Appendix D
Epidemiology: Select Diseases
SJÖGREN’S DISEASE
Epidemiology Overview
Few studies have examined the incidence or prevalence of Sjögren’s disease, and the data are limited to “primary” Sjögren’s disease (i.e., Sjögren’s disease occurring alone), which excludes individuals with co-occurring systemic rheumatic disease such as SLE, rheumatoid arthritis, systemic sclerosis, or dermatomyositis. A recent study estimated the annual incidence of primary Sjögren’s disease in New York County (Manhattan) as 3.5 cases per 100,000 persons (Izmirly et al., 2019). This estimate does not include children with Sjögren’s disease and therefore grossly underestimates the overall incidence of Sjögren’s disease. The same study estimated the prevalence of primary Sjögren’s disease in this population to be 13.1 per 100,000 persons (Izmirly et al., 2019). Again, however, children were not included, and Sjögren’s disease commonly co-occurs with other autoimmune diseases, so while this prevalence may reflect primary Sjögren’s disease in adults, it fails to capture the full extent of individuals in the United States with Sjögren’s disease. In recent studies, 30 percent of individuals with rheumatoid arthritis also had Sjögren’s disease (Harrold et al., 2020), while about 15 to 20 percent of individuals with SLE had Sjögren’s disease (Aggarwal et al., 2015; Baer et al., 2010). In addition, a study has shown that patients with primary Sjögren’s disease are often initially misdiagnosed with SLE or rheumatoid arthritis (Rasmussen et
al., 2016). Thus, the overall prevalence of Sjögren’s disease in the United States is likely greater than 0.3 percent of the population.
Sex, Age, Racial, and Ethnic Disparities
Diagnosis of Sjögren’s disease most commonly occurs in the fifth to seventh decades of life, though it may occur from early childhood through late adulthood (Brito-Zerón et al., 2020; Ramos-Casals et al., 2020; Sjögren’s Foundation, 2021). Sjögren’s disease is diagnosed more commonly in females, with a female-to-male ratio of 6:1 based on small studies in the United States (Izmirly et al., 2019; Maciel et al., 2017), though large global studies have found an even higher female-to-male ratio of 14:1 in adults and a slightly decreased ratio of 5:1 in children (Basiaga et al., 2020; Brito-Zerón et al., 2020; Ramos-Casals et al., 2020). Sjögren’s disease occurs across races and ethnicities but is diagnosed more commonly in White individuals. Among over 10,000 adults with Sjögren’s disease in a multinational study, 77 percent were White individuals, 14 percent Asian individuals, 6 percent Hispanic individuals, and 1.4 percent Black individuals (Brito-Zerón et al., 2020). Measures of disease activity indicated that disease activity was highest in Black individuals, followed by White, Asian, and Hispanic individuals.
Specific disease manifestations differed according to ethnicity and age (Ramos-Casals et al., 2021). For example, children had a higher frequency of glandular swelling, rashes, and cytopenias but a lower frequency of dryness, whereas the frequency of pulmonary and neuromuscular manifestations increased with age at presentation. In a recent U.S.-based study, the American Indian population was disproportionately affected by Sjögren’s disease, displaying a higher risk of diagnosis and increased disease activity (Scofield et al., 2020). Additional studies are needed to further define racial and ethnic disparities.
SYSTEMIC LUPUS ERYTHEMATOSUS
Epidemiology Overview
A recent meta-analysis based on data from a Centers for Disease Control and Prevention (CDC) network of four national lupus registries estimated overall SLE incidence in the United States as 5.1 per 100,000 person years for 2002 to 2009 (Izmirly et al., 2021a).1 Some of the most comprehensive data on SLE are from studies based in Michigan, Georgia, and California associated with the CDC national lupus registries. The
___________________
1 This study was published around the time the report was going into review.
registries provide incidence and prevalence data for select state areas. Incidence of SLE was 4.6 per 100,000 persons in San Francisco County in California (Dall’Era et al., 2017), 5.5 per 100,000 in southeastern Michigan (Somers et al., 2014), and 5.6 per 100,000 in Fulton and DeKalb counties in Georgia (Lim et al., 2014). The Manhattan Lupus Surveillance Program (Izmirly et al., 2017), a registry of residents in New York County (Manhattan), found an annual incidence of 4.6 cases per 100,000 persons for 2007 to 2009 and a prevalence rate of 62.2 cases per 100,000 persons for the year 2007.
Sex, Age, Racial and Ethnic Disparities
A meta-analysis of CDC national lupus registries found the U.S. incidence of SLE in women to be approximately seven times higher than in men for the years 2002–2009 (Izmirly et al., 2021a). In each of the Michigan (Figure D-1A), Georgia, and California studies, incidence rates for women were 5 to 10 times higher than for men across all ages and in each racial or ethnic group studied (Dall’Era et al., 2017; Lim et al., 2014; Somers et al., 2014). For women, each of these studies also found considerably higher rates of the disease in Black patients than in White patients, with a marked increase in incidence between the ages of 20 and 60 years, as represented by Georgia data in Figure D-1B. For White, Asian-Pacific Islander, and Hispanic patients, however, incidence rates among women showed less variation across these age groups (Figure D-1C).
Similarly, in the New York study (Izmirly et al., 2017), the incidence and prevalence rates of SLE were around nine times higher in women than in men. When examining only women, incidence and prevalence rates were highest for non-Hispanic Black women, and showed a marked increase in the 20–59 age groups (Figure D-2). White, Hispanic, and Asian women showed less variation across these age groups.
Investigators have conducted a meta-analysis of data from the California, Georgia, Michigan, and New York studies and from the Indian Health Service and estimated a prevalence of SLE for the United States of 72.8 per 100,000 (Izmirly et al., 2021b). Between 80 and 90 percent of individuals with SLE are female, and SLE is two to three times more common in Black, Indigenous, and people of color. The highest prevalence of SLE was seen in American Indian and Alaska Native women at 270.6 per 100,000 women, and Black women at 230.9 per 100,000 women, while the rate for Hispanic women was lower at 120.7 per 100,000 women but still 43 percent higher than the rate for White women (84.7 per 100,000 women) and Asian and Pacific Islander women (84.4 per 100,000 women) (Izmirly et al., 2021b).
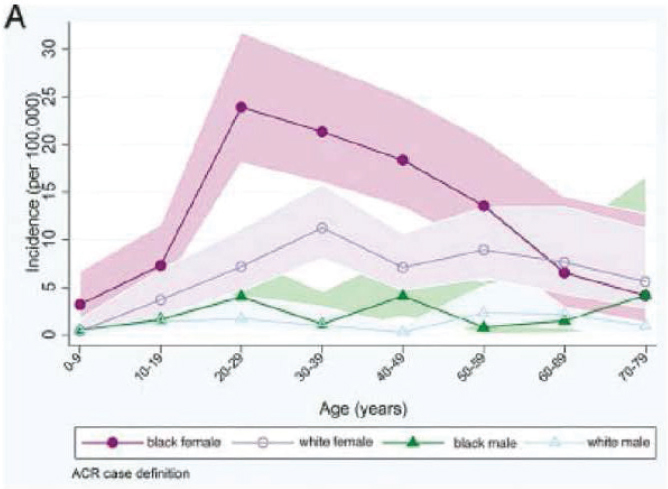
Incidence of Systemic Lupus Erythematosus in Men and Women in southeastern Michigan, 2002–2004 by age, sex, and race. Somers et al., 2014.
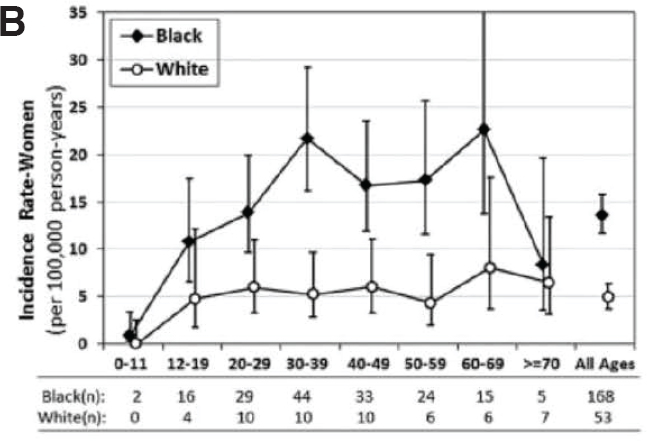
Incidence of Systemic Lupus Erythematosus in Women in Fulton and DelKalb Counties, Georgia, 2002–2004 by age and race. Lim et al., 2019.
SOURCES: Dall’Era et al., 2017; Lim et al., 2014; Somers et al., 2014.
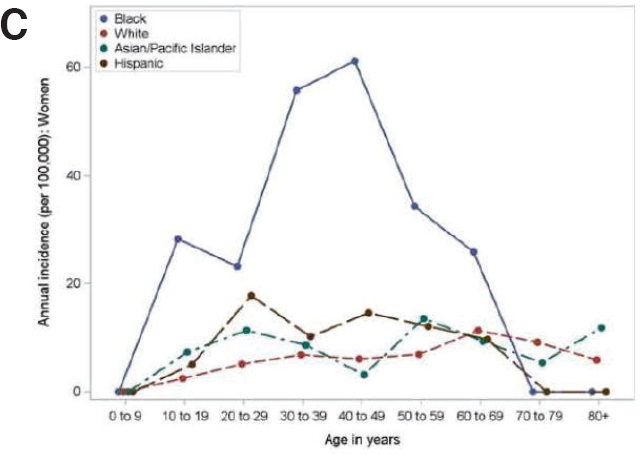
Incidence of Systemic Lupus Erythematosus in Women in San Francisco County, California, 2007–2009, by age and race-ethnicity. Dall–Era et al., 2017.
