The third session of the workshop centered on models of the human embryo based on stem cells. Panelists described the latest scientific and technical developments with models of human embryos and considered directions for future research and applications of this work. They also examined the fidelity of human embryo model systems to bona fide human embryos and explored methods for validation of the model systems. The session was moderated by Arnold Kriegstein, a professor of neurology and the director of the developmental and stem cell biology program at the University of California, San Francisco.
GASTRULOIDS: MODELING HUMAN EMBRYOS AND EMBRYONIC TISSUES
Ali Brivanlou, the Robert and Harriet Heilbrunn Professor at The Rockefeller University, introduced synthetic self-organizing models of human embryos called “gastruloids” and highlighted lessons about the basic science of human development that can be gleaned from these models. From a utilitarian perspective, he also made the case for the use of synthetic models as a tool for addressing diseases whose roots can be traced to very early human embryonic development. He began by describing the rationale for human embryological studies more broadly (see Box 4-1).
In Vitro Attachment and Self-Organization in Early Human Embryonic Development
Brivanlou opened by presenting a series of images to trace the development of the embryo over the first 2 weeks of development, noting that very few pictures are available that represent this early developmental
stage. At day 8, the in vitro attached human embryo displays an amazing ability to self-organize in radially symmetrical patterns. The inner cell mass (ICM) can be observed with the trophectoderm around it. He said that his assumption had been that as soon as the embryo attaches, inductive interactions between maternal tissue and embryonic tissue would be required for the proper induction and patterning of cell fate, so observing this happening independently without apparent extrinsic influences was unexpected for him. At day 12, this self-organization continues to progress, with increasingly aesthetically attractive attributes. He highlighted the ratio between trophectoderm and the ICM at day 10 and then at day 12, noting that a large fraction of the attached embryos lack epiblast cells. Again, he emphasized the aesthetic features of this self-organization: the cells are radially patterned, and the yolk sac and amniotic cavity are nicely proportioned with similar numbers of cells.
Rationale for Human Embryo Models
Motivated by these types of observations about in vitro attachment and self-organization in early human embryonic development, Brivanlou and colleagues began exploring the extent to which knowledge gained from model systems is valid in humans by investigating whether these types of attributes are (1) human-specific or (2) attributes that have been evolutionarily conserved. In exploring these questions, they were able to detect human biological features that had never before been identified in the embryo of any other mammalian species. For example, they discovered the novel human cell type yolk sac trophectoderm (ysTE), which has a unique molecular signature (OCT4LO GATA6LO GATA3+ CDX2+) never before seen in any other mammalian embryo (Deglincerti et al., 2016; Zhou et al., 2019). This type of discovery underscores the importance of working with human tissue, he said.
Brivanlou delved further into the rationale for studying human embryo models—“gastruloids”—that are derived from human embryonic stem cells (hESCs) (Warmflash et al., 2014). One reason for using human embryo models is the 14-day limit.1 However, even in the absence of such a guideline, there will never be sufficient sources of biological material available to thoroughly investigate our human origins, such as the molecular basis of human cell pattern formation and the emergence of organs and the tissues, Brivanlou said. Surmounting this barrier will require developing model systems that can provide the large numbers of biological samples that are required to conduct this work. Mouse models and nonhuman primate
___________________
1 The 14-day rule refers to a policy that restricts the culture of human embryos past 14 days of development.
models have value, he said, but making meaningful progress will require exploring species-specific facets of development with human models. From a utilitarian perspective, clinical breakthroughs are dependent on this type of research, particularly the development of therapies for diseases with a developmental origin and especially for those diseases that develop after the 14-day point. Model human embryos can facilitate those types of clinical breakthroughs for diseases that affect the early embryo or fetus—which he characterized as long overdue—without the need for human embryos. “The only way to move forward where that system has mostly failed us is to bite the bullet and move forward with human studies,” Brivanlou said.
Creating Model Human Gastruloids Derived from Human Embryonic Stem Cells
Brivanlou described how these model human gastruloids are created using hESCs. When the hESCs are cultured under standard conditions, the variations in size and density of colonies—some colonies are touching other colonies, some colonies are completely independent of one another—and the presence of individual randomly dispersed cells make it impossible to replicate the system and quantify it at a subcellular level. This may be a reason why different laboratories performing the same experiment generate inconsistent results under identical experimental conditions, he noted. To address this issue, Aryeh Warmflash, who would speak next in the session, created a process for imposing a standard on hESCs by developing micropattern technology whereby the hESCs would only adhere to a circular substrate (85–1,000 microns in size) (Warmflash et al., 2014). At a minimum, this allows for imposing size and dimension homogeneity in the culture, with the potential to bring in more quantitative approaches as well. Warmflash and colleagues also developed the programming technology to track and evaluate single-cell resolution response to a given stimuli. When this process is performed over thousands of cells and hundreds of colonies, cell behaviors can be measured in a way that is highly reproducible. This technology has moved forward in Brivanlou’s laboratory, where they are applying deep neural networks and artificial intelligence to track the behavior of these cells.
Self-Organization of hESCs into Gastruloids by Geometrical Confinement in Response to Bone Morphogenetic Protein 4
Brivanlou described a seminal result of those first experiments with geometrical confinement (Warmflash et al., 2014). When the hESC colony maintained in pluripotency media was stimulated only with BMP4 for 48 hours, the result was dramatic. The hESC colony self-organized into
gastruloids containing ectoderm (SOX2), mesoderm (BRACHYURY, or BRA), and trophectoderm (CDX2). This can be reproduced over many thousands of colonies with exactly the same precision. Brivanlou maintained that this process of geometrical confinement settles any debate over whether bone morphogenetic protein 4 (BMP4) induces trophectoderm or mesoderm. Geometrical confinement is sufficient to give rise to all the cell types in response to a single stimulus. “Confinement of geometry … can induce everything as long as you give it an edge,” he said. This holds true not only for ectoderm, mesoderm, and extraembryonic tissue, but also for endoderm (SOX17), primitive streak (zinc finger protein SNAI1, or SNAIL), and primordial germ cells (B-lymphocyte-induced maturation protein 1, or BLIMP1). The embryo contains no cell type that does not self-organize in this type of symmetry, he added. He explained that this powerful tool has enabled new avenues of research into how cell fate is established in these self-organizing patterns. It also eliminates the challenge of embryo-to-embryo variation because the process creates homogeneity not only in size, density, and cell number, but also in the cells’ genotypes.
