7
Infrastructure for Scaling and Spreading Whole Health
Chapter 6 described the contextual conditions needed to scale and spread whole health, including systemic change and social movement, advances in structures and processes, and enhanced integration of services. New health ecosystems can only be created and sustained if a strong foundational infrastructure supports and promotes these conditions (see Figure 6-1 in Chapter 6). Health informatics, workforce training and education, the measurement of whole health for learning and accountability, and financing are pillars of health care at the national level. For that reason they are often the focus of debate on health care reform. Changing them will not be easy or quick, but it is equally clear that, with inspired leadership and appropriate resources, change is possible. Meaningful advances to these infrastructural elements can occur through the public policy process. Advances can also come from the private sector, particularly in the case of payment reform. This chapter describes the infrastructural elements necessary to scale and spread whole health throughout the United States.
HEALTH INFORMATICS
A substantial share of clinical and personal health information now lives online or in electronic platforms. While paper records persist in many small practices, most larger practices and provider organizations in the United States now use electronic medical records. In this era, health informatics is part of the foundational infrastructure for supporting the scale and spread of whole health. Health informatics act as the train tracks upon which information flows among clinicians, managers, planners, payors,
and researchers as well as from the health care system to public health, community programs, and social services. It also supports the patient, interprofessional care team, delivery system, and broader care ecosystem. Existing health informatics functionality can be part of delivering whole health, but improvements in functionality are needed for whole health to fully succeed. The changes that are needed extend far beyond simple technical advances and will need to include changes to core functions, data governance, human factors design, and even definitions concerning who is included in the care team that has access to data and information systems (Gamache et al., 2018). To help people, families, and communities achieve whole health, health informatics will need to support all five foundational elements of whole health—being people-centered, comprehensive and holistic, upstream-focused, equitable and accountable, and grounded in team well-being (see Chapter 2 for a detailed description of the committee’s five foundational elements of whole health).
This committee agrees that implementing the two health informatics–related recommendations from the 2021 report, Implementing High-Quality Primary Care: Rebuilding the Foundation of Health Care, would help meet the needs of whole health systems. These recommendations were (1) to develop the next phase of digital health certification standards that support relationship-based, continuous, people-centered care; simplify user experience; ensure equitable access; and hold vendors accountable; and (2) to adopt a comprehensive aggregate patient data system usable by any certified digital health tool (Krist et al., 2021; NASEM, 2021). Over the past decade the National Academies of Sciences, Engineering, and Medicine have repeatedly called for needed informatics changes. These changes also need to be implemented to support whole health care. Examples of reports include Health IT and Patient Safety: Building Better Systems for Better Care (IOM, 2012), Taking Action Against Clinician Burnout: A Systems Approach to Professional Well-Being (NASEM, 2019), and Building Data Capacity for Patient-Centered Outcomes Research: Priorities for the Next Decade (NASEM, 2022).
While these advances are necessary to support primary care, they are not sufficient for effectively scaling and spreading whole health because whole health care will require more robust, people-centered tools that gather and sort information about what matters most to individuals, families, and communities and mapping progress toward their goals; extending the use of technology to all whole health interprofessional team members; supporting teamwork through communication, collaboration, and coordination tools; addressing upstream determinants of health which are currently segregated from clinical record systems; promoting accessible and proactive care through virtual technologies and automated functions; and ensuring that technology facilitates care team well-being by being user friendly and
capable of synthesizing and presenting data that supports, instead of adding to, clinicians’ work. Both the Department of Veterans Affairs (VA) and the broader health care system nationally will need to make changes in their health informatics infrastructure to promote whole health. Changes should span conventional medical settings, comprehensive and integrative health settings, community settings, and social care settings. VA health information systems will need to include Veterans Benefits Administration (VBA) services as well as community and social care systems that care for veterans across systems of care.
Health Informatics Infrastructure at VA
VA has long been an early adopter and leader in health informatics (VA, 2022a). It began researching electronic health records (EHRs) in the 1970s; made the Veterans Health Information Systems and Technology Architecture (VistA) content open source in the 1980s; developed information exchanges between VA, the Department of Defense, and the Indian Health Service in the early 2000s; launched its My HealtheVet patient portal in 2003; led electronic prescribing in 2009; and pioneered information sharing in 2010 with Blue Button functionality and additional information exchanges (Hogan et al., 2014; Klein et al., 2015). In recent decades, as the rest of the health care system struggled with a fragmented ecosystem of health informatics platforms, clinicians often regarded the VA EHR as exemplifying a high-functioning system that contains standardized health care information for patients and that clinicians from any VA facility nationwide can access. Looking to the future, VA can continue to be a leader and transform health informatics to better support whole health.
VA’s legacy EHR system, VistA, consisted of over 170 clinical, financial, and administrative applications which supported millions of veteran medical encounters per year at VA facilities nationwide (Rose et al., 2021). Several modernization initiatives have led to more than 130 unique iterations of VistA, requiring sequential “patches” which needed to be installed, tested, and adjusted at facilities across VA’s national system. While VistA led the field for decades, it also created challenges in standardizing processes, coordinating care across medical facilities, and information sharing with other health care providers. To address this, VA is transitioning its legacy systems to a commercial EHR, Cerner. The first deployment of the system-wide transformation occurred in October 2020 and is slated to be completed in 2028. While this transition may better support seamless care across health care settings that span active duty to veteran status and include community health partners, it will also introduce new health informatics challenges associated with commercial products and vendors (Colicchio et al., 2019; Marwaha et al., 2022).
VA has laid out its proposed digital transformation with a clear roadmap that will modernize and consolidate digital tools into an enterprise-wide, self-service platform accessible from a single place on the VA website (VA, 2020). The roadmap includes support to prioritize the delivery of comprehensive and integrated whole health care, which includes functions such as capturing, managing, and sharing personalized health plans. VHA’s Health Information Strategic Plan (HISP) for 2022–2026 defines the direction for the future informatics environment. Although whole health is not mentioned in HISP as a driver, meeting the plan’s goals and objectives will provide some of the infrastructure needed to spread and scale whole health, including enterprise-wide data and information standardization, secure information exchange, and new agility in meeting the health care needs of a diverse veteran population. As the strategic plan is updated, it will be important for the HISP to ensure that all five foundational elements of whole health are being addressed (VA, 2021a).
National Health Informatics Infrastructure
Most health care systems, clinical practices, and patients have adopted EHRs and patient portals. In 2017, 86 percent of all office-based physicians and 95 percent of health systems in the United States reported using an EHR (ONC, 2019a,b). In a 2022 survey of physicians, nearly all clinicians (93 percent) reported that technology enabled them to provide better care for people (AMA, 2022). Research has shown that EHRs improve patient care overall, increase clinician access to patient information, identify potential medication errors, alert teams to critical laboratory values, remind patients and clinicians about recommended care, improve the ordering of appropriate tests, and improve patient–clinician communication, among other things (King et al., 2014; Krist et al., 2012, 2017).
Despite these advantages, health informatics has yet to live up to its full potential for advancing health for several reasons. Health informatics systems remain siloed; patient access is often limited and non-transferable; information exchanges between systems are rudimentary; there has been insufficient attention to usability and making users’ lives easier; systems have not incorporated many of the technological innovations available to improve functionality; and EHR vendors and health systems can maximize market power by limiting or even blocking the flow or exchange of health information (Everson et al., 2021; ONC, 2015; Vest and Kash, 2016). Clinicians commonly cite EHRs as a primary driver of burnout, as they often add to workload and detract from connecting with patients (NASEM, 2019). As clinical data amasses in a person’s health record, the risk of data overload increases. Most EHRs currently lack tools to help make sense of clinical information and trend data, and they have only basic mechanisms
to sort and filter information (Furlow, 2020; Krist et al., 2021; Singh et al., 2013). Patient engagement tools have not advanced much beyond patient portals that mainly allow patients to email clinicians, schedule appointments, and see their test results. Functions to help patients understand their health information and activate and engage patients as partners in care are only just beginning to emerge (Krist and Woolf, 2011; Odendaal et al., 2020; Sawesi et al., 2016).
Recent policies will support the health informatics transformation needs for whole health. Specifically, the 21st Century Cures Act mandates that health informatics systems adopt standardized application programming interfaces (APIs), which will allow individuals to access structured electronic health information using smartphone applications securely and easily (ONC, 2022a). The Cures Act also mandates implementation of Trusted Exchange Framework and Common Agreement interoperability standards to ease information sharing across networks, and it penalizes health systems and EHR vendors for blocking information. This policy can improve information sharing among clinicians and patients, which is essential to creating an infrastructure that will support whole health systems of care. Similarly, the United States Core Data for Interoperability is a standardized set of health data classes and constituent data elements for nationwide, interoperable health information exchange, and it is now required as part of the new API certification criterion (ONC, 2022b). Among its data classes that could apply to whole health are patient demographics, smoking status, health concerns, patient goals, and assessment and treatment plans. These data classes and constituent data elements will need to be expanded to fully support whole health care.
People-Centered Tools
As described throughout this report, whole health is fundamentally people-centered. It starts by identifying what matters to people and tailoring care to a person, family, and community’s needs, preferences, and goals. This type of information is not routinely part of the conventional medical record. Some EHRs have started to include a new field to record “what matters most to people,” but this is not yet integrated as an essential, structured, or sharable element of the EHR. Furthermore, EHRs lack processes to identify and create care plans and goals or to track how people are doing with respect to these goals. There are emerging applications to support goal setting and guide teams in supporting people to achieve their goals, but more is needed (Glasgow et al., 2019; Krist et al., 2020b).
Moreover, scaling and spreading whole health will require that people have access to their personal records and that people’s records follow them over time, regardless of the clinician or organization providing
health services. As people move between providers and locations, enhanced interoperability will enable systems to communicate with each other and clinicians to understand each individual’s history, data, and health and well-being priorities.
Another important people-centered health informatics need for scaling and spreading whole health is more people-facing supports and tools. These are necessary for engaging and activating people as partners in their care. VA has developed several digital tools to support its whole health efforts, including a mobile application and web-based resource library (VA, 2022b). Additional resources developed by VA include the #LiveWholeHealth self-care blog series, links to the My HealtheVet online personal health record, and a mindfulness podcast series (Box 7-1). Other integrative health resources that VA integrative health coordinating centers offer include websites; videos; audio/podcasts; journals; online classes for yoga, meditation, Pilates, and tai chi; biofeedback experiential resources; and meditation/guided imagery experiential resources.
Digital health and the move to people-centered care has also been accelerated since the COVID-19 pandemic. While gaining experience in developing digital health platforms, a new emphasis has been placed on seeking out digital health tools developed by trustworthy sources. The Defense Health Agency offers a wide variety of free apps and podcasts grounded in clinical research and vetted to ensure quality and safety (Apple Inc, 2022a,b). Furthermore, telehealth has been expanded, promoting virtual access to clinical
and social services (Perlin et al., 2004). These innovations are components of an accelerating shift from developing information technology (IT) applications that address the needs of health care providers and institutions to a rapid propagation of patient-centered applications.
Including All Interprofessional Care Team Members and Promoting Teamwork
As this report has emphasized, it takes an interprofessional team to deliver whole health services and care. Team members span clinical and community care settings and include conventional medical care, new medical services, integrative health approaches, and social services delivery. Veterans may receive whole health care services in both VA and civilian settings. Technology is needed to share information, support communications, coordinate services, and promote teamwork across all interprofessional team members and in all settings. This can occur partially through the interoperability of health informatics systems and real-time health information exchange. While there have been significant advances in interoperability, creating complete and real-time health information exchanges remains an ongoing challenge across the health care system.
Mandates to use the Substitutable Medical Applications and Reusable Technologies on Fast Health Interoperability Resources (SMART on FHIR) have helped with data sharing. FHIR provides a common format for sharing health information, allows medical applications to run unmodified across different health informatics systems, and facilitates data flow across otherwise incompatible systems (Mandel et al., 2016). The Centers for Medicare & Medicaid Services (CMS) has further reinforced the national movement toward promoting interoperability with its proposed rule to move the health care ecosystem in the direction of interoperability and commitment to the 21st Century Cures Act’s mandate to improve the quality and accessibility of information for people to make informed health care decisions (HHS, 2020). In addition, health care providers, health informatics developers, and health systems have been prohibited since April 2021 from information blocking and face substantial fines if they cannot electronically share on demand patient demographics, smoking status, health concerns, patient goals, assessment and plan of treatment, social needs assessment, encounter information, clinical notes, and vital signs (ONC, 2020).
For its part, VA has published its strategic plan to create seamless and secure interoperability and health information exchanges of veterans’ health information among VA, the Department of Defense, and the community (Office of Information and Technology, 2020). The strategic plan includes developing and implementing the FHIR data architecture, the use of APIs and cloud technologies, and the incorporation of cybersecurity and
privacy tools. The goal of the strategic plan is to enable veterans to access their secure personal health information in any setting. To scale and spread whole health, health systems can use this infrastructure to support the entire interprofessional team.
In May 2021 the Office of the National Coordinator for Health Information Technology (ONC) engaged the public to gather input on measurable health interoperability outcome statements for 2030 (ONC, 2021). ONC is the principal federal entity charged with coordinating nationwide efforts to implement and use the most advanced health information technology and exchange health information electronically. If these outcome statements are implemented effectively, they would further advance the interoperability of health informatics functions needed to support whole health in VA and nationally (Box 7-2).
There are unique challenges to including community and social service providers—essential whole health care team members—in health information exchanges that interoperability alone will not solve. Community and social service providers often use information and communication systems that are different from those used by health care providers. In general, these community and social informatics systems do not exchange information
with health informatics platforms. Adding to this siloing of care and information, community and social service providers do not have access to health informatics systems for their clients, and health care providers do not have access to the community and social services information systems.
VA has made some initial advances to break down the silos between the health care information system and the community and social services information system. The VA Maintaining Internal Systems and Strengthening Integrated Outside Networks (MISSION) Act of 2018 (see Chapter 6) included language aimed at improving the sharing of previously protected health information between VA and the community partners. The MISSION Act’s focus is primarily on providing veterans access to community health care partners, including those that provide integrative health approaches, but not to community and social services providers. VA Direct is a secure system that allows the exchange of select health information to and from community partners. For the most part this system has been used for communication between conventional medical providers, but it could be used for community and social services providers (VA OIG, 2020). The fact that the VHA’s health informatics systems and the VBA’s benefits information systems are not integrated and do not communicate—a capability essential for coordinating the delivery of whole health care—exemplifies the scope of this problem within VA.
Nationally, there have been multiple efforts to integrate social determinants of health into EHRs (Cantor and Thorpe, 2018; IOM, 2014, 2015). Most health informatics efforts to date have focused on collecting and documenting social needs as part of conventional care, although developers have created and tested several closed-loop referral and communication systems, including CommunityRx, Unite US, and HelpFinder (Curt et al., 2021; Help Finder, 2022; Lindau et al., 2019; Tung et al., 2020; Unite US, 2018). Studies have shown that these closed-loop referral systems are feasible, increase referrals, improve patient knowledge and confidence in addressing needs, and fulfill social needs. Yet without further investment in the social programs themselves, creating these informatics linkages may not be scalable or sustainable. The eHealth Exchange, a network of networks, has also proven successful in seamlessly sending millions of COVID-19 testing and diagnoses reports between health care and public health settings (eHealth Exchange, n.d.). This exchange extends to a broader range of whole health providers not participating in current health information exchanges. A number of systems have also used accreditation through DirectTrust to determine which partners with whom they will share health information securely (DirectTrust, n.d.).
Beyond the first step of data sharing, technology is needed to promote teamwork. Historically, health information technologies have been primarily designed to support the tasks of individual people—individual clinicians
and individual patients. Whole health care is team based and relies on a coordinated and effective interprofessional team. Technologies will need to continue to support task work but also support teamwork with communication, collaboration, and coordination tools (Carayon and Hoonakker, 2019; Walker and Carayon, 2009).
Health Informatics that Supports New Whole Health Services
Health informatics are needed to support whole health services that extend beyond conventional medical services, such as personal goal setting and care planning tools, wellness programs, health behavior change support, community and social services delivery, educational resources, home-based biometric/telemedicine monitoring systems, and complementary and integrative health services and programs. A key health informatics feature will be to include the interprofessional team members that will deliver these services, but health informatics is also needed to alert and inform clinicians and patients that these services may be of benefit, make these services more accessible, and coordinate the delivery of these services. Existing alert, reminder, and care gap type functionality within health informatics systems can be harnessed for some of these new functions. In addition, patient portals can be entry points for patients to get educational materials and access new services.