Although the rates of some autoimmune diseases are lower in children than adults, these diseases have a significant impact on the children and their families. Juvenile-onset SLE, which occurs before age 18, is estimated to affect about 15 to 20 percent of people with SLE (Charras et al., 2021; Hersh et al., 2010). Data on U.S. studies suggest that the incidence of juvenile SLE ranges from 0.36 to 2.5 per 100,000 children and the estimated prevalence is from 1.9 to 34.1 per 100,000 children (Charras et al., 2021). Childhood-onset SLE shows population-level differences in incidence and prevalence similar to those seen in adult-onset disease. In a study of the U.S. Medicaid beneficiary population from 2000 to 2004, the annual incidence of SLE in children 3 to <18 years old was 2.22 per 100,000 Medicaid-enrolled children and that of lupus nephritis, a frequent complication of SLE, was 0.72 per 100,000 (Hiraki et al., 2012). In the same population, prevalence estimates for children with SLE were 9.73 per 100,000 Medicaid enrolled children, of whom 84 percent were female, 40 percent were Black, 25 percent were Hispanic, and 21 percent were White. In addition, 37 percent of children with SLE had lupus nephritis, representing a prevalence of 3.64 per 100,000 Medicaid-enrolled children. Both prevalence and incidence rates of SLE and lupus nephritis increased with age, were higher in girls than in boys, and were higher in all non-White racial and ethnic groups (Hiraki et al., 2012).
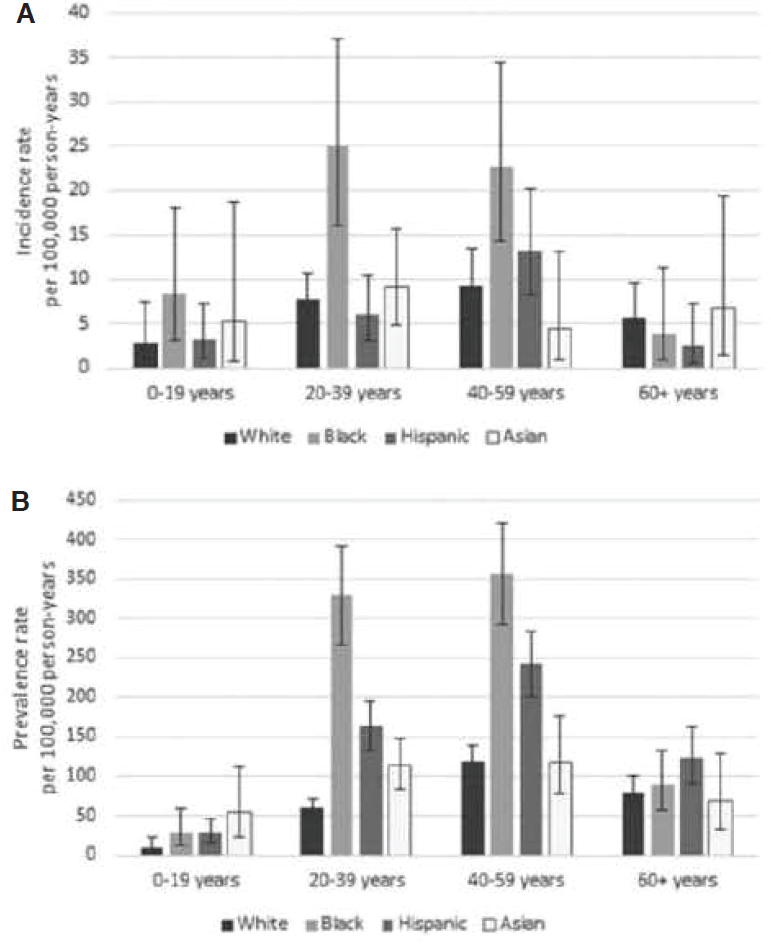
SOURCE: Izmirly et al., 2017.
SLE exemplifies important disparities in disease severity, physical and mental morbidity, and mortality across racial and ethnic groups. Indeed, SLE is the fifth leading cause of death in Black and Hispanic females aged 15 to 24 years old in the United States (Yen and Singh, 2018). Furthermore, research has identified disparities in outcomes of SLE in children, including care delivery and SLE-related conditions such as mental health disorders (Rubinstein and Knight, 2020). These disparities may exist according to race and ethnicity, income, and geography.
Some of the most severe SLE manifestations are lupus nephritis and neuropsychiatric lupus (NPSLE). Although lupus nephritis and NPSLE typically occur early in the disease course, they can occur years after the onset of disease in some patients. NPSLE has many manifestations, including seizures, cerebrovascular disease, demyelinating syndrome, psychosis, cognitive dysfunction, acute confusional state, chorea, myelopathy, mononeuritis multiplex, and peripheral neuropathies (Ford et al., 1999; Gulinello et al., 2012). Both nephritis and NPSLE manifestations may remit, or more commonly, result in long-standing damage.
One U.S. study of end-stage renal disease (ESRD) resulting from SLE found that rates of ESRD in Black individuals ages 5 to 39 with lupus nephritis increased during the 1995 to 2006 period (Costenbader et al., 2011). Researchers, using data from the same study, further observed that Black children were half as likely to receive a kidney transplant as White children and twice as likely to die from ESRD caused by lupus nephritis. Hispanic children and adults, compared with non-Hispanic children and adults, were also less likely to receive kidney transplants. Researchers also found that children living in the U.S. West and Northwest were placed on kidney transplant wait lists more often that children living in the South (Hiraki et al., 2011). A large study of hospitalized children with SLE in the United States, using data from the Kids’ Inpatient Database,2 found both regional and racial disparities in mortality, with Black children and children from southern states having twice the risk of death compared with White children and children from northeastern states (Knight et al., 2014). While these studies do reveal disparities among certain U.S. subpopulations, the committee believes that efforts to understand these disparities would benefit from large epidemiologic studies covering a wide range of demographic characteristics.
___________________
2 Additional information is available at https://www.hcup-us.ahrq.gov/kidoverview.jsp.
ANTIPHOSPHOLIPID SYNDROME
Epidemiology Overview
Because of technical differences in assessing antiphospholipid antibodies and APS’s overlap with SLE and other autoimmune diseases, epidemiology data for this illness are scarce, but the prevalence is thought to be 40 to 50 cases per 100,000 persons (Cervera, 2017; Duarte-Garcia et al., 2019). Reports on demographics, incidence, prevalence, and population outcome often come from retrospective studies that are not generalizable (Erkan et al., 2012, 2021). A recent population-based study in Minnesota addresses that bias, but is limited in its generalizability in that it is based in a relatively small and homogenous population with a low incidence of SLE (Duarte-Garcia et al., 2019). Prospective studies and clinical trials with diverse participants and large sample sizes could yield more accurate and relevant results (Erkan et al., 2012; FDA, 2021).
The 2022 International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) code for antiphospholipid syndrome (D68.61) became effective on October 1, 2021 (ICD10 Data, 2021). This code excludes the common finding of a positive lupus inhibitor test, as measured by specialized coagulation tests, in an asymptomatic person, suggesting use of the code R76.02 instead.
Sex, Age, Racial and Ethnic Disparities
Isolated thrombotic APS affects both sexes equally (NORD, 2016). When APS occurs with another autoimmune disease, that disease is most often lupus, and because the majority of individuals with lupus are female, APS associated with other illnesses is female predominant. Considering obstetric and thrombotic APS together, APS is female predominant. In a population-based study of incidence of APS in ages ≥18 years, the female-to-male ratio was 1.0 (Duarte-Garcia et al., 2019); this low ratio may result from the relative lack of representation of people at higher risk of SLE in this study. Unlike SLE, the risk of APS is greatest in White populations and relatively low in Black populations (Duarte-García et al., 2019; Izmirly et al., 2021b).
RHEUMATOID ARTHRITIS
Epidemiology Overview
Rheumatoid arthritis is one of the most common autoimmune diseases, affecting an estimated 2.4 million adults in the United States
(Kawatkar et al., 2019). Although incidence rates for rheumatoid arthritis decreased in the United States and Europe in the latter part of the 20th century, the trends since the 1990s have demonstrated relatively stable or somewhat rising rates (Myasoedova et al., 2010; Safiri et al., 2019). During the period 1995 to 2007, based on the Olmsted County, MN, population, overall age- and sex-adjusted annual incidence was 40.9 per 100,000 population (Myasoedova et al., 2010).
