9
Response Measures for Resilient Medical Product Supply Chains
In a sense, response measures are options of last resort, as they depend on various awareness, mitigation, and preparedness measures and include actions taken postevent to minimize harm from the shortage. However, steps can also be taken before an event to facilitate a more effective response after it occurs. Various institutional measures can be implemented to cultivate a general awareness of emergency preparedness actions, enable the right people to talk to one another in a productive manner, provide resources for adapting to medical product shortages, and facilitate steps for reducing harm to people from the shortages while they persist.
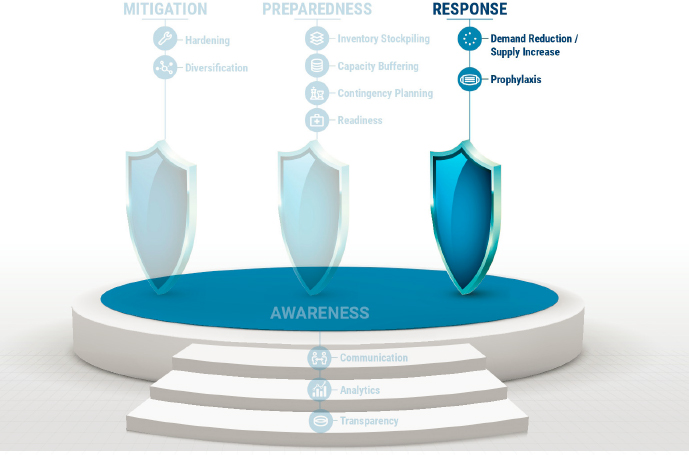
As described in Chapter 5 and in the medical product supply chains resilience framework (reproduced in Figure 9-1), response measures include (1) reducing the demand or increasing the supply, and (2) prophylaxis measures, which protect human health while the shortage persists. While international cooperation and coordinated response can help minimize the effect of medical product shortages globally, response measures by front-line clinicians and other medical product end users are essential when mitigation and preparedness measures fail to protect patients from supply chain failures. To address these, this chapter focuses on both the global level, by addressing issues raised by international production of medical products, and the local level, by addressing last-mile issues in medical product delivery. As discussed in Chapter 3, the globalization of the U.S. medical product supply chains brings substantial benefits to American consumers and producers in the form of lower costs, greater access to a diversity of medical products, increased efficiencies, and more innovation. However, the globalization of medical product supply chains also comes with costs and risks, particularly
during emergency conditions. These were cast into sharp relief during the coronavirus disease (COVID-19) pandemic, when access to products that are predominantly manufactured abroad was disrupted. The committee concluded in Chapter 3 that market forces create powerful incentives for medical product supply chains to remain globalized. These global supply chains provide efficiency, innovation, and, in some cases, diversification benefits. However, such global supply chains also pose transparency and coordination challenges. International cooperation can help address these challenges, strengthen the resilience of medical product supply chains, and minimize the effect of shortages. To achieve this, nations and manufacturers must be better equipped to understand and manage the challenges of global medical product supply chains, including issues related to transparency, regulatory authorities, and national security.
Having the ability to implement response measures and adapt to a current disaster situation or shortage until supply chains have returned to normal is critical to protecting public health and human life. Indeed, it is common for the most difficult and complex part of supply chains to exist in the final delivery and distribution of medical products to end users, so it is logical that disruptions to medical product supply chains often arise in, or are worsened by, problems in the last mile of distribution. This is especially problematic during chaotic emergency situations, such as that experienced during the recent pandemic. Therefore, to protect public health it is vital to be ready to manage this final stage properly in an emergency.
RESPONSE MEASURES FOR GLOBAL MEDICAL PRODUCT SUPPLY CHAINS
In response to the COVID-19 pandemic, several countries, including the United States, implemented measures that restricted exports of certain medical products, including personal protection equipment (PPE) such as face masks and shields, pharmaceuticals, and medical equipment, such as ventilators. According to a 2020 World Trade Organization (WTO) report, by May 2020, 85 countries and separate customs territories had introduced pandemic-related trade restrictions, such as export bans (WTO, 2020). Roughly half of these measures did not include a specific duration for the trade restriction. While many of the restrictions have since been lifted, as of August 2021, more than 45 countries still have over 70 export restrictions in place, many of which apply to medical products (CRS, 2021). Domestically, U.S. agencies (e.g., U.S. Department of Health and Human Services [HHS], Federal Emergency Management Agency [FEMA]) began using the Defense Production Act (DPA) to place priority ratings on medical supply contracts for agency orders to receive preference over others. Use of the DPA also provided medical supply manufacturers with incentives to expand domestic production of medical supplies to reduce the United States’ dependence on foreign sources of supply (GAO, 2020).
