5
A Framework for Resilient Medical Product Supply Chains
As described in Chapter 1, the primary goal of resilient medical product supply chains is to prevent public health and safety from being compromised by shortages of medical products. However, because resources are limited, costs must be considered. Excess spending to enhance resilience of one supply chain may mean inadequate resources for improving resilience of another supply chain or other actions to promote public health and safety. Therefore, the aim must be to find a cost-effective mix of measures to promote a socially desirable level of medical product supply chain resilience.
This chapter presents a framework for increasing the resilience of medical product supply chains. First, the chapter describes a method for determining which medical products are supply chain critical, and therefore in need of resilience interventions. Second, the chapter explains a process for determining the level of protection needed for a given product on the supply chain critical list. And third, the chapter shows how to use the framework for systematically enumerating options to enhance supply chain resilience for a given medical product.
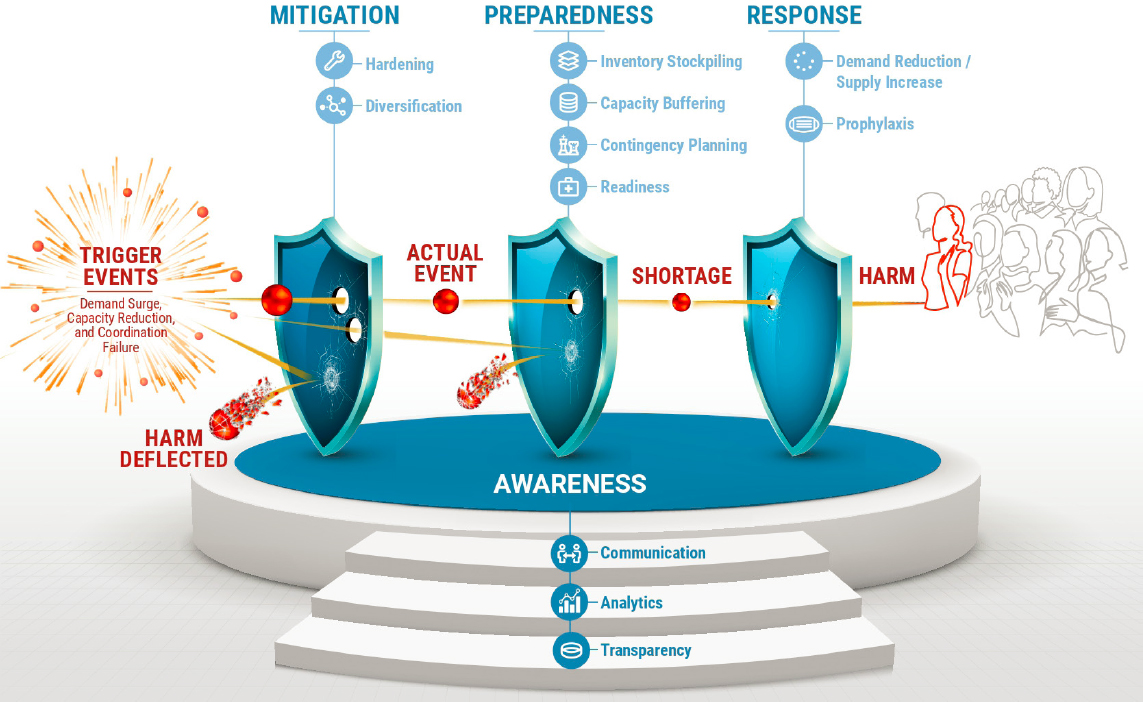
The resulting framework is depicted in Figure 5-4, which shows that policies for enhancing medical product supply chain resilience can be classified into four categories: awareness, mitigation, preparedness, and response. Although this framework is described as if it will be used to analyze and remediate each product individually, it can also be used to gain insights into general policies, such as information disclosure requirements, that affect many medical products. The framework will be used in this latter context in Chapters 6–9 to justify several specific recommendations that address the most significant gaps between current and desired protection levels. Taken
together, these recommendations form the basis for an integrated strategy to build resilience of critical medical product supply chains.
DEFINING RESILIENCE FOR MEDICAL PRODUCT SUPPLY CHAINS
In Chapter 1, supply chain critical medical products were defined as those that are both medically essential and vulnerable to supply chain shortages. A clear definition of resilience is needed to identify which medical products warrant supply chain resilience interventions. This section refines and quantifies a definition of resilience in medical product supply chains using a standardized procedure that takes into account past data and future forecasts to provide reasonable estimates. This should be adequate for focusing attention on the products for which combined clinical and supply chain risks justify intervention. Toward this end, the subscript (i) is used as a product index in the following definitions:
These definitions allow the total expected harm from all medical products to be expressed as the sum of the individual product risks:
___________________
1 Health and financial costs could be combined by attaching a dollar value to QALYs. However, since the model is being used conceptually, rather than computationally, it will focus only on human health for simplicity.
2 Note that (HiSi) is mathematical shorthand for the multiplication of (Hi) and (Si), or (Hi í Si) (Bergman, n.d.).
| Total Expected Harm = ΣiRi = ΣiHiΣi | (Equation 5-1) |
If the supply chain resilience problem is framed as one of reducing total expected harm by as much as possible for a given level of investment, Equation 5-1 indicates that focus should be on products with high Hi Si values—particularly if they are amenable to inexpensive intervention. Products with high Hi Si values are of concern because they present substantial clinical and supply chain risks. Therefore, such products should be on the supply chain critical list.
However, there are two reasons why the supply chain critical list should not be limited to the products with the highest Hi Si values, both of which have to do with the fact that Hi and Si are expectations. First, the expected value Hi could be low because product i has very few users, even if the medical importance of product i to each user is very great. For example, a drug for treating a rare cancer may be essential for the patients using it, but a shortage might result in the loss of relatively few QALYs because of the small patient population. Leaving such a product off the supply chain critical risk would cause these patients to bear an undue portion of the total risk, violating equity. Furthermore, protecting these patients from harm might be relatively inexpensive because a small stockpile would cover demand for a significant time period. Therefore, it is appropriate to include products that are extremely medically essential on the supply chain critical risk, even if the expected harm from a shortage is low.
