4
Causes and Consequences of Medical Product Supply Chain Failures
In preceding chapters, this report articulated an overarching goal of increasing the resilience of medical product supply chains and it described key characteristics of these supply chains. This chapter turns to the first step in identifying a strategy for achieving this goal, which is to understand why medical product supply chains fail. Medical product shortages can represent a significant threat across the landscape of public health and health care delivery by undermining the ability to provide timely and high-quality care to patients. This has been brutally clear in the context of the COVID-19 pandemic. However, long before COVID-19, medical product supply chains contended with a series of product shortages. The persistence of shortages in the United States and worldwide—caused by a confluence of complex factors—underscores the need to better understand the root causes and effects of shortages to inform more effective strategies to mitigate and protect against shortages, and respond to them when they do arise.
This chapter begins by outlining the mechanics of shortages in medical product supply chains and then explores the three main causes of shortages: demand surges, capacity reductions, and coordination failures. Finally, the chapter examines the effects of these shortages from the perspectives of patients, health care facilities and providers, and medical product manufacturers and suppliers. These discussions will provide the foundation for the development of the medical product supply chains resilience framework described in Chapter 5.
MECHANICS OF MEDICAL PRODUCT SUPPLY CHAIN FAILURES
As noted in Chapter 2, medical product supply chains facilitate the flow of medical products from raw material or component suppliers to producers to distributors to providers and finally to patients. See Figure 4-1 for a graphical representation of a typical supply chain under normal, nondisrupted conditions.
The field of supply chain management views a network like the one shown in Figure 4-1 as an end-to-end system, with the overarching goal of managing the flow of goods from the supplier to the end consumer through cooperation of the supply chain as a unified entity (Mentzer et al., 2001). However, it is rare to find supply chains for almost any product type that are contained entirely within a single vertically integrated organization. For medical products, the levels in Figure 4-1 are usually operated by separate entities—often with multiple entities at some levels. This means that supply chains are not centrally controlled systems but are instead distributed networks with multiple decision makers that have different objectives. This can lead to mismatches in supply and demand, even under normal conditions. The challenges presented by decentralization during
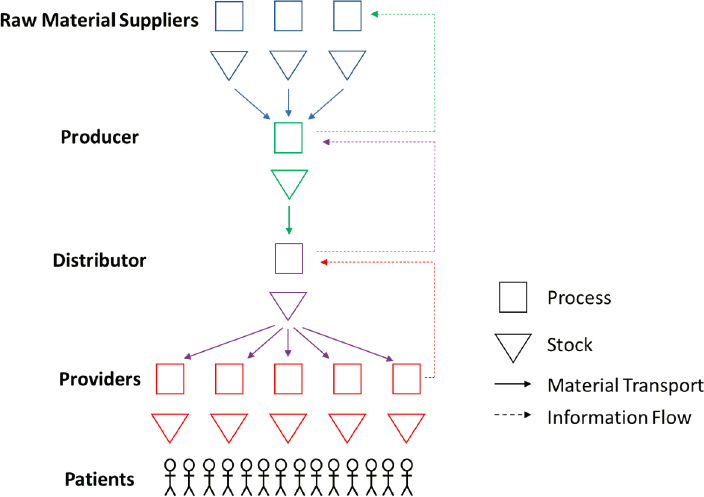
SOURCE: Adapted from NASEM, 2020.
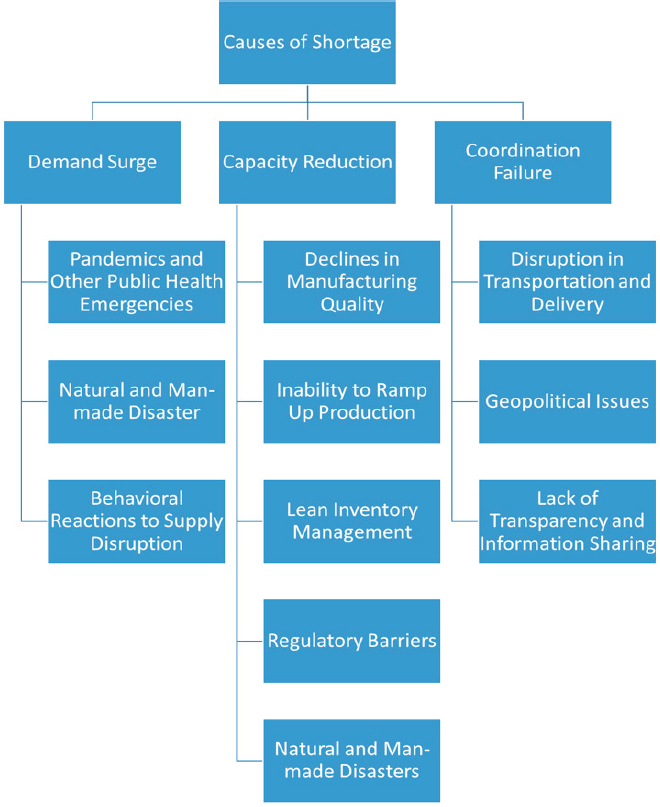
normal times become even greater when the supply chain is disrupted by a natural disaster, infectious disease outbreak, manufacturing process problem, or other event. At a high level, a disruptive event can cause a mismatch between supply and demand—a shortage—in medical product supply chains in three ways:
- Demand surge: An event drives demand for a medical product well above the normal level for an extended period of time. For example, a major natural disaster, such as a tornado or earthquake, can spike regional demand for certain medical products if these events result in a significant number of casualties requiring medical care. As seen during COVID-19, a pandemic can drive up global demand for many medical products.
- Capacity reduction: One or more production or transport processes are impeded by lack of assets, power, or people. For example, a natural disaster could cause a factory to lose power and halt production or regulatory barriers or manufacturing quality problems could restrict the output of a supplier or producer, and could even eliminate inventory stock if a product is recalled. As seen during the COVID-19 pandemic, production of some products may have decreased because of lockdown measures or the need for workers to quarantine or be on sick leave.
- Coordination failure: Events that prevent supply from being matched to demand can cause shortages of medical products even when total supply is sufficient to meet total demand. For example, geopolitical issues or communication system failures during a hurricane or other natural disaster can reduce or obstruct the delivery of emergency supplies to the people that need them.
A central concept for describing the capacity of flow in a supply chain is a bottleneck. In general, bottleneck is the stage in a supply chain with the highest utilization (demand as a percentage of capacity) (Hopp, 2008). Under normal conditions, the bottlenecks in medical product supply chains can be the supply of raw materials, a production process of a supplier, a final assembly plant, or a distribution process. Under normal conditions, all stages of a supply chain, including the bottleneck, remain safely below 100 percent of capacity. However, an event that causes a demand surge or capacity reduction can push the use of one or more production or transport resources above 100 percent. A resource with use above 100 percent capacity is said to be overloaded (NASEM, 2020). When this happens, supply will not be able to keep up with demand. At first, this will cause stock levels at various points in the supply chain to decline. When this
eventually depletes stock at the health care provider level, patients experience shortages (NASEM, 2020).1
Figure 4-2 illustrates a snapshot of a supply chain that is experiencing a shortage. In this scenario one of the raw material suppliers has become overloaded. This could be the result of a capacity reduction affecting that supplier, such as an earthquake damaging a facility or a manufacturing quality problem that has restricted shipments. It could also be the result of a demand surge that caused the supplier, which was already the bottleneck, to become overloaded. In either case, the bottleneck resource will define the overall capacity of the supply chain. If that capacity is insufficient to keep up with demand, patients will experience a shortage once any inventories in the supply chain are exhausted. Finally, note that Figure 4-2 indicates that some providers are experiencing shortages, while others are not. This could be the result of some nodes holding more buffer inventory or some receiving higher priority in the distribution of limited supplies.
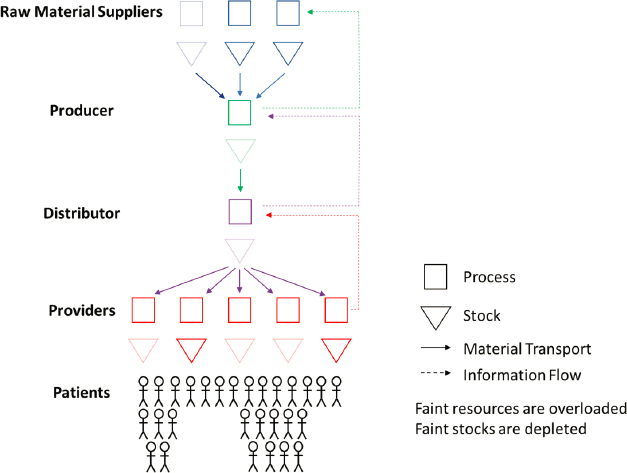
SOURCE: Adapted from NASEM, 2020.
___________________
1 Note that a disruptive event can cause the bottleneck to move. For example, under normal conditions, the bottleneck of a supply chain might be the final manufacturing step, which limits the amount of product that can be produced. However, if a fire damages a component plant, vastly reducing its output, this plant may become the bottleneck. Worse, it could become an overloaded bottleneck that prevents the entire supply chain from keeping up with demand.
It is important to note that disruptions can overlap and interact—demand surges and capacity reductions may occur simultaneously, or capacity reductions may precipitate subsequent demand increases and vice versa. Additionally, disruptions do not cause medical product supply chains to shift instantly from the normal conditions of Figure 4-1 to the shortages of Figure 4-2. Because all supply chains contain at least some amount of inventory, and because decision makers will take steps to increase capacity, such as run overtime shifts, in response to an impending shortage, it will take time for a disruption to turn into a shortage experienced by patients. Figure 4-3 gives a graphic illustration of how a capacity overload of a raw material supplier might propagate through the supply chain to affect patients as a shortage.
The time-phased nature of failures in medical product supply chains has important implications for building supply chain resilience. First, steps that increase the time from a disruption to a product shortage may help avoid shortages altogether; for example, if the length of the disruption is shorter than the timeframe covered by the inventory held in the supply chain, then inventory provides a buffer against a shortage. Second, even if a shortage does occur, inventory buffers delay the shortage from reaching patients and thereby provide time to execute contingency plans and response measures to protect patients. Chapter 5 will build on these insights to create a medical product supply chains resilience framework. This chapter will first explore types of demand surges, capacity reductions, and coordination failures, along with examples of each (see Figure 4-4).
