3
Glycoscience in Health, Energy, and Materials
Glycoscience contributes in fundamental ways to three key areas on which the committee focused: the understanding of human health and disease, the search for alternative sources of energy, and the development of new materials. The committee selected these areas because they illustrate the range and diversity of research encompassed by glycoscience as a field. They also help illustrate how glycoscience knowledge will be embedded in efforts to address fundamental challenges in health and sustainability.
The chapter begins with examples and questions related to human health because this has been a major focus of efforts in the field of glycoscience and glycomics, particularly in the United States. Indeed, many scientists may automatically think of health when they think of glycans and their functions. Although other researchers actively study carbohydrates and their uses (e.g., in polymer engineering), the terminology and techniques used by these fields may vary. As a result, the scientific community may not immediately think of the totality of glycan research as part of a unified field of glycoscience. One goal of this report is to provide a view of glycoscience that encompasses a broader range of topics. Indeed, although health care remains an important driver for research, increased attention is being paid to other drivers, including the environment and energy security (Johnson 2012), and glycoscience will be relevant in multiple contexts.
Over the past several decades, research from many laboratories has established that glycans are directly involved in normal physiology and in the etiology of every major disease afflicting mankind (Varki et al. 2009). Deciphering the glycome creates an expanding frontier for knowledge and discovery about human health. The section begins with an explanation of the roles of glycans in fundamental biological processes, such as inflammation and immune system activation, and moves on to consider examples from infectious diseases and vaccine development. It then turns to chronic diseases such as diabetes and cardiovascular disease and to a discussion of cancer and congenital genetic disorders. Finally, the significance of glycans in the development of new pharmaceuticals is discussed. Examples of the diverse roles that glycans play in human health are provided to illustrate the breadth and importance of glycoscience to this field. The section does not attempt to comprehensively address all glycan functions. As it illustrates, however, the development of a more complete understanding of glycans can impact the diagnosis and treatment of infectious, chronic, and genetic diseases. (See Figure 3-1 for a partial summary of some of the roles played by glycans in biological systems.)
3.1.1 Glycans’ Regulation of Inflammation
Inflammation, both chronic and acute, underlies the pathology of a broad range of diseases, including diabetes, cancer, arthritis, asthma, heart disease, and infectious disease (Barreiro and Sanchez-Madrid 2009; Celie et al. 2009; Kobayashi et al. 2009a; Korpos et al. 2009; Langer and Chavakis 2009; Schauer 2009; Sperandio et al. 2009; McEver 2010; McEver and Zhu 2010; Sorokin 2010; Zarbock et al. 2011). Glycans play a key role in inflammation at many levels.
Inflammation begins with the generation of multiple cytokines by various cell types that react to pathogen-associated molecular patterns (PAMPs) or danger-associated molecular patterns (DAMPs) arising from damaged tissues. Many of these molecular patterns are glycoconjugates, and many cytokines themselves bind to endogenous glycans. More recently recognized is the fact that glycans found in an individual host, such as a human, can serve as self-associated molecular patterns (SAMPs) that dampen inflammation and that SAMPs can be mimicked by microbes (mSAMPs; Varki 2011). The signaling associated with these molecular patterns is part of the multistep process that results in leukocyte homing into affected tissues, a process initiated when leukocytes adhere to activated endothelial cells lining blood vessel walls. This Velcro-like adhesion slows the rapidly flowing leukocytes, causing them to roll along the surfaces of the endothelial cells. Rolling leukocytes are
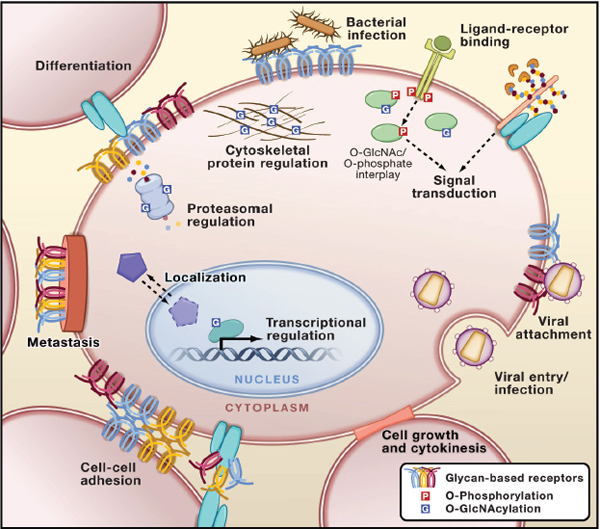
FIGURE 3-1 Glycans play diverse roles within biological systems. SOURCE: Reprinted from Hart and Copeland 2010, with permission from Elsevier.
able to bind tightly to glycoprotein receptors, called integrins, which can lead to penetration of the endothelial cell monolayer and its basement membrane. The mechanisms for the initial binding and rolling of leukocytes have been studied extensively and involve transient expression of highly regulated glycan-binding proteins, called selectins, that decorate the surfaces of leukocytes, activated platelets, and activated endothelial cells. Selectins are exquisitely specific at binding to certain glycan structures. One of the primary determinants for their binding specificity is the tetrasaccharide known as sialyl Lewis x. The unique properties of the interactions between a selectin protein and its specific glycan ligand are critical to slowing leukocytes down so that they can bind to and then extravasate into inflamed tissue. Selectins and their glycan ligands also play a role in tumor metastasis (Dube and Bertozzi 2005; Laubli and Borsig 2010; St Hill 2011).
Glycoconjugates such as collagens, laminins, and sulfated proteoglycans that surround endothelial and other cells also play critical roles in immune cell infiltration of tissues. Leukocytes secrete hydrolases, enzymes that degrade extracellular matrix glycoconjugates and release bioactive glycan-containing fragments in the extracellular milieu. These glycan-containing fragments help perpetuate inflammation by affecting leukocyte chemotaxis, activation, and differentiation. For example, fragments of the polysaccharide hyaluronan are pro-inflammatory as a result of their ability to bind to a class of receptors called Toll-like receptors and serve as “danger signals” of acute cell injury or infection.
The steps in inflammation that are controlled by glycans and glycoconjugates represent potential novel targets for therapeutics that could improve on current treatments like broad-acting steroids. In addition, advances in medicinal chemistry now allow for the rational design of a class of drugs—glycomimetics—that are based on the bioactive conformations of functional glycans (Imberty et al. 2008; Ernst and Magnani 2009; Magnani and Ernst 2009; Garber et al. 2010; Chabre et al. 2011; Drozdova et al. 2011; Jandus et al. 2011). For example, cell adhesion mediated by selectins underlies the vascular occlusion crises that characterize sickle cell anemia. One company has recently designed a small-molecule antagonist that binds to selectins and is now in clinical trials as a treatment for this disease. Other companies are investigating the use of antibodies directed against a selectin and its ligand. Meanwhile, the already approved drug heparin is known to block inflammation by blocking selectin interactions. As a result of these advances, the study of functional glycans represents a source of leads for novel therapeutics for treating inflammation and a variety of other human diseases. The key roles played by glycans in inflammation and the trafficking of white blood cells to tissues also helped stimulate interest in understanding the physiological importance of glycans in cell adhesion and cell signaling more broadly.
3.1.2 Glycans’ Essential Role in Regulation of the Immune System
Inflammation is one result of the immune system’s response to danger. Immunity is the other. Antibodies themselves are glycosylated proteins, and gycans can also be the targets (antigens) for antibody binding and the generation of immune responses.
The general importance of glycans in immunity has been appreciated for many years, and the early discovery that the ABO blood groups derive from specific glycan structures is just one example (Morgan and Watkins 1969). While in most cases the glycans that form part of antibodies do not play a direct role in their binding to antigens, they do play a critical role in their effector functions to activate components of the immune
system (Raju 2008; Lux and Nimmerjahn 2011). Mammals have developed sophisticated glycan recognition systems to recognize pathogen-associated molecular patterns, including specific types of glycan-binding proteins such as lectins, and Toll-like receptors. In fact, the most common phenotype in mice that survive the selective deletion of glycosyltransferases is defective immune cell function (Marth and Grewal 2008). In humans, dendritic cells play a primary role in the presentation of foreign antigens to the immune system (Erbacher et al. 2009). The glycans on dendritic cell surfaces and multiple lectins are involved in dendritic cell functions, including antigen uptake, immune modulation, detection, processing of viral antigens, and trafficking (Silva et al. 2012). Meanwhile, host glycans such as sialic acids serve as self-associated molecular patterns. It is becoming evident that many of these glycan-protein interactions are involved in the critical balance between immune tolerance and the generation of a strong immune response.
Some human autoimmune diseases involve auto-antibodies that recognize self-glycans. The glycans on a pathogen may be sufficiently similar to a human glycan such that an immune response to the pathogen leads to the generation of cross-reactive antibodies. For example, this is thought to be the case in the development of Guillain-Barré syndrome and Miller Fisher syndrome (Kaida et al. 2009). The significance of antiglycan antibodies is also being explored in other autoimmune diseases, such as systemic lupus erythematosus and rheumatoid arthritis (Dai and Gao 2011; Fattal et al. 2010; Louthrenoo et al. 2010).
