise in health communication, as well as in environmental and infectious inhalation hazards. The agency also has the necessary infrastructure and established channels for dissemination, including to state, local, tribal, and territorial public health agencies. Consequently, the committee believes CDC is best positioned to lead the development of guidance and training related to public’s use of respiratory protection.
Specifically, the committee suggests the following considerations:
- CDC should serve as the focal point for assurance that guidance materials are accessible to and appropriate for the general population and its many subgroups. While other federal agencies (e.g., EPA, the Federal Emergency Management Agency) will be involved in developing the guidance, harmonization will be necessary to avoid conflicting information. CDC should develop formalized mechanisms for that purpose.
- In addition to partner federal agencies, CDC should gather input from other key stakeholders, such as local, state, and tribal partners and nongovernmental groups, including medical care providers and health systems, faith leaders, community leaders, and community-based organizations with reach to specific population groups.
- CDC should test the guidance as part of ongoing preparedness activities and periodically following its dissemination to ensure that it is appropriate for diverse population groups and determine whether it needs to be refined as information and contexts change.
- CDC should ensure that mechanisms for reaching critical partners for dissemination, including local, state, territorial, and tribal agencies, as well as community organizations, have been explored and tested, along with routes for direct dissemination to individuals. CDC should map the reach of its communications to ensure that they can reach all segments of the population.
- CDC should partner with medical and public health professionals and societies, as well as device manufacturers and retailers, to develop, test, and disseminate training modules and guidance appropriate for health care and public health professionals and community health workers so they will be prepared to provide guidance to and respond to questions from the public about health risks associated with inhalation hazards and respiratory protective devices.
- If CDC is made aware of the spread of misinformation, it should develop and disseminate improved versions of the guidance to counter such misinformation.
To support the efforts of public health agencies on the ground in responding to the needs of their diverse communities, guidance from CDC should
- be sufficient for incorporation into emergency response plans at the local, state, tribal, territorial, and federal levels;
- be easily tailored for specific populations by local, state, territorial, and tribal agencies and usable by community organizations;
- be tested with diverse end-user groups and periodically over time to determine whether the guidance needs further refinement, particularly as environmental and political contexts change;
- take into account health literacy principles embodied in the National CLAS Standards to increase the information’s acceptability and usability; and
- apply principles in CDC’s Clear Communication Index and the National Academies publication Communicating Science Effectively: A Research Agenda (NASEM, 2017).
To ensure effective and adequate dissemination and implementation, trainings for public health and medical professionals should
- provide continuing education credits;
- be free and easily accessible;
- acknowledge the diverse needs of the American public and potential inequities in the public’s access to health care and respiratory protection; and
- be incorporated into the medical and public health education system where feasible so that future generations have this training as part of their foundational skill set.
Function F5: Incorporate Lifecycle Learning and Continuous Improvement
Function F5 is responsible for ongoing learning and continuous improvement that are critical to catalyzing advances in each of the core functions (F0 through F4) and ensuring the continued robustness of the framework over time to anticipate, prepare for, and respond to evolving needs for respiratory protection for the public. Just as learning and continuous improvement is the underlying operational paradigm for the various steps in the product lifecycle depicted in Figure 5-1 in Chapter 5, Function F5 is responsible for executing the activities associated with lifelong learning and continuous improvement of the framework. Function F5 also serves as the pathway to activating Function F0 (e.g., product design, standards development, conformity assessment, and manufacturing) when the need for a respiratory protective device to protect the public against an inhalation hazard has been identified. The various feedback loops that make up Function F5 and drive continuous improvement are critical because the framework must evolve continuously while anticipating and preparing for future changes. Thus, the ultimate objective of Function F5 is to create a “learning organization” (Garvin, 1993; Senge, 1990) that will address present needs while creating a robust system to anticipate and continuously enhance national preparedness in respiratory protection for the public.
Addressing Gaps in Responsibilities and Authorities
The major activities associated with Function F5 for the public are as follows:
- Promote collaboration among stakeholders in the framework by developing and utilizing appropriate channels of communication in a coordinated and timely manner.
- Define and implement streamlined processes with milestones to monitor the activities in the framework, track progress, and modify the functions as necessary to ensure that their goals are being met.
- Institute a process for capturing learning and experience over time to create a rich knowledge base that can be harnessed by all stakeholders.
- Track and analyze technological advances related to respiratory protection, and work with relevant stakeholders in using these advances to improve existing and create new respiratory protective devices for the public.
- Create a roadmap for the future of respiratory protection for the public, and institute processes for ongoing updating of the roadmap to keep it robust and responsive to evolving inhalation hazards and technological advances.
- Use the roadmap to develop a strategic research agenda for both filling identified gaps and laying the foundation for meeting respiratory protection needs in the future.