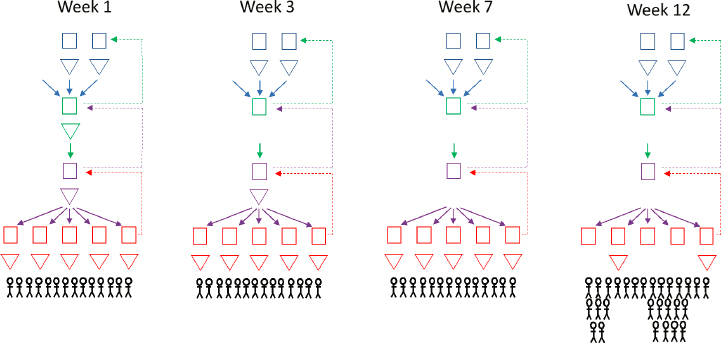
SOURCE: Adapted from NASEM, 2020.
DEMAND SURGES
When an event drives demand for a medical product well above the normal level for an extended period of time, a demand surge occurs. Whether a demand surge results in shortages or not depends on whether the various actors in the medical product supply chains have enacted mitigation and preparedness measures, such as hardening of the supply chain, building sufficient inventory or capacity buffers, and planning for contingencies (as will be discussed in detail in Chapter 5). Three types of events can cause demand surges: pandemics and other public health emergencies, natural and
man-made disasters, and behavioral reactions to supply chain disruptions (FDA Drug Shortages Task Force, 2020; Socal et al., 2020).
Pandemics and Other Public Health Emergencies
As seen during the COVID-19 pandemic and previous infectious disease outbreaks, a pandemic or other public health emergency can severely strain medical resources at different levels—locally, regionally, nationally, and internationally—when the demand for health care services increases dramatically (IOM, 2009). The COVID-19 pandemic directly led to a demand surge for personal protection equipment (PPE), ventilators, and medications required for critical care (Ammar et al., 2021; Farrell et al., 2021; Ranney et al., 2020). Subsequent shortages led to intense competition for products in short supply, which in turn caused price increases and in some cases price gouging (Butler et al., 2020; Linskey et al., 2020). See Box 4-1 for a case study on the surge in demand for N95 masks. These shortages highlighted the effect of demand surges in the context of a major public health emergency (Schondelmeyer et al., 2020), the risks of having a single source for an item, the need to better manage supply risk, and the importance of end-to-end transparency across the supply chain (NASEM, 2021).
Natural and Man-Made Disasters
Natural disasters—such as fires, floods, earthquakes, and hurricanes—and man-made disasters—such as war and bioterrorism—have the potential to cause demand surges for health care services that overwhelm local and regional medical and public health systems as well as the capacity of medical product supply chains (IOM, 2009). Even prior to the COVID-19 pandemic, there were shortages of N95 masks and air filters in areas where wildfire smoke caused poor air quality (CBS News, 2018). In 2011, a tsunami triggered by an earthquake off the coast of Japan led to a surge in demand for basic food items, blankets, sanitary pads, diapers, and toilet paper (Cavallo et al., 2014). Disasters are major disruptions to the supply chain, so without adequate preparedness and planning measures in place, demand surges can quickly turn into shortages.
Behavioral Reactions to Supply Disruption
Behaviors such as hoarding and panic buying can create demand surges and exacerbate existing supply disruptions. Hoarding can be both a result of a shortage and increase the likelihood that a disruption will become a shortage (Childs, 2019; Fox and McLaughlin, 2018). During a shortage, hoarding by institutions or individuals may be driven by concerns about maintaining or acquiring an adequate supply of products, particularly if there is a lack of awareness and transparency about the severity of the shortage, its expected duration, and any ongoing mitigation strategies. A national survey of more than 700 hospital pharmacy managers looked at rationing practices during shortages at U.S. hospitals. Eighty one percent of respondents reported using hoarding of the available supply of a drug in shortage as a mitigation strategy (Hantel et al., 2019). In turn, hoarding exacerbates a shortage by making the product unavailable for others, which can result in black and gray markets that can be counterproductive and potentially dangerous. Furthermore, frequent updates to clinical guidance and touting of potential treatments during a public health emergency can make demand difficult to predict (Callaway Kim et al., 2021). For example, the 2001 anthrax attacks led to a demand surge attributable to panic buying of ciprofloxacin (Weschsler, 2001). The ciprofloxacin shortage was not caused by widespread medical use of the drug to treat anthrax victims, it was the result of panic buying and limits on expanded production owing to intellectual property rights (Herper, 2001). See Box 4-2 for an additional example of health care providers hoarding saline solutions as a response to, and an exacerbation of, an existing shortage.
CAPACITY REDUCTIONS
A capacity reduction occurs when one or more production or transport processes are impeded by lack of assets, power, or people. Capacity reductions can arise at any part of the supply chain, including raw material production, component production, final assembly, and transportation (FDA Drug Shortages Task Force, 2020). Each of these steps can be disrupted by factors such as declines in manufacturing quality, inability to ramp up production, lean inventory management practices, regulatory barriers, and natural and man-made disasters.
Declines in Manufacturing Quality
The single biggest cause of drug product supply chain disruptions under routine conditions is a failure to maintain manufacturing quality, according to an analysis by FDA (FDA Drug Shortages Task Force, 2020). The 2020 update of the FDA Drug Shortages Report found that quality problems are responsible for 62 percent of the drugs that went into shortage between 2013 and 2017. Declines in quality are often attributable to factors such as the use of older facilities, the introduction of new products that require companies to share production facilities, contracting practices that lead to poor oversight, and increased price competition (Woodcock and Wosinska, 2013). In a market with a small number of manufacturers,
a capacity reduction or market exit by a single manufacturer due to quality problems places pressure on the other manufacturers to fill the gap in supply, which they may not be able to do (Childs, 2019). Furthermore, the market currently does not recognize or incentivize robust quality management systems. The entry of low-cost competitors can cause a ripple effect that affects established manufacturers and ultimately forces some of them to exit the market because of price decreases (Childs, 2019).
Declines in manufacturing quality and consequent supply disruptions occur frequently among generic manufacturers—particularly disruptions at the finished dosage level. Generic drugs administered by injection are the most common products in shortages, largely because there are fewer manufacturers of these drugs and because the manufacturers that do produce them typically operate at full capacity, making it difficult to respond to and recover from disruptions (Fox and McLaughlin, 2018). Box 4-3 provides a case study of the anticoagulant heparin, which illustrates how quality issues led to both direct and indirect health effects owing to a supply shortage. This case also highlights difficulties in monitoring and regulating quality in global supply chains and some challenges and successes in both diversifying and on-shoring supplies.
All manufacturers must adhere to FDA’s current good manufacturing practice regulations, which include “minimum requirements for methods, facilities, and controls used in manufacturing, processing, and packing of a drug product” (FDA, 2020a). Buyers, therefore, assume the product is safe for use and contains the ingredients at the strength it claims to have and do not distinguish between the quality of any given generic drug. As a result, the market does not reward for production quality. Without a price premium for quality, and with low margins that minimize the value of protecting continuity of supply, generic manufacturers typically do not invest enough in quality systems or equipment modernization to prevent quality failures.
The expansion of multiple foreign manufacturing sites presents new challenges in FDA’s work to ensure the quality and safety of imported products entering the domestic market (Woo et al., 2008). To address these challenges, FDA relies on a number of measures to ensure quality and safety of medical products in facilities around the world including preapproval inspections, surveillance inspections, and for-case inspections.2 FDA also relies on automated systems to screen import shipments of drugs, biologics, and medical devices at the U.S. port of entry for compliance with regulatory requirements. These electronic systems review import entries and flag products that may require additional information, a physical exam, or sample analysis (FDA, 2017).
The lack of visibility into the quality of the manufacturing processes used for medical products, and therefore the quality of the products them-
___________________
2 Information given by testimony of Judith A. McMeekin (Senate Committee on Finance, 2020).
selves, prevents the market from rewarding quality manufacturers, and causes supply disruptions and shortages (FDA Drug Shortages Task Force, 2020). The committee’s examination of quality-related shortages highlights important observations that must be included in thinking about how to make medical product supply chains safer and more resilient: For nearly a decade, analyses have found that quality problems are responsible for a majority of the drugs that go into shortage. As a result, there have been repeated calls for a robust and mature quality management system to ensure consistent and reliable drug manufacturing and quality performance. However, there is still no such system and the quality problems persist. Purchasers of medical products lack access to sourcing and quality information, which limits their ability to incorporate supply chain resilience when making contracting, purchasing, and inventory decisions.
Counterfeit Medical Products
Counterfeiting, or the proliferation of substandard or falsified medical products, is also a challenge with a globalized supply chain. The complex, and at times circuitious, distribution routes that medical products follow from manufacturer to patient can lead to unregistered/unlicensed, substandard, and falsified (SF) medicines entering the market (The PEW Health Group, 2011). Diverted, stolen, or counterfeited medical products may make their way to American consumers from both domestic and foreign sources (The PEW Health Group, 2011). The American market is particularly vulnerable to the entrance of SF medical products during shortages, when wholesalers and distrubtors may exchange products multiple times, complicating the tracability of the products and raising concerns about the authenticity and quality of the medical products being sold (Rockefeller et al., 2012).
Counterfeiting in the global supply chain is more than a safety issue; it can become a national security issue if individuals introduce counterfeit drugs into the supply chain with the intention to cause harm (Marucheck et al., 2011). For instance, if a counterfeit drug contains contaminated components, it can cause serious harm beyond the lack of therapeutic effect (Bos, 2009). While the World Health Organization’s (WHO’s) Global Surveillance and Monitoring System3 for SF medical products maintains a database of suspected and validated SF reports, the data cannot be extrapolated to country-specific prevalence because regulatory authorities are not always willing, able, or incentivized to report the incidents (NASEM, 2021). One option to address counterfeiting used for pharmaceuticals is serialization, whereby individual drug units are assigned unique identification numbers. This allows
___________________
3 WHO Global Surveillance and Monitoring System is available at https://www.who.int/who-global-surveillance-and-monitoring-system (accessed January 19, 2022).
for tracking the product, authenticating the product within the distribution chain, as well as identifying counterfeits (Pascu et al., 2020). However, in order for such a system to be successful for globalized supply chains, coordination between international health authorities, such as the World Health Organization, and regional or national authorities is crucial to maintaining the integrity and transparency of supply chains (Pascu et al., 2020).
Inability to Ramp Up Production
Manufacturers’ inability to quickly ramp up production is a factor that compounds capacity disruptions. Manufacturers may face multiple barriers to ramping up production in response to intermittent supply disruptions (NASEM, 2021). For most companies, maintaining standing capacity to ramp up production rapidly is neither logistically nor financially feasible. Generic drug manufacturing, in particular, is a low-margin business that does not allow for idle capacity. If a competitor experiences a supply disruption or demand surge, there will likely be insufficient excess capacity available for other manufacturers to increase production to fill the gap, as was the case during a 2010 propofol shortage (FiercePharma, 2010; Jensen and Rappaport, 2010; Woodcock and Wosinska, 2013).