The specific branching pattern of N-glycans on T-cell antigen receptors regulates their threshold of activation, and a deficiency of a key N-glycan branching glycosyltransferase known as GnT5 may contribute to autoimmune disease (Lee et al. 2007). During their biosynthesis, B- and T-cell antigen receptors are glycosylated in a manner reflective of the cell’s physiological state. This glycan plasticity alters their molecular interactions at the cell surface, associations with signaling complexes, and internalization via endocytosis. These glycans also appear to control the spatial organization of receptors laterally in the plasma membrane. Galectins, which are multivalent glycan-binding proteins, and their glycan ligands have numerous roles in varied immune processes, including pathogen recognition, regulation of inflammation, and modulation of the adaptive immune response (Rabinovich and Toscano 2009). There are currently more than 15 different galectins known in the human genome, each with varied glycan-binding specificity and distinct cellular distributions. Specialized galectins also modulate the threshold of T-cell receptor activation during T-cell development (Demotte et al. 2008). In addition to their role in regulating immune functions, galectins mediate cellular interactions with parasites, viruses, bacteria, and fungi. Recent studies
have indicated that glycan-galectin lattices on the surfaces of immune cells modulate receptor signaling and play a role in modulating effector functions (Rabinovich et al. 2007).
Another class of glycan-binding proteins—siglecs—also have critical functions in immunity. Siglecs are membrane-bound, sialic-acid-binding, immunoglobulin-like, glycan-binding proteins that play a key role in regulating immune cell adhesion, signaling, and endocytosis (Crocker et al. 2007; Crocker and Redelinghuys 2008). Sialic acids are negatively charged monosaccharides that often appear at the terminus of glycan structures. Siglec interactions with immune cell receptors at the cell surface help inhibit abnormal immune cell activation. Siglecs are important for preventing autoimmunity, and they influence the responses of almost every cell in the immune system. There are currently 17 known siglecs encoded in primate genomes, each with a different immune cell type distribution and function. The types of sialyl-oligosaccharides and the structures of the sialic acids on the surfaces of immune cells also play key roles in the activity of siglec regulation of immune cell functions. Given the very recent finding of sialic acids as self-associated molecular patterns that help regulate these immune reactions, it is reasonable to suggest that other self-glycan patterns might yet be discovered.
For high-affinity binding, siglecs require the glycans to be clustered in specific patterns that are not well understood. Critical to their functions is the ability to bind sialic acid glycans both on the same cell (cis) and on a different cell or microorganism (trans). The molecular mimicry of host sialo-glycans by a variety of pathogens and beneficial microorganisms takes advantage of siglecs. As is the case for the selectins, siglec-specific agonists and antagonists represent potentially powerful, but as yet untapped, targets for the development of therapeutics to treat autoimmune and inflammatory diseases. Meanwhile, siglecs already serve as targets for delivering chemotherapeutic agents to specific cell types. For example, CD33, also known as Siglec-3, is the target of an antibody approved for treatment of acute myeloid leukemia, and antibodies targeting CD22/Siglec-2 are in clinical trials for treatment of B-cell non-Hodgkin’s lymphoma and autoimmune diseases (O’Reilly and Paulson 2009).
Glycans in the nucleus and cytoplasm also play a critical role in the regulation of immunity. Recent studies have shown that O-GlcNAcylation, a ubiquitous monosaccharide modification of nuclear and cytoplasmic proteins (Hart et al. 2011), plays a key role in both T- and B-lymphocyte activation (Golks et al. 2007). O-GlcNAc transferase is required for early activation of B-lymphocytes via the B-cell receptor. Data suggest that O-GlcNAcylation is required for nuclear translocation and functions of key transcription factors regulating B-lymphocyte activation and functions.
Although there is still much to be learned about the roles of glycans in immunity, new insights could yield important advances in treating a wide variety of human diseases (Kolarich et al. 2012).
3.1.3 Glycans’ Key Role in Infectious Diseases and Vaccine Development
While glycans are important in regulating immunity, they are also key actors in the constant battle between our cells and invading pathogens, including viruses, bacteria, and parasites. Indeed, glycans are the dominant molecules at this interface. Glycans and glycan-binding proteins, in part because of their plasticity and rapid evolution, play a critical role on both sides of this battle in nearly every species of pathogen (Bardoel and van Strijp 2011). Not only are glycans commonly used by microbes and viruses to bind to and infect host cells but also nearly all of the vaccines for infectious diseases recognize glycans present on the disease-causing organism. The complex mucin-bound glycans lining the epithelial surfaces of the human body not only block invasion by pathogens but also provide binding sites essential to colonization by beneficial bacteria that reside in our bodies and are required for our survival.
Many, if not most, bacteria have adhesins on their surfaces that bind to cells via glycans. These protein-glycan interactions often determine the tissue selectivity of bacterial pathogens (Pieters 2011). For example, lung and airway pathogens, such as Pseudomonas aeruginosa, Haemophilus influenza, and Staphylococcus aureus, primarily recognize glycans terminating in GalNAcβ1-4Gal structures. Recent studies of the bacteria responsible for gastric ulcers (Helicobacter pylori) indicate that these bacteria bind sialylated glycans, such as those found on mucins and gangliosides in the stomach (Kobayashi et al. 2009b). Conversely, the innate immune system of humans, which is a major line of defense against pathogens, has evolved to deal with millions of species of bacteria, fungi, and viruses primarily by recognizing their foreign glycoconjugate structures (Bardoel and van Strijp 2011). For example, the lipid A component of bacterial lipopolysaccharide and the complex mannan structures on fungi are particularly potent elicitors of an immune response. The C-type lectins made by our cells are an important component of our innate immunity in that they recognize a wide variety of glycans on pathogens.
Research has confirmed only recently that the complex glycans present in human milk play a role in protecting newborns from infections and represent a major form of innate immunity (Newburg et al. 2005). The variety of different glycan structures in human milk is enormous, and recently developed glycomic methods are beginning to elucidate the human milk glycome (Chichlowski et al. 2011; Tao et al. 2011). Milk
glycans serve as soluble receptors for pathogens, in much the same way that glycans on epithelial mucins function to inhibit pathogens from binding to the mucosal surface of the gastrointestinal tract. These and other findings suggest that more detailed knowledge about milk glycans might lead to novel antimicrobial agents to prevent, rather than treat, infections.
Because all human cells are covered with a thick glycocalyx, nearly all pathogens, including viruses, must gain entry to their target cells by interacting with glycans. The importance of glycans in influenza infection has been known since the 1940s (Karlsson 1998). In recent years, however, the critical roles of specific glycans in viral infections have been highlighted by fears of a new influenza pandemic. The first step in flu virus infection is the binding of a viral coat glycoprotein—hemagglutinin (HA)—to glycan structures on the host cell. Small mutations in HA enable it to bind to differently shaped glycans on a cell (e.g., to a human cell glycan rather than to a bird cell glycan; see Box 3-1). Thus, a remarkably small change in the ability of a protein to bind to a specific linkage of a single monosaccharide on cell surfaces can have a huge effect on society.
BOX 3-1
Pandemic Influenza
Four major human pandemics—in 1918, 1957, 1968, and 2009—were due to influenza viruses from birds and swine crossing into the human population, causing widespread disease because of a lack of preexisting immunity. Concern about pandemics from new viruses, such as the highly pathogenic H5N1 avian flu virus, has drawn increased attention to the potential for influenza to cross species’ barriers. The designation H5N1 is a classification based on two proteins on the surface of the virus that interact with host receptors. The H stands for hemagglutinin, which attaches the virus to sialic acid receptors on cells. The N stands for neuraminidase, which cleaves sialic acids to allow release of newly formed virus from the infected cell. The neuraminidase is the target for current antiinfluenza medicines such as Tamiflu and Relenza, which blocks the cycle of influenza replication.
Over 60 years ago influenza virus was found to bind to sialic acids on host cells (Karlsson 1998). It is now known that the hemagglutinin of avian flu viruses recognizes sialic acid receptors that differ from those recognized by human viruses. Avian viruses recognize 2-3 linked sialic acids found in susceptible cells in birds, while human influenza viruses recognize 2-6 linked sialic acids, which are found in human airway cells (Stevens et al. 2006; Viswanathan et al. 2010). This small difference is crucial for the transmission of influenza virus in humans. As a result, receptor specificity represents a barrier for transmission of new animal viruses to humans (Tumpey et al. 2007). Owing to the importance of receptor specificity, it is now tracked by the Centers for Disease Control and Prevention as a risk factor for the emergence of new human pandemics from animal influenza viruses.
