5
Solar, Astronomical, and Atmospheric Effects on Climate
JAMES B.POLLACK
NASA Ames Research Center
INTRODUCTION
Significant climatic changes have taken place on the Earth over a very broad range of time scales ranging from decades to billions of years [Figures 5.1(a) and 5.1(b)]. There is probably not a single cause for these changes but rather a number, which, however, are individually effective over limited time domains (Pollack, 1979). Here, we will be concerned with solar, atmospheric, and astronomical factors that may have played a role in some of the climatic variability that characterizes pre-Pleistocene times.
The sections of this paper are organized by the climatic factors of interest. In each case, we define the manner in which the factor may have influenced past climates, provide an estimate for the characteristic time scales over which the factor is thought to vary, and give a summary of some of the research that has been done in relating this factor to climatic change, with an assessment of its likely importance. Spacecraft missions and ground-based observations have provided evidence that climatic changes have occurred on other objects in the solar system. For example, liquid water may have once flowed across the now desertlike surface of Mars. Therefore, where relevant, we also consider the possible influence of the above factors for the climate and its variability on other solar-system objects.
SOLAR VARIABILITY
From almost the beginning of the solar system, 4.6 billion years (b.y.) ago, the Sun’s luminosity has been steadily increasing with time, according to almost all models of the Sun’s evolution. Recent calculations suggest that the fractional increase in the Sun’s output over the entire period is about 25 to 30 percent (Newman and Rood, 1977; R.Strothers as quoted in Canuto and Hsieh, 1978). A representative calculation of the long-term change in the Sun’s output is given in Figure 5.2.
If no other factor varied, the lower solar output in the past would imply that the temperature of the Earth was progressively lower as we proceed further back in time. In fact, quantitative assessments of the amount of cooling give rise to a serious paradox, as first pointed out by Sagan and Mullen (1972). Global radiation models, in which the CO2 content and

FIGURE 5.1 (a) Left Estimates of the change in the globally averaged surface temperature of the Earth over time scales ranging from the last century to the last 60 million years. These estimates are based on a variety of proxy records except for those of the last century, which are based on direct measurements [from Pollack (1979)]. (b) Above Surface temperature of the Earth over the last several billion years. The filled circles show temperatures inferred from isotopic studies of cherts by Knauth and Epstein (1976). The shaded tracks represent three possible evolutionary tracks for the temperature, as given by Sagan and Mullen (1972).
relative-humidity profile of the atmosphere are held constant with time, indicate that the Earth should have been totally covered with ice from 4.5 to 2.3 b.y. ago as a result of the lower solar luminosity then. But microfossils and stromatolites provide evidence that life has existed on the Earth for the last 3.5 b.y., and the occurrence of sedimentary rocks in the oldest geologic provinces implies that there have been water oceans on the Earth for the past 3.8 b.y. (W. Schopf, University of California, Los Angeles, and H.D. Holland, Harvard University, private communication). Allowance for some climatic feedback processes, such as the ice-albedo mechanism of Budyko (1969), only accentuates this paradox (e.g., Ghil, 1976). Unfortunately, even the sign of other key feedback mechanisms, such as one involving the radiative properties of water clouds, is unknown. Hence, the net response of the complete climate system to a change in solar luminosity cannot be calculated with precision at present.
Before discussing possible solutions to this problem in the next section, it is important to assess how well the Sun’s evolution can be predicted. It is almost universely agreed among astronomers that fusion of hydrogen into helium in the deep interior of the Sun is the ultimate source of the energy it radiates to space. Detailed descriptions of these nuclear transformations have been worked out, with the so-called p-p chain being the dominant sequence for the Sun. An observational test of this theory is provided by attempts to detect neutrinos that are emitted at several steps of the transformation chain. Unfortunately, while solar neutrinos have apparently been detected (Rowley et al., 1980), the flux of them is about a factor of 3 or 4 smaller than that predicted by conventional models of the Sun (Newman and Rood, 1977). This problem is somewhat alleviated by the fact that the neutrinos studied come from a relatively minor branch of the fusion chain. It has been suggested recently that neutrinos may have a small nonzero mass, in which case they may partially transmute from one type of neutrino to other types on the path from the Sun to the Earth. Consequently, fewer “standard” neutrinos may be detected. Finally and most to the point, virtually all solar models, including some very exotic ones that have been devised to resolve the neutrino problem, are characterized by long-term changes
in solar output that are very similar to that of the standard models. This degree of agreement is due to the luminosity variation’s being fundamentally related to the change of the Sun’s mean molecular weight that accompanies the conversion of hydrogen into helium. Hence, once we accept that the fusion of hydrogen into helium has been the source of the Sun’s output over almost its entire history, then it is difficult to avoid the temporal variations of output shown in Figure 5.2.
There is, however, one additional factor that needs to be considered before we accept the above long-term changes in solar output. These changes occur over such long time scales that the universe itself has varied significantly over them. There is the possibility that such cosmological factors as the universal gravitational constant, G, has varied as the universe has expanded. Canuto and Hsieh (1978) have developed cosmologies of this type and have found that the temporal history of the solar flux at the orbit of the Earth is quite different for these models than for the more conventional ones. In one model, in which G varies and matter is created, the Sun’s luminosity increases even more rapidly with time than for the “standard” case. In a second model in which G varies but no matter is created, the Sun’s luminosity decreases with time. In this latter case, we may have just the opposite problem to the one considered above: too high a temperature (greater than 373 K) for life to exist on the early Earth.