Similarly, the expected value Si could be low because the events that would trigger a shortage are extremely unlikely. Bioterror and nuclear attacks are examples of events that are unlikely but serious. Furthermore, extremely rare events are precisely the ones for which estimating likelihoods is most difficult, which means expectations will be subject to error. For these reasons, the expected outcome is not a very helpful characterization of highly unlikely events with extreme consequences. Products with such unlikely, but large, risks warrant consideration of mitigation measures. However, unlike the case of an essential drug for a small population, where protection costs are likely to be low, the cost of protection against a large but unlikely event could be high because of the volumes of product required. Therefore, the assessment of whether the risks justify the costs of protection will be more nuanced and difficult than when considering essential but small market products.
The conclusion here is that highlighting medical products with high Hi Si values is a good start to developing a supply chain critical list. But equity and extreme risk considerations must be factored in. Including products that are vital to life and without viable alternatives is important from an equity perspective, while including products that are subject to unlikely but extreme events is important from a security standpoint. For the remainder of this chapter, it will be assumed that these nuanced assessments
have been made and that a list of supply chain critical medical products has been generated.
Individual Medical Product Targets
The next step is to determine the socially desirable level of protection needed for each medical product on the supply chain critical list. To do this, a characterization of the distribution of shortages a product might face is needed in addition to the expected shortage Si. If shortages are measured in weeks of supply, such a distribution would look something like Figure 5-1—which depicts the likelihood that a disruption, given that one occurs, will result in a shortage equivalent to n weeks of supply for various values of n. As n grows larger, the probability of a disruption lasting n weeks approaches 1 (or 100 percent). Indeed, if it is assumed that capacity could be repaired, expanded, or replaced within n weeks, then the cumulative distribution will reach 1 (100 percent) at n.
Getting data to construct a shortage distribution like that in Figure 5-1 is nontrivial. Options for obtaining and using such data are described in Appendix C. These include using statistical analysis of past disruptions for products, such as generic drugs, that are prone to routine disruption by process failures, and subjective scenario analysis of disruptions by major emergencies such as pandemics. It is important to note, however, that the goal of such analysis is not precise characterization of shortage distributions. Rather it is to provide a sense of the range of shortage risks a product
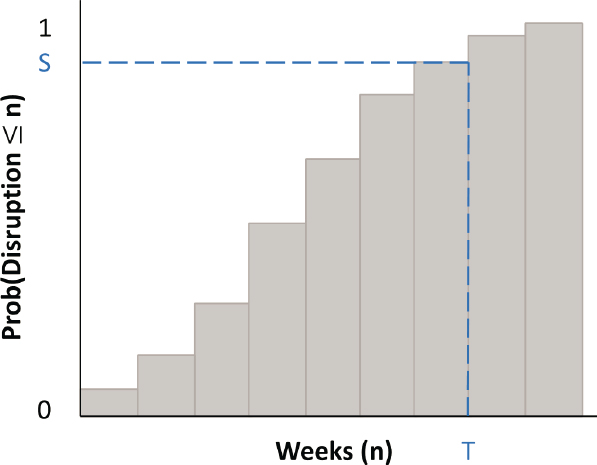
faces so that these risks can be addressed in a balanced manner across the medical products on the supply chain critical list.
Figure 5-1 helps describe the fundamental trade-off that must be considered when determining an appropriate protection target. Suppose for the sake of discussion that one is limited to using an inventory stockpile as protection against a supply shortage for a given product. As the weeks of supply (indicated by T) increase in the stockpile, the protection (defined as the probability of having enough stock to offset a shortage and indicated by S) will also increase. The stockpiling cost will increase in proportion to the amount of inventory. However, while the cost of inventory will increase linearly, the protection will increase at a decreasing rate as it gets closer and closer to 100 percent. This in turn implies that the harm (measured in QALYs) avoided becomes increasingly expensive as the protection level increases. In economics terms, investments in supply chain protection will exhibit diminishing returns to scale.
This diminishing returns insight is essential to achieving a balanced supply chain resilience strategy. For example, imagine that there are stocks of two products in the Strategic National Stockpile (SNS) that are clinically similar (i.e., have similar Hi values) and have similar unit costs (and therefore similar costs to carry inventory). However, after evaluating shortage scenarios, it is determined that the stock level of the first product is sufficient to provide a protection level of 75 percent, while the stock level for the second product is enough for a protection level above 99 percent. When this is the case, increasing the stockpile of the first product, while decreasing the stockpile of the second product to keep the cost constant, will increase overall protection. The reason is that the diminishing returns property implies the added investment in the first product stockpile will result in a relatively large increase in the number of QALYs, while the reduced investment in the second product stockpile will result in a much smaller reduction in the number of QALYs.
The point here is not that inventory of one product should literally be sold to buy inventory of another product. Rather, it is that protection targets should be set consistently in a manner that accounts for clinical criticality, product cost, and disruption risks.3Appendix C offers additional details on this calculation. But providing a comprehensive manual for setting stockpile
___________________
3 Investing in measures to protect against medical supply shortages is directly analogous to buying insurance. Increasing the amount of insurance (i.e., the payout limit) protects us against increasingly unlikely events (e.g., large liability assessments). Just as we reasonably choose different levels of insurance protection against different types of losses (e.g., we might want greater coverage of medical expenses than of property damage), we should set different protection targets for medical products with different implications for health and safety. But when insuring against comparable risks (e.g., liability coverage for different automobiles), protection levels should be consistent. The same is true in a balanced supply chain resilience strategy.
levels is beyond the scope of this report. Therefore, for the remainder of this chapter, it will be assumed that sensible protection targets for the products on the list of supply chain critical medical products have been set. This poses the most important question addressed in this chapter, which is how to systematically identify potential actions for achieving these targets.
