3
Approaches to Closing the Gaps
OVERVIEW
Efforts to close the gaps among research, treatment, and policy traditionally focus on education, training, and/or dissemination of information within each separate arena. Even when such activities are effective, they have the potential to change only one group. Thus they generally fall far short of producing systemic change. Changing the system (as Figure 3.1 is meant to illustrate) will require the three groups working together to ask and answer the right questions and to jointly commit to implementation. Consequently, while this report proposes changes within each area, it also proposes joint activities that are needed to produce systemic changes.
Other areas of medical care have developed strategies to facilitate an integration of treatment, research, and policy. These include models for technology transfer, financial and other incentives to encourage organizational change, as well as methods to develop consensus on evidence-based practices and promote their use. The committee also found examples of collaboration that included the development of infrastructures and promoted trust-building between researchers and providers. Based on findings from the committee's workshops, site visits, briefings, and review of the
This chapter was edited by James L. Sorensen with contributions by Kathleen T. Brady,Thomas Crowley, Emily Jean Hauenstein, A. Thomas McLellan, and Steven M. Mirin.
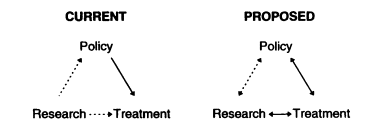
FIGURE 3.1 Need for bidirectional communication.
literature, several models are described here and in the following chapters that could help to bridge the gaps among stakeholders in this field:
TECHNOLOGY TRANSFER MODELS
The knowledge base on technology transfer has grown rapidly in the last fifteen years. By one estimate the citations in this field exceeded 10,000 by 1995.1 Experts make several distinctions that are useful in considering how to close the gaps between research, treatment, and policy in the drug-abuse area. They distinguish between technology transfer that is "hard" (e.g., equipment) and "soft" (e.g., counseling methods), and between technologies that are "high" (requiring substantial capital) and "low" (requiring relatively little investment). They differentiate between ''embodied" technologies (involving a physical entity like a new drug) and "disembodied" technologies (e.g., a new counseling procedure) (Backer, 1991). Technology experts also distinguish between "information dissemination" activities (e.g., information clearinghouses) and "knowledge utilization" activities that provide assistance in adoption efforts after information is available.
A recent Institute of Medicine report (IOM, 1994) makes another use-
ful distinction when the goal is bidirectional communication, as it is in this study. In Reducing the Risks for Mental Disorders, the IOM committee used the term "knowledge exchange" because it emphasizes the need for two-way communication, including feedback. This report will do the same. By contrast, the term "dissemination" has a connotation of directionality, and is used when only a one-way flow of information is implied. Studies in the 1960s and 1970s established that information dissemination alone is usually insufficient to stimulate change in individuals or in organizations. Studies in the 1970s and 1980s explored more active methods of promoting information utilization and developed strategies to aid that end. Recent work has been concerned with consolidating these principles into programmatic strategies (Backer et al., 1995).
The National Institute on Drug Abuse and the Center for Substance Abuse Treatment both have technology transfer programs which include knowledge exchange as well as dissemination activities. NIDA dissemination tools include videotapes, program assessment packages, and clinical reports (see for example NIDA [1993]). Treatment topics (e.g., relapse prevention, methadone, and special population treatment issues) are addressed in videotapes. The NIDA web site (http://www.nida.nih.gov) includes "NIDA capsules," which describe the effect of individual drugs, extent of current use by age groups, and new research findings (see Appendix G). NIDA Infofax (1-888-NIH-NIDA) provides quick access to science-based facts on drug abuse and addiction. NIDA is placing increasing emphasis on knowledge exchange activities. The goal of the NIDA Treatment Initiative is to improve the quality of drug addiction treatment through reciprocal exchanges of ideas and information among the research, treatment and policy communities, and the public; and to stimulate research in areas of treatment most relevant to the public health. Treatment Initiative activities include national conferences on research and practice, and online town meetings to bring the latest drug abuse research findings to communities and to receive feedback about community needs (Leshner, 1997; NIDA, 1996).