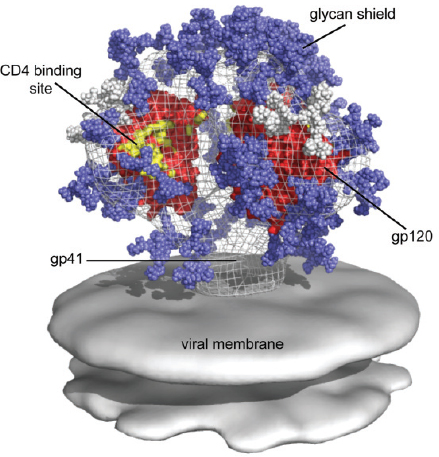
FIGURE 3-2 The glycan shield of HIV. Glycans (blue) cover the HIV gp120 protein, which is responsible for virus binding to the CD4 receptor on T cells. The binding and entry of HIV result in infection of the T cells and, ultimately, in immunodeficiency. SOURCE: William Schief, The Scripps Research Institute, used with permission.
The human immunodeficiency virus (HIV) that causes AIDS has had an enormous impact on human health worldwide. The HIV coat protein (Env) is among the most heavily glycosylated proteins known. As with many viruses, the glycans are assembled using the host glycosylation machinery (Raska and Novak 2010). The HIV virus uses this “glycan shield” to prevent attack by the human immune system (see Figure 3-2). However, recent findings show that humans immune to HIV produce antibodies that bind to the glycan shield and neutralize infection by most HIV strains (McLellan at al. 2011; Pejchal et al. 2011; Walker et al. 2011). These insights are providing new hope for developing vaccine strategies to prevent the disease. The glycans covering pg120 obscure it from recog-
nition by the host immune system. The most potent antibodies identified to date that neutralize HIV infection are those that bind to the glycan shield.
Glycan-containing vaccines were first reported as early as 1929 (Tillett and Francis 1929). Some of the most effective vaccines against infectious organisms are directed toward glycans, and successful glycan-based vaccines include those against Haemophilus influenza and Streptococcus pneumoniae; others are in development. As of 2010, more than 30 glycan-based vaccines were in preclinical and clinical trials (Astronomo and Burton 2010). Recent advances in glycomics, glycan synthesis, glycan arrays, and methods for structural determination have resulted in a quantum leap in glycan vaccine development (Seeberger and Werz 2007; Huang and Wun 2010; Lepenies and Seeberger 2010). Advances in synthetic glycan chemistry are also allowing researchers to create fully synthetic vaccines, a development that may eliminate some safety concerns (Huang and Wun 2010). Still, many issues and challenges remain, such as the identification of epitopes on glycans as a function of a pathogen’s life cycle, stimulation of both humoral and cellular immunity without triggering tolerance, and the design of antigen presentation to generate high-avidity neutralizing antibodies.
More than 15 million deaths occur annually because of parasitic diseases. Even though the immune responses to parasites are almost always directed against their unusual glycans (Nyame et al. 2004), there are no effective vaccines against major parasitic diseases such as malaria, trypanosomiasis, schistosomiasis, and amebiasis. Given the success of glycan-based vaccines against bacteria and the growing knowledge with respect to novel lineage-specific glycans in parasites, this area also represents a promising target for future vaccine development
While only a few examples are given here, it is clear that glycans play a central role in our battle against invading organisms of all types. As antibiotic resistance continues to rise, and the need for antivirals and antiparasitics becomes acute, more focused efforts to understand the central roles of glycans in infectious disease and vaccine development will be increasingly important.
3.1.4 Glycans’ Multifaceted Role in Cardiovascular Disease
Cardiovascular disease is the leading cause of death worldwide (Nieuwdorp et al. 2005; Broekhuizen et al. 2009). It has recently become clear that the glycocalyx of vascular endothelial cells plays a critical role in the etiology of cardiovascular disease. This glycocalyx is comprised of membrane glycoproteins, proteoglycans such as syndecans, and associated glycosylated plasma proteins. The molecules hyaluronan and hepa-
ran sulfate glycosaminoglycans are major components of the endothelial glycocalyx. Normally, these glycans protect the vasculature from damage, but disruption or damage of the endothelial glycocalyx contributes directly to the onset of atherogenesis. The endothelial glycocalyx regulates important enzymes such as nitric oxide synthase and superoxide dismutase and serves as a barrier to macromolecules. In noninflammatory states it also prevents the adherence of platelets and leukocytes (Nieuwdorp et al. 2005).
Diabetes, another chronic disease of increasing prevalence and concern, is a major cause of atherosclerosis. One current model suggests that abnormal expression of proteoglycans or glycosaminoglycans in diabetics contributes to the binding of cholesterol-rich lipoprotein particles at sites in the vascular endothelium. Localized inflammation results in infiltration of macrophages mediated by selectins, which then take up the lipoprotein particles to become foam cells, leading to a plaque that eventually occludes the blood vessel (Tannock and King 2008). In contrast, heparan sulfate proteoglycans play an important role in the clearance of lipoprotein particles from circulation by the liver (Stanford et al. 2009). Nuclear and cytoplasmic protein glycosylation by the molecule O-GlcNAc also plays an important role in diabetic cardiomyopathy (Clark et al. 2003; Jones 2005; Fulop et al. 2007). Recent studies show that the contractile machinery in the heart is heavily modified by O-GlcNAc and that both the modification and association of O-GlcNAc cycling enzymes are strikingly increased in diabetes (Ramirez-Correa et al. 2008). Both the glycocalyx of endothelial cells and the modification of contractile machinery and transcription factors regulating the expression of key cardiac proteins represent novel targets for therapeutic discovery.
3.1.5 Glycans and the Molecular Mechanisms of Chronic Diseases
The prevalence of chronic diseases such as diabetes and Alzheimer’s disease is on the rise, and glycans appear to play critical roles in the etiology of these and other chronic illnesses. Hyperglycemia and hyperlipidemia are the root cause of the biochemical events leading to the morbidity and mortality associated with diabetes, and glycans are involved in regulating a variety of cellular processes. Within the nucleus and cytoplasm, modification of proteins by glycans is surprisingly abundant (Hart et al. 2007; Copeland et al. 2008; Hart et al. 2011). Throughout the cell a system for glycosylating proteins with the monosaccharide O-GlcNAc has extensive cross talk with protein phosphorylation to regulate many cellular processes in response to nutrients and stress, including transcription, signaling, and cell division. Recent data from several laboratories indicate that hyperglycemia-induced increases in O-GlcNAc modification
of regulatory proteins affect insulin signaling (Teo et al. 2010) and are a major mechanism of glucose or lipotoxicity in diabetes. This cellular toxicity results from hyper-O-GlcNAcylation’s effects on signaling as well as from its regulation of gene expression, in which extensively modified transcription factors do not function properly (Clark et al. 2003; Kudlow 2006; Solomon et al. 2008). There is also growing evidence that increased O-GlcNAcylation of mitochondrial proteins in diabetes plays a role in hyperglycemia-induced cellular toxicity (Hu et al. 2009).
Research has shown that elevated free fatty acids or a high-fat diet cause nuclear exclusion and loss of expression of key transcription factors regulating the expression of a glycosyltransferase, called GnT-4a, in the beta cells of the pancreas (Ohtsubo et al. 2011). This loss of transcription factor trafficking and expression is likely related to O-GlcNAcylation, but this possibility has not yet been investigated. Nonetheless, GnT-4a activity is critical for proper glycosylation and expression of the glucose transporters that are key to pancreatic beta cell function. These studies not only serve as another example of how subtle changes in glycosylation can have an impact on human disease in unexpected ways but also suggest a novel target for the possible prevention of beta cell destruction in type 2 diabetes.
Another glycan implicated in diabetes is hyaluronan, a large polysaccharide belonging to the family of glycosaminoglycans made by all cells (Wang et al. 2011). This glycan’s synthesis is exquisitely sensitive to hyperglycemia via both the levels of the donor nucleotide sugars, such as UDP-GlcNAc, required for its synthesis and hyaluronan synthase’s regulation by O-GlcNAcylation (Jokela et al. 2011). The abnormal expression of hyaluronan in diabetes likely plays a critical role in inflammation and in extracellular matrix abnormalities in diabetes, particularly in diabetic nephropathy. Hyaluronan and hyaluronan-binding proteins are directly involved in inflammation, tissue injury and repair, release of cytokines, and cell migration in lung diseases, kidney disease, brain injury, and heart disease (Jiang et al. 2011a).
Other studies have shown that dysregulation of O-GlcNAcylation plays a major role in neurodegenerative diseases of aging, such as Alzheimer’s disease (Dias and Hart 2007; Lazarus et al. 2009). Recent investigations have shown that O-GlcNAcylation plays an important role in brain and neuronal functions, including learning and memory (Tallent et al. 2009; Skorobogatko et al. 2011; Rexach et al. 2012), and pharmaceutical companies are exploring drugs that increase O-GlcNAcylation as possible therapeutics for late-onset Alzheimer’s disease (Yuzwa et al. 2012). The glycan polysialic acid also plays a role in the central nervous system. It is attached to the neural cell adhesion molecule and is involved in such functions as cell adhesion and signaling, which can affect neuron
growth, synaptic plasticity, and leaning. The roles of polysialic acid and neural adhesion molecules in such disorders as Alzheimer’s disease and schizophrenia are also being explored (Kochlamazashvili et al. 2010).