WTO rules generally ban export prohibitions and restrictions (WTO, 1994a). However, there are exceptions that allow WTO members to temporarily impose measures “to prevent or relieve critical shortages” of essential products, such as food and medical products, or to protect human, animal, or plant life or health (WTO, 1994a, Article XI). These exceptions give member countries the flexibility to impose trade restrictions in response to health emergencies or national security threats (WTO, 1994a, Articles XI, XX).
In a joint statement, WTO and the International Monetary Fund raised concerns about the use of export restrictions on food and medical products in response to the COVID-19 pandemic:
Taken collectively, export restrictions can be dangerously counterproductive. What makes sense in an isolated emergency can be severely damaging in a global crisis. Such measures disrupt supply chains, depress production, and misdirect scarce, critical products and workers away from where they are most needed. Other governments counter with their own restrictions. The result is to prolong and exacerbate the health and economic crisis — with the most serious effects likely on the poorer and more vulnerable countries (IMF and WTO, 2020).
The committee concludes that export restrictions may temporarily increase the availability of domestic supplies, but there are short- and
long-term costs associated with such measures. Such measures disrupt supply chains, depress production, and misdirect scarce, critical products and workers away from where they are most needed.
For example, in the European Union (EU), export bans on PPE coupled with demand surges early on in the pandemic led to shortages in non-EU countries in February and March 2020, resulting in hampered effective medical care in these countries (Bown, 2020; Keynes, 2020). Export bans can also have a long-term effect on global supply chains. For example, in response to the 2007-2008 global food crisis, temporary export bans and other trade restrictions implemented by major rice exporters contributed to record high global rice prices, which exacerbated hoarding, panic buying, food riots, and a breakdown of food supply chains (FAO, 2020).
The Need for International Cooperation
International cooperation can help minimize the effect of medical product shortages and strengthen supply chain resilience. The 1994 Agreement on Trade in Pharmaceutical Products (known as the Pharmaceutical Agreement) eliminates tariffs and other duties and charges on a number of pharmaceutical products and the substances used to produce them, permanently binding them at duty-free levels (WTO, 1994b). Canada, the EU, Japan, Norway, China, Switzerland, the United Kingdom, and the United States currently participate in this agreement (Hallak, 2020). Emerging markets, including Brazil, China, India, and Mexico, have increased imports along with their purchasing power (Donoghoe et al., 2012; Mariadoss, 2018). These countries have benefited from zero duties when importing from signatories of the Pharmaceutical Agreement. While this agreement has helped enable the growth of international trade in pharmaceutical products (European Commission, 2020a; Sunesen et al., 2016), additional cooperation is needed to curb export prohibitions or restrictions on medical products during public health crises.
Proposals to restrict export prohibitions and restrictions have been discussed among like-minded countries. In response to the ongoing trade response to the COVID-19 pandemic, a group of WTO members (Australia, Brazil, Canada, Chile, the EU, Japan, Kenya, South Korea, Mexico, New Zealand, Norway, Singapore, and Switzerland) called for restraint on the use of export restrictions, implementation of trade-facilitating measures, and improved transparency (European Commission, 2020b). This proposal was circulated among WTO members for consideration in May 2021 as a draft declaration, “COVID-19 and Beyond: Trade and Health” (WTO, 2021). In June 2021, the EU submitted a WTO proposal for a multilateral trade response to the COVID-19 pandemic. Among other commitments, the proposal called on WTO members to “subscribe to the G20 commitment to
ensure that any export restrictive measures relating to health products are targeted, transparent, proportionate, and temporary, and consistent with WTO obligations” (European Union, 2021).
Industry trade associations and trade policy experts have voiced their support for such proposals. On the industry side there is the Association for Accessible Medicines (AAM), a trade association representing manufacturers and distributors of generic pharmaceutical products; it has called for an international pharmaceutical supply chain agreement:
To promote the benefits of a globally diverse supply chain, the United States Trade Representative, working with the U.S. Department of Health and Human Services (HHS), should negotiate a plurilateral1 agreement with U.S. allies to promote a cooperative approach to securing the U.S. supply chain, ensuring diversity of supply and responding to global health care challenges and natural disasters, without resorting to export controls or other trade barriers. In addition, coordinating the expansion of pharmaceutical manufacturing with U.S. allies will allow for economies of scale and a coordinated approach to global pandemics. Possible signatories would include U.S. allies such as Canada, Europe, India, Israel, Japan, Jordan, and Mexico. (AAM, 2020)
Trade experts have argued for proactive international cooperation to boost global surge capacity for PPE, encourage supply chain transparency, and prevent knee-jerk export restrictions in response to public health emergencies (Bown, 2021).
As discussed in Chapter 6, publicly available medical product supply chain data would enable stakeholders to proactively detect and respond to supply chain disruptions, improve coordination on a national and global level, and enhance situational awareness. Furthermore, previous National Academies’ work acknowledges how information sharing among regulatory authorities is critical to ensure that patients and health care providers receive quality-assured, safe, and effective medical products (see Box 9-1) (IOM, 2013).