CSAT produces a technical Treatment Assistance Publication Series (TAPS) that includes detailed clinical guidelines for such clinical activities as relapse prevention. CSAT by FAX is a one-page newsletter featuring recent developments in treatment (see Appendix G). It is faxed to all treatment programs that receive any CSAT funding and was regularly mentioned by providers participating in the committee workshops and site visits as something they valued. The CSAT dissemination program includes a Treatment Improvement Protocol (TIPS) series that covers a wide variety of treatment topics ranging from infectious disease screening to drug specific treatment recommendations (see Appendix H). Providers attending the workshops were familiar with the TIPS publications but considered their
length and lack of a standard format to be a barrier in clinic use. An evaluation of this program is currently under way. A CSAT knowledge exchange activity is the Treatment Improvement Exchange (TIE) program to promote information exchange between CSAT staff and state and local alcohol and drug abuse agencies. TIPS and CSAT by Fax are both available on the Treatment Improvement Exchange. TIE is accessible via the CSAT web site (http://www.samhsa.gov/csat/csat.htm) or directly (http://www.treatment.org). While workshop participants who use the Internet appreciated this availability, it was evident that a significant number of providers still do not have effective access to this resource.
ORGANIZATIONAL CHANGE MODELS
An obvious goal of any organization is to maintain its viability. Organizational survival depends on the ability to provide a service or product that someone will buy or support. Increasingly, organizations must anticipate market forces and be able to accommodate rapid changes in their environment. Health care organizations, particularly those that are not-for-profit, traditionally have been somewhat sheltered from severe environmental and market forces. However, recent rapid changes in the financing of health care, including behavioral health care, are affecting community-based drug treatment providers.
As organization size increases, jobs within the organization become more differentiated. The workforce tends to be more stable because larger organizations are more likely to offer full-time employment, benefits, and other employee incentives. Organizational operations become formalized and may include specific procedures for innovation and implementation of new programs. Larger organizations are more likely to have adequate technology and other resources to sustain the extra work efforts that go into the adoption and implementation of new programs.
Many health care organizations have been unable to accommodate to a rapidly changing health care environment and have failed. This is particularly true of small to medium-sized mental health and drug abuse services that are poorly financed compared with organizations that provide mainstream health services. As a consequence, managers of CBOs, especially those that are small in size, focus primarily on maintaining organizational viability. This focus calls for a conservative organizational culture, a trim work force, and the ability to deliver a competitive product. The focus also stimulates attention to health care financing and other environmental changes that affect the resource base of the organization. In this climate, investing in innovation makes organizational sense only if it promotes organizational survival.
Not all organizations can support the kind of innovation necessary to
implement the evidenced-based practice guidelines discussed below, for instance. For organizations with appropriate resources, however, being an early adopter of research findings may facilitate recruiting and maintaining a satisfied, high-quality workforce. Even when the innovation supports important organizational goals, innovative programming requires managerial support, adequate financial and human resources, and an organizational culture that values scientifically based practice, problem solving, and creativity (Crump et al., 1996). It is not only small CBOs that are finding it hard to meet these tests.2 A description of the attributes associated with successful innovation is shown in Box 3.1.
The explicit goals of the organization may support innovation in treatment, but the organizational culture affects its outcome. Organizational culture has been described as the pattern of behaviors developed by groups to solve work-related problems and survive in their jobs (Coeling and Simms, 1993). It is manifested in the organization's beliefs and values, in its normative structure, and through artifacts or symbols (Seago, 1996). It is within this culture that the implicit goals of the organization take root. The culture's strength is determined by the degree of consensus among all levels of workers about which norms dominate and prevail in the actions of the organization. A strong organizational culture among the staff workers which is incongruent with management can defeat management efforts to introduce change (Nystrom, 1993).
Successful adoption of research findings in CBOs depends on careful matching of organizational characteristics, culture, and stages of development. This is discussed further in Chapter 4. Several authors have described an orderly process for adopting new treatments into CBOs when the implementation requires significant change within the organization (Altman, 1995; Nutbeam, 1996; Orlandi, 1996).
|
BOX 3.1 Attributes Associated With Innovations Likely to Be Implemented |
PRACTICE GUIDELINES AND SCORECARDS IN ADDICTIONS TREATMENT
The development of practice guidelines might help close the gap among the three segments of the drug abuse field, as well as improve clinical outcomes and enhance the credibility of caregivers. Both payers and policymakers have voiced skepticism about the efficacy of treatment for substance use disorders. In part, this skepticism is based on anecdotal experience, along with biases rooted in stigma and a history of perceived abuses of the reimbursement system by some providers. To some extent these same barriers operate at the interface between the substance treatment community and the rest of health care system.
