The report was requested by the federal Health Resources and Services Administration Division of Injury Compensation Programs (DICP), which houses the Vaccine Injury Compensation Program and the Countermeasures Injury Compensation Program. DICP uses reports from the National Academies as an important scientific contribution to its compensation decisions. This report includes research published before mid-October 2023; future research will bring more clarity to the conclusions as researchers continue to study these important vaccines.
The National Academies committee also investigated the causal relationships between intramuscular vaccine administration and shoulder injuries.
Learn more about Shoulder Injuries from Intramuscular Administration of Vaccines
These definitions of causation describe the degrees of certainty that a condition has a causal relationship to a vaccine. The committee adopted the categories and definitions of causation used by previous National Academies committees. The conclusions are asymmetric. A conclusion that there is no causal relationship can never be as certain as a conclusion that there is a causal relationship.
Read more about how the committee assessed causality in Chapter 1
The totality of the evidence suggests that vaccination can cause this harm. Further research is unlikely to lead to a different conclusion.
The totality of the evidence suggests that vaccination might cause this harm, but meaningful uncertainty remains. Studies that better minimize bias and confounding, and studies that estimate effects more precisely, could lead to a different conclusion.
The available evidence is too limited (e.g., few studies in humans, biased, imprecise) or inconsistent to draw meaningful conclusions in support of or against causality. Future research could lead to a different conclusion. This conclusion also applies to situations in which no studies were identified.
The totality of the evidence suggests that vaccination does not cause this harm, but meaningful uncertainty remains. The committee acknowledges that individual causal effects are difficult to ascertain and the limitations of applying population average effects to draw conclusions about the causes of specific events in individual people. For example, it is possible that both vaccination and disease cause certain harms. Thus, (1) an event could be more common in an unvaccinated than a vaccinated population and (2) some of the events in the vaccinated population could be caused by vaccination. Research demonstrating a clear mechanism of action, or research demonstrating increased risk among vaccinated people compared with unvaccinated people, could lead to a different conclusion.
Select a vaccine, and/or an adverse effect, and/or a causal category to view conclusions on evidence linking each of the four COVID vaccines to each of the adverse effects and their level of causation.
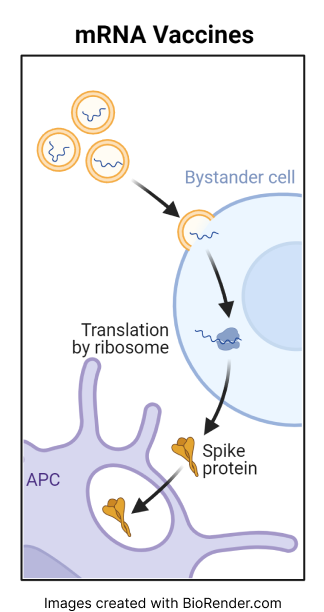
The Pfizer-BioNTech COVID-19 vaccine contains mRNA packaged inside a lipid nanoparticle. After injection, the mRNA enters cells and instructs them to produce a portion of the spike protein normally found on the surface of the SARS-CoV-2 virus. This leads the body to generate an immune response against the spike protein that protects the individual if they are later exposed to the virus.
It was granted Emergency Use Authorization (EUA) from the FDA on December 11, 2020. Full approval for individuals 16 years and older was granted on August 23, 2021. For pediatric use (ages 12-15), full approval was granted on May 10, 2021. For pediatric use (ages 5-11), full approval was granted on October 29, 2021.
The Moderna COVID-19 vaccine is an mRNA vaccine that contains mRNA packaged inside a lipid nanoparticle. After injection, the mRNA enters cells and instructs them to produce a portion of the spike protein normally found on the surface of the SARS-CoV-2 virus. This leads the body to generate an immune response against the spike protein that protects the individual if they are later exposed to the virus.
It was granted Emergency Use Authorization (EUA) by the FDA on December 18, 2020. Full approval for use in individuals 18 years and older was granted on August 23, 2021. For pediatric use (ages 12-17), full approval was granted on October 29, 2021.
BNT162b2 and mRNA-1273 COVID-19 vaccines are very similar in how they work with minor differences in how they are constructed. Neither vaccine integrates into the human genome.
mRNA vaccines, like other vaccines, are designed to trigger an immune response in the body without causing COVID infection.
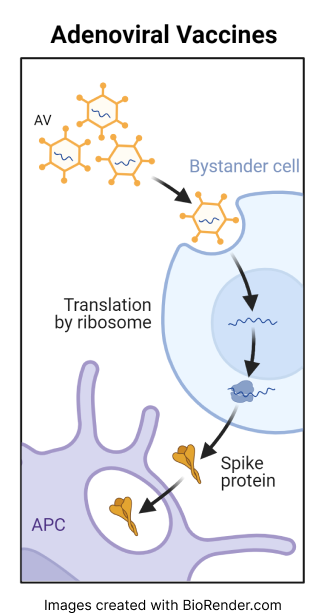
The Janssen Pharmaceuticals’ (J&J) viral vector vaccine delivers genetic material (DNA) inside a modified (harmless) adenovirus. The DNA contains instructions for cells to produce a portion of the spike protein normally found on the surface of the SARS-CoV-2 virus. This leads the body to generate an immune response against the spike protein that protects the individual if they are later exposed to the virus.
It was granted Emergency Use Authorization (EUA) by the FDA on February 27, 2021. However, its EUA was revoked on July 12, 2021, due to concerns about a rare side effect involving blood clots.
Adenovirus vector, like other vaccines, are designed to trigger an immune response in the body without causing COVID infection.
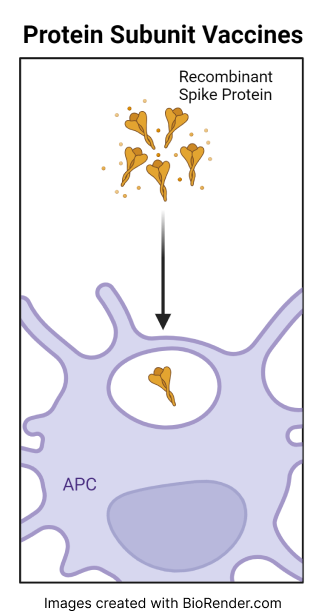
The Novavax COVID-19 vaccine is a protein subunit vaccine that contains portions of the spike protein, a protein that is normally found on the surface of the SARS-CoV-2 virus. The vaccine also contains Matrix-M® (an “adjuvant”) that boosts the body’s immune response. This immune response against the spike protein then protects the individual if they are later exposed to the virus.
It was granted Emergency Use Authorization (EUA) by the FDA on July, 13 2022.
Protein subunit vaccines, like other vaccines, are designed to trigger an immune response in the body without causing COVID infection.