Additional factors that limit the ability of manufacturers to ramp up production can include operational and regulatory risks as well as packaging and labeling capacity (Jung, 2020). Operational delays may include limits on production, constraints on plant or shipping capacity, and problems with transportation and distribution. Particularly if the disruption is caused by a disaster or public health emergency, operational risks could include human resource deficits (e.g., absenteeism), restrictions of work shift capacity that limit output, or constraints in certain parts of supply chains (e.g., cold-chain storage), especially if multiple manufacturers are attempting to ramp up production to meet demand signal fluctuations (Jung, 2020). Among the potential regulatory risks that manufacturers may face when increasing production are facility inspection delays for new products, generics, and biosimilars, which can lead to longer approval times, as well as inspection requirements following importation (Jung, 2020). The packaging and labeling needed to contain, transport, distribute, and administer products often relies on foreign sources for components, such as resin-based bottles and films, paper cartons and labels, and stainless steel needles. If those components are not consistently available, it can contribute to disruptions in the supply of the finished product (Jung, 2020). Furthermore, decisions to manufacture a new product or ramp up production of an existing product can force companies to make production trade-offs if they are already operating at full capacity, as many generic manufacturers do.
Manufacturers may also have other reasons not to increase their manu-
facturing capacity during a shortage. For example, after the 2009 H1N1 influenza pandemic, when the mask manufacturer Prestige Ameritech ramped up production during the emergency, they were left with excess product and financial problems when demand suddenly disappeared. Consequently, the company was initially reluctant to ramp up N95 production during the early weeks of the COVID-19 pandemic. The company did eventually install several new mask-making machines and secured investments and multiyear contracts from facilities to make additional masks, which enabled them to ramp up production significantly (Noguchi, 2021).
The above observations lead the committee to conclude that manufacturers often lack incentives to maintain surge capacity in their supply chains, as it is neither financially nor logistically feasible. This limits the ability of supply chain managers to react to disruptions and leads to shortages.
Lack of Investment in Modern Technology
The use of outdated technology is common in medical product manufacturing, and it contributes to manufacturers’ ability to ramp up production. It also introduces product errors that can subsequently lead to shortages. While other industries typically function at an error rate below 3.4 defects per million opportunities, the pharmaceutical manufacturing industry operates at a rate of around 66,000 defects per million opportunities—owing in part to out-of-date manufacturing processes (Woodcock and Kopcha, 2020). Public and private investment in advanced manufacturing is needed to improve the reliability of medical products and reduce the opportunity for quality defects.
Advanced manufacturing techniques, such as continuous manufacturing, and additive manufacturing, such as 3D printing, can improve product quality and address shortages (FDA, 2021a). However, a survey of medical product manufacturers, conducted by Manufacturing x Digital (MxD) and the International Academy of Automation Engineering (IAAE), found that surveyed manufacturers largely still relied on manual, paper-based processes and were only just beginning to advance to more automated and integrated process stages (MxD and IAAE, 2021).
The committee concludes that advanced manufacturing techniques can improve product quality and reliability, thereby addressing frequent causes of shortages. However, medical product manufacturers have little incentive to harden supply chains through updated techniques, processes, and controls that promote reliability and quality in medical products. The medical product industry will likely require additional incentives to spur investment in updated manufacturing technologies that promote reliability. Namely, health systems will need information regarding products’ manufacturing processes to provide manufacturers the incentive to invest in quality.
Lean Inventory Management
Lean inventory management practices can lead to capacity reductions and increase the risk of medical product shortages. It is common practice among manufacturers, distribution centers, and health care organizations to use a just-in-time (JIT) inventory management system (Fox and McLaughlin, 2018). In many cases, JIT leads to smaller inventory levels across the supply chain, which, as illustrated in Figure 4-3, reduces the time it takes for a shortage to reach patients and thereby makes shortages more likely to occur. Lean practices are an example of policies that optimize profits under normal conditions but can also make supply chains less able to respond to disruptions and emergencies. A lack of transparency of inventory practices across supply chain actors and regulators further compounds these challenges (NASEM, 2018). Lean systems are especially vulnerable during major disruptions caused by a natural disaster or public health emergency (NASEM, 2020). The committee therefore concludes that market forces incentivize lean inventory management in medical product manufacturing and ordering, which limits the ability of the health care system to withstand shortages and increases the likelihood that shortages will affect patients.
Regulatory Barriers
In 2020, the FDA Drug Shortages Task Force found that logistical and regulatory challenges can make market recovery difficult following a supply disruption (FDA Drug Shortages Task Force, 2020). Furthermore, as drug supply chains have become more complex, it has become increasingly difficult for manufacturers to respond to shortages because of additional regulations involved, gaps in API procurement, and FDA application requirements for new market entry (FDA Drug Shortages Task Force, 2020). If U.S. medical product supply chains depend heavily upon a few manufacturers, and one manufacturer exits the market, there can be a significant time lag before a new manufacturer can enter the market and increase the available supply. FDA is taking steps to bring greater efficiency to the generic drug review process and respond to shortages. For example, when a shortage is reported, FDA can expedite review of new production lines or new raw material sources. The agency can also consider extending the expiration date for a product experiencing shortage if expired stock is available (FDA, 2013) and thereby increase the available supply. FDA has also taken steps to expedite the development and marketing of generic drug products. For example, in 2019 the agency announced a process to expedite U.S. Pharmacopeial Convention small molecule monograph updates (FDA, 2019b). However, the approval of a new manufacturer can still take 1 to 2 years (FDA, 2021a; Sullivan, 2018).
Other forms of regulation can also lead to unintended capacity reductions in medical product supply chains. In 2019, the firm Sterigenics closed a sterilization plant after the Illinois Environmental Protection Agency prohibited the firm from using the carcinogen ethylene oxide in its sterilization process because of the resulting chemical emissions from the plant (Goldberg, 2019). Closure of the plant severely reduced the total medical device sterilization capacity in the United States. The closure caused several devices to go into shortage (Crotti, 2019) and FDA warned that widespread shortages were possible (Maxwell, 2019). At the time of the closure, about 50 percent of devices requiring sterilization used ethylene oxide (Maxwell, 2019), and only 2 percent of those had an alternate validated sterilization method available for use (Lim, 2019). FDA is now working to advance innovation in ways to sterilize medical devices using lower levels of ethylene oxide and other currently available agents, and it is funding research into alternative sterilization techniques (FDA, 2020b).
Natural and Man-Made Disasters
In addition to causing demand surges, natural and manmade disasters can also cause capacity reductions. The past three decades have seen drug shortages result from natural disasters, either by affecting product availability, damaging production facilities, and/or creating unexpected demands for drugs needed to treat the injured (Fox et al., 2009). In 2017, for example, Hurricane Maria severely disrupted the production of medical use saline (NASEM, 2020). At the time, the majority of saline produced for the U.S. market was manufactured by a single firm, Baxter International, at three facilities in Puerto Rico. Hurricane Maria disrupted power at these facilities in September 2017, and power was not completely restored until January 2018. The 4-month supply disruption resulted in widespread saline shortages at hospitals in the United States throughout 2018 (NASEM, 2020). See Box 4-2 for a case study on saline, which illustrates the vulnerabilities created by concentrated production (even when that production is located on U.S. territory), how hoarding exacerbated the shortage, and the importance of being able to tap the global supply chain for backup supplies in an emergency.
COORDINATION FAILURE
A coordination failure leads to shortages of medical products when total supply is available to meet demand, but circumstances prevent the supply from being matched to the demand. Examples of coordination failures include disruption in transportation and delivery, geopolitical issues, and lack of transparency and information sharing. In addition to exacerbating shortages, coordination failures can also inhibit response and recovery.
Disruption in Transportation and Delivery
Particularly in emergency conditions, supply may be available but is unable to reach the people who need it because of challenges in transportation and delivery. This happens frequently in hurricanes and other natural disasters. When Hurricane Maria hit Puerto Rico in 2017, there was extensive damage to the electrical grid, roads, airports, and fuel supplies, disrupting delivery of raw materials and components to Baxter International’s saline production facilities, halting their production lines and causing a shortage (AEP, 2018). Bottlenecks in shipping have caused widespread delays during the COVID-19 pandemic. The cost of a shipping container has quadrupled since 2020 (Page, 2021), and ports have been backed up, unable to load and unload items waiting to be shipped (Page, 2021). A survey of medical technology manufacturers revealed that over 70 percent of firms had experienced delays in their semiconductor supply chains (Murray and Bradley, 2021). Semiconductors are in high demand across many industries, and as a result, manufacturers are competing for the same supply. The Advanced Medical Technology Association appealed to the secretary of the Department of Commerce to help medical device manufacturers gain priority access to the limited supply (AdvaMed, 2021). Many medical products such as vaccines must be kept in temperature-controlled environments from the time they leave the production facility to the time they are delivered to patients. This poses particular challenges in transportation and delivery to rural and remote locations. This challenge was evident during the distribution of certain vaccines against COVID-19, but it has also been an ongoing challenge in delivering medical products around the globe for many decades (Lloyd and Cheyne, 2017; UNICEF, n.d.).
Geopolitical Issues
Geopolitical risks can arise because of the consequences of national politics or societal disruption and have the potential to prevent supply from reaching demand and causing a shortage. Political crises often occur due to the economic consequences of the domestic political milieu, legislative actions, or the instability of governments and other institutions, such as political transitions, corruption, policy shifts, inadequate law enforcement, societal conflict, and “buy national” policies (Cline et al., 2019, 2020). Foreign sources of supply could also become unavailable, not because of a deliberate strategic choice on the part of the government, but because that government’s own ability to maintain order within its territory is somehow threatened. Societal disruption can arise when groups such as nongovernmental organizations, trade unions, and consumer bodies develop a collective political identity and engage in activism or activities—such as boycotts, protests, supply disruptions, or corporate espionage—that can
affect manufacturing firms who are operating both nationally and transnationally (Cline et al., 2019, 2020).
Trade restrictions, a heavy reliance on foreign manufacturing in certain product classifications and areas, including APIs, and the inability to get foreign supply to the United States have been related to shortages during the COVID-19 pandemic (Mullen, 2020). Geopolitical tensions may also arise when a country limits access to raw or finished materials as a matter of deliberate strategic choice, as observed following the U.S. use of the Defense Production Act, thereby limiting access to materials. The drug supply chain is also increasingly vulnerable to cyberattacks by malicious geopolitical actors. These cybersecurity vulnerabilities justified the military’s involvement in Operation Warp Speed as an effort to prevent the sabotage of critical infrastructure (Florko, 2020).