Guidelines are relatively new in this field. The American Society of Addiction Medicine (ASAM) has published placement criteria, and the American Psychiatric Association (APA) has published comprehensive practice guidelines for this patient population (APA, 1996). The psychiatric practice guidelines are based on review and synthesis of the currently available treatment literature, complimented where appropriate by the experience of a group of skilled clinician reviewers. Sequential drafts of the guidelines were reviewed by a national sample of individual clinicians and researchers, as well as numerous professional organizations and governmental agencies in the addictions field.
The psychiatric practice guidelines include principles of treatment applicable to all forms of substance use disorder, as well as sections on the assessment and management of patients with alcohol, cocaine, and opioid related disorders. They provide a framework for choosing among treatment
options and make specific recommendations wherever possible, based on the strength of available research findings as well as the perceived degree of clinical consensus among practicing clinicians. Treatments that have not been adequately tested in well-controlled trials, or treatments where there are conflicting reports about efficacy but which are consistent with expert opinion and generally accepted treatment principles, are recommended with a lower level of clinical confidence or alternatively, recommended to be applied only in specific clinical circumstances. These guidelines leave the ultimate judgment to the clinician, based on data presented by the patient and on the diagnostic and treatment options available. It is anticipated that the guidelines will be revised every three to five years to incorporate emerging research and clinical experience.
Despite the potential benefits of incorporating advances in clinical research into clinical care delivery, many barriers exist to the successful dissemination and adoption of evidence-based practice guidelines within the drug abuse treatment community. A number of factors may contribute to this situation. Chief among them is the heterogeneity in the background, training, and clinical perspectives of clinicians practicing within the addiction treatment community. With the notable exception of methadone maintenance, the relative paucity of clinically effective, medically based treatments for this patient population has helped foster a treatment culture in which many treatment approaches, including self-help and therapeutic communities, have flourished. Heavily influenced by both the experience and world views of recovering drug abusers, this segment of the treatment system has embraced a treatment philosophy and approaches to care that depend more on the motivational power of group support and on spiritual beliefs, than on methodologically sound studies of treatment effectiveness. In this context, guidelines based on data from clinical research, particularly research carried out in medical settings, may be seen as undermining treatment approaches less amenable to study by scientific methods.
Both the American Medical Association (Office of Quality Assurance, 1996) and the Institute of Medicine (IOM, 1992) have developed principles for practice guideline development and implementation. Not surprisingly, AMA recommends that guidelines be developed by, or in conjunction with, physician organizations. In addition, AMA recommends that guideline dissemination be coupled with a plan for measuring their impact on short- and long-term treatment outcome. Testing is important for guidelines in any field because of the potential for obtaining useful new information as well as avoiding unintended bad effects (Weingarten, 1997).
The IOM report on Clinical Guidelines for Practice (1992) recommended that guidelines should evolve as a result of a multidisciplinary process "that includes participation by representatives of key affected groups" who can identify, critically evaluate, and incorporate all important
clinical and scientific evidence into the guidelines. The latter seems particularly relevant in developing clinical guidelines in the drug abuse field, if the target audience (i.e., community-based treatment programs) is to view the guidelines as credible. Multidisciplinary participation maximizes the chances for addressing practical problems in their use. A recent report recommended that guidelines be accompanied by a timetable for scheduled review and revision (IOM, 1997).
A number of studies have demonstrated that merely publishing guidelines does not change the practice patterns of targeted clinicians, but that there are strategies which enhance the likelihood of this occurring (Greco and Eisenberg, 1993; Rogers, 1995b). Strategies that work include direct endorsement by respected professional associations and clinical "opinion leaders," coupled with teaching sessions under their aegis, and the incorporation of guidelines into training and continuing education programs, as well as self-assessment, certification, and recertification examinations. The use of practice guidelines by third party payers and managed care organizations to inform decision making on benefit utilization also enhances their dissemination and ultimate acceptance. Incorporating measures of dissemination and incorporating guideline use in HEDIS surveys and JCAHO standards would facilitate this goal. It will be necessary to devise strategies by which the acceptability and ultimate utility of practice guidelines in drug and alcohol abuse treatment can be measured.