3.1.6 Glycans’ Roles in Cancer Progression and Early Detection
If heart disease is the leading cause of death in the world, cancer is number two and gaining ground. Again, glycans appear to play a central role in this complex set of diseases. If nuclear and cytoplasmic glycosylation is included, it is likely that greater than 80 to 90 percent of all proteins are covalently modified by glycans (Hart and Copeland 2010). Altered or abnormal protein glycosylation is universal in cancer cells of all types (Hakomori 2002; Varki et al. 2009), making such glycans potentially important cancer biomarkers (Reis et al. 2010). Indeed, many current clinical biomarkers in both tissue and serum assays are based on glycans. Much current research, such as that sponsored by the National Cancer Institute’s Alliance of Glycobiologists for Detection of Cancer (http://glycomics.cancer.gov), has already recognized the value of glycans as biomarkers for the early detection of cancer. In addition, recent advances in glycan synthetic chemistry and chemical biology, as well as the development of novel analytical tools, have produced a convergence of proteomics and glycomics to develop improved biomarkers in this area (Drake et al. 2010).
Many of the oldest and most widely used clinical tests for cancer, such as those for carcinoembryonic antigen (colon cancer), prostate-specific antigen (prostate cancer), and cancer antigen 125 (ovarian cancer), rely on the detection of glycoproteins. All of these glycoproteins have both altered polypeptide expression and altered glycosylation in cancer. The concept of using specific glycoforms of glycoproteins to improve the sensitivity and specificity of biomarkers has already been validated by the use of core-fucosylated alpha-fetoprotein as a marker of hepatocellular carcinoma (HCC; Li et al. 2001). Elevation of this specific glycoform in HCC is due to increased activity of a specific fucosyltransferase by HCC cells. While today’s Food and Drug Administration (FDA)—approved assays rely on detecting only a single biomolecule, it is likely that the true diagnostic power of cancer-specific glycoforms will result from multiplexing several glycoprotein biomarkers.
It has been suggested that abnormal expression of specific glycosyltransferases themselves might serve as useful biomarkers for cancer (Meany and Chan 2011). In fact, certain glycosyltransferases, such as GlcNAc-T5 and GALNT5, have been linked directly to the etiology of cancer and are potential targets for therapy. There are numerous examples in which simple overexpression of a glycosyltransferase leads to tumorigen-
esis (Dennis et al. 1999; Meany and Chan 2011). Thus, improvements to existing biomarker assays for nearly all types of cancers will likely involve the specific cancer-associated glycans attached to the polypeptide as well as the polypeptide expression. By combining data analyses from genomics, proteomics, metabolomics, and glycomics with pathway analyses, it should be possible to make great strides in cancer biomarker discovery as well as to reveal novel therapeutic targets (Adamczyk et al. 2011).
Many tumor-associated antigens were discovered by nonchemical methods and were later determined to be glycans (Heimburg-Molinaro et al. 2011). Adult tumor cells often display glycan structures that are normally found only on specific cells of embryos. There is even an instance in which a diet-derived nonhuman sialic acid Neu5Gc is incorporated into tumor cell glycans as a “xeno-autoantigen,” driving chronic inflammation via interactions with “xenoautoantibodies.” Over many years, glycan tumor-associated antigens have been studied and used as potential cancer vaccines (Li et al. 2010). It has been suggested that as more is learned about glycomics and about how the immune system recognizes cancer-associated glycans, effective development and use of glycan vaccines against cancers may come to fruition.
Altered glycosylation also underlies many of the properties of the cancer cell, including the propensity to metastasize. Tumor metastasis is a complex process requiring adhesive interactions, many of which are mediated by cell surface glycans and lectins, particularly the selectins (Rambaruth and Dwek 2011). N-glycan branching on the epithelial cell adhesion molecule E-cadherin (Pinho et al. 2011) and selectin ligands play a role in critical tumor cell interactions with platelet and endothelial selectins. Laminin and the hyaluronan-binding protein CD44 play a role in invasion and migration of tumor cells through connective tissues. Lectins and heparan sulfates, another class of glycans, play a role in both angiogenesis and in interactions with vascular endothelial cells. The glycocalyx of tumor cells plays a key role in masking surface antigens and evading immune surveillance mechanisms. While it is clear that cell surface glycans and glycan-binding proteins contribute to all stages of cancer progression and metastasis, and thus may provide novel targets for therapy, there is still much to be learned about the specific roles of glycans in cancer.
Nuclear and cytoplasmic glycosylation also plays a role in carcinogenesis by directly affecting signaling and transcription processes that contribute to cell growth and neoplastic transformation (Slawson et al. 2010; Slawson and Hart 2011). In fact, every nuclear oncogene or tumor suppressor protein examined to date is dynamically O-GlcNAcylated, and every cancer type examined to date has strikingly elevated O-GlcNAcylation on many different proteins. Cancer cells also have increased levels of the
enzymes that control O-GlcNAc cycling in cells. Nonetheless, the roles of O-GlcNAcylation in cancer remain a relatively unexplored area.
3.1.7 Critical Roles of Glycans in Human Development
Genetic diseases of both glycan biosynthesis and glycan degradation clearly establish the critical roles of glycans in human development. Currently, there are some 4,500 identified genetic disorders, with the biochemical cause unknown for 2,700. It is estimated that 2 percent of the genome encodes glycosylation-related genes. As a result, many genetic diseases whose etiology is unknown today may prove to be tied to glycosylation. In fact, research over the past 15 years has identified more than 65 glycosylation disorders (Hennet 2012), many of which did not have defects in known genes. These disorders encompass all known glycosylation pathways but saturate none of them, which suggests that there are likely more glycosylation disorders yet to be identified. The value of research aimed at identifying these disorders is shown by the development of enzyme replacement therapies for enzymes that act on glycans. Gaucher’s disease, which results from defective degradation of glycosphingolipids, and mucopolysaccharide storage diseases that result from defects in degradation of glycosaminoglycans are examples of glycan-processing disorders now successfully treated with enzyme replacement therapy (Butters 2007).
Congenital diseases of glycosylation (CDGs) can result from mutations in almost all aspects of the glycosylation machinery, including the biosynthesis and transport of nucleotide sugars, proteins controlling vesicular trafficking in the endoplasmic reticulum and Golgi apparatus, and glycosyltransferases (Freeze and Ng 2011; Jaeken 2011). CDGs are rare in the human population because defects in glycosylation are almost always fatal in development, but when the fetus survives to birth the defects are often quite severe. History has shown that rare diseases have served as Rosetta stones for physiology. These rare genetic diseases of glycosylation have already revealed critical functions of glycans in human development. But to fully capitalize on the data that studies on CDGs will generate will require informatics and systems biology-based approaches to map the links between glycobiology and physiological function, which in turn could generate new therapies and new leads for finding other glycosylation-related genes.
Severe forms of muscular dystrophy result when a genetic mutation causes abnormal glycosylation of alpha-dystroglycan, a protein that links the extracellular matrix to the actin cytoskeleton in muscle cells (Muntoni et al. 2011). Mutations in any of the glycosyltransferases involved in synthesis of the glycan structure on alpha-dystroglycan result in various
forms of muscular dystrophy, clearly supporting an essential role for these glycans in muscle physiology (Godfrey et al. 2011). Another example of how specific glycosylation regulates a critical developmental signaling pathway comes from the study of Notch signaling, which is regulated by the glycans attached to the Notch receptor.
Many lysosomal storage diseases result from the mis-targeting of lysosomal glycohydrolase enzymes to the lysosome (Cox and Cachon-Gonzalez 2012). The targeting of lysosomal enzymes to the lysosome by glycans containing mannose-phosphate moieties has long been a model of glycan-specific functions (Kornfeld 2010). These storage diseases are characterized by severe developmental abnormalities, especially in the brain. Many glycan storage diseases involve proteoglycan or glycosaminoglycan accumulation (Coutinho et al. 2012) or result in the accumulation of gangliosides (Schulze and Sandhoff 2011), perhaps as a result of their abundance and their complex structures. There are seven known forms of glycosaminoglycan storage diseases, all sharing some clinical features, such as organomegaly, and central nervous system malfunctions (Coutinho et al. 2012).
3.1.8 Bioactivity and Pharmacokinetics of Drugs
With notable exceptions such as insulin and growth hormone, the great majority of biotherapeutics are produced as glycoproteins. Attempts to express such proteins in prokaryotic systems, such as E. coli, often result in misfolded, unstable, or inactive products. To circumvent these issues, multiple platforms have been developed for the manufacture of glycoproteins, including the recombinant expression of biotherapeutics in yeast, insect, plant, and mammalian cell systems. However, expression in such eukaryotic systems often results in incorporation of nonhuman glycosylation, which can alter efficacy, half-life, antigenicity, and other features of the biotherapeutic (see Box 3-2). Glycan engineering approaches to “humanize” the glycans attached to proteins produced in these systems are a strategy that is actively being explored (Gomord et al. 2005; Chiba and Jigami 2007; Zhang et al. 2011).
