2
Instrumentation
LOGISTICS FOR USING THE INTERNATIONAL SPACE STATION AS A BIOTECHNOLOGY RESEARCH PLATFORM
The International Space Station (ISS) is currently under construction; assembly is scheduled to be complete in 2005. However, NASA plans to begin research on the facility as early as 2000, using equipment that has been flown on the shuttle and that can be temporarily installed in modules of the ISS as they are completed. As the ISS grows and more station-specific hardware is ready, the research program will expand and more permanent instrumentation will be fitted into the ISS. The present schedule calls for a specialized biotechnology facility to be one of the last units installed on the ISS in 2005, so until that date, the hardware for protein crystal growth and cell science research in space will be fitted into EXPRESS racks in whatever laboratory modules have been completed.1 A more detailed outline of the schedule for research on the ISS is included in Appendix A.
Although equipment from the space shuttle will be used, temporarily, on the ISS, there are several key differences between the logistics of experiments on the shuttle and on the ISS. First are the time scales. On the shuttle, experiments lasted no more than 2 weeks at a stretch. On the ISS, the microgravity environment will be available almost indefinitely. Current plans call for three or four shuttle trips to the ISS each year for the purpose of transporting new experiments up and returning samples and results from completed work.2 Accordingly, NASA anticipates that research efforts will be organized into approximately 100-day groupings, known as “increments,” the time between shuttle flights. Assuming adequate storage space for multiple samples, many more investigations can be conducted in an increment than have been conducted on past shuttle flights with science missions. However, since physical items, such as samples or film from cameras, will not be returned to Earth until the shuttle trip at the end of each increment, alternative methods for communicating results to researchers in a timely fashion—such as real-time computer links to data acquisition software on orbit or ground-based control of experiments—will become critical.
The second difference between science on the shuttles and on the ISS is the reduction in crew involvement in experiments. The total number of crew members will not be much different, but the amount of research will have greatly expanded. While the astronaut pool from which crew members will be selected includes people with a
|
1 |
EXPRESS racks are skeleton structures that can provide basic resources like power to a variety of modular experiments. Eight separate units, called middeck locker equivalents (MLEs), fit into each EXPRESS rack. |
|
2 |
More shuttle trips may occur for resupply and construction purposes. |
variety of scientific backgrounds (biologists, physicists, engineers, etc.), it is unlikely that the crew expertise in a given increment will match the large variety of experiments taking place. In addition, through 2005, the primary occupation of all crew members will be ISS assembly. In this situation, any technical innovations that permit the automation of routine tasks or the robotic manipulation of experiments will greatly increase efficiency. The task group also notes that another way to maximize the scientific output of the ISS-based research would be to allow investigators to participate directly in experiments. Scientists could spend time on the ISS as short-term residents or travel with shuttle crews during routine supply and transfer missions. This approach might not be immediately realistic owing to the stringent demands on crew time and expertise during ISS construction. However, as the assembly phase approaches completion, the demands on personnel time should become more flexible, allowing greater crew involvement in research projects and the possibility of having nonastronaut scientists aboard the ISS.
PROTEIN CRYSTAL GROWTH
Since the beginning of the NASA protein crystal growth program in 1985, a variety of equipment has been used to grow and observe crystals in the microgravity environment. Useful and innovative hardware development on systems for the ISS continues today. A complete description of the equipment that is or will soon be available is provided in Appendix A. Options for investigators range from liquid-nitrogen-cooled dewars capable of holding large numbers of samples but providing minimal environmental control or observation to refrigerated trays aligned with Michleson-Morley phase-shift interferometers. The task group was particularly impressed with the prototype of the X-ray Crystallography Facility (XCF), which can grow and cryopreserve a reasonable number of samples as well as provide important monitoring capabilities, such as video feedback and X-ray diffraction data. While this approach of monitored growth appeared to the task group very promising, it is important to recognize that in some situations large numbers of samples might be an effective alternative to a few carefully chosen and observed samples.
The Hardware Development Process
It is clear that NASA's microgravity crystallization program and its associated crystallography hardware are not yet mature. However, it is important that researchers interested in exploiting the microgravity environment on the ISS have access to hardware that is state of the art and the most efficient available. To achieve that goal, collaboration and communication between the various laboratories involved in hardware development should be established. Many of the key pieces of hardware so far have been innovated by external investigators, so it is not necessary to have centralized hardware development within NASA. Multiple developers will encourage variety and creativity, while preventing NASA from getting locked in to a single hardware approach. However, the efforts of hardware developers must be coordinated and communication between them improved to ensure that different programs are not producing instruments with duplicative capabilities and that technological advances are quickly shared and integrated into all equipment where appropriate. In addition, since the modular structure of ISS racks will permit instruments from multiple hardware designers to coexist, it is important that the systems be compatible to allow experimenters to take advantage of the full variety of equipment (e.g., samples could be grown in one type of hardware yet monitored by another developer 's system). Finally, the most vital step in hardware development is for the research community at large to have input into the instrumentation development process, as cutting-edge science problems can drive the development of innovative new technologies. An example of the critical nature of the collaboration between science and engineering for effective use of large facilities can be seen in how synchrotron sources and the instrumentation for beam lines have evolved most successfully when bureaucratic structures are not allowed to divorce scientific goals from the work on the technology needed to explore those goals.
The most important characteristic of NASA-sponsored hardware development should be flexibility. If a variety of equipment types are available, investigators, with the help of hardware developers and NASA staff, can match their experiments to the instruments best suited to their needs and goals. A modular approach should be emphasized so that individual systems can be upgraded as technology advances. Finally, NASA needs to be prepared to abandon completed hardware or hardware under development if it becomes clear that better systems or
new technologies are available. The evaluation of such hardware should be driven by scientific criteria—“better” hardware is that which is more effective in growing crystals and answering key scientific questions.