Extensive reliance on foreign sources for medical products, their key components, or even transportation could leave the United States vulnerable to the geopolitical and trade decisions of those nations. When seen through the lens of national security, other foreign actors could use these market concentrations to put undue influence on American policy makers when negotiating bilateral and multilateral agreements (NASEM, 2021). Although geopolitical issue-driven supply chain disruptions are rare and difficult to identify as the root cause of a shortage, the concern remains among lawmakers, federal agencies, and the private sector (Chalfant, 2017; DoD et al., 2021). Greater situational awareness to anticipate geopolitical shocks could support better recognition and response to emerging concerns.
Lack of Transparency and Information Sharing
Lack of transparency and information sharing is a coordination failure that has ramifications throughout supply chains, and is frequently highlighted as a major shortcoming in the current system. U.S. drug supply chain vulnerabilities are not sufficiently transparent to support timely management of drug shortages, which makes it “nearly impossible” to predict shortages (Schondelmeyer et al., 2020). See Box 4-3 for a case study on heparin, highlighting the dangerous consequences of a lack of transparency in a medical product supply chain. Similarly, there are also issues of transparency in medical device supply chains. For example, Dai and colleagues (2020) examined the past 5 years’ financial disclosure of three major PPE manufacturers (3M Company, Honeywell International Inc., and MSA Safety Inc.) and conducted an exhaustive search of 2020 media reports about the PPE supply chain, and found no basic supply chain data, including, for example, the exact domestic versus foreign capacity of N95 masks. Basic PPE supply chain data (e.g., the production quantity in each facility)
is treated as confidential and not disclosed to any government agency, the public, or the companies’ shareholders.
As will be discussed in Chapter 6, many of the measures needed to address the medical product shortage problem in the United States will require a dramatic increase in transparency at every step of the supply chain, particularly regarding sourcing, quality, volume, and capacity information. Improved transparency and information sharing with FDA and other health authorities provides the means to mitigate and prepare for shortages (Tolomeo et al., 2020). For example, industry stakeholders identified limited market insights into future demand as a major issue in their ability to manage shortages (The PEW Charitable Trusts; International Society for Pharmaceutical Engineering, 2017). Companies reported being reluctant to invest in setting up additional manufacturing capacity to protect against shortages in the absence of accurate information about expected demand—particularly for low-volume, low-margin products. They suggested that insight into future demand could be improved through better management of internal operations such as sales and operations planning and demand forecasting, as well as better practices for sharing information about the market landscape, such as how long other manufacturers are expected to be out of the market.
Drug manufacturers, foreign or domestic, that wish to sell products in the United States must register with FDA and report limited information on what products are manufactured at specific facilities.4 However, manufacturers are not required to report information such as the fraction of product produced at any given facility or the fraction of an API sourced from a particular supplier. The Coronavirus Aid, Relief, and Economic Security Act (CARES Act) improved upon previous reporting requirements by requiring manufacturers to report the amount of drug manufactured.5 However, many gaps remain in FDA’s ability to collect and analyze data in a timely manner to react to sudden disruptions and shortages (The White House, 2021). It is therefore not possible for FDA to identify when a supply chain is dangerously concentrated or otherwise vulnerable to disruption (The White House, 2021). It is also important to note that although FDA has these limited data from drug manufacturers, the agency receives very little information from medical device manufacturers on supply chain and shortages.
Transparency is an issue for regulatory agencies, manufacturers, and consumers as medical product supply chains become more global and the supply chain networks become more complex. Regardless of supply chain type (e.g., global versus domestic), there is little insight into the where and how of medical product manufacturing. In many cases, the specific factory,
___________________
4 Information given by testimony of Janet Woodcock (House Committee on Energy and Commerce, 2019).
5 CARES Act, 15 U.S.C. § 116-136.
or even the country in which the drug is produced, is kept confidential as proprietary information. Manufacturers are also hindered by lack of transparency into foreign sources of API and other components. A disruption that affects a single API manufacturer could create a major problem by halting production across multiple finished dosage form (FDF) manufacturers. If a manufacturer needs to find a new API source—either to increase production or because of a disrupted supply from another API source—it can be challenging to find a foreign API source that complies with global regulatory requirements (FDA Drug Shortages Task Force, 2020). FDA compiles lists of approved API suppliers and recently inspected API manufacturing facilities.6 However, the agency does not provide a centralized source of information about API suppliers. Consumers have even less information about the sources of drugs and devices. In the case of prescription drugs sold in the United States, labels are not required to disclose the identity or location of the API or FDF manufacturer (Conti et al., 2020). Regulations regarding country of origin that apply to other imported products are not typically enforced by U.S. Customs for prescription drug imports (Schondelmeyer et al., 2020).7 This undermines the ability of both purchasers and policy makers to analyze data to identify and mitigate supply chain vulnerabilities (Schondelmeyer et al., 2020).
Although it is widely accepted that better visibility across the supply chain into manufacturers’ sources, locations, and volumes of raw materials, APIs, and finished products could help to prevent and mitigate supply disruptions, many manufacturers are reluctant to provide that information (NASEM, 2021). Barriers to increasing transparency in manufacturers’ supply chains include
- competition among manufacturers;
- lack of (accurate) data collection;
- concerns about privacy and confidentiality such as proprietary and trade secrecy concerns on the part of industry;
- concerns about counterfeiting;
- lack of incentive structures;
___________________
6 “API facilities are included in FDA Inspections Classification Database, which includes final classifications for surveillance inspections of all API facilities in the human pharmaceutical program. This database includes results of FDA inspections and where FDA has made use of an inspection conducted by a capable inspectorate under the mutual recognition agreement (MRA). API facilities not currently supplying the U.S. market would not be included in this database. Furthermore, the database does not list the products being made at the facilities” (FDA Drug Shortages Task Force, 2020).
7 “Country of origin” is defined as “the country of manufacture, production, or growth of any article of foreign origin entering the United States” (Code of Federal Regulations. Title 19, Part 134, Subpart A, Section 134.1, Country of Origin Marking, General Provisions, Definitions. April 1, 2011).
- potential risk to shareholders;
- lack of data collection mechanisms and supporting infrastructure, including guidance on what data are needed and the tools needed to make the data accessible to the people that need them;
- interest in maintaining security of sources; and
- complexity and multiplicity of components (NASEM, 2021).
The committee concludes that a lack of transparency in medical product supply chains has led to limited empirical evidence regarding best strategies for addressing supply chain issues. The current practice of keeping medical product supply chains confidential conflicts with public health needs and puts the public’s health at risk. Improving the public’s access to data that are important to their health and well-being is critical.
EFFECTS OF MEDICAL PRODUCT SHORTAGES
Shortages of medical products have pervasive consequences that extend from the individual patient level through health care facilities, manufacturers, and suppliers (FDA Drug Shortages Task Force, 2020). For patients, shortages can potentially lead to poor clinical outcomes that are caused by substitutions, medical errors, treatment delays, or even lack of treatment if no alternative is available (Fox et al., 2014; Phuong et al., 2019; Tucker et al., 2020). Health care facilities across the United States must also contend with the effects of increasingly frequent product shortages that impose a variety of costs, from higher drug budgets to lost revenue to additional labor costs required to mitigate those effects (Kaakeh et al., 2011). During periods of shortage, manufacturers and suppliers of critical medical products may face intense pressure to compensate for supply chain disruptions, but they are often unable to ramp up production to meet unpredicted surges in demand (Ventola, 2011).
Effects on Patients
Effect on Clinical Outcomes and Patient Safety
Medical product shortages can affect patients in myriad ways, from poor clinical outcomes to other detrimental experiences that affect their quality of life. A 2019 global scoping review synthesized literature on the economic, clinical, and humanistic effects of drug shortages on patient outcomes in the United States and other countries (Phuong et al., 2019). The review found that during times of shortages, clinical outcomes associated with the effects of shortages included increases in drug errors, adverse events, and mortality. Drug shortages at health care facilities can undermine
the quality of patient outcomes through delays in inpatient medication treatment, delays or cancellations of outpatient infusions, delays in immunization, and delays in medical procedures (Vizient, 2019). When a heparin shortage occurred in 2008, elective surgeries were canceled to conserve supply in an attempt to reduce demand for the product (Zhu et al., 2019) (see Box 4-3 for more on heparin shortages). Facilities faced with shortages may need to ration drugs, devices, and other medical products that are in short supply based on patient characteristics and clinical evidence, potentially giving rise to difficult ethical decisions about how to prioritize patients (Fox and McLaughlin, 2018; Grimm, 2020; Hantel, 2014).
Drug shortages can have potentially devastating effects on individual patients, particularly those who depend on the medications for lifesaving treatment. A 2013 survey of U.S. oncology pharmacists found that in 2011, frequent shortages of oncology drugs contributed to delays and changes in chemotherapy regimens, as well as increasing the risk of medication errors and adverse outcomes (McBride et al., 2013). In 2019, Teva Pharmaceuticals, one of two manufacturers of vincristine—used to treat a variety of pediatric cancers—stopped production for economic reasons; the remaining manufacturer, Pfizer, was unable to cover the unmet demand (Caruso, 2020). For high-risk patients living with chronic conditions, shortages can cause sudden treatment interruptions that quickly escalate into acute emergency situations that require them to move from their homes to acute care settings for treatment (NASEM, 2021). In some cases, shortages can leave patients with no treatment options at all. For instance, shortages of lidocaine can make it unavailable for treating patients undergoing anesthesia and sedation with propofol—which can cause a burning sensation on induction—leading to pain and agitation for the patient (FDA Drug Shortages Task Force, 2020).
Medical device shortages can have equally devastating effects on patients. During the COVID-19 pandemic, hospitals did not have enough mechanical ventilators to meet the demand (Jacobs et al., 2020). These shortages prompted many U.S. hospitals to develop triage protocols to decide which patients would receive ventilation—a decision of who lives and who dies for patients with severely compromised breathing (Kerr and Schmidt, 2021; Truog et al., 2020). Other hospitals resorted to placing multiple patients on the same ventilator (Bernstein and Cha, 2020). This comes with increased risk to patients and increased challenges to clinical care (FDA, 2021c). Twenty-six states provided guidance to providers on how to allocate ventilators in the case of a severe shortage (Piscitello et al., 2020).