Compared to practice guidelines that have been developing for more than a decade, the science of consumer scorecards in health care is in an early state (Hanes and Greenlick, 1998). However, the movement is growing and a useful purpose could be served in the development of scorecards providing information about community-based treatment programs. Included in such scorecards would be information from consumer satisfaction and quality of life surveys, as well as other data on short- and long-term treatment outcomes assessing the effectiveness of the treatment program.
CONSENSUS CONFERENCES AND EVIDENCE-BASED REVIEWS
Closely linked to practice guidelines are consensus conferences and the development of evidence-based reviews, two other mechanisms that are being widely tested in other areas of the health care delivery to reduce the communication gaps among research, practice, and policy segments. The experience of the Agency for Health Care Policy Research (AHCPR) can provide some guidance in this area. As the question of practice variation and inefficiency in the health care system became a major focus at AHCPR, their first approach was the creation of a set of Patient Outcome Research Teams (PORT) and the implementation of a guidelines development process within the Agency. Several PORTs were funded, each with a focus on
practice methods within a particular disease entity (Goldberg et al., 1994). The PORTs, studying practice in such areas as stroke, acute myocardial infarction, low-back pain, and knee replacement did some extraordinary work studying care in the various areas, and an extensive literature is emerging. The output from the PORTs was to fuel AHCPR's guideline development process.
The guideline development process did emerge and AHCPR became the official government agency creating guidelines in many important areas of clinical practice. But there were a variety of problems with the federal approach to guideline creation, including the evidence cited above that governmentally created guidelines was not the most effective way to influence clinical practice. Moreover, there was significant political fall-out from this process, including a move by one group of medical specialists to abolish AHCPR as a result of their unhappiness with the contents of a guideline. Cooler heads prevailed and the threat to the agency dissipated.
With experience came a rethinking of the guidelines/PORT model and AHCPR created a new model. The current thinking follows from the approaches discussed above, that guidelines are best created by sponsors closer to the actual clinical care, including managed care programs, medical specialty groups and the like. But the major impediment to guidelines creating is still the paucity of evidence reviews in many clinical areas. So AHCPR has now named twelve Evidence-Based Practice Centers to produce the evidence-based reviews intended to facilitate improvement in clinical practice. For the current status of this effort, see the AHCPR web site (http://www.ahcpr.gov). Further, AHCPR has created a national nomination process for assisting in determining priorities for the particular evidence-based reviews to be created. It is likely that a similar process would be extremely helpful in the area of substance abuse treatment.
Numerous impediments make it difficult for counselors, program managers, and state agency staff to sift through the research literature, critique it effectively, and select findings to implement in treatment. Techniques such as the consensus conference mechanism and the evidence-based reviews approach might begin to close the gap and to improve treatment, as well as to enhance the potential for broader use of treatment guidelines in drug abuse treatment. The first NIH Consensus Development Statement on drug abuse treatment is included as Appendix F and may also be found on the NIH Consensus Development Statement web site (http://consensus.nih.gov).
TOP-DOWN INCENTIVES MODELS
Workshop participants described a variety of "top-down" models, most of which could be fairly described as "money with strings" that would
require recipients to do something that the proposer viewed as salutary. It was clear, however, that many treatment providers believe that money with strings can make it harder for them to successfully compete in an increasingly difficult world, and when that was the case they rejected the concept.
The discussion and deferral of plans to link federal funding for substance abuse treatment to performance objectives under the Performance Partnership Grants (PPG) Program illustrates the problem. In the opinion of the National Association of State Alcohol and Drug Abuse Directors and a National Academy of Sciences panel, performance measures for public health, substance abuse, and mental health are not ready for prime time. Both concluded that the science of performance measurement and the data available to support such a link are major stumbling blocks (Gustafson and Sheehan, 1997).
Recognizing that data resources and measurement methods need improvement, the panel recommends that DHHS continue to work with states toward several infrastructure goals: developing common definitions and measurement methods; encouraging efficient development of data resources that support multiple public health, mental health, and substance abuse needs; incorporating state data priorities in national infrastructure development efforts; and promoting states' data collection and analytic capabilities.