Despite the many roles of glycans in biology, there are still relatively few glycan-based drugs (Galan et al. 2011), with a recent review identifying only nine on the market (Shriver et al. 2004). This small number is a consequence of the difficulties in synthesizing glycans and their high polarity, which typically results in poor pharmacokinetics. Instead, most companies synthesize molecules to mimic glycans (Ernst and Magnani 2009; Magnani and Ernst 2009). However, with the right pharmacokinetic properties, glycan-based drugs can prove incredibly useful. For example, one of the oldest and most used set of drugs known is the polysaccharide
BOX 3-2
Influence of Glycosylation on Drugs: Erythropoietin
The anemia drug erythropoietin (EPO) provides an example of a recombinant biotherapeutic in which differences between normal human glycosylation and nonhuman glycosylation have functional significance and can be used as the basis for testing. The glycosylation of recombinant erythropoietin affects clearance by the liver and influences the pharmacokinetic interactions of EPO with its receptor, which affects uptake into tissues and clearance from the circulation. Nonhuman glycosylation, such as the presence of Galα1-3Gal in recombinant biotherapeutics, has been reported to cause antigenicity, while the capping of glycans with sialic acid is known to dramatically increase circulating half-life. The glycosylation difference between natural human erythropoietin (made by the kidneys) and the recombinant form (made in Chinese hamster ovary cells) is the primary basis for the World Anti-Doping Agency test to detect illicit erythropoietin use by elite athletes, such as cyclists in the Tour de France. Despite a potential illicit market in the billions of dollars, no one has yet succeeded in mimicking the natural human glycosylation in cultured cells.
The importance of glycosylation on biotherapeutic agents goes beyond these biological factors. First, because of differential activity, claims of unique intellectual property can sometimes be made based on differential glycosylation even considering the same underlining polypeptide. The glycosylation difference between erythropoietin made in different types of cells was partly the basis for the successful defense of intellectual property rights challenges against recombinant erythropoietin. This has also been the case with Fc effector functions of antibodies. Second, because of nontemplate addition of glycosylation, a biotherapeutic is actually a mixture of glycoforms rather than a single active ingredient. Biotherapeutic characterization and lot-to-lot comparison are made more difficult because of this microheterogeneity. As a consequence, regulatory agencies such as the FDA must develop definable and “acceptable” ranges for variations of this microheterogeneity, to define process control and comparability across different manufacturing systems. Finally, changes in culture conditions can result in significant changes to glycosylation. Thus, glycosylation is an independent way of monitoring process control. Our ability to understand, analyze, control, and modify glycosylation has and is likely to continue to have a huge impact on the quality and application of biotherapeutic agents. The market for these drugs is in the billions of dollars, and this is an aspect of glycoscience’s impacts on not only human health but also economics and world regulatory standards.
heparin and fragments of heparin, which are used in the clinic as an anticoagulant and as an anti-inflammatory agent.
It has been estimated that as few as 20 thousand glycan structures might represent most of the binding specificities of known human glycan-binding proteins (Cummings 2009; Smith et al. 2010). Indeed, it soon will be in the realm of possibility to construct a glycan array with this number
of different glycan structures. These arrays can impact the field of drug discovery by allowing the specificities of lectins, growth factors, cytokines, antibodies, and toxins to be analyzed rapidly (Liang et al. 2008; Wu et al. 2009). This would be the first step in the design and development of glyco-mimetic drugs.
Antibodies, which are glycoproteins, have been used as therapeutics for more than 100 years and the development of monoclonal antibodies in the early 1970s revolutionized their therapeutic potential (Yamada 2011). In recent years, the pharmaceutical industry has put significant effort into the development of antibody therapeutics. However, only recently have they realized the importance of understanding the types of glycans attached to antibodies, which play roles in their efficacy and safety. For example, antibodies made in cultured cells or animals often have glycan structures attached to them that differ from those made by humans, and these changes can elicit deleterious immune responses and affect therapeutic effectiveness. For example, antibodies synthesized in nonhuman cells often are modified by nonhuman sialic acids known as Neu5Gc (Ghaderi et al. 2010) or nonhuman alpha-galactose residues, both of which can elicit an immune response (Lux and Nimmerjahn 2011). Recent advances in glycoengineering of therapeutic monoclonal antibodies should greatly advance the development of useful therapeutic antibodies, and this area represents a substantial investment in the field of drug development. A large number of other therapeutic agents also are glycoproteins, and the glycosylation of these biological drugs plays various roles, ranging from initial protein folding, to stability in circulation, to the criteria used by the FDA to monitor process control.
3.1.9 Key Messages on Glycoscience and Health
As the preceding discussion has demonstrated, glycans play critical roles across all aspects of human health. The importance of glycans to so many areas of biology and the diversity of roles that glycans play are not surprising, because they are one of the four fundamental classes of biological molecules and are abundant on cell surfaces, as well as being major modifiers of proteins and lipids. Significant advances in understanding the functions of glycans in human health and disease, and unlocking ways in which they may contribute to the development of new medical treatments, have already been made. In many cases, glycans provide an additional layer of biological information that builds on what can be learned through genomics and proteomics. Investments in understanding the human genome and proteome have already yielded insights. Because it is so entwined in biology, however, incorporating an improved understand-
ing of glycoscience will be required to fully realize the goals of genomic and proteomic research to improve human health.
As a result, the committee finds that:
- Glycans are directly involved in the pathophysiology of every major disease.
- Additional knowledge from glycoscience will be needed to realize the goals of personalized medicine and to take advantage of the substantial investments in human genome and proteome research and its impact on human health.
- Glycans are increasingly important in pharmaceutical development.
The need for increased energy sources to power modern society is a major area of research and discovery. The U.S. Department of Energy has estimated that the United States derives 37.13 quads of energy from petroleum sources (Greene 2011) and that in 2010 U.S. oil consumption was more than 19 million barrels per day (CIA World FactBook; https://www.cia.gov/library/publications/the-world-factbook/). In 2009, fossil fuels (petroleum, coal, and natural gas) were used to meet approximately 83 percent of this country’s energy needs, whereas only 8 percent came from a range of renewable energy sources (Greene 2011).
Although the global economic recession and continued development of new oil fields and new oil extraction techniques influence the cost and accessibility of petroleum resources, global energy consumption is expected to continue to increase, and there is ongoing interest in exploring a range of energy solutions. Lynd (2010) has written that twice in human history—the Neolithic Revolution and the Industrial Revolution—major changes in resource use by humans have transformed day-to-day life and societal organization. Today, there are indications that a third revolution, a Sustainability Revolution, may be required. Biomass, which currently provides only an estimated 3.88 quads of U.S. energy (Greene 2011), is one option as an energy source, particularly for the production of liquid fuels that will always supply as much as two-thirds of the demands from the transportation sector. A major underlying theme for the development of biomass resources is the role of sugars in the formation of the complex glycans that make up the bulk of plant cell walls. In principle, these complex glycans, if processed efficiently, can serve as a renewable source of high-value biofuels and bioproducts in much the same way that oil serves as the source of high-value fuels and petroleum products. However, achieving this sustainability revolution will be a challenge because
it must be global in scale, occur over a shorter period of time than the previous two revolutions, and will require overcoming the recalcitrance to degradation of plant cell walls that make up the bulk of what is collectively known as biomass (Himmel 2008; Lynd 2010; Lynd and Cruz 2010).
3.2.1 Biomass—Plant Cell Walls
Plant cells walls represent Earth’s dominant biological carbon sequestration system. However, although plant cell walls are a terminal carbon sink, they are not merely a storage depot. Rather, they are a highly complex set of polysaccharides with structural proteins and lignin. It is important to recognize that there is no such thing as a “cell wall.” The cell wall structures of plant species, grasses, and trees, for example, have different levels of structural heterogeneity (Avci et al. 2011). In addition, there can be 35 to 40 tissues in a single plant, each with a cell wall containing structural differences. Even in a given tissue or cell, cell wall layers and domains may have very different structures or polysaccharide distributions (Albersheim et al. 2010).
During photosynthesis, plants use sunlight to “fix” carbon from carbon dioxide into sugars, a form of chemical energy. In this process the photons in sunlight are used to produce molecules that contain high-energy chemical bonds, such as ATP and NADPH. These molecules in turn are involved in a series of reactions that create carbon-containing sugars. It is these sugars in biomass that are considered a source of energy for the production of biofuels. However, little is known about how the cell converts those sugars into complex polysaccharides and assembles the completed polysaccharides into networks and then into the overall architecture of the cell wall. Cellulose, hemicellulose, and pectins—all complex polysaccharides—plus lignin, and often structural proteins, all contribute to the overall architecture of the cell wall (see Figure 3-3). Once the networks are formed, there are further processing steps, such as hydrolase processing, about which little is known but that are major contributors to polysaccharide heterogeneity. How this whole system is regulated is still largely a mystery (Albersheim et al. 2010) and remains a major challenge in glycoscience.