Lessons from Studying Human Gastruloids
Brivanlou highlighted several lessons learned by studying human gastruloids that would be impossible to glean from studying human embryos within the current guidelines. The first is that gastruloids self-organize through mechanisms of edge sensing and reaction diffusion (Etoc et al., 2016). Cells establish their fate in these circular colonies by measuring their distance from the edge, he explained. This can be demonstrated with various techniques, including the use of CRISPR/Cas9, to show that the wave of transcriptional response patterning starts from the edge and moves inward to crash in the center (i.e., the reverse of throwing a pebble in water). The second lesson is that Wnt signaling memory is required for Activin to function as a morphogen (Yoney et al., 2018). History matters for cells’ fate, Brivanlou explained. A cell knows who it is in the gastruloid based on the history of signaling it has been exposed to. This could not have been learned without manipulating the sequence in which the ligand stimulus is presented at different times. The third lesson is that a wave of Wnt signaling controls primitive streak formation downstream of the BMP4 signaling, which can be functionally demonstrated using grafting in a chick embryo (Martyn et al., 2019). Single-cell RNA sequencing of gastruloids can reveal novel human-specific molecular signatures, he said. For example, his laboratory has recently discovered a group of genes in humans that have never before been associated with primordial germ cells. Among the most unexpected was DOCK2, a gene that previously was associated only with the migration of white blood cells and lymphocytes from the blood cells out
to the target. This finding suggests that the early migration of primordial germ cells in humans follows pathways and molecular hierarchies that are already known, he said, which creates the potential for developing interventions in the future.
Near Horizon: Three-Dimensional Gastruloids and Symmetry Breaking
The two-dimensional (2D) embryo modeling systems are useful, Brivanlou said, but the human embryo is three dimensional (3D). Fortunately, a generation of 3D gastruloids that undergo symmetry breaking is on the near horizon. A method has already been developed to change substrate stiffness in 3D colonies to generate what looks like primitive endoderm (GATA6) and trophectoderm (GATA3) (Deglincerti et al., 2016; Simunovic et al., 2019). The method involves dissociating the cells and combining them with a 3D epiblast grown in a combination of Matrigel and hydrogel. High-fidelity modeling is then used to monitor the development. At day 1 post-mixing, the geometries of epiblast, amniotic cavity, and yolk sac cavity that emerge in the in vitro attached embryo appear similar to embryos developed in vivo. After 3 days, the phenomenon of symmetry breaking—indicated by GATA3, BRA, and SOX2—was observed in the self-organizing 3D embryos. Work is under way to validate this model by comparing 3D embryos to real embryos attached in vitro at day 8, he said. Morphologically they appear similar; their molecular signatures indicate that they are close but not identical. Single-cell RNA sequencing is in progress to compare the specimens and evaluate the similarities and differences between their respective cell types in the hope that new cell types might be identified.2
Modeling Human Disease in Gastruloids and Neuruloids
In the final part of his presentation, Brivanlou commented on the application of human embryo models to cure human diseases. This utilitarian aspect is important to consider, particularly in the context of using public funding, he said. To explore whether diseases can be modeled in gastruloids, his laboratory decided to target Huntington’s disease. It is relatively straightforward to investigate because it is caused by a single dominant mutation in the HTT gene.3 It is considered to be a neuro-
___________________
2 Brivanlou noted that the ysTE discovered in the attached human embryo does not exist in the model embryo, which is a positive finding.
3 The mutation that results in Huntington’s disease causes a polyglutamine (polyQ) expansion in the HTT protein.
degenerative disease and currently has no therapy or cure. Despite the fact that it was the first gene cloned with 100 percent penetrance for the state of the disease, model systems have failed to accurately replicate the disease thus far. The discovery of an isoform of Huntington’s disease only present and expressed in hominids—but not marmosets or lower primates—suggests that the reliance on mouse or lower nonhuman primate models may be a reason that the model systems have failed (Ruzo et al., 2018). That is, they may have been working with systems that do not represent the molecular signature of the target disease. To investigate, Brivanlou’s laboratory generated a series of isogenic human ESC lines to introduce the Huntington’s disease mutation in gastruloids. Immediately it created a phenotypic signature that was also dependent on the length of the repeating CAG (cytosine-adenine-guanine) section segment in the gene (i.e., gastruloids with different insertions displayed related but different phenotypes). This finding suggested for the first time that Huntington’s disease is actually a developmental disease, not a neurodegenerative disease. The center section of gastruloids can be used to generate another type of self-organizing structure called neuruloids, which can also be used to model Huntington’s disease. Neuruloids are self-organizing ectodermal structures that display radial distribution of cell types, with brain cells at the center, surrounded by neural crest and neural placodes (sensory organs) and epidermis covering the entire structure. Neuruloids also display a specific phenotypic signature for Huntington’s disease. The researchers are now selecting for compounds that can rescue the phenotype from the neuruloid and gastruloid back to the non-Huntington’s disease phenotype. High-throughput screening has identified five compounds that can rescue the phenotypes seen in Huntington neuruloids, and the researchers are ready to move to the next stage of medicinal chemistry. Brivanlou added that similar disease modeling is in progress for fragile X (FMR1), tuberous sclerosis (TSC1 and 2), and autism (MECP2). Because neuruloids can be pushed to self-organized circuitry, they are being used to look at psychiatric diseases and circuitries in different tissues and organs. Another line of research is using these models to look at infectious diseases such as Zika virus, HIV, and cytomegalovirus 1, in which the fetal and early embryo are known targets of the viral infection.
MODELING HUMAN DEVELOPMENT IN 2D WITH MICROPATTERNS
Aryeh Warmflash, a principal investigator and an assistant professor in the Department of Biosciences at Rice University, discussed how human development can be modeled in 2D using micropatterns. He offered insights into how gastruloid systems generate patterns and considered ways for-
ward to generate models that can help to answer specific developmental questions.