Assessment of Performance Measures for Public Health, Substance Abuse, and Mental Health, Phase 1 Report; NRC (1997)
One top-down model that was discussed would have suggested changes in the incentives that currently are attached to the state block grant money used to support treatment programs in most communities. The block grant program has requirements that states pass on to service providers. For example, states are required to assure expenditures for services to pregnant and parenting women, to injection drug users, and to provide access to HIV and TB services for testing and medication (GAO, 1995). In order to meet the requirements, states may offer treatment programs additional funds to deliver new services and to serve consumers with specified characteristics (e.g., using injection drugs, caring for children). States could also use funding strings to promote collaborations among community-based organizations and research groups. The Department of Veterans Affairs approach includes a money-with-strings strategy (see Box 3.2).
The introduction of managed care into the drug abuse treatment field has produced a new, and particularly difficult, set of requirements. Most treatment providers already are quite concerned with the "strings" that come with managed care money, but the quick response by treatment pro-
|
BOX 3.2 The VA Model-Top-Down Incentives Model For the past seven years, the VA has used an in-service program of education and training to integrate research-based treatments into its approach to substance dependent patients. This program has included national meetings for program leaders, interactive video teleconferencing for presentation of curriculum materials, the development of Centers of Excellence in Substance Abuse Treatment and Education as national education resource centers, quarterly conference calls with program leaders across the country, and small meetings to introduce technical treatments such as LAAM. VA officials have learned that certain things do work in this process of change: Richard Suchinsky Associate Chief for Addictive Disorders, Department of Veterans Affairs Committee Workshop, July 27, 1997, Washington, DC |
grams to the requirements of managed care programs, albeit against their wishes and desires, is an example of the power of this approach.
Top-down incentives were viewed by the committee as a powerful approach, but also a dangerous one. Money with dumb strings can lead to inappropriate actions and services, of which the committee heard several examples. The committee did agree that when top-down models are proposed, it must be with careful consideration of the potential negative consequences.
MODELS THAT INCORPORATE TRUST-BUILDING EXPERIENCES
Knowledge exchange and the development of two-way communication between treatment personnel and researchers requires the development of trust. Trust takes time to develop. Trust between researchers and practitio-
ners builds over several years of shared experience in designing and carrying out service delivery research and is the key ingredient in establishing successful linkage between research and practice. Researchers who develop and implement interventions in the community need to design interventions that are useful to community systems after the formal phase of research ends. Thus in technology transfer it is essential to collaborate with the people who will need to live with the results of change and to foster effective long-term relationships between researchers and the community programs in which the research takes place (Altman, 1995).
Most drug abuse innovations involve procedural knowledge, such as treatment doses or behavioral change requirements, not hard technology such as a new medical device (Tenkasi and Mohrman, 1995). As a result, most innovations are not adopted literally. They are put into use through a process of ''contextual adaptation" that matches the innovation to the environment. This is a human process involving creative synthesis by workers, a process of "reinventing innovations" by modifying them to fit varying local circumstances (Rogers, 1995a). Personal contact can also be a key to the adoption of new technology. For example, approaches that involve personal contact were found to result in greater adoption of a job seekers' workshop in drug treatment programs than dissemination approaches that provided only written materials (Sorensen et al., 1988). Personal consultation was similarly important in the VA successes described to the committee.
Once an innovation has been targeted for program adoption it may be necessary to have a period of transition in which the program adapts the innovation to its own culture (Diamond, 1995). The rituals of bureaucracy, such as organizational missions, policy statements, staff meetings, and in-service training—which exist in part to provide a way of reducing anxiety—can also be used to stimulate the transfer of the targeted innovation.
It is probably impossible for those removed from the work to appreciate subtle differences in the work task. And so technology, defined broadly as the procedures and equipment we use, is always local.
Ann Lennarson Greer in Greer (1995), p. 329
However, the "pull" for making local adaptations to a treatment model must be balanced with a concern for maintaining the efficacy of the treatment. For example, there has been considerable research over 20 years on the replication of the Program of Assertive Community Treatment (PACT).
A review of the research on this comprehensive community-based service delivery model for the seriously mentally ill has shown that positive client outcomes are achieved when the original model is followed with regard to organization, staffing, and practice patterns. The reviewer concludes that not implementing the program fully and not providing the necessary staff training will jeopardize the ability of the program to assist clients in becoming fully functioning members of their communities (Allness, 1997).