Sex, Age, Racial and Ethnic Disparities
The incidence of rheumatoid arthritis begins to rise in women and men aged 35 to 54, with the highest rates in both women and men among those aged 65 to 74. Female-to-male incidence ratios are about 3:1 during reproductive and perimenopausal years, and decrease to about 1.5:1 for individuals over the age of 55 (Myasoedova et al., 2010). Incidence is higher in the United States and northern European countries (Alamanos et al., 2006). Studies in specific American Indian and Alaska Native communities (Blackfeet, Yakima, Chippewa, and Pima) have reported higher prevalence rates of rheumatoid arthritis than reported for the general U.S. population estimate (Kawatkar et al., 2019; McDougall et al., 2017).
With respect to health outcomes and disability measures among people with rheumatoid arthritis, studies have observed higher disease activity and lower rates of remission among Black and Hispanic populations compared with White populations (Barton et al., 2011; Greenberg et al., 2013). Early treatment with the newer, more targeted therapies is recommended (Fraenkel et al., 2021), but use of these medications may depend on insurance coverage and access to care.
PSORIASIS
Epidemiology Overview
The annual incidence of psoriasis in the United States is estimated to be 78.9 new cases per 100,000 persons (Icen et al., 2009), though a more recent study based on health administrative data for Ontario, Canada, reported an incidence rate of 69.9 cases per 100,000 persons in 2015 (Eder et al., 2019). A medical records-based study in a large health maintenance organization in northern California estimated the prevalence of psoriasis to be 939 per 100,000, which translates to approximately 2.5 million U.S. adults (Asgari et al., 2013). The prevalence among U.S. children is lower, with 128 cases per 100,000 (Paller et al., 2018).
Sex, Age, Racial and Ethnic Disparities
The incidence of psoriasis is similar among men and women; however men may experience more severe forms of the disease (Hägg et al., 2013). The prevalence of psoriasis increases over the life course, with prevalence of 0.12 percent at age 1 year and 1.2 percent at age 18 years (Augustin et al., 2010). A 2015 study of psoriasis in U.S. children and adolescents estimated the prevalence at 128 per 100,000 persons. The prevalence was higher in females than in males—146 per 100,000 versus 110 per 100,000—and increased with age, ranging from 30 per 100,000 in the 0 to 3 age group to 205 per 100,000 in the 12 to 17 age group (Paller et al., 2018). The location of lesions and the type of psoriasis may also vary by age. For example, psoriasis is more likely to affect the face in children, and guttate psoriasis, which manifests as red, scaly, small, teardrop-shaped spots, is more common among children than adults (Boehncke and Schön, 2015).
White individuals had the highest prevalence of psoriasis at 3.6 percent, followed by other racial/ethnic groups (including multiracial) at 3.1 percent, and Hispanic and Black individuals at 1.9 percent and 1.5 percent, respectively (Armstrong et al., 2021).
INFLAMMATORY BOWEL DISEASE
Epidemiology Overview
Ulcerative colitis and Crohn’s disease have emerged as global diseases in the 21st century (Ananthakrishnan et al., 2020). Estimates of the prevalence of these diseases in the United States, obtained using U.S. medical records, range from 1.3 to 2.3 million persons. Another study using less rigorous, self-report methods suggest estimates as high as 3.9 million persons. The true prevalence is unknown (see Box 2-1 in Chapter 2).
Sex, Age, Racial and Ethnic Disparities
Epidemiologic studies from Olmsted County, MN, found that the incidence of Crohn’s disease among women and men was about the same, though the incidence of ulcerative colitis was slightly higher for males than females (Shivashankar et al., 2017). The highest incidence of both ulcerative colitis and Crohn’s disease occurs in the second to fourth decades, and particularly in the 20 to 29 age range.
Pediatric IBD accounts for approximately 25 percent of cases of IBD, and approximately 18 percent of children with IBD will present before age 10 (Rosen et al., 2015). The prevalence of IBD in children under 17 years of age is about 77.0 per 100,000 people in the United States, which
translates into 58,000 children between the ages of 2 and 17 affected by IBD (Ye et al., 2020). The prevalence of pediatric IBD in the United States has increased by 133 percent from 2007 to 2016, from 33.0 to 77.0 cases per 100,000 people, with Crohn’s disease being twice as prevalent as ulcerative colitis at 45.9 cases of Crohn’s disease per 100,000 children versus 21.6 cases of ulcerative colitis per 100,000 children (Ye et al., 2020). Studies have observed geographic variability in IBD rates, with the incidence rising in developing countries and urban areas. Changes in diet that affect the intestinal microbiota, exposure to sunlight or temperature differences, socioeconomic status, and hygiene are among the environmental variables that most likely explain that geographic variability (Benchimol et al., 2017; Bernstein et al., 2019). In infantile IBD or very-early-onset IBD, researchers have identified genetic mutations as the basis for this susceptibility in up to 10 percent of cases, which suggests a simple monogenic origin of the disease in these cases (Ouahed, 2021).
White individuals and people of Ashkenazi Jewish descent have the highest incidence of IBD, though the incidence of IBD is increasing in Hispanic and Asian populations (Hou et al., 2009; Santos et al., 2018). IBD is more prevalent in urban areas and higher socioeconomic populations compared with rural areas and lower socioeconomic populations (Benchimol et al., 2017; Bernstein et al., 2019). IBD is a familial disorder in up to 12 percent of those affected, and the strongest risk factor for the development of IBD is a first-degree relative with the disease. There is an approximately 4-fold increased risk of ulcerative colitis in the children of mothers or fathers with ulcerative colitis and an almost 8-fold increased risk of Crohn’s disease in the children of mothers or fathers with Crohn’s disease (Agrawal et al., 2021; Moller et al., 2015), as well as similar patterns regarding which parts of the intestinal system the disease affects. Some children of parents with IBD develop disease during the first decade of life. In a small study, the relative risk for concordant disease in monozygotic twins was estimated at 95.4 for Crohn’s disease and 49.5 for ulcerative colitis. The relative risk in dizygotic twins was 42.2 and 0.0 for Crohn’s disease and ulcerative colitis, respectively (Bengtson et al., 2010).
CELIAC DISEASE
Epidemiology Overview
Celiac disease is characterized by the presence of tissue transglutaminase autoantibodies (tTGA) that often precedes the biopsy-proven diagnosis of celiac disease (Gandini et al., 2021). Using the presence of tTGA in serum samples to provide an estimate of disease in the United States, a study based on data from the National Health and Nutrition
Examination Survey estimated a seroprevalence of 790 per 100,000 persons over 5 years of age (Mardini et al., 2015), which corresponds to 2.3 million people. Estimates place the worldwide seroprevalence of celiac disease at 1.4 percent, and the worldwide prevalence of biopsy-proven celiac disease at 0.7 percent (Singh et al., 2018).
Sex, Age, Racial and Ethnic Disparities
Celiac disease is 1.5 times more common in females than in males, and approximately 2 times more common in children than in adults (Singh et al., 2018). The prevalence and incidence of celiac disease has been increasing over time, which suggests that changing environmental factors are influencing the development of this disease (Lohi et al., 2007; Ludvigsson et al., 2013). Studies have found the prevalence of celiac disease to be highest in Europe and Oceania (0.8 percent) and lowest in South America (0.4 percent), with Africa (0.5 percent), North America (0.5 percent), and Asia (0.6 percent) being intermediate (Singh et al., 2018). Studies estimate that the prevalence of celiac disease in the United States is four to eight times higher among non-Hispanic Whites compared with other racial and ethnic groups (Mardini et al., 2015).
PRIMARY BILIARY CHOLANGITIS
Epidemiology Overview
The incidence of PBC in the United States is 4 to 5 per 100,000 persons per year (Lu et al., 2018). Incidence rates have been relatively stable during the past 20 years (Kanth et al., 2017; Lu et al., 2018), but the prevalence has increased during this time from approximately 22 per 100,000 persons to 39 per 100,000 persons (Lu et al., 2018). This difference may reflect earlier treatment leading to improved patient outcomes. The 5-year mortality rate is approximately 15 to 20 percent.
Sex, Age, Racial and Ethnic Disparities
PBC is predominantly a disorder of women, who account for approximately 90 percent of all cases. The typical age of onset is in the fifth or sixth decades of life, and the highest reported incidence and prevalence is in northern Europe and the northern United States (Lv et al., 2021).
Studies have reported increased prevalence in some Indigenous groups in the United States and Canada (Yoshida et al., 2006). In comparison to White patients with PBC, Indigenous Canadians present with advanced disease and have worse long-term outcomes (Roberts et al.,
2022). Although rates are higher among White and Asian and Pacific Islanders compared with Black populations (Lu et al., 2018), the severity of disease at diagnosis may be greater in Black and Hispanic compared with White populations (Peters et al., 2007), which has implications for treatment options and outcomes.
MULTIPLE SCLEROSIS
Epidemiology Overview
Multiple sclerosis is the most common CNS inflammatory disorder, though incidence data for the United States do not exist. The overall 2010 U.S. prevalence of multiple sclerosis was estimated at 309 per 100,000 persons (Wallin et al., 2019), equating to approximately 775,000 cases of multiple sclerosis in the United States in 2020. The prevalence of multiple sclerosis has increased worldwide between 1990 and 2016 (Walton et al., 2020), while studies have not consistently found an increase in incidence. Therefore, the increasing multiple sclerosis prevalence is likely a result of a combination of increased awareness leading to increased detection, better diagnostic technology, the increasing age of the populations studied, and increased survival of those with the disease.