MULTILAYERED PROTECTION
The above discussion of protection targets used inventory stockpiling to illustrate the trade-offs involved in investments to protect people from the consequences of medical product shortages. But stockpiling is only one of a wide array of policies that could be used as protection. In order to enumerate options in a systematic way, a framework is needed. To construct one, the committee notes that building resilience into medical product supply chains is an example of a reliability problem. Admittedly, it is a complicated and difficult reliability problem because there are many resilience options available that fit together in intricate ways. There are three major reasons for this:
- Medical product supply chains are complex systems that involve people, processes, technologies, and policies. Consequently, their resilience can be addressed in many ways by focusing on different aspects of the system.
- The Redundancy Principle, which states that “independent layers of protection increase the reliability of a system” (Hopp and Lovejoy, 2012), implies that multiple safeguards are useful in achieving a high level of reliability.
- Interventions to improve resilience can be made at different points in the timeline because disruptive events and their consequences play out over time. Consequently, these interventions can complement one another by addressing public health and safety in a time-phased manner.
James Reason (2000) introduced the Swiss cheese model as a graphical illustration of the Redundancy Principle (Figure 5-2). In this model, slices of cheese represent layers of defense against a hazard causing harm or losses. Just as Swiss cheese has holes, all defense systems (e.g., alarms, warning lights, checklists, and human oversight) are fallible. If a hazard penetrates all the layers of defense (slices of cheese), harm will occur (Box 5-1).
In medical product supply chains, a disruptive trigger event will only result in harm to people if multiple layers of defense fail. For example, if a drug manufacturer has a quality problem that interrupts production (failure of layer 1), there is insufficient inventory in the supply chain (failure of layer 2), there are no other production facilities with additional capacity (failure of layer 3), and health systems have no substitution strategy (fail-
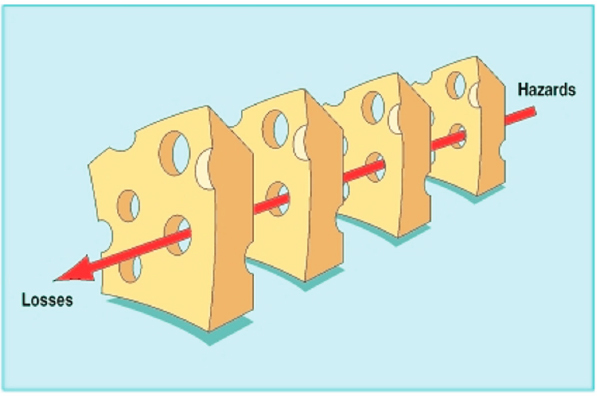
SOURCE: Reason, 2000. Reproduced from Human error: Models and management, James Reason, Vol 320, 769, ©2000 with permission from BMJ Publishing Group Ltd.
ure of layer 4), patients could be harmed. Invoking the Swiss cheese model description of the Redundancy Principle, the resilience of this supply chain can be improved by adding more layers of defense (more slices of cheese) or by improving the effectiveness of individual layers (fewer holes).

SOURCE: Reason, 1997. Copyright (© 1997) From Managing the risks of organizational accidents by James Reason. Reproduced by permission of Taylor and Francis Group, LLC, a division of Informa plc.
It is important to note that additional layers of defense only improve reliability if they are at least partially independent. In the Swiss cheese analogy, an additional slice of cheese whose holes align with those of an existing slice does not add any protection. In a real-world example, adding an additional fill-and-finish plant for a drug will do little to improve reliability if it relies on the same source of active pharmaceutical ingredient (API) supply as all other plants and the API supplier is the main source of disruption risk. For medical product supply chains, which interventions are appropriate, independent, and compatible will depend on both the type of medical product and the disruptive event(s). The implication, therefore, is that different interventions will be appropriate to different medical product supply chains.
THE COMMITTEE’S MEDICAL PRODUCT SUPPLY CHAINS RESILIENCE FRAMEWORK
With the Swiss cheese representation of the Redundancy Principle in mind, the committee developed a framework for enumerating supply chain resilience interventions in a systematic manner. Because the steps that lead to a supply shortage play out over time, it is logical to consider options with respect to their position on the disruption timeline, from preknowledge of risks to postdisruption of medical product supply chains. Figure 5-4 depicts the path from a potential trigger event to public harm and shows the categories of ways to increase the resilience of medical product supply chains.
Clearly, the medical product supply chain resilience framework depicted in Figure 5-4 is a version of the Swiss cheese model of system reliability. The layers of defense (depicted here as shields, rather than cheese) are specifically related to medical product supply chains. These protective layers are grouped into mitigation, preparedness, and response—the standard phases of emergency response (FEMA, 2021). According to the Federal Emergency Management Agency (FEMA), mitigation takes place prior to an emergency and focuses on “preventing future emergencies or minimizing their effects” (FEMA, 2021). Preparedness measures also take place prior to an emergency and focus on “preparing to handle an emergency” (FEMA, 2021). Response measures take place during an emergency and focus on “responding safely to an emergency” (FEMA, 2021). FEMA also includes a fourth phase, recovery, which includes measures after an emergency. The recovery phase has been omitted here; however, a fourth category of awareness has been added and serves as the support base for the three layers of defense.
The basic sequence in the framework of Figure 5-4 is as follows. A trigger event occurs, which creates a potential event; for example, a fire breaks out in a pharmaceutical plant. Whether this potential event becomes an actual supply chain disruption will depend on the mitigation measures. Therefore, if the sprinkler system functions properly and puts out the fire before heat and flames damage the production process, the harm has been deflected and no disruption occurs. But if the fire is not stopped, and the plant is seriously damaged, production stops and an actual event begins. Whether disruptions become shortages affecting providers and patients depends on the preparedness measures in place. For instance, if there is sufficient inventory of the drug in emergency stockpiles to cover the shortfall until the plant is back online, then patients and providers do not see a shortage; otherwise, a shortage occurs. If that happens, the magnitude of the harm caused will depend on the response measures. Crisis standards of care that prioritize the drug for some patients and provide substitutes for others might reduce the health effects. Finally, mitigation, preparedness, and response measures all depend on awareness measures, which make information available to all decision makers.