Several experts have recommended that researchers gain a deeper understanding of the treatment programs they hope to influence (Kavanaugh, 1995). Sobell adapts business techniques and encourages behavioral scientists to get "close to the customer" in developing and fostering close working relationships (Sobell, 1996). Brown suggests that, at a minimum, to develop effective technology transfer, the innovator must obtain input from potential adopters about the relevance, clarity, credibility, and adaptability of an intervention (Brown, 1995). Trust-building experiences can include site visits, jointly sponsored seminars and staff development activities, and short-term exchanges of staff.
The building of successful research-treatment partnerships, which recognize the contribution of both the research and treatment communities, is one way to build trust. Partnerships can be successfully organized with the community group as senior partner, the academic group as senior partner, or in a balanced partnership (Mittelmark, 1990). The committee heard from several administrators of community-based drug treatment programs who emphasized the need to work for a collaborative relationship. One pointed out that who takes the lead in a proposal depends on the funding agency: if it is SAMHSA, the CBO leads, if the funder is NIH, the leader is the university-based researcher.
These issues are not unique to drug abuse. In the area of cancer treatment, 80 percent of care is provided in the community and the quality of care can be quite variable. In an attempt to improve the quality of care provided in the local communities, various organizations have issued guidelines for effective treatment procedures, but like the Treatment Improvement Protocols in the drug abuse area, writing a guideline does not guarantee that providers will comply (Czaja et al., 1997; Ford et al., 1987; Klabunde et al., 1997).
The National Cancer Institute's Community Clinical Oncology Program (CCOP) provides a model for researchers and clinicians seeking to collaborate (Cobau, 1994; Kaluzny et al., 1993, 1996). To better integrate policy, research, and treatment and thereby assure access to improved care within local communities, CCOP involves primary care physicians and oncologists in the conduct and management of clinical trials, in cooperation with NCI-funded centers and clinical cooperative groups. CCOP has become a valuable resource to NCI for performance of a wide variety of
investigational treatment, prevention, and control activities. The potential for adaptation of this model to the drug abuse treatment field is discussed in Chapter 5, with a recommendation following in Chapter 6. Chapter 5 includes other collaboration models as well. The practice-based research networks described in the next chapter (see Box 4.3) provides an alternative model developed in several medical specialties to involve clinicians in the development of knowledge to guide their practice.
Another trust-building model, the Agricultural Extension Service, has had a far-reaching impact on U.S. farm productivity in the past 50 years. As described to the committee by Everett Rogers of the University of New Mexico School of Communication, the agriculture extension model consists of a set of assumptions, principles, and organizational structures for diffusing the results of agricultural research to farmers. The success of the model is based on farmer participation in identifying local needs, serving on county-level committees to develop the research agenda, providing test plots for the agricultural research, and providing feedback to the state university researchers on the applicability of the results. It has built-in reward systems for farmers and researchers to encourage utilization of the new information. Agriculture extension specialists are in close social, political, and spatial contact with these county research committees and with agricultural researchers, which allows them to facilitate linking research-based knowledge to farmer problems. This model, funded by the U.S. Department of Agriculture, worked particularly well in diffusing agricultural production technology to family farmers in the early development of scientific farming (Rogers, 1995a; Rogers et al., 1976).
SUMMARY
Many of the approaches to closing the gap rely on infrastructure changes within both treatment and research organizations. The next chapter focuses on the issue from the perspective of the treatment programs, the following one focuses on changes needed within the research enterprise. Even when effectiveness studies document that a treatment can be successfully implemented in a clinical setting, technology transfer to local drug abuse treatment centers is difficult. New treatments typically are adopted and implemented by trained staff, who may be in short supply in many CBOs. Challenges in the final stage of treatment transfer include training staff in delivering the new treatment, changing attitudes of the providers so they embrace the new treatment, and providing evidence that the new treatment is effective in improving the health status of drug abusers.
Each of these components of training must be planned, systematically delivered, and protective of the fidelity of the treatment. Many have suggested that the transfer of new treatment knowledge occurs best in the
context of a long-term relationship between a researcher and the sponsoring CBO (Nurco and Hanlon, 1996). In some places well trained and respected clinicians who have established trust with community treatment colleagues may be the best to transfer new knowledge. In either case, a collaborative model of community-based research appears to be the most appropriate model to facilitate the design of treatment research that is relevant to the CBO's values and mission, sensitive to its fiscal and human resources, and respectful of its culture and that of the population it serves. The conduct of community-based research is an intensely interpersonal enterprise, and these relationships must be cultivated at different levels of the organization, with community residents, and often with members of other agencies related to the CBO.