Recommendation: The efforts of external hardware developers should be coordinated to ensure that instruments are compatible, to prevent duplication of efforts, to ensure that technical innovations are shared, and to facilitate input from the scientific community in defining the goals and capabilities of protein crystal growth equipment for the ISS. NASA must also be prepared to discontinue development projects that do not use cutting-edge technologies or that are out of tune with the most current scientific goals.
A significant factor affecting equipment development is the instability in the budget for the ISS. Several hardware design and engineering projects have been put on hold as a result of fiscal uncertainties or shortages. If this situation continues, the greatest long-term negative consequence will be the reduction or elimination of a high-quality user community for the NASA protein crystal growth program. This will occur in part because continual uncertainty is demoralizing and discouraging; researchers will avoid a program that cannot offer reliably scheduled research or funding opportunities. In addition, if money is repeatedly siphoned off from the hardware development work, the quality of the equipment on the ISS will be significantly below that of the cutting-edge hardware available on the ground, and researchers will not be interested in using the outdated equipment or willing to entrust precious samples to it. If support originally designated for scientific instrumentation or research becomes a convenient contingency fund for ISS construction, NASA will send a clear message that science on the ISS has a low priority and will alienate the research community even further.
Key Characteristics of Protein Crystal Growth Hardware on the ISS
Designing and performing an experiment for the ISS will be an arduous task for researchers, especially those unfamiliar with the constraints imposed when working in a microgravity environment. The equipment developed by and for NASA should aim to provide a high level of control over samples, equipment, and procedures. On the ISS, crew time will be limited, and the human access to samples and the feedback to the investigators via shuttle trips will be infrequent (once every 3 or 4 months). In this environment, two related hardware characteristics will be essential: automation and ground-based control. Minimizing the number of crew actions required will allow flexibility in scheduling and increase the options open to investigators. In addition, if the principal investigators are able to make decisions about experimental parameters (temperature, time for growth, which samples to preserve or examine) and then are able to adjust experiments in real time from Earth (as they would in their own laboratories), the research produced in each experiment will be of higher quality, and the NASA program will become more attractive. Therefore, hardware development efforts should emphasize the importance of automation, monitoring, real-time feedback, telemanagement, and sample recovery (via mounting and freezing). NASA-sponsored efforts in these areas could be accelerated by increased interactions with engineers developing instrumentation for synchrotron beam lines. This community is interested in very similar issues, including remote control of diffraction experiments, better measures of crystal quality, automation of crystal selection, freezing and mounting, and the rapid characterization of many crystals via routine experiments. Sharing experiences and technologies would benefit both communities.
Effective analysis, preservation, and reentry of promising crystal samples are especially necessary given the key role synchrotrons are playing in protein structure determination. Hardware designers should be sensitive to how synchrotrons define the needs of crystallographers; standard crystal sizes accommodated in ISS equipment should reflect advances in the brillance of synchrotron sources, and techniques for environmental control (pH, temperature, vibrations, etc.), freezing methods, and sample reentry should be fine-tuned to maximize crystal quality. The production and safe return of high-quality crystals from the ISS are crucial if the NASA program is to attract researchers interested in important and challenging biological problems.
While most successful microgravity experiments will probably conclude with diffraction studies at a synchrotron, it is not NASA's responsibility to arrange or guarantee this step. Building a synchrotron beam line is expensive and would not be the most efficient use of NASA's scarce resources. In addition, there are strong user
programs at the many beam lines throughout the United States, where user opportunities are allocated by peer review processes. Assuming that NASA's peer review process is selecting the most scientifically rigorous and interesting projects, successful crystallization should enable researchers to compete effectively for the necessary beam time, and success in this extra layer of peer review should further validate the NASA program within the scientific community. Therefore, NASA should not contribute to the construction of a new synchrotron beam line and should not apply for a block grant of time to be reserved for NASA investigators.
The X-ray Crystallography Facility
The X-ray Crystallography Facility (XCF) is a multipurpose facility designed to provide and coordinate all elements of protein crystal growth experiments on the ISS: sample growth, monitoring, mounting, freezing, and X-ray diffraction. A module for the growth phase is designed to house vapor diffusion experiments. The visualization unit uses magnified still photographs of samples that have completed growth to determine whether the resulting crystals are worth preserving. The Crystal Preparation Prime Item (CPPI) is a robotic system that mounts the crystals on hair loops for cryopreservation or on hair loops inside a capillary, unfrozen. Finally, the X-ray diffraction module employs a low-power (24 W) X-ray source and has a maximum resolution of 1.1 Å. The various modules are controlled remotely from the ground; crew time is required to move samples from unit to unit. The various components of XCF are described more fully in Appendix A. The growth phase of research occurs in modular units located in an EXPRESS rack reserved for this purpose, while the CPPI and the X-ray diffraction instrumentation are located in a separate, specially designed rack (see Figure 2.1).
The task group was impressed by the XCF, by the robotics, the remote control, and the range of experimental capabilities provided. The automation is vital on the ISS, where crew time for scientific experiments will be limited and crew expertise may not match up with the experimental tasks. In addition, the ability to observe results in real time and choose the next experimental steps accordingly is attractive, given the length of time between shuttle trips and the limited opportunities for reflight of an experiment. The X-ray diffraction module provides valuable information about whether a given crystal will diffract —this real-time feedback is key to making decisions about the success or failure of a particular crystallization experiment and will help allocate scarce freezer resources by ensuring that the most promising crystals are preserved and returned to Earth. Any diffraction data gained from the ISS X-ray system will be a bonus. The task group offers one specific piece of technical advice: a step in which the sample is swept through a cryopreservative before freezing should be added to the CPPI module; this addition should greatly increase the success rate for freezing.