The Office of Disease Prevention and Health Promotion of the U.S. Department of Health and Human Services has developed the Healthy People 2030 objectives to set data-driven national objectives to improve health and well-being over the next decade (ODPHP, 2022). A small subset of high-priority objectives, focused on upstream measures such as risk factors and behaviors, were selected to drive action toward improving health and well-being. Information technology was identified as a key driver of change, and the Healthy People 2030 objectives can serve as a roadmap for new needed informatics functions.
An added complexity is that new services also require new data architecture. Standardized coding terminologies serve as universal languages for health informatics, and multiple coding standards are built into health informatics systems, such as SNOMED Clinical Terms, LOINC, ICD-10, CPT, NDC, RxNorm, and others (AHRQ, n.d.; NLM, 2016). These standardized codes address conventional medical elements such as diagnoses, medications, allergies, tests and procedures, and they are accepted as a common global language for health terms in over 50 countries. Similar value sets need to be created and standardized on a national and international level to support new whole health functions (NLM, 2021). Without an agreed-upon standard coding terminology, it will not be possible to
longitudinally track, analyze, or share whole health information across systems and settings.
Health Informatics that Supports Accessible Proactive Care
To support the whole health foundational element of equity and accountability, health informatics needs functionality to make care delivery more accessible and more proactive. The COVID-19 pandemic highlighted health informatics’ potential for making care more accessible with the rapid adoption and implementation of virtual care (Ferguson et al., 2021; Krist et al., 2020a). It is likely that not every community will have all the programs necessary for whole health, and remote access can allow people to get services from their home that would not otherwise be available, extending care beyond the traditional office visit. During the COVID-19 pandemic, VA was able to rapidly transform many of its whole health services, such as the Taking Charge of My Life and Health program, into virtual group sessions. These programs were found to be as effective as the in-person programs and were widely accepted by participants and clinicians (Anderson et al., 2022; Dryden et al., 2021). VA and most health systems’ EHRs and patient portals currently include functionality to support virtual visits, but this functionality will need to be extended to more of the unique whole health services, such as goal setting, yoga, tai chi, or health behavior change classes; include more community and social service providers; and be delivered from more settings, such as community programs.
Access to the internet is a prerequisite for access to virtual care. Unfortunately, 15 percent of veteran households do not have access to an internet connection (FCC, 2019). The Federal Communications Commission reports that 6 percent of the entire U.S. population (approximately 19 million Americans) continue to lack access to fixed broadband service at threshold speeds. In rural areas, nearly one-fourth of the population—14.5 million people—lack access to this service. In tribal areas, nearly one-third of the population lacks access (FCC, 2012). Even in areas where broadband is available, approximately 100 million Americans still do not subscribe. In summary, many older, poor, rural, and other vulnerable populations lack access to high-speed internet and even lack the hardware needed for virtual care (Chiou et al., 2020).
Local governments, community and veterans’ organizations, and industries continue to work in underserved and rural areas to bring affordable broadband, digital skills courses, and computers to people in need. Virtually 100 percent of public libraries in the United States provide free broadband to members of the public, including rural and low-income veterans (Bertot et al., 2006). One VA program provided qualifying veterans with cellular-enabled iPads to access telehealth services. Veterans who received
tablets reported high levels of satisfaction with care, were less likely to miss appointments, and found it easier and more convenient to access VA care (Zulman et al., 2019). These findings have profound implications for a whole health model that seeks to use telehealth touchpoints to serve a patient’s physical, emotional, spiritual, and environmental needs. Many efforts are under way to break down digital literacy and relevance barriers and thereby spurring broadband adoption, but considerable progress must be made before virtual care is accessible to all.
Whole health care includes a large breadth of services provided to entire communities. Effectively delivering these services requires telehealth and remote monitoring for people to access care but also other virtual technologies that have not been well developed or utilized in health care. Examples include but are not limited to virtual group meetings, interactive sessions to support health behavior changes, and novel strategies to deliver high touch complementary and integrative health services.
For health systems and clinicians delivering whole health care, health informatics is needed to both define the community that the system is accountable for and to proactively identify needs. Existing functionality such as registries and electronic alerts that have been shown to improve the delivery of recommended conventional medical care can be applied to new whole health needs (McNeil et al., 2010; Nelson et al., 2016; Shojania et al., 2010), but they will need to be framed and applied differently. For example, registries will need to include not just the individuals seen in the system, but also the people and communities that the system should be accountable for, that are not being seen, and that need care. Similarly, electronic alerts can be reconfigured from conventional medical services, such as the need for colon cancer screening, and applied to whole health services as supports for achieving a patient goal. This will require new triggers and algorithms to identify needs, which will likely be more complex than delivering a simple medical service.
Usability
Given that the use of health informatics applications—EHRs in particular—is the leading factor that clinicians cite as contributing to burnout, it is important to ensure that health informatics applications are easy to use, if not time saving for clinicians, and that they improve the care delivery experience (Muhiyaddin et al., 2022; Robertson et al., 2017). Using human-centered design (HCD) principles can help to ensure the usability of health informatics applications for clinicians and patient-facing health informatics tools as well as promote the whole health concept of team well-being (GSA, 2018; Melles et al., 2021; Zahid et al., 2021). Ensuring that systems are
easy to use and intuitive will promote their uptake and also better engage patients in their care (Vaisson et al., 2021).
HCD is a methodology and a best practice that puts the end user of the provided service at the center of the design efforts. VA currently uses HCD in its digital modernization strategy, and some health informatics vendors have used it in their development processes (VA, 2019). The basic HCD process includes research to genuinely understand user needs and behaviors, design and prototyping of solutions to meet those needs, and iterative testing for usability with representative users (Seftel-Kirk, 2019). HCD should consider a diverse group of users, including veterans, families, caregivers, clinicians, and community partners, and it should also consider social risk factors, race, gender, trust in health care, and comfort with using technology. Machine learning, artificial intelligence, and data science approaches also offer promise for enhancing user experience and patient care by improving information representation and decision-making capacity.
Training interprofessional team members to use health informatics systems will also be essential to ensure that they are using the tools as efficiently and effectively as possible, further reducing burnout and improving team well-being. What can happen when this is not done is exemplified by the experience with an automated texting system to support patient self-management that VA implemented in 2016. Onboarding the texting system was difficult, the staff implemented and used it inconsistently, and there was a lack of clarity on staff roles and responsibilities. While VA thought the new functionality had potential to further engage patients and expand access to care, additional support and training was needed before the system could have the desired effect (Yakovchenko et al., 2021).
Health informatics systems can promote team member well-being and improve care delivery experience by saving time. Possibilities for making health informatics systems time-saving include automating documentation of clinical activities, synthesizing large quantities of data across inputs and over time to assist with, e.g., monitoring patients’ condition, detecting when clinical activities deviate from treatment plans, predicting patients’ clinical course, and assisting with clinical decision making.
WORKFORCE
Successful scaling and spreading of whole health will depend on a workforce that is available, accessible, and supported in its effort to meet the needs of the population it serves. Achieving this, both within and outside of VA, will require strengthening the clinical and allied workforce in both individual and organizational competencies and capacities.
Ensuring that such a workforce receives the necessary training and support to think holistically and care for populations using a whole health
approach will require contributions from the policy side and the health care delivery system. Policy makers will need to expand opportunities for training primary care clinicians, nurse practitioners, physician assistants, social workers, health coaches, integrative health providers, community health workers (CHWs), and other allied health professionals to deliver care through a team-based whole health approach. Provider organizations can build and nurture high-performing interprofessional teams designed to meet the specific needs of the communities that they serve while maintaining an orientation toward whole health of the population rather than health care services rendered. One important aspect of this is ensuring that interprofessional teams represent the populations they serve with respect to cultural characteristics, socioeconomic status, and shared life experiences as such teams will be more likely to exhibit enhanced patient communication, better patient satisfaction, and improved clinical outcomes (Cooper and Powe, 2004).
Training team members to work effectively within a whole health system is a necessary but not sufficient component of scaling and spreading whole health. As described in Chapter 6, team members—clinicians and nonclinicians within and outside health care settings—will need to embrace the paradigm shift represented by whole health. This will require a culture change in the workforce itself as well as innovations in team-based care design by systems leaders. Scaling and spreading whole health will require a willingness to change and the necessary investment of resources. Because of the unique, overarching approach of whole health from the community and societal perspective, the consumers of health care, their families, and their communities should be part of this process.
Human Resources
Although not unique to whole health, health systems of all types and sizes currently face challenges related to hiring, retention, professional shortages, and over- and under-representation of clinicians across race and ethnicity. The COVID-19 pandemic, which has led to clinician burnout and turnover, has exacerbated many of these challenges. VHA employs more than 371,000 employees across nearly 1,300 facilities, making it the largest health system in the United States (VA, 2022c). This is, however, just a fraction of the nation’s 22-million-person health care workforce.
Today, the nation faces a health care workforce shortage (ASPE, 2022a). While the acute need to staff the nation’s hospitals during the pandemic has lessened somewhat since its peak, the size of the health care workforce today remains below expected levels. Shortages in nursing care and elder care remain particularly striking, and outside of hospitals the return of women to the health care labor force has lagged behind that of men (Wager
et al., 2021). Moreover, the mental health workforce is currently meeting only 28 percent of mental health needs nationwide, with wide variability by state (Kaiser Family Foundation, 2021). Projections indicate that the situation in the future many be even more dire. The shortage of low-wage health care workers is predicted to reach three million by 2027, with a shortage of between 200,000 and 450,000 registered nurses predicted by 2025 as well as a shortfall of up to 140,000 physicians by 2033 (Berlin et al., 2022; HHS, 2022). Although the country overall has struggled, VA has maintained consistent growth of its health care workforce throughout the COVID-19 pandemic (Bur, 2020; VA, 2021b, 2022d).
While burnout and its effects on the health care workforce threaten our nation’s ability to scale and spread whole health, whole health has the potential to address burnout. A 2021 health care workforce survey found that 55 percent of the workforce reported feeling burned out and 29 percent reported having considered leaving their profession because of burnout (Kirzinger et al., 2021). A separate survey found that nearly one in five health care workers quit their jobs between February 2020 and October 2021. Of those who stayed, 19 percent said they were considering leaving the health care profession altogether (Galvin, 2021). These pressures on an already fragile health care workforce will be difficult for any health system to manage, whole health or otherwise. However, integration efforts (see Chapter 6), diversifying the workforce, and team-based approaches that engage more professionally diverse teams (see below) can improve delivery efficiency and reduce worker burnout, which has the potential to make a career in health care more attractive and help mitigate some of these strains on the workforce.
Diversity and Equity in the Workforce
It is imperative that any effort to scale and spread whole health be careful to attend to diversity and equity in the whole health workforce. While diversity and equity issues are not unique to whole health, having a diverse workforce that reflects the population that it serves can improve health equity, reduce health care disparities, improve access to care, improve health outcomes, strengthen patient communication, and improve patient satisfaction in underserved communities (COGME, 2016; Cohen et al., 2002; Cooper and Powe, 2004; Muhiyaddin et al., 2022; Poma, 2017; Robertson et al., 2017; Wakefield, 2014). As the Implementing High-Quality Primary Care report states:
Health profession education is a common good, so programs should be expected to supply graduates prepared to care for their immediate and regional communities. To the extent that they fail to do this, they are
failing their public mission. Health disparities are a long-standing, well-recognized problem in the United States, perpetuated by a health care workforce that does not come from, represent, or commit to the population it purports to serve. (NASEM, 2021, p. 201)
In a 2021 study looking at representation across 10 health care professions, Black individuals, Hispanic individuals, and Native Americans were under-represented in all 10 professions compared with the overall population. While the trends indicate that the education pipeline is more diverse than the current workforce and that some professions had better representation than others, the authors concluded that ensuring that the workforce has more equal representation across race and ethnicity for the health care professions will require additional policy supports (Salsberg et al., 2021). A 2017 Health Resources and Services Administration (HRSA) report found similar trends, revealing that white workers represent the majority of all 30 health professions studied and were over-represented in 23 of the 30 professions relative to their representation in the overall U.S. workforce (HRSA, 2017). A report by the Association of American Medical Colleges about Black men in medicine concluded that persistent structural racism and stereotyping that leads to widespread implicit and explicit bias can create exclusionary environments and de facto segregation, limiting the effectiveness of efforts to diversify the workforce pipeline. Many minority medical students who do make it into the medical training pipeline report experiencing racial discrimination, prejudice, and feelings of isolation during training (NASEM, 2021). Students who reported these experiences were more likely to have burnout, depressive symptoms, and lower quality of life than those who did not (Dyrbye et al., 2007).
These racial and ethnic representation issues are symptoms of longstanding systemic racism which is unfortunately woven into the fabric of the United States and is present in all sectors of society. Even with the most supportive and thoughtful policy supports, it could take decades of intentional and persistent efforts to build a workforce that reflects the diversity of populations at the local level throughout the United States. Health care systems, however, can rapidly expand opportunities for some professions, such as CHWs, health coaches, care coordinators, and health educators, all of which are generally more diverse professions, require less training than clinical professions do, and play key roles in the scaling and spreading of whole health (IOM, 2003; Jackson and Gracia, 2014). CHWs, for example, are a diverse reflection of underserved populations: 65 percent are Black or Hispanic, 23 percent are white, 10 percent are American Indian or Alaska Native, and 2 percent are Pacific Islander (Arizona Prevention Research Center, 2015). They primarily serve the local communities they reside in and can carry out a number of roles relevant to whole health, including care coordination, case management, health coaching, health education,
health assessment and screening, resource linking, medication management, remote care, patient follow-up, and social support (HRSA, 2017).
Making recommendations to address these pressures on the U.S. health care workforce is beyond the committee’s charge. However, any effort to scale and spread a whole health system of care will have to work within this reality and consider these factors when building interprofessional teams to implement whole health systems.
Preparation
Training the health care workforce is unlikely to lead to sufficient cultural transformation if the workforce does not offer informed commitment and ongoing support at all levels of the organization or if health care delivery organizations are not committed to act on whole health principles when the initial training is rolled out. This includes readiness to engage the population it serves as well as its own workforce. In addition, the way in which the vast majority of health professionals train today is not well aligned with interprofessional team-based approaches to care. Although most health care professions require some level of interprofessional competency as a graduation requirement (Health Professions Accreditors Collaborative, 2019), most trainees in the United States spend most of their time within their profession—physician trainees train with other physicians, nurses with nurses, social workers with social workers, and so on. This arrangement is counter to the team-based approach of whole health models and other team-based approaches. To prepare a workforce to adequately deliver a whole health approach to care, team members will have to learn how to function and perform in integrated, team-based settings and to integrate, communicate, and coordinate with community-based team members that may not be physically co-located within their employer organization.
There are four major core competencies of interprofessional practice: values and ethics, roles and responsibilities, interprofessional communication, and teams and teamwork (Schmitt et al., 2011). However, it is difficult to incorporate these competencies into classroom instruction, experiential learning, and, ultimately, practice (Goldman et al., 2018). Additional challenges arise in that most clinical settings today are not functioning within a team-based model, particularly as training moves online and to simulated platforms, limiting the availability of interprofessional training opportunities. Often, students receive an introduction to the core competencies of interprofessional training in the classroom but then move on to clinical training settings that are not interprofessional. Lacking reinforcement, the students are unable to truly develop the practical experience of working on teams, collaborating across professions, and engaging with community services (NASEM, 2021).
As the single largest educator of the health care workforce in the United States, VA is well positioned to shift its approach to health care workforce training to focus on interprofessional practice as it concurrently spreads and scales the Whole Health System (WHS) throughout VA. Upward of 120,000 clinical trainees receive at least some of their training at a VA facility every year. Ninety-five percent of medical schools and 100 percent of schools of osteopathic medicine have VA training collaborations. VA also has partnerships with 18 nursing schools. About 1,800 colleges and universities have VA affiliations, including many historically Black colleges and universities as well as many Hispanic-serving institutions. In addition, VA provides training across more than 40 clinical health profession education programs (Sells and McQuaid, 2021). Because so many health professionals train within VA’s walls, it is an ideal setting to fully embrace interprofessional training as the organization concurrently scales and spreads whole health. Doing so should strengthen the interprofessional workforce within and outside of VA.