How this whole system is regulated is still largely a mystery (Albersheim et al. 2010) and remains a major challenge in glycoscience. Although the process of how plants make sugars from carbon dioxide and water is fairly well understood, little is known about how the cell partitions that carbon into different sugars and then into the 14 different nucleotide diphosphate sugars (NDP sugars) needed for the biosynthesis of cell wall polysaccharides. In most cases the enzymes involved in these processes are known, but it is not known how they are regulated. Also,
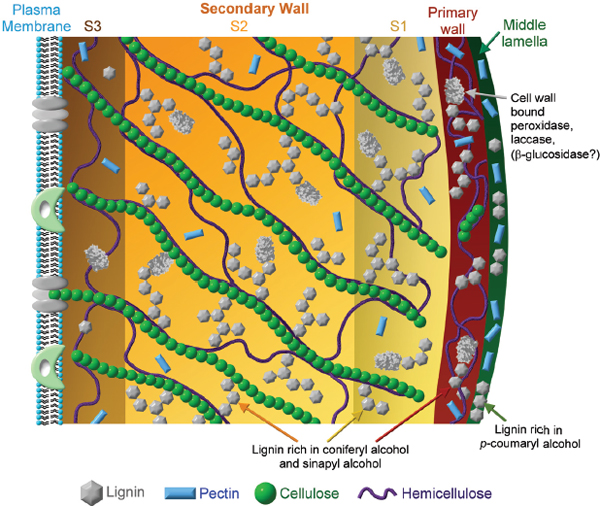
FIGURE 3-3 Schematic of a plant cell wall. SOURCE: Achyuthan et al. 2010, used with permission.
yet to be clarified is the role that Golgi-localized NDP-sugar transporters play in the subsequent synthesis of polysaccharides and the identity of all of the glycan syntheses and glycosyltransferases involved in the production of the component polymers of plant cell walls (Bar-Peled and O’Neill 2011). Even when the identity of these enzymes is known, it is still unclear how they choose their substrates, whether they need a primer to start the synthesis of a polymer chain, whether they work in protein complexes, and what determines the ultimate length of a synthesized polysaccharide. To date, based on the glycosidic linkages found in any given plant cell wall, a good estimate is that about 120 different glycosyltranferases are needed to synthesize the plant wall, of which only about seven are known. Another important unknown regarding cell wall structure is how a plant actually assembles the completed polysaccharides into networks and then into the overall architecture of the insoluble cell wall. Once the
networks are formed, there are more processing steps that are major contributors to polysaccharide heterogeneity (Albersheim et al. 2010).
3.2.2 Recalcitrance to Degradation of Biomass Feedstock
The major obstacle to realizing a future in which liquid fuels and other high-value products and materials are produced from biomass as a supplement to, or in place of, petroleum is the recalcitrance of the cell wall to degradation and release of sugars for fermentation (Himmel 2008). Cellulosic biomass’s recalcitrance to degradation is related to the need for a plant’s cell walls to support its morphology and biological functions, including upright growth habit, resistance to internal turgor pressure, and water and nutrient transport. It also arises from the plant’s need to evolve resistance to attack by the elements and from microbes and their enzymes.
There are several approaches to overcoming this recalcitrance, including:
- creation of a modified cell wall architecture that would be more susceptible to deconstruction by microbes or enzymes,
- reduction of the inhibitors in plant cell walls that reduce the efficiency of microbial and enzymatic deconstruction,
- reduction or modification of the cell wall lignin content, and
- increase in the amorphous cellulose and hemicellulose content of the cell wall and increase in susceptibility to breakdown.
This is an area where glycoscience understanding will play an essential role in bioenergy science, given the large number of unknowns that exist concerning how a plant makes a cell wall and in understanding the enzymes and microbial interactions that can aid in overcoming this cell wall recalcitrance to degradation (Himmel et al. 2007).
Because of the wide variation in cell wall composition and structure across the plant kingdom, the type of feedstock used as a source of sugars for biofuel production has a pervasive impact on biofuel performance metrics. Perennial biomass cellulosics (e.g., switchgrass) score highly except in terms of cost-efficient conversion of biomass to sugars as a result of cell wall recalcitrance. A number of companies that are currently developing commercial conversion facilities to produce liquid fuels from lignocellulose believe that it will be possible to sell such fuels at the same price as petroleum without any subsidies (DOE 2006; Lynd et al. 2008). As noted above, however, the grand challenge to achieving that kind of price drop is overcoming the recalcitrance of cellulosic biomass to conversion into the sugars that are needed to produce liquid fuel via fermentation. This is the most costly processing step or set of steps in producing liquid
fuel from biomass, but there is potential for research and development–driven cost reductions. Such advances are not only necessary but also would be sufficient to create a cellulosics biofuels industry (DOE 2006).
Understanding cell wall recalcitrance at this basic level would further the achievement of several goals. Efforts are under way to develop improved enzymes and other catalysts for decomposing cell walls, and an understanding of plant recalcitrance helps in both the design of new molecules and investigating how and why different cell walls respond differently to them (Himmel et al. 2007; Rubin 2008; Carroll and Somerville 2009; Van de Vyver et al. 2011). Genomic investigations have identified a large number of potential carbohydrate-active enzymes, and the CAZy database for carbohydrate-active enzymes (http://www.cazy.org/) is an important resource in this area. For example, more than 27,000 putative carbohydrate-active enzymes were identified in a recent study of a cow rumen (Hess et al. 2011). It remains largely unknown whether or how this range of potential genetic and structural enzyme variations translates to a range of useful functional variations, and research will be required to decipher the functions and possible applications of these potential enzymes. As this area demonstrates, glycoscience will indeed continue to build on new discoveries and developments in fields such as genomics.
Basic knowledge about cell walls can also be used to reconstitute or, more likely, modify the cell wall to make it less recalcitrant to processing for energy production. This may happen by altering existing pathways in plants or introducing new ones that lead to the creation of less recalcitrant polysaccharides or by altering lignin-polysaccharide interactions (Albersheim et al. 2010). The creation or modification of cell walls with altered chemical compositions or properties will draw on such techniques as genetic engineering. Although the scientific and economic drivers for these developments are clear, the potential ecological impacts and social perceptions of genetic engineering will need to be assessed as part of these and other biofuels research efforts.
Cell wall recalcitrance manifests itself at surfaces where microbial enzymes and the plant cell wall interact (Himmel et al. 2007). Given that both the plant cell wall and microbe surfaces are composed primarily of glycans, glycoscience will play a central role in understanding these interactions. While understanding how isolated enzymes or other catalysts interact with the plant cell wall as part of deconstruction is an important avenue of investigation, investigations are also under way to determine whether microbes can be more effective than enzymes at deconstructing the plant cell wall for lignocellulose feedstocks (Lu et al. 2006; Olson et al. 2011). Theories being explored include the suggestion that microbes alter the structured layers of water that form along the cell wall surface. This water layer may present a barrier to the approach of free enzymes and
inhibit the escape of soluble products, which in turn would slow hydrolysis rates. Thanks to 1 billion years of evolution, microbes have surfaces that may enable them to disrupt this water layer, effectively solubilize the plant cell wall, and capture solubilization products. Molecular mechanics simulations appear to support this hypothesis (Himmel et al. 2007; Ding et al. 2008; Lynd 2010). Manipulating or modifying such microbes may lead to the generation of more effective microorganisms for consolidated bioprocessing (Olson et al. 2011).
Another challenge in creating biofuels from plant biomass is to increase production of biomass per hectare and to increase the yield of fermentable sugars from each ton of biomass. Increased production can be done by choosing the right crop for a given set of environmental conditions and by increasing the resource efficiency of these plants by decreasing water and fertilizer needs (Tilman et al. 2009; Lynd and Cruz 2010). From a biological perspective, one possibility is delaying the time when plants flower. Keeping plants in a juvenile stage will also increase biomass production. At least one research team has found that introducing a micro-RNA into a plant that caused it to remain in its juvenile stage increased biomass production but also caused the plant to reduce lignin production, leaving the resulting biomass more susceptible to degradation and conversion (Chuck et al. 2011).
3.2.3 Key Messages on Glycoscience and Energy
There is increasing interest in how to make use of the carbon in plant biomass as an alternative source of energy to petroleum. However, plants have evolved to resist destruction by the environment and by microbes, and so efficient deconstruction of cell walls into carbohydrates and sugar intermediates that can be fermented into biofuels is a challenge. Understanding how plant cell walls are synthesized and constructed and, conversely, how they can be effectively broken down remains a major goal for improving biomass-derived energy. Addressing this challenge will require continued research into the cell wall formation and breakdown process and forms an important part of the glycoscience field.
As a result, the committee finds that:
- Plant cell walls, made mostly of glycans, represent the planet’s dominant source of biological carbon sequestration, or biomass, and are a potentially sustainable and economical source of non-petroleum-based energy.
- Understanding cell wall structure and biosynthesis and overcoming the recalcitrance of plant cell walls to conversion into feedstocks that can be transformed into liquid fuels and other energy
-
sources will be important to achieving a sustainable energy revolution. Glycoscience research will be necessary to advance this area.