Finding: Automation, monitoring, real-time feedback, telemanagement, and sample recovery (via mounting and freezing) will be vital for successful protein crystal growth experiments on the ISS. The XCF, through its use of robotics and a variety of experimental and observational capabilities, provides many of the tools researchers need to take full advantage of the microgravity environment.
The task group was impressed by the ground-based control capabilities of the XCF. Current plans call for the technical equipment that performs remote experimental management to be situated at the University of Alabama at Birmingham, rather than at one of the NASA centers. This location is appropriate in the near term, as the expertise of the engineers who developed the system will be invaluable for troubleshooting when the XCF is installed on the ISS, as well as for maintenance and possible future improvements once the system is up and running. It is also important that NASA personnel be involved in these activities. Being allowed to share in the developers' knowledge base about XCF will assist NASA staff in coordinating the XCF instruments with NASA 's various other crystal growth hardware and in training principal investigators from a variety of institutions to use the ground-based control systems.
The XCF is typical of several hardware development projects for NASA in that the technologies it employs can be applied to ground-based research capabilities as well as to those based in space. For XCF, the application of robotics to the cryofreezing process and the low-power, compact X-ray source both have the potential to be useful in laboratories on Earth. Currently, however, the scientific community is mostly unaware of the quality of
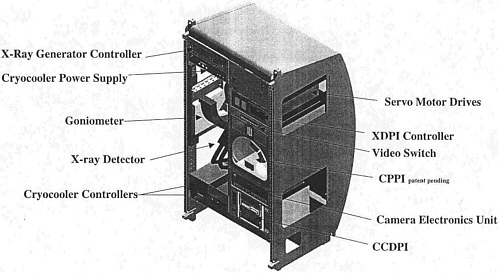
FIGURE 2.1 Schematic diagram of one of the two racks of the X-ray Crystallography Facility (XCF). This rack contains the hardware for mounting and freezing crystal samples and for performing X-ray diffraction studies; the other rack (not shown) contains hardware for growing crystals and observing the growth process. The XCF is in development at the Center for Macromolecular Crystallography at the University of Alabama at Birmingham.
automation displayed in the prototype of the CPPI and of the combined capabilities of the X-ray optics and the low-power source that together yield a high-intensity X-ray beam. While two companies did exhibit this technology at the 1999 Congress of the International Union of Crystallography, the scientific progress made during development of this new product was not widely publicized. While commercial entities may need to be reticient about revealing information about products in development to protect their proprietary work, the task group strongly recommends that full information about technologies and equipment to be used on the ISS be made available to all researchers. This recommendation is based on three factors. The first is that only if the capabilities of the hardware are fully understood will experiments be designed to take advantage of the equipment and the microgravity environment. The second is that well-publicized information about unique or cutting-edge hardware available only through the NASA program will attract a new and broader set of researchers to apply for NASA grants. The third is that scientists will not be willing to entrust precious samples having biologically important implications to equipment unless they are completely aware of both the risks and the potential benefits. The primary goal of the NASA protein crystal growth program should be to serve the research community, not commercial entities. Indeed, NASA's commercial program will be best served by applying the instrumentation to highly visible frontline academic investigations.
CELL SCIENCE
A variety of instruments are being developed to support cell science research on the ISS. Many of them are based on earlier generations of equipment flown on space shuttles or on Mir, but some new approaches are also being investigated. The hardware for cell and tissue culturing falls into three main categories: basic incubators, perfused stationary culture systems, and rotating wall vessels. There is also a variety of supporting equipment,
including refrigeration, monitoring, and analytical instruments. A full list and description of various pieces of hardware relevant to cell biology work on the ISS is provided in Appendix A. The task group focused on the most advanced versions of each type of equipment. Its comments in this chapter on the culturing equipment and the support systems take into account the scientific issues facing the research community and the specialized logistical requirements imposed when experiments are performed so far away from the investigators.
Cell and Tissue Culture Hardware
The hardware for cell and tissue culturing falls into three main categories: basic incubators, perfused stationary culture systems, and rotating wall vessels. Currently, NASA intends to use these instruments to support two types of experiments. First, experiments using the perfused rotating bioreactor and, later, a perfused passive system will investigate tissue morphogenesis for the production of normal and neoplastic tissue for research and biomaterial production. Second, the basic incubators and perfused stationary culture systems will be used to understand the cellular response to the changes in force resulting from decreased gravity and to establish microgravity as a tool for understanding fundamental cellular processes. The three instruments described below are all in the late stages of development and scheduled to be deployed between late 2000 and early 2002.
-
Biotechnology Temperature Controller (BTC). This unit is designed to provide refrigeration on orbit as well as to allow preserving and incubating of multiple cell cultures simultaneously. The cell culture bags are transparent to allow visualization of the samples by light microscopy. While the BTC does not have capability for automated medium exchange, the cultures can be fed using special needleless “penetration” connectors on the bags that provide for multiple aseptic connections. The BTC can accommodate 120 7-ml cultures, or fewer larger samples, within one middeck locker equivalent (MLE).
-
Cell Culture Unit (CCU). This unit is a modular cassette-style bioreactor that can accommodate multiple cell culture chambers (see Figure 2.2). The CCU provides temperature and pH control and allows for continual feeding and waste medium harvest from perfused stationary cultures (Searby et al., 1998). Mixing occurs via medium recirculation. The CCU also provides automated sample collection and injection and high-quality video microscopy. Individual perfused culture chambers can be replaced on orbit. Specimens are loaded in chambers on the ground; inoculation and subculture can occur in space. Bubbles must be manually prevented from accumulating in the chambers; it would be better if an automated system could be developed to handle this task. The CCU can accommodate 8 large (30 ml) to 24 small (3 ml) samples and the associated support and observation equipment within 2.5 MLEs. This piece of hardware is under development by Payload Systems, Inc., in conjunction with the Massachusetts Institute of Technology for the Life Sciences Division of NASA. The cellular biotechnology program within the Microgravity Sciences Division is funding early development work on a Perfused Stationary Culture System, which is expected to be a small-volume (5 to 50 ml), multivessel system for on-orbit cell culture and tissue engineering investigations. This system is in the early stages of development, has a role similar to that of the CCU, and may not be developed if CCU development is successful.