Geometric Confinement Triggers Self-Organized Patterning Along the Colony Axis
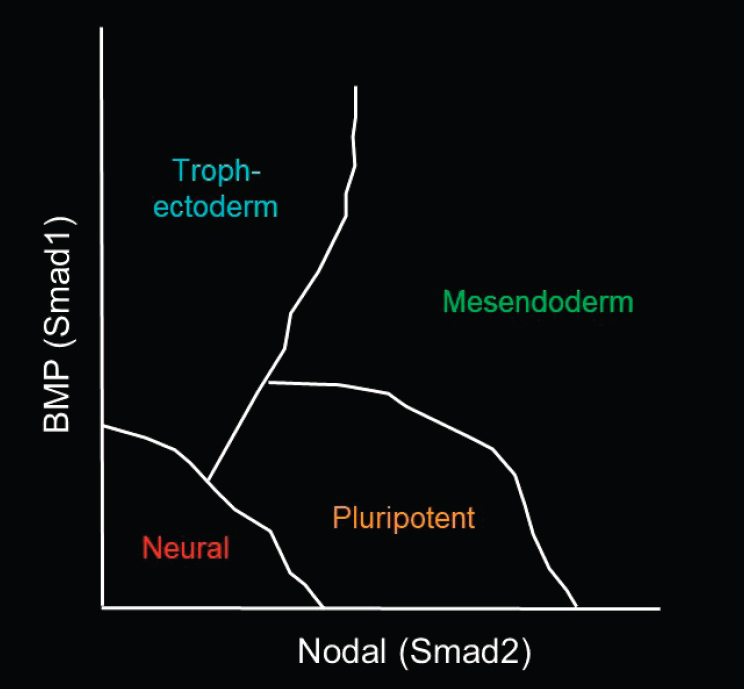
As Brivanlou described, Warmflash and his colleagues found that by geometrically confining human ESCs and adding a single growth factor, BMP4, the cells self-organize into trophectoderm-type cells with all three germ layers—endoderm, mesoderm, and ectoderm (Warmflash et al., 2014). His laboratory is now seeking to understand how the cells actually generate those patterns. He presented Figure 4-1 to illustrate the functions of the
SOURCE: Aryeh Warmflash workshop presentation, January 17, 2020.
signaling pathways that are thought to be involved in differentiating hESCs to each germ layer. BMP (Smad1) is thought to push cells toward the trophectoderm, and some Nodal (Smad2) signaling is required to maintain the pluripotent state. Up-regulating Nodal signaling, together with Wnt, can generate mesoendoderm cells. Down-regulating all of those signals generates neural cells, a subset of the ectodermal germ layer. Warmflash noted that the specific fates within each germ layer can depend on other signals as well. The question is how all of those cell fates are generated along the y-axis (or the BMP signaling) of the diagram in the figure.
Measuring Signaling Dynamics of Key Morphogen Pathways
Warmflash explained how CRISPR-engineered reporters can be used to measure the signaling dynamics of key morphogen pathways, the TGFβ superfamily (BMP and Nodal) and Wnt. It has been established that both Wnt and Nodal signaling are required downstream of BMP4 in order to pattern the gastruloid (Chhabra et al., 2019; Martyn et al., 2018; Warmflash et al., 2014). Time-lapse imaging of hESCs with CRISPR-engineered reporters can be used to create “movies” to watch how these endogenous signals, which are activated downstream of BMP, play out in space and time to generate the patterns observed in hESC colonies. He noted that this technology allows for toggling between sparser culture conditions, in which the cells are mostly responsive to exogenous signals that can be well controlled, and denser micropattern systems, in which the primary controls are endogenous signals.
For the TGFβ superfamily, CRISPR/Cas-tagged alleles were generated for the signal transducer Smad4, which is downstream of both BMP and Activin/Nodal (Heemskerk et al., 2019; Nemashkalo et al., 2017). For the Wnt pathway, β-catenin was tagged to watch Wnt signaling (Massey et al., 2019). Warmflash’s team found that BMP signaling is sustained over time (as long as BMP is not inhibited) and that this sustained signaling is necessary for differentiation of cells to CDX2-positive trophectoderm fates (Heemskerk et al., 2019; Nemashkalo et al., 2017). Wnt and Activin/Nodal signaling behave differently than BMP signaling in that they respond adaptively to ligand stimulation, Warmflash said. The Wnt reporter β-catenin starts out at the adherence junctions, and fills the cells when Wnt is added. Then the cells respond adaptively. Despite the constant presence of Wnt in the media, the cells stop Wnt signaling to various degrees; this adaptation is complete at low doses and partial at higher doses (Massey et al., 2019). Activin/Nodal also creates an adaptive signaling response, but with less dependence on the ligand dose—the cells adapt almost completely within about 4 hours, independent of the dose (Heemskerk et al., 2019).
To understand how these patterns are generated, Warmflash and his colleagues looked into the hESC colonies where these signals play out. Warmflash noted that BMP signals are exogenous, but Wnt and Activin/Nodal signals are endogenous. He presented a movie quantifying what happens when a cell colony is treated with BMP4. BMP signaling is initially widespread, but it is then restricted to the colony border where the signaling is sustained (Heemskerk et al., 2019). Initially, the entire colony responds to this high dose of growth factor, but then there is a hollowing out at the center of the colony, with the signal only sustained at the edge, he said. This is consistent with the finding that sustained BMP signaling is needed to generate CDX2-positive trophectoderm. Because BMP signaling is only sustained at the edge of the colony, CDX2-positive trophectoderm can only be generated there. Another movie demonstrated the dynamics of Wnt signaling, which is initially activated close to the edge and spreads inward, with linear propagation of the signal through the colony at a constant rate (Chhabra et al., 2019). In other words, a wave of Wnt signaling moves through the colony from the edge inward. Wnt signaling reaches well beyond the region of mesoderm differentiation, he added. Following the wave of Wnt signaling, Nodal is activated. A wave of Nodal signaling then propagates through the colony and induces mesodermal differentiation in its wake in a certain region of the colony (Heemskerk et al., 2019).