- Glycoscience can also contribute significantly to bioenergy development by advancing the understanding of how to increase biomass production per hectare and how to increase the yield of fermentable sugar per ton of biomass.
3.3 GLYCOSCIENCE AND MATERIALS
As discussed above, plant cells walls represent the planet’s dominant biological carbon sequestration system. They are estimated to account for 120 billion to 170 billion tons per year. In comparison, the annual global production of chemicals, including fertilizers but not pharmaceuticals, was 1.2 billion tons in 2010. In other words, it takes plants three days to produce biomass in the form of cell walls equal to the total annual output of the world’s chemical industry (DOE 2011).
Many of today’s most widely used materials are petroleum based. With the world’s population growing, consumer-based economies expanding, and petroleum resources ultimately being stretched in many directions, there is a global need for the development of renewable and sustainable resources that can serve as the source of a new generation of materials. This is a surmountable task, one that could be at least partly accomplished using polysaccharides produced by a wide variety of living species, including plants, algae, fungi, and even insects and arthropods. This diverse collection of organisms produces sufficient quantities of harvestable polysaccharides to potentially meet the world’s demand both for energy and synthetic materials (Perlack et al. 2005). Investigating glycan-based materials further opens the possibility of designing new materials with tailored properties that can expand on the range of materials currently available and may find new applications.
Materials based on polysaccharides, particularly cellulose-based polysaccharides, have a long history as a source of functional materials used by human society. Wood, cotton, linen, hemp, and other cellulose-based polysaccharides have been used as engineering materials for thousands of years, and their use continues today, as evidenced by the enormity of the worldwide industries in forest products, paper, and textiles, among others. As a chemical raw material, cellulose has been used for about 150 years (Klemm et al. 2005). Regenerated cellulose has been used in the processing of synthetic cellulose films (cellophane) and fibers (rayon), while cellulose derivatives, made, for example, by replacing hydroxyl groups with other functional groups, have been used to produce a wide variety of cellulosic polymers, including cellulose esters and cellulose ethers, which
have many industrial and pharmaceutical applications. Commercial products made from cellulose derivatives include coatings, inks, binders, thickening and gelling agents, and controlled-release drug tablets (Klemm et al. 2005). Recently, the availability of cellulose-based nanoparticles has begun changing the paradigm of what is achievable with natural materials, including the production of biodegradable transparent films that are stronger than steel (Moon et al. 2011). For two examples, see Box 3-3.
Further advances in glycoscience could drive the discovery of a wide range of new sustainably produced polysaccharide-based materials in three general categories: fine chemicals and feedstocks, polymeric materials, and nanomaterials. Glycoscience has a real opportunity to positively impact the progress of the baseline technologies needed to develop cost-effective materials from polysaccharides that can compete, in terms of cost and performance, with petroleum-based chemicals and polymers. This vision can be achieved by providing mechanisms to develop an increased understanding of polysaccharide biosynthesis in plants and trees, new characterization tools and methods for understanding polysaccharide structure, new methods for polysaccharide isolation, synthetic process and chemical modification, and improved predictive modeling capabilities.
3.3.1 Fine Chemicals and Feedstocks
A variety of polysaccharides are being investigated in the production of functional chemical precursors that are then subsequently used to make industrially relevant chemicals and engineering polymers (Bozell and Petersen 2010). Examples of functional chemical precursors include alcohols, such as ethanol, propanol, butanol, xylitol, and sorbitol; furans, such as furfural and hydroxymethylfurfural; biohydrocarbons, such as isoprene and long-chained hydrocarbons; and organic acids, such as lactic acid, succinic acid, and levulinic acid. Carbohydrates are one of a handful of natural products that can be used for production of many chiral compounds with defined stereochemistries. Research has focused on optimization of the bioconversion of polysaccharides in terms of yield, rate, separation, titer, and product specificity. Much of this work has focused on identifying and engineering improved fermentation organisms, fermentation processes, and catalysts for converting sugars into chemical precursors (Bozell and Petersen 2010). Significant advances will require further development of methods to improve our ability to engineer specific enzymes and organisms, such as yeast and fungi to produce large yields of single materials. Advances in materials development will also require a better understanding of how to achieve specific chemical
BOX 3-3
Examples of Carbohydrate-Based Materials: Flexible Displays and Artificial Blood Vessels
Polysaccharides can be used in the development of a wide range of new materials with very diverse properties. For example, cellulose nanocomposites from wood can be used as the basis for flexible, optically transparent materials that can be used as substrates in the creation of luminescent organic light-emitting diodes. Such materials have the potential for new applications in flexible electronics and displays. Cellulose, in this case derived from bacteria, can also be used to make thin, flexible tubes for use as implanted blood vessels.

Glycan-based materials have multiple uses. SOURCES: Top, Reprinted from Okahisa et al. 2009, with permission from Elsevier; Botttom, Klemm et al. 2011, used with permission.
reactions with organisms and catalysts in a way that enables selective reductions, conversions, and chemical bond formation or bond breaking.
As discussed earlier, polysaccharides represent a broad range of natural polymers consisting of repeating sugar monomer units joined together by glycosidic bonds. They can be linear or highly branched and can have a wide variety of side groups. As a result of the variety of polysaccharides produced in living species through their biosynthesis processes, the diversity of polysaccharide materials available in nature creates an almost limitless range of possibilities for creating useful new materials. For example, trees produce cellulose and hemicelluloses, whereas nonwoody plants produce cellulose, pectins, and starches, and various bacteria can synthesize polysaccharides that include glycogen, alginate, xanthan, dextran, curdlan, gellan, colanic acid, K30 antigen, hyaluronic acid, and cellulose (Rehm 2010). Each of these polysaccharides has a unique chemical structure comprising different combinations of sugars linked together in different configurations, all of which influence the properties of the given polysaccharide in terms of its structural configuration, thermal stability, reactivity, rheology, and mechanical properties.
With these natural materials as starting points, extensive research has gone into the development of new reaction pathways that modify the existing polysaccharide backbone structure or the side groups that branch off the backbone. The goal of these efforts is to create new polymeric materials with novel properties and functions (Klemm et al. 2005; Roy et al. 2009). For example, cellulose can be dissolved and the glucan chains reassembled to produce regenerated cellulose, or cellulose II, which has been used to produce cellulose films, known as cellophane, and fibers including rayon. Research has also yielded reaction pathways that create cellulose derivatives such as cellulose acetate, cellulose acetate propionate, cellulose acetate butyrates, carboxymethyl cellulose, cellulose butyrate succinate, and cellulose acetate propionate (Klemm et al. 2005). Each of these cellulose-based polymers was created by replacing accessible hydroxyl groups with other chemical groups to produce a material with novel performance characteristics. Given the wide range of polysaccharides produced by nature, these examples represent only a fraction of the potentially novel and useful materials that could be produced. But the complexity and variability of polysaccharides represent a challenge that requires fundamental research to develop:
- faster and more accurate methods for structural characterization of polysaccharides,
- improved technology for separating and isolating polysaccharides from their natural sources,
- novel chemical reactions to target chemical modifications to specific locations on the polysaccharides in order to create regioselective functionalization, and
- synthesis pathways for long-chain polysaccharides.
Polysaccharides with linear or minimally branched backbone structures can self-assemble into ordered bundles in which the polymer chains stack in parallel with each other along the chain axis. Such parallel-stacked chains from a crystalline structure that can be characterized by x-ray, neutron, and electron scattering techniques, among others. Two primary examples of polysaccharides that show this crystalline structural behavior are cellulose and chitin, a polysaccharide isolated from fungi and from the exoskeleton of crustaceans and insects. The structures of cellulose and chitin are similar, the difference being that chitin has one hydroxyl group on each sugar replaced with an acetyl amine group. However, the two polysaccharides have considerably different physical and mechanical properties. Nonetheless, during the biosynthesis process, both of these linear polysaccharides form fibril structures containing both crystalline and amorphous arrangements of polymer chains. These fibrils serve as the base reinforcement unit that provides the high mechanical strength, strength-to-weight ratio, and toughness of plants, trees, crustaceans, and insects.
Using specialized chemical-mechanical extraction methods, these fibril structures and their crystalline regions can be isolated and used to develop the next generation of plastics. To date, most work has been completed with cellulose, largely because of its availability and the extensive scientific and technological expertise developed by the pulp and paper industry. With this in mind, the next sections focus on cellulose to illustrate some of the opportunities for creating such nanomaterials, along with the associated challenges. The issues discussed are relevant to other ordered polysaccharides as well.