-
Rotating-Wall Perfused System (RWPS): This unit houses a single 125-ml rotating wall perfused vessel in a controlled environment along with associated equipment for medium infusion/perfusion, temperature control, gas exchange, and independent wall rotation control (see Figure 2.3 for a schematic diagram of the RWPS's precursor). Unlike ground-based, rotating-wall bioreactors, in which laminar flow is set up to randomize the force vectors and to minimize the shear stress, space-based vessels have rotating walls in order to produce Couette flow, which augments mass transport. Observation and video recording are possible through a large window in the front of the unit. The RWPS can be inoculated on the ground just before launch or on orbit, but once it has been powered and the experiment initiated, it remains powered throughout the increment until landing. Cell and media samples can be removed on orbit through sample ports located on the side and front panels.
Overall, the NASA-funded cell science work to date has emphasized the use of bioreactors to support three-dimensional tissue growth. While the development of rotating-wall vessels has had, and should continue to have, a significant impact on cell and tissue culturing methodology on the ground (see Box 2.1), the task group has a
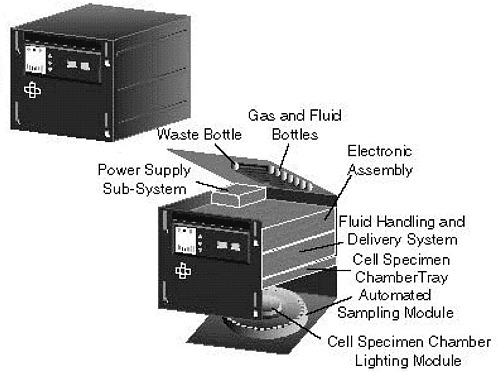
FIGURE 2.2 Schematic of the Cell Culture Unit under development by Payload Systems, Inc. in conjunction with the Massachusetts Institute of Technology for the Life Sciences Division of NASA. Illustration from the NASA Web site, <http://quest.arc.nasa.gov/neuron/photos/images/CCUhabitat.gif>.
variety of concerns about the effectiveness and appropriateness of this approach for research in the microgravity environment. One is that the large volume required for the bioreactor and supporting equipment will yield relatively small amounts of data per unit volume. Other hardware scheduled for use on the ISS (such as the BTC and the CCU) contain smaller specimen units and therefore can serve multiple investigators and house sufficient replicates to permit a complete experiment series in one increment; the potential downside of at least the BTC is the lesser degree of control over environmental conditions. Other concerns about the RWPS relate to the difficulty of accessing the vessel on orbit: sampling is not an easy process and requires a significant amount of equipment manipulation and crew training. Also, bubbles tend to form in the RWPS but cannot be removed without disassembling the entire reactor. A hydrodynamic focusing bioreactor (HFB) is currently being developed at NASA; in this system the shape and rotation of a rotating wall vessel have been redesigned to focus bubbles at one end of the system for easy removal. It may be possible to adapt this approach to allow focusing of cell clusters and, thereby, easy sampling of cell or tissue aggregates of varying sizes from the HFB. If this can be done, the bioreactors could be used as a cell/tissue source for other hardware—e.g., the bioreactor would generate large aggregates that could then be aliquotted and tested in the smaller units for response to a manipulable parameter. Such an approach could generate more data in the same volume based on tissue constructs initiated in a controlled bioreactor. This approach would facilitate the kinetic analysis of cell growth and differentiation processes and would greatly increase the amount of data that could be obtained from a single RWPS reactor experiment. However, the HFB is still in early development, and the current RWPS setup is not appropriately configured for a sample generation role. Furthermore, other ground-based methods for generating three-dimensional tissue constructs, such as the use of scaffolding constructed from biomaterials or micropatterned substrates, may prove to be

FIGURE 2.3 Schematic diagram of the front view (upper panel) and the top view (lower panel) of the Engineering Development Unit (EDU), the precursor of the Rotating-Wall Perfused System. The EDU has housed rotating-wall vessel experiments on the space shuttle and on Mir. Illustrations from the NASA Web site, <http://spaceflight.nasa.gov/shuttle/archives/sts-70/orbit/payloads/bio/bds/bds201.html > and <http://spaceflight.nasa.gov/shuttle/archives/sts-70/orbit/payloads/bio/bds/bds 101.html>.