Warmflash summarized this sequence of events in the following way: (1) BMP signaling is initially widespread but then hollows out; (2) a wave of Wnt is triggered through the colony; (3) a wave of Nodal signaling follows the wave of Wnt; and (4) mesoderm differentiation occurs immediately after the wave of Nodal signaling. Both Wnt and Nodal signals reach farther into the colony than primitive streak differentiation, so the signal that controls the positioning of the streak cannot be a simple threshold in either of these pathways. Instead, the cells combinatorially interpret the dynamics of both pathways in making a decision. One hypothesis is that the order in which the cell sees these signals is important, he added. If they see the Wnt signaling and then the Nodal signaling, they will differentiate. If they see the Nodal signaling and then the Wnt signaling, they will not differentiate. This is consistent with data from other laboratories showing directly that the Wnt signal needs to precede the Nodal signal for differentiation.
Molecular Mechanisms That Propagate the Signaling
Warmflash and colleagues have also gained insight into the molecular mechanisms by which these signals propagate. Inhibitor of Wnt Production-2 (IWP2), a small molecule inhibitor of Wnt secretion, is a useful reagent because it inhibits the secretion of Wnt signaling without inhibiting the response to Wnt signaling. Adding IWP2 at different times
reveals how the signal propagates through the colony (Chhabra et al., 2019). Unsurprisingly, the earlier that IWP2 is added, the less Wnt signaling is observed. However, they were surprised to find that the Wnt signal travels just as far when the inhibitor is added, even though there is not as much Wnt signaling and new Wnt cannot be produced. This suggests that Wnt is produced near the edge of the colony and then moves inward into the colony via some means of active transport, in a way that is linear in time and does not require production of new Wnt. Therefore, the secretion of new Wnt ligands is dispensable for movement of Wnt signaling, but it increases Wnt levels. Nodal behaves differently when Wnt is inhibited, he said. Wnt is needed to activate the Nodal signaling, but at some point the Nodal signaling becomes self-propagating and independent of upstream pathways, activating itself as it goes through the colony. Nodal has a binary-type phenotype: either it is not activated and stays mostly at the edge, or it is activated everywhere.
Differences Between Micropatterned Cultures and the Embryo
Warmflash observed that the insights they have gained about cell differentiation from dissecting these signals, observing how they regulate each other, and tracking how they propagate through the colony in time would be difficult to obtain using any in vivo system. He provided a brief overview of the differences between the types of micropatterned cultures he described and an actual embryo.
- In micropatterned cultures the germ “layers” that form are adjacent in the same 2D plane, rather than layered in 3D.
- The hESC colonies fail to break symmetry: rather than having a primitive streak that forms on the posterior side, as in an embryo, the primitive streak forms as a ring that circles the entire colony.
- Some extraembryonic lineages are missing in micropatterned cultures: trophectoderm-like cells are present, but not visceral endoderm cells.
- The glass surface constrains the movements of cells, resulting in a loss of order as cells delaminate.
- In terms of organismal potential, the system would not develop further if implanted.
Future Directions for Synthetic Embryology
Warmflash emphasized that the goal of this work is not to build embryos from stem cells; rather, the goal is to build systems to answer specific questions about how the embryo works. The current state of knowledge and
technical capability warrants simplified systems that can be quantitatively manipulated, measured, and modeled. There is still much to be learned from 2D simplified systems, he said. To learn about later stages or other aspects of development, new systems can then be created that focus on relevant aspects without modeling the entire embryo. For example, patterns can be made at other developmental stages with more complex protocols. This has already been done with the four different states of ectoderm (Britton et al., 2019). Strides have also been made in studying morphogenesis and patterning in 3D, he added. His laboratory has developed controlled ways of taking cells into 3D to model the neural tube. He and his colleagues are also interested in interactions between embryonic and extraembryonic cells. He conceded that it is controversial to put those cell types together, but they are studying the interaction in 2D without developing the cells further. When BMP-differentiated trophectoderm-like cells and ESCs are juxtaposed, the trophectoderm cells induce the gastrulation-like events, including the primitive streak marker Brachyury, in ESCs without the need to add exogenous ligands.
BLASTOIDS: MODELING THE PRE-IMPLANTATION EMBRYO
Nicolas Rivron, a group leader at the Institute of Molecular Biotechnology, Austrian Academy of Sciences, described how his laboratory models early embryonic development in 3D by combining trophoblast stem cells and embryonic stem cells to form blastoids, which model some features of the pre-implantation embryo. He emphasized that the work he would describe is strictly based on mouse stem cells, not human stem cells.
Reciprocity Between Embryo and Placenta
During pregnancy, the placenta is the lifeline that sustains the development of the embryo and the fetus. However, Rivron and colleagues discovered that during the very early developmental stages—before implantation in the uterus—the embryo actually takes care of the future placenta by fueling the proliferation, self-renewal, and morphogenesis of the trophoblast stem cells in the placenta. The embryonic compartment feeds the trophoblast in order to set it apart from the embryo and form the trophectoderm cyst. Through a complex combination of signals, it pushes the trophoblasts to develop and prepares them for implantation inside the uterus. This is interesting from an evolutionary perspective, Rivron remarked, because “at this early stage, the embryo invests in its own future.”
Creating a Blastoid
To frame his laboratory’s work on blastoids, Rivron presented a movie depicting the very early stages of mammalian development. At day 4.5, the mouse blastocyst comprises about 100 cells and is still floating inside the uterus. At this point it already contains three cell types: the epiblast that will form the full embryo, the primitive endoderm, and the trophectoderm (see Figure 4-2). Although this blastocyst could implant inside the uterus, Rivron and colleagues put it in culture and expand the stem cells that it contains. ESCs are dependent on Wnt and LIF, and trophoblast stem cells (TSCs) are dependent on fibroblast growth factor 4 (FGF4) and TGFβ ligands, Rivron said. Those cells are frozen in time and can be reinjected back into an embryo, where they will contribute to their respective embryonic and placental compartments. In Rivron’s laboratory, those cells are recombined in a dish to promote self-organization into a structure that is remarkably similar to the blastocyst. Rivron calls this structure a “blastoid.” He underlined two elements that are experimental advantages to blastoids: (1) they can generate large numbers of blastoids because this process does not require embryos every time, and (2) they can genetically edit the stem cells before the blastoid is formed.

