Metabolism and Bioenergetics:
Linking Energy Use to Health

Building on research conducted during the first seven decades of the 20th century, scientists have made dramatic advances in understanding metabolism and bioenergetics since 1970. Defined as the chemical processes that maintain life, metabolism is highly individual, with both genetic and environmental influences. But research has identified larger patterns and factors that affect metabolism, providing new insights into body weight, metabolic disorders, and the development and progression of disease.

1980s
Understanding Photosynthesis
The conversion of light from the sun into energy to drive biological processes, known as photosynthesis, involves the transport of electrons among proteins bound in specialized membranes. Before the early 1980s, the structure of these membrane-bound proteins remained unknown. Then scientists determined the three-dimensional structure of a protein complex that performs the primary energy conversion reaction in a purple bacterium. Though photosynthesis in bacteria is simpler than in algae and green plants, the commonalities among organisms have resulted in greatly increased understanding of how living things capture energy from light.
1980s
A Multifaceted Regulatory Mechanism
Thousands of structurally different proteins within the body regulate biochemical processes. A critical mechanism in this regulation is phosphorylation, a process probed in the late 1980s, in which enzymes attach a phosphate group to targeted proteins. This chemical alteration is responsible for such vital mechanisms as the regulation of blood sugar, the battle against infection, and the development of cancers. The purification and characterization of proteins that carry out phosphorylation or become phosphorylated themselves launched a wave of research into regulatory processes within the body.

1980s
The Formation of Adenosine Triphosphate
The molecule adenosine triphosphate (ATP) functions as the carrier of energy in all living organisms. An enzyme known as ATP synthase, characterized in the 1980s, uses the energy derived from nutrients to add a phosphate group to the molecule adenosine diphosphate. The resulting ATP molecule then transfers this energy to other biochemical molecules, driving the fundamental processes of life.

1990s
Rapid-Effect Steroid Hormones
For a long time, scientists believed that steroid hormones, which regulate many physiological and developmental processes, act by binding to specific receptors in target cells. But by the mid-1990s they had learned that steroid hormones can act much more quickly through such mechanisms as biochemically altering cell membranes. These fast-acting mechanisms are involved in many cell functions and in the development of hormone-responsive cancers, offering new opportunities for chemotherapeutics.

1990s
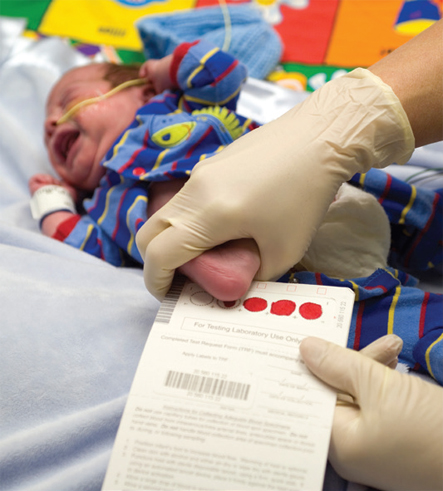
Expanding the Reach of Newborn Screening
By 1970 it was possible to diagnose a handful of treatable diseases in newborn babies who appeared healthy by examining their blood a day or two after birth. Technological advances in mass spectrometry in the 1990s made it possible to diagnose dozens of these diseases at once by examining metabolite levels in a drop of blood. These expanded newborn screening assays are now performed in millions of babies each year, allowing physicians to begin lifesaving therapies before the babies even appear sick.
2008
Cancer-Causing Metabolites
Tumors frequently display altered metabolism, but until recently it was unknown whether these changes could initiate cancer. Starting in 2008, two related metabolic enzymes were discovered to be frequently mutated in cancers of the brain, bone marrow, and other organs. The mutant enzymes produce large quantities of a metabolite that prevents cells from activating genes needed for cells to mature properly. Some leukemias can now be treated with drugs that block these mutant enzymes and cause cells to lose their malignant properties.

2010
The Progression of Multifactorial Disorders
With multifactorial diseases, heterogeneous combinations of genetic and environmental factors account for the origins of the disease, but how this happens and how it varies from one individual to another have been difficult to unravel. Nevertheless, progress is being made. In 2010, for example, scientists discovered that people have inherited differences in the ability of cells to oxidize nutrients and release chemical energy to be stored in adenosine triphosphate. These differences in cellular energetics may contribute to diseases subject to environmental factors such as diabetes, cancer, and neurodegenerative diseases, and better understanding of these linkages could open new avenues for treatment.

Improved knowledge of metabolic processes will enable personalized medicine, in which therapies are tailored to individual patients. Rising rates of obesity and diabetes make this approach especially critical to prevent an onslaught of diseases related to metabolic dysfunction.