|
BOX 2.1 Rotating-Wall Vessels on the Ground Rotating-wall vessel systems are an important tool for ground-based investigators exploring cellular and tissue responses to low-stress growth environments and simulated microgravity. These systems have been used primarily to produce three-dimensional self-assembling aggregates that retain some of the cell-cell interactions present in tissues. The most important advantage of these bioreactors is the ease with which investigators can form, sample, and feed the aggregates. Several versions of this equipment are available commercially, including a high-aspect rotating vessel (HARV) and a slow-turning lateral vessel (STLV). Scientists at a number of institutions have used the bioreactors to study a wide variety of culture systems, leading to several advances (e.g., propagation of parasites such as cyclospora or those associated with Lyme disease and studies on impaired locomotion of lymphocytes in space). This technology is now mature and new applications are being investigated in a collaboration between NASA and the National Institutes of Health (NIH). Investigators have compiled a large list of tissues that have been propagated in the bioreactor, including cancer cells, cartilage, liver, kidney, lymphoid tissue, thyroid, skin, pancreatic islet cells, neuroendocrine cells, hematopoietic cells, and intestinal epithelium, as well as tissues from the Bowhead whale and microorganisms.1 While the rotating-wall vessel systems have been important tools for generating aggregates in cell culture for three-dimensional tissue constructs, the rotating wall vessel is limited in many respects. First, even when co-cultures are used, the tissue synthesized is apt to lack many of the minor cells and elements formed within the intact organism. Second, in cell cultures, the cells that die are not generally removed, creating some artifacts. Third, tissues grown in bioreactors are not subject to the environmental signals that they might sense in situ (growth factors, vascular changes, neuromuscular changes), yet these signals are apt to change in the microgravity environment. In addition to these systemic and environmental drawbacks, the rotating-wall vessel has technical limitations. The limited oxygen transfer capabilities make bioreactors inappropriate for systems with high oxygen demand. Also, it has not yet been determined if rotating-wall vessel bioreactors can provide an appropriate environment for tissues such as osteoblasts that only grow properly when the distances between the cells are maintained. Analyses of which cells propagate better than others and why they do might allow the selection of more appropriate cells for development in the bioreactor and increase the effectiveness of the bioreactor's use for ground-based study. Through a joint program with NIH, NASA has made the bioreactor technology and the expertise of NASA scientists experienced with the hardware available to researchers at NIH, the Food and Drug Administration, and other government laboratories. While this program has expanded the use and appreciation of rotating-wall vessel technologies, a broader outreach program that included scientists from universities and other research institutions might increase the communities' familiarity with the technology and improve understanding of which systems benefit most from the bioreactor's low-shear, low-turbulence environment.
|
more effective sources of samples for multiple-chamber hardware such as the CCU and the BTC. In addition, the amount of data produced by these systems in a given period of time and amount of volume on the ISS will be significantly greater than would be produced by a bioreactor system.
Recommendation: Given the current status of equipment in development, finite fiscal resources at NASA, and the limited amount of volume on the ISS, the task group recommends that future research on the ISS should deemphasize the use of rotating-wall vessel bioreactors, which are already established, and continue to encourage the development of new technologies such as miniaturized culture systems and compact analytical devices.
Another factor to be considered in selecting hardware for cell science research on the ISS is the equipment's ability to contribute to efforts to distinguish between the direct impacts of microgravity, where the low level of gravity alters cell behavior, and indirect effects, where space changes the local culture environment (e.g., variations in gas exchange or nutrient and mass transport rates), which in turn affects the cells. If investigations on the ISS are to tackle this question, the instrumentation should be able to manage and monitor culture conditions, such
as cell feeding, environmental stresses, and convective flows, as well as facilitate the comparison of samples with those obtained from a variety of ground- and space-based experimental control groups. For example, methodologies such as micromanipulation could allow researchers to apply controlled mechanical stresses to cells in a microgravity environment to clearly discern whether all mechanosensing capabilities are lost in spaceflight conditions or if the absence of gravity per se has a specific effect on cell behavior.
While the task group believes that of the present instruments, the CCU and the BTC are the most practical tools for research on the ISS, what sort of instrumentation will be most effective for cell and tissue growth in microgravity has yet to be determined. It is important that the relative merits of various pieces of instrumentation be carefully evaluated and that NASA maintain the administrative and engineering flexibility it needs to adopt the most effective systems employing the most advanced technologies and to discontinue hardware development projects that are not attuned to cutting-edge scientific needs of the cell science community. The long-term nature of design and construction of equipment for ISS may be a limiting factor, because the issues and the technology for ground-based research are changing so rapidly that the priorities and hardware selected today may be out of date by the time the cell culture facility is fully operational. Flexibility will be essential, as new culture units, cooling methodologies, and sensors are developed. A modular approach to hardware design must be used to allow for the innovations in technology and the scientific breakthroughs that are likely to occur in the five or more years before the installation of a specialized biotechnology facility on the ISS. Many of the hardware development process issues discussed in the section on protein crystal growth also apply to the development of hardware for cell science. Within NASA's cell science program, close interaction is needed between researchers and in-house operational personnel responsible for developing and constructing hardware to ensure maximum flexibility and responsiveness to evolving scientific goals. Recently, the flight hardware engineering part of the cell science work was placed in an administrative unit separate from that housing the biological research work. Direct communication between these two groups is necessary to construct a facility that can handle relevant, cutting-edge research problems (NRC, 1998). The trend toward separating personnel responsible for the science mission from personnel providing engineering support is, on balance, deleterious to the program. The task group discourages such divisions; instead, NASA should emphasize that the main goal of engineering support is to surmount the challenges faced by space-based science and optimize the research environment on the ISS.
Experiment Management
Cellular systems are very sensitive to environmental perturbations. A continuous power supply to maintain appropriate and stable environments during experiments and for sample storage and transport is essential to ensure valid results.3 The possibility of brownouts during construction of the ISS and the devastating effects that limitations of power would have on cell culture experiments must count heavily in the selection and design of experiments that will be deployed while the ISS is still being assembled. Because the main activity in this phase will be building on existing structures and attaching new modules, there will be severe vibrations throughout the ISS and significant depletion of an already limited power supply. In addition, the amount of power available for research will fluctuate, increasing every time more solar panels are added and decreasing as new modules are attached. The uncertainty surrounding the power supply (and the variations in the total power available for research) suggests that the shuttle itself might provide a more reliable and predictable base for cell science experiments until the ISS is fully operational. However, volume on the shuttle will be devoted mainly to transporting ISS construction materials during assembly, so research activities will occur only infrequently and in the limited volume available in the shuttle's middeck lockers. Experiments on the shuttle would also be limited with respect to time, and short-term tests of research equipment being developed for the ISS may not supply enough information. Flight of cell science hardware aboard Mir provided crucial lessons about long-term experi-
|
3 |
This requirement is relatively unusual among the wide range of research projects being planned for the ISS. |
mentation and hardware reliability and limitations. If it would be possible to perform some experiments on the station while ISS hardware is still in development, potential problems with equipment and experiments could be identified and resolved at an early stage. To gather this information effectively, it would be very useful to schedule periods during the construction phase of the ISS where stable power levels are guaranteed.