Building increased resilience into supply chains depends on each of these four categories of actions, which are described below.
Awareness
As depicted in Figure 5-4, awareness is the foundation for resilience in medical product supply chains. Here, awareness is the possession by the appropriate people of the information needed to assess, mitigate, prepare for, and respond to risks of medical product shortages. This includes informa-
tion about where medical products come from, where in the system medical products are, and many other characteristics of medical product supply chains. Without being able to identify and evaluate risks that exist in medical product supply chains, actions cannot be taken to mitigate, prepare for, or respond to them. Hence, the awareness category includes actions that promote understanding of risks and vulnerabilities across the entire medical product supply chain ecosystem from raw material or component suppliers (e.g., makers of ingredients, subassemblies) to producers (e.g., final assembly plants, fill-and-finish facilities), to distributors (e.g., wholesalers), to providers (e.g., health systems, pharmacies, retailers), and finally to patients so the focus and priority can be given to the appropriate resilience efforts.
Awareness measures can be broken into three subcategories: (1) transparency activities that make data available—which includes both surveillance/collection to obtain data and disclosure to make it available; (2) analytics activities that process those data into useful information, which includes compilation, graphical display, statistical analysis, and any other data processing to reveal their meaning; and (3) communication activities, including report writing, database construction and management, and anything else that gets the information into the hands of the people responsible for mitigation, preparedness, and response.
An example of an awareness initiative involving all three subcategories is mapping the supply chains of drugs and devices from production to patient to develop a full picture of the capacity and diversification of production and the profile of inventories across the supply chain. This might reveal that certain drugs or devices have highly limited or concentrated steps that pose risks of disrupting the supply or preventing a response to a spike in demand.
Awareness measures can be promoted at all levels of the supply chain, as well as by the government and third parties that monitor and influence medical supply chains. For example, a potential source of vital information is a hospital pharmacy. This is typically where a health care system would first notice a shortage of a drug. To mitigate a drug shortage within their own hospital, the pharmacy team could inform prescribers of the issue and recommend they use alternative drugs; the team could also contact other suppliers for the product, substitute the prescribed medication, and update their formulary (Shukar et al., 2021). As such, this represents the medical product supply chain resilience framework in a microcosm, with the pharmacy promoting awareness that underpins multiple layers of protective measures.
Mitigation
This category includes actions taken prior to a disruptive event to avoid the event altogether or reduce its magnitude. Mitigation can be divided into
two subcategories: (1) hardening activities, that reduce the likelihood or magnitude of disruptive events within stages of the system, and (2) diversification activities, which create parallel versions of stages to reduce the risk of catastrophic failure. In terms of the medical product supply chain resilience framework, hardening strengthens the layers, by reducing the number of penetrating holes in a single shield, while diversification creates more layers of shields. For example, a possible hardening measure for a medical device production process that is prone to interruptions by quality control problems could be product redesign. By reducing the number of components and/or assembly steps, a redesign could reduce opportunities for error (holes in the cheese). An example of a diversification measure for the medical device could be to set up a second production line to share the production load. If one line encounters process problems, the other can continue producing and may even make up some or all of the lost production from the disrupted line.
Mitigation measures do not need to directly address the physical elements of supply chains. For example, regulatory action by the U.S. Food and Drug Administration (FDA) to require better quality process documentation by manufacturers of generic drugs could push manufacturers to scrutinize and improve their processes, thereby reducing the number of quality problems, and hence the number of shortages caused by recalls. FDA (FDA Drug Shortages Task Force, 2020) estimated that quality and process issues are responsible for 62 percent of drug shortages; therefore, steps to reduce these would have a significant effect on both routine drug shortages and shortages during major emergencies. In addition to preventing such shortages, better quality control processes could remediate problems more quickly, thereby reducing the duration of shortages that do occur.
Preparedness
This category includes actions taken prior to a disruptive event that will reduce negative effects on health and safety should an event occur. Preparedness can be grouped into four subcategories, two physical and two virtual. Physical preparedness measures include inventory buffering and capacity buffering, in which actual stock or productive capacity are held in readiness to fill a supply shortfall. Virtual preparedness measures include contingency planning, which establishes plans for dealing with specific scenarios, and readiness, which builds capabilities for dealing with scenarios without specific plans made in advance.
Like the shields in the medical product supply chain resilience framework, the subcategories of the preparedness category can be viewed in terms of their distance from the point where products are used by providers to treat patients. Finished product inventory stockpiling is the closest because, subject to a transport step to move products to the location needed,
it is ready to be used by providers. Capacity buffering is an additional step removed, since the product must be made and then shipped to the desired location. Contingency planning to scale up some form of capacity is one more step removed from providers and patients since the plan must be carried out to create the capacity to produce inventory. Finally, a readiness strategy that builds organizational capabilities is yet another step removed. Thinking carefully about the time sequence involved in medical product shortages and in shortage prevention can help in enumerating a broad range of interventions for enhancing supply chain resilience. This observation also highlights the fact that measures that are closer to being ready to use can be accessed more quickly, which, as discussed below, is important in matching different measures as part of a comprehensive resilience strategy.
Inventory Stockpiling and Capacity Buffering
Although inventory stockpiling and capacity buffering are both physical forms of preparedness, they are different in character. Inventory stockpiling involves maintaining a store of medical products, components, or raw materials that are immediately available. Capacity buffering involves maintaining a capability to produce medical products, components, or raw materials. Strictly speaking, a physical capacity buffer refers to ready-to-use production. For example, a medical device manufacturing plant might have the equipment, labor, and components to immediately produce 20 percent more product than its normal volume. If so, then this extra capacity represents a physical buffer that can compensate for a shortage, subject only to the production and delivery lead times.