VA is maximizing its status as the largest health care workforce educator in the country through its learning health system (LHS) training programs. These span VA programs for health services research, quality improvement, implementation, and training. Through LHS, real-world data are continuously transformed into new knowledge which, in turn, generates best practices to address community problems (Kilbourne et al., 2022). As a national health care system answerable to and actively informed by policy-maker, clinical, and consumer priorities, VA is focused on developing and applying advanced methods to address the organization, financing, delivery, and quality of its care. Its emphasis on addressing real-world problems through training in organizational acumen, informatics, implementation, quality improvement, and effective engagement of multiple interested parties offers a powerful driver for WHS principles and practices. LHSs facilitate ongoing input and engagement from interested parties who are brought into a learning community which determines the prioritization, design, and communication of research and its impacts. For example, VA’s Senior Innovation Fellowship augments the ability of established leaders to leverage the efforts of internal and external interested parties throughout VA, other governmental bodies, academia, and industry. Fellows engage in experiential learning in core competencies including communication strategies, storytelling, building and expanding professional networks, developing a business case, and systems thinking which helps them to develop and scale innovative initiatives and practices at the national level. VA’s LHS programs are also designed to reach beyond clinical settings to increase the diversity of the health care workforce and address persistent gaps in health outcomes among populations that have been historically marginalized. Participants without terminal degrees are eligible to participate in some programs,
reducing economic and academic barriers for many. For these reasons, VA’s LHS is a helpful model for the scale and spread of whole health by improving health care research, training, and implementation both within and beyond VA itself.
Experience to date in VA’s scaling and spreading its WHS illustrates the importance of ensuring that, before formal training even begins, clinicians, administrators, and system executives have already developed and tested the tools required to implement whole health practices (Bokhour et al., 2020). Having programmatic, administrative, and clinical champions can facilitate implementation. All VA staff members, including clinicians and other professionals, should be familiar with a whole health approach to caring for the veteran population. This requires developing new courses and supporting materials designed to reorient staff toward delivering whole health care, from clinic- and clinician-centered care to patient-centered and patient-driven care. Local adaptations in training programs, such as whether training occurs all at once or over multiple sessions and at what level of detail, are required to ensure that programs address the training needs of different cohorts. If the staff members are not provided with adequate preparation, a significant lag may ensue between them being trained and their having the ability to act on lessons learned, which may blunt their enthusiasm and decrease their confidence in applying what they have learned.
Supporting Workforce Well-Being
As Chapter 3 described, whole health systems need a healthy, interprofessional, team-based workforce to ensure that they address the five foundational elements of whole health. Toward that end, systems should actively foster conditions to enable individuals—both employees of the system and those who seek their services—to achieve their personal whole health and well-being goals. As Chapters 4 and 5 describe, VA’s WHS, as well as most of the other systems the chapters highlight, have developed employee wellbeing components in their whole health approaches. However, most program components addressing employee well-being are individual-level programs and interventions designed to help employees improve their personal wellbeing through stress management techniques, town hall meetings, and other activities. This emphasis on individual-level intervention and action, while effective to some degree, is not as effective as organizational interventions and approaches to build well-being systems and to prevent employee burnout by addressing the systems-level causes of it (NASEM, 2019; Panagioti et al., 2017). Systems-level causes of workforce burnout (see Chapter 3) include excessive job demands, administrative burden, inefficient workflows, time
pressure and encroachment on personal time, poor usability of health IT, moral distress, and patient factors (NASEM, 2019).
Two systematic reviews looked at both individual-level interventions, such as mindfulness, stress management, and small group discussions, and organizational-level interventions including changes in duty hour requirements and practice-based delivery changes, to better understand their effect on reducing or preventing burnout. One review found that both individual and organizational-level interventions can reduce burnout but did not specify which particular interventions might be most effective for different populations (West et al., 2016). The second review found that while both types of interventions had value, the systems-level interventions have a greater impact on reducing burnout and symptoms of burnout (Panagioti et al., 2017).
Most research on health care workforce well-being examines interventions designed to prevent or reduce burnout, rather than interventions designed to promote individual well-being. The published literature is also fairly unclear regarding which systems-level interventions hold the most promise for reducing burnout or strengthening well-being (NASEM, 2019). Recognizing this, the 2019 National Academies report Taking Action Against Clinician Burnout: A Systems Approach to Professional Well-Being (NASEM, 2019) created guidelines to help health care organizations integrate well-being systems into their overall care delivery system (Box 7-3). The values, systems approach, leadership, and implementation aspects of these guidelines align well with the processes supporting the scale and spread of whole health (see Chapter 6). The evidence in the well-being section of Chapter 3 supports these work system redesign guidelines.
Team Well-Being Lessons from the Pandemic
VA’s roll out of its WHS coincided with the emergence of the COVID-19 pandemic, but rather than slowing implementation, the heightened stress that both patients and health system employees experienced during that crisis actually helped promote whole health. One study found that because patients and staff came to embrace whole health principles and practices as means of a support, they “leaned into [w]hole [h]ealth rather than retreating to former processes and practices” (Dryden et al., 2021, p. 7). Similarly, medical center leadership at multiple levels recognized the value of whole health activities in addressing stress among their employees and actively modeled and encouraged whole health activities in response. For example, facility leaders began to open staff meetings with “mindful moments” aimed at better managing stress and anxiety; daily employee newsletters and emails from leadership regularly included endorsements of whole health principles and practices; and town hall meetings focused
on how whole health was as relevant for health center employees as it was for the patients they served. While there is documented value in these approaches to enhance well-being and to mitigate burnout, and while there was clearly an urgent need to do something as COVID-19 was sweeping rapidly through the health care workforce, systems-level interventions to promote well-being and to manage burnout may be more effective than individual-level interventions (Lebares et al., 2021; Montano et al., 2014).
Nevertheless, these efforts at VA, combined with the universal perception of a pressing need, the availability of high-quality whole health training programs and materials, and the concerted efforts of health coaches, peer-support specialists, clinical champions, mental health professionals, chaplains, employee health workers, and facility leaders helped to advance robust whole health implementation at VA facilities where earlier efforts had stalled. Still more impressive, flagship sites within at least some VA regions took the initiative of exporting whole health principles and practices to their neighbors and networks in response to the burden of the pandemic. One study of VA employees found that those involved in the VA’s WHS were more engaged and less likely to experience burnout than their colleagues (Jonas and Rosenbaum, 2021). Clinical staff working in the WHS were also less likely to resign from VA. As a group, these employees reported that they had better leadership, had higher levels of intrinsic motivation, and felt more engaged in their mission.
Such benefits of whole health are not confined to VA health care systems. The Southcentral Foundation’s Nuka System of Care (see Chapter 4) reported that following its transition to a relationship-based, whole health approach, staff turnover decreased by 15 percent (Eby and Ross, 2016). A 2019 study of 1,298 family physicians in ambulatory outpatient settings found that physicians who perceived that they were able to meet their patients’ social needs were less likely to experience burnout, suggesting that care settings in which clinicians are able to address some upstream factors of health may have a positive effect on clinician burnout (De Marchis et al., 2019).
As concern about burnout among health care workers rises within medical circles and among the public at large as a result of the COVID-19 pandemic (Jalili et al., 2021), whole health transformation may become increasingly attractive across different sectors of society and, in particular, among health care system employees and leaders. As a 2021 study observed, whereas large health system transformation often has negative effects on employees, engagement in VA’s WHS has had a positive effect on employees (Reddy et al., 2021). The study’s authors suggest that this benefit springs from an alignment between the principles of whole health and core values held by health care employees regarding the importance of providing individualized, people-centered care. As experience during the pandemic has demonstrated, benefits may also derive from the uptake of whole health
practices among health care employees in the face of marked workplace stressors. Ironically, the greatest medical crisis of the century may become a powerful driver of whole health scale and spread across the nation.
Policy Considerations
While individual health care delivery systems may choose to transform to whole health, workforce development at the national level will require action at the federal level. More primary care clinicians are needed to anchor whole health systems (NASEM, 2021). Residency training positions are largely funded by CMS, and there have been minimal changes in the number of positions it has funded for the last several decades. When CMS added more positions 10 years ago, primary care physician leadership groups such as the American Academy of Family Physicians requested that CMS allocate the new slots to primary care disciplines and met significant resistance from groups representing subspecialists (Kaufman and Alfero, 2015). The HRSA-supported Teaching Health Center initiative funded by the Affordable Care Act authorized ambulatory organizations to sponsor primary care residencies, but the frequent reauthorizations required by this initiative stand in contrast to the more stable CMS funding for residency training (HRSA, 2022). More recently, VA allocated 1,500 new residency positions devoted to primary care, although at first it mandated that trainees in these positions spend 100 percent of their time in VA space. When it subsequently provided flexibility in how these positions were structured, that allowed VA to recruit more family medicine residents, who have mandatory requirements for pediatric and obstetrical experiences that are not available in VA. These challenges in increasing the capacity to train primary care physicians and other primary care clinicians need to be addressed by legislation at the federal and state levels.
Policy makers may also consider increasing the number of residency training slots in U.S. teaching hospitals for certain physician specialties that are critical for whole health but currently face rather large shortages (AAMC, 2021b). Given recent declines in the number of U.S. medical graduates who choose a career in primary care, geriatrics, infectious disease, and other less-well-paid specialties, investing in training slots would be a long-term strategy for preparing the workforce to be ready to deliver whole health. For example, CMS could fund an increase in primary care or geriatrics positions (Lester et al., 2020; West and Dupras, 2012). Outside of changing the number of training slots, policy makers could also use incentives to encourage U.S. teaching hospitals to train more physicians in needed specialties. Such incentives could take the form of positive or negative financial incentives tied to the percentage or number of physicians in such specialties (Song et al., 2015).
MEASUREMENT, LEARNING, AND ACCOUNTABILITY FOR WHOLE HEALTH
To scale and spread whole health, measuring progress and outcomes will be central to the learning and adaptive process for health care organizations and policy makers. Measurement has long been a core goal of health systems and payers, with several decades of history and empirical study focused on measuring health care quality and the implications of quality measures on clinician behavior and patient outcomes.
The foundation of measurement in health care is rooted in the seminal Donabedian framework (Blumenthal, 1996). Among other key contributions, this framework distinguished structures, processes, and outcomes as distinct targets of measurement (Donabedian, 1980, 1988). Structural measures include counts of necessary inputs for delivering care, such as physical infrastructure, as exemplified in safe and accessible facilities. Process measures—by far the measure that insurers use most commonly—focus on whether clinicians delivered guideline-based services and include appropriate tests at appropriate intervals for chronic disease monitoring (e.g., annual kidney function tests in patients with diabetes) and age-appropriate cancer screening. Outcome measures, in turn, are arguably most important, as they address the outcomes faced by patients. To date, these have included intermediate outcomes such as blood pressure levels in patients with hypertension and cholesterol levels in patients with high cholesterol as well as defined end points such as functional outcomes and mortality. Economic outcomes, including overall per capita and population spending and benefit-to-cost ratios, are also important outcomes to consider in the context of the new spending required to scale and spread whole health. In particular it will be important to demonstrate the benefits that accrue in return for additional spending in order to overcome skepticism about whole health.
Over the past 30 years, numerous quality measurement programs and incentives have been implemented and studied. The mission of the federal Agency for Healthcare Research and Quality (AHRQ) is to produce evidence to make health care safer, higher quality, and more accessible, equitable, and affordable and to work within the U.S. Department of Health and Human Services and with other partners to ensure that the evidence is understood and used. AHRQ receives funding from the Office of the Secretary’s Patient-Centered Outcomes Research Trust Fund to disseminate evidence to practitioners (ASPE, 2022b). Other national bodies such as the National Committee for Quality Assurance and the National Quality Forum have taken leading roles in designing and approving quality measures. A large body of literature is aimed at understanding both the state of performance on quality measures and to what extent financial incentives can improve the quality of care. In general, the quality of care delivered in
the United States has been suboptimal, with adults and children receiving only about 50 percent of recommended care (Mangione-Smith et al., 2007; McGlynn et al., 2003). Efforts by insurers to improve quality through financial incentives have led to some improvements in performance on quality measures, but have also introduced important unintended consequences (Berwick, 1989; Rosenbaum, 2022b). Among the latter have been measures of fatigue among clinicians, the nearsighted focus on quality measure performance at the cost of intrinsic motivation and unmeasured dimensions of quality, and the exacerbation of racial or ethnic inequities through pay-for-performance incentives (Rosenbaum, 2022a).
Learning and accountability are different and potentially unaligned processes; yet scaling and spreading of whole health require both. Learning systems—those that provide data comparing the efforts and outcomes of peers over time—help providers, teams, organizations, and systems understand how to move from whole health aspirations to effective operations at mico, meso, and macro levels. Accountability systems—those that assess the adequacy and completeness of whole health activities—can motivate providers and systems to embrace and achieve whole health. They can also promote communication and cooperation among stakeholders (Berwick and Shine, 2020), enabling local adaptation to recommended approaches, the evaluation of scale and spread over time, and adaptation in response to changing conditions. Accountability systems complement but do not replace professional norms and values that lead to high-quality outcomes for patients (Kanter et al., 2013; Starr, 2017).
Learning and accountability may sometimes be at odds, as when widespread under-reporting (necessary for learning) confounds efforts to measure human error (for purposes of accountability) (Edmondson, 1996). For providers to be willing to report errors from which a team or organization may learn, they must believe that the group is safe for taking interpersonal risk and that reporting will lead to change (Edmondson, 2019). Attempting to use such measures for accountability risks undermining providers’ sense of psychological safety, which results in under-reporting. In addition, because learning processes take time, they often cause delays and short-term performance decrements (Repenning and Sterman, 2002). For this reason, the costs of learning (e.g., error identification) may be initially more visible than its benefits (Singer and Edmondson, 2008). Holding providers accountable for required learning that is not yet completed can therefore be unhelpful. Unintended consequences of accountability tied to incentives can include motivating inappropriate levels of effort toward achieving rewarded metrics while ignoring other desired outcomes for tasks that may be more difficult to measure (Holmstrom and Milgrom, 1991) and turning quality measurement into a box-checking exercise that promotes burnout instead of motivating improvement (Berenson, 2016; McWilliams, 2020; NASEM,
2019, 2021; Phillips, 2020; Philips et al., 2019). For these reasons, the process of identifying and developing metrics and measurement systems to assess progress toward whole health should be pursued with sensitivity to the differences between learning and accountability goals.
While specifying a standard set of learning and accountability metrics for whole health is beyond the scope of this report, the committee emphasizes that identifying such measures should occur through a coordinated process involving key stakeholders, including community members. Such efforts should seek to identify existing measures that reflect whole health foundational elements as well as important gaps that may require new measures for advancing whole health. Public and private systems fielding a whole health approach need to evaluate how to implement and adapt whole health care and its outcomes, which will require partnerships with academic health researchers. Learning from this kind of evaluation is needed to inform continual adaptation and improvement, and this learning should be disseminated so it may inform others who are also implementing or considering implementing a whole health approach.
A set of core principles should drive the process of defining effective metrics for whole health. However, most existing health care quality metrics focus on specific components of health care, such as hemoglobin A1c levels in diabetes control, and thus are poorly suited to measuring whole health systems, which emphasize the integration of multiple components of care; many of these components exist beyond what is traditionally considered the domain of medical practice or responsibility (NASEM, 2021; Stange et al., 2014).
The principles that the committee recommends for promoting the scale and spread of whole health systems and improved performance on dimensions of whole health are largely consistent with those that the 2021 National Academies report Implementing High-Quality Primary Care: Rebuilding the Foundation of Health Care describes. Modifying the criteria (using brackets to identify differences) established by the Starfield Summit for superior primary care measures (Etz et al., 2017) and reported in the Implementing High-Quality Primary Care report, the committee suggests that superior whole health measures
- Are meaningful—to patients, families, [communities], health systems, policy makers, and clinicians.
- Assess [whole health] as defined, practiced, experienced, and cocreated between patients, [families, communities,] clinicians, and teams.
- Assess the intended outcomes of [whole health] (e.g., achievement of health and health goals, illness prevention and health promotion,
- healing, avoidance of unnecessary pain and suffering, equity, [and overall health spending]).
- Balance the tensions endemic to [whole] health care: standardization alongside customization, predictability alongside ambiguity.
- Are flexible—adaptive to setting (from the individual to national levels), lifespan (infant to elderly), health state (changing health status), and individual differences (context, family, and preferences).
- Provide evaluation and improvement information actionable at the local, regional, and national levels.
- Support self-assessment, self-learning, and aspiration.
- Are feasible, reliable, and without undo data collection burden.
- Point out and establish the importance of things that cannot yet be counted.
- Inform evaluation of a broad vision that understands health and illness exist within a social and cultural framework.
- Reflect the complexity of the [system]—the whole is more than an additive sum of parts. Embrace interconnectivity, reject reduction to cause and effect of individual elements, assess and support emergence—where just adding up what happens to parts (diseases, individuals) does not equal the whole (people, populations).