The health and well-being of the U.S. population is inextricably linked to the health and well-being of populations in other countries around the world. Public health crises, such as the current COVID-19 pandemic, are not restricted by country borders. As described in previous chapters, market forces, stockpiling, and on-shoring may resolve some barriers, but they are not a one-size-fits-all solution. For example, short-term medical product shortages may be managed through stockpiling, but this may not address the need for longer-term production capacity (Finkenstadt et al., 2020). Shifting
___________________
1 WTO member countries are given the choice to agree to new rules in a plurilateral agreement on a voluntary basis (WTO, n.d.).
toward more domestic manufacturing capabilities may offer some relief for aspects of medical product supply chain vulnerabilities, but would likely be insufficient to meet the demand if and when the next pandemic arises. Furthermore, it would be unrealistic for the United States to shift to a fully self-reliant domestic medical product supply chain given the significant time, cost, and complexity of drug and device inputs and manufacturing. For these reasons, international cooperation to faciliate global trade in medical products remains essential for maintaining the health and security of the United States.
Establishing an International Trade Agreement
To address the need for international cooperation, inside and outside of public health crises, there should be agreement among the major exporters of medical products to refrain from export bans or other interventions that would fragment or limit the global supply response, and to cooperate to avoid excessive concentration of key products or components in a single location or from a single producer. Such an agreement would also serve to strengthen supply chain resilience in the absence of a public health emergency by facilitating the manufacturing and distibution of lifesaving drugs and devices.
To ensure cooperation, this agreement would have to be binding and should include reputational and legal sanctions. Any country that violates the terms of the agreement would be subject to sanctions by other signatories of the agreement. Providing incentives for countries to uphold commitments and cooperate in the event of another public health crisis and disincentives for violating the terms of the agreement would help ensure that the treaty is self-enforcing.
Chapter 6 highlights the benefits of information sharing in supply chain management and provides recommendations for U.S. government actions to collect and publicly share data. Given the globalization of U.S. medical supply chains, information sharing across international borders will also be essential for stakeholders—including government and the private sector—to mount effective response measures to future supply chain disruptions. Therefore, in addition to constraining export bans in the face of global health emergencies, negotiators of this treaty could consider adding provisions to facilitate the sharing of relevant information regarding global supply chains, such as information on demand, inventory, capacity, and supply.
Trade policy experts have suggested that another important component of effective international policy coordination is a mechanism to enable U.S. investment in the production capacity of suppliers located abroad (Bown and Bollyky, 2021). While the DPA enabled the United States to invest in nation-wide coordinated expansion of medical product production capicity during the COVID-19 pandemic (GAO, 2020), similar investments in global manufacturing in response to future public health emergencies would benefit the United States as well as other countries. Such a concept merits
further exploration and could be considered as an added provision within an international trade agreement.
It is important to recognize that multilateral trade agreements are complex, difficult to negotiate, and often take years to implement (Moser and Rose, 2012). Recent analysis finds that negotiations of trade agreements may take additional time when the United States, the EU, or Japan is a signatory (Arroyo and Castillo-Ponce, 2019; Moser and Rose, 2012). Additionally, the effect of the COVID-19 pandemic on international trade as described above coupled with the recent rise in nationalist economic policies on the part of advanced and emerging-market economies may make it more challenging for countries to pursue multilateral agreements, which depend on global cooperation.
Given the duration and considerable negotiations required to establish multilateral agreements within WTO, one approach for advancing this concept could be for a subset of WTO members comprising the major exporters of medical products—United States, EU, China, and India—to negotiate a plurilateral agreement (Statista Research Department, 2016). For example, the Information Technology Agreement, which eleminated tariffs on information technology products covered by the agreement, was first negotiated in 1996 and included 29 participants. As of October 2021 there were 82 participants in the Information Technology Agreement, and in December 2015, over 50 members expanded the agreement to cover an additional 201 products (WTO, n.d.).
The U.S. government must take on the task of better managing and reducing these risks, while maximizing the benefits of globalization. Recommendation 6 (international treaty) advocates a plurilateral agreement by major exporters of medical products, including the United States, under the WTO that prohibits export bans on components of critical medical products. Although such an agreement cannot prevent a worldwide shortage from occurring, it can limit the risk to any individual country by “spreading the pain” across the global economy. Furthermore, if such an agreement increases the collective trust in global supply chains during an emergency, then that can be used to strengthen Recommendation 5 (Capacity Buffering). It is almost certainly more cost-efficient to build virtual capacity globally than locally. Hence, using the treaty as the basis for collaboration, major medical product exporting countries could further cooperate on providing capacity buffering for medical products likely to be in short supply during global emergencies.