In summary, according to almost all models, the Sun’s luminosity has varied by several tens of percent over the history of the Earth. For cosmologies in which G does not vary, the lower solar output of the Sun in the past needs some other variation to prevent the Earth from being entirely covered with ice over much of its lifetime, contrary to the geologic record. Even cosmologies with varying G are characterized by significant temporal changes in the Sun’s output.
The evolution of the Sun’s luminosity also presents a paradox for Mars, at least for constant-G cosmologies. Certain types of channels that are present on the surface of that planet appear to have been carved by running water in the past,
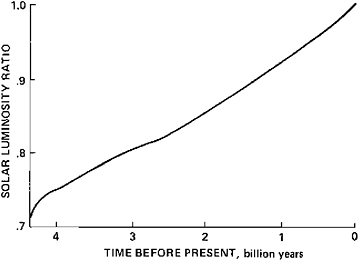
FIGURE 5.2 Solar luminosity as a function of time. The luminosity values have been normalized by the current value. From Stothers as quoted in Canuto and Hsieh (1978).
although they are now dry. While it is not necessary to invoke an altered climate to explain the occurrence of certain classes of fluvial channels, especially the large “outflow” channels, a much warmer climate may be required by other classes, especially the smaller gullies, which are found ubiquitously on the older terrains (Pollack, 1979). But since these terrains were formed several billion years ago, a colder and not a warmer climate is expected, owing to the long-term changes in solar output.
CHANGES IN ATMOSPHERIC COMPOSITION
The composition of the Earth’s atmosphere is controlled by a series of thermodynamic, biological, and geologic factors. For example, the amount of water vapor in the atmosphere is buffered by the much larger amount of water in the oceans, with the partitioning between these two reservoirs being controlled by the saturation vapor-pressure curve of water. Hence, the amount in the atmosphere depends exponentially on the surface temperature. Almost all the oxygen in the Earth’s atmosphere is the result of photosynthesis by living organisms, chiefly phytoplankton, with its abundance being controlled over long time scales (>108 yr) by a balance between losses suffered during chemical weathering of reduced surface material and gains resulting from the burial of reduced carbon compounds [see Figure 5.3(a)]. Much more carbon dioxide is locked up in carbonate rocks than exists in the atmosphere, with the latter being determined over long time scales (>108 yr) by a balance between losses due to the chemical weathering of silicate rocks and gains due to volcanic outgassing of juvenile CO2 and CO2 derived from the thermalization of buried carbonate rocks [cf., Figure 5.3(b)].
There are good reasons for believing that the atmospheric composition has varied with time. First, very little oxygen was presumably present in the early Earth’s atmosphere at epochs prior to the evolution of photosynthetic organisms: the reduced compounds contained in volcanic effluents probably overwhelmed the oxygen produced from the photodissociation of water vapor followed by the escape of hydrogen from the top of the atmosphere (Kasting et al., 1979). Interpretations of the geologic record suggest that little oxygen was present prior to 2.1×109 yr ago, with the oxygen partial pressure subsequently rising more or less monotonically from that point up until a time close to the end of the Pre-Cambrian (~6×108 yr ago), when a partial pressure close to the present value was achieved (Windley, 1977). The rise in oxygen was also accompanied by a progressive buildup in the ozone content of the atmosphere (Kasting et al., 1979). Thus, during much of the Pre-Cambrian, a lot more biologically damaging solar UV light reached the surface than today, although organisms vary considerably in the doses they can tolerate. The rise of oxygen may have set the stage for the invasion of the continents by plants about 4×108 yr ago both by leading to increased shielding of the surface from UV radiation and by providing the basis for an energy efficient metabolism that permitted the development of complex organisms (Kasting et al., 1979; Pollack and Yung, 1980).
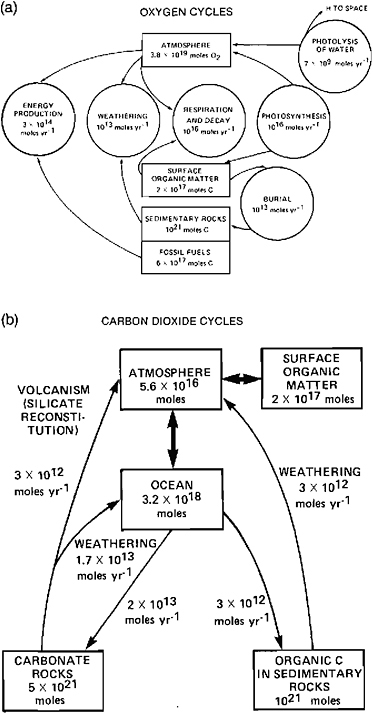
FIGURE 5.3 Geochemical cycles of oxygen (a) and carbon (b). The rectangular boxes show the amount of material stored in major reservoirs: the numbers within circles in (a) and along narrows in (b) indicate the fluxes between reservoirs. From Walker (1977).