Even when the ISS is fully operational, management of the culture environment will still be a key issue. NASA is currently developing the Experiment Control System module to provide the interfaces required for communication and control of experimental equipment, execution of the investigators' experimental protocols, and the recording and archiving of experiment and equipment performance data. Work is also continuing on an on-demand control system to manage resources (such as power and gas delivery) so that equipment can exploit scarce resources efficiently and frugally while maintaining appropriate cell culture environments. Such a system would be installed in a specialized biotechnology facility rack dedicated at least in part to cell science work and would juggle resource delivery so that the experiments grouped in the special rack would consume less power than if they were housed in an EXPRESS rack. Also, the system could reroute power within the facility in crisis situations. Since construction on the ISS was just beginning and much of the research equipment was still in development during the course of this study, the task group was unable to quantitatively compare the power availability and the power demand for biotechnology experimentation on the ISS. As plans for volume and resource allotment crystallize in the coming years, careful efforts must be made to coordinate supply and demand for scarce power resources. Even once construction is complete and the Biotechnology Facility has been installed, the modular approach, whereby equipment can be exchanged between increments, will require continued analysis and coordination of the various instruments' power demands.
Another issue that will be problematic particularly during ISS construction but also after the station is complete, is the limited amount of crew time available for research. Crew will often be trained many months before the experiments and personnel are actually flown, and the astronauts' backgrounds cannot always be coordinated with all of the research under way in a given increment. Therefore, a high priority of equipment developers should be to automate routine tasks and to facilitate remote management of the experiments. For automation, the Experiment Control System has the potential to be very useful. For remote management, a variety of factors must be coordinated to allow investigators on the ground to control and adjust their ISS experiments in real time. The development and effective use of robotics for sample procurement and analysis could markedly increase throughput per unit crew time while also increasing data observation by investigators. Since the lead time before space-based experiments and the time before a follow-up experiment can be flown are both long, any approaches that enable investigators to run a complete experiment, including midcourse corrections, within one increment will greatly improve the quality of research performed on the ISS and will make the program significantly more attractive to the scientific community.
Two key supports for ground-based control of ISS research are (1) sensors to enable physiological control of the cell/tissue culture media environment and (2) analytical equipment to provide feedback about the status of cell and tissue samples. NASA on-site contractors are focusing on sensors that will help investigators monitor and adjust their experiments. Development work on sensors for pH and glucose, as well as a pH control system, is quite advanced, while sensors to measure oxygen and carbon dioxide concentration levels are still in the early stages. A commercial off-line blood gas analyzer system will also be available. To ensure that these instruments closely match the scientific tasks of remote experiment monitoring and management, there must be frequent interaction and feedback between NASA scientists and the on-site contractors.
It will be advantageous to have analytical instrumentation on the ISS to characterize the cells that will be subjected to experimental protocols under microgravity conditions. For example, since microgravity affects the cell cycle, a flow cytometer could permit evaluation of cell cycle markers or apoptosis markers or sorting of cells at specific stages. It is recognized that the conventional technologies will have to be modified for such an analytical instrument to operate in the absence of gravity. Since visualization of the cells will be important, in addition to the light microscope that is planned, confocal microscopy (perhaps digital, instead of laser-based) and phase-contrast microscopy would be beneficial, particularly for the analysis of the three-dimensional constructs produced in tissue engineering experiments. (Confocal microscopy might also be useful for protein crystal growth experiments.) Recent innovations such as the “lab-on-a-chip, ” which utilizes microchip and DNA technologies to
analyze biological samples automatically, would enable the miniaturization of facilities, as would devices for single-cell transfer. Gas chromatograph-mass spectrometry will also be important for the comprehensive characterization of natural products.
Because the new generation of analytical instrumentation is very user friendly, requires small samples, and provides rapid output, the potential is very good for significant analytical capacity on the ISS. Recent advances in miniaturization and automation (Henry, 1999) could allow significant enhancements of ISS analytical capabilities with minimal weight and volume penalties. Some Department of Energy facilities, such as the Sandia and Oak Ridge National Laboratories, are developing relevant miniaturized analytical technologies; NASA would benefit from communication with the personnel in those programs. Other relevant technological advances include DARPA-sponsored work 4 on microfabricated substrates and microfluidic systems (biochips) for cell biology. When miniaturized systems are used, many replicas can be analyzed in parallel, more variables can be explored, less weight is required, and many more experiments can be contained within a single unit. In order to efficiently produce the data needed for cell science publications, the equipment on the ISS should enable researchers to perform multiple studies that are repeated on many different occasions and are focused on distinct, but related, aspects of a single scientific question addressed at the single-cell level.
During selection and development of the analytical systems for the ISS, NASA must recognize the vital role of real-time feedback and acknowledge the limited volume available for sample and information storage. The task group was particularly concerned to note that current plans call for both film and digital still cameras and only a general-purpose video camera to be available on the ISS. Since archiving and transmitting data electronically are easier and less expensive, the task group recommends that digital cameras be the primary technology for both video and static microphotography of cell science on the ISS. This approach, when coupled with use of automated analytical techniques (e.g., lab-on-a-chip), will facilitate archiving and allow immediate transmission of information and results to investigators on the ground. Real-time access to data from the sensors and analytical equipment on the ISS is necessary to enable ground-based control of experiments and to provide scientists with real-time records of the progress of experiments. In the construction phases of the ISS, refrigeration and freezer capability and transport space will be limited, so it will be necessary to analyze the samples immediately before and after the experiments. Even when the ISS is complete, preservation, or reentry forces, can change key characteristics of the samples, so on-orbit analytical capabilities will continue to be essential. If digitized data from on-orbit analyses were available to investigators in real time, they would be less dependent on shuttle trips for results, could select the most important samples for the scarce storage space, and study the changes wrought in samples by freezing and reentry. The value of real-time data should not be underestimated, but neither should digital data from on-orbit sensors be considered a comprehensive substitute for the information that can be gathered on samples when they are returned to Earth and undergo thorough investigation with the wide array of instruments available in ground-based laboratories.