Sex, Age, Racial and Ethnic Disparities
Women are more than twice as likely as males to live with multiple sclerosis as males, with a 2.8-fold higher prevalence (Wallin et al., 2019). For adult-onset multiple sclerosis, the mean age at clinical diagnosis is 30 years (Mayo Clinic, 2021). Onset of multiple sclerosis in childhood is uncommon, occurring in 2 to 10 percent of individuals with multiple sclerosis (Yan et al., 2020; Yeh et al., 2009). Sex and age at diagnosis differ by the type of multiple sclerosis, however, with nearly equal proportions of men and women having primary progressive multiple sclerosis and showing a later onset—approximately 10 years—than those with relapsing-remitting multiple sclerosis (Tremlett et al., 2005, Table 2, p. 1921).
A study of racial and ethnic variability in multiple sclerosis in the multi-ethnic community-dwelling members of a California health plan showed a higher annual incidence in Black members, with 10.2 cases per 100,000 persons, compared with White members, with 6.9 cases per 100,000 persons, and lower rates in Hispanic members, with 2.9 cases per 100,000 persons, and Asian and Pacific Islander members, with 1.4 cases per 100,000 persons (Langer-Gould et al., 2013). The prevalence of multiple sclerosis is higher in non-Hispanic Black compared with non-Hispanic White populations, and there is even lower prevalence in Asian,
Pacific Islander, and Hispanic populations (Romanelli et al., 2020). There is a dearth of data on delay in diagnosis in racial and ethnic minority populations, however, with several studies suggesting that delays result from a lack of access to a multiple sclerosis specialist and to perceptions among physicians that the disease is more common in White populations (Stuifbergen et al., 2021).
TYPE 1 DIABETES
Epidemiology Overview
A study in Olmstead County, MN, between 1994 and 2010 reported an overall annual incidence of type 1 diabetes of 9.2 per 100,000 people per year among all ages, and 19.9 per 100,000 people for those younger than 20 years (Cartee et al., 2016). A population-based registry across five U.S. sites reported an annual incidence of 21.7 per 100,000 in people under the age of 20 in 2011 to 2012 (Mayer-Davis et al., 2017).
An estimated 178,000 individuals under the age of 20 have type 1 diabetes in the United States (Dabelea et al., 2014). One study based on non-verified, self-reported data collected through the National Health Interview Study in 2016 and 2017 estimated that 0.5 percent of the U.S. population had a diagnosis of type 1 diabetes. In 2020, this would have represented about 1,485,000 adults living with the disease (Bullard et al., 2018; Xu et al., 2018).
Sex, Age, Racial and Ethnic Disparities
The annual incidence of type 1 diabetes in individuals 19 years and younger is almost twice that of adults 20 to 64 years of age (Rogers et al., 2017). In studies of individuals ages 0 to 19, type 1 diabetes often occurs during two peaks: at ages 5 to 7 and during puberty (Atkinson et al., 2014). In the 0 to 19 age group, the prevalence of type 1 diabetes is similar in males and females (Dabelea et al., 2014), though the incidence of the disease increased among boys but not among girls between 2002 and 2012 (Mayer-Davis et al., 2017). In adults, there is a higher incidence of type 1 diabetes in males compared with females (Rogers et al., 2017).
During the period from 2001 to 2009, a large U.S. study showed an increase in type 1 diabetes prevalence in the 0–19 age group from 1.48 per 1,000 individuals to 1.93 per 1,000 individuals (Dabelea et al., 2014). A subsequent study of the U.S. Medicaid pediatric population showed an increase in annual type 1 diabetes prevalence from 1.29 per 1,000 individuals to 2.34 per 1,000 individuals during 2002 to 2016 (Chen et al.,
2019). This trend was not seen, however, in a study spanning 1994 to 2010 in Olmsted County, MN (Cartee et al., 2016).
In the United States, the prevalence of type 1 diabetes in the age 0 to 19 population is highest among non-Hispanic White persons compared with Hispanic, African American, American Indian, and Native Alaskan persons (Dabelea et al., 2014). The incidence of type 1 diabetes in that age group increased 1.4 percent annually, from 19.5 cases per 100,000 youths per year in 2002 and 2003 to 21.7 cases per 100,000 youths per year in 2011 and 2012. There was a greater rate of increase among Hispanic persons compared with non-Hispanic White persons, with a 4.2 percent annual increase among Hispanic individuals versus a 1.2 percent annual increase in non-Hispanic White individuals (Mayer-Davis et al., 2017), suggesting the influence of environmental factors that may differentially influence risk in different ethnic and racial groups.
AUTOIMMUNE THYROID DISEASES
Epidemiology Overview
Despite being among the most common autoimmune disorders (Bülow Pedersen and Laurberg, 2009), there is a lack of detailed epidemiologic data on Hashimoto’s thyroiditis and Graves’ disease for the past 20 years for the United States. Historically, data from Rochester, MN, have served as a key source for U.S. data on autoimmune thyroid diseases (Furszyfer et al., 1972), although these data predated current methods for thyroid function testing. Nonetheless, an important finding based on the Rochester data from 1935 to 1967 was documenting a sharp rise in incidence over the three decades, an increase that appeared most pronounced among those younger than 40 years old. Data in males were too sparse to characterize temporal trends, and the study did not find evidence of a cohort effect.
European annual incidence estimates of autoimmune thyroid disease have varied widely, from 51 to 490 cases per 100,000 persons (Galofré et al., 1994; Vanderpump et al., 1995). Differences in methodologies and nomenclature across studies likely contribute to the wide range of estimates, although true regional variation should not be ruled out. A study in 2008 to 2013 in Sheffield, United Kingdom, reported an annual incidence of 24.8 cases per 100,000 persons, with a median age of 44 years old at diagnosis. Women accounted for 80 percent of those affected (Hussain et al., 2017). A large cohort study in the Netherlands examined thyroid medication use and thyroid hormone levels to classify overt and subclinical thyroid diseases. Thyroid medication was used by 3.1 percent of participants, the majority (98.2 percent) of whom used levothyroxine.
Subclinical hypothyroidism was detected in 9.4 percent of the people who were not taking thyroid medications (Wouters et al., 2020).
Sex, Age, Racial and Ethnic Disparities
Onset of autoimmune thyroid diseases typically occurs in mid- to late-adulthood (Cooper and Stroehla, 2003). A strong female preponderance of up to 95 percent has been reported (Cooper and Stroehla, 2003); epidemiologic data are lacking for males as a result of how rare thyroid disease is in men. U.S. data indicate that the prevalence of Hashimoto’s thyroiditis is highest in White populations. A histologic study in Baltimore from 1955 to 1960 documented an approximately four times greater prevalence in White females compared with Black females (Masi, 1965), and a study based on data from the third National Health and Nutrition Examination Survey conducted from 1988 to 1994 found that prevalence of anti-thyroid antibodies was highest among White populations, followed by Mexican American populations, and was lowest in Black populations (Hollowell et al., 2002). Trends related to race and ethnicity have been less well characterized for Graves’ disease. However, a 2014 report of active-duty U.S. military personnel found that the incidence of Graves’ disease was significantly higher among individuals from Black and Asian or Pacific Islander populations compared with those from White populations, and trended higher in Hispanic populations compared with White populations (McLeod et al., 2014). This study also corroborated the earlier race and ethnicity trends observed for Hashimoto’s thyroiditis (McLeod et al., 2014).
REFERENCES
Aggarwal, R., J.-M. Anaya, K. A. Koelsch, B. T. Kurien, and R. H. Scofield. 2015. Association between secondary and primary Sjögren’s syndrome in a large collection of lupus families. Autoimmune Diseases 2015:298506. https://doi.org/10.1155/2015/298506.
Agrawal, M., G. Corn, S. Shrestha, N. M. Nielsen, M. Frisch, J.-F. Colombel, and T. Jess. 2021. Inflammatory bowel diseases among first-generation and second-generation immigrants in Denmark: A population-based cohort study. Gut 70(6):1037–1043. https://doi.org/10.1136/gutjnl-2020-321798.
Alamanos, Y., P. V. Voulgari, and A. A. Drosos. 2006. Incidence and prevalence of rheumatoid arthritis, based on the 1987 American College of Rheumatology criteria: A systematic review. Seminars in Arthritis and Rheumatism 36(3):182–188. https://doi.org/10.1016/j.semarthrit.2006.08.006.
Ananthakrishnan, A. N., G. G. Kaplan, and S. C. Ng. 2020. Changing global epidemiology of inflammatory bowel diseases: Sustaining health care delivery into the 21st century. Clinical Gastroenterology and Hepatology 18(6):1252–1260. https://doi.org/10.1016/j.cgh.2020.01.028.
Armstrong, A. W., M. D. Mehta, C. W. Schupp, G. C. Gondo, S. J. Bell, and C. E. M. Griffiths. 2021. Psoriasis prevalence in adults in the United States. Journal of the American Academy of Dermatology 157(8):940–946. https://doi.org/10.1001/jamadermatol.2021.2007.
Asgari, M. M., J. J. Wu, J. M. Gelfand, C. Salman, J. R. Curtis, L. R. Harrold, and L. J. Herrinton. 2013. Validity of diagnostic codes and prevalence of psoriasis and psoriatic arthritis in a managed care population, 1996–2009. Pharmacoepidemiology and Drug Safety 22(8):842–849. https://doi.org/10.1002/pds.3447.