The very early atmosphere of the Earth (~3.8–4.6×109 yr ago) may have been reducing in character in order for the chemical steps that led to the origin of life to have occurred (Miller and Orgel, 1974). While a number of years ago this biological requirement was thought to imply a fully reducing atmosphere consisting only of H2, NH3, CH4, and other hydrogen-containing gases, it is now known that complex organic molecules can be biologically synthesized in mildly reducing atmospheres. There is even a minority opinion that such syntheses are possible in neutral atmospheres that interact with catalytic clays (Baur, 1978). Crude estimates of the early composition of the Earth’s atmosphere can be obtained from considerations of constraints placed by thermodynamic equilibrium on the composition of volcanic gases and by the loss of certain gases through the top and bottom boundaries of the atmosphere. These studies suggest that N2 and CO2 were the dominant N- and C-containing gases in the Earth’s early atmosphere, with minor amounts of H2 (~10−3) and CO also being present (Pollack and Yung, 1980).
Variations in the CO2 content of the atmosphere can occur owing to changes in the factors that control its geochemical cycle. For example, the lithosphere may have been thinner and the volcanic outgassing rate higher during the early history of the Earth (Holland. 1978). As a result, the CO2 content of the atmosphere may have been higher then.
We now consider the way in which altered atmospheres may have helped to counter the lower solar luminosity in the past and hence have prevented the Earth from being totally covered with ice. In order for this to occur, there needs to be an augmentation in the concentration of gases that are optically active in the thermal infrared region. Such an augmentation results in an enhanced greenhouse effect. Water vapor is not able by itself to fill this role because its abundance is determined by its saturation vapor curve, however it will roughly double the warming caused by the increase in another gas: the initial warming leads to more water vapor in the atmosphere and thus an even stronger greenhouse effect.
Three types of altered atmospheres have been suggested to counteract the lower solar luminosity in the past: ones containing large amounts of hydrogen (1 bar) (Sagan and Mullen, 1972), trace amounts of ammonia (few to tens of parts per million), and enhanced amounts of CO2 (up to a 1000-fold increase) (Owen et al., 1979). Because hydrogen can readily escape from the top of the atmosphere, with the loss rate being proportional to its mixing ratio, it seems unlikely that hydrogen was a major component of the Earth’s atmosphere over any extended period, and hence the first of these altered atmospheres has a serious problem (Pollack and Yung, 1980). The trace amounts of ammonia cited above for the second model are consistent with its large solubility in water. However, at the above concentration, photodissociation by solar-UV radiation quickly will irreversibly convert NH3 to N2. For example, the total N2 content of the present atmosphere can be generated from NH3 photolysis in only 107 yr (Kuhn and Atreya, 1979). Furthermore, much of the initial N-containing gases may have been N2 rather than NH3, as indicated above. Conceivably, ways of shielding NH3 from solar-UV radiation can be found (see e.g., Pollack and Yung, 1980). But clearly, this second model faces serious difficulties.
The third model, the one involving enhanced CO2, appears to be the most attractive one at present. The factors that control the geochemical cycle of CO2 are such that one can imagine ways in which much more CO2 was present in past atmospheres of the Earth, e.g., an enhanced amount of volcanic outgassing, as mentioned above. Furthermore, the geologic record places only very loose constraints on the level of CO2 in the atmosphere over the last 3.8×109 yr (H.D.Holland,
Harvard University, personal communication). Specifically, the absence of alkali-containing carbonates implies that the CO2 partial pressure was less that about 0.1 bar during this interval. Naturally, it still remains to be demonstrated that there were orders of magnitude more CO2 in the Earth’s atmosphere several billion years ago than today (see Figure 5.4, which summarizes the results of Owen et al., 1979).
An increased abundance of CO2 in the past atmospheres of the Earth may have been due to the combination of the way the Earth’s interior has evolved with time and to feedback relationships between surface temperature and the atmospheric mixing ratio of CO2. With regard to the former, we have already cited the possibility of the Earth’s lithosphere being thinner in the past. Such a possibility is indicated by modern
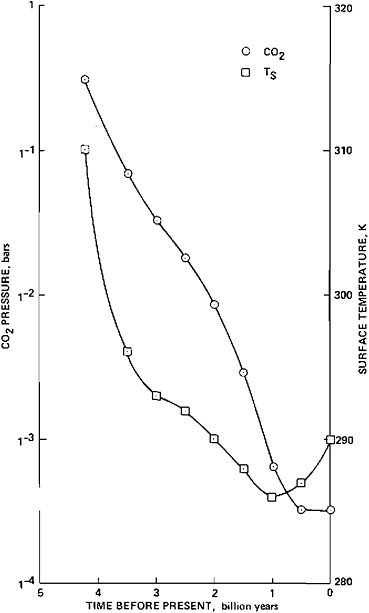
FIGURE 5.4 One possible time history of the partial pressure of CO2 and the resulting variation in the mean surface temperature of the Earth. This figure is based on the model proposed by Owen et al. (1979) to resolve the solar-luminosity paradox.
models of the temperature history of the Earth’s interior (Schubert et al., 1979). An extreme example of a feedback relationship between surface temperature and the amount of atmospheric CO2 is provided by considering a totally ice-covered Earth (Moroz and Mukhin, 1980). In this event, little chemical weathering of silicate rocks would occur and essentially no carbonate rocks would be formed. Hence, the CO2 content of the atmosphere would steadily rise as volcanoes injected new CO2 into it. The surface temperature would increase, because of the enhanced greenhouse effect, to the melting point of water. Walker et al. (in press) have suggested a similar type of feedback process for an Earth dominated by liquid water. They suggest that weathering rates depend monotonically on both the partial pressure of CO2 and temperature. Hence, a lowering of surface temperature leads to a higher partial pressure of CO2, in order to keep the geochemical cycle in balance; in turn, the enhanced CO2 results in a stronger greenhouse effect, which partially counteracts the reduced temperature.