In addition, effective measurement systems should
- Account for people-centered needs, not just patient-centered needs. Beyond readily available measures of disease control and life expectancy, whole health measures should address how individuals live and evaluate their own life experiences. This will likely require greater use of a broader range of patient-reported assessments as well as the inclusion of new measures (e.g., of well-being, happiness, and/or flourishing) not often addressed in clinical settings.
- Distinguish measures valuable for learning from those intended for accountability. As explained, comingling measurement objectives will undermine both learning and accountability. Instead, these must be distinguished. To encourage accurate reporting, learning-oriented measures must protect reporters from potential punitive repercussions and should be accompanied by organizational capacity for taking action based on results. Safety climate is an example of a learning-oriented measure. What’s important is that a team or organization receives information that allows improvement. If used for accountability, such a measure could be easily gamed and its value for improvement would be lost.
- Address macro, meso, and micro levels and balance individual, family, and team-based, and community accountability for whole
- health. Achieving whole health requires cooperation and integration across multiple levels, including individuals’ primary providers, care teams, whole health delivery systems, payers, and community resources. Measurement for accountability should therefore address multiple levels, for example, through report cards comparing individual, team, and organizational performance against peers. While delivering whole health requires collaboration and integration across teams, team- and community-level accountability do not diffuse individual accountability, including that of individuals’ primary providers, who are ultimately accountable for patients’ care.
- Include few measures. Parsimony is a critical goal of effective and efficient measurement. Frugal, well-focused measures increase focus, reduce burden and burnout, and promote alignment across payers, patients, clinicians, health systems, and communities.
- Where possible, draw on measures that have been previously developed and tracked, which relate to the foundational elements of whole health, either directly or indirectly.
- Be flexible in reflecting people and populations’ needs and goals, keeping individual, community, and cultural values in mind.
- Anticipate, whenever possible, potential unintended, unproductive consequences of proposed metrics (given that systems tend to get the results that they measure for).
- Encompass both clinical and social outcomes such as homelessness, occupational, financial or nutritional insecurity, and level of family stability and social support.
- Balance objective and subjective outcome measures when available, including validated process and patient experience measures.
- Be easily understood by the target audience, and consistent over time.
Scaling and spreading whole health will require new measures that better reflect the five foundational elements of whole health, but these measures may need to be developed over time within iterative quality improvement and translational science processes. Consideration should be given to structural and processual measures, such as team-based care and care integration; cognitive measures, such as trust (Lynch, 2020); measures of the quality of the therapeutic relationship (Greenhalgh and Heath, 2010); and measures of well-being (Vogt et al., 2019), in addition to clinical outcomes. Equity is also an important goal of whole health, for which measures should be developed, as should measures appropriate for specific subpopulations.
Evaluations should be prospective and longitudinal, use a mix of methods, include information on how to achieve whole health, and not be overly
burdensome on clinicians or people receiving care. Because whole health centers around people and their communities, the committee anticipates that illuminating progress toward certain foundational elements will require population and member surveys as well as focused community assessments, such as the American Census Bureau’s American Community Assessment (NRC, 2007). Findings should be shared openly and transparently using narratives and numbers, with details to assess the strengths, limitations, and potential biases so that all can learn and adapt approaches based on the results. These evaluations should focus on learning and be separate and distinct from evaluations used to ensure accountability and quality whole health care.
Action to scale and spread whole health based on new measures of learning and accountability will be required at both organizational and systemic levels. Systemic action requires policy change, especially to ensure that action promotes health equity. Given their expertise, clinicians can be effective advocates for change at both organizational and systemic levels, particularly when they combine empirical evidence of outcomes based on validated measures with compelling stories of the difference whole health makes in the lives of individuals.
FINANCING AND PAYMENT FOR WHOLE HEALTH
The concept of whole health extends beyond health care and the institutions that deliver health care. As Chapter 3 details, the holistic health and well-being of individuals and communities often depend more on upstream factors outside of the health care system than those within it (Bradley et al., 2013, 2016; Willard-Grace et al., 2014). Nevertheless, the health care system itself—including not only the human capital necessary for delivering care but also the physical and now increasingly virtual structures of care delivery—retains an important role in determining whether and how a vision of whole health can be achieved. Notably, this role is shaped by the policies and markets that govern or are embedded within the health care system.
In the context of a health care system, how care is financed and paid for goes a long way toward determining how patients and communities access care and what services they ultimately receive (Chandra et al., 2011; Cutler, 2005; Newhouse, 2002). Not only do professional ethos and cultures of medical practice matter, economic incentives and regulatory structures also matter, often even more so. Financing and payment for care are related, but they are not the same. Financing refers to the way that society raises the funds to pay for health care and is often linked with health insurance. From a public perspective, financing occurs through key mechanisms such as taxation, general revenues, and deficit spending or borrowing. These
mechanisms largely fund public programs including Medicare, Medicaid, the Indian Health Service, Tricare, and the Veterans Health Administration (VHA). In the private sphere, the financing of health care comes from business revenues and the insurance premiums that employees pay, which largely come out of their wages (Baicker and Chandra, 2006; Clemens and Cutler, 2014). The private financing of health care also includes philanthropy, a substantial source of revenue for many health care institutions, and extends to the personal and family resources of individual patients (AAMC, 2021a). Although philanthropic spending accounts for only about 7 percent of overall health spending (Shaw-Taylor, 2016), the historical examples of the Rockefeller Foundation’s support for the National Committee for Mental Hygiene and the Carnegie Foundation’s support for the Flexner Report (as described in Chapter 6) demonstrate that strategic philanthropy can exert a powerful influence on the trajectory of health care concepts and programs. For those with health insurance, public and private financing mechanisms typically establish the pool of resources that makes insurance coverage possible. In turn, insurance protects people from the financial risk of needing health care (Cutler and Zeckhauser, 2000), although it is not free for individuals or for society. Whether one considers a universal, single-payer model of providing insurance or a decentralized, multipayer scenario like in the United States, the fundamental fact that health care needs are, to a significant degree, difficult to predict renders some system of health insurance necessary.
Payment refers to the mechanisms that reimburse (or sometimes prospectively pay) health care providers—including clinicians, facilities, hospitals, post–acute care providers, and other entities—for delivering health care services. Payment models include reimbursement for each service rendered according to a fee (or price) schedule, often known as “fee-for-service”; bundled or episode-based payments determined prospectively by a target or budget for a defined set of services over a period of time; and payment determined prospectively for all services across the spectrum of care for a defined population over a period of time. The latter approach of prospective payment is often referred to as global payment, global budget, capitation, risk contracts, or accountable care organization contract models. Within health care organizations, whether paid through fee-for-service or prospective payment, individual clinicians can receive payment via salary (a form of prospective payment) or volume-based “productivity” incentives (a form of fee-for-service), although many salary models are mixed with productivity incentives based on the number of services rendered. Outside of these main payment mechanisms, health care providers may also receive payments for quality of care, sometimes called pay-for-performance or performance-based bonuses. Years of evidence have demonstrated that each type of
payment incentive affects clinician behavior with intended and unintended implications for health care use, spending, and patient outcomes.
The following section discusses the financing of care in VA and evidence pertaining to various payment models that may be appropriate for whole health both within VA and scaled to environments beyond VA. In response to the statement of task, the committee also describes payment and financing models used in the private sector that could facilitate value-based, whole person care within a population health orientation beyond VA.
Financing and Payment in the VA
VA delivers care through its VHA at 172 medical centers and more than 1,100 outpatient clinics. Federal budgetary appropriations finance this integrated delivery system, which comprises 18 regional Veterans Integrated Service Networks (VISNs) which contain medical centers. Congress makes these appropriations through four major categories or “accounts:” (1) Medical Services, which covers health care services; (2) Medical Community Care, which comprises services provided by community providers; (3) Medical Support and Compliance, which covers administration and research activities; and (4) Medical Facilities, which addresses the operation and maintenance of physical infrastructure. From fiscal year 2015 to 2019, federal appropriations to the VHA increased from $56 billion to $81 billion a year, financing care for roughly 7 million veterans (GAO, 2019a).
The statement of task asked the committee to examine how the Veterans Equitable Resource Allocation (VERA) system could facilitate the transformation to whole health within VA and inform models in the private sector. The federal government appropriates a budget to the VHA every 2 years. In turn, the VHA uses the VERA system to allocate about two-thirds of its funding ($49 billion in 2019) to VISNs and medical centers for general patient care—called “general purpose funds”—based on patient workload (GAO, 2002). VHA allocates the remaining third of its funds ($23 billion in 2019) to specific program offices—termed “specific purpose funds”—through the Medical Center Allocation System (MCAS). Such programs include those focused on prosthetics, hepatitis C drugs, homelessness, medical residency education, and community care. These program offices, in turn, pass on the funds to medical centers based on their own financing models, which could also be workload based.
The largest share of specific purpose funds (46 percent) goes to programs for community care, which through the MISSION Act (see Chapter 6) enable veterans to receive care outside of VA when care would be challenging to receive because of wait times, long travel distances, or the unavailability of a service inside VA. While the rules of the MISSION Act
govern the current iteration of community care, it represents a longstanding mechanism for the VA to purchase care from providers outside of VA, which it has done since the 1940s. In recent years, however, spending on community care has steadily grown, prompting VA to request additional appropriations—$1.2 billion and $2.2 billion in fiscal years 2017 and 2018, respectively—to pay for that care (GAO, 2019b).
VA aims to titrate financing based on measures of need as reflected through clinical use. The VERA and MCAS allocation systems differ in how they adjust financing to patient workload. The VERA model considers the number of veterans served, the types of veterans served as defined based on use patterns, the complexity of services, and geographic factors such as local labor costs. The MCAS model uses a workload measure called “patient-weighted work” which is derived from patient volume, case mix, and specialized services. Both models adjust funding based on usage, the complexity of services, and patient characteristics. In essence, this reflects a form of risk adjustment, whereby medical centers that demonstrate plausibly greater need could receive additional funding. Throughout the year, VA monitors the status of funds, and it has the authority to redistribute funds toward needs that arise.
Payment in VA
Once funding has been appropriated and allocated, VA hospitals are essentially paid based on a budget, and clinicians are generally salaried. In this way the VA’s hospitals and clinicians are paid in a more prospective manner than are most community providers in the broader health care system today, who remain paid through a largely fee-for-service system that incentivizes volume. By paying its hospitals according to a budget and its clinicians via salary, VA has a basic foundational payment infrastructure that encourages its delivery system to care for patients holistically and that emphasizes prevention, as additional use does not immediately garner additional revenue.
Paying hospitals and clinicians in a more budgeted or prospective manner aligns with the philosophy behind value-based payment models that emphasize prevention, primary care, and reducing low-value care. Fundamentally, if VA could avoid morbidity and expensive care, it would have a better chance to stay within its federally allocated budget. This would ease the pressure on the federal budget and help relieve some of the fiscal pressure generated by the growth in health care spending of recent decades. Paying for the health care of defined populations prospectively also resonates with the ethos of whole health, which emphasizes attention to upstream risk factors and opportunities to affect health, including addressing the social determinants of health. For example, within a budget, health systems
would be better able to prospectively invest in housing, transportation, or social services to keep patients healthy and maintain spending under the budget. In such sense, VA is an ideal fertile ground for the development and advancement of a whole health approach to caring for populations.
Within the structure of an overall budget, however, volume incentives are not absent in VA. This is because the volume of care and complexity of care are key inputs into the workload adjustment used to determine future funding allocations in the VERA and MCAS systems. Therefore, increases in volume in one year could lead to a larger budget allocation in future years. In this way, a meaningful volume incentive still exists, even though it is a less direct and less immediate incentive than traditional fee-for-service. Moreover, if a medical center spends less than its allocated budget, VA could redistribute its funds to other needs that arise, creating an incentive for every VA medical center to use the full extent of funding it receives.
Financing and Payment Outside of VA
Most health care services delivered outside of VA are reimbursed on a fee-for-service basis. Fee-for-service arrangements reimburse clinicians and health care professionals at a negotiated price or an administratively set price per unit of health care service rendered. At its core, fee-for-service rewards volume of care, rather than explicitly paying for quality, appropriateness, or outcomes (De Brantes et al., 2009; Rosenthal et al., 2004). Moreover, fee-for-service payment is likely a key contributor to the fragmentation of the U.S. health care delivery system, as it does not directly reward patient co-management or coordination of care among clinicians (Ginsburg, 2012; Laugesen and Glied, 2011). As a result, fee-for-service rewards the development and proliferation of technologically intensive and higher-priced services, which are often in the domain of procedural specialties, rather than encouraging clinicians and other health care professionals to actively prioritize whole health. For example, there is no defined fee for instilling empathy into a relationship, building community for a patient, or preventing an illness from occurring. There is a fee, however, for treating the illness once it has emerged.
In recent years, both private and public insurers, including Medicare and Medicaid, have begun to move payment arrangements away from fee-for-service and toward “prospective” or “alternative payment models” that provide clinicians and health care delivery organizations a spending target or budget for a set of services or the care of an attributed population of patients (Burwell, 2015; Emanuel et al., 2012; Rajkumar et al., 2014). The Center for Medicare and Medicaid Innovation has tested about 50 alternative payment models in its first decade (Smith, 2021); meanwhile, states, including California, Massachusetts, Oregon, and Washington, have started
to use 1115 Waiver authority to innovate away from pure fee-for-service payment.
Such alternative payment models include bundled and episode-based payment models that give clinicians and hospitals a prospectively estimated target or budget for a defined episode of care, such as 30 days after a joint replacement or 6 months after the onset of chemotherapy (CMS, 2022a,b; Press et al., 2016; Song and Colla, 2016). Alternative payment models also include global budget or accountable care organization (ACO) contracts which provide health care delivery organizations a prospective, risk-adjusted spending target or budget for the entire continuum of care of a defined population of patients (Pham et al., 2015; Rajkumar et al., 2015; Song and Lee, 2013). In these models, provider organizations including physicians, hospitals, and other providers, such as post–acute care providers, have financial accountability for the quality of care and spending within a prospectively determined spending target or budget. By 2021, about 40 percent of U.S. health care spending flowed through such alternative payment models (MITRE Corporation, 2021; Health Care Payment Learning & Action Network, 2022).
Spending Target (or Budget) vs. Accountability
Central to the whole health approach is a health care system that takes accountability for the health outcomes of its population and how scarce resources are allocated to achieve those health outcomes. Payment via a prospectively determined target or budget is a necessary, but not sufficient, condition for this accountability.
Accountability for quality of care is generally established through quality measures, with financial incentives tied to performance on those measures. While imperfect and sometimes with notable unintended consequences on provider behavior (Rosenbaum, 2022a,b), quality measures generally seek to capture how well clinicians and hospitals perform on process measures (e.g., appropriate intervals of testing for patients with chronic diseases), outcome measures (e.g., intermediate health outcomes such as blood pressure, cholesterol, or hemoglobin A1c levels), and patient experience.
Accountability for spending occurs through providers bearing financial risk for spending, which typically occurs in two ways. First, “upside risk,” a type of financial carrot, rewards providers with a share of the savings if actual spending on health care in a contractual period is less than the prospectively set budget or spending target. Second, “downside risk”—a financial stick—penalizes providers by omitting payment (or a percentage of payment) for any spending that exceeds the budget or spending target.
In the extreme, this downside risk can be “full” risk, where providers are not paid for any services billed for in excess of the budget. In most cases, however, the risk is shared between the payer and providers, whereby the payer reimburses the provider for a portion of excess spending (referred to as “shared risk”). A payment model based on a spending target or budget with only upside risk is often denoted a “one-sided” alternative payment model, whereas a model with both upside and downside risk is often dubbed a “two-sided” alternative payment model. Figure 7-1 provides an illustration of the spending target and implications of one-sided and two-sided financial incentives for providers.
Moreover, these financial incentives can be linked. For example, the magnitude of shared savings or shared risk faced by the provider organization could be conditioned upon a certain level of performance on quality measures. In such a way, better quality could earn the provider a larger share of any savings, should there be savings, and a smaller share of any risk, should there be excess spending. Accountability could also be reflected through non-financial incentives to deliver higher-value care, including clinicians’ intrinsic sense of professionalism, peer pressure, or organizational leadership or culture (McWilliams, 2020). These non-financial incentives could also move clinical decision making toward higher-value care and may be more effective than financial incentives in certain situations.