Execution of these activities will need to be purposefully driven and reinforced by mechanisms for accountability. No single agency or other federal entity currently holds this responsibility. Addressing this gap will require oversight by the coordinating entity and implementation by those stakeholders with lead roles across the other framework functions, as described in Box 7-6.
Development of the nascent knowledge base related to public use of respiratory protective devices will have to be a focused, expansive science- and policy-driven effort guided by a coordinated research agenda. Research activities related to respiratory protection for the public are currently fragmented, with CDC and EPA leading intra- and extramural research. Other stakeholders include NIH, particularly the National Institute for Environmental Health Sciences (NIEHS), and DHS. NIOSH’s National Occupational Research Agenda (NORA)55 provides a useful model that could be considered in the efforts to establish a research agenda.
___________________
55 Information on NORA is available at https://www.cdc.gov/nora/default.html (accessed December 12, 2021).
- develop and update plans with objective and transparent milestones, and ensure that changes needed to continuously enhance the framework are made;
- regularly assess and publicly report on progress;
- ensure that available funding and its allocation enable goals to be achieved;
- coordinate linkages across partners in government, academia, nonprofits, and industry;
- conduct periodic exercises to evaluate national preparedness for respiratory protection for the public; and
- lead the development of a coordinated, strategic research agenda to develop the knowledge base and address knowledge gaps in respiratory protection for the public.
Function F6: Coordination
The coordinating entity for the public framework will be charged with integrating the efforts of all stakeholders to ensure that the six preceding functions are carried out seamlessly. To this end, the coordinating entity will have an oversight role for each function, as described in Box 7-7.
REFERENCES
Alfano-Sobsey, E., B. Kennedy, F. Beck, B. Combs, W. Kady, S. Ramsey, A. Stockweather, and W. Service. 2006. Evaluation of a model training program for respiratory-protection preparedness at local health departments. Journal of Environmental Health 68(8):38-42.
ASPR (Office of the Assistant Secretary for Preparedness and Response). 2015. PHEMCE mission components. https://www.phe.gov/Preparedness/mcm/phemce/Pages/mission.aspx (accessed September 21, 2021).
Barratt, R., M. Wyer, S. Y. Hor, and G. L. Gilbert. 2020. Medical interns’ reflections on their training in use of personal protective equipment. BMC Medical Education 20(1):328.
Bell, M. 2021. Discussion at the October 6, 2021, public meeting of the National Academies of Sciences, Engineering, and Medicine Committee on Respiratory Protection for the Public and Workers Without Respiratory Protection Programs at Their Workplaces, virtual. https://www.nationalacademies.org/event/10-06-2021/respiratory-protection-for-the-public-and-workers-without-respiratory-protection-programs-at-their-workplaces-meeting-8 (accessed November 9, 2021).
Buchner, K., J. Chuong, C. Paredes-Gomez, Z. Saeed, N. Wilkens, and B. Yang. 2021. Presentation at the January 25, 2021, meeting of the National Academies of Sciences, Engineering, and Medicine Committee on Respiratory Protection for the Public and Workers Without Respiratory Protection Programs at Their Workplaces, virtual.
CDC (Centers for Disease Control and Prevention). 2014. Receiving, distributing, and dispensing Strategic National Stockpile assets: Guide to preparedness, version 11. https://stacks.cdc.gov/view/cdc/77036 (accessed October 7, 2021).
CDC. 2021. Guidance for COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/communication/guidance.html (accessed September 21, 2021).
Chughtai, A., S. Stelzer-Braid, W. Rawlinson, G. Pontivivo, Q. Wang, Y. Pan, D. Zhang, y. Zhang, L. Li, and C. Macintyre. 2019. Contamination by respiratory viruses on outer surface of medical masks used by hospital healthcare workers. BMC Infectious Diseases 19:491.
Clayman, M. L., J. A. Manganello, K. Viswanath, B. W. Hesse, and N. K. Arora. 2010. Providing health messages to Hispanics/Latinos: Understanding the importance of language, trust in health information sources, and media use. Journal of Health Communication 15(Suppl 3):252-263.
Cohen, J., and Y. v. d. M. Rodgers. 2020. Contributing factors to personal protective equipment shortages during the COVID-19 pandemic. Preventive Medicine 141:106263.
Council of Canadian Academies. 2015. Health product risk communication: Is the message getting through? The expert panel on the effectiveness of health product risk communication. Ottawa, ON: Council of Canadian Academies.
CRS (Congressional Research Service). 2020. COVID-19 and domestic PPE production and distribution: Issues and policy options. https://crsreports.congress.gov/product/pdf/R/R46628 (accessed October 8, 2021).