Effect of Substitutions and Alternatives
Drug shortages can result in poor outcomes for patients—especially those who are most vulnerable—when their substitutes are less efficacious, have a worse adverse-event profile, or require a less common or more difficult dosing regimen (Fox and McLaughlin, 2018). A 2017 survey found that 71 percent of providers could not provide patients with a treatment of choice because of drug shortages, and almost half of those providers believed that their patients received a less effective treatment as a result (Institute for Safe Medication Practices, 2018). A retrospective cohort study reported that during the 2011 hospital-level norepinephrine shortage in the United States, patients admitted with septic shock who were treated with an alternative vasopressor (e.g., phenylephrine) in hospitals affected by the shortage had higher in-hospital mortality compared to those treated in hospitals during periods when the first-line norepinephrine treatment was not in shortage (Vail et al., 2017). In another example, a 2016 shortage of bleomycin—a palliative treatment for patients with cancer—warranted the use of alternative regimens that required inpatient treatment, which is more stressful for patients and families, increases the risk of patients’ nosocomial exposure to pathogens, and is more costly (FDA Drug Shortages Task Force, 2020).
Effect of Medication Errors
Drug shortages can undermine patient safety in a host of ways. For example, they can contribute to medication errors when pharmacies are required to change their practices around how drugs are prescribed, prepared, or dispensed (Fox and McLaughlin, 2018). According to a survey of hospital pharmacy staff about the effect of drug shortages on outcomes for hospitalized patients from March 2011 to March 2012, 16 medications in short supply were each involved in more than one report of patient harm, including prolonged disease duration, disease progression, injuries, and death (Institute for Safe Medication Practices, 2012). Patient harms reported in the survey fell into four general categories:
- Inadequate treatment because the alternative medication provided was not the drug of choice,
- Medication errors when an alternative drug or form/strength of drug was used as a substitute,
- Omission of vital medication (nontreatment), and
- Errors in attempts to compound unavailable drugs (Institute for Safe Medication Practices, 2012).
Effect on Patients’ Experiences
Shortages can negatively affect other types of patient experiences, as well. The 2019 scoping review by Phuong and colleagues found that patients were more likely to report increased out-of-pocket costs during shortages. Patient outcomes affected by shortages included patient complaints, frustration, and anger, as well as increased travel time (Phuong et al., 2019). According to a 2013 survey of pharmacy directors, 38 percent of respondents reported patient complaints caused by drug shortages at their institutions (McLaughlin et al., 2013). For the health care providers who care for patients, drug shortages can also lead to burnout, stress, and frustration; these are the secondary effects of shortages, and they negatively affect providers’ ability to deliver high-quality care to patients (NASEM, 2021).
Patients are typically unaware of the risks and realities of shortages (NASEM, 2021). However, a 2015 survey focused on the patient perspective regarding the effects of drug shortages in the United States and Canada, finding that about three-quarters of respondents reported wanting to be notified about drug shortages that could affect their care—prior to elective surgery, for example (Hsia et al., 2015).
Effect on Researchers and the Conduct of Clinical Trials
Drug shortages can also undermine clinical trials for novel therapeutics in cases where the drug that is the current standard of care is unavailable. These unforeseen drug shortages may necessitate substitutions and result in deviations from the study protocol. Shortages of oncology drugs affected the conduct of clinical trial research at 44 percent of represented institutions in the 2013 survey of U.S. oncology pharmacists (McBride et al., 2013). For instance, drug shortages can substantially delay patient enrollment in clinical trials. One oncology trial enrolled less than 60 percent of the expected number of patients because of a shortage of one of the drugs in the comparison arm (Goozner, 2012).
Shortages of medical supplies are another threat to clinical trials and drug research and development. A series of supply chain disruptions in early 2021 precipitated a global shortage of pipette tips—disposable plastic tips commonly used in the conduct of biomedical research (Sheridan, 2021). The shortage forced research institutions to begin contemplating which studies to prioritize and threatened to prompt delays in clinical research.
Effect on Health Systems
Medical product shortages have a near-universal effect on all types of health care facilities and most of their personnel, from clinicians and pharmacists to buyers and facility administrators. For example, severe shortages
of PPE at the onset of the COVID-19 pandemic endangered health care workers (WHO, 2020). Vizient conducted a study across more than 6,000 U.S. facilities,8,9 all of which reported being affected by drug shortages and more than half of which reported having managed a minimum of 20 shortages during the 6-month survey period (Vizient, 2019). At the facility and hospital level, critical medical product shortages are associated with substantial costs that affect budgets, resources, and the ability to provide high-quality and efficient care (Shaban et al., 2018).
For facilities, the direct and indirect costs of drug shortages include increased drug budgets, lost revenue from canceled treatments and procedures, greater need for pharmacy and technician employees, and lost productivity owing to reallocation of pharmacy resources (Vizient, 2019). According to the survey, critical drug shortages cost facilities an estimated $360 million per year for the additional labor required to mitigate the effects of shortages on patient care (Vizient, 2019). Further, to manage the effects of drug shortages, U.S. hospitals are estimated to spend an additional 8.6 million personnel hours per year.
The effects of drug shortages have been documented at private-sector health care facilities as well as government health systems. A 2018 study investigating the effects of drug shortages on the U.S. Department of Veterans Affairs (VA) health care system found that—similar to the private sector—drug shortages are a major barrier to patient care for the larger facilities within the VA system, affecting both quality and efficiency of care, as well as increasing staff workloads and institutional operational costs (Shaban et al., 2018).
The example of the medical oxygen shortages encountered in California in December 2020 and January 2021 depict the broad effect of medical product shortages on health care facilities. Aside from the effects on COVID-19 patients and other individuals with respiratory conditions that require the use of medical oxygen, the oxygen shortage caused patient intake backlogs, reduced the capacity of ambulances to transport patients, tested hospitals’ aging oxygen infrastructure, and precipitated the need for makeshift field hospitals (Nirappil and Wan, 2021). The effects of this shortage rippled outward as hospital’s low oxygen supply forced them to turn away patients, crippling the ambulances that were unable to find hospitals accepting patients, and exacerbating the shortage of portable oxygen tanks. This, in turn, further stressed
___________________
8 Vizient is a company that partners with health care organizations throughout the United States to help improve health care performance by providing data, insights, and purchasing power to their members. For more information see https://www.vizientinc.com/what-we-do.
9 Including nonacute facilities and health systems; academic medical centers; self-governed children’s hospitals; small-, medium- and large-sized hospitals; critical access hospitals; behavioral facilities; long-term care facilities; specialty hospitals; and ambulatory care facilities.
ambulances in addition to delaying the discharge of COVID-19 patients who would otherwise be able to leave the hospital with a portable oxygen tank, thus furthering hospitals’ oxygen shortages.
Effects on Manufacturers and Suppliers
During medical product shortages, manufacturers and suppliers may receive purchase orders substantially above historical levels and come under pressure to ramp up supply quickly. However, this degree of agility is challenging and may be infeasible for many of them, depending on the cause of the shortage. For example, manufacturers that are reliant on single sources of key components may have no recourse to expand production sufficiently to meet the demand. Drug manufacturers may be unable to source an API at all (e.g., because it is not being produced), or they may be dependent upon an API that is not available or is shipped in insufficient quantities to the finished dosage manufacturer to meet demand.
Ramping up and expanding production also tends to be very capital and time intensive, without a guaranteed return on that investment. As previously described, N95 manufacturer Prestige Ameritech was left with a surplus of product and financial instability when demand abruptly fell after ramping up production during the 2009 H1N1 outbreak (Noguchi, 2021).
A serious concern that faces hospitals, health care facilities, and other purchasers of medical products are rogue distributors that operate in the gray market (Rockefeller et al., 2012). These distributors charge exorbitant sums of money for products that are in shortage, and knowingly or unknowingly sell counterfeit or substandard medical products. Gray market distributors often seize on the uncertainty in the supply chain once a shortage arises and health care facilities can no longer reliably purchase products from their normal suppliers. However, even compliant distributors may need to increase their prices when upstream supply chain disruptions increase the price of raw materials (Sheridan, 2021).
CONCLUDING REMARKS
Although it can be difficult to identify the cause of a specific medical product shortage owing to the lack of transparency throughout supply chains, the root causes of shortages can generally be classified into one of three categories: demand surges, capacity reductions, and coordination failures. No matter the root cause, medical product shortages can have devastating effects on patients by undermining the ability of the health care system to provide high-quality care. Some supply chain resilience measures, such as stockpiling inventory, provide protection against a broad range of disruptions, while others, such as improving oversight of quality assurance systems,
reduce the risk of very specific types of disruptions. To select the appropriate elements of a cost-effective strategy for increasing the resilience of critical medical product supply chains, a framework is needed for systematically enumerating and evaluating options. This is the subject of the next chapter.
REFERENCES
AdvaMed (Advanced Medical Technology Association). 2021. AdvaMed response to the Department of Commerce’s request for public comments on risks in the semiconductor supply chain. https://www.advamed.org/wp-content/uploads/2021/11/AdvaMed-Response-to-the-Department-of-Commerce.pdf (accessed February 15, 2022).
AEP (Analytic Exchange Program). 2018. Public-privateanalyticexchange program (AEP). https://www.dhs.gov/sites/default/files/publications/2018_AEP_Threats_to_Pharmaceutical_Supply_Chains.pdf. (accessed November 2, 2021).
AMA (American Medical Association). 2020. Advocacy resource center - recently enacted medical liability immunity statutes related to COVID-19. https://www.ama-assn.org/system/files/2020-12/medical-liability-immunity-statutes-chart.pdf (accessed December 7, 2021).
Ammar, M. A., G. L. Sacha, S. C. Welch, S. N. Bass, S. L. Kane-Gill, A. Duggal, and A. A. Ammar. 2021. Sedation, analgesia, and paralysis in COVID-19 patients in the setting of drug shortages. Journal of Intensive Care Medicine 36(2):157-174. https://doi.org/10.1177/0885066620951426.
Armour, S., and T. M. Burton. 2018. Hospitals wrestle with shortage of IV bags, linked to hurricane. https://www.wsj.com/articles/hospitals-wrestle-with-shortage-of-iv-bags-linked-to-hurricane-1515349271 (accessed October 25, 2021).
Bernstein, L., and A. E. Cha. 2020. A New York hospital is treating two patients on a device intended for one. The Washington Post, March 29, A08.
Bos, J. 2009. Globalization of the pharmaceutical supply chain: What are the risks?–the FDA’s difficult task. Society of Actuaries 23-26.
Bradsher, K., and L. Alderman. 2020. The world needs masks. China makes them, but has been hoarding them. The New York Times, March 14, B05.
Brunell, N. 2020. What happened to California’s medical supply stockpile for a pandemic. https://spectrumnews1.com/ca/la-west/health/2020/04/08/what-happened-to-california-s-big-medical-supply-stockpile-for-a-pandemic (accessed October 25, 2021).
Butler, D., J. Eilperin, and T. Hamburger. 2020. “No offense, but is this a joke?” Inside the underground market for face masks. The Washington Post, May 18.
California ACEP (American College of Emergency Physicians). 2021. COVID-19 PPE resources. https://californiaacep.org/page/COVID-19_PPE (accessed December 6, 2021).