It is worth noting that changes in the CO2 content of the Earth’s atmosphere may not only have occurred secularly over the entire history of the planet but may have also occurred episodically in amounts that could have caused shorter-term climatic fluctuations. If, hypothetically, the outgassing rate of CO2 was doubled and the weathering rate stayed the same, the CO2 content of the atmosphere and ocean would double in only about 4×105 yr (Walker et al., in press). The comparable response time for the CO2 rock reservoir is about 3× 108 yr. Thus, changes in the level of volcanism and/or the characteristics of continents (total area, location, and topography) may also have been accompanied by changes in the amount of CO2 in the atmosphere and thus the globally averaged surface temperature. Changes in the CO2 content of the atmosphere of a factor of 2 or more are required to produce sizable changes in surface temperature. For example, doubling the CO2 content of the present atmosphere, as may occur over the next several decades or century because of man’s activities, would cause the surface temperature to increase on the average by several degrees Celsius, with larger changes occurring in the polar regions.
An enhanced amount of CO2 in past atmospheres of Mars may be the means by which the surface temperature of that planet was increased to the melting point of water, thus setting the stage for the formation of certain types of fluvial channels on the older terrain (Pollack, 1979). Greenhouse calculations suggest that about 1 bar of CO2 is required, i.e., an amount that is a factor of 150 larger than the current atmospheric content of CO2 and an amount that is comparable with the total quantity of CO2 outgassed over the history of the planet (Pollack, 1979; Cess et al., 1980; Pollack and Yung, 1980). Such a large partitioning of CO2 into the early atmosphere of Mars is not implausible, since, in the absence of liquid water, the weathering rates there may have been much smaller than for the Earth. Indeed, the ultimate consequence of the large buildup of CO2—the occurrence of liquid water—may have brought about the demise of the large atmosphere through increased weathering. Because Mars is less massive than the Earth by a factor of about 10, its lithosphere may have thickened much more rapidly than the Earth’s litho-
sphere, and, as a result, little recycling of CO2 may have occurred for Mars (Pollack and Yung, 1980).
OSCILLATIONS IN ORBITAL AND AXIAL CHARACTERISTICS
As a result of gravitational perturbations by the other planets, most notably Jupiter and Saturn, the obliquity of the Earth’s axis of rotation, i, and the eccentricity, e, of its orbit undergo quasi-periodic variations. Also, the orientation of its axis steadily precesses around the normal to its orbital plane because of solar and lunar torques. The characteristic period over which i varies is 41,000 yr, with i varying between about 22.0 and 24.5° (its current value is 23.4°). The dominant period for e is 413,000 yr, with there also being a number of secondary periods clustered around 100,000 yr. e varies between 0 and 0.06, with its current value being 0.017. The precession period is 25,000 yr (Imbrie and Imbrie, 1980).
The above astronomical variations result in seasonal and latitudinal redistributions of solar energy, although only a minor change in the annually and globally averaged solar energy results (Pollack, 1979; Imbrie and Imbrie, 1980). In particular, the obliquity variations lead to a 10 percent peak-to-peak change in the annually averaged solar insolation near the poles, and eccentricity variations result in a 25 percent peak-to-peak modulation in the summertime insolation in a given hemisphere, with the two hemispheres being out of phase. These modulations may be particularly important at times when the continents in one hemisphere are marginally able to have ice sheets as a result of their location and the mean surface temperature.
The last several million years have been characterized by a succession of alternating glacial and interglacial epochs. Indeed, the last major ice age, the Wisconsin, ended only about 12,000 yr ago. Strong evidence that the above astronomical variations are in part responsible for these ice ages is given by comparisons of the characteristic frequencies and phases found in well-dated sea cores with those expected from the astronomical theory (Hays et al., 1976). In particular, periods of 41,000, 23,000, and 19,000 yr characterize certain climate indicators in the cores, which correspond quite closely to those expected from the obliquity variations and the combined eccentricity-precession variations. However, there is still a good deal of uncertainty as to whether the dominant period of the cores, a 100,000-yr period, should be associated with the corresponding eccentricity period. According to linear theories of the relationship between the astronomical variations and climate, only the combined eccentricity-precession variable is relevant. Conceivably, the nonlinear response characteristics of ice sheet growth and decay permit the eccentricity alone to affect climate, although details of the relationship need to be worked out (Imbrie and Imbrie, 1980).
Regardless of the outcome of the above problem, the astronomical variations are at most a necessary, but not a sufficient, cause of ice ages. Prior to several million years ago for a time span of several hundred million years, no large continental glaciations in subpolar and mid-latitude regions occurred, despite the occurrence of astronomical variations with about the same amplitudes as those for the last several million years. In all probability, the drift of the continents toward the poles set the stage for the Pleistocene ice ages and presumably for earlier ones as well (Pollack, 1979).