RESPONSE MEASURES FOR LAST-MILE DELIVERY AND END USERS
At a global level, supply chains focus on matching total supply to total demand. The awareness, mitigation, and preparedness recommendations
of this report are predominantly aimed at enabling medical product supply chains to accomplish this in normal and emergency conditions. At the local level—often termed the “last mile”—supply chains focus on getting products to the individuals who need them. As the COVID-19 pandemic highlighted, supply shortages, whether the result of global disruptions or local imbalances such as hoarding or misallocation, present enormous challenges to end users, including public health workers, health care providers, and pharmacists. In the chaos of a crisis, these key personnel may not know what supplies exist or when they are coming. They may lack clear guidelines on how to allocate limited supplies to those who need them most. And, they may be unaware of best practices for protecting health in the face of medical supply shortages.
As described in Chapter 5, response measures can either close the gap between supply and demand or protect people from harm due to the gap (i.e., via prophylaxis measures). Both of these can be addressed by those managing the last mile of medical product supply chains. For example, an effective response by hospitals and clinics across the United States to reduce the serious shortage of N95 masks was to reuse masks with multiple patients (CDC, 2021; Chaudhuri, 2020; Hufford, 2020). Although reuse entailed some risk, it allowed the American health care system to function while dramatically reducing the required number of N95 masks. This provided a large and necessary “cushion” while global supply chains struggled to increase production. As this example illustrates, end users of medical supplies such as hospitals, clinicians, pharmacies, and patients have important roles to play in addressing disruptions to medical product supply chains. Although workarounds like this do not solve the larger supply shortage, they can alleviate the impact of shortages and provide much needed time for supply chains to catch up.
Protecting patient health through careful management of the last mile so that much needed medical products reach those who most need them, the end users need information, evidence-based best practices, and training. This section discusses ways to provide these types of last-mile support through resource sharing, development of a national framework for scarce medical product allocation, and the engagement of end users in planning and training for medical product shortages.
Mechanisms to Increase the Sharing of Critical Supplies
Resource sharing is a fundamental way to ensure that limited medical supplies go to those who need them most. This involves transferring supplies from one locality, state, or region to another. A key prerequisite for resource sharing is the transparency of critical supply availability across organizations (Devereaux et al., 2020).
Other key elements of a resource sharing system are mechanisms that facilitate (or require) the transfer of supplies and other resources when necessary. These can be established through the collaboration of local, state, and federal public health officials and clinicians, as well as public–private partnerships. For example, during the COVID-19 pandemic, regions developed innovative systems such as transfer centers to ensure patients and supplies were optimally matched (NRCC Healthcare Resilience Task Force and FEMA, 2020; Valin et al., 2020), but these systems typically required a voluntary willingness to share information with competitors about the presence or absence of critical supplies. Early in the COVID-19 pandemic such voluntary cooperation was relatively common, but in many less dire supply shortages it is not.
Recent experience suggests that regional and national multidisciplinary teams that proactively plan ahead for alternatives and prepare recommendations for substitution or conservation strategies prior to supplies becoming exhausted are more robust than single-center efforts (Devereaux et al., 2020; Tosh and Burry, 2020). When each facility creates its own strategies for responding to shortages, and when shortages are unevenly distributed among facilities, there is a significant risk of inequitable effects. For instance, in the last few decades, health care coalitions (HCCs) have become core components of regional emergency and disaster response, facilitating transparency about resource shortages and the sharing of scarce resources (Barnett et al., 2020). Additionally, some well-formed and well-organized HCCs play fundamental roles in the health of their communities during nonemergency times. HCCs like this could also play a role in developing standardized plans for responding to routine medical product shortages, including strategies for moving resources to where they are most needed, to help balance the disparate effects of shortages on different facilities. Indeed, HHS’s Office of the Assistant Secretary for Preparedness and Response (ASPR) has already created several regional disaster response coalitions, which provide important opportunities to explore the possibility of sharing disaster response capabilities across multiple states (ASPR, 2018). Regional and national dashboards that include drug quantities available could provide valuable information when drug shortages arise and can facilitate moving drugs to areas of greatest need (Tosh and Burry, 2020).
A National Framework for Scarce Medical Product Allocation
Local resource sharing can work if the end users know and trust one another, but sharing resources on a larger scale, where personal relationships cannot be relied on, is extremely challenging. Even deciding who is most deserving of a scarce medical product presents a difficult question. Clinical need, age, equity, and a wide range of other factors can enter into
this question. The COVID-19 pandemic highlighted an alarming number of inequities and challenges in the resource allocation process.
The allocation problem is well known to clinicians and has been the subject of a great deal of work at the National Academies of Science, Engineering, and Medicine (the National Academies) (NASEM, 2020a, 2021). However, even with this body of research, health care and public health officials were left without clear guidance on the best frameworks to use when allocating scarce medical products, resulting in inconsistent, and at times inequitable, use between and within states and within health care systems. For example, some implemented the Centers for Disease Control and Prevention’s recommended Social Vulnerability Index and Area Deprivation Index frameworks (Ndugga et al., 2021; Schmidt et al., 2021) while others used different approaches for allocating medial products like the Fair Priority Model (Emanuel et al., 2020), all without a clear consensus on whether one worked better or more effectively than the other. It may also be that different frameworks are needed for different medical products.