Finding: The limited amount of crew time available for research-related work and the infrequency with which investigators will have access to their samples via shuttle trips mean that automation of routine tasks, ground-based control of experiments, on-orbit analytical capabilities, and real-time transmission of digital data are vital for conducting effective cell science research on the ISS.
It is important to recognize that, while equipment is being designed now, the primary use of the cell science facilities on the ISS will begin 2 to 5 years into the future, and researchers will require access to the most up-to-date analytical instrumentation for their space-based experiments. The focus on quantitative measures of cell and tissue behavior in microgravity will require new forms of genomic, proteomic, and metabolic analysis. Relevant instruments could include fluorescence microscopes using both real-time and time-lapse imaging at up to 100×
|
4 |
The DARPA MicroFlumes program is described on the Web at <http://www.darpa.mil/MTO/mFlumes/>. |
magnification in order to take advantage of techniques that analyze dynamic changes in cell structure and function. Microinjection, micromanipulation, microfluorimetry, use of optical tweezers, and magnetic manipulation are other potential methods to consider as ways to address many of the mechanistic questions relating to gravisensing in cells.
Storage, Transport, and Throughput of Samples
In the initial stages of ISS construction, the generation and retrieval of samples will be limited by the availibility of refrigeration volume. In the absence of low-temperature refrigeration, initial studies will require fixation, and perhaps storage, at ambient temperatures. This approach will require additional ground controls. During flight, multiple experiments will have to be run concurrently, but even comparisons between these systems on orbit will not be able to isolate the effects of launch on the cell and tissue cultures. This limitation will force investigators to have analyses performed immediately after the cultures arrive on the ISS, and then again at predetermined intervals. Reentry will cause other problems, making facilities for on-station analyses doubly useful. Before analytical capabilities are fully installed on the ISS, preservation of samples will be especially important, so the task group recommends a more detailed investigation of methods to optimize sample preservation and recovery in the construction phases of the ISS. Since the paucity of freezer and storage space will impose serious limitations on the type and number of experiments performed, the task group believes that higher priority should be given to acquiring a freezer or other device (e.g., a dewar of liquid nitrogen) for transporting and storing cryopreserved cells during the early phases of ISS construction. This will allow cultures to be initiated on-orbit, eliminating launch effects and increasing reproducibility and throughput. For example, cryocapabilities will permit scientists to assess the influences of varied gravitational forces on parameters of cell structure and function.
Once the ISS is complete, plans call for a variety of refrigeration capabilities: cryogenic (−183°C), fixed samples (−80°C), reagents (−20°C), and storage systems (+4°C). To maximize throughput, plans call for preparing cultures from frozen batches of cells, so these refrigerators and freezers are needed for storing cells for the inoculation of cultures in the middle of an increment. There is some concern that the amount of storage or transport facilities might not be adequate for a sufficient throughput of samples to support an active and viable research program. To make the most of limited volume, miniaturization of samples (even single-cell studies) might be considered. Effective miniaturization of experimental hardware could substantially increase the number of samples analyzed and, hence, the amount of data generated and the number of primary articles published per year.
Another factor affecting the throughput of cell science experiments at all phases of ISS construction and use is access, the ability to get samples and equipment to and from the station. Cell science work requires late loading (14 to 24 hours before launch) and early unloading (3 to 8 hours after landing) from the shuttles to and from the ISS. Cell science samples must also be transported on the shuttle middeck. This will limit access to the ISS in the early phases, when middeck space is at a premium. Thus, there is an incentive to transport specimens that require tight temperature control separately from large equipment such as the CCU and BTC modules, which could fly in the shuttle bay or even be left on the ISS. In this scenario, specimen loading and removal would occur on-orbit, assuming crew time would be available for sample transfer while the shuttle is docked. One factor that must be considered in transporting equipment in the shuttle bay is that the bay can reach 120°F during launch.
Refrigeration and transport are not the only factors limiting the throughput of cell science research on the ISS. Other factors include crew time required for the experiments, the amount and reliability of the power supply, adequate storage space and appropriate environments for samples and supplies, shuttle flight schedules to and from the ISS, the volume of materials to be transported, and, of course, the size of the budget provided for cell science hardware development and research support. Many of these factors are affected by competition from other activities for resources on the ISS, and some of the problems are not specific to cell science but affect programs throughout NASA.