In developing a typology linking specific treatment strategies with amenable research approaches, it becomes clear that community-based research will be most likely to happen within the context of a structured collaboration between CBOs and researchers. This analysis favors approaches designed to develop such long-term collaborations, within which both investigators and providers become stakeholders and, consequently, become committed to the appropriate implementation of successful innovations created and tested within the collaboration.
REFERENCES
Allness DJ. 1997. The Program of Assertive Community Treatment (PACT): The model and its replication. New Directions for Mental Health Services 74:17-26.
Altman DG. 1995. Sustaining interventions in community systems: On the relationship between researchers and communities. Health Psychology 14:526-536.
APA (American Psychiatric Association). 1996. American Psychiatric Association: Practice Guidelines. Washington, DC: American Psychiatric Association Press.
Backer TE. 1991. Drug Abuse Technology Transfer. Rockville, MD: National Institute on Drug Abuse.
Backer TE, Liberman RP, Kuehnel T. 1986. Dissemination and adoption of innovative psychosocial interventions. Journal of Consulting and Clinical Psychology 1:111-118.
Backer TE, David SL, Soucy G. 1995. Introduction. In: Backer TE, David SL, Soucy G eds. Reviewing the Behavioral Science Knowledge Base on Technology Transfer. NIDA Research Monograph 155. Rockville, MD: National Institute on Drug Abuse. Pp. 1-20.
Brown BS. 1995. Reducing impediments to technology transfer in drug abuse. In: Backer TE, David SL, Soucy G eds. Reviewing the behavioral science knowledge base on technology transfer. NIDA Research Monograph 155. Rockville, MD: National Institute on Drug Abuse. Pp. 169-185.
Cobau CD. 1994. Clinical trials in the community: The Community Clinical Oncology Program experience. Cancer Supplement 74(9):2694-2700.
Coeling HVE, Simms LM. 1993. Facilitating innovation at the nursing unit level through cultural assessment: Part 1, how to keep management ideas from falling on deaf ears . Journal of Nursing Administration 23(4):46-53.
Crump CE, Earp JA, Kozma CM, Hertz-Picciotto I. 1996. Effect of organization-level variables on differential employee participation in 10 federal worksite health promotion programs. Health Education Quarterly 23(2):204-223.
Czaja R et al. 1997. Preferences of community physicians for cancer screening guidelines. Annals of Internal Medicine 120(7):602-608.
Diamond MA. 1995. Organizational change as a human process, not technique. In: Backer TE, David SL, Soucy G eds. Reviewing the behavioral science knowledge base on technology transfer. NIDA Research Monograph 155. Rockville, MD: National Institute on Drug Abuse. Pp. 119-132.
Ford L et al. 1987. The effects of patient management guidelines on physician practice patterns: The Community Hospital Oncology Program. Journal of Clinical Oncology 5:504-511.
GAO (General Accounting Office). 1995. Block Grants: Characteristics, Experience and Lessons Learned. GAO/HEHS-95-74. Washington, DC: General Accounting Office.
Glaser EM, Abelson HH, Garrison KN. 1983. Putting Knowledge to Use: Facilitating the Diffusion of Knowledge and the Implementation of Planned Change. San Francisco, CA: Jossey-Bass.
Goldberg HI, Cummings MA, Steinberg EP, Ricci EM, ST, Soumerai SB, Mittman BS, Eisenberg J, Heck DA, Kaplan S, Kenzora JE, Vargus AM, Mulley AG, Rimer BK. 1994. Deliberations on the dissemination of PORT products: Translating research findings into improved patient outcomes. Medical Care 32(7):JS90-JS110.
Greco PJ, Eisenberg JM. 1993. Changing physician practices. New England Journal of Medicine 329:1271-1273.
Greer AL. 1995. The shape of resistance . . . the shapers of change. Journal on Quality Improvement 21(7):328-332.
Gustafson JS, Sheehan K. 1997. Linking substance abuse treatment funding to performance measurement will take time. Connection April:1-2, 5.
Hanes PP, Greenlick MR. 1998. The alchemy of accountability: Science and art of consumer scorecards . In: Hanes PP, Greenlick MR eds. Grading Health Care: The Science and Art of Developing Consumer Scorecards. San Francisco: Jossey-Bass. Pp. 119-129.