The amplitude of the astronomical variations in eccentricity are set by the initial orbits of the planets in the early history of the solar system and is not expected to change in any significant way subsequently (W.R.Ward, Jet Propulsion Laboratory, personal communication). However, the amplitudes of the obliquity variations depend on the relationship between the precession frequency, ωp, and the characteristic frequencies of the planetary perturbations. perturbations, ωi. When ωp≫ωi for all i, the amplitudes of the obliquity variations are relatively small and can be significantly augmented only if ωp decreases to a value comparable with ωi. At present, the torque exerted by the Moon on the Earth’s equatorial bulge accounts for about two thirds of its precession rate, with the rest due to the Sun’s torque. During past epochs when the Moon was closer to the Earth, ωp was presumably larger and hence the obliquity variations were somewhat smaller. A dramatic event may occur in the future when the moon moves somewhat further from the Earth. ωp will increase to a 50,000-yr period, becoming comparable with one of the ωi. When that occurs (~few× 108 yr from now), the amplitude of the obliquity oscillations will increase by more than an order of magnitude and enormous climatic changes will occur (W.R.Ward, Jet Propulsion Laboratory, personal communication).
It is highly unlikely that the mean obliquity has varied significantly over the age of the Earth. In principle, such a change could occur because of core-mantle coupling: the mantle has a bigger bulge and is more strongly affected by the lunar and solar torques. Only very gradually through viscous interactions does the mantle drag the core along. Such a coupling would gradually decrease the mean obliquity with time. However, the time constant for the coupling may well exceed the age of the Earth.
Because its ωp is comparable with some of the ωi, the obliquity variations for Mars have a peak-to-peak amplitude of about 20°, i.e., almost an order of magnitude larger than that for the Earth (Ward, 1974). Furthermore, its eccentricity variations are about a factor of 2.5 larger than those for the Earth. These much larger astronomical variations for Mars may have played an important role in generating a quasi-periodic sequence of sedimentary layers in its polar regions (Cutts, 1973; Pollack, 1979). These layers are believed to consist of a mixture of water ice and suspendable dust particles, with the astronomical variations modulating both the deposition rate and the ratio of the two constituents. For example, at times of low obliquity, the atmospheric pressure may drop because of absorption of CO2 on surface dust grains (CO2 is the dominant constituent of the Martian atmosphere). As a result, the frequency of dust storms may decline sharply (Pollack, 1979).
COLLISIONS WITH APOLLO ASTEROIDS
Apollo asteroids are small, stray bodies whose orbits cross the orbital plane of the Earth. At present, some 30 such objects are known, with diameters ranging from 0.2 to 8 km. When al-
lowance is made for the large incompleteness of the telescopic searches for them, it is estimated that there are approximately 750 Apollo asteroids whose diameters exceed 1 km (Wetherill, 1979).
It is inevitable that some of the Apollo asteroids will collide with the Earth; because of orbital perturbations by the planets, the semimajor axes of their orbits and the normals to their orbital planes precess. As a result, every few thousand years their orbits intersect that of the Earth. Almost all of the time, the Apollo asteroid intersects the Earth’s orbit when the Earth is not at the intersection point. But there is a well-defined, nonzero probability for the Earth being at the intersection point, in which case the asteroid collides with the Earth. The average lifetime of a given Apollo asteroid before such a collision occurs is about 2×108 yr (Wetherill, 1979). Hence, collisions with Apollo asteroids with diameters of 1 km or greater happen about once every 2.5×105 yr (Wetherill, 1979) and with ones having a diameter of at least 10 km about once every 108 yr. Because the current population of Apollo asteroids represents one in which an equilibrium has been established between losses through collisions with planets and ejection from the solar system and gains from objects added from the asteroid belt and old cometary nuclei, the above collision frequencies hold throughout most of the Earth’s history. However, during the first 7×108 yr of the Earth’s history, the bombardment rate was probably orders of magnitude larger, based on our knowledge of lunar chronology (Hartmann, 1972).
A collision on land with an Apollo object results in the creation of a crater, whose diameter is about 20 times that of the impacting object and whose initial depth at the center of the crater is about one quarter the crater’s diameter. The Apollo object is almost totally volatized, and a mass equal to about 100 times that of the object is ejected from the crater. Some of the ejecta is highly comminuted rock, and this material may reach high altitudes and stay in the atmosphere for an extended time. Much water as well as pulverized rock may be injected into the atmosphere if the impact occurs in an ocean.
While collision with an Apollo asteroid is clearly a catastrophic event in the immediate vicinity of the impact, the more important issue is whether such collisions can perturb the climate in a significant way on a global scale. Some evidence that collisions with the largest Apollo objects (~10 km) had a profound impact on the global climate has been given by Alvarez et al. (1980). In an effort to detect the presence of extraterrestrial material in the geologic record, they determined the abundance of the element iridium in sedimentary sections that spanned the time period from the Upper Cretaceous through the Lower Tertiary. This element was chosen because its abundance in the Earth’s crust is orders of magnitude smaller than its “solar abundance” value (presumably much of the Earth’s iridium has been segregated to its deep interior) and because very small amounts of it can be accurately measured with neutron activation techniques. Alvarez et al. (1980) found very large enhancements of the concentration of iridium in the acid insoluble clay fraction at precisely the Cretaceous-Tertiary boundary (see also Chapter 8). An enhancement factor of 30 and 160 characterized sections from Italy and Denmark, respectively.