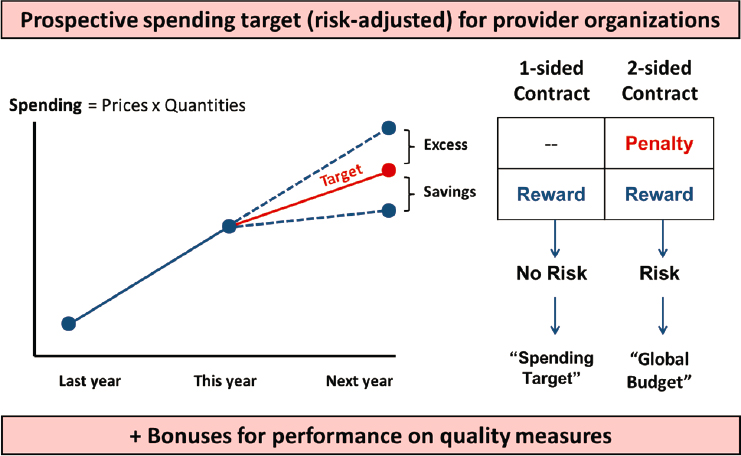
SOURCE: Song, 2014.
Recent Evidence from Traditional Medicare
To date, the Center for Medicare & Medicaid Innovation (CMMI) has designed and tested over 50 payment and delivery models intended to improve the value of care—an effort directionally aligned and substantively consistent with the philosophy of whole health. To date, nearly all of these models have been voluntary for providers to consider and join. In the first decade, about a handful of these CMMI models have generated what appears to be net savings in health care spending (McWilliams et al., 2019; Smith, 2021). Net savings can be understood as savings generated for the payer or society after accounting for incentive payments to providers, which include the shared savings, quality-based performance bonuses, and other non-claims-based or per-member-per-month payments to providers to help them adapt to the new payment model (e.g., lump-sum care management fees or investments to help providers establish electronic health records). In Medicare, where the prices of care are generally similar across the country, changes in volume or use explain the savings. Evidence to date suggests that in many Medicare alternative payment models, savings have come out of post–acute care (Barnett et al., 2019a; McWilliams et al., 2013, 2017). Some of these Medicare models appear to improve the quality of care without savings.
The Medicare Shared Savings Program, the largest Medicare ACO program and one that uses a mostly one-sided incentive model, has demonstrated modest, though meaningful savings to date (McWilliams et al., 2018, 2020; Trombley et al., 2019). The Medicare Pioneer ACO program, which used a two-sided model, also demonstrated modest savings in the initial years, although participation waned due to the financial consequences of downside risk (McWilliams et al., 2015; Nyweide et al., 2015; Trombley et al., 2022). In these ACO models, provider organizations are attributed to a defined population of Medicare beneficiaries for whom it is accountable for total spending and quality. This attribution can either take place prospectively—before a contract year, whereby an organization knows the roster of covered lives it is responsible for—or retrospectively based on where beneficiaries received most of their primary care in a given year. These ACO models provide an example for the VA context, in which additional accountability for whole health may be integrated into the budgets allocated to the medical centers.
Other examples for the VA context include federal payment models that were similarly designed to improve the quality of care, slow spending, or both. In primary care, the federal Comprehensive Primary Care initiative featured lump-sum payments to primary care practices that were designed to support care management. In the first few years, the model was associated with fewer emergency department visits. However, the monthly
payments to primary care practices exceeded savings on emergency department visits, and no net savings were evident (Peikes et al., 2018). In fact, during years 1, 2, and 3, the model was associated with reductions of $16, $10, and $2 per beneficiary per month, respectively; however, the $16 care management fee, on average, exceeded those savings on claims (Peikes et al., 2016). The more recent Comprehensive Primary Care Plus model, which began in 2017 and also provided lump-sum payments to practices to support care management, similarly did not reduce net Medicare spending after accounting for the care management fees, although it was connected to improvement on some quality measures (Swankoski at al., 2022).
Outside of primary care, Medicare’s largest mandatory bundled payment model, the Comprehensive Care for Joint Replacement Model for hip and knee replacements, demonstrated modest savings on claims in the first 2 years which were driven by changes in post–acute care, but those savings were largely offset by incentive payments (shared savings and quality bonuses) paid to participating providers (Barnett et al., 2019b; Finkelstein et al., 2018; Haas et al., 2019). Medicare’s Oncology Care Model, an episode-based model that gives providers a prospective spending target for 180 days of care after starting chemotherapy, was associated with some reductions in spending on claims in the first 3 years, which were offset by its monthly payments for enhanced oncology services, resulting in no net savings; there were no associated changes in the quality of care or patient experience (Brooks et al., 2019; Keating et al., 2021, 2022). The Maryland All-Payer model, a unique state-based global budget model for hospitals, also did not demonstrate clear evidence of changes in use or savings in the first 3 years attributable to the payment model (Roberts et al., 2018a,c), although some debate remains over its effect (Emanuel et al., 2022; Roberts et al., 2018b; Sharfstein et al., 2018).
Recent Evidence from Medicare Advantage
The Medicare program features another type of prospective financing, Medicare Advantage, which now enrolls about half of the Medicare population (Freed et al., 2022). Commercial insurers issue Medicare Advantage plans, and these plans accept a prospective, risk-adjusted payment from the federal government to offer the Medicare insurance benefit in U.S. counties of their choosing. The plans can modify the base Medicare benefit within regulatorily defined bounds, based on how much in government subsidies it needs. By requiring fewer subsidies than those that CMS is willing to offer, a plan can add additional benefits—for example, vision, dental, or hearing coverage, or lower premiums for Part B or prescription drug coverage—to the base Medicare benefit, which may appeal to Medicare beneficiaries. To control spending, plans can restrict enrollees to a narrow network of
providers, in contrast to traditional Medicare. Plans can also use managed care strategies such as prior authorization and usage review. Beneficiaries can choose to enroll in traditional Medicare or a Medicare Advantage plan, based on the plans available in their county of residence.
By accepting a prospective payment, Medicare Advantage plans have a strong financial incentive to adopt a whole health philosophy in their population health management. Every unnecessary medical service averted is potential savings in the budget, and evidence suggests that Medicare Advantage plans indeed contain service use more effectively than traditional Medicare (Duggan et al., 2018; Schwartz et al., 2021), in part through their use of narrow provider networks and managed care strategies such as prior authorization. Studies have also found that Medicare Advantage beneficiaries receive more recommended preventive services and report higher scores for some patient experience measures than traditional Medicare beneficiaries (Meyers et al., 2022). However, whether Medicare Advantage plans deliver better value overall for taxpayers and the country relative to traditional Medicare remains less clear. Medicare Advantage patients often receive care from lower-quality providers, including hospitals and nursing homes, compared to traditional Medicare patients (Meyers et al., 2018). Quality of care in certainly clinical situations have been modestly better in Medicare Advantage than traditional Medicare (Landon et al., 2022), although non-random selection by Medicare beneficiaries into Medicare Advantage plans have complicated some of these comparisons to traditional Medicare (Agarwal et al., 2021). However, what is more clear is that Medicare Advantage plans often attain generous additional federal subsidies through intensive coding of clinical diagnoses, which has led to substantial overpayments through risk adjustment (Jacobs and Kronick, 2021; Kronick, 2017). At present, definitive evidence on whether a whole health approach is better suited for private insurers administering a public benefit relative to public insurers administering the benefit remains elusive.
Despite this lack of clarity in the evidence, physician groups—notably primary care physicians—have increasingly entered the Medicare Advantage market (Song et al., 2022). Some have chosen to bear the financial risk of the prospective payment from CMS—by purchasing that risk from the insurer—enabling physicians themselves to keep the savings if care is managed effectively within the budget. Others have chosen to join or form larger cooperatives of practices that receive support from a private convening company, which supports the member practices and guides them toward savings under the budget through population health management, coding support, utilization management, or other strategies aimed at high- and low-value services. Still other physician groups have become Medicare Advantage plans themselves, mirroring the trend of larger health systems launching their own Medicare Advantage plans. Behind this evolution is
the idea that a prospective, risk-adjusted budget creates the incentives for managing population health rather than maximizing volume. At the same time, the strong financial incentive to retain any savings generated under a risk-adjusted budget that is susceptible to coding intensity must also be recognized as a salient motivation (Gilfillan and Berwick, 2021; Ginsburg and Lieberman, 2022). The risk of inappropriate rationing of care, if budgets are not sufficient to support necessary services or if the focus on generating savings under the budget is inappropriate, also remains a concern (GAO, 1996).
Recent Evidence from Commercial Insurers
Analogous alternative payment models, notably ACO contracts, have also proliferated between commercial insurers and provider organizations. One of the largest examples is the Blue Cross Blue Shield of Massachusetts ACO model called the Alternative Quality Contract (AQC), implemented in 2009. In the commercial insurance context, savings could arise from reductions in use, as in Medicare, or reductions in prices given the differences in the prices of care commanded by different providers resulting from their market power relative to commercial insurers. This latter reality affords providers in the commercial context an alternative avenue for pursuing savings—referring patients to lower-priced providers or settings. In theory, this ability to shift referrals, an important lever in the pursuit of higher-value care (Song et al., 2014b), could encourage price competition among providers (especially specialists) in a given market, as organizations in the AQC had an incentive to reconsider their patient referral patterns.
Evaluations of this model revealed modest savings on claims in the first 2 years which were driven by changes in referral patterns—a price effect rather than a usage effect—although these savings were similarly offset by incentive payments (shared savings and quality bonuses) to the provider organizations. However, by year 4 and through year 8, net savings emerged after accounting for incentive payments; these savings began to be increasingly explained by lower use in areas such as high-cost drugs, emergency department visits, imaging, and tests. This was accompanied by improvements in some quality measures (Song et al., 2014a, 2019). Evaluations of other commercial ACO contracts have also demonstrated some meaningful changes in provider behavior (Zhang et al., 2019, 2021). To date, rigorous evidence on the effects of alternative payment models on patient experience is lacking (Graham et al., 2021).
Paying for Whole Health
Consistent with the vision of whole health, alternative payment models have been developed with the goal of improving quality and efficiency in
health care delivery while moving clinicians toward higher-value decisions and less wasteful care. With the reality that health care spending society-wide is one of the most important drivers of persistent budget deficits and national debt, much promise has been put on payment reform. Slowing the growth of health care spending, as is the hope by public and private payers, will enable society to invest in other valuable services, both health and non-health related. Taken together, evidence to date suggests that payment reform models have likely led to modest savings, including some evidence of net savings after incentive payouts to providers after the first few years, along with some improvements in the observable quality of care as measured by performance on mostly claims-based process measures. The general dearth of net savings from alternative payment model programs to date could be explained by the following.
Providers generated savings on claims that were fully offset by incentive payments, including shared savings, quality bonuses, or up-front payments (Peikes et al., 2018). Inflated spending benchmarks (or spending targets), implementation challenges, and voluntary provider participation have also contributed (McWilliams et al., 2019). Some models may not have changed provider behavior enough to reduce usage, likely because programs affect a small portion of the provider group’s patient panel.
Despite the modest savings, however, the evidence on payment reform to date suggests that financial incentives can be effective in modifying clinician or provider organization behavior toward an orientation of improved efficiency and quality as reflected through available quality measures. Even though the estimated effects have been modest, from a societal view a more prospectively designed or value-based approach toward provider payment appears to be clearly preferable to than pure fee-for-service. A decade into payment reform, health systems are still learning to adapt to the evolving payment landscape. Moreover, even as improvements in the quality of care in such models to date have focused on process measures—with less evidence on improvements in patient experience or hard outcome end points—there has generally been no evidence of decreases in quality or stinting on high-value or necessary care. Therefore, financial incentives for quality within the context of payment reform appear not to have negatively affected quality.
Public and private payers hope that as payment reform eventually slows the growth of health care spending while improving the quality of care, more of society’s resources will be liberated for other needs—from housing and food to wages and social services that are important for health. Indeed, provider organizations and some payers, including Medicare Advantage plans, that operate under a global budget model have increasingly contemplated or sought to invest in such social services. For example, some
hospitals have invested directly in housing to support homeless populations in their catchment areas. Some Medicare Advantage organizations have invested in transportation as a social determinant of health (Powers et al., 2016). Such activities resonate with the vision of whole health.
Despite some encouraging evidence and examples to date, much work remains to design and implement payment models that best support whole health. Global budget and other prospective payment models can be improved from a contract design perspective in several ways. First, on the dimension of quality of care, such models could measure and track observable whole health activity, as laid out in the earlier section on Measurement, Learning, and Accountability for Whole Health. Second, a payment model that supports whole health could prioritize financial incentives toward such measures that are most related to whole health, either in addition to or in place of traditional process measures. Given that payments for quality measures offset any savings on claims generated through changes in clinician behavior, the financing available for quality, from a fiscal perspective, is naturally constrained. Moreover, evidence suggests that payments for quality measures (an extrinsic incentive) may suppress more important internal motivation (intrinsic incentives) of health care providers to improve quality and patient outcomes (McWilliams, 2020). For both of these reasons and as described in the Measurement, Learning, and Accountability for Whole Health subsection above, the design and implementation of quality measures require careful thought and judgment of tradeoffs.
Third, from the financial risk perspective, prospective payment models that support whole health could enable provider entities within VA, such as hospitals and clinics, to assume more accountability for quality and spending that veterans incur within their federally allocated budget. This accountability could include incentives for providers to retain savings under the budget. To reduce the risk of shortfalls in the quality of care, a larger proportion of shared savings could be earned by the hospital or clinic conditional on a certain level of quality performance (a carrot), or else a larger proportion of shared risk could be borne by the providers should quality performance be lower than desired (a stick).
Fourth, a prospective payment model could build in larger incentives for the patient. For example, rather than providers keeping all savings under the budget or quality bonuses, a model could require that patients receive a share of the savings or quality bonuses. After all, patients’ own health behaviors may be crucial for reducing unnecessary usage, such as via adherence to chronic disease medications, and for improving performance on quality measures, such as by obtaining guideline-based preventive care. By sharing savings with their patients, hospitals and clinics may also better align their financial incentives with patient health behaviors (improved diet and exercise, for example). This patient share could be placed in a health
savings account, which the patient could draw on for out-of-pocket costs or other health-related services, such as yoga, food, or even housing, within the framework of whole health.
FINDINGS AND CONCLUSIONS
Following the previous chapter’s description of contextual conditions necessary for scaling and spreading whole health, this chapter described four types of foundational infrastructure, critical for creating and sustaining those conditions. The four elements of foundational infrastructure are (1) supportive health informatics, (2) workforce training and education, (3) the measurement of whole health for learning and accountability, and (4) the financing of whole health. Recognizing the need for these elements is not new; in fact, the committee draws on several previous reports to describe the state of and recommendations for these foundational elements. However, the need to scale and spread whole health care underscores their urgency.
Advancing infrastructure related to health informatics will require developing systems that are more patient centered; enable data sharing among interprofessional team members and end users; foster teamwork through communication, collaboration, and coordination tools; support data collection for whole health services beyond conventional medical services; support more accessible and proactive care, including care at home, through virtual technologies and automated functions; foster team wellbeing; and improve the care delivery experience for patients, families and communities. Interoperability across all information systems is essential. VA systems need to develop the ability to interact with non-VA systems and national mandates to continue to support interoperability need to be enforced. Health informatics systems need to develop new data entry fields that identify and track social care needs and whole health services as part of electronic medical records.
Developing a whole health workforce that is available, accessible, and supported in its efforts will require strengthening individual and organizational competencies and capacities for both clinical and social services across VA and non-VA systems. Workforce shortages that affect health care will affect the nation’s ability to scale whole health with the diversity required to improve health and equity outcomes. Current forms of training do not reflect a commitment to whole health and are not aligned with interprofessional team-based approaches to care. To successfully field whole health teams, training centers will need to train groups of professionals from diverse disciplines as teams, emphasize all foundational elements of whole health, and reinforce training by creating care settings that rely
upon interprofessional teams. The appropriate approach for training whole health teams will vary, depending on the size, needs, and resources of communities they serve, but all should foster conditions that enable workforce well-being. Federal actions, such as increasing training slots and using incentives to direct trainees to needed positions, will be required to develop sufficient workforce capacity to support whole health.
Measuring progress and outcomes is essential to the learning and adaptive process for scaling and spreading whole health. Health care organizations and policy makers need measures that enable both learning and accountability; such measures should address mico, meso, and macro levels and must remain distinct from one another in order to achieve either goal. The committee has recommended a set of principles and guidelines to help ensure that measurement promotes whole health transformation rather than devolving to a tick box exercise. Scaling and spreading whole health will require new measures that capture and track the five foundational elements of whole health. Making progress toward the goal of whole health will depend on the ability to measure change in response to new efforts, investments, and innovations.