Callaway Kim, K., M. Tadrous, S. L. Kane-Gill, I. J. Barbash, S. D. Rothenberger, and K. J. Suda. 2021. Changes in purchases for intensive care medicines during the COVID-19 pandemic: A global time series study. Chest 160(6):2123-2134. doi:10.1016/j. chest.2021.08.007.
Caruso, C. 2020. Oncology drug shortages persist. Cancer Discovery 10(1):6.
Caspi, H. 2015. Non-sterile saline enters hospital supply chain, sickens 40. https://www.health-caredive.com/news/non-sterile-saline-enters-hospital-supply-chain-sickens-40/353333/ (accessed October 25, 2021).
Cavallo, A., E. Cavallo, and R. Rigobon. 2014. Prices and supply disruptions during natural disasters. Review of Income and Wealth 60:S449-S471.
CBS News. 2017. IV bags in short supply after Hurricane Maria disrupted production in Puerto Rico. https://www.cbsnews.com/news/iv-bags-in-short-supply-after-hurricane-maria-disrupted-production/ (accessed October 25, 2021).
CBS News. 2018. Protective masks fly off store shelves in wildfire-ravaged California. https://www.cbsnews.com/news/fires-in-california-protective-masks-high-demand-camp-fire-bay-area/ (accessed October 21, 2021).
CDC (Centers for Disease Control and Prevention). 2021a. Personal protective equipment: Questions and answers. https://www.cdc.gov/coronavirus/2019-ncov/hcp/respirator-use-faq.html (accessed December 6, 2021).
CDC. 2021b. Strategies for optimizing the supply of n95 respirators. https://www.cdc.gov/coronavirus/2019-ncov/hcp/respirators-strategy/index.html (accessed October 26, 2021).
Cha, A. E. 2007. Pig disease in China worries the world. https://www.washingtonpost.com/wp-dyn/content/article/2007/09/15/AR2007091501647.html (accessed October 25, 2021).
Chalfant, M. 2017. Lawmakers worry cyberattacks could cause drug shortages. https://the-hill.com/policy/cybersecurity/351797-lawmakers-worry-cyberattacks-on-health-industry-could-produce-drug (accessed October 21, 2021).
Chaudhuri, S. 2020. Coronavirus prompts hospitals to find ways to reuse masks amid shortage. The Wall Street Journal, March 31.
Childs, B. 2019. Re: FDA-2018-n-3272, identifying the root causes of drug shortages and finding enduring solutions; public meeting; request for comments, edited by S. Gottlieb. Premier. https://www.premierinc.com/downloads/FDA-Drug-Shortage-Task-Force_Premier-Comments_FINAL.PDF (accessed December 16, 2021).
CivicaRx. 2019. Civica Rx and Hikma announce shipments of heparin and seven other essential injectable medicines. https://civicarx.org/civica-rx-and-hikma-announce-shipments-of-heparin-and-seven-other-essential-injectable-medicines/ (accessed October 25, 2021).
Clark, D. B. 2021. Inside the chaotic, cutthroat gray market for N95 masks. The New York Times Magazine, November 17, 2020.
Cline, M., J. Shames, and C. Rickert-McCaffrey. 2020. How to manage political risk in a post-pandemic world. https://riskcenter.wharton.upenn.edu/lab-notes/how-to-manage-political-risk-in-a-post-pandemic-world/ (accessed December 20, 2021).
Cline, M. K., W. Henisz, S. Behrendt, K. P. Lawless, and R. Abdurakhim. 2019. Political risk and corporate performance: Mapping impact. Ernest & Young and The Wharton School. https://riskcenter.wharton.upenn.edu/wp-content/uploads/2019/09/EY-Geostrategic-Business-Group_Impact-of-Political-Risk_Academic_Draft.pdf (accessed December 20, 2021).
Conti, R. M., Berndt, E. R., Kaygisiz, N. B., Shivdasani, Y. 2020. We still don’t know who makes this drug. Health Affairs Blog. https://www.healthaffairs.org/do/10.1377/hblog20200203.83247/full/ (accessed December 20, 2021).
Crotti, N. 2019. More device shortages pegged to sterilization plant shutdown. https://www.medicaldesignandoutsourcing.com/more-device-shortages-pegged-to-sterilization-plant-shutdown/ (accessed October 21, 2021).
Dai, T., G. Bai, and G. F. Anderson. 2020. PPE supply chain needs data transparency and stress testing. Journal of General Internal Medicine 35:2748-2749. https://doi.org/10.1007/s11606-020-05987-9.
DoD (U.S. Department of Defense), HHS (U.S. Department of Health and Human Services), DHS (U.S. Department of Homeland Security), and VA (U.S. Department of Veterans Affairs). 2021. National strategy for a resilient public health supply chain, edited by Department of Health and Human Services, Department of Defense, Department of Homeland Security, Department of Commerce, Department of State, Department of Veterans Affairs, and The White House Office of the COVID-19 Response. Washington, DC. https://www.phe.gov/Preparedness/legal/Documents/National-Strategy-for-Resilient-Public-Health-Supply-Chain.pdf (accessed October 29, 2021).
Edney, A. 2020. Hurricane season could threaten saline solution production. Bloomberg. https://www.bloomberg.com/news/articles/2020-07-16/hurricane-season-could-threaten-saline-solution-production (accessed October 25, 2021).
Evans, J., and J. Meisenheimer. 2020. COVID-19—suffocating the global medical supply chain while breathing life into its future. Site Selection Magazine, April.
Evstatieva, M. 2021. U.S. Companies shifted to make N95 respirators during COVID. Now, they’re struggling. https://www.npr.org/2021/06/25/1009858893/u-s-companies-shifted-to-make-n95-respirators-during-covid-now-theyre-struggling (accessed October 25, 2021).
Fairclough, G., and T. M. Burton. 2008. China’s role in supply of drug is under fire. https://www.wsj.com/articles/SB120354600035281041 (accessed October 25, 2021).
Fareed, J., W. Jeske, and E. Ramacciotti. 2019. Porcine mucosal heparin shortage crisis! What are the options? Clinical and Applied Thrombosis/Hemostasis 25:1076029619878786.
Farrell, N. M., B. D. Hayes, and J. A. Linden. 2021. Critical medication shortages further dwindling hospital resources during COVID-19. The American Journal of Emergency Medicine 40:202-203.
FDA (U.S. Food and Drug Administration). 2013. Strategic plan for preventing and mitigating drug shortages. Silver Spring, MD: FDA. https://www.fda.gov/media/86907/download (accessed December 20, 2021).
FDA. 2017. Entry screening systems and tools. https://www.fda.gov/industry/import-systems/entry-screening-systems-and-tools#screened (accessed January 19, 2022).
FDA. 2019a. FDA encourages reintroduction of bovine-sourced heparin. https://www.fda.gov/drugs/pharmaceutical-quality-resources/fda-encourages-reintroduction-bovine-sourced-heparin (accessed October 25, 2021).
FDA. 2019b. FDA in brief: FDA announces new efforts to expedite generic drug development and marketing to improve patient access to medicines. Silver Spring, MD: FDA. https://www.fda.gov/news-events/fda-brief/fda-brief-fda-announces-new-efforts-expedite-generic-drug-development-and-marketing-improve-patient (accessed December 16, 2021).
FDA. 2020a. Current good manufacturing practice (CGMP) regulations. https://www.fda.gov/drugs/pharmaceutical-quality-resources/current-good-manufacturing-practice-cgmp-regulations (accessed July 22, 2021).
FDA. 2020b. Ethylene oxide sterilization for medical devices. https://www.fda.gov/medical-devices/general-hospital-devices-and-supplies/ethylene-oxide-sterilization-medical-devices#actions (accessed October 21, 2021).
FDA. 2021a. Activities report of the generic drugs program | GDUFA II quarterly performance. https://www.fda.gov/industry/generic-drug-user-fee-amendments/activities-report-generic-drugs-program-gdufa-ii-quarterly-performance (accessed October 22, 2021).
FDA. 2021b. FAQs on shortages of surgical masks and gowns during the COVID-19 pandemic. https://www.fda.gov/medical-devices/personal-protective-equipment-infection-control/faqs-shortages-surgical-masks-and-gowns-during-covid-19-pandemic (accessed December 6, 2021, 2021).
FDA. 2021c. Using ventilator splitters during the COVID-19 pandemic—letter to health care providers. https://www.fda.gov/medical-devices/letters-health-care-providers/using-ventilator-splitters-during-covid-19-pandemic-letter-health-care-providers (accessed October 21, 2021).
FDA Drug Shortages Task Force. 2020. Drug shortages: Root causes and potential solutions. https://www.fda.gov/drugs/drug-shortages/report-drug-shortages-root-causes-and-potential-solutions (accessed December 16, 2021).
FiercePharma. 2010. Teva to stop making propofol. https://www.fiercepharma.com/manufacturing/teva-to-stop-making-propofol (accessed December 16, 2021).
Florko, N. 2020. New document reveals scope and structure of Operation Warp Speed and underscores vast military involvement. STAT, September 28.
Fox, E. R., and M. M. McLaughlin. 2018. ASHP guidelines on managing drug product shortages. American Journal of Health-System Pharmacy 75(21):1742-1750.
Fox, E. R., A. Birt, K. B. James, H. Kokko, S. Salverson, and D. L. Soflin. 2009. ASHP guidelines on managing drug product shortages in hospitals and health systems. American Journal of Health-System Pharmacy 66(15):1399-1406.
Fox, E. R., B. V. Sweet, and V. Jensen. 2014. Drug shortages: A complex health care crisis. Mayo Clinic Proceedings 89(3):361-373.
Fry, E. 2015. There’s a national shortage of saline solution. Yeah, we’re talking salt water. Huh? https://fortune.com/2015/02/05/theres-a-national-shortage-of-saline/ (accessed October 25, 2021).
GAO (Government Accountability Office). 2010. FDA response to heparin contamination. http://www.gao.gov/new.items/d1195.pdf (accessed December 16, 2021).
Goldberg, S. 2019. Sterigenics to close suburban Chicago plant. https://www.modernhealthcare.com/safety/sterigenics-close-suburban-chicago-plant (accessed October 21, 2021).
Goodnough, A. 2020. Some hospitals are close to running out of crucial masks for coronavirus. New York Times, March 9.
Goozner, M. 2012. Drug shortages delay cancer clinical trials. Journal of the National Cancer Institute 104(12):891-892.
Grimm, C. A. 2020. Hospital experiences responding to the COVID-19 pandemic: Results of a national pulse survey March 23–27, 2020. U.S. Department of Health and Human Services, Office of Inspector General.