Inventory stockpiling and capacity buffering also differ with regard to their dependence on one another. While finished goods inventory is ready to be used, inventory held at the intermediate level (components for devices, APIs for drugs) or the raw material level depend on downstream capacity to be made into finished products. In contrast, buffer capacity at any level requires upstream inventory. For example, the final fill-and-finish stage of drug manufacturing requires API inventory, while API production requires raw material inventory, to carry out their respective manufacturing steps. This mirror-type interdependence—combined with the differences in speed of availability—makes inventory and capacity buffers natural complements, with inventory providing the first line of protection and capacity providing additional layers of protection subject to ramp-up lead times.
Relative Economics of Inventory Stockpiling and Capacity Buffering
In addition to depending on lead times, the right combination of inventory stockpiling and capacity buffering for a given medical product also de-
pends on the relative costs. In general, inventory stockpiling involves a high up-front cost to establish a stockpile and an ongoing maintenance cost to store and rotate it. These costs are incurred regardless of whether the inventory is used or not. Capacity buffering costs are more varied. Since all plants have some excess capacity, they can provide some buffering with no up-front cost or ongoing maintenance cost. Typically, a plant can provide additional buffering by adding labor and/or equipment, with an up-front cost and a longer lead time to ramp up capacity. A production process that can ramp up quickly and efficiently is scalable. But whether or not a process is scalable, the production costs (e.g., materials, labor, energy, etc.) for manufacturing products or inputs are incurred only if the extra capacity is needed. This implies that buffer capacity will be more economical than buffer inventory for products or components that are expensive and/or rarely needed.
The characteristics of inventory stockpiling and capacity buffering suggest a natural pairing in which enough inventory is held to cover the lead time to ramp up enough additional capacity to achieve the target protection. For production stages where capacity can be ramped up enough to provide the target protection without excessive up-front costs or lead time this could be a highly effective strategy. But there may be production stages where capacity cannot be ramped up quickly or economically. If so, different buffers may be appropriate for different production stages of the supply chain. For example, suppose a pharmaceutical fill-and-finish plant is capable of ramping up capacity to the level needed to achieve the desired protection target, but the API plant that feeds it would require a very large cost and a very long lead time to increase capacity sufficiently. In this case, holding finished goods inventory to cover the lead time to increase final production, and holding API inventory sufficient to cover the full protection target, may be appropriate. In general, the most economical mix of inventory stockpiling and capacity buffering, as well as the form in which inventory should be held (finished goods, components, raw materials), depends on the details of the manufacturing and supply chain processes for a given medical product. See Box 5-2 for a blueprint for making economic sense of preparedness measures: inventory stockpiling and capacity buffering.
The economics that govern effective use of inventory stockpiling and capacity buffering as protection against medical product shortages to enhance public health also govern the use of these buffers as protection against loss of sales to enhance private profits. Consequently, producers and marketers of medical products maintain buffer inventory and buffer capacity at various levels of their supply chains to ensure business continuity. However, because private profit incentives differ from public health incentives, protection investments by firms may or may not produce socially beneficial outcomes. For example, consider the relatively likely occurrence of short disruptions due to process problems or demand fluctuations. Such disruptions can be
covered with modest amounts of inventory that will be tapped relatively often. For high-margin products, holding inventory as protection against losing sales or harming one’s brand are easy to justify. Consequently, supply chains for brand-name drugs and patented devices, which have large margins, typically contain inventory stockpiles to protect against variations in supply and demand. They may even contain some form of buffer capacity to protect against larger, less likely disruptions. However, low-margin products, for which cost control is vital, often make use of lean practices that reduce inventory stockpiles to minimal levels.4 Consequently, even relatively modest disruptions, such as a short-term process problem, can produce supply shortages for these products. This explains why generic drugs account for the majority of routine drug shortages (ASHP, 2018). A high-profile example is vincristine, a chemotherapy drug for childhood cancers, which experienced a severe and prolonged shortage when the primary manufacturer encountered a quality control problem in 2019. The implication is that where profit margins are low, public intervention to spur greater use of inventory stockpiling and/or capacity buffering may be needed to provide socially acceptable levels of protection against medical product shortages.
Buffer Flexibility
As noted above, scalability makes capacity more effective as protection against a supply shortage. Another characteristic that can make both inventory stockpiling and capacity buffering more effective is flexibility. A resource is flexible if it can be used to satisfy more than one source of demand. Just as the layers of protection in the medical product supply chain resilience framework need to be independent to reduce risks of a shortage, flexibility is only effective for independent sources of demand. For example, if surges of patients requiring intubation in different regions is perfectly correlated, then resource sharing of ventilators is not helpful. Similarly, if a global pandemic surges demand for N95 masks everywhere at the same time, then reciprocal import/export agreements will be irrelevant. Fortunately, even in widespread events that eventually affect everyone, demand often occurs in waves that are offset in time. This enables flexibility and resource sharing to provide some level of protection. But for major global emergencies, flexibility will not be enough. A combination of inventory stockpiling and capacity buffering will be needed to provide protection in these rare but extreme events.
___________________
4 Lean practices are a set of management practices to improve efficiency and effectiveness by eliminating waste. The core principle of these is to reduce and eliminate non-value adding activities and waste (Crawford, 2016).
Contingency Planning and Readiness
In addition to the physical protections of inventory stockpiling and capacity buffering, preparedness measures also include the virtual protections of contingency planning and readiness. The difference between contingency planning and readiness is that a contingency plan addresses a specific scenario, while a readiness measure prepares for a general class of scenarios or a completely unspecified scenario.