As the evidence clearly shows, the manner in which health care is financed and paid often determines how patients and communities access care and which services they receive. The financing and payment processes found in most systems, especially fee-for-service systems with no accountability for spending or quality, fail to support the whole health continuum of services and often incentivize the delivery of low-value health care services. Prospective payment models, including bundled and global payments that aim to improve the value of care, align better with the philosophy of whole health. However, substantial evidence suggests that the savings are fairly modest and that the observed quality improvements are generally limited to process measures rather than improvements in health outcomes. Despite some encouraging examples of global payment models, providing support for whole health will require further improvements in the design and implementation of financing and payment models. In particular, the MISSION Act as currently implemented remains difficult for veterans, VA staff, and community networks to navigate, and its spiraling costs threaten to destabilize funding for VA’s in-house health care programs. Putting these critical elements of foundational infrastructure in place will require experimentation and incremental change, combined public- and private-sector initiative and collaboration, and resources and leadership at multiple levels, ranging from the macro levels of the regional health systems and state and federal government to the micro levels of individuals, families, and communities. If the past truly is prologue, even seismic change on this scale is possible with persistent, coordinated, and thoughtful efforts across sectors.
REFERENCES
AAMC (Association of American Medical Colleges). 2021a. Annual development survey: 2021 fundraising data. https://www.aamc.org/data-reports/faculty-institutions/interactive-data/2021-fundraising-data (accessed November 10, 2022).
AAMC. 2021b. AAMC report reinforces mounting physician shortage. https://www.aamc.org/news-insights/press-releases/aamc-report-reinforces-mounting-physician-shortage (accessed November 10, 2022).
Agarwal, R., J. Connolly, S. Gupta, and A. Navathe. 2021. Comparing Medicare Advantage and traditional Medicare: A systematic review. Health Affairs 40(6):937–944.
AHRQ (Agency for Healthcare Research and Quality). n.d. Architecture of health IT. https://digital.ahrq.gov/architecture-health-it (accessed November 10, 2022).
AMA (American Medical Assocation). 2022. AMA digital health care 2022 study findings. https://www.ama-assn.org/about/research/ama-digital-health-care-2022-study-findings (accessed December 6, 2022).
Anderson, E., K. Dvorin, B. Etingen, A. M. Barker, Z. Rai, A. Herbst, R. Mozer, R. P. Kingston, and B. Bokhour. 2022. Lessons learned from VHA’s rapid implementation of virtual whole health peer-led groups during the COVID-19 pandemic: Staff perspectives. Global Advances in Health and Medicine 11:21649561211064244.
Apple Inc. 2022a. Apple podcasts preview. https://podcasts.apple.com/us/podcast/military-meditation-coach-podcast/id1313813296 (accessed October 7, 2022).
Apple Inc. 2022b. Defense Health Agency iPad & iPhone apps. https://apps.apple.com/us/developer/defense-health-agency/id1264687017?see-all=i-phonei-pad-apps (accessed October 7, 2022).
Arizona Prevention Research Center. 2015. 2014 National Community Health Worker Advocacy Survey. Tuscon, AZ: University of Arizona.
ASPE (Assistant Secretary for Planning and Evaluation). 2022a. Impact of the COVID-19 pandemic on the hospital and outpatient clinician workforce. https://aspe.hhs.gov/reports/covid-19-health-care-workforce (accessed November 10, 2022).
ASPE. 2022b. OS-patient-centered outcomes research trust fund (OS-PCORTF). https://aspe.hhs.gov/collaborations-committees-advisory-groups/os-pcortf (accessed October 4, 2022).
Baicker, K., and A. Chandra. 2006. The labor market effects of rising health insurance premiums. Journal of Labor Economics 24(3):609–634.
Barnett, M. L., A. Mehrotra, and D. C. Grabowski. 2019a. Postacute care—The piggy bank for savings in alternative payment models? New England Journal of Medicine 381(4):302–303.
Barnett, M. L., A. Wilcock, J. M. McWilliams, A. M. Epstein, K. E. Joynt Maddox, E. J. Orav, D. C. Grabowski, and A. Mehrotra. 2019b. Two-year evaluation of mandatory bundled payments for joint replacement. New England Journal of Medicine 380(3):252–262.
Berenson, R. A. 2016. If you can’t measure performance, can you improve it? JAMA 315(7):645.
Berlin, G., M. Lapointe, M. Murphy, and J. Wexler. 2022. Assessing the lingering impact of COVID-19 on the nursing workforce. https://www.mckinsey.com/industries/healthcare-systems-and-services/our-insights/assessing-the-lingering-impact-of-covid-19-on-the-nursing-workforce (accessed November 10, 2022).
Bertot, J. C., C. R. McClure, P. T. Jaeger, and J. Ryan. 2006. Public libraries and the internet 2006: Study results and findings. Bill and Melinda Gates Foundation and the American Library Association.
Berwick, D. M. 1989. Continuous improvement as an ideal in health care. New England Journal of Medicine 320(1):53–56.
Berwick, D. M., and K. Shine. 2020. Enhancing private sector health system preparedness for 21st-century health threats. JAMA 323(12):1133–1134.
Blumenthal, D. 1996. Quality of care—What is it? New England Journal of Medicine 335(12):891–894.
Bokhour, B. G., J. Hyde, S. Zeliadt, and D. Mohr. 2020. Whole Health System of Care evaluation—A progress report on outcomes of the WHS pilot at 18 flagship sites. Bedford, MA: VA Center for Evaluating Patient-Centered Care in VA.
Bradley, E., M. Canavan, E. Rogan, K. Talbert-Slagle, C. Ndumele, L. Taylor, and L. Curry. 2016. Variation in health outcomes: The role of spending on social services, public health, and health care, 2000–09. Health Affairs 35(5):760–768.
Bradley, E. H., L. A. Taylor, and H. V. Fineberg. 2013. The American health care paradox: Why spending more is getting us less. New York: PublicAffairs.
Brooks, G. A., S. Jhatakia, A. Tripp, M. B. Landrum, T. J. Christian, G. Newes-Adeyi, S. Cafardi, A. Hassol, C. Simon, and N. L. Keating. 2019. Early findings from the Oncology Care Model evaluation. Journal of Oncology Practice 15(10):e888–e896.
Bur, J. 2020. Will the VA’s hiring gains be maintained post-pandemic? Federal Times, July 2. https://www.federaltimes.com/management/hr/2020/07/02/will-the-vas-hiring-gains-be-maintained-post-pandemic/ (accessed July 18, 2022).
Burwell, S. M. 2015. Setting value-based payment goals—HHS efforts to improve U.S. health care. New England Journal of Medicine 372(10):897–899.
Cantor, M. N., and L. Thorpe. 2018. Integrating data on social determinants of health into electronic health records. Health Affairs (Millwood) 37(4):585–590.
Carayon, P., and P. Hoonakker. 2019. Human factors and usability for health information technology: Old and new challenges. Yearbook of Medical Informatics 28(1):71-77.
Chandra, A., D. Cutler, and Z. Song. 2011. Who ordered that? The economics of treatment choices in medical care. Chapter 6 in M. V. Pauly, T. G. McGuire, and P. P. Barros (eds.), Handbook of Health Economics, Vol. 2. Amsterdam: Elsevier. Pp. 397–432.
Chiou, L., C. Tucker, and National Bureau of Economic Research. 2020. Social distancing, internet access and inequality. NBER working paper no. 26982. Cambridge, MA: National Bureau of Economic Research.
Clemens, J., and D. M. Cutler. 2014. Who pays for public employee health costs? Journal of Health Economics 38:65–76.
CMS (Centers for Medicare & Medicaid Services). 2022a. Comprehensive care for joint replacement model. https://innovation.cms.gov/innovation-models/cjr (accessed date here).
CMS. 2022b. Oncology care model. https://innovation.cms.gov/innovation-models/oncology-care (accessed November 10, 2022).
COGME (Council on Graduate Medical Education). 2016. Supporting diversity in health professions. Rockville, MD: Council on Graduate Medical Education.
Cohen, J. J., B. A. Gabriel, and C. Terrell. 2002. The case for diversity in the health care workforce. Health Affairs 21(5):90–102.
Colicchio, T. K., J. J. Cimino, and G. Del Fiol. 2019. Unintended consequences of nationwide electronic health record adoption: Challenges and opportunities in the post-meaningful use era. Journal of Medical Internet Research 21(6):e13313.
Cooper, L. A., and N. R. Powe. 2004. Disparities in patient experiences, health care processes, and outcomes: The role of patient–provider racial, ethnic, and language concordance. New York: The Commonwealth Fund.
Curt, A., H. Khidir, G. Ciccolo, C. A. Camargo, Jr., and M. Samuels-Kalow. 2021. Geographically indexed referral databases to address social needs in the emergency department. Western Journal of Emerging Medicine 22(2):218–224.
Cutler, D., and R. Zeckhauser. 2000. The anatomy of health insurance. Chapter 11 in A. Culyer and J. Newhouse (eds.), Handbook of Health Economics. Amsterdam: Elsevier. Pp. 563–643.
Cutler, D. M. 2005. Your money or your life: Strong medicine for America’s health care system. New York: Oxford University Press.
De Brantes, F., M. B. Rosenthal, and M. Painter. 2009. Building a bridge from fragmentation to accountability—The Prometheus payment model. New England Journal of Medicine 361(11):1033–1036.
De Marchis, E., M. Knox, D. Hessler, R. Willard-Grace, J. N. Olayiwola, L. E. Peterson, K. Grumbach, and L. M. Gottlieb. 2019. Physician burnout and higher clinic capacity to address patients’ social needs. Journal of the American Board of Family Medicine 32(1):69–78.
DirectTrust. n.d. Who is DirectTrust? https://directtrust.org/who-we-are (accessed November 10, 2022).
Donabedian, A. 1980. Explorations in quality assessment and monitoring: The definition of quality and approaches to its assessment. Chicago: Health Administration Press.
Donabedian, A. 1988. The quality of care: How can it be assessed? JAMA 260(12):1743–1748.
Dryden, E. M., R. E. Bolton, B. G. Bokhour, J. Wu, K. Dvorin, L. Phillips, and J. K. Hyde. 2021. Leaning into whole health: Sustaining system transformation while supporting patients and employees during COVID-19. Global Advances in Health and Medicine 10:21649561211021047.
Duggan, M., J. Gruber, and B. Vabson. 2018. The consequences of health care privatization: Evidence from Medicare Advantage exits. American Economic Journal: Economic Policy 10(1):153–186.
Dyrbye, L. N., M. R. Thomas, A. Eacker, W. Harper, F. S. Massie, Jr., D. V. Power, M. Huschka, P. J. Novotny, J. A. Sloan, and T. D. Shanafelt. 2007. Race, ethnicity, and medical student well-being in the United States. Archives of Internal Medicine 167(19):2103–2109.
Eby, D., and L. Ross. 2016. Internationally heralded approaches to population health driven by Alaska Native/American Indian/Native American communities IHI 28th annual national forum. Presentation for IHI 28th annual national forum. http://app.ihi.org/events/SessionDetail.aspx?EventId=2760&SessionId=39156 (accessed March 29, 2022).
Edmondson, A. C. 1996. Learning from mistakes is easier said than done: Group and organizational influences on the detection and correction of human error. Journal of Applied Behavioral Science 32(1):5–28.
Edmondson, A. C. 2019. The fearless organization: Creating psychological safety in the workplace for learning, innovation, and growth. Hoboken, NJ: John Wiley & Sons, Inc.
eHealth Exchange. n.d. Home page. https://ehealthexchange.org/ (accessed November 10, 2022).
Emanuel, E., N. Tanden, S. Altman, S. Armstrong, D. Berwick, F. De Brantes, M. Calsyn, M. Chernew, J. Colmers, D. Cutler, T. Daschle, P. Egerman, B. Kocher, A. Milstein, E. Oshima Lee, J. D. Podesta, U. Reinhardt, M. Rosenthal, J. Sharfstein, S. Shortell, A. Stern, P. R. Orszag, and T. Spiro. 2012. A systemic approach to containing health care spending. New England Journal of Medicine 367(10):949–954.
Emanuel, E. J., D. Johnson, M. Guido, and M. Goozner. 2022. Meaningful value-based payment reform, part 1: Maryland leads the way. Health Affairs Forefront, February 9. https://www.healthaffairs.org/do/10.1377/forefront.20220205.211264/ (accessed November 10, 2022).
Etz, R., and the Starfield Writing Team. 2017. Guidance for developing PC measures. Conference brief. Washington, DC: Starfield Summit III.
Everson, J., V. Patel, and J. Adler-Milstein. 2021. Information blocking remains prevalent at the start of 21st Century Ccures Act: Results from a survey of health information exchange organizations. Journal of the American Medical Informatics Association 28(4):727–732.
FCC (Federal Communications Commission). 2012. Eighth broadband progress report. https://www.fcc.gov/reports-research/reports/broadband-progress-reports/eighth-broadband-progress-report (accessed November 10, 2022).
FCC. 2019. Report on promoting internet access service for veterans. https://www.fcc.gov/document/report-promoting-internet-access-service-veterans (accessed November 10, 2022).
Ferguson, J. M., J. Jacobs, M. Yefimova, L. Greene, L. Heyworth, and D. M. Zulman. 2021. Virtual care expansion in the Veterans Health Administration during the COVID-19 pandemic: Clinical services and patient characteristics associated with utilization. Journal of the American Medical Informatics Association 28(3):453–462.
Finkelstein, A., Y. Ji, N. Mahoney, and J. Skinner. 2018. Mandatory Medicare bundled payment program for lower extremity joint replacement and discharge to institutional postacute care. JAMA 320(9):892–900.
Freed, M., J. Biniek, A. Damico, and T. Neuman. 2022. Medicare Advantage in 2022: Enrollment update and key trends. https://www.kff.org/medicare/issue-brief/medicare-advantage-in-2022-enrollment-update-and-key-trends/ (accessed November 10, 2022).
Furlow, B. 2020. Information overload and unsustainable workloads in the era of electronic health records. The Lancet Respiratory Medicine 8(3):243–244.
Galvin, G. 2021. Nearly 1 in 5 health care workers have quit their jobs during the pandemic. https://morningconsult.com/2021/10/04/health-care-workers-series-part-2-workforce/ (accessed July 19, 2022).
Gamache, R., H. Kharrazi, and J. Weiner. 2018. Public and population health informatics: The bridging of big data to benefit communities. Yearbook of Medical Informatics 27(01):199–206.
GAO (U.S. Government Accountability Office). 1996. Veterans’ health care facilities resource allocations could be more equitable. GAO/HEHS-96-48. Washington, DC: U.S. Government Accountability Office.
GAO. 2002. VA health care: Allocation changes would better align resources with workload. GAO-02-338. Washington, DC: U.S. Government Accountability Office.
GAO. 2019a. VA health care: Estimating resources needed to provide community care. GAO-19-478. Washington, DC: U.S. Government Accountability Office.
GAO. 2019b. Veterans health care: VA needs to improve its allocation and monitoring of funding. GAO-19-670. Washington, DC: U.S. Government Accountability Office.
Gilfillan, R., and D. Berwick. 2021. Medicare Advantage, direct contracting, and the Medicare “money machine,” part 1: The risk-score game. Health Affairs Forefront. https://www.healthaffairs.org/do/10.1377/forefront.20210927.6239/ (accessed November 10, 2022).
Ginsburg, P. 2012. Fee-for-service will remain a feature of major payment reforms, requiring more changes in medicare physician payment. Health Affairs (Millwood) 31(9): 1977–1983.
Ginsburg, P., and S. Lieberman. 2022. The debate on overpayment in Medicare Advantage: Pulling it together. Health Affairs Forefront. https://www.healthaffairs.org/do/10.1377/forefront.20220223.736815/ (accessed November 10, 2022).
Glasgow, R. E., A. G. Huebschmann, A. H. Krist, and F. V. Degruy. 2019. An adaptive, contextual, technology-aided support (ACTS) system for chronic illness self-management. Milbank Quarterly 97(3):669–691.
Goldman, J., S. Kitto, and S. Reeves. 2018. Examining the implementation of collaborative competencies in a critical care setting: Key challenges for enacting competency-based education. Journal of Interprofessional Care 32(4):407–415.
Gottgens, I., and S. Oertelt-Prigione. 2021. The application of human-centered design approaches in health research and innovation: A narrative review of current practices. JMIR mHealth and uHealth 9(12):e28102.
Graham, J. M., D. W. Cowling, and H. Zhang. 2021. Findings from a commercial ACO patient experience survey. Journal of Patient Experience 8:237437352110078.
Greenhalgh, T., and I. Heath. 2010. Measuring quality in the therapeutic relationship—Part 1: Objective approaches. BMJ Quality & Safety 19(6):475–478.
GSA (U.S. General Services Administration). 2018. Human centered design (HCD) discovery stage field guide v.1. https://www.gsa.gov/cdnstatic/HCD-Discovery-Guide-Interagency-v12-1.pdf (accessed November 10, 2022).