SOURCE: Nicolas Rivron workshop presentation, January 17, 2020.
Rivron noted that since their first publication describing the creation of mouse blastoids in 2018 (Rivron et al., 2018b), three subsequent publications have presented slightly different ways of making mouse blastoids (Kime et al., 2019; Li et al., 2019; Sozen et al., 2019). The approach used by Rivron and colleagues was to first seed ESCs on non-adherent microwells made of a hydrogel (diameter: 200 micrometers). Because the surface is non-adherent, the cells have no other choice but to adhere with each other and aggregate. Once small clusters were formed, they seeded the TSCs on top of them. When they screened for factors that are present during blastocyst formation, they found that the cells remember where they come from and start talking to each other, spontaneously organizing into the structures.
Features of Blastoids
Rivron provided an overview of the features of the blastoid. He explained that the blastoids make analogs of the three cell types that form the blastocyst—epiblast, trophoblast, and primitive endoderm—which later develop into the placenta, yolk sac, and body to form the whole organism. His view is that blastoids do not have organismic potential because development is a historical process that starts with the fertilization of an egg and ends up with the delivery 21 days later (for the mouse). Blastoids are used to model a very short window of time of only 1–3 days. However, the cells at this stage do have the NANOG-positive cells and OCT4-positive cells that will eventually form the body as well as the CDX2-positive cells that will form the placenta. The niche is also permissive for the generation of PDGFalpha positive cells and GATA6-positive primitive endoderm cells that will form the yolk sac, which is the second extraembryonic tissue.
Blastoids are morphologically similar to blastocysts, Rivron said. They also form very efficiently, with beautiful structures emerging within just 48 hours. In his first series of studies using the microwells containing the appropriate numbers of cells (i.e., 8 ESCs and 20 TSCs), he observed a rate of self-organization to blastoid structure of greater than 70 percent. Subsequently, he has attained levels greater than 90 percent. Rivron added that the yield of blastoids is highly dependent on both the absolute numbers and the ratio of ESCs to TSCs. A comparison of blastoid systems with the actual blastocyst reveals that the blastoid systems have high fidelity and can implant when transferred into the uterus of mice, where the blastoids induce the formation of the uterine cocoon, termed decidua. However, the blastoids collapse and cannot develop further after 2 days. In investigating why the blastoids lack the potential for future development, Rivron’s group found interesting reactions occurring in the decidua. For example, his group observed the formation of ALDH3A1-positive cells, which has been shown to be specific to the implantation of blastocysts, a marker that suggests a
similarity to the implantation process of blastoids. Further characterization revealed CDX2-positive cells, which form the placenta, as well as many other relevant cell types. He concluded that, in the uterus, blastoids can initiate implantation and development in a way that is similar to blastocysts, including the formation of decidua-like structures. However, the cell types in the blastoids were far from properly organized. Box 4-2 is an overview of the blastoid’s defining features as presented by Rivron.
The Cooperative Life of Embryo Cells
Rivron’s laboratory focuses on understanding the cooperation between cell types in the embryo. More specifically, he and his colleagues look at the autonomous, self-organizing, and cooperative behaviors that lead to the compartmentalization of the conceptus.4 A flow of information between the future embryo and the extraembryonic tissues facilitates synchronization and establishes developmental checkpoints of development.
Systematic Inventory of Embryonic Inductions
Rivron explained that his group is able to generate trophospheres using the same process for generating blastoids, but without the presence of the embryonic cells. Comparing the blastoids and trophospheres allows for establishing a proxy for the influence of the embryonic cells on the trophoblast cells. He presented single-cell RNA sequencing experiments and a T-distributed stochastic neighbor embedding plot of single blastoid cells, single trophosphere cells, and single TSCs in 2D, which illustrated the
___________________
4 The term conceptus was defined in a recent publication as “the products of conception at all stages of development from zygote to birth. These include the embryo proper, the fetus, the placenta, and all extraembryonic membranes” (Hyun et al., 2020, p. 170).
transcriptome shifts occurring upon crosstalk between the trophoblast and embryonic cells. This shift was used, he said, to deeply analyze the effects of the embryonic cells on the trophoblast cells—so-called embryonic inductions. By developing a list of signaling pathways, the group discovered that the embryonic cells strongly influence the development of the trophoblast cells. The embryonic cells produce a range of different factors (e.g., FGF4, Nodal, BMP4, BMP7, and some Wnts) that activate the MAP kinase and TGFβ and Wnt signaling pathways, among others, in the trophoblast.
Further functional analysis of these embryonic inductions demonstrated that embryonic cells regulate trophoblast proliferation and self-renewal, Rivron said. Previous work showed that FGF is inducing the proliferation of the trophoblast cells that are in contact with the embryonic cells and that go on to form the placenta. On the other side of the blastocyst cavity, the cells that are away from the embryonic inductions are differentiating and acquire functions that allow them to mediate implantation inside the uterus. Rivron and colleagues were able to show that the presence of the embryonic cells induces proliferation and self-renewal of the trophoblast in blastoids (Rivron et al., 2018b). By contrast, proliferation was not induced in trophospheres in which those cells were absent. Thus, the blastoids were clearly recapitulating the only known induction, he noted.
In an experiment looking at the TGFβ pathway, Rivron and colleagues compared the diameter of the cystic structures between blastoids and trophospheres (Rivron et al., 2018b). Although trophospheres are smaller than blastoids, they determined that it was possible to apply the factors produced by ESCs (BMP4 and Nodal) on top of the trophospheres. This causes the trophospheres to swell. By counting the number of cells, they determined that Nodal and BMP4 were not inducing proliferation of the cells; rather, they were just inducing the spreading of the structure. Because the researchers were able to perform this analysis on a huge number of blastoids and trophospheres, they were able to identify small effects that would be difficult to pinpoint by analyzing limited numbers of actual blastocysts. Rivron highlighted this as a strong example of an advantage of embryo models. Embryo models can also be used to generate specific knockouts only in one compartment, he added. The trophectoderm stopped swelling when Nodal was specifically knocked out in the embryonic cells, indicating that Nodal is an embryonic inducer that regulates trophectoderm swelling. Furthermore, they discovered that one of the targets of Nodal in the trophoblast, KLF6-positive cells, regulates trophectoderm swelling and likely plays a role in epithelial morphogenetics.