Atkinson, M. A., G. S. Eisenbarth, and A. W. Michels. 2014. Type 1 diabetes. The Lancet 383(9911):69–82. https://doi.org/10.1016/s0140-6736(13)60591-7.
Augustin, M., G. Glaeske, M. A. Radtke, E. Christophers, K. Reich, and I. Schäfer. 2010. Epidemiology and comorbidity of psoriasis in children. British Journal of Dermatology 162(3):633–636. https://doi.org/10.1111/j.1365-2133.2009.09593.x.
Baer, A. N., J. W. Maynard, F. Shaikh, L. S. Magder, and M. Petri. 2010. Secondary Sjögren’s syndrome in systemic lupus erythematosus defines a distinct disease subset. Journal of Rheumatology 37(6):1143–1149. https://doi.org/10.3899/jrheum.090804.
Barton, J. L., L. Trupin, D. Schillinger, S. A. Gansky, C. Tonner, M. Margaretten, V. Chernitskiy, J. Graf, J. Imboden, and E. Yelin. 2011. Racial and ethnic disparities in disease activity and function among persons with rheumatoid arthritis from university-affiliated clinics. Arthritis Care & Research 63(9):1238–1246. https://doi.org/10.1002/acr.20525.
Basiaga, M. L., S. M. Stern, J. J. Mehta, C. Edens, R. L. Randell, A. Pomorska, N. Irga-Jaworska, M. F. Ibarra, C. Bracaglia, R. Nicolai, G. Susic, A. Boneparth, H. Srinivasalu, B. Dizon, A. A. Kamdar, B. Goldberg, S. Knupp-Oliveira, J. Antòn, J. M. Mosquera, S. Appenzeller, K. M. O’Neil, S. A. Protopapas, C. Saad-Magalhaes, J. D. Akikusa, A. Thatayatikom, S. Cha, J. C. Nieto-Gonzánlez, M. S. Lo, E. B. Treemarcki, N. Yokogawa, and S. M. Lieberman, for the Childhood Arthritis and Rheumatology Research Alliance and the International Childhood Sjögren Syndrome Workgroup. 2020. Childhood Sjögren syndrome: Features of an international cohort and application of the 2016 ACR/EULAR classification criteria. Rheumatology (Oxford). https://doi.org/10.1093/rheumatology/keaa757.
Benchimol, E. I., G. G. Kaplan, A. R. Otley, G. C. Nguyen, F. E. Underwood, A. Guttmann, J. L. Jones, B. K. Potter, C. A. Catley, Z. J. Nugent, Y. Cui, D. Tanyingoh, N. Mojaverian, A. Bitton, M. W. Carroll, J. deBruyn, T. J. B. Dummer, W. El-Matary, A. M. Griffiths, K. Jacobson, M. E. Kuenzig, D. Leddin, L. M. Lix, D. R. Mack, S. K. Murthy, J. N. P. Sanchez, H. Singh, L. E. Targownik, M. Vutcovici, and C. N. Bernstein. 2017. Rural and urban residence during early life is associated with risk of inflammatory bowel disease: A population-based inception and birth cohort study. The American Journal of Gastroenterology 112(9):1412–1422. https://doi.org/10.1038/ajg.2017.208.
Bengtson, M.-B., G. Aamodt, M. H. Vatn, and J. R. Harris. 2010. Concordance for IBD among twins compared to ordinary siblings — a Norwegian population-based study. Journal of Crohn’s and Colitis 4(3):312–318. https://doi.org/10.1016/j.crohns.2009.12.008.
Bernstein, C. N., C. Burchill, L. E. Targownik, H. Singh, and L. L. Roos. 2019. Events within the first year of life, but not the neonatal period, affect risk for later development of inflammatory bowel diseases. Gastroenterology 156(8):2197.e2191–2197.e2110. https://doi.org/10.1053/j.gastro.2019.02.004.
Boehncke, W.-H., and M. P. Schön. 2015. Psoriasis. The Lancet 386(9997):983–994. https://doi.org/10.1016/s0140-6736(14)61909-7.
Brito-Zerón, P., N. Acar-Denizli, W.-F. Ng, I. F. Horváth, A. Rasmussen, R. Seror, X. Li, C. Baldini, J.-E. Gottenberg, D. Danda, L. Quartuccio, R. Priori, G. Hernandez-Molina, B. Armagan, A. A. Kruize, S.-K. Kwok, M. Kvarnstrom, S. Praprotnik, D. Sene, R. Gerli, R. Solans, M. Rischmueller, T. Mandl, Y. Suzuki, D. Isenberg, V. Valim, P. Wiland, G. Nordmark, G. Fraile, H. Bootsma, H. Nakamura, R. Giacomelli, V. Devauchelle-Pensec, B. Hofauer, M. Bombardieri, V. F. M. Trevisani, D. Hammenfors, S. G. Pasoto, S. Retamozo, T. A. Gheita, F. Atzeni, J. Morel, C. Vollenweider, M. Zeher, K. Sivils, B. Xu, S. Bombardieri, P. Sandhya, S. De Vita, A. Minniti, J. Sánchez-Guerrero, L. Kilic, E. van der Heijden, S.-H. Park, M. Wahren-Herlenius, X. Mariette, and M. Ramos-Casals, for the Sjögren Big Data Consortium. 2020. Epidemiological profile and north–south gradient driving baseline systemic involvement of primary Sjögren’s syndrome. Rheumatology (Oxford) 59(9):2350–2359. https://doi.org/10.1093/rheumatology/kez578.
Bullard, K. M., C. C. Cowie, S. E. Lessem, S. H. Saydah, A. Menke, L. S. Geiss, T. J. Orchard, D. B. Rolka, and G. Imperatore. 2018. Prevalence of diagnosed diabetes in adults by diabetes type - United States, 2016. MMWR: Morbidity and Mortality Weekly Report 67(12):359–361. https://doi.org/10.15585/mmwr.mm6712a2.
Bülow Pedersen, I., and P. Laurberg. 2009. Chapter 60 - antibodies to thyroid peroxidase and thyroglobulin in iodine deficiencies. In Comprehensive handbook of iodine, edited by V. R. Preedy, G. N. Burrow, and R. Watson. San Diego: Academic Press. Pp. 575–585.
Cartee, A. K., L. A. Owens, B. D. Lahr, B. P. Yawn, J. A. Murray, and Y. C. Kudva. 2016. Incidence of type 1 diabetes is not increasing in a population-based cohort in Olmsted County, Minnesota, USA. Mayo Clinic Proceedings 91(8):1066–1073. https://doi.org/10.1016/j.mayocp.2016.05.019.
Cervera, R. 2017. Antiphospholipid syndrome. Thrombosis Research 151(Suppl 1):S43–S47. https://doi.org/10.1016/S0049-3848(17)30066-X.
Charras, A., E. Smith, and C. M. Hedrich. 2021. Systemic lupus erythematosus in children and young people. Current Rheumatology Reports 23(3):20. https://doi.org/10.1007/s11926-021-00985-0.
Chen, Y., T. Wang, X. Liu, and R. R. Shankar. 2019. Prevalence of type 1 and type 2 diabetes among US pediatric population in the MarketScan Multi-State Database, 2002 to 2016. Pediatric Diabetes 20(5):523–529. https://doi.org/10.1111/pedi.12842.
Cooper, G. S., and B. C. Stroehla. 2003. The epidemiology of autoimmune diseases. Autoimmunity Reviews 2(3):119–125. https://doi.org/10.1016/s1568-9972(03)00006-5.
Costenbader, K. H., A. Desai, G. S. Alarcón, L. T. Hiraki, T. Shaykevich, M. A. Brookhart, E. Massarotti, B. Lu, D. H. Solomon, and W. C. Winkelmayer. 2011. Trends in the incidence, demographics, and outcomes of end-stage renal disease due to lupus nephritis in the US from 1995 to 2006. Arthritis and Rheumatism 63(6):1681–1688. https://doi.org/10.1002/art.30293.
Dabelea, D., E. J. Mayer-Davis, S. Saydah, G. Imperatore, B. Linder, J. Divers, R. Bell, A. Badaru, J. W. Talton, T. Crume, A. D. Liese, A. T. Merchant, J. M. Lawrence, K. Reynolds, L. Dolan, L. L. Liu, R. F. Hamman, and Search for Diabetes in Youth Study. 2014. Prevalence of type 1 and type 2 diabetes among children and adolescents from 2001 to 2009. JAMA 311(17):1778–1786. https://doi.org/10.1001/jama.2014.3201.
Dall’Era, M., M. G. Cisternas, K. Snipes, L. J. Herrinton, C. Gordon, and C. G. Helmick. 2017. The incidence and prevalence of systemic lupus erythematosus in San Francisco County, California: The California Lupus Surveillance Project. Arthritis & Rheumatology 69(10):1996–2005. https://doi.org/10.1002/art.40191.
Duarte-Garcia, A., M. M. Pham, C. S. Crowson, S. Amin, K. G. Moder, R. K. Pruthi, K. J. Warrington, and E. L. Matteson. 2019. The epidemiology of antiphospholipid syndrome: A population-based study. Arthritis & Rheumatology 71(9):1545–1552. https://doi.org/10.1002/art.40901.