CRS. 2021. The PREP Act and COVID-19: Limiting liability for medical countermeasures. https://crsreports.congress.gov/product/pdf/LSB/LSB10443 (accessed October 7, 2021).
de Perio, M. 2021. Discussion at the October 6, 2021, public meeting of the National Academies of Sciences, Engineering, and Medicine Committee on Respiratory Protection for the Public and Workers Without Respiratory Protection Programs at Their Workplaces, virtual. https://www.nationalacademies.org/event/10-06-2021/respiratory-protection-for-the-public-and-workers-without-respiratory-protection-programs-at-their-workplaces-meeting-8 (accessed November 9, 2021).
Delaney, L. 2021. Discussion at the October 6, 2021, public meeting of the National Academies of Sciences, Engineering, and Medicine Committee on Respiratory Protection for the Public and Workers Without Respiratory Protection Programs at Their Workplaces, virtual. https://www.nationalacademies.org/event/10-06-2021/respiratory-protection-for-the-public-and-workers-without-respiratory-protection-programs-at-their-workplaces-meeting-8 (accessed November 9, 2021).
EPA (Environmental Protection Agency). 2020. Process of reviewing the national ambient air quality standards. https://www.epa.gov/criteria-air-pollutants/process-reviewing-national-ambient-air-quality-standards (accessed September 21, 2021).
EPA, California Air Resources Board, California Office of Environmental Health Hazard Assessment, and CDC (Centers for Disease Control and Prevention). 2019. Wildfire smoke: A guide for public health officials. https://www.airnow.gov/sites/default/files/2021-09/wildfire-smoke-guide_0.pdf (accessed October 5, 2021).
FDA (Food and Drug Administration). 2009. Summary fact sheet for disposable respirators for use during the swine flu emergency. https://www.fda.gov/media/76708/download (accessed October 7, 2021).
Fischer, E. P., M. C. Fischer, D. Grass, I. Henrion, W. S. Warren, and E. Westman. 2020. Low-cost measurement of face mask efficacy for filtering expelled droplets during speech. Science Advances 6(36):eabd3083.
Garritty, C., G. Gartlehner, B. Nussbaumer-Streit, V. J. King, C. Hamel, C. Kamel, L. Affengruber, and A. Stevens. 2021. Cochrane rapid reviews methods group offers evidence-informed guidance to conduct rapid reviews. Journal of Clinical Epidemiology 130:13-22.
Garvin, D. A. 1993. Building a learning organization. Harvard Business Review. https://hbr.org/1993/07/building-a-learning-organization (accessed October 7, 2021).
Gerstein, D. M. 2020. The Strategic National Stockpile and COVID-19: Rethinking the stockpile. Testimony presented before the Senate Committee on Homeland Security and Governmental Affairs on June 24, 2020. RAND Corporation. https://www.rand.org/pubs/testimonies/CTA530-1.html (accessed October 7, 2021).
Gruley, B., and R. Clough. 2020. How 3M plans to make more than a billion masks by end of the year. Bloomberg Businessweek, March 25. https://www.bloomberg.com/news/features/2020-03-25/3m-doubled-production-of-n95-face-masks-to-fight-coronavirus (accessed October 7, 2021).
Harber, P., S. Barnhart, B. A. Boehlecke, W. S. Beckett, T. Gerrity, M. A. McDiarmid, E. Nardbell, L. Repsher, L. Brousseau, T. K. Hodous, and M. J. Utell. 1996. Respiratory protection guidelines. American Journal of Respiratory and Critical Care Medicine 154:1153-1165.
HHS (Department of Health and Human Services), DoD (Department of Defense), DHS (Department of Homeland Security), Department of Commerce, Department of State, VA (Department of Veterans Affairs), and White House Office of the COVID-19 Response. 2021. National strategy for a resilient public health supply chain. https://www.phe.gov/Preparedness/legal/Documents/National-Strategy-for-Resilient-Public-Health-Supply-Chain.pdf (accessed October 7, 2021).
John, A., M. E. Tomas, A. Hari, B. M. Wilson, and C. J. Donskey. 2017. Do medical students receive training in correct use of personal protective equipment? Medical Education Online 22(1):1264125.
Kyung, S. Y., Y. Kim, H. Hwang, J. W. Park, and S. H. Jeong. 2020. Risks of N95 face mask use in subjects with COPD. Respiratory Care 65(5):658-664.
Lakhina, S. F., and B. Lebeda. 2021. Wildfires in a pandemic: What we learned from 2020 and how to plan for what’s next. https://fireadaptednetwork.org/wildfires-in-a-pandemic (accessed October 8, 2021).
Leonesio, M. V., B. Bridges, R. Gesumaria, and L. Del Bene. 2012. The increasing labor force participation of older workers and its effect on the income of the aged. Social Security Bulletin 72(1):1-24.
Mason, D. J., and C. R. Friese. 2020. Protecting health care workers against COVID-19—and being prepared for future pandemics. JAMA Health Forum 1(3):e200353.