Rivron emphasized that embryonic inductions are important in inducing trophoblast proliferation and self-renewal by (1) maintaining trophectoderm core transcription factors, (2) preventing differentiation, and (3) regulating trophectoderm epithelial morphogenesis. Together,
these embryonic inductions also prepare and maintain the potential of trophoblasts to implant inside the uterus. He explained that there is a window of opportunity for trophoblasts to implant and that if the cells differentiate into post-implantation placental cell types, they lose the capacity to implant into the uterus. This is demonstrated in the largely diminished potential to implant observed in trophospheres, which stop proliferating and begin to differentiate in the absence of those embryonic inductions. These findings have implications for addressing the high rate of implantation failure in human pregnancies, he noted. These types of embryo models also hold potential for addressing clinical issues—for example, treating infertility, improving in vitro fertilization, designing new contraceptives, and preventing disease (Hyun et al., 2020; Rivron et al., 2018a).
MICROFLUIDIC STEM CELL MODEL OF PERI-IMPLANTATION
Jianping Fu, an associate professor of biomedical and mechanical engineering and an associate professor of cell and developmental biology at the University of Michigan, discussed stem cell models of peri-implantation human development. His laboratory uses human pluripotent stem cells (hPSCs)—both induced pluripotent stem cells and hESCs—to develop sensitive models for studying pre-implantation human development. He said that investigating many important questions about human development does not require generating complete synthetic structures that include all of the embryonic or extraembryonic images.
Overview of Peri-Implantation Human Embryonic Development
Fu began with an overview of peri-implantation human development. He highlighted the initial notable embryonic events that occur after the blastocyst implants into the maternal uterus. First is the spontaneous formation of the pro-amniotic cavity. The epiblast next to the invading trophoblast then spontaneously differentiates into the amniotic ectoderm, while the remaining epiblast next to the hypoblast may remain pluripotent. Soon after these symmetry-breaking events, the anterior–posterior axis is established, and the posterior end of the epiblast transitions to establish the three different germ layers. After implantation but before gastrulation, another cell type appears—the primordial germ cell, which is a precursor for all of the sex cells that are responsible for human reproduction as well as human evolution.
Self-Organized Development of Stem Cell Models of Peri-Implantation
When the researchers in Fu’s laboratory initiated this work, they were not planning to develop systems to model peri-implantation in human development. However, as they developed a 3D culture environment for the control experiment (in which they did not add any exogenous signals to the culture), they observed that hPSCs began to self-organize. The cells developed multicellular structures and underwent morphogenetic events similar to those expected during peri-implantation of human development. First, the hPSCs undergo lumenogenesis to form a cavity. Soon after, the cells begin to change shape, becoming more squamous and flattened, with protrusions beginning to extend from the basal surfaces at the same time (Shao et al., 2017a,b). The researchers determined that the cells were differentiated from hPSCs, and they ultimately characterized these cells as amniotic ectoderm–like cells. The 3D culture environment drove most of the cells to differentiate into uniformly squamous amniotic ectoderm-like cysts, but in about 5–10 percent of the cysts, they observed asymmetry as well as spontaneous symmetry-breaking events. Fu presented an example of this asymmetry. After forming a cavity, some of the cells at one pole of the cyst started to differentiate into squamous amniotic ectoderm–like cells, while the rest of cells remained undifferentiated columnar epiblast-like cells.
Co-staining the cells for pluripotency markers (e.g., NANOG, OCT4, SOX2) suggested that the undifferentiated cells would remain as undifferentiated pluripotent cells, Fu said. When the differentiated cells were stained, only the squamous differentiated cells stained positive for what are thought to be amniotic ectoderm markers. Yet another subpopulation of cells appeared to be mimicking gastrulation-like events that were occurring after lumenogenesis, cavity formation, symmetry breaking, and cell differentiation. These cells, which were disseminating from the undifferentiated epiblast compartment, stained positive for canonical markers associated with gastrulation and began to lose expression for NANOG.
Autonomous BMP Signaling Drives Amniotic Differentiation
Fu and colleagues investigated the molecular mechanisms for driving amniotic cell differentiation from hPSCs in their 3D culture environment. Their model did not have any exogenous external signals driving the stem cells to differentiate, so they looked at BMP signaling pathways. Staining the cells for phosphorylated SMAD, the downstream effector for BMP signaling, suggested that when the cells start to differentiate, they autonomously activate BMP signaling. Blocking BMP signaling enabled the
researchers to rescue the pluripotency of the cells in the cadre. Fu said that this observation about autonomous expression or autonomous activation of BMP signaling in amniotic ectoderm–like cells is consistent with available data generated from post-implantation monkey embryos. BMP4 is almost exclusively expressed in the amniotic ectoderm compartment of those monkey embryos, and MSX2—a downstream BMP target gene—is also clearly expressed (Sasaki et al., 2016).
Controlled Microfluidic Modeling of Human Embryo Development
Conventional 3D culture environments can be used to generate self-organizing multicellular structures that bear similarities to certain facets of human development, Fu said. However, these systems offer limited controllability and reproducibility. He illustrated this using data from some of his group’s experiments. Although there are clear examples of asymmetric tissue in some cysts, different degrees of progressive development and differentiation are observed in the cysts overall. It is this heterogeneity that limits the conventional 3D culture’s controllability and reproducibility and, by extension, limits the utility of these systems for mechanistic investigations.