The above result is of considerable interest because the iridium anomaly occurs precisely at the point in the geologic record where mass extinctions, including those of the dinosaurs, occurred 65 million years ago. Thus, the extraterrestrial event responsible for the iridium enrichment may have been responsible for the extinction. A supernova origin for the enrichment was ruled out because no observable amount of 244Pu was detected and because the isotopic ratio, 191Ir/193Ir, was essentially identical to the terrestrial one. The enrichment was attributed by Alvarez et al. (1980) to an impact of a 10-km Apollo asteroid, whose iridium abundance was presumably comparable with the solar-abundance value, The above size is consistent with both the total amount of iridium found and the expected frequency of such collisions.
Alvarez et al. (1980) offer the following scenario for the manner in which an impact of a large Apollo object could have resulted in the extinction at the Cretaceous-Tertiary boundary: Some fraction of the ejecta was very fine dust, which reached the stratosphere and remained there for 3 to 5 yr, by analogy with the large enhancement of the stratospheric aerosol population following the Krakatoa volcanic explosion. So much dust was placed in the stratosphere by the Apollo impact event that essentially no sunlight reached the surface for the next 3 to 5 yr. As a result, photosynthesis ceased for this time period, causing whole food chains to collapse and leading to the extinction of organisms dependent directly or indirectly on living plants and plants, such as phytoplankton, that lacked seeds or their equivalent. Unfortunately, the above scenario rests on a misunderstanding of the enhancement of the stratospheric aerosol population following the Krakatoa event: the fine dust remained in the stratosphere for only a month or two, with almost all of the subsequent perturbation being due to the slow conversion of injected sulfur gases to sulfuric acid aerosols (Lazrus et al., 1971; Pollack et al., 1976). Nevertheless, it is quite clear that study of the various effects that accompany the impact of a large Apollo object— changes in atmospheric temperature and ozone abundance as well as opacity—is a fruitful topic of research and that it may have relevance to the extinction of the dinosaurs as well as possibly other similar occurrences.
PASSAGE THROUGH DENSE INTERSTELLAR CLOUDS
The density of the interstellar medium of gas and dust varies considerably. with gas densities ranging from about 1 atom or molecule/cm3 to about 106 atoms or molecules/cm3. As a very crude approximation, about 1 percent of the mass is partitioned into solid grains, whose characteristic size is about 0.1 µm. During the course of its motion around the galactic center, the solar system may pass through a spiral arm, in which dense interstellar clouds are preferentially located and, in the process, encounter one or several dense clouds. Hydrogen is the most abundant element in the interstellar medium, with it being principally in the form of molecular hydrogen in the dense clouds. Below, we briefly consider the possible climatic consequences of accretion of molecular hydrogen by, alternatively, the Sun and the Earth, It is to be noted that, even dur-
ing the passage through a region having a density of 106 H2/cm3, the opacity of dust grains between the Earth and the Sun is sufficiently small so as not to cause a noticeable reduction in the solar flux at the orbit of the Earth.
Hoyle and Lyttleton (1939) and McCrea (1975) have considered the effect on the Sun’s luminosity of its accreting molecular hydrogen during the solar system’s passage through a dense interstellar cloud. The Sun’s luminosity increases at such times because of the release of gravitational energy by the accreted matter, with the amount of enhancement being proportional to the rate of accretion and thus the density of the cloud. For typical velocities of the Sun relative to the interstellar medium (about 5–25 km/sec), McCrea (1975) finds that cloud densities of about 105 to 107 H2/cm3 are required in order to increase the Sun’s luminosity by 1 to 100 percent. Oddly enough, the increased luminosity is invoked as a means of triggering an ice age, in accord with a theory of Sir George Simpson. Whereas Simpson’s theory is not currently widely held, there is no doubt that an increase in the Sun’s luminosity of 1 percent or more would be climatically significant. McCrea (1975) claims that the solar system passes through an interstellar cloud whose density exceeds 105 H2/cm3 at intervals of about 108 yr. However, more recent considerations of the properties of the interstellar medium by Talbot and Newman (1977) indicate that the above interval may be significantly longer, in which case accretion of interstellar matter by the Sun may not have climatic relevance.
The statistical analysis of Talbot and Newman (1977) indicates that the Earth passes through an interstellar cloud with a density of 103 H2/cm3 about once every 3×108 yr. McKay and Thomas (1978) have examined the possible chemical and climatic consequences of the Earth accreting H2 from such a cloud. During such time, the duration of which is about 105 yr, the solar wind is confined to within the Earth’s orbit and the Earth accretes H2 at a globally average rate of about 7× 109 H2/cm2/sec. This value may be compared with the present rate of 108 H2/cm2/sec at which H is escaping from the top of the Earth’s atmosphere. According to McKay and Thomas (1978), the interstellar H2 molecules are thermalized and diffuse down to an altitude of 140 km, below which they are destroyed by reactions with O, leading to the formation of oddhydrogen compounds, including H, H2O, OH, and HO2.