McCullough, N. V. 2021. Presentation at the January 26, 2021, meeting of the National Academies of Sciences, Engineering, and Medicine Committee on Respiratory Protection for the Public and Workers Without Respiratory Protection Programs at Their Workplaces, virtual.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016. The nation’s medical countermeasure stockpile: Opportunities to improve the efficiency, effectiveness, and sustainability of the CDC strategic national stockpile: Workshop summary. Washington, DC: The National Academies Press.
NASEM. 2017. Communicating science effectively: A research agenda. Washington, DC: The National Academies Press.
NASEM. 2019. Reusable elastomeric respirators in health care: Considerations for routine and surge use. Washington, DC: The National Academies Press.
NASEM. 2020. Airborne transmission of SARS-CoV-2: A virtual workshop. August 26–27, 2020. https://www.nationalacademies.org/event/08-26-2020/airborne-transmission-of-sars-cov-2-a-virtual-workshop#sectionWebFriendly (accessed October 9, 2020).
OSHA (Occupational Safety and Health Administration). 2011. Small entity compliance guide for the respiratory protection standard. https://www.osha.gov/sites/default/files/publications/3384small-entity-for-respiratory-protection-standard-rev.pdf (accessed October 7, 2021).
Patel, A., M. M. D’Alessandro, K. J. Ireland, W. G. Burel, E. B. Wencil, and S. A. Rasmussen. 2017. Personal protective equipment supply chain: Lessons learned from recent public health emergency responses. Health Security 15(3):244-252.
Ready.gov. 2021. Build a kit. https://www.ready.gov/kit (accessed September 17, 2021).
Regli, A., A. Sommerfield, and B. S. von Ungern-Sternberg. 2021. The role of fit testing N95/FFP2/FFP3 masks: A narrative review. Anaesthesia 76(1):91-100.
Rice, M. B., S. B. Henderson, A. A. Lambert, K. R. Cromar, J. A. Hall, W. E. Cascio, P. G. Smith, B. J. Marsh, S. Coefield, J. R. Balmes, A. Kamal, M. I. Gilmour, C. Carlsten, K. M. Navarro, G. W. Collman, A. Rappold, M. D. Miller, S. L. Stone, and D. L. Costa. 2021. Respiratory impacts of wildland fire smoke: Future challenges and policy opportunities. An official American Thoracic Society workshop report. Annals of the American Thoracic Society 18(6):921-930.
Robinson, R. 2021. Presentation at the July 9, 2021, meeting of the National Academies of Sciences, Engineering, and Medicine Committee on Respiratory Protection for the Public and Workers Without Respiratory Protection Programs at Their Workplaces, virtual.
Samet, J. M., K. Prather, G. Benjamin, S. Lakdawala, J. M. Lowe, A. Reingold, J. Volckens, and L. Marr. 2021. Airborne transmission of SARS-CoV-2: What we know. Clinical Infectious Diseases 73(10):1924-1926. https://doi.org/10.1093/cid/ciab039.
Santana, F. N., D. J. X. Gonzalez, and G. Wong-Parodi. 2021. Psychological factors and social processes influencing wildfire smoke protective behavior: Insights from a case study in northern California. Climate Risk Management 34:100351.
Senge, P. 1990. The fifth discipline: The art & practice of the learning organization. New York: Doubleday/Currency.
Shaffer, R. M. 2021. Environmental health risk assessment in the federal government: A visual overview and a renewed call for coordination. Environmental Science & Technology 55(16):10923-10927.
Stout, S. S., L. A. Simpson, and P. Singh. 2019. Trust between health care and community organizations. Journal of the American Medical Association 322(2):109-110.
Tanne, J. H. 2020. COVID-19: CDC publishes then withdraws information on aerosol transmission. British Medical Journal 370:m3739.
Think Cultural Health. 2018. National culturally and linguistically appropriate services standands. Department of Health and Human Services. https://thinkculturalhealth.hhs.gov/clas/standards (accessed October 7, 2021).
Udow-Phillips, M., and P. M. Lantz. 2020. Trust in public health is essential amid the COVID-19 pandemic. Journal of Hospital Medicine 15(7):431-433.
van der Sande, M., P. Teunis, and R. Sabel. 2008. Professional and home-made face masks reduce exposure to respiratory infections among the general population. PLoS ONE 3(7):e2618.
Watney, C., and A. Stapp. 2020. Masks for all: Using purchase guarantees and targeted deregulation to boost production of essential medical equipment. Mercatus Center Policy Briefs. https://www.mercatus.org/publications/covid-19-crisis-response/masks-all-using-purchase-guarantees-and-targeted-deregulation (accessed October 7, 2021).
This page intentionally left blank.
















