Over the past several years Fu and colleagues have leveraged their expertise in microengineering and microfluidics to enhance controllability through controlled microfluidic modeling of human embryo development (Zheng et al., 2019). This system features a simple microfluidic structure, he explained. The microfluidic device contains an induction channel and a cell-loading channel; gels are loaded into the central channel between the other two. During gelation, the gel contracts due to surface tension, causing the gel to autonomously generate gel pockets that form between the supporting posts. hPSCs are loaded into the cell-loading channel and the device is tilted 90 degrees for 10 minutes. Many of the cells spontaneously settle into each of the gel pockets; later, the floating cells can gently be removed. In this way microfluidics makes it possible to create assays that offer superior controllability, reproducibility, and scalability.
Fu described the first set of experiments he and his colleagues performed with the microfluidic modeling system. No exogenous inductive signals were added to jog the stem cells to differentiate, so all the cells would remain pluripotent, he noted. As soon as the hPSCs started touching each other, they began to coalesce into a community and spontaneously underwent lumenogenesis, forming cavities of epiblast-like cysts in a synchronized way.5 He said that this type of microfluidic system is scalable and compatible with live imaging, enabling investigation of the cells’ signaling
___________________
5 Fu noted that this is an established lumenogenic property of hPSCs.
dynamics, signaling propagation, and the inductive signals that drive the formation of different compartments.
Asymmetric BMP Stimulation Induces Embryonic-Like Sac Formation
In the second set of experiments, Fu and colleagues decided to add BMP4 into one of the top channels (Zheng et al., 2019). They had established in their previous studies that BMP4 signaling is sufficient to drive hPSCs to differentiate into amniotic ectoderm–like cells. In the bottom cell-loading channel, there were no exogenous inductive signals. When the cells underwent lumenogenesis, the cells directly exposed to BMP4 stimulation spontaneously differentiated into amniotic ectoderm–like cells. However, because only a portion of the cells in each cell colony were directly exposed to BMP4, they form asymmetrical bipolar embryonic-like structure. Once the top portion of cells differentiated into amniotic ectoderm–like cells, these amniotic cells began secreting inductive signals to drive the continuous development of the bottom portion of undifferentiated cells to spontaneously undergo gastrulation-like events. At certain time points, molecular asymmetry was very evident in the stem cells: only the cells that were directly exposed to BMP4 stimulation expressed TFAP2A, the gastrulating cells expressed T/Brachyury and were continuously differentiating, and all of the cells expressed CDX2. (CDX2 is an extraembryonic marker, but it also expresses in the posterior primitive streak.) Fu showed a video demonstrating this patterning. The cells formed a cavity and the top cells exposed to BMP4 became increasingly squamous and flattened. Soon after, the cells started to disseminate from the opposite pole. Fu said that he and his colleagues cease all experiments at day 3 or 4 because after that point the entire structure will disassemble and collapse, with gastrulating cells disseminating in an uncontrollable fashion.
Emergence of Primordial Germ Cell–Like Cells in Embryonic-Like Sac
Fu’s laboratory has also used the microfluidic model system to look at primordial germ cells, the important cell lineage developed in embryonic tissue that appears post-implantation but before gastrulation (Kobayashi et al., 2017; Sasaki et al., 2016). In that case cells were stained with canonical markers SOX17, NANOG, and TFAP2C, and the triple-positive cells were identified as primordial germ cell–like cells, which began to appear at early time points in a scattered fashion. Soon after, these cells began to accumulate toward the junction between the amniotic ectoderm compartment and the gastrulating epiblast-like cell compartment. The researchers also observed some interesting cell dynamics, suggesting that this type of system
would be informative for examining the origin and lineage development of primordial germ cells in early human embryonic development.
Comparative Transcriptome Analysis with Primate Monkey Embryo Data
As an in vitro system, the system developed by Fu’s laboratory is compatible with the use of downstream genomic analysis and single-cell transcriptome profiling to identify cell types and lineage markers. The researchers have also used comparative transcriptome analysis to compare the cell types generated by their synthetic system with cell types developed from primate monkey embryos (Nakamura et al., 2016). According to Fu, the gastrulating cells generated by their system are similar to the gastrulating cells from the monkey embryos. The researchers also compared the primordial germ cell–like cells generated by the system with those generated in monkey and human embryos and other in vitro culture models (Kobayashi et al., 2017; Kojima et al., 2017; Sasaki et al., 2016). The primordial–germ cell–like cells generated by the microfluidic system compared well with those generated in other in vitro culture models, Fu said.
PANEL DISCUSSION
Toward More Unified Terminology
Given the range of the terminology being used in these models (e.g., blastoids, gastruloids, neuruloids, synthetic embryos, embryoids), Clark suggested trying to unify the terms that scientists use to discuss this work—and to do it in a way that does not cause alarm among the general public. Warmflash described “synthetic embryo” as an inappropriate term, for instance; embryo should not be part of the name because these models do not have organismal potential. He suggested promoting uniform use of the “-oid” suffix to indicate that something is an in vitro model that aims to capture features of a stage of development (e.g., a blastoid is an in vitro model intended to capture something about the blastocyst stage, but it is not a real blastocyst). Rivron agreed that names should be based on what the system is presumed to be modeling, with the “-oid” suffix used to clarify that it is only a model. However, a challenge is that at the outset, the window of development to which the cells are equivalent is unknown. Fu suggested using “stem cell–based models of human development” if a single umbrella term including all of the models is needed.
Molecular-Controlling Influences of Size
A workshop participant asked Brivanlou about the influence of the diameter change on the RNA-sequencing profiles. A major consideration in biology in general and in embryology in particular is the molecular-controlling influences of size: everything has a given thickness and diameter that sometimes changes over time, Brivanlou said. When the size of the cell colonies is shrunk, the center fate is always lost (i.e., ectoderm is lost when the size shifts from 1,000 to 500 microns; mesoderm is lost and only trophectoderm remains when the size shifts from 500 to 125 microns). Further work in Brivanlou’s laboratory has shown that the cells have a “magic ruler” that measures the distance from the edge, indicating that the patterns are controlled from the edge, not from the center. The researchers in Brivanlou’s lab have defined two features of that ruler: (1) physical edge sensing that is dictated and imposed by the BMP receptor and (2) a reaction diffusion whereby BMP4 induces the direct expression of its own inhibitor, NOGIN. In no other species, including nonhuman primates, is BMP4 able to directly induce the expression of its own inhibitor. This creates a Turing pattern, in which NOGIN diffuses much faster from the edge toward the center and completely blocks the effect of BMP4 coming from trophectoderm.