Given the variety of frameworks to choose from and the lack of guidance around which one to use and when, it is clear that a standard ethical framework for scarce medical product allocation is needed at the national level if outcomes like those seen during COVID-19 are to be avoided. Such a framework must be designed in a way that is acceptable to all major actors—distributors, producers, end users—without undue burden placed on any one entity, and it must be flexible enough to provide guidance for decision making and deliberation within and across health sectors, health institutions, and health professionals in response to medical product shortages (Gibson et al., 2012). It must reconcile the actors’ competing values and be accepted as ethical, not just seen as an assertion of power (Emanuel et al., 2020). In an ideal setting, use of this common ethical framework for scarce medical product allocation would have a cascading effect: producing less product waste would increase producer confidence that medical products will be fairly allocated to benefit people, which would in turn motivate an increase in production for continued distribution (Emanuel et al., 2020).
Furthermore, because of the vital role local health care professionals play in enabling the efficient delivery of medical products to the end user, it is essential that they be involved in the development of allocation frameworks. They can offer perspectives on how the framework would or would not be useful based on their clinical experiences (Gibson et al., 2012). Although training for disaster response preparedness is common for health care professionals, it is less common for it to include instruction regarding their potential roles in addressing population health concerns, such as health disparities, health inequities, and hesitancy receiving medical treatment (Aruru et al., 2021; Ducatman et al., 2020; Jacobsen et al., 2020; Johnson et al., 2020; Thibault, 2020). These elements should be included in trainings to address
inequitable resource distribution during supply shortages so medical professionals can more effectively treat and advocate for their patients.
Engaging End Users in Last-Mile Planning
There is a close relationship between the virtual preparedness measures (Chapter 8) and response measures. Contingency planning and readiness activities taken prior to a disruptive event can improve the speed and accuracy of response activities. For instance, a contingency plan that identifies supplemental producers for specific supply chain critical medical products and readiness steps, such as sharing supplier and bill-of-material information with these producers, would make ramping up capacity in an emergency faster and more reliable.
Yet beyond a narrow role in devising a framework for scarce medical product allocation, end users must be involved more broadly in planning for medical product shortages. The reason for this is that, in order to respond more effectively to the next public health crisis, end users need a number of institutional capabilities, particularly those related to managing medical product shortages. These include clearly defined roles, standardized crisis standards and contingency operations, better communication channels, standards of practice that provide documentation of such things, and training of medical personnel to equip them to act more effectively when they are without the standard level of routine supplies. An end-user perspective is absolutely vital to develop guidelines and tools for building these capabilities.
An essential element of last-mile planning is clearly articulating who has authority and responsibility for coordinating health care systems and suppliers, particularly during an emergency or crisis situation. As discussed in Chapter 8, the National Academies has explored and developed systems for ethically addressing circumstances where usual care is unattainable due to resource shortages and crisis standards of care (CSC) are required (IOM, 2009), including resources and tools for implementing and executing CSC. A group, or appropriate set of teams, needs to have command and control responsibility for the last-mile response system when there is a medical product shortage and resource allocation is unbalanced. The recent development of ASPR-funded regional disaster response systems serving multiple states in geographic proximity presents opportunities for the exploration of systems to improve cross-state sharing of information on supply levels and resources (ASPR, 2021). This could help alleviate implicit rationing and hoarding of medical products during a crisis. Additional resources on CSC can be found in Chapter 8 in Box 8-4.
Building the institutional capabilities to support last-mile, contingency, and crisis planning will require extensive training. Because of their knowl-
edge of both the institutions and the challenges of managing the last mile, health care professionals must be involved in the development and execution of training to prepare for medical product shortages. Although such resources are limited at present, there is an emerging trend in health professional education toward teaching students and trainees about the importance of medical ethics during emergency situations, including medical product shortages (Aruru et al., 2021; Ducatman et al., 2020; Jacobsen et al., 2020; Johnson et al., 2020; Thibault, 2020). This trend should be encouraged and could be used to improve workforce readiness and coordination to address supply shortages and problems of inequitable resource distribution.
Practicing clinicians and the leaders of health care delivery organizations can and should be encouraged to participate in planning for disruptions of medical product supply chains. This can be achieved in part by recognizing that supply chain disruptions, unlike pandemics and most other public health emergencies, are remarkably common. In fact, many health systems operate in a state of chronic contingency, with various supplies routinely being conserved, reused, and repurposed due to persistent shortages of critical resources (NASEM, 2018). However, these chronic contingency situations can provide health systems with opportunities to practice using CSC frameworks during less severe shortages and at much lower risk and cost to their operations and patient lives. As such, there is an ongoing need to establish platforms to facilitate communication during disruptions and platforms for sharing best practices among clinical end users facing supply shortages.