Eder, L., J. Widdifield, C. F. Rosen, R. Cook, K.-A. Lee, R. Alhusayen, M. J. Paterson, S. Y. Cheng, S. Jabbari, W. Campbell, S. Bernatsky, D. D. Gladman, and K. Tu. 2019. Trends in the prevalence and incidence of psoriasis and psoriatic arthritis in Ontario, Canada: A population-based study. Arthritis Care & Research 71(8):1084–1091. https://doi.org/10.1002/acr.23743.
Erkan, D., M. D. Lockshin, and on behalf of APS ACTION members. 2012. APS Action--Antiphospholipid Syndrome Alliance for Clinical Trials and International Networking. Lupus 21(7):695–698. https://doi.org/10.1177/0961203312437810.
Erkan, D., S. Sciascia, M. L. Bertolaccini, and H. Cohen. 2021. Antiphospholipid syndrome alliance for clinical trials and international networking (APS action): 10-year update. Current Rheumatology Reports 23(6):45. https://doi.org/10.1007/s11926-021-01008-8.
FDA (Food and Drug Administration). 2021. Clinical trial diversity. https://www.fda.gov/consumers/minority-health-and-health-equity/clinical-trial-diversity (accessed March 16, 2022).
Ford, H., E. Waterhouse, D. K. Wallace, and J. B. Winer. 1999. The American College of Rheumatology nomenclature and case definitions for neuropsychiatric lupus syndromes. Arthritis and Rheumatism 42(4):599–608. https://doi.org/10.1002/1529-0131(199904)42:4<599::AID-ANR2>3.0.CO;2-F.
Fraenkel, L., J. M. Bathon, B. R. England, E. W. St Clair, T. Arayssi, K. Carandang, K. D. Deane, M. Genovese, K. K. Huston, G. Kerr, J. Kremer, M. C. Nakamura, L. A. Russell, J. A. Singh, B. J. Smith, J. A. Sparks, S. Venkatachalam, M. E. Weinblatt, M. Al-Gibbawi, J. F. Baker, K. E. Barbour, J. L. Barton, L. Cappelli, F. Chamseddine, M. George, S. R. Johnson, L. Kahale, B. S. Karam, A. M. Khamis, I. Navarro- Millán, R. Mirza, P. Schwab, N. Singh, M. Turgunbaev, A. S. Turner, S. Yaacoub, and E. A. Akl. 2021. 2021 American College of Rheumatology guideline for the treatment of rheumatoid arthritis. Arthritis Care & Research 73(7):924–939. https://doi.org/10.1002/acr.24596.
Furszyfer, J., L. T. Kurland, W. M. McConahey, L. B. Woolner, and L. R. Elveback. 1972. Epidemiologic aspects of Hashimoto’s thyroiditis and Graves’ disease in Rochester, Minnesota (1935–1967), with special reference to temporal trends. Metabolism: Clinical and Experimental 21(3):197–204.
Galofré, J. C., R. V. García-Mayor, E. Fluiters, L. Fernàndez-Calvet, A. Rego, C. Pàramo, and M. A. Andrade. 1994. Incidence of different forms of thyroid dysfunction and its degrees in an iodine sufficient area. Thyroidology 6(2):49–54.
Gandini, A., M. P. Gededzha, T. De Maayer, P. Barrow, and E. Mayne. 2021. Diagnosing coeliac disease: A literature review. Human Immunology. https://doi.org/10.1016/j.humimm.2021.07.015.
Greenberg, J. D., T. Spruill, Y. Shan, G. Reed, J. M. Kremer, J. Potter, Y. Yazici, G. Ogedegbe, and L. R. Harrold. 2013. Racial and ethnic disparities in disease activity in rheumatoid arthritis patients. American Journal of Medicine 126(12):1089–1098. https://doi.org/10.1016/j.amjmed.2013.09.002.
Gulinello, M., J. Wen, and C. Putterman. 2012. Neuropsychiatric symptoms in lupus. Psychiatric Annals 42(9):322–328. https://doi.org/10.3928/00485713-20120906-05.
Hägg, D., M. Eriksson, A. Sundström, and M. Schmitt-Egenolf. 2013. The higher proportion of men with psoriasis treated with biologics may be explained by more severe disease in men. PlOS One 8(5):e63619. https://doi.org/10.1371/journal.pone.0063619.
Harrold, L. R., Y. Shan, S. Rebello, N. Kramer, S. E. Connolly, E. Alemao, S. Kelly, J. M. Kremer, and E. D. Rosenstein. 2020. Prevalence of Sjögren’s syndrome associated with rheumatoid arthritis in the USA: An observational study from the Corrona registry. Clinical Rheumatology 39(6):1899–1905. https://doi.org/10.1007/s10067-020-05004-8.
Hersh, A. O., L. Trupin, J. Yazdany, P. Panopalis, L. Julian, P. Katz, L. A. Criswell, and E. Yelin. 2010. Childhood-onset disease as a predictor of mortality in an adult cohort of patients with systemic lupus erythematosus. Arthritis Care & Research 62(8):1152–1159. https://doi.org/10.1002/acr.20179.
Hiraki, L. T., C. H. Feldman, J. Liu, G. S. Alarcón, M. A. Fischer, W. C. Winkelmayer, and K. H. Costenbader. 2012. Prevalence, incidence and demographics of systemic lupus erythematosus and lupus nephritis among Medicaid-enrolled U.S. Children, 2000–2004. Arthritis and Rheumatism 64(8):2669–2676. https://doi.org/10.1002/art.34472.
Hiraki, L. T., B. Lu, S. R. Alexander, T. Shaykevich, G. S. Alarcon, D. H. Solomon, W. C. Winkelmayer, and K. H. Costenbader. 2011. End-stage renal disease due to lupus nephritis among children in the US, 1995–2006. Arthritis and Rheumatism 63(7):1988–1997. https://doi.org/10.1002/art.30350.
Hollowell, J. G., N. W. Staehling, W. D. Flanders, W. H. Hannon, E. W. Gunter, C. A. Spencer, and L. E. Braverman. 2002. Serum tsh, t(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). The Journal of Clinical Endocrinology & Metabolism 87(2):489–499. https://doi.org/10.1210/jcem.87.2.8182.
Hou, J. K., H. El-Serag, and S. Thirumurthi. 2009. Distribution and manifestations of inflammatory bowel disease in Asians, Hispanics, and African Americans: A systematic review. The American Journal of Gastroenterology 104(8):2100–2109. https://doi.org/10.1038/ajg.2009.190.
Hussain, Y. S., J. C. Hookham, A. Allahabadia, and S. P. Balasubramanian. 2017. Epidemiology, management and outcomes of Graves’ disease—real life data. Endocrine 56(3):568–578. https://doi.org/10.1007/s12020-017-1306-5.
ICD10 Data. 2021. 2022 ICD-10-CM diagnosis code D68.61 https://www.icd10data.com/ICD10CM/Codes/D50-D89/D65-D69/D68-/D68.61 (accessed November 18, 2021).
Icen, M., C. S. Crowson, M. T. McEvoy, F. J. Dann, S. E. Gabriel, and H. Maradit Kremers. 2009. Trends in incidence of adult-onset psoriasis over three decades: A population-based study. Journal of the American Academy of Dermatology 60(3):394–401. https://doi.org/10.1016/j.jaad.2008.10.062.
Izmirly, P. M., J. P. Buyon, I. Wan, H. M. Belmont, S. Sahl, J. E. Salmon, A. Askanase, J. M. Bathon, L. Geraldino-Pardilla, Y. Ali, E. M. Ginzler, C. Putterman, C. Gordon, C. G. Helmick, and H. Parton. 2019. The incidence and prevalence of adult primary Sjögren’s syndrome in New York County. Arthritis Care & Research 71(7):949–960. https://doi.org/10.1002/acr.23707.
Izmirly, P. M., E. D. Ferucci, E. C. Somers, L. Wang, S. S. Lim, C. Drenkard, M. Dall’Era, W. J. McCune, C. Gordon, C. Helmick, and H. Parton. 2021a. Incidence rates of systemic lupus erythematosus in the USA: Estimates from a meta-analysis of the Centers for Disease Control and Prevention national lupus registries. Lupus Science & Medicine 8(1). https://doi.org/10.1136/lupus-2021-000614.
Izmirly, P. M., H. Parton, L. Wang, W. J. McCune, S. S. Lim, C. Drenkard, E. D. Ferucci, M. Dall’Era, C. Gordon, C. G. Helmick, and E. C. Somers. 2021b. Prevalence of systemic lupus erythematosus in the United States: Estimates from a meta-analysis of the centers for disease control and prevention national lupus registries. Arthritis & Rheumatology:1–6. https://doi.org/10.1002/art.41632.
Izmirly, P. M., I. Wan, S. Sahl, J. P. Buyon, H. M. Belmont, J. E. Salmon, A. Askanase, J. M. Bathon, L. Geraldino-Pardilla, Y. Ali, E. M. Ginzler, C. Putterman, C. Gordon, C. G. Helmick, and H. Parton. 2017. The incidence and prevalence of systemic lupus erythematosus in New York County (Manhattan), New York: The Manhattan Lupus Surveillance Program. Arthritis & Rheumatology 69(10):2006–2017. https://doi.org/10.1002/art.40192.
