Infection, Immunity, and Inflammation:
Unraveling and Applying the Capacities of the Immune System
Tremendous advances in understanding of the immune system and inflammation have led to powerful new treatments against infectious and autoimmune diseases. This golden age of immunology research continues today, offering promise that the body’s immune system could be harnessed to fight a wide range of diseases.
1974
Differentiating Self from Non-Self
Receptors on cells known as T cells are responsible for recognizing foreign molecules in the body and triggering an immune system response. In 1974, scientists discovered that these receptors can only recognize foreign molecules that are bound to proteins generated by what is known as the major histocompatibility complex (MHC), a set of genes that varies from person to person and population to population. This discovery heightened understanding of the immune system by revealing the link between people’s immune responses and their genetic profiles.
1976
Understanding the Vast Diversity of Antibodies
B cells help regulate the immune system by producing antibodies and more. Our bodies can generate many millions of structurally different antibodies that defend us against a host of foreign molecules, organisms, or substances. For many years, scientists were unsure whether the genetic diversity required to produce millions of antibodies was generated during evolution (and therefore is carried in sperm and egg cells) or occurs during development (and is generated in other body cells). In 1976, researchers answered this question by showing that the rearrangement of genes in cells other than sperm and egg cells allows the generation of a great diversity of antibodies from a finite number of genes, solving the mystery and opening the way to further discoveries involving gene rearrangements.
1982
Infectious Proteins
In 1982, the discovery that variants of normal proteins can act as infectious agents expanded understanding of disease mechanisms and transmission. These proteins, named “prions” (short for proteinaceous infectious particles), are misfolded forms of proteins that act as templates to to promote misfolding among other proteins. Once thought to cause only relatively rare brain diseases, similar aggregates of misfolded proteins are found in Alzheimer’s disease, some cancers, and other disorders, suggesting that protein misfolding may play a larger role in human diseases than previously suspected.
1986
Two Types of Immune Help
A balanced immune response requires the proper regulation of immune cells known as helper T cells. In 1986, scientists identified two types of helper T cells that differ in how they respond to foreign molecules. One type orchestrates an inflammatory response that leads to direct killing of invading microbes, while the other helps stimulate the production of antibodies that ultimately destroy the invader. Recently, the discovery of new subsets of helper T cells has further illuminated the remarkable complexity of the human immune system.
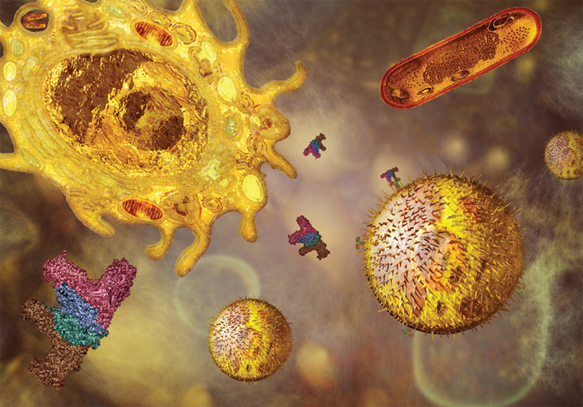
1990s
Recognizing Microbial Invaders
Roughly a decade after the 1985 discovery of a protein known as Toll in the fruit fly Drosophila melanogaster, many laboratories discovered Toll-like receptors (TLRs) on human cells that play a vital role in activating the immune system in response to danger signals. TLRs are a class of proteins that are primarily produced by white blood cells and recognize white blood cells and recognize molecules broadly shared by pathogenic microbes. The specificity of TLRs and an elaborate regulatory system associated with these receptors ensure that they only recognize molecules associated with these microbial threats, do not activate an inappropriate response to benign microbes, and are highly specific in their response to the different types of antagonistic microbes.
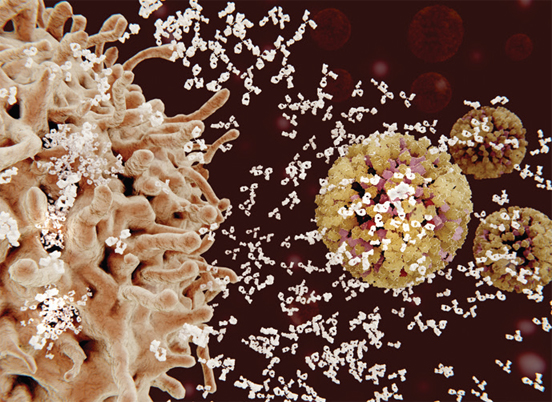
2001
Triggering the Death of Cancer Cells
In 2001, scientists discovered the FOXO3 gene, which plays a pivotal role in causing cell death. Mounting evidence suggests that FOXO3 normally functions as a tumor suppressor by detecting cancerous cells and triggering a variety of responses to these abnormalities, including cell death, but is dysregulated in several types of cancer. Better understanding of the protein encoded by this gene may provide new possibilities for cancer treatment and may have implications for human longevity.

2006
A Cell That Kills Other Cells
Natural killer (NK) cells are a type of white blood cell that provides a rapid response to cells that have been invaded by a virus or have become cancerous. For many years, NK cells were thought to lack specific cell surface receptors that, if present, would enable them to recognize and kill infected or abnormal cells that they have previously encountered. However, in 2006, a subset of NK cells was found to possess memories of past exposure to a threat, demonstrating a new immunological capacity independent of B and T cells.
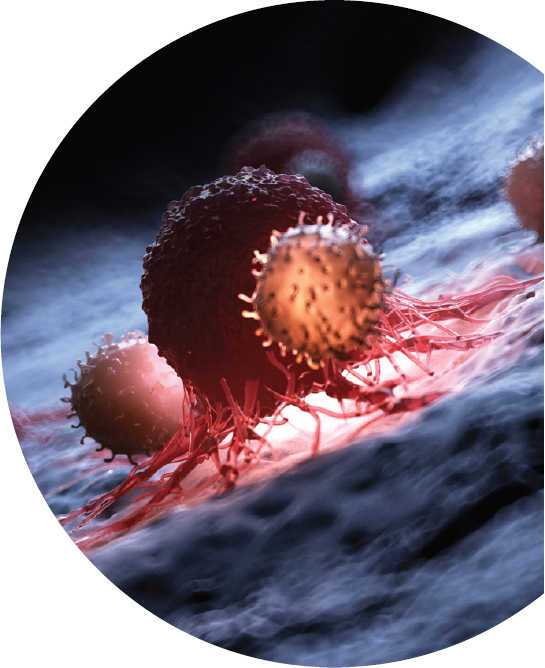
2011
Engineering the Immune System to Defeat Cancer
T cells were first used in 2011 to treat advanced cancers. By modifying the cells to express particular receptors, they could be targeted at malignancies that display the protein they were engineered to recognize. Treatment using these engineered immune cells delayed cancer progression and launched a wave of research into modulating the immune system to fight disease.

2017
Adoptive Cell Transfer to Treat Cancer
In 2017, the U.S. Food and Drug Administration approved a therapy for certain kinds of leukemia and lymphoma that uses genetically engineered T cells. In an approach known as adoptive cell transfer, T cells from a person with cancer are removed, genetically altered to target tumor cells, and then transferred back into the person. The technique heralds a new era of individualized cancer therapy.
Continued research into the immune system will drive continual improvements in the diagnosis, prevention, and treatment of disease. At the same time, better understanding of inflammation will provide key insights into widespread diseases, such as dementia and obesity, where inflammation plays a major role.