RECOMMENDATIONS
A critical component of supply chain resilience is the ability to recover from disruptions quickly and effectively. While disruptions at the global and end-user levels appear disparate and can be complex to manage, the key to the solution for both sets of problems is effective communication and cooperation. The specific and actionable recommendations listed below are intended to help stakeholders better manage and reduce the risks associated with the globalization of medical product supply chains and disruptions at the last mile of delivery. Recommendation 6 (International Treaty) does this at the global level by promoting open communication and cooperative efforts by medical exporting nations. Recommendation 7 (Last-Mile Management) does this at the local level by establishing a working group to detail and develop the tools needed by medical professionals to manage medical product shortages.
Recommendation 6 (International Treaty). Major exporters of medical products, including the United States, should negotiate a plurilateral
treaty under the World Trade Organization that prohibits export bans and restrictions on key components of global medical product supply chains. Any country that violates the terms of this agreement should be subject to sanctions by other signatories of the agreement. Specifically,
- The treaty should provide incentives for countries to uphold commitments and cooperate in the event of a public health crisis.
- The treaty should provide disincentives or sanctions, such as reputational, economic, and legal sanctions, for violating the terms of the agreement.
- Treaty negotiators could consider adding provisions to this treaty that facilitate information sharing, particularly during medical emergencies.
Recommendation 7 (Last-Mile Management). The Office of the Assistant Secretary for Preparedness and Response, in collaboration with the Centers for Disease Control and Prevention, should convene a working group of key stakeholders to examine and identify effective last-mile strategies to ensure end users are able to respond in the event of medical product shortages. The working group should
- Determine what information needs to be shared, with whom and in what form, in order for end users to be able to execute resource sharing, supply redistribution, substitution, adaptation, and other strategies for responding to medical product shortages at the local level.
- Develop a standard national ethical framework for allocating scarce medical products, building in previous crisis standards of care work, including attention to equity, efficiency, and additional ethical values.
- Develop and incorporate response plans and training for medical product shortages into public health and health care professional capabilities.
REFERENCES
AAM (Association for Accessible Medicines). 2020. A blueprint for enhancing the security of the U.S. pharmaceutical supply chain (2nd edition). https://accessiblemeds.org/sites/default/files/2020-04/AAM-Blueprint-US-Pharma-Supply-Chain.pdf (accessed October 20, 2021).
Arroyo, L., and R. A. Castillo-Ponce. 2019. The duration of trade agreement negotiations. Applied Econometrics and International Development 19(2):19-36.
Aruru, M., H.-A. Truong, and S. Clark. 2021. Pharmacy emergency preparedness and response (PEPR): A proposed framework for expanding pharmacy professionals’ roles and contributions to emergency preparedness and response during the COVID-19 pandemic and beyond. Research in Social and Administrative Pharmacy 17(1):1967-1977.
ASPR (Assistant Secretary for Preparedness and Response). 2018. HHS selects pilot projects to demonstrate better approach to disaster medical care. HHS (U.S. Department of Health and Human Services). https://public3.pagefreezer.com/browse/HHS.gov/31-12-2020T08:51/https://www.hhs.gov/about/news/2018/09/27/hhs-selects-pilot-projects-demonstrate-better-approach-disaster-medical-care.html (accessed October 21, 2021).
ASPR. 2021. Partnership for disaster health response cooperative agreement. HHS. https://www.phe.gov/Preparedness/planning/PDHRCA-FOA/Pages/default.aspx (accessed February 17, 2021).
Barnett, D. J., L. Knieser, N. A. Errett, A. J. Rosenblum, M. Seshamani, and T. D. Kirsch. 2020. Reexamining health-care coalitions in light of COVID-19. Disaster Medicine and Public Health Preparedness 1-5.
Bown, C. 2020. E.U. limits on medical gear exports put poor countries and Europeans at risk. In Trade and investment policy watch. Peterson Institute for International Economics. https://www.piie.com/blogs/trade-and-investment-policy-watch/eu-limits-medical-gear-exports-put-poor-countries-and (accessed December 20, 2021).
Bown, C., and T. Bollyky. 2021. The world needs a COVID-19 vaccine investment and trade agreement. In Trade and investment policy watch. Peterson Institute for International Economics. https://www.piie.com/blogs/trade-and-investment-policy-watch/world-needs-covid-19-vaccine-investment-and-trade-agreement (accessed November 10, 2021).
Bown, C. P. 2021. How COVID 19 medical supply shortages led to extraordinary trade and industrial policy. Asian Economic Policy Review 17:114-135. https://doi.org/10.1111/aepr.12359.
CDC (Centers for Disease Control and Prevention). 2021. Strategies for optimizing the supply of N95 respirators. https://www.cdc.gov/coronavirus/2019-ncov/hcp/respirators-strategy/index.html (accessed October 26, 2021).
Chaudhuri, S. 2020. Coronavirus prompts hospitals to find ways to reuse masks amid shortage. The Wall Street Journal, March 31.