Kanth, R., R. B. Shrestha, I. Rai, J. J. VanWormer, and P. K. Roy. 2017. Incidence of primary biliary cholangitis in a rural midwestern population. Clinical Medicine & Research 15(1–2):13–18. https://doi.org/10.3121/cmr.2017.1351.
Kawatkar, A. A., S. E. Gabriel, and S. J. Jacobsen. 2019. Secular trends in the incidence and prevalence of rheumatoid arthritis within members of an integrated health care delivery system. Rheumatology International 39(3):541–549. https://doi.org/10.1007/s00296-018-04235-y.
Knight, A. M., P. F. Weiss, K. H. Morales, and R. Keren. 2014. National trends in pediatric systemic lupus erythematosus hospitalization in the United States: 2000–2009. Journal of Rheumatology 41(3):539–546. https://doi.org/10.3899/jrheum.130592.
Langer-Gould, A., S. M. Brara, B. E. Beaber, and J. L. Zhang. 2013. Incidence of multiple sclerosis in multiple racial and ethnic groups. Neurology 80(19):1734–1739. https://doi.org/10.1212/WNL.0b013e3182918cc2.
Lim, S. S., A. R. Bayakly, C. G. Helmick, C. Gordon, K. A. Easley, and C. Drenkard. 2014. The incidence and prevalence of systemic lupus erythematosus, 2002–2004: The Georgia Lupus Registry. Arthritis & Rheumatology 66(2):357–368. https://doi.org/10.1002/art.38239.
Lohi, S., K. Mustalahti, K. Kaukinen, K. Laurila, P. Collin, H. Rissanen, O. Lohi, E. Bravi, M. Gasparin, A. Reunanen, and M. Mäki. 2007. Increasing prevalence of coeliac disease over time. Alimentary Pharmacology and Therapeutics 26(9):1217–1225. https://doi.org/10.1111/j.1365-2036.2007.03502.x.
Lu, M., Y. Zhou, I. V. Haller, R. J. Romanelli, J. J. VanWormer, C. V. Rodriguez, H. Anderson, J. A. Boscarino, M. A. Schmidt, Y. G. Daida, A. Sahota, J. Vincent, C. L. Bowlus, K. Lindor, T. Zhang, S. Trudeau, J. Li, L. B. Rupp, S. C. Gordon, and for the Fibrotic Liver Disease Consortium Investigators. 2018. Increasing prevalence of primary biliary cholangitis and reduced mortality with treatment. Clinical Gastroenterology and Hepatology 16(8):1342–1350.e1341 https://doi.org/10.1016/j.cgh.2017.12.033.
Ludvigsson, J. F., A. Rubio-Tapia, C. T. van Dyke, L. J. Melton, 3rd, A. R. Zinsmeister, B. D. Lahr, and J. A. Murray. 2013. Increasing incidence of celiac disease in a North American population. American Journal of Gastroenterology 108(5):818–824. https://doi.org/10.1038/ajg.2013.60.
Lv, T., S. Chen, M. Li, D. Zhang, Y. Kong, and J. Jia. 2021. Regional variation and temporal trend of primary biliary cholangitis epidemiology: A systematic review and meta-analysis. Journal of Gastroenterology and Hepatology 36(6):1423–1434. https://doi.org/10.1111/jgh.15329.
Maciel, G., C. S. Crowson, E. L. Matteson, and D. Cornec. 2017. Prevalence of primary Sjögren’s syndrome in a population-based cohort in the United States. Arthritis Care & Research 69(10):1612–1616. https://doi.org/10.1002/acr.23173.
Mardini, H. E., P. Westgate, and A. Y. Grigorian. 2015. Racial differences in the prevalence of celiac disease in the US population: National health and nutrition examination survey (NHANES) 2009–2012. Digestive Diseases and Sciences 60(6):1738–1742. https://doi.org/10.1007/s10620-014-3514-7.
Masi, A. T. 1965. Hashimoto’s disease. An epidemiological study based on a community-wide hospital survey. Journal of Chronic Diseases 18:35–57. https://doi.org/10.1016/0021-9681(65)90052-4.
Mayer-Davis, E. J., D. Dabelea, and J. M. Lawrence. 2017. Incidence trends of type 1 and type 2 diabetes among youths, 2002-2012. New England Journal of Medicine 376(15):1419–1429. https://doi.org/doi:10.1056/NEJMoa1610187.
Mayo Clinic. 2021. Multiple sclerosis: Overview. https://www.mayoclinic.org/diseases-conditions/multiple-sclerosis/symptoms-causes/syc-20350269 (accessed September 1, 2021).
McDougall, C., K. Hurd, and C. Barnabe. 2017. Systematic review of rheumatic disease epidemiology in the indigenous populations of Canada, the United States, Australia, and New Zealand. Seminars in Arthritis and Rheumatism 46(5):675–686. https://doi.org/10.1016/j.semarthrit.2016.10.010.
McLeod, D. S., P. Caturegli, D. S. Cooper, P. G. Matos, and S. Hutfless. 2014. Variation in rates of autoimmune thyroid disease by race/ethnicity in US military personnel. JAMA 311(15):1563–1565. https://doi.org/10.1001/jama.2013.285606.
Moller, F. T., V. Andersen, J. Wohlfahrt, and T. Jess. 2015. Familial risk of inflammatory bowel disease: A population-based cohort study 1977-2011. American Journal of Gastroenterology 110(4):564–571. https://doi.org/10.1038/ajg.2015.50.
Myasoedova, E., C. S. Crowson, H. M. Kremers, T. M. Therneau, and S. E. Gabriel. 2010. Is the incidence of rheumatoid arthritis rising?: Results from Olmsted County, Minnesota, 1955-2007. Arthritis and Rheumatism 62(6):1576–1582. https://doi.org/10.1002/art.27425.
NORD. 2016. Antiphospholipid syndrome. https://rarediseases.org/rare-diseases/antiphospholipid-syndrome/ (accessed November 18, 2021).
Ouahed, J. 2021. Expanding contributions of monogenic very early onset inflammatory bowel disease. Inflammatory Bowel Diseases 27(11):1870–1872. https://doi.org/10.1093/ibd/izab145.
Paller, A. S., R. Singh, M. Cloutier, M. Gauthier-Loiselle, B. Emond, A. Guérin, and A. Ganguli. 2018. Prevalence of psoriasis in children and adolescents in the United States: A claims-based analysis. Journal of Drugs in Dermatology 17(2):187–194.
Peters, M. G., A. M. Di Bisceglie, K. V. Kowdley, N. L. Flye, V. A. Luketic, S. J. Munoz, G. Garcia-Tsao, T. D. Boyer, J. R. Lake, M. Bonacini, B. Combes, and for the PUMPS Group. 2007. Differences between Caucasian, African American, and Hispanic patients with primary biliary cirrhosis in the United States. Hepatology 46(3):769–775. https://doi.org/10.1002/hep.21759.
Ramos-Casals, M., N. Acar-Denizli, A. Vissink, P. Brito-Zerón, X. Li, F. Carubbi, R. Priori, N. Toplak, C. Baldini, E. Faugier-Fuentes, A. A. Kruize, T. Mandl, M. Tomiita, S. Gandolfo, K. Hashimoto, G. Hernandez-Molina, B. Hofauer, S. Mendieta-Zerón, A. Rasmussen, P. Sandhya, D. Sene, V. F. M. Trevisani, D. Isenberg, E. Sundberg, S. G. Pasoto, A. Sebastian, Y. Suzuki, S. Retamozo, B. Xu, R. Giacomelli, A. Gattamelata, M. Bizjak, S. Bombardieri, R.-E. Loor-Chavez, A. Hinrichs, P. Olsson, H. Bootsma, S. M. Lieberman, and the Sjögren Big Data Consortium. 2021. Childhood-onset of primary Sjögren’s syndrome: Phenotypic characterization at diagnosis of 158 children. Rheumatology (Oxford) 60(10):4558–4567. https://doi.org/10.1093/rheumatology/keab032.
Ramos-Casals, M., P. Brito- Zerón, S. Bombardieri, H. Bootsma, S. De Vita, T. Dörner, B. A. Fisher, J.-E. Gottenberg, G. Hernandez-Molina, A. Kocher, B. Kostov, A. A. Kruize, T. Mandl, W.-F. Ng, S. Retamozo, R. Seror, Y. Shoenfeld, A. Sisó-Almirall, A. G. Tzioufas, C. Vitali, S. Bowman, and X. Mariette, and on behalf of the EULAR-Sjögren Syndrome Task Force Group. 2020. EULAR recommendations for the management of Sjögren’s syndrome with topical and systemic therapies. Annals of the Rheumatic Diseases 79(1):3–18. https://doi.org/10.1136/annrheumdis-2019-216114.