For cellulose the particles that are isolated after chemical and mechanical extraction have dimensions on the nanoscale and are generically called cellulose nanomaterials (CNs) (see Figure 3.4). CNs can have either a rod and whisker–like structure or a fibril particle morphology, with the dimensions varying depending on cellulose source, extraction methods, and extraction conditions (Habibi et al. 2010; Moon et al. 2011). Typically, CNs range from 3 to 30 nanometers (nm) in diameter and from 50 nm to several microns (μm) in length, with length-to-width (aspect) ratios of 20
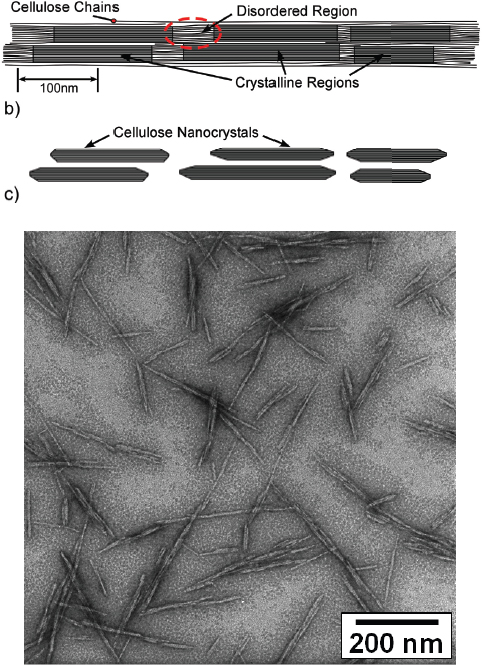
FIGURE 3-4 Cellulose nanocrystals. Top, Stacking of cellulose chains showing areas of “order” and “disorder.” During one type of cellulose nanomaterial extraction process that uses acid hydrolysis, the amorphous regions are preferentially dissolved and only the crystalline regions are left. Bottom, Transmission electron micrograph of cellulose nanocrystals produced by acid hydrolysis. SOURCE: Moon et al. 2011, used with permission.
to 100. CNs have high stiffness, low density, low thermal expansion, and thermal stability up to about 200 to 300oC, and their surfaces can be readily modified using a variety of chemical methods.
The sources from which CNs are extracted are themselves sustainable, biodegradable, and carbon neutral, and they generally have low environmental, health, and safety risks. CNs have the potential to be processed at industrial-scale quantities at low cost, although reaction conditions, feedstock crystallinity, and other factors influence achievable yield (Habibi et al. 2010; Qua et al. 2011). Preliminary testing shows minimal environmental, health, and safety risks for CNs (Vartiainen et al. 2011), although investigations into the environmental and health effects of all types of nanoparticles continue to be an area of research and discussion.
With these advantages in mind, CNs are being considered for use in the development of a variety of new plastics and composite structures, including films, fibers, aerogels, and hydrogels. These materials have potential applications in barrier films, separation membranes, antimicrobial films, transparent films, flexible displays, cardiovascular implants, wound and burn dressings, tissue regeneration scaffolding, drug delivery vehicles, fibers and textiles, templates for electronic components, batteries, supercapacitors, electroactive polymers, and body armor.
Glycoscience could have a transformative impact on the CN bioplastics industry by advancing the technologies necessary to control the crystalline structure, properties, surface chemistry, particle morphology, and particle size distribution of CNs. Such technologies could then be used to produce “tailored” CNs to meet specific performance metrics at low cost. This vision can be achieved with advances in the following areas, each of which are described in the sections that follow:
- understanding cellulose nanomaterial extraction processes,
- CN characterization,
- atomistic modeling of cellulose, and
- cellulose synthesis.
3.3.3.1 Understanding cellulose nanomaterial extraction processes
Unlike the extractions generally needed to produce fine chemicals, feedstocks, and new polymers, the techniques needed to extract completely ordered polysaccharide particles from natural cellulose source materials demand particular delicacy, and developing suitable processing technologies requires new techniques and methodologies. Extracting CNs from cellulose requires several steps, each of which influences particle morphology or shape, particle size and size distribution, and interfacial properties (Moon et al. 2011). Current processing technologies afford min-
imal control of each of these physical properties, which in turn affects the performance traits of the resulting nanoparticles. Because they are derived from cellulose, a better understanding of the mechanisms involved in plant cell wall construction and destruction can help provide insight into new mechanisms that could be exploited to improve CN extraction methods to decrease the internal damage in CNs, narrow the particle size range for a given CN processing methodology, improve extraction efficiencies, increase CN yields, and scale production to industrial quantities.
3.3.3.2 Cellulose nanomaterial characterization
In addition to the structural and chemical characterization needed to produce fine chemicals, feedstocks, and new polymers, novel structural, chemical, and mechanical characterization techniques are needed for ordered polysaccharides. In particular, there is a need for techniques that can characterize the configuration of the parallel-stacked polysaccharide chains found in CNs. The properties of any given CN depend on the arrangement of the cellulose chains, defects in the ordering of chains, local changes in the chemistry internal to the CN, and changes in the chemistry on the external CN surface. Characterizing these aspects is important for understanding CN properties and for developing the means to rationally design new CNs with specific properties. In addition, improved characterization of structure, nanomechanical properties, and surface chemistry will provide the opportunity to better understand processing-structure-property relationships as they relate to the CN particles themselves and to CN-CN and CN-water interactions, all of which are important for the design of composite materials with improved performance.
While the cellulose polymorph structures of CNs are generally known, characterization of individual CNs is currently lacking in such areas as the percentage of crystallinity, the location of amorphous regions on the CN surface or throughout the CN core, the fraction of a given cellulose polymorph structure and its location in the particle, the identification of defects such as missing cellulose chains, and the hydrogen bonding networks both within and between cellulose chains in the CN. Characterizing the elastic and tensile strength and other nanomechanical properties of CNs remains a huge challenge, because their small size pushes the limits of sensitivity of current methodologies such as atomic force microscopy (Wagner et al. 2011). While the reported properties for CNs are on par with atomistic model predictions, these models are too variable for conducting fundamental research on structure-property relationships, let alone for assessing the influence of different extraction processes or cellulose source on the quality of the resulting nanomaterial. Also lacking is the ability to pinpoint the particular location of a given chemically func-
tionalized side group along the CN surface. This information is important to assess the accessibility of the given side group and how these groups can interact with neighboring CNs or with the surrounding liquid in CN-liquid suspensions.
3.3.3.3 Atomistic models of cellulose
New methods for atomistic modeling can contribute to better understanding of CN properties by providing insights into how to characterize and describe the configuration of the parallel stacking of the polysaccharide chains and how that configuration affects assembly properties. When linked with experiments for direct comparison and validation, atomistic modeling is a useful tool to help develop a better understanding of the structure, mechanical properties, and surface chemistry of nanomaterials such as CNs. Atomistic modeling can also provide information on the interactions of CNs with neighboring nanoparticles, which affects the composite properties of a CN-based material, and the interactions within a particle suspension, which affects rheology. In addition, atomistic models that could accurately represent surface interactions and preferential bonding site location would provide insights into composite properties and aid efforts to tailor CN properties.
3.3.3.4 Cellulose synthesis
Today, it is particularly challenging to synthesize ordered polysaccharides with the desired surface chemistry that self-assembles with controlled crystalline properties. Through detailed understanding of the polysaccharide biosynthesis process, it will ultimately be possible to develop processing routes that facilitate tailoring both the side-group chemistries of the polysaccharide backbone structure and the assembly of individual chains into an ordered chain structure.
3.3.4 Key Messages on Glycoscience and Materials
Plastics and many other materials are currently derived from petroleum resources. Carbohydrates derived from plants, microorganisms, insects, and other biological sources can provide a wealth of new options for materials with diverse chemical and physical properties. Glycoscience knowledge is helping unlock the development of these new materials and the control of their design. The example of cellulose nanoparticles was described in greater detail previously in this section to help illustrate some of the research questions involved in characterizing and designing new materials derived from carbohydrates. However, the need to under-
stand chemical structures and interactions, to characterize and modify materials, and to control production processes is important to developing functional and economically feasible new materials and products more broadly. The application of glycoscience to materials science and engineering represents an expanding area for the field and one for which continued research and development will be needed.
As a result, the committee finds that:
- By fostering a greater understanding of the properties of glycans and of plant cell wall construction and deconstruction, glycoscience can play an important role in the development of nonpetroleum-based sustainable new materials.
- Glycan-based materials have wide-ranging uses in such areas as fine chemicals and feedstocks, polymeric materials, and nanomaterials.
- There are many pathways to create a variety of functionalities on a glycan, creating a wide range of options for tailoring material properties.
This chapter explores three significant ways in which carbohydrates contribute to society—in human health, in energy, and in materials. Glycans are closely linked to both normal physiological function and to the genesis and development of disease. They play promising roles in the discovery of new diagnostic biomarkers for such diseases as cancer and for new therapeutic targets. But glycans and glycoscience also have important roles to play in the improved conversion of biofuels and the design and creation of new carbohydrate-based materials. As a result, glycoscience knowledge will contribute to the development of new energy and materials science solutions that can replace some of the roles currently played by petroleum-based products. This chapter attempts to provide a sense of some of the unanswered questions in glycoscience as well as the exciting potential that may be realized through future research. The next chapter continues this discussion by exploring a set of questions that we may not yet have the necessary tools to fully address but the pursuit of whose answers remains an exciting challenge.


