CRS (Congressional Research Service). 2021. Export restrictions in response to the COVID-19 pandemic. CRS. https://sgp.fas.org/crs/natsec/IF11551.pdf (accessed December 20, 2021).
Devereaux, A., H. Yang, G. Seda, V. Sankar, R. C. Maves, N. Karanjia, J. S. Parrish, C. Rosenberg, P. Goodman-Crews, L. Cederquist, F. M. Burkle, J. Tuteur, C. Leroy, and K. L. Koenig. 2020. Optimizing scarce resource allocation during COVID-19: Rapid creation of a regional health-care coalition and triage teams in San Diego County, California. Disaster Medicine and Public Health Preparedness 1-7.
Donoghoe, N., A. Gupta, R. Linden, P. Mitra, and I. Beyer von Morgenstern. 2012. Medical device growth in emerging markets: Lessons from other industries. In Vivo: The Business and Medicine Report, June 2012.
Ducatman, B. S., A. M. Ducatman, J. M. Crawford, M. Laposata, and F. Sanfilippo. 2020. The value proposition for pathologists: A population health approach. Academic Pathology 7:2374289519898857.
Emanuel, E. J., G. Persad, A. Kern, A. Buchanan, C. Fabre, D. Halliday, J. Heath, L. Herzog, R. J. Leland, E. T. Lemango, F. Luna, M. S. McCoy, O. F. Norheim, T. Ottersen, G. O. Schaefer, K. C. Tan, C. H. Wellman, J. Wolff, and H. S. Richardson. 2020. An ethical framework for global vaccine allocation. Science 369(6509):1309-1312.
European Commission. 2020a. Communication from the European Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions: Pharmaceutical strategy for Europe. Brussels: European Commission. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:52020DC0761 (accessed December 20, 2021).
European Commission. 2020b. Ottawa group proposes a global trade and health initiative. Brussels. https://trade.ec.europa.eu/doclib/press/index.cfm?id=2215 (accessed December 20, 2021).
European Union. 2021. Communication from the European Union to the WTO general council: Urgent trade policy responses to the COVID-19 crisis. Brussels. https://trade.ec.europa.eu/doclib/docs/2021/june/tradoc_159605.pdf (accessed December 20, 2021).
Finkenstadt, D., R. Handfield, and P. Guinto. 2020. Why the U.S. still has a severe shortage of medical supplies. Harvard Business Review, September–October 2020, September 17,.
FAO (Food and Agriculture Organization of the United Nations). 2020. Why export restrictions should not be a response to COVID-19: Learning lessons from experience with rice in Asia and the Pacific. https://www.fao.org/3/ca9362en/CA9362EN.pdf (accessed October 20, 2021).
GAO (Government Accountability Office). 2020. Defense Production Act: Opportunities exist to increase transparency and identify future actions to mitigate medical supply chain issues. https://www.gao.gov/assets/gao-21-108.pdf (accessed October 12, 2021).
Gibson, J. L., S. Bean, P. Chidwick, D. Godkin, R. W. Sibbald, and F. Wagner. 2012. Ethical framework for resource allocation during a drug supply shortage. Healthcare Quarterly (Toronto, ON) 15(3):26-35.
Hallak, I. 2020. EU imports and exports of medical equipment. European Parliamentary Research Service. https://www.europarl.europa.eu/RegData/etudes/BRIE/2020/649387/EPRS_BRI(2020)649387_EN.pdf (accessed December 20, 2021).
Hufford, A. 2020. Why are N95 masks so important? The Wall Street Journal, June 1.
IMF and WTO (International Monetary Fund and World Trade Organization). 2020. IMF and WTO heads call for lifting trade restrictions on medical supplies and food: World Trade Organization. https://www.wto.org/english/news_e/news20_e/igo_15apr20_e.htm (accessed December 20, 2021).
IOM (Institute of Medicine). 2009. Guidance for establishing crisis standards of care for use in disaster situations: A letter report. Washington, DC: The National Academies Press.
IOM. 2013. International regulatory harmonization amid globalization of drug development: Workshop summary. Washington, DC: The National Academies Press.
Jacobsen, K., M. Hay, J. Manske, and C. Waggett. 2020. Curricular models and learning objectives for undergraduate minors in global health. Annals of Global Health 86(1):102.
Johnson, S. B., M. A. Fair, L. D. Howley, J. Prunuske, S. B. Cashman, J. K. Carney, Y. S. Jarris, L. R. Deyton, D. Blumenthal, N. K. Krane, N. H. Fiebach, A. H. Strelnick, E. MortonEggleston, C. Nickens, and L. Ortega. 2020. Teaching public and population health in medical education: An evaluation framework. Academic Medicine 95(12):1853-1863.
Keynes, S. 2020. New trade barriers could hamper the supply of masks and medicines. The Economist, March 7-13 2020, March 11.