Examples of contingency planning include crisis standards of care planning, such as a policy to use N95 masks for up to five shifts, with nightly sterilization between shifts, to address a supply shortage. An example of a contingency plan at a different point in the supply chain would be a contract with an auto manufacturer to assemble ventilators in an emergency. Note that such a plan would provide additional production capacity, albeit with a longer lead time than physical buffer capacity. A contingency plan that identifies supplemental producers for specific supply chain critical medical products and readiness steps, such as sharing supplier and bill-of-material information with these producers, would make ramping up capacity in an emergency faster and more reliable.
Readiness activities are usually in the form of organizational preparation at multiple levels (i.e., top-down, bottom-up). An example of a readiness practice is the establishment of forums for sharing information and ideas. For instance, the Supply Chain Risk Leadership Council (SCRLC) was set up to encourage supply chain managers to discuss awareness and preparedness practices during routine times and to pool expertise to respond during emergency events (SCRLC, n.d.). A similar council could be established specifically for medical product supply chain managers. Training activities to expose individuals and groups to previous emergency events and collaborative exercises to build relationships among them also act as readiness measures. Each of these measures will help to ensure response is quick and effective to whatever scenario arises.
Response
The response category depends on various awareness, mitigation, and preparedness measures and includes actions taken postevent to minimize harm from the shortage and to resolve the shortage. These actions can be subdivided into measures to close the supply gap through (1) reducing the demand or increasing the supply, and (2) prophylaxis measures, which protect human health while the shortage persists. Taken together, response measures seek to return supply chains to normal (or a “new normal”) with as little harm as possible to patients.
Measures to reduce demand and increase supply must be implemented at the global and local levels to minimize harm from medical product short-
ages once they occur. As such, they must deal with both the big picture of global medical product supply chains and the small picture of local distribution and delivery of medical products to the end users (i.e., the last mile of supply chains). These twin approaches are needed to help national supply chains respond to global emergencies and to aid local end users in reducing the effect of shortages on the health of individual patients and communities. International cooperation and coordinated response, such as international agreements or treaties, can help minimize the effects of medical product shortages and strengthen resilience in supply chains.
Examples of prophylaxis measures include the many improvised or inventive ways medical professionals adapted to medical product shortages during the COVID-19 pandemic. One such innovation was the development of an aerosol box that protected health care workers from exhaled aerosols emitted during patient intubation (Begley et al., 2020). In general, prophylaxis activities are front-line, last-mile measures rather than upstream actions as they resolve the immediate issue but do not solve the supply shortage. However, some improvised and inventive activities can have supply chain implications. For instance, use of 3D-printed components to modify ventilators to allow multiple patients to share a single machine was a demand reduction measure during the early days of the COVID-19 pandemic (Ayyıldız et al., 2020; NIH 3D Print Exchange, 2020; Rosen, 2020).
Finally, it should be noted that there is a very close relationship between virtual preparedness measures and response measures. Contingency planning and readiness activities taken prior to a disruptive event can improve the speed and accuracy of response measures. Therefore, in the planning process it makes sense to consider these categories of options in tandem.
Different Taxonomies and Perspectives
There are other taxonomies and perspectives for building resilient medical product supply chains beyond the categories and measures presented in Figure 5-4. Each category can consider interventions that make use of regulations, economics, technology, and so forth. For example, within the preparedness category, capacity buffering measures are identified as potentially attractive options. However, capacity buffering is not something that can be implemented directly. Instead, it needs to be cultivated by a combination of regulatory changes, economic incentives, and technological advances, such as continuous drug manufacturing that would make domestic production and scale-up practical.
Other taxonomies, like one considering options from the perspective of different actors, such as government, private firms, nonprofits, international organizations, and so forth, may also be helpful in thinking through options
and helping to evolve an effective resilience strategy for medical product supply chains.
No One-Size-Fits-All Strategy: A Discussion on Cost-Effectiveness and Finding Balance within an Integrated Resilience Strategy
Finally, a fundamental take-away from Figure 5-4 and the logic behind it is that there is no one-size-fits-all strategy for increasing the resilience of supply chains for all medical products. Different medical product supply chains, different markets, and different risk profiles all require different interventions. The key challenge is to match measures to products in a cost-effective manner.
At the level of individual medical products, matching appropriate measures to products can be done using the medical product supply chain resilience framework to help select candidate options and then using detailed information about the specific medical product to evaluate the practicality of each option. For medical products identified as supply chain critical, such detailed scrutiny may be warranted. For example, determining the right level of inventory of a given product to hold in the SNS is a policy intervention at the individual product level. The above take-away and the discussion leading to it imply that such a stockpiling decision should take into account the individual characteristics, such as the profit incentive for the market to provide protections, the scalability of the production technology, etc., of the product.
However, it is not possible or practical to make individual analyses of every supply chain critical medical product. Therefore, general purpose regulations and requirements are also needed that enhance supply chain resilience for broad sets of medical products. The implication of the above take-away for this type of policy making is that, where possible, interventions should focus on incentives rather than actions. A useful analogy is the Clean Air Act of 1970, which required use of scrubbers to remove effluents such as sulfur dioxide from the smokestack emissions of power plants. At the time, many economists objected to this requirement, arguing that (1) scrubbers might be a good alternative for some plants but not for others, and (2) the “technology forcing” nature of the requirement removed all incentive for utilities to find more effective ways to control pollution. A better policy would have been to impose an effluent tax that would charge utilities for every pound of effluent they emitted. If the tax was set high enough to make scrubbers more economical than paying the tax, utilities would have incentive to use them. But plants for which other methods (e.g., changes in fuel or process) could reduce pollution more cheaply could use these. And everyone would have incentive to find more efficient means for reducing their effluents. The result would be lower costs, which would translate into
lower prices for consumers, and an innovative culture that would promote ongoing progress in pollution control.