Hantel, A. 2014. A protocol and ethical framework for the distribution of rationed chemotherapy. Journal of Clinical Ethics 25(2):102-115.
Hantel, A., M. Siegler, F. Hlubocky, K. Colgan, and C. K. Daugherty. 2019. Prevalence and severity of rationing during drug shortages: A national survey of health system pharmacists. JAMA Internal Medicine 179(5):710-711.
Herper, M. 2001. Cipro, anthrax and the perils of patents. Forbes, October 17.
Hopp, W. J. 2008. Supply chain science. New York: McGraw-Hill Irwin.
Hsia, I. K., F. Dexter, I. Logvinov, N. Tankosic, H. Ramakrishna, and S. J. Brull. 2015. Survey of the national drug shortage effect on anesthesia and patient safety: A patient perspective. Anesthesia & Analgesia 121(2):502-506.
Hufford, A. 2020a. 3M CEO on N95 masks: ‘Demand exceeds our production capacity’. The Wall Street Journal, April 2.
Hufford, A. 2020b. Why are N95 masks so important? The Wall Street Journal, June 1.
Hufford, A., and M. Evans. 2020. Coronavirus outbreak strains global medical-mask market. The Wall Street Journal, February 6.
Institute for Safe Medication Practices. 2012. A shortage of everything except errors: Harm associate with drug shortages. ISMP. https://www.ismp.org/resources/shortage-everything-except-errors-harm-associated-drug-shortages (accessed Feburary 12, 2021).
Institute for Safe Medication Practices. 2018. Drug shortages continue to compromise patient care. https://www.ismp.org/resources/drug-shortages-continue-compromise-patient-care (accessed March 1, 2021).
IOM (Institute of Medicine). 2009. Guidance for establishing crisis standards of care for use in disaster situations: A letter report, edited by B. M. Altevogt, C. Stroud, S. L. Hanson, D. Hanfling and L. O. Gostin. Washington, DC: The National Academies Press.
Jacobs, A. 2021. Can’t find an N95 mask? This company has 30 million that it can’t sell. The New York Times, February 10.
Jacobs, A., N. E. Boudette, M. Richtel, and N. Kulish. 2020. Amid desperate need for ventilators, calls grow for federal intervention. The New York Times, March 25.
Jacobsen, J. 2020. 3M responds to Trump’s invocation of Defense Production Act to obtain N95 masks. https://www.kare11.com/article/news/health/coronavirus/pres-trump-invokes-defense-production-act-to-obtain-n95-masks-from-3m/89-d3c72df1-a9d2-445f-93af-b49291b14a0f (accessed October 25, 2021.
Jensen, V., and B. A. Rappaport. 2010. The reality of drug shortages — The case of the injectable agent propofol. New England Journal of Medicine 363(9):806-807.
Jung, A. 2020. COVID-19: Risks and resiliency in the drug supply chain. https://www.ey.com/en_us/strategy-transactions/covid-19-risks-and-resiliency-in-the-drug-supply-chain (accessed January 5, 2021).
Kaakeh, R., B. V. Sweet, C. Reilly, C. Bush, S. DeLoach, B. Higgins, A. M. Clark, and J. Stevenson. 2011. Impact of drug shortages on U.S. health systems. American Journal of Health-System Pharmacy 68(19):1811-1819.
Kerr, W., and H. Schmidt. 2021. COVID-19 ventilator rationing protocols: Why we need to know more about the views of those with most to lose. Journal of Medical Ethics 47(3):133-136.
Khazan, O. 2020. Why we’re running out of masks. https://www.theatlantic.com/health/archive/2020/04/why-were-running-out-of-masks-in-the-coronavirus-crisis/609757/ (accessed October 25, 2021).
King, R. 2020. HHS defends falling short of PPE goals on Strategic National Stockpile as some shortages linger. https://www.fiercehealthcare.com/hospitals/hhs-defends-falling-short-ppe-goals-strategic-national-stockpile-as-some-shortages-linger (accessed October 25, 2021).
Kishimoto, T. K., K. Viswanathan, T. Ganguly, S. Elankumaran, S. Smith, K. Pelzer, J. C. Lansing, N. Sriranganathan, G. Zhao, Z. Galcheva-Gargova, A. Al-Hakim, G. S. Bailey, B. Fraser, S. Roy, T. Rogers-Cotrone, L. Buhse, M. Whary, J. Fox, M. Nasr, G. J. Dal Pan, Z. Shriver, R. S. Langer, G. Venkataraman, K. F. Austen, J. Woodcock, and R. Sasisekharan. 2008. Contaminated heparin associated with adverse clinical events and activation of the contact system. New England Journal of Medicine 358(23):2457-2467.
Langreth, R., and C. Koons. 2017. This simple liquid is suddenly in short supply. https://www.bloomberg.com/news/articles/2017-11-14/this-simple-lifesaving-liquid-is-suddenly-in-short-supply (accessed October 25, 2021).
Le, P., L. Grund, J. Marwa, W. Ojo, J. Otts, Jr., and F. Arab. 2018. Combating substandard and counterfeit medicines by securing the pharmaceutical supply chain: The Drug Supply Chain Security Act (DSCSA) of 2013. Innovations in Pharmacy 9(2):1-11.
Lee, J. 2014. Growing IV saline shortage has providers scrambling during bad flu season. https://www.modernhealthcare.com/article/20140123/NEWS/301239970/growing-iv-saline-shortage-has-providers-scrambling-during-bad-flu-season (accessed October 25, 2021).
Lim, D. 2019. Ethylene oxide plant closures put us on ‘cusp of a major medical logistical failure’. https://www.medtechdive.com/news/ethylene-oxide-plant-closures-place-united-states-on-cusp-major-medical-logistical-failure/566922/ (accessed October 21, 2021).
Linskey, A., J. Dawsey, I. Stanley-Becker, and C. Janes. 2020. As feds play ‘backup,’ states take unorthodox steps to compete in cutthroat global market for coronavirus supplies. The Washington Post, April 11.
Lloyd, J., and J. Cheyne. 2017. The origins of the vaccine cold chain and a glimpse of the future. Vaccine 35(17):2115-2120.
Manufacturing x Digital (MxD) and the International Academy of Automation Engineering (IAAE). 2021. Analysis of the advantages of and barriers to adoption of smart manufacturing for medical products. Food and Drug Administration. https://www.fda.gov/media/152568/download (accessed January 27, 2022).
Marsa, L. 2020. Tackling dangerous drug shortages. https://health.usnews.com/health-care/patient-advice/articles/tackling-dangerous-drug-shortages (accessed October 25, 2021).
Marucheck, A., N. Greis, C. Mena, and L. Cai. 2011. Product safety and security in the global supply chain: Issues, challenges and research opportunities. Journal of Operations Management 29(7):707-720.
Maxwell, Y. L. 2019. FDA warns of potential medical device shortage due to sterilization woes. https://www.tctmd.com/news/fda-warns-potential-medical-device-shortage-due-sterilization-woes (accessed October 21, 2021).
Mazer-Amirshahi, M., and E. R. Fox. 2018. Saline shortages—Many causes, no simple solution. New England Journal of Medicine 378(16):1472-1474.
McBride, A., L. M. Holle, C. Westendorf, M. Sidebottom, N. Griffith, R. J. Muller, and J. M. Hoffman. 2013. National survey on the effect of oncology drug shortages on cancer care. American Journal of Health-System Pharmacy 70(7):609-617.
McCarthy, C. P., M. Vaduganathan, E. Solomon, R. Sakhuja, G. Piazza, D. L. Bhatt, J. M. Connors, and N. K. Patel. 2020. Running thin: Implications of a heparin shortage. Lancet 395(10223):534-536.
McLaughlin, M., D. Kotis, K. Thomson, M. Harrison, G. Fennessy, M. Postelnick, and M. H. Scheetz. 2013. Effects on patient care caused by drug shortages: A survey. Journal of Managed Care Pharmacy 19(9):783-788.
Mentzer, J. T., W. DeWitt, J. S. Keebler, S. Min, N. W. Nix, C. D. Smith, and Z. G. Zacharia. 2001. Defining supply chain management. Journal of Business Logistics 22(2):1-25.
Mullen, R. 2020. COVID-19 is reshaping the pharmaceutical supply chain. Chemical and Engineering News 98(16):10.
Murray, B., and S. Bradley. 2021. The semiconductor chip shortage hits medtech: Strategies to build resilient supply chains. https://www.advamed.org/2021/09/23/the-semiconductor-chip-shortage-hits-medtech-strategies-to-build-resilient-supply-chains/ (accessed October 21, 2021).
NASEM (National Academies of Science, Engineering, and Medicine). 2018. Medical product shortages during disasters: Opportunities to predict, prevent, and respond: Proceedings of a workshop—in brief, edited by A. Nicholson, L. Runnels, C. Giammaria, L. Brown, and S. Wollek. Washington, DC: The National Academies Press.
NASEM. 2020. Strengthening post-hurricane supply chain resilience: Observations from Hurricanes Harvey, Irma, and Maria. Washington, DC: The National Academies Press.
NASEM. 2021. The security of America’s medical product supply chain: Considerations for critical drugs and devices: Proceedings of a workshop—In brief, edited by A. Nicholson, E. Randall, L. Brown, C. Shore, and B. Kahn. Washington, DC: The National Academies Press.
Nirappil, F., and W. Wan. 2021. Los Angeles is running out of oxygen for patients as COVID hospitalizations hit record highs nationwide. The Washington Post, April 28.
Noguchi, Y. 2021. Why N95 masks are still in short supply in the U.S. Washington, DC: NPR. https://www.npr.org/sections/health-shots/2021/01/27/960336778/why-n95-masks-are-still-in-short-supply-in-the-u-s (accessed December 16, 2021).
Paavola, A. 2017. 4 ways the FDA is addressing the saline bag shortage. https://www.beckershospitalreview.com/supply-chain/4-ways-the-fda-is-addressing-the-saline-bag-shortage.htm (accessed October 25, 2021).
Page, P. 2021. Container shipping prices skyrocket as rush to move goods picks up. https://www.wsj.com/articles/container-ship-prices-skyrocket-as-rush-to-move-goods-picks-up-11625482800 (accessed October 22, 2021).
Palmer, E. 2019. B. Braun commits $1B to expand in face of IV fluid shortages. https://www.fiercepharma.com/manufacturing/b-braun-commits-1b-to-expand-face-iv-fluid-shortages (accessed October 29, 2021).
Parshley, L. 2020. The mask shortage is forcing health workers to disregard basic coronavirus infection control. https://www.vox.com/2020/4/3/21206726/coronavirus-masks-n95-hospitals-health-care-doctors-ppe-shortage (accessed October 25, 2021).