Mariadoss, B. J. 2018. Chapter 5: Emerging markets. In Core principles of international marketing. Vancouver, WA: Washington State University. https://opentext.wsu.edu/cpim/chapter/5-5-emerging-markets (accessed November 8, 2021).
Moser, C. and A. K. Rose. 2012. Why do trade negotiations take so long? KOF Working Papers 295. Zurich: KOF Swiss Economic Institute. http://dx.doi.org/10.3929/ethz-a-006844132.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2018. Medical product shortages during disasters: Opportunities to predict, prevent, and respond: Proceedings of a workshop—in brief. Washington, DC: The National Academies Press. doi: https://doi.org/10.17226/25267.
NASEM. 2020a. A framework for equitable allocation of COVID-19 vaccine. In Framework for equitable allocation of COVID-19 vaccine. Washington, DC: The National Academies Press. Pp. 89-144.
NASEM. 2020b. Regulating medicines in a globalized world: The need for increased reliance among regulators. Washington, DC: The National Academies Press. https://doi.org/10.17226/25594.
NASEM. 2021. Rapid expert consultation on allocating COVID-19 monoclonal antibody therapies and other novel therapeutics (January 29, 2021), edited by L. Brown, A. Downey, S. Wollek, C. Shore, E. Fine, and B. Kahn. Washington, DC: The National Academies Press.
Ndugga, N., S. Artiga, and O. Pham. 2021. How are states addressing racial equity in COVID-19 vaccine efforts? Kaiser Family Foundation. https://www.kff.org/racialequity-and-health-policy/issue-brief/how-are-states-addressing-racial-equity-in-covid-19-vaccine-efforts/ (accessed August 27, 2021).
NRCC Healthcare Resilience Task Force, and the Federal Emergency Management Agency (FEMA). 2020. Medical operations coordination cells toolkit: Department of Homeland Security (DHS). https://files.asprtracie.hhs.gov/documents/fema-mocc-toolkit.pdf (accessed October 14, 2021).
Schmidt, H., R. Weintraub, M. A. Williams, K. Miller, A. Buttenheim, E. Sadecki, H. Wu, A. Doiphode, N. Nagpal, L. O. Gostin, and A. A. Shen. 2021. Equitable allocation of COVID-19 vaccines in the United States. Nature Medicine 27(7):1298-1307.
Statista Research Department. 2016. Main medical device exporters worldwide in 2015, by country, edited by Statista. https://www.statista.com/statistics/619607/medical-device-exporters-worldwide-by-country/ (accessed Novemebr 10, 2021).
Sunesen, E., T. Jeppesen, and M. H. Thelle. 2016. How a strong pharma chapter in TTIP will benefit the EU. Copenhagen Economics. https://www.efpia.eu/media/25874/how_a_strong_pharmaceutical_chapter_in_ttip_will_benefit_the_eu_policy_brief.pdf (accessed Novemebr 8, 2021).
Thibault, G. E. 2020. The future of health professions education: Emerging trends in the United States. Federation of American Societies for Experimental Biology (FASEB) Bio-Advances 2(12):685-694.
Tosh, P. K., and L. Burry. 2020. Essential institutional supply chain management in the setting of COVID-19 pandemic. The American College of Chest Physicians (CHEST). https://www.chestnet.org/resources/essential-institutional-supply-chain-management-in-the-setting-of-covid-19-pandemic (accessed October 14, 2021).
Valin, J. P., S. Gulley, B. Keidan, K. Perkins, C. Savor Price, W. Neff, G. Winfield, and D. Tad-y. 2020. Physician executives guide a successful COVID-19 response in Colorado. New England Journal of Medicine Catalyst Innovations in Care Delivery 1(5).
WTO (World Trade Organization). 1994a. General agreement on tariffs and trade 1994. Geneva, Switzerland: WTO. https://www.wto.org/english/docs_e/legal_e/06-gatt_e.htm (accessed December 8, 2021).
WTO. 1994b. The WTO’s pharma agreement. In Trade topics. The World Trade Organization. https://www.wto.org/english/tratop_e/pharma_ag_e/pharma_agreement_e.htm (accessed October 20, 2021).
WTO. 2020. Report on G20 trade measures (mid-October 2019 to mid-May 2020). WTO. https://www.wto.org/english/news_e/news20_e/report_trdev_jun20_e.pdf (accessed October 20, 2021).
WTO. 2021. COVID-19 and beyond: Trade and health. WTO. https://docs.wto.org/dol2fe/Pages/SS/directdoc.aspx?filename=q:/Jobs/GC/251R1.pdf&Open=True (accessed January, 21, 2022).
WTO. n.d. Information technology agreement. In Trade topics. The World Trade Organization. https://www.wto.org/english/tratop_e/inftec_e/inftec_e.htm (accessed October 21, 2021).
__________________
This page intentionally left blank.


