Using Blastoids to Model Developmental Potential
Rivron was asked if he had tried adding primitive endoderm lineage to the blastoid model in order to look at the effect on implantation developmental potential. His team did so in the original protocol, he answered, and primitive endoderm did form, but the numbers were not sufficient. His team’s approach is to push ESCs to form primitive endoderm cells by exposing cells to certain factors using specific “cocktails” that can efficiently form primitive endoderm cells. Next, they developed an assay to functionally test whether more primitive endoderm cells would help to support further development. They observed a higher propensity for the egg cylinder to develop after the induction of primitive endoderm cells, suggesting that the epiblast may develop in synergy with the primitive endoderm. Primitive endoderm may be one of many elements required for blastoids to take the next developmental step, Rivron surmised. He noted that this type of work demonstrates certain advantages of using stem cell–based embryo models over the classical approach to embryology, which involves knocking out genes in actual embryos and looking for a phenotype. This approach is limited to genes that provide a good phenotype and can actually be studied using the genetic knockout; those genes have now been largely exhausted. Embryo models offer a new approach that works in the opposite direction:
starting with a minimal structure (i.e., rudimentary blastoid) and building it up with increasing complexity to see if it progresses through the developmental steps. This bottom-up approach to embryology allows for identifying minimal features that are necessary for development but could not be seen before.
Stable Endpoints Versus Continuous Differentiation in Model Systems
A workshop participant asked whether stem cell–based systems end at defined states or have continuous differentiation and developmental processes, which has implications for the propagation and development of different cell types. Warmflash said that the systems he uses will continue to develop, but their fidelity diminishes over time because the systems have been engineered to model a specific stage. The cell types will mature because they are programmed to keep developing, but they will lose organization due to the limitations of 2D culture systems. In his work on frog development, Brivanlou observed a linear progression over time in transitions of cell fates. The germ layers were produced, and then posterior patterning kicked in within the germ layers; morphogenesis added another layer of complexity. Cell fates continue to change as a function of time—frogs can develop from a single cell to 40 million cells in only 48 hours. In addition to this transition of cell states, other issues in embryology include the ability of a cell to acquire a fate or to undergo a transition in time and space. In contrast to the natural environment, in which a developing system progresses without extrinsic information, experimental conditions involve adding extrinsic information (e.g., BMP4, Wnt, and/or Nodal) and then measuring what the cells can do in that specific milieu within a very small window of transition time.
Human Versus Nonhuman Model Systems
Speakers were asked whether the models that had been discussed might eventually obviate the need to study human embryonic material directly. A workshop participant went on to ask which elements might be essential to continue studying using human material versus those that might be replaced with models in nonhuman primates or other species. Brivanlou replied that limits on the length of modeling notwithstanding, there will never be enough biological material—from humans, nonhuman primates, or other species—available to perform quantitative measurements with the level of resolution offered by synthetic model systems.
Validating Human-Specific Features of Synthetic Models
Kriegstein asked the panelists to consider how it will be possible to validate the human-specific features of these models without the use of animal models and given the limited access to human tissue. RNA-sequencing approaches can be used, Brivanlou answered. For example, his laboratory has used this approach to provide direct evidence that a splice isoform of the Huntington gene only exists in hominids. More difficult to test are, for example, species-specific signaling interactions and reaction diffusions. However, Brivanlou predicted that some of the models described by the panelists would ultimately allow for that type of testing. CRISPR/Cas9 can already be used to perform tracing at resolutions never achieved before, he added. He expressed hope that quantitative tools would be developed to demonstrate the speed of NOGIN with BMP4 in real time in a human model.
Huntington’s Disease Phenotype in the Gastrula Stage
Kriegstein remarked that Huntington’s disease tends to manifest several decades after birth, so the gastrula-stage phenotype identified by Brivanlou seems to appear earlier than expected. Brivanlou replied that this was also a surprise to him. Huntington’s disease is caused by an insertion of CAG-repeats in a gene called Huntingtin (HTT). Different individuals have CAG-repeats of different lengths in their HTT gene, and the onset of the disease is CAG-length-dependent, with longer insertions corresponding to an earlier manifestation of the disease. The earliest recorded death from Huntington’s disease is 18 months of age, Brivanlou said. Comparative studies in a variety of species suggest that a maternal rescue component kicks in and mediates many of the early effects of the mutation, which may account for why the disease tends to manifest later in life. The loss of both Huntingtin alleles (HTT–/–) leads to embryonic lethality, demonstrating that the gene is necessary for gastrulation and that it has independent functions beyond the nervous system. The gene is expressed in the fertilized egg and then expressed ubiquitously in all adult cell types. If the mutation were a dominant mutation, it follows that the earliest effect of the mutant would occur at the earliest time point of biological activity, Brivanlou said. Furthermore, a mouse model study found that if a pulse of Huntington mutant is given to an early embryo and then the activity is retracted, the adult will manifest Huntington’s disease symptoms, he noted.
Markers of Peri-Implantation Development
Fu was asked how he determines that he is not actually studying trophoblast differentiation instead of peri-implantation development. A key diagnostic marker to identify developmental stage is the appearance of primordial germ cell–like stem cells in the amniotic ectoderm compartment, Fu said, because primordial germ cells only appear post-implantation and before gastrulation. This suggests that the amniotic ectoderm compartment corresponds to the amniotic compartment in the peri-implantation human embryo. However, the underlying question of whether his lab has established the equivalency of the cell types generated in their in vitro models cannot be answered until data become available that allow them to perform comparisons with in vivo human or primate embryos.
Precursor of Germ Cells
Martin Pera asked Fu if lineage tracing had been used to look for the precursor of the germ cells. Fu replied that he has a grant pending with the aim of answering that specific question. He added that these synthetic models provide powerful experimental platforms for advancing fundamental understanding about human development, particularly for discovering essential features of developmental mechanisms at a detailed molecular level.


