Haas, D. A., X. Zhang, R. S. Kaplan, and Z. Song. 2019. Evaluation of economic and clinical outcomes under Centers for Medicare & Medicaid Services mandatory bundled payments for joint replacements. JAMA Internal Medicine 179(7):924–931.
Health Care Payment Learning & Action Network. 2022. APM measurement effort. https://hcp-lan.org/apm-measurement-effort/2022-apm/2022-infographic/ (accessed December 7, 2022).
Health Professions Accreditors Collaborative. 2019. Guidance on developing quality interprofessional education for the health professions. Chicago, IL: Health Professions Accreditors Collaborative.
Help Finder. 2022. Home page. https://helpfinder.com/ (accessed November 10, 2022).
HHS (Department of Health and Human Services). 2020. Interoperability and patient access. https://www.cms.gov/files/document/cms-9115-f.pdf (accessed June 22, 2022).
HHS. 2022. New Surgeon General advisory sounds alarm on health worker burnout and resignation. https://www.hhs.gov/about/news/2022/05/23/new-surgeon-general-advisorysounds-alarm-on-health-worker-burnout-and-resignation.html (accessed November 10, 2022).
Hogan, T. P., K. M. Nazi, T. M. Luger, D. J. Amante, B. M. Smith, A. Barker, S. L. Shimada, J. E. Volkman, L. Garvin, S. R. Simon, and T. K. Houston. 2014. Technology-assisted patient access to clinical information: An evaluation framework for Blue Button. JMIR Research Protocols 3(1):e18.
Holmstrom, B., and P. Milgrom. 1991. Multitask principal-agent analyses: Incentive contracts, asset ownership, and job design. Journal of Law, Economics, & Organization 7(Spec Iss):24–52.
HRSA (Health Resources and Services Administration). 2017. Sex, race, and ethnic diversity of U.S. health occupations (2011–2015). Rockville, MD: Health Resources and Services Administration.
HRSA. 2022. Apply for a grant. https://www.hrsa.gov/grants/apply-for-a-grant (accessed September 18, 2022).
IOM (Institute of Medicine). 2003. Unequal treatment: Confronting racial and ethnic disparities in health care. Washington, DC: The National Academies Press.
IOM. 2012. Health IT and patient safety: Building safer systems for better care. Washington, DC: The National Academies Press.
IOM. 2014. Capturing social and behavioral domains in electronic health records: Phase 1. Washington, DC: The National Academies Press.
IOM. 2015. Capturing social and behavioral domains and measures in electronic health records: Phase 2. Washington, DC: The National Academies Press.
Jackson, C. S., and J. N. Gracia. 2014. Addressing health and health-care disparities: The role of a diverse workforce and the social determinants of health. Public Health Reports 1292(Suppl 2):57–61.
Jacobs, P. D., and R. Kronick. 2021. The effects of coding intensity in Medicare Advantage on plan benefits and finances. Health Services Research 56(2):178–187.
Jalili, M., M. Niroomand, F. Hadavand, K. Zeinali, and A. Fotouhi. 2021. Burnout among healthcare professionals during COVID-19 pandemic: A cross-sectional study. International Archives of Occupational and Environmental Health 94(6):1345–1352.
Jonas, W. B., and E. Rosenbaum. 2021. The case for whole-person integrative care. Medicina 57(7):677.
Kaiser Family Foundation. 2021. Mental health care health professional shortage areas (HPSAs). https://www.kff.org/other/state-indicator/mental-health-care-health-professional-shortage-areas-hpsas/ (accessed July 18, 2022).
Kanter, M. H., M. Nguyen, M. H. Klau, N. H. Spiegel, and V. L. Ambrosini. 2013. What does professionalism mean to the physician? Permanente Journal 17(3):87–90.
Kaufman, A., and C. Alfero. 2015. A state-based strategy for expanding primary care residency. Health Affairs Forefront. https://www.healthaffairs.org/do/10.1377/forefront.20150731.049707 (accessed November 10, 2022).
Keating, N. L., S. Jhatakia, G. A. Brooks, A. S. Tripp, I. Cintina, M. B. Landrum, Q. Zheng, T. J. Christian, R. Glass, V. D. Hsu, C. M. Kummet, S. Woodman, C. Simon, A. Hassol, G. Norton, S. McClellan, J. Gerteis, M. A. Manetas, N. West, A. Evans, M. Juergens, S. Shao, N. S. Kapedia, L. Riedel, M. P. H. Liu, R. Wolf, H. Uno, M. Davidson, R. Acanfora, N. Ume, D. Zhang, D. Davis, A. Braendle, S. Khan, C. Wong, and S. Negrusa. 2021. Association of participation in the Oncology Care Model with Medicare payments, utilization, care delivery, and quality outcomes. JAMA 326(18):1829–1839.
Keating, N. L., G. A. Brooks, M. B. Landrum, P. H. Liu, R. Wolf, L. E. Riedel, N. S. Kapadia, S. Jhatakia, A. Tripp, C. Simon, V. D. Hsu, C. M. Kummet, and A. Hassol. 2022. The Oncology Care Model and adherence to oral cancer drugs: A difference-in-differences analysis. Journal of the National Cancer Institute 114(6):871–877.
Kilbourne, A. M., J. Schmidt, M. Edmunds, R. Vega, N. Bowersox, and D. Atkins. 2022. How the VA is training the next‐generation workforce for learning health systems. Learning Health Systems 6(4). https://doi.org/10.1002/lrh2.10333.
King, J., V. Patel, E. W. Jamoom, and M. F. Furukawa. 2014. Clinical benefits of electronic health record use: National findings. Health Services Research 49(1 Pt 2):392–404.
Kirzinger, A., A. Kearney, L. Hamel, and M. Brodie. 2021. KFF/The Washington Post frontline health care workers survey. https://www.kff.org/coronavirus-covid-19/poll-finding/kff-washington-post-health-care-workers/ (accessed July 18, 2022).
Klein, D. M., G. M. Fix, T. P. Hogan, S. R. Simon, K. M. Nazi, and C. L. Turvey. 2015. Use of the Blue Button online tool for sharing health information: Qualitative interviews with patients and providers. Journal of Medical Internet Research 17(8):e199.
Krist, A. H., and S. H. Woolf. 2011. A vision for patient-centered health information systems. JAMA 305(3):300–301.
Krist, A. H., S. H. Woolf, S. F. Rothemich, R. E. Johnson, J. E. Peele, T. D. Cunningham, D. R. Longo, G. A. Bello, and G. R. Matzke. 2012. Interactive preventive health record to enhance delivery of recommended care: A randomized trial. Annals of Family Medicine 10(4):312–319.
Krist, A. H., S. H. Woolf, C. Hochheimer, R. T. Sabo, P. Kashiri, R. M. Jones, J. E. Lafata, R. S. Etz, and S. P. Tu. 2017. Harnessing information technology to inform patients facing routine decisions: Cancer screening as a test case. Annals of Family Medicine 15(3):217–224.
Krist, A. H., J. E. DeVoe, A. Cheng, T. Ehrlich, and S. M. Jones. 2020a. Redesigning primary care to address the COVID-19 pandemic in the midst of the pandemic. Annals of Family Medicine 18(4):349–354.
Krist, A. H., K. O’Loughlin, S. H. Woolf, R. T. Sabo, J. Hinesley, A. J. Kuzel, B. D. Rybarczyk, P. L. Kashiri, E. M. Brooks, R. E. Glasgow, A. G. Huebschmann, and W. R. Liaw. 2020b. Enhanced care planning and clinical-community linkages versus usual care to address basic needs of patients with multiple chronic conditions: A clinician-level randomized controlled trial. Trials 21(1):517.
Krist, A. H., R. Phillips, L. Leykum, and B. Olmedo. 2021. Digital health needs for implementing high-quality primary care: Recommendations from the National Academies of Sciences, Engineering, and Medicine. Journal of the American Medical Informatics Association 28(12):2738–2742.
Kronick, R. 2017. Projected coding intensity in Medicare Advantage could increase Medicare spending by $200 billion over ten years. Health Affairs 36(2):320–327.
Landon, B. E., T. S. Anderson, V. E. Curto, P. Cram, C. Fu, G. Weinreb, A. M. Zaslavsky, and J. Z. Ayanian. 2022. Association of Medicare Advantage vs traditional Medicare with 30-day mortality among patients with acute myocardial infarction. JAMA 328(21):2126.
Laugesen, M., and S. Glied. 2011. Higher fees paid to U.S. physicians drive higher spending for physician services compared to other countries. Health Affairs 30(9):1647–1656.
Lebares, C. C., A. L. Greenberg, N. L. Ascher, K. L. Delucchi, L. M. Reilly, M. van der Schaaf, F. Baathe, P. O’Sullivan, and K. Isaksson Rø. 2021. Exploration of individual and system-level well-being initiatives at an academic surgical residency program: A mixed-methods study. JAMA Network Open 4(1):e2032676.
Lester, P. E., T. S. Dharmarajan, and E. Weinstein. 2020. The looming geriatrician shortage: Ramifications and solutions. Journal of Aging and Health 32(9):1052–1062.
Lindau, S. T., J. A. Makelarski, E. M. Abramsohn, D. G. Beiser, K. Boyd, C. Chou, M. Giurcanu, E. S. Huang, C. Liao, L. P. Schumm, and E. L. Tung. 2019. CommunityRx: A real-world controlled clinical trial of a scalable, low-intensity community resource referral intervention. American Journal of Public Health 109(4):600–606.
Lynch, T. 2020. Building trust & health equity during COVID-19. Background paper. ABIM Foundation. https://abimfoundation.org/wp-content/uploads/2020/07/Foundation2020-Forum-Background-Paper.pdf (accessed November 10, 2022).
Mandel, J. C., D. A. Kreda, K. D. Mandl, I. S. Kohane, and R. B. Ramoni. 2016. SMART on FHIR: A standards-based, interoperable apps platform for electronic health records. Journal of the American Medical Informatics Association 23(5):899–908.
Mangione-Smith, R., A. H. Decristofaro, C. M. Setodji, J. Keesey, D. J. Klein, J. L. Adams, M. A. Schuster, and E. A. McGlynn. 2007. The quality of ambulatory care delivered to children in the United States. New England Journal of Medicine 357(15):1515–1523.
Marwaha, J. S., A. B. Landman, G. A. Brat, T. Dunn, and W. J. Gordon. 2022. Deploying digital health tools within large, complex health systems: Key considerations for adoption and implementation. npj Digital Medicine 5(1):13.
McGlynn, E. A., S. M. Asch, J. Adams, J. Keesey, J. Hicks, A. Decristofaro, and E. A. Kerr. 2003. The quality of health care delivered to adults in the United States. New England Journal of Medicine 348(26):2635–2645.
McNeil, J. J., S. M. Evans, N. P. Johnson, and P. A. Cameron. 2010. Clinical-quality registries: Their role in quality improvement. Medical Journal of Australia 192(5):244–245.
McWilliams, J. M. 2020. Professionalism revealed: Rethinking quality improvement in the wake of a pandemic. NEJM Catalyst 1(5). https://catalyst.nejm.org/doi/full/10.1056/CAT.20.0226 (accessed November 10, 2022).
McWilliams, J. M., M. E. Chernew, A. M. Zaslavsky, and B. E. Landon. 2013. Post-acute care and ACOs—Who will be accountable? Health Services Research 48(4):1526–1538.
McWilliams, J. M., M. E. Chernew, B. E. Landon, and A. L. Schwartz. 2015. Performance differences in year 1 of pioneer accountable care organizations. New England Journal of Medicine 372(20):1927–1936.
McWilliams, J. M., L. G. Gilstrap, D. G. Stevenson, M. E. Chernew, H. A. Huskamp, and D. C. Grabowski. 2017. Changes in postacute care in the Medicare shared savings program. JAMA Internal Medicine 177(4):518–526.
McWilliams, J. M., L. A. Hatfield, B. E. Landon, P. Hamed, and M. E. Chernew. 2018. Medicare spending after 3 years of the Medicare Shared Savings Program. New England Journal of Medicine 379(12):1139–1149.
McWilliams, J. M., B. E. Landon, V. K. Rathi, and M. E. Chernew. 2019. Getting more savings from ACOs—Can the pace be pushed? New England Journal of Medicine 380(23):2190–2192.
McWilliams, J. M., L. A. Hatfield, B. E. Landon, and M. E. Chernew. 2020. Savings or selection? Initial spending reductions in the Medicare Shared Savings Program and considerations for reform. Milbank Quarterly 98(3):847–907.
Melles, M., A. Albayrak, and R. Goossens. 2021. Innovating health care: Key characteristics of human-centered design. International Journal for Quality in Health Care 33(Suppl 1):37–44.
Meyers, D., A. M. Ryan, and A. N. Trivedi. 2022. How much of an “advantage” is Medicare Advantage? JAMA Network 328(21):2112-2113.
Meyers, D. J., V. Mor, and M. Rahman. 2018. Medicare Advantage enrollees more likely to enter lower-quality nursing homes compared to fee-for-service enrollees. Health Affairs 37(1):78-85.
MITRE Corporation. 2021. APM measurement: Progress of alternative payment models, 2020–2021 methodolgoy and results report. Health Care Payment Learning & Action Network. http://hcp-lan.org/workproducts/APM-Methodology-2020-2021.pdf (accessed November 10, 2022).
Montano, D., H. Hoven, and J. Siegrist. 2014. Effects of organisational-level interventions at work on employees’ health: A systematic review. BMC Public Health 14(1):135.
Muhiyaddin, R., A. Elfadl, E. Mohamed, Z. Shah, T. Alam, A. Abd-Alrazaq, and M. Househ. 2022. Electronic health records and physician burnout: A scoping review. Studies in Health Technology and Informatics 289:481–484.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2019. Taking action against clinician burnout: A systems approach to professional well-being. Washington, DC: The National Academies Press.
NASEM. 2021. Implementing high-quality primary care: Rebuilding the foundation of health care. Washington, DC: The National Academies Press.
NASEM. 2022. Building data capacity for patient-centered outcomes research: Priorities for the next decade. Washington, DC: The National Academies Press.
Nelson, E. C., M. Dixon-Woods, P. B. Batalden, K. Homa, A. D. Van Citters, T. S. Morgan, E. Eftimovska, E. S. Fisher, J. Ovretveit, W. Harrison, C. Lind, and S. Lindblad. 2016. Patient focused registries can improve health, care, and science. BMJ 354:i3319.
Newhouse, J. P. 2002. Pricing the priceless: A health care conundrum. Cambridge, MA: The MIT Press.
NLM (National Library of Medicine). 2016. Overview of SNOMED CT. https://www.nlm.nih.gov/healthit/snomedct/snomed_overview.html (accessed November 10, 2022).
NLM. 2021. VSAC authoring best practices. https://www.nlm.nih.gov/vsac/support/author-guidelines/bestpractices.html (accessed July 23, 2022.
NRC (National Research Council). 2007. Using the American Community Survey: Benefits and challenges. Washington, DC: The National Academies Press.
Nyweide, D. J., W. Lee, T. T. Cuerdon, H. H. Pham, M. Cox, R. Rajkumar, and P. H. Conway. 2015. Association of pioneer accountable care organizations vs traditional Medicare fee for service with spending, utilization, and patient experience. JAMA 313(21):2152–2161.
Odendaal, W. A., J. Anstey Watkins, N. Leon, J. Goudge, F. Griffiths, M. Tomlinson, and K. Daniels. 2020. Health workers’ perceptions and experiences of using mHealth technologies to deliver primary healthcare services: A qualitative evidence synthesis. Cochrane Database of Systematic Reviews 2020(3):CD011942.
ODPHP (Office of Disease Prevention and Health Promotion). 2022. Healthy People 2030 https://health.gov/healthypeople (accessed October 7, 2022).
Office of Information and Technology. 2020. Fy2020-2022 IT (IRM) Strategic Plan. https://www.ea.oit.va.gov/EAOIT/docs/Oct2020/IRM-Strategic-Plan-FY2020-2022-v26.pdf?msclkid=9a8523a4be9a11ec95fbe9b3209f6e4f (accessed August 15, 2022).
ONC (Office of the National Coordinator for Health Information Technology). 2015. Report on health information blocking. Washington, DC: Department of Health and Human Services. https://www.heal-thit.gov/sites/default/files/reports/info_blocking_040915.pdf (accessed November 10, 2022).
ONC. 2019a. Office-based physician electronic health record adoption. https://www.healthit.gov/data/quickstats/office-based-physician-electronic-health-record-adoption (accessed November 10, 2022).