Analogously, if policies are adopted that require firms to hold certain amounts of inventory or to adopt certain flexible manufacturing technologies, it is likely that policies that work well to promote supply chain resilience for some products but are highly inefficient or ineffective for other products will be implemented. If instead policies are used that create incentives for supply continuity and emergency readiness, the market will respond by crafting systems appropriate to their supply chains and by innovating to find more efficient and effective ways to enhance medical supply chain resilience.
To make this general recommendation to leverage the market more concrete, the committee forward references two pieces of recommendations. First, in Recommendation 3 (Health System Actions), health systems are encouraged to incorporate quality and supply continuity into their contracts, so that suppliers pay penalties for defects, recalls, and delays that meet specified criteria. The penalties will provide incentive for suppliers to enhance their reliability but leave it to the firms to find the best way to do this. Second, in Recommendation 5 (Capacity Buffering), the committee advocates for publication of “crisis prices” by the federal government that specify premiums to be paid for critical medical products under specified emergency conditions. These provide incentives for firms to find ways to provide “pop-up” capacity for an emergency but leave it to the market to identify which firms will participate and how. In both recommendations, the policies create incentives for firms to enhance medical product supply chain resilience rather than specifying actions that may be suboptimal or become so over time as manufacturing technologies and practices evolve.
In the following sections the medical product supply chain resilience framework is leveraged and general insights and guidelines that may be helpful in identifying incentive-oriented policies to promote supply chain resilience are discussed.
Early Intervention
In general, the earlier that a harm is prevented, the lower the total cost will be to society. In the language of Figure 5-4, mitigation is generally cheaper than preparedness, which is generally cheaper than response. Although all three will be needed to protect the public from harm by medical product shortages during emergencies, it is usually less expensive to avoid disruption events via mitigation measures than to protect people from events that occur through preparedness measures. Similarly, it is usually less costly to avoid public harm through preparedness measures than it is to address that harm through response measures. Furthermore, since
awareness is a precursor to mitigation, preparedness, and response, investments in information gathering are particularly cost-effective because they enhance the effectiveness of all other measures.
Clearly, the above observation does not imply that every investment in an early shield (or the awareness foundation) of the medical product supply chain resilience framework is more effective than any investment in a later shield. Foolish investments to collect unnecessary information or to implement ineffective mitigation measures are clearly not better than sensitive and appropriate response measures. The best options early in the timeline, when there is time to plan and prepare, are typically better than the best options later in the timeline, when a crisis is under way. Policies that promote proactive behavior in favor of reactive behavior should not be overlooked.
However, although early interventions are important, they are almost never sufficient on their own. The reason is that many measures exhibit decreasing returns to scale. Each additional increment of protection from a given measure becomes less effective or more expensive. For example, each increment of inventory added to a stockpile becomes less and less likely to be needed, and hence provides fewer and fewer expected health benefits. The cost-effectiveness of inventory investments therefore decreases with scale. Similarly, investments in capacity become less valuable with scale, but they also become more expensive. The first increment of buffer capacity may be very cheap to achieve by simply scheduling overtime. A second increment may be more expensive because additional workers must be hired and trained to staff an additional shift. A third increment may be extremely expensive (and slow to achieve) if additional production facilities are needed. Consequently, as with inventory, the cost-effectiveness of capacity investments decreases with scale. Because of this, the most efficient measures—the low-hanging fruit—should be prioritized in all of the protective layers of the medical product supply chain resilience framework to find the most cost-effective mix of measures for promoting resilience in medical product supply chains.
Virtual Protection
Virtual measures are cheaper but slower than concrete measures. For example, the expected cost of a contingency plan to obtain extra inventory will be less than that of an inventory stockpile because the contingency plan might not need to be implemented, while the stockpile will cost whether or not it is needed. Analogously, general readiness measures that address a broad range of scenarios may be cheaper than many specific contingency plans to address each scenario because more time and expense will be invested to generate concrete plans than to be ready in a general sense.
However, the more concrete the resilience measure is, the more rapidly it can be deployed. For example, inventory is immediately available, while
a contingency plan must be carried out before it becomes effective. The implication is that concrete measures are generally best suited for short, frequent disruptions, while virtual measures are better suited for long, infrequent disruptions. As noted earlier, while it may be easy to justify the high up-front cost of inventory stockpiling to cover expected disruptions, it is more pragmatic to rely on capacity buffering to cover large disruptions that are highly unlikely.
Figure 5-5 provides a graphical illustration of how to align resilience measures on different points of the virtual/concrete scale to medical products with different risk profiles. Consistent with the discussion above, the most concrete physical measures of inventory and capacity buffering (along with measures to make them flexible) are suited to products at high risk of supply disruption. Intermediate contingency planning measures, which do not create physical assets but do create explicit plans for specific scenarios, are suited to products having an intermediate risk of disruption. The most virtual measures of response are the options of last resort for dealing with products at low risk of disruption. Finally, the willingness to use a more concrete measure should increase with the medically essential score of a product because the higher cost of human harm justifies more expensive investments to provide faster and more reliable protection. On the other end of the scale, for medical products that are sufficiently nonessential, it may be optimal to do nothing so that resources can be used to secure supplies of more supply chain critical medical products.
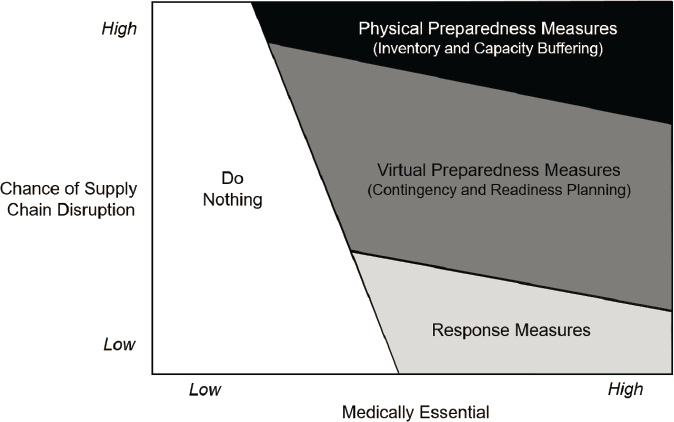
SOURCE: Adapted from Hopp, 2008.