Rasmussen, A., L. Radfar, D. Lewis, K. Grundahl, D. U. Stone, C. E. Kaufman, N. L. Rhodus, B. Segal, D. J. Wallace, M. H. Weisman, S. Venuturupalli, B. T. Kurien, C. J. Lessard, K. L. Sivils, and R. H. Scofield. 2016. Previous diagnosis of Sjögren’s syndrome as rheumatoid arthritis or systemic lupus erythematosus. Rheumatology (Oxford) 55(7):1195–1201. https://doi.org/10.1093/rheumatology/kew023.
Roberts, S. B., G. M. Hirschfield, L. J. Worobetz, C. Vincent, J. A. Flemming, A. Cheung, K. Qumosani, M. Swain, D. Grbic, H. H. Ko, K. Peltekian, N. Selzner, L. Abrahamyan, B. Aziz, E. Lytvyak, K. Tirona, A. F. Gulamhusein, H. L. A. Janssen, A. J. Montano-Loza, A. L. Mason, B. E. Hansen, and D. Canadian Network for Autoimmune Liver. 2022. Ethnicity, disease severity, and survival in Canadian patients with primary biliary cholangitis. Hepatology 00:1–14. https://doi.org/10.1002/hep.32426.
Rogers, M. A. M., C. Kim, T. Banerjee, and J. M. Lee. 2017. Fluctuations in the incidence of type 1 diabetes in the United States from 2001 to 2015: A longitudinal study. BMC Medicine 15(1):199. https://doi.org/10.1186/s12916-017-0958-6.
Romanelli, R. J., Q. Huang, J. Lacy, L. Hashemi, A. Wong, and A. Smith. 2020. Multiple sclerosis in a multi-ethnic population from northern California: A retrospective analysis, 2010–2016. BMC Neurology 20(1):163. https://doi.org/10.1186/s12883-020-01749-6.
Rosen, M. J., A. Dhawan, and S. A. Saeed. 2015. Inflammatory bowel disease in children and adolescents. JAMA Pediatrics 169(11):1053–1060. https://doi.org/10.1001/jamapediatrics.2015.1982.
Rubinstein, T. B., and A. M. Knight. 2020. Disparities in childhood-onset lupus. Rheumatic Disease Clinics of North America 46(4):661–672. https://doi.org/10.1016/j.rdc.2020.07.007.
Safiri, S., A. A. Kolahi, D. Hoy, E. Smith, D. Bettampadi, M. A. Mansournia, A. Almasi-Hashiani, A. Ashrafi-Asgarabad, M. Moradi-Lakeh, M. Qorbani, G. Collins, A. D. Woolf, L. March, and M. Cross. 2019. Global, regional and national burden of rheumatoid arthritis 1990–2017: A systematic analysis of the global burden of disease study 2017. Annals of the Rheumatic Diseases 78(11):1463–1471. https://doi.org/10.1136/annrheumdis-2019-215920.
Santos, M. P. C., C. Gomes, and J. Torres. 2018. Familial and ethnic risk in inflammatory bowel disease. Annals of Gastroenteroly 31(1):14–23. https://doi.org/10.20524/aog.2017.0208.
Scofield, R. H., R. Sharma, N. Pezant, J. A. Kelly, L. Radfar, D. M. Lewis, C. E. Kaufman, S. Cioli, J. Harris, K. Grundahl, N. L. Rhodus, D. J. Wallace, M. H. Weisman, S. Venuturupalli, M. T. Brennan, K. A. Koelsch, C. J. Lessard, C. G. Montgomery, K. L. Sivils, and A. Rasmussen. 2020. American Indians have a higher risk of Sjögren’s syndrome and more disease activity than European Americans and African Americans. Arthritis Care & Research 72(8):1049–1056. https://doi.org/10.1002/acr.24003.
Shivashankar, R., W. J. Tremaine, W. S. Harmsen, and E. V. Loftus, Jr. 2017. Incidence and prevalence of Crohn’s disease and ulcerative colitis in Olmsted County, Minnesota from 1970 through 2010. Clinical Gastroenterology and Hepatology 15(6):857–863. https://doi.org/10.1016/j.cgh.2016.10.039.
Singh, P., A. Arora, T. A. Strand, D. A. Leffler, C. Catassi, P. H. Green, C. P. Kelly, V. Ahuja, and G. K. Makharia. 2018. Global prevalence of celiac disease: Systematic review and meta-analysis. Clinical Gastroenterology and Hepatology 16(6):823–836.e822. https://doi.org/10.1016/j.cgh.2017.06.037.
Sjögren’s Foundation. 2021. Living with Sjögren’s—Sjögren’s in children. https://www.sjog-rens.org/living-with-sjogrens/sjogrens-in-children (accessed March 21, 2022).
Somers, E. C., W. Marder, P. Cagnoli, E. E. Lewis, P. DeGuire, C. Gordon, C. G. Helmick, L. Wang, J. J. Wing, J. P. Dhar, J. Leisen, D. Shaltis, and W. J. McCune. 2014. Population-based incidence and prevalence of systemic lupus erythematosus: The Michigan Lupus Epidemiology and Surveillance Program. Arthritis & Rheumatology 66(2):369–378. https://doi.org/10.1002/art.38238.
Stuifbergen, A., H. Becker, C. Phillips, S. Horton, J. Morrison, and F. Perez. 2021. Experiences of African American women with multiple sclerosis. International Journal of MS Care 23(2):59–65. https://doi.org/10.7224/1537-2073.2019-068.
Tremlett, H., D. Paty, and V. Devonshire. 2005. The natural history of primary progressive MS in British Columbia, Canada. Neurology 65(12):1919–1923. https://doi.org/10.1212/01.wnl.0000188880.17038.1d.
Vanderpump, M. P. J., W. M. G. Tunbridge, J. M. French, D. Appleton, D. Bates, F. Clark, J. Grimley Evans, D. M. Hasan, H. Rodgers, F. Tunbridge, and E. T. Young. 1995. The incidence of thyroid disorders in the community: A twenty-year follow-up of the Whickham survey. Clinical Endocrinology 43(1):55–68. https://doi.org/10.1111/j.1365-2265.1995.tb01894.x.
Wallin, M. T., W. J. Culpepper, J. D. Campbell, L. M. Nelson, A. Langer-Gould, R. A. Marrie, G. R. Cutter, W. E. Kaye, L. Wagner, H. Tremlett, S. L. Buka, P. Dilokthornsakul, B. Topol, L. H. Chen, N. G. LaRocca, and on behalf of the US Multiple Sclerosis Prevalence Workgroup. 2019. The prevalence of MS in the United States: A population-based estimate using health claims data. Neurology 92(10):e1029–e1040. https://doi.org/10.1212/WNL.0000000000007035.
Walton, C., R. King, L. Rechtman, W. Kaye, E. Leray, R. A. Marrie, N. Robertson, N. La Rocca, B. Uitdehaag, I. van der Mei, M. Wallin, A. Helme, C. Angood Napier, N. Rijke, and P. Baneke. 2020. Rising prevalence of multiple sclerosis worldwide: Insights from the atlas of MS, third edition. Multiple Sclerosis 26(14):1816–1821. https://doi.org/10.1177/1352458520970841.
Wouters, H. J. C. M., S. N. Slagter, A. C. Muller Kobold, M. M. van der Klauw, and B. H. R. Wolffenbuttel. 2020. Epidemiology of thyroid disorders in the Lifelines Cohort Study (the Netherlands). PLOS ONE 15(11):e0242795. https://doi.org/10.1371/journal.pone.0242795.
Xu, G., B. Liu, Y. Sun, Y. Du, L. G. Snetselaar, F. B. Hu, and W. Bao. 2018. Prevalence of diagnosed type 1 and type 2 diabetes among US adults in 2016 and 2017: Population based study. BMJ 362:k1497. https://doi.org/10.1136/bmj.k1497.
Yan, K., C. Balijepalli, K. Desai, L. Gullapalli, and E. Druyts. 2020. Epidemiology of pediatric multiple sclerosis: A systematic literature review and meta-analysis. Multiple Sclerosis and Related Disorders 44:102260. https://doi.org/10.1016/j.msard.2020.102260.
Ye, Y., S. Manne, W. R. Treem, and D. Bennett. 2020. Prevalence of inflammatory bowel disease in pediatric and adult populations: Recent estimates from large national databases in the United States, 2007–2016. Inflammatory Bowel Diseases 26(4):619–625. https://doi.org/10.1093/ibd/izz182.
Yeh, E. A., T. Chitnis, L. Krupp, J. Ness, D. Chabas, N. Kuntz, E. Waubant, and for the U. S. Network of Pediatric Multiple Sclerosis Centers of Excellence. 2009. Pediatric multiple sclerosis. Nature Reviews: Neurology 5(11):621–631. https://doi.org/10.1038/nrneurol.2009.158.
Yen, E. Y., and R. R. Singh. 2018. Brief report: Lupus-an unrecognized leading cause of death in young females: A population-based study using nationwide death certificates, 2000-2015. Arthritis & Rheumatology 70(8):1251–1255. https://doi.org/10.1002/art.40512.
Yoshida, E. M., M. Riley, and L. T. Arbour. 2006. Autoimmune liver disease and the Canadian First Nations Aboriginal communities of British Columbia’s Pacific Northwest. World Journal of Gastroenterology 12(23):3625–3627. https://doi.org/10.3748/wjg.v12.i23.3625.
