There are several possible major effects of the above flux of H2 into the Earth’s atmosphere and its subsequent chemical conversions (McKay and Thomas, 1978). First, the ionospheric F-region may be largely destroyed owing to the great efficiency of H2 in affecting electron recombination. Second, there may be a substantial depletion of ozone at altitudes above 50 km owing to the great increase in the abundance of odd-hydrogen species, which dominate the catalytic destruction of ozone there. Third, and most important, there may be a large increase in the water-vapor mixing ratio near the mesopause (about 100 ppm), which may engender the appearance of a dense, global cloud of water and ice in the upper mesosphere. At present, only a very thin ice cloud occurs near the mesopause, and it is located only in the polar regions. According to McKay and Thomas’s calculations, the optical depth of the enhanced mesospheric ice clouds reaches a value on the order of 0.1, resulting directly in a decrease in the globally averaged surface temperature of about 1°C. They claim that such a cooling may trigger an ice age. However, as noted earlier, the position of the continents plays an important role in setting the stage for ice ages. The probability of the Earth passing through a dense interstellar cloud when the continents are properly aligned is extremely small. Nevertheless, the above cooling is still climatically significant and merits further study. Finally, the above chemistry and its consequences apply to times when oxygen was present in substantial quantities in the Earth’s atmosphere and, thus, is not relevant prior to about 2×109 yr ago.
SUPERNOVA EXPLOSIONS
Supernova explosions are cataclysmic events in the lives of some stars whereby their outer envelopes are ejected at high velocities into space. During the first hundred days or so following the explosion, the light output from the supernova exceeds by many orders of magnitude that of the pre-explosion star, and it spans a broad range of the electromagnetic spectrum. In addition, a large flux of cosmic rays accompanies the expanding envelope or supernova remnant.
The climate of the Earth may be affected by a nearby supernova explosion, principally through its effects on the ozone layer. Ruderman (1974) suggested that the terrestrial ozone layer would be severely depleted by both the gamma rays generated during the early phases of the supernova event (approximately first 100 days) and by the cosmic rays generated in the envelop of the remnant, when they swept past the Earth. However, Whitten et al. (1976) have pointed out some significant revisions to Ruderman’s proposal brought about by newer astrophysical data and have carried out a more detailed calculation of the possible ozone reduction. They suggest that the amount of energy contained in the gamma-ray portion of the supernova’s spectrum is too small to affect the ozone layer appreciably and that the cosmic rays contained in the supernova envelope are chiefly due to the trapping of cosmic rays from the interstellar medium. As a consequence of the latter, the Earth is exposed to an enhanced level of cosmic rays over a longer period of time (about 103–104 yr) than suggested by Ruderman.
The amount of ozone in the atmosphere is reduced by the cosmic rays because of their producing large quantities of nitrogen oxides in the stratosphere, which act to destroy the ozone catalytically. According to the calculations of Whitten et al. (1976), ozone reductions of 20 and 50 prcent result from enhancements of the cosmic-ray flux at the Earth of factors of 102 to 103, respectively, which can be expected from supernova explosions situated 10 and 5 parsecs from the Earth. Such reductions will increase the amount of potentially biologically damaging solar-UV radiation reaching the Earth’s surface and will affect the atmospheric temperatures, as indicated below. However, Whitten et al. (1976) estimate that the mean interval between such nearby supernova explosions is about 2× 109 and 2×1010 yr for the 20 and 50 percent depletions, respectively.
Hunt (1978) has calculated the effects of the reduction in ozone on the atmospheric temperature structure. He finds that
temperatures in the stratosphere are reduced by 5 and 11°C and surface temperatures are reduced by 0.1 and 0.3°C, by ozone reductions of 20 and 50 percent, respectively. He also points out that the large temperature changes in the stratosphere may affect the troposphere through dynamical coupling of these two regions.
CONCLUSIONS
Of the various factors discussed above, the ones that would appear to have most profoundly affected the Earth’s climate during the pre-Pleistocene period are changes in the solar output due to long-term (about 109 yr), evolutionary effects, long-term changes in the composition of the terrestrial atmosphere, and impacts by large Apollo asteroids. The first of these factors tended to cause a progressive cooling with increasing time into the past, which may have been counteracted by the second factor. Large increases in the carbon dioxide content of the early Earth appears to be the most promising means of engendering the desired enhanced greenhouse effect. In addition, episodic variations in the carbon dioxide content of the atmosphere appear to be possible due to variations in tectonic processes, with shorter-term climatic oscillations accompanying these changes. Collisions with 10-km-sized Apollo objects about every 108 yr may have been the determining factor for the mass extinctions that characterize the geologic record, although details of the mechanism for this relationship need to be worked out and additional cores need to be studied.
The finding that climatic changes have occurred on other solar system objects, particularly Mars, adds a new and important dimension to studies of long-term climatic changes on the Earth. Furthermore, some of the same climatic factors that are important for the Earth also appear to have been involved in these extraterrestrial climate changes.
REFERENCES
Alvarez, L.W., W.Alvarez, F.Asaro, and H.V.Michel (1980). Extraterrestrial cause for the Cretaceous-Tertiary extinction: Experiment and theory, Science 208, 1095–1108.