IOM (Institute of Medicine). 1992. Guidelines for Clinical Practice from Development to Use. Washington, DC: National Academy Press.
IOM. 1994. Reducing Risks for Mental Disorders: Frontiers for Preventive Intervention Research. Washington, DC: National Academy Press.
IOM. 1997. Managing Managed Care: Quality Improvement in Behavioral Health. Washington, DC: National Academy Press.
Kaluzny AD, Lacey LM, Warnecke R, Hynes DM, Morrissey J, Ford L, Sondik E. 1993. Predicting the performance of a strategic alliance: An analysis of the Community Clinical Oncology Program. Health Services Research 28(2):159-182.
Kaluzny AD, Warnecke RB et al. 1996. Managing a Health Care Alliance: Improving Community Cancer Care. San Francisco, CA: Jossey-Bass.
Kavanaugh KH. 1995. Collaboration and diversity in technology transfer. In: Backer TE, David SL, Soucy G eds. Reviewing the Behavioral Science Knowledge Base on Technology Transfer. NIDA Research Monograph 155. Rockville, MD: National Institute on Drug Abuse. Pp. 42-64.
Klabunde C, O'Malley M, Kaluzny A. 1997. Physicians' reactions to changing recommendations for mammography screening. American Journal of Preventive Medicine 13(6):432-438.
Leshner Al. 1997. Taking drug abuse research to the community. NIDA Notes 12(1):3-4.
Mittelmark MB. 1990. Balancing the requirements of research and the needs of communities. In: Bracht N ed. Health Promotion at the Community Level. New York: Sage Publications.
NIDA (National Institute on Drug Abuse). 1993. How Good is Your Drug Abuse Treatment Program? A Guide to Evaluation. NIH Pub. No. 93-3609. Rockville, MD: National Institute on Drug Abuse.
NIDA. 1996. National Institute on Drug Abuse Community-Based HIV Prevention Research. Rockville, MD: National Institute on Drug Abuse.
NRC (National Research Council). 1997. Assessment of Performance Measures for Public Health, Substance Abuse, and Mental Health. Washington, DC: National Academy Press.
Nutbeam D. 1996. Improving the fit between research and practice in health promotion: Overcoming structural barriers. Canadian Journal of Public Health 87(Suppl 2):S18-S23.
Nurco DN, Hanlon TE. 1996. The linking of research and service. Substance Use and Misuse 31(8):1059-1062.
Nystrom PC. 1993. Organizational cultures, strategies, and commitments in health care organizations. Health Care Management Review 18(1):43-49.
Office of Quality Assurance, American Medical Association. 1996. Attributes to Grade the Development of Practice Parameters. Chicago, IL: American Medical Association.
Orlandi MA. 1996. Health promotion technology transfer: Organizational perspectives. Canadian Journal of Public Health 87(Suppl. 2):S28-S33.
Rogers EM. 1995a. Diffusion of Innovations. 4th Edition. New York: Free Press.
Rogers EM. 1995b. Lessons for guidelines from the diffusion of innovations. Joint Commission Journal on Quality Improvement 21(7):324-328.
Rogers EM et al. 1976. Extending the Agricultural Extension Model. Preliminary Draft. Washington, D.C.: National Science Foundation.
Seago JA. 1996. Work group culture, stress, and hostility. Correlations with organizational outcomes. Journal of Nursing Administration 26(6):39-47.
Sobell LC. 1996. Bridging the gap between scientists and practitioners: The challenge before us. Behavior Therapy 27:297-320.
Sorensen JL, Hall SM, Loeb P, Allen T, Glaser EM, Greenberg PD. 1988. Dissemination of a job seekers' workshop to drug treatment programs. Behavior Therapy 19:143-155.
Tenkasi RV, Mohrman SA. 1995. Technology transfer as collaborative learning. In: Backer TE, David SL, Soucy G eds. Reviewing the Behavioral Science Knowledge Base on Technology Transfer. NIDA Research Monograph 155. Rockville, MD: National Institute on Drug Abuse. Pp. 262-279.
Weingarten S. 1997. Practice guidelines and prediction rules should be subject to careful clinical testing. Journal of the American Medical Association 277(24):1977-1978.

