Hybrid Strategies
The detailed goal set for this chapter was to be able to cover a shortage of size Xi for each product i on the supply chain critical list. Based on the development and discussion of the resulting medical product supply chain resilience framework, it is clear that it will be more cost-effective to do this with a combination of supply chain resilience measures, rather than with a single measure. A main reason is because the size of the supply disruption is uncertain. By the logic of Figure 5-5, the fast, concrete measures of inventory stockpiling and capacity buffering should be used to cover smaller shortage amounts that are likely to occur, but the slower response measures should be relied on to deal with large but very unlikely shortages. Contingency planning can be used for intermediate cases. The implication is that a layered protection strategy, with inventory to cover the first Xi units of shortage and virtual measures to provide additional coverage if needed, is likely to be a sound approach for most products.
The proper mix of physical and virtual protection depends on lead times. If the time to bring backup capacity online is very short, such as a plant simply scheduling extra shifts, then it would be wasteful to hold a large amount of inventory in a stockpile. Hence, the most cost-effective hybrid strategy for building supply chain resilience of a given medical product will depend on how critical it is, as indicated in Figure 5-5, and also on the characteristics of its supply chain, which influence how quickly capacity can be ramped up. Furthermore, measures in the different layers can be strongly synergistic. For example, measures that promote better process control (a mitigation measure) may be the most cost-effective way to avoid shortages caused by manufacturing quality problems.
CONCLUDING REMARKS AND OVERVIEW OF COMMITTEE RECOMMENDATIONS
To achieve the goal of promoting resilient medical product supply chains that are cost-effective and protect public health in both normal and emergency conditions, all protective layers of the medical product supply chains resilience framework must be used.
In the remaining chapters (Chapters 6–9), the committee articulates seven recommendations within the four protective layers (or shields)—awareness, mitigation, preparedness, and response—of the medical product supply chains resilience framework. While the recommendations may address more than one layer, the list in Box 5-3 shows the recommendations under the layer with which they are primarily aligned. Taken together, the seven recommendations shown below will increase the resilience of medical product supply chains at all four protective layers of
the resilience framework. Furthermore, where possible, the committee has articulated ways to address these layers by means of incentives that promote actions to enhance supply chain resilience but leaves specifics to the market to allow the power of market competition and innovation to generate the best available solutions. Given balanced attention and coordination, these recommendations will substantially improve the nation’s ability to maintain supplies of medical products and prevent harm during normal and emergency conditions.
REFERENCES
ASHP (American Society of Health-System Pharmacists). 2018. ASHP guidelines on managing drug product shortages. American Journal of Health-System Pharmacists 75:1742-1750.
Ayyıldız, S., A. M. Dursun, V. Yıldırım, M. E. Ince, M. A. Gülçelik, and C. Erdöl. 2020. 3D-printed splitter for use of a single ventilator on multiple patients during COVID-19. 3D Printing and Additive Manufacturing 7(4):181-185.
Begley, J. L., K. E. Lavery, C. P. Nickson, and D. J. Brewster. 2020. The aerosol box for intubation in Coronavirus Disease 2019 patients: An in-situ simulation crossover study. Anaesthesia 75(8):1014-1021.
Bergman, G. M. n.d. Order of arithmetic operations. https://math.berkeley.edu/~gbergman/misc/numbers/ord_ops.html (accessed November 2, 2021).
Crawford, M. 2016. 5 lean principles every engineer should know. The American Society of Mechanical Engineers. https://www.asme.org/topics-resources/content/5-lean-principles-every-should-know (accessed November 2, 2021).
FDA (U.S. Food and Drug Administration) Drug Shortages Task Force. 2020. Drug shortages: Root causes and potential solutions. https://www.fda.gov/drugs/drug-shortages/report-drug-shortages-root-causes-and-potential-solutions (accessed December 16, 2021).
FEMA (Federal Emergency Management Agency). 2021. The four phases of emergency management. https://training.fema.gov/emiweb/downloads/is10_unit3.doc (accessed December 16, 2021).
Hopp, W. J. 2008. Supply chain science. New York: McGraw-Hill Irwin.
Hopp, W. J., and W. S. Lovejoy. 2012. Hospital operations: Principles of high efficiency health care. Pearson Education. Upper Saddle River, NJ: FT Press.
NIH 3D Print Exchange. 2020. LRTee: Ventilator splitter. NIH. https://3dprint.nih.gov/discover/3dpx-013734 (accessed November 3, 2021).
Reason, J. 1997. Managing the risks of organizational accidents. Burlington, VT: Ashgate Publishing.
Reason, J. 2000. Human error: Models and management. BMJ (Clinical Research Edition) 320(7237):768-770.
Rosen, J. 2020. Johns Hopkins engineers developing 3D-printed ventilator splitter, edited by Johns Hopkins University and Office of Communications. Johns Hopkins University. https://releases.jhu.edu/2020/04/02/johns-hopkins-engineers-developing-3d-printedventilator-splitter/ (accessed November 3, 2021).
SCLRC (Supply Chain Risk Leadership Council). n.d. Supply Chain Risk Leadership Council. http://scrlc.com/about.php (accessed December 9, 2021).
Shukar, S., F. Zahoor, K. Hayat, A. Saeed, A. H. Gillani, S. Omer, S. Hu, Z.-U.-D. Babar, Y. Fang, and C. Yang. 2021. Drug shortage: Causes, impact, and mitigation strategies. Frontiers in Pharmacology 12:1772.
Smetzer, J., and M. Cohen. 1998. Lesson from the Denver medication error/criminal negligence case: Look beyond blaming individuals. Hospital Pharmacy 33(6).
__________________
This page intentionally left blank.