ONC. 2019b. Hospitals’ use of electronic health records data, 2015–2017. https://www.healthit.gov/sites/default/files/page/2019-04/AHAEHRUseDataBrief.pdf (accessed November 10, 2022).
ONC. 2020. Information blocking and the ONC Health IT Certification Program: Extension of compliance dates and timeframes in response to the COVID-19 public health emergency: A rule by the Health and Human Services Department on 11/04/2020. Federal Register 85:70064–70085.
ONC. 2021. Health Interoperability Outcomes 2030. https://www.healthit.gov/topic/interoperability/health-interoperability-outcomes-2030 (accessed July 22, 2022).
ONC. 2022a. Information blocking. https://www.healthit.gov/topic/information-blocking (accessed July 22, 2022).
ONC. 2022b. United States Core Data for Interoperability (USCDI). https://www.healthit.gov/isa/united-states-core-data-interoperability-uscdi (accessed October 6, 2022).
Panagioti, M., E. Panagopoulou, P. Bower, G. Lewith, E. Kontopantelis, C. Chew-Graham, S. Dawson, H. van Marwijk, K. Geraghty, and A. Esmail. 2017. Controlled interventions to reduce burnout in physicians: A systematic review and meta-analysis. JAMA Internal Medicine 177(2):195–205.
Peikes, D., G. Anglin, E. Taylor, S. Dale, A, O’Malley, and A. Ghosh. 2016. Evaluation of the comprehensive primary care initiative: Third annual report. Cambridge, MA: Mathematica Policy Research.
Peikes, D., S. Dale, A. Ghosh, E. F. Taylor, K. Swankoski, A. S. O’Malley, T. J. Day, N. Duda, P. Singh, G. Anglin, L. L. Sessums, and R. S. Brown. 2018. The Comprehensive Primary Care initiative: Effects on spending, quality, patients, and physicians. Health Affairs (Millwood) 37(6):890–899.
Perlin, J. B., R. M. Kolodner, and R. H. Roswell. 2004. The Veterans Health Administration: Quality, value, accountability, and information as transforming strategies for patient-centered care. American Journal of Managed Care 10(11 Pt 2):828–836.
Pham, H. H., J. Pilotte, R. Rajkumar, E. Richter, S. Cavanaugh, and P. H. Conway. 2015. Medicare’s vision for delivery-system reform—The role of ACOs. New England Journal of Medicine 373(11):987–990.
Phillips, R., A. Bazemore, W. Newton, and R. Byyny. 2019. Medical professionalism: A contract with society. The Pharos, Autumn: 2–7. https://www.alphaomegaalpha.org/wp-content/uploads/2021/03/19_Autumn_Editorial.pdf (accessed November 10, 2022).
Phillips, R. L. 2020. The built environment for professionalism. Journal of the American Board of Family Medicine 33(Suppl):S57–S61.
Poma, P. A. 2017. Race/ethnicity concordance between patients and physicians. Journal of the National Medical Association 109(1):6–8.
Powers, B. W., S. Rinefort, and S. H. Jain. 2016. Nonemergency medical transportation: Delivering care in the era of Lyft and Uber. JAMA 316(9):921–922.
Press, M. J., R. Rajkumar, and P. H. Conway. 2016. Medicare’s new bundled payments. JAMA 315(2):131.
Rajkumar, R., P. H. Conway, and M. Tavenner. 2014. CMS—Engaging multiple payers in payment reform. JAMA 311(19):1967–1968.
Rajkumar, R., M. J. Press, and P. H. Conway. 2015. The CMS innovation center—A five-year self-assessment. New England Journal of Medicine 372(21):1981–1983.
Reddy, K. P., T. M. Schult, A. M. Whitehead, and B. G. Bokhour. 2021. Veterans Health Administration’s Whole Health System of Care: Supporting the health, well-being, and resiliency of employees. Global Advances in Health and Medicine 10:216495612110226.
Repenning, N. P., and J. D. Sterman. 2002. Capability traps and self-confirming attribution errors in the dynamics of process improvement. Administrative Science Quarterly 47(2):265-295.
Roberts, E. T., J. M. McWilliams, L. A. Hatfield, S. Gerovich, M. E. Chernew, L. G. Gilstrap, and A. Mehrotra. 2018a. Changes in health care use associated with the introduction of hospital global budgets in Maryland. JAMA Internal Medicine 178(2):260–268.
Roberts, E. T., A. Mehrotra, and M. E. Chernew. 2018b. Maryland’s hospital global budget program—Comment and response. JAMA 320(19):2040.
Roberts, E., L. Hatfield, M. McWilliams, M. Chernew, N. Done, S. Gerovich, L. Gilstrap, and A. Mehrotra. 2018c. Changes in hospital utilization three years into Maryland’s global budget program for rural hospitals. Health Affairs 37(4):644–653.
Robertson, S. L., M. D. Robinson, and A. Reid. 2017. Electronic health record effects on work–life balance and burnout within the I3 population collaborative. Journal of Graduate Medical Education 9(4):479–484.
Rose, L., M. Aouad, L. Graham, L. Schoemaker, and T. Wagner. 2021. Association of expanded health care networks with utilization among veterans affairs enrollees. JAMA Network Open 4(10):e2131141.
Rosenbaum, L. 2022a. Metric myopia—Trading away our clinical judgment. New England Journal of Medicine 386(18):1759–1763.
Rosenbaum, L. 2022b. Reassessing quality assessment—The flawed system for fixing a flawed system. New England Journal of Medicine 386(17):1663–1667.
Rosenthal, M. B., R. Fernandopulle, H. R. Song, and B. Landon. 2004. Paying for quality: Providers’ incentives for quality improvement. Health Affairs (Millwood) 23(2):127–141.
Salsberg, E., C. Richwine, S. Westergaard, M. Portela Martinez, T. Oyeyemi, A. Vichare, and C. P. Chen. 2021. Estimation and comparison of current and future racial/ethnic representation in the U.S. health care workforce. JAMA Network Open 4(3):e213789.
Sawesi, S., M. Rashrash, K. Phalakornkule, J. S. Carpenter, and J. F. Jones. 2016. The impact of information technology on patient engagement and health behavior change: A systematic review of the literature. JMIR Medical Informatics 4(1):e1.
Schmitt, M., A. Blue, C. A. Aschenbrener, and T. R. Viggiano. 2011. Core competencies for interprofessional collaborative practice: Reforming health care by transforming health professionals’ education. Academic Medicine 86(11):1351.
Schwartz, A. L., K. Zlaoui, R. P. Foreman, T. A. Brennan, and J. P. Newhouse. 2021. Health care utilization and spending in Medicare Advantage vs traditional Medicare. JAMA Health Forum 2(12):e214001.
Seftel-Kirk, H. 2019. How agencies like CMS and HHS are embracing HCD and why it’s worth the time. https://www.fedhealthit.com/2019/08/how-agencies-like-cms-and-hhs-are-embracing-hcd-and-why-its-worth-the-time/ (accessed October 13, 2022).
Sells, J. R., and J. R. McQuaid. 2021. VA academic affiliations matter in the era of community care: A model from California. Federal Practitioner 38(4):174–182.
Sharfstein, J. M., E. A. Stuart, and J. Antos. 2018. Global budgets in Maryland. JAMA 319(24):2475–2476.
Shaw-Taylor, Y. 2016. Nongovernment philanthropic spending on public health in the United States. American Journal of Public Health 106:58–62.
Shojania, K. G., A. Jennings, A. Mayhew, C. Ramsay, M. Eccles, and J. Grimshaw. 2010. Effect of point-of-care computer reminders on physician behaviour: A systematic review. CMAJ 182(5):E216–E225.
Singer, S. J., and A. C. Edmondson. 2008. When learning and performance are at odds: Confronting the tension. In P. Kumar and P. Ramsey (eds.), Learning and performance matter. Singapore: World Scientific Publishing. Pp. 33–60.
Singh, H., C. Spitzmueller, N. J. Petersen, M. K. Sawhney, and D. F. Sittig. 2013. Information overload and missed test results in electronic health record-based settings. JAMA Internal Medicine 173(8):702–704.
Smith, B. 2021. CMS innovation center at 10 years—Progress and lessons learned. New England Journal of Medicine 384(8):759–764.
Song, Z. 2014. Accountable care organizations in the U.S. health care system. Journal of Clinical Outcomes Management 21(8):364–371.
Song, Z., and C. H. Colla. 2016. Specialty-based global payment. JAMA 315(21):2271–2272.
Song, Z., and T. H. Lee. 2013. The era of delivery system reform begins. JAMA 309(1):35–36.
Song, Z., S. Rose, D. G. Safran, B. E. Landon, M. P. Day, and M. E. Chernew. 2014a. Changes in health care spending and quality 4 years into global payment. New England Journal of Medicine 371(18):1704–1714.
Song, Z., T. D. Sequist, and M. L. Barnett. 2014b. Patient referrals: A linchpin for increasing the value of care. JAMA 312(6):597–598.
Song, Z., V. Chopra, and L. F. McMahon, Jr. 2015. Addressing the primary care workforce crisis. American Journal of Managed Care 21(8):e452–e454.
Song, Z., Y. Ji, D. G. Safran, and M. E. Chernew. 2019. Health care spending, utilization, and quality 8 years into global payment. New England Journal of Medicine 381(3):252–263.
Song, Z., D. A. Chokshi, and M. J. Press. 2022. Primary care and financial risk—Navigating the crossroads. New England Journal of Medicine 387(4):292–294.
Stange, K. C., R. S. Etz, H. Gullett, S. A. Sweeney, W. L. Miller, C. R. Jaén, B. F. Crabtree, P. A. Nutting, and R. E. Glasgow. 2014. Metrics for assessing improvements in primary health care. Annual Review of Public Health 35:423–442.
Starr, P. 2017. The social transformation of American medicine: The rise of a sovereign profession and the making of a vast industry. New York: Basic Books.
Swankoski, K., A. O’Malley, and H. Tu. 2022. Independent evaluation of Comprehensive Primary Care Plus (CPC+): Fourth annual report. Princeton, NJ: Mathematica.
Trombley, M., M. McWilliams, B. Fout, and B. Morefield. 2022. ACO investment model produced savings, but the majority of participants exited when faced with downside risk. Health Affairs 41(1):138–146.
Trombley, M. J., B. Fout, S. Brodsky, J. M. McWilliams, D. J. Nyweide, and B. Morefield. 2019. Early effects of an accountable care organization model for underserved areas. New England Journal of Medicine 381(6):543–551.
Tung, E. L., E. M. Abramsohn, K. Boyd, J. A. Makelarski, D. G. Beiser, C. Chou, E. S. Huang, J. Ozik, C. Kaligotla, and S. T. Lindau. 2020. Impact of a low-intensity resource referral intervention on patients’ knowledge, beliefs, and use of community resources: Results from the CommunityRx trial. Journal of General Internal Medicine 35(3):815–823.
Unite US. 2018. Unite US launches patent-pending integration between EHRs and its social care coordination platform. https://uniteus.com/unite-us-launches-patent-pending-integration-between-ehrs-and-its-social-care-coordination-platform/ (accessed July 23, 2022).
VA (U.S. Department of Veterans Affairs). 2019. VA human-centered design and digital product management. https://www.oit.va.gov/tech-incubator/product-management (accessed July 23, 2022).
VA. 2020a. Digital transformation. VA enterprise roadmap. FY 2020–2026. https://www.ea.oit.va.gov/EAOIT/docs/Apr2021/FY-2020-2026-VA_OIT_Enterprise-Roadmap-20201211.pdf (accessed November 10, 2022).
VA. 2021a. Veterans Health Administration: Health Information Strategic Plan fiscal year 2022-2026. https://www.ea.oit.va.gov/EAOIT/docs/VHA-Health-Information-Strategic-Plan_FY22-26.pdf (accessed November 10, 2022).
VA. 2021b. Annual report on the steps taken to achieve full staffing capacity. https://www.va.gov/EMPLOYEE/docs/Section-505-Annual-Report-2021.pdf (accessed November 10, 2022).
VA. 2022a. History of IT at the VA. https://www.oit.va.gov/about/history.cfm (accessed November 10, 2022).
VA. 2022b. Mobile apps and online tools. https://www.va.gov/WHOLEHEALTH/veteran-resources/MobileApps-OnlineTools.asp (accessed July 22, 2022).
VA. 2022c. About VHA. https://www.va.gov/health/aboutvha.asp (accessed March 18, 2022).
VA. 2022d. Annual report on the steps taken to achieve full staffing capacity. https://www.va.gov/EMPLOYEE/docs/Section-505-Annual-Report-2022.pdf (accessed November 10, 2022).
VA OIG (Veterans Affairs Office of Inspector General). 2020. Improving VA and select community care health information exchanges. https://www.va.gov/oig/pubs/VAOIG-20-01129-220.pdf (accessed November 10, 2022).
Vaghefi, I., and B. Tulu. 2019. The continued use of mobile health apps: Insights from a longitudinal study. JMIR mHealth and uHealth 7(8):e12983.
Vaisson, G., T. Provencher, M. Dugas, M. E. Trottier, S. Chipenda Dansokho, H. Colquhoun, A. Fagerlin, A. M. C. Giguere, H. Hakim, L. Haslett, A. S. Hoffman, N. M. Ivers, A. S. Julien, F. Legare, J. S. Renaud, D. Stacey, R. J. Volk, and H. O. Witteman. 2021. User involvement in the design and development of patient decision aids and other personal health tools: A systematic review. Medical Decision Making 41(3):261–274.
Vest, J. R., and B. A. Kash. 2016. Differing strategies to meet information-sharing needs: Publicly supported community health information exchanges versus health systems’ enterprise health information exchanges. Milbank Quarterly 94(1):77–108.
Vogt, D., E. C. Taverna, Y. I. Nillni, B. Booth, D. F. Perkins, L. A. Copeland, E. P. Finley, F. A. Tyrell, and C. L. Gilman. 2019. Development and validation of a tool to assess military veterans’ status, functioning, and satisfaction with key aspects of their lives. Applied Psychology: Health and Well-Being 11(2):328–349.
Wager, E., K. Amin, C. Cox, and P. Hughes-Cromwick. 2021. What impact has the coronavirus pandemic had on health care employment? https://www.kff.org/coronavirus-covid-19/issue-brief/what-impact-has-the-coronavirus-pandemic-had-on-health-care-employment/ (accessed July 28, 2022).
Wakefield, M. 2014. Improving the health of the nation: HRSA’s mission to achieve health equity. Public Health Reports 129(1 Suppl 2):3–4.
Walker, J. M., and P. Carayon. 2009. From tasks to processes: The case for changing health information technology to improve health care. Health Affairs (Millwood) 28(2):467-477.
West, C. P., and D. M. Dupras. 2012. General medicine vs subspecialty career plans among internal medicine residents. JAMA 308(21):2241–2247.
West, C. P., L. N. Dyrbye, P. J. Erwin, and T. D. Shanafelt. 2016. Interventions to prevent and reduce physician burnout: A systematic review and meta-analysis. The Lancet 388(10057):2272–2281.
Willard-Grace, R., D. Hessler, E. Rogers, K. Dubé, T. Bodenheimer, and K. Grumbach. 2014. Team structure and culture are associated with lower burnout in primary care. Journal of the American Board of Family Medicine 27(2):229–238.
Yakovchenko, V., D. K. McInnes, B. A. Petrakis, C. Gillespie, J. M. Lipschitz, M. B. McCullough, L. Richardson, B. Vetter, and T. P. Hogan. 2021. Implementing automated text messaging for patient self-management in the Veterans Health Administration: Qualitative study applying the nonadoption, abandonment, scale-up, spread, and sustainability framework. JMIR mHealth and uHealth 9(11):e31037.
Zahid, A., J. K. Poulsen, R. Sharma, and S. C. Wingreen. 2021. A systematic review of emerging information technologies for sustainable data-centric health-care. International Journal of Medical Informatics 149:104420.
Zhang, H., D. W. Cowling, J. M. Graham, and E. Taylor. 2019. Five-year impact of a commercial accountable care organization on health care spending, utilization, and quality of care. Medical Care 57(11):845–854.
Zhang, H., D. W. Cowling, J. M. Graham, and E. Taylor. 2021. Impact of a commercial accountable care organization on prescription drugs. Health Services Research 56(4):592–603.
Zulman, D. M., E. P. Wong, C. Slightam, A. Gregory, J. C. Jacobs, R. Kimerling, D. M. Blonigen, J. Peters, and L. Heyworth. 2019. Making connections: Nationwide implementation of video telehealth tablets to address access barriers in veterans. JAMIA Open 2(3):323–329.
























