Pascu, G. A., G. Hancu, and A. Rusu. 2020. Pharmaceutical serialization, a global effort to combat counterfeit medicines. Acta Marisiensis-Seria Medica 66(4):132-139.
Patton, D. 2021. Analysis: African Swine Fever inflicts renewed toll on northern China’s hog herd. https://www.reuters.com/business/healthcare-pharmaceuticals/african-swine-fever-inflicts-renewed-toll-northern-chinas-hog-herd-2021-04-01/ (accessed October 25, 2021).
The PEW Charitable Trusts and International Society for Pharmaceutical Engineering. 2017. Drug shortage: An exploration of the relationship between U.S. market forces and sterile injectable pharmaceutical products—Interviews with 10 pharmaceutical companies. PEW and ISPE. https://www.pewtrusts.org/-/media/assets/2017/01/drug_shortages.pdf (accessed October 25, 2021).
The PEW Health Group. 2011. After heparin: Protecting consumers from the risks of substandard and counterfeit drugs. https://www.pewtrusts.org/en/research-and-analysis/reports/2011/07/12/after-heparin-protecting-consumers-from-the-risks-of-substandard-and-counterfeit-drugs (accessed October 25, 2021).
Phuong, J. M., J. Penm, B. Chaar, L. D. Oldfield, and R. Moles. 2019. The impacts of medication shortages on patient outcomes: A scoping review. PLoS ONE 14(5):e0215837.
Piscitello, G. M., E. M. Kapania, W. D. Miller, J. C. Rojas, M. Siegler, and W. F. Parker. 2020. Variation in ventilator allocation guidelines by US state during the Coronavirus Disease 2019 pandemic: A systematic review. JAMA Network Open 3(6):e2012606.
Ranney, M. L., V. Griffeth, and A. K. Jha. 2020. Critical supply shortages—the need for ventilators and personal protective equipment during the COVID-19 pandemic. New England Journal of Medicine 382(18):e41.
Raymond, N. 2019. U.S. Closes IV solution shortage antitrust probe, Baxter says. https://www.reuters.com/article/us-baxter-intl-antitrust-idUSKCN1QB25K (accessed October 25, 2021).
Rockefeller, J. D., T. Harkin, and E. E. Cummings. 2012. Shining light on the “gray market”: An examination of why hospital are forced to pay exorbitant prices for prescription drugs facing critical shortages. https://www.commerce.senate.gov/services/files/AFA98935-2FF5-4004-88DC-BE70D1C22B5D (accessed December 16, 2021).
Sacks, C. A., A. S. Kesselheim, and M. Fralick. 2018. The shortage of normal saline in the wake of Hurricane Maria. JAMA Intern Medicine 178(7):885-886.
Sathya, D. 2021. How the N95 masks shortage evolved to a supply-demand disconnect. https://getusppe.org/how-the-n95-masks-shortage-evolved-to-a-supply-demand-disconnect/ (accessed October 24, 2021).
Schondelmeyer, S. W., J. Seifert, D. J. Margraf, M. Mueller, I. Williamson, C. Dickson, D. Dasararaju, C. Caschetta, N. Senne, and M. T. Osterholm. 2020. Part 6: Ensuring a resilient us prescription drug supply. In COVID-19: The CIDRAP viewpoint, edited by J. Wappes. Minneapolis, MN: Regents of the University of Minnesota. https://www.cidrap.umn.edu/sites/default/files/public/downloads/cidrap-covid19-viewpoint-part1_0.pdf (accessed December 16, 2021).
Scutti, S. 2018. IV bags in short supply across US after Hurricane Maria. https://www.cnn.com/2018/01/16/health/iv-bag-shortage/index.html (accessed October 25, 2021).
Senate Committee on Finance. 2020. COVID-19 and beyond: Oversight of the FDA’s foreign drug manufacturing inspection process: Congressional testimony of Judith A. McMeekin. June 2. https://www.fda.gov/news-events/congressional-testimony/covid-19-and-beyond-oversight-fdas-foreign-drug-manufacturing-inspection-process-06022020 (accessed January 25, 2022).
Shaban, H., C. Maurer, and R. J. Willborn. 2018. Impact of drug shortages on patient safety and pharmacy operation costs. Federal Practitioner for the Health Care Professionals of the VA, DoD, and PHS 35(1):24-31.
Sheridan, K. 2021. How blackouts, fires, and a pandemic are driving shortages of pipette tips—and hobbling science. https://www.statnews.com/2021/04/28/pipette-tips-shortage/ (accessed April 30, 2021).
Socal, M. P., J. M. Sharfstein, and J. A. Greene. 2020. Critical drugs for critical care: Protecting the US pharmaceutical supply in a time of crisis. American Journal of Public Health 110(9):1346-1347.
Sullivan, T. 2018. FDA under pressure to speed up generic approvals. https://www.policymed.com/2017/04/fda-under-pressure-to-speed-up-generic-approvals.html (accessed October 25, 2021).
Szajek, A., E. Gray, D. Keire, B. Mulloy, A. A. Hakim, C. Chase, M. Da Luz Carvalho Soares, D. Cairatti, J. Hogwood, P. Mourão, and T. S. Morris. 2015. Diversifying the global heparin supply chain: Reintroduction of bovine heparin in the United States? https://www.pharmtech.com/view/diversifying-global-heparin-supply-chain-reintroduction-bovine-heparin-united-states (accessed October 25, 2021).
Tolomeo, D., K. Hirshfield, and D. L. Hustead. 2020. Engage with health authorities to mitigate & prevent drug shortages. In ISPE Special Reports. ISPE. https://ispe.org/pharmaceutical-engineering/july-august-2020/engage-health-authorities-mitigate-prevent-drug (accessed October 25, 2021).
Truog, R. D., C. Mitchell, and G. Q. Daley. 2020. The toughest triage—Allocating ventilators in a pandemic. New England Journal of Medicine 382(21):1973-1975.
Tucker, E. L., Y. Cao, E. R. Fox, and B. V. Sweet. 2020. The drug shortage era: A scoping review of the literature 2001–2019. Clinical Pharmacology & Therapeutics 108(6):1150-1155.
UNICEF. n.d. What is a cold chain? https://www.unicef.org/supply/what-cold-chain (accessed October 25, 2021).
Usdin, S. 2009. The heparin story. International Journal of Risk & Safety in Medicine 21(1-2):93-103.
Vail, E., H. B. Gershengorn, M. Hua, A. J. Walkey, G. Rubenfeld, and H. Wunsch. 2017. Association between us norepinephrine shortage and mortality among patients with septic shock. JAMA 317(14):1433-1442.
van der Meer, J. Y., E. Kellenbach, and L. J. van den Bos. 2017. From farm to pharma: An overview of industrial heparin manufacturing methods. Molecules 22(6):1025.
Ventola, C. L. 2011. The drug shortage crisis in the United States: Causes, impact, and management strategies. Pharmacy and Therapeutics 36(11):740-757.
Vilanova, E., A. M. F. Tovar, and P. A. S. Mourao. 2019. Imminent risk of a global shortage of heparin caused by the African Swine Fever afflicting the Chinese pig herd. Journal of Thrombosis and Haemostasis 17(2):254-256.
Vizient. 2019. Drug shortages and labor costs mesuring the hidden costs of drug shortages on U.S. hospitals. In Vizient Drug Shortages Impact Report. Irving, TX: Vizient Inc. https://www.vizientinc.com/-/media/documents/sitecorepublishingdocuments/public/vzdrugshortageslaborcost_fullreport.pdf?la=en&hash=19172F274A8B03817E042C2EF423BED98CDF0401 (accessed December 20, 2021).
Ward, N. 2019. Crystalloids: An overview. https://hospitalhealthcare.com/clinical/albumin/2019-focus/crystalloids-an-overview/ (accessed October 25, 2021).
Weber, L. 2018. Hurricane Maria’s effect on the health care industry is threatening lives across the U.S. https://www.huffpost.com/entry/iv-bag-drug-shortage-puerto-rico-hurricane-maria_n_5ba1ca16e4b046313fc07a8b (accessed October 25, 2021).
Weber, L., and Kaiser Health News. 2020. State mask stockpiling orders are hurting nursing homes, small clinics. https://www.nbcnews.com/news/us-news/state-mask-stockpiling-orders-are-hurting-nursing-homes-small-clinics-n1245395 (accessed October 25, 2021).
Wendelbo, M., and C. C. Blackburn. 2018. A saline shortage this flu season exposes a flaw in our medical supply chain. https://www.smithsonianmag.com/innovation/saline-shortage-this-flu-season-exposes-flaw-in-our-medical-supply-chain-180967879/ (accessed October 25, 2021).
Weschsler, J. 2001. Bioterrorism threat shines spotlight on drug manufacturing. Pharmaceutical Technology 25(12):14-22. https://cdn.sanity.io/files/0vv8moc6/pharmtech/69918b66616ce9011d3997e5ec39c710fdd68eab.pdf/article-3397.pdf (accessed February 15, 2022).
Westly, E. 2011. A more modern blood thinner. https://www.technologyreview.com/2011/10/28/189986/a-more-modern-blood-thinner/ (accessed October 25, 2021).
The White House. 2021. Building resilient supply chains, revitalizing American manufacturing, and fostering broad-based growth. Department of Commerce, Department of Energy, Department of Defense, Department of Health and Human Services. https://www.whitehouse.gov/wp-content/uploads/2021/06/100-day-supply-chain-review-report.pdf (accessed October 25, 2021).
WHO (World Health Organization). 2020. Shortage of personal protective equipment endangering health workers worldwide. https://www.who.int/news/item/03-03-2020-shortage-of-personal-protective-equipment-endangering-health-workers-worldwide (accessed October 25, 2021).
Woo, J., Wolfgang, S., Batista, H. 2008. The effect of globalization of drug manufacturing, production, and sourcing and challenges for American drug safety. Clinical Pharmacology & Therapeutics 83(3):494-497.
Woodcock, J., and M. Wosinska. 2013. Economic and technological drivers of generic sterile injectable drug shortages. Clinical Pharmacology and Therapeutics 93(2):170-176.
Woodcock, J., and M. Kopcha. 2020. Quality: The often overlooked critical element for assuring access to safe and effective drugs. HealthAffairs. https://www.healthaffairs.org/do/10.1377/hblog20200311.912049/full/ (accessed October 19, 2021).
Zhu, Y., F. Zhang, and R. J. Linhardt. 2019. Heparin contamination and issues related to raw materials and controls. In The science and regulations of naturally derived complex drugs. Cham, Switzerland: Springer. Pp. 191-206.
This page intentionally left blank.








