Current plans call for NASA to support between 5 and 15 cell science flight investigations on the ISS each year, and it has been suggested that this level of activity should result in the publication of between two and five primary articles each year. The task group believes that the impact of the papers is more important than the
number of papers. A window of opportunity has been created by the advances in molecular, cellular, and biochemical approaches (e.g., functional genomics and proteomics) that are occurring as the ISS research platform becomes available. The task group recommends that to most efficiently exploit this opportunity, emphasis should be placed on integration of the different approaches and on collaboration between principal investigators and other intra- and extra-NASA investigators. The impact of a given number of experiments could be increased by greater collaboration between investigators interested in different aspects of the same experiment and by more effective management of experiments. For example, a portion of the tissue samples from experiments designed to evaluate the effects of microgravity on three-dimensional tissue formation could be analyzed for changes in gene expression by a collaborating investigator. In this regard, it may make sense to issue NASA Research Announcements (NRAs) requesting proposals for additional analyses that could be piggybacked onto planned flight experiments (these would be described in the NRA). It may not even be necessary to fully fund these piggyback investigations, as the costs associated with flight would be covered under the primary investigator 's grant and the main cost to the secondary researcher would be for sample analyses. Of course, care would have to be taken to avoid conflicts between different experimental protocols. Collaborations between the Life Sciences Division and the cell science investigators could also enhance the scientific output of space station research; these opportunities should be advertised on the NASA Web site to encourage cooperative efforts. NRAs for flight experiments should be modified to encourage applicants to establish multiinvestigator collaborative projects in advance. If this is done, part of the proposal evaluation should include assessing the total amount of information that would be obtained from a flight experiment, including that from collaborators.
Recommendation: Mechanisms should be developed to enable collaborative research projects that maximize the amount of data obtained from each cell or tissue sample by executing multiple analyses on each sample.
Even after experiments are complete and have returned to Earth, methods for archiving the results will be an additional challenge. Current plans do not call for the retrieval/reentry of viable samples from the ISS, but preserved samples and the data obtained both on orbit and during post-flight analysis, such as gene sequences, would be valuable resources for the scientific community. It is important that samples and data from all experiments be archived and the information made readily available, particularly for unsuccessful work that does not result in published articles but probably contains lessons about the difficulties of on-orbit research. Issues related to archiving include what kind of storage will be available, who will own data such as gene sequences (especially data obtained in international collaborations), and how access to archived specimens and data will be advertised to the general science community.
OVERALL VOLUME ALLOTMENT FOR BIOTECHNOLOGY RESEARCH ON THE ISS
Currently, plans call for biotechnology research funded by NRAs to occur within one rack on the ISS. This rack would be shared by protein crystal growth and cell science work. In addition, two racks are reserved for the hardware associated with the X-ray Crystallography Facility (XCF). The task group considered this arrangement and the needs of the various research communities and recommends a shift in the allotments. Of the two XCF racks, one is filled with equipment related to the automated cryofreezing of protein crystal samples and the X-ray diffraction of these samples. The other rack is devoted to growth of the crystals and visual monitoring of the results, and is currently reserved for the equipment and users of XCF and the Space Product Development Division. The task group recommends that this latter rack be officially dedicated to the protein crystal growth program of NASA's Microgravity Research Division, where experiments are selected by a centralized peer-review process and where all protein crystal growth hardware can be used. The rack currently scheduled to be shared by cell science and protein crystal growth can then be dedicated entirely to cell science research.
The task group makes this recommendation based on several considerations. A primary issue is the basic incompatibility between the technical needs of cell science and protein crystal growth equipment on the ISS. For the culture growth hardware, the primary concern is environmental control, which is most effectively maintained
by connecting the cell science experiments to the ISS cooling loop to take advantage of ISS-wide efficiencies.5 For the protein crystal growth experiments, minimal vibration is important and can be obtained by installing an active rack isolation system (ARIS), which cushions the rack from ISS motion and the effects of surrounding equipment. ARIS and the cooling loop cannot be installed on the same rack. The cell science experiments require frequent exchanges of samples, which would also negatively affect the vibrational quiet of the protein crystal growth modules. If cell science and protein crystal growth equipment are housed within one rack, one or both of the disciplines will be forced to operate under suboptimal conditions. Another technical consideration is coordination with other related equipment on the ISS. It would be more efficient for the cell science work to be located near associated analytical equipment or storage facilities, while the importance of the monitoring, mounting, and freezing capabilities of the XCF means that the protein crystal growth experiments need to be closely coordinated with this facility.
The task group also carefully considered the needs of the various research communities expected to use the biotechnology facilities on the ISS. For cell science, there was concern that the amount of data and results generated from half a rack would not be substantial enough to maintain interest within the scientific community, whereas a full rack's worth of instrumentation could raise the program to a critical threshold. For protein crystal growth, the research community is still uncertain about the benefits of growing crystals in a microgravity environment, so the guest investigator program is undersubscribed and commercial interest is low. By focusing the protein crystal growth research efforts on biologically challenging problems and by emphasizing hardware capable of monitoring and preserving samples (the XCF, for instance), NASA could direct its resources to validating the program. The current volume commitment of half a rack of general macromolecular research is insufficient to establish the value of the crystal growth program, but a full rack, filled with peer-reviewed experiments that employ all types of available hardware and have access to the capabilities of the XCF, should be adequate to give the program a fair chance of success. If after several years the results from the protein crystal growth work have provided sufficient proof of microgravity's benefits and the academic and commercial demand for facilities on the ISS increases, then high-throughput hardware should be developed and the allotment of space on the ISS reconsidered based not only on the demand for macromolecular crystallography research volume but also on the results to that point from the cell science program. Alternatively, if the work done through the augmented commitment suggested here fails to clearly demonstrate the value of microgravity for work in structural biology, then the protein crystal growth program can justifiably be terminated.
Recommendation: The volume allotment for biotechnology work on the ISS should be redistributed as follows:
-
The mounting, freezing, and diffracting equipment of the X-ray Crystallography Facility (XCF) should occupy one rack (as currently planned).
-
The cell science work should occupy the entirety of what is currently designated the Biotechnology Facility.
-
The rack currently assigned to the XCF growth equipment and managed by NASA Space Product Development should be officially dedicated to the peer-reviewed macromolecular research run out of the NASA Microgravity Research Division.
|
5 |
This loop circulates water at low (3 to 6°C) and moderate (16 to 18°C) temperatures; the water flows through heat exchangers attached to individual payloads in order to reject heat generated by the various experimental instruments. |