Baur, M.E. (1978). Thermodynamics of heterogeneous iron-carbon systems: Implications for the terrestrial reducing atmosphere, Chem. Geol. 22, 189–206.
Budyko, M.I. (1969). Climatic change, Sov. Geogr. 10, 429–457.
Canuto, V., and S.H.Hsieh (1978). Scale covariant cosmology and the temperature of the Earth, Astron. Astrophys. 65, 389–391.
Cess, R.D., V.Ramanathan, and T.Owen (1980). The Martian paleoclimate and enhanced carbon dioxide, Icarus 41, 159–165.
Cutts, J.A. (1973). Nature and origin of layered deposits of the Martian polar regions, J. Geophys. Res. 78, 4231–4249.
Ghil, M. (1976). Climatic stability for a Sellers-type model, J. Atmos. Sci. 33, 3–20.
Hartmann, W.K. (1972). Paleocratering of the moon: Review of post-Apollo data, Astrophys. Space Sci. 17, 48–64.
Hays, J.D., J.Imbrie, and N.J.Shackelton (1976). Variation in the Earth’s orbit; Pacemaker of the ice ages, Science 194, 1121–1132.
Holland, H.D. (1978). The evolution of seawater, in The Early History of the Earth, B.F.Windley, ed., Wiley, New York, pp. 559–568.
Hoyle, F., and R.A.Lyttleton (1939). Proc. Cambridge Philos. Soc. 35, 405–415.
Hunt, G.E. (1978). Possible climatic and biological impact of nearby supernovae, Nature 271, 430–431.
Imbrie, J., and J.Z.Imbrie (1980). Modelling the elimatic response to orbital variations, Science 207, 943–953.
Kasting, J.F., S.C.Liu, and T.M.Doanhue (1979). Oxygen levels in the prebiological atmosphere, J. Geophys. Res. 84, 3097–3107.
Knauth, L.P., and S.Epstein (1976), Hydrogen and oxygen isotope ratios in nodular and bedded charts, Geochim. Cosmochim. Acta 40, 1095–1108.
Kuhn, W.R., and S.K.Atreya (1979). Ammonia photolyses and the greenhouse effect in the primordial atmosphere of the Earth, Icarus 37, 207–213.
Lazrus, A., B.Gandrud, and R.D.Cadle (1971). Chemical composition of air infiltration samples of the stratospheric sulfate layer, J. Geophys. Res. 76, 8083–8088.
McCrea, W.H. (1975). Ice ages and the galaxy, Nature 255, 607–609.
McKay, C.P., and G.E.Thomas (1978). Consequences of a past encounter of the Earth with an interstellar cloud, Geophys. Res. Lett. 5, 215–218.
Miller. S.L., and L.E.Orgel (1974). The Origins of Life on Earth, Prentice-Hall, Englewood Cliffs, N.J.
Moroz, V.I., and L.M.Mukhin (1980). About the initial evolution of atmosphere and climate of the Earth type planets, report of U.S.S.R. Space Research Institute.
Newman, M.J., and R.T.Rood (1977). Implications of solar evolution for the Earth’s early atmosphere, Science 198, 1035–1037.
Owen, T., R.D.Cess, and V.Ramanathan (1979). Enhanced CO2 greenhouse to compensate for reduced solar luminosity on early Earth, Nature 277, 640–642.
Pollack, J.B. (1979). Climatic change on the terrestrial planets, Icarus 37, 479–553.
Pollack, J.B., and Y.L.Yung (1980). Origin and evolution of planetary atmospheres, Ann. Rev. Earth Planet. Sci. 8, 424–487.
Pollack, J.R., O.R.Toon, C.Sagan, A.Summers, B.Baldwin, and W.Van Camp (1976). Volcanic explosions and climatic change: A theoretical assessment, J. Geophys. Res. 81, 1071–1083.
Rowley, J.K., B.T.Cleveland, and R.Davis, Jr. (1980). Brookhaven National Laboratory preprint 27190.
Ruderman, M.A. (1974). Possible consequences of nearby supernova explosions for atmospheric ozone and terrestrial life, Science 184, 1079–1081.
Sagan, C., and G.Mullen (1972). Earth and Mars: Evolution of atmospheres and surface temperature, Science 177, 52–56.
Schubert, G., P.Cassen, and R.E.Young (1979). Subsolidus convective cooling histories of terrestrial planets, Icarus 38, 192–211.
Talbot, R.J., and M.J.Newman (1977). Encounters between stars and dense interstellar clouds, Astrophys. J. Suppl. 34, 295–308.
Ward, W.R. (1974). Climatic variations on Mars. 1. Astronomical theory of insolation, J. Geophys. Res. 79, 3375–3386.
Walker, J.C.G. (1977). Evolution of the Atmosphere, Macmillan, New York, 318 pp.
Walker, J.C.G., P.B.Hays, and J.F.Kasting (in press). Long-term stabilization of Earth’s surface temperature by the greenhouse effect of carbon dioxide.
Wetherill, G. (1979). Apollo objects, Sci. Am. 240, 54–65.
Whitten, R.C., J.Cuzzi, W.J.Borucki, and J.H.Wolfe (1976). Effect of nearby supernova explosions on atmospheric ozone, Nature 263, 398–400.
Windley, B.F. (1977). The Evolving Continents, Wiley, New York.









