5
Influenza Vaccine Access and Financing
Despite new governance facilities aimed at promoting equitable access to COVID-19 vaccines—such as the COVID-19 Vaccines Global Access (COVAX) pillar of the Access to COVID-19 Tools (ACT) Accelerator—high-income countries (HICs) have cornered the market to the extent that major disparities in global access have emerged. In January 2021, the World Health Organization (WHO) Director-General Tedros Adhanom Ghebreyesus warned that in terms of ensuring access to COVID-19 vaccines, the world was “on the brink of a catastrophic moral failure—and the price of this failure will be paid with lives and livelihoods in the world’s poorest countries” (WHO, 2021a). This mirrors the inequities that undercut access to vaccines during the 2009 H1N1 influenza pandemic (see Chapter 2). He echoed this sentiment in his opening remarks at the World Health Assembly (WHA) in May 2021, describing the ongoing vaccine crisis as a “scandalous inequity that is perpetuating the pandemic” (WHO, 2021b). Without improved coordination, incentivization, and investments to promote access in low-income countries (LICs), a similar pattern will likely undermine the rollout of vaccines during a future influenza pandemic.
THE COVAX FACILITY AND ITS SHORTFALLS
In April 2020, COVAX was formed jointly by WHO, the Coalition for Epidemic Preparedness Innovations (CEPI), and Gavi to invest in several vaccine candidates and ensure that the vaccines would be equitably distributed among participant countries largely by funding distribution to the 92 lowest-income countries. Box 5-1 provides an overview of its aims
and financing structures. Almost every country signed up for COVAX, and it delivered its first doses in late February of 2021. The following month, due to the rapidly escalating number of cases in India, the Serum Institute of India (SII)—which COVAX had presumed would be its main vaccine supplier—stopped exporting its vaccine, which was made in collaboration with AstraZeneca and the University of Oxford (Cohen and Kupferschmidt, 2021). This move was predicted to create a 190-million dose shortage by the end of June 2021. COVAX had delivered just 4 percent of all vaccine doses administered worldwide as of May 2021, although its latest supply forecast (September 2021, WHO, 2021c) suggests that approximately 1.2 billion doses will be available for lower-income countries participating in its advance market commitment (AMC), enough to protect 40 percent of all adults in the 92 AMC countries, with the exception of India.
The COVAX experience highlights the difficulty facing global health coordination, particularly around broadening global access to vaccines in an era of geopolitical transformation and increasing nationalism. COVAX has certainly improved vaccine equity compared to a scenario without a multilateral procurement or allocation mechanism. However, progress toward equitable global vaccine allocation remains disappointing 18 months into the pandemic. The facility is unlikely to deliver doses representing 20 percent of each country’s population by the end of 2021, as originally committed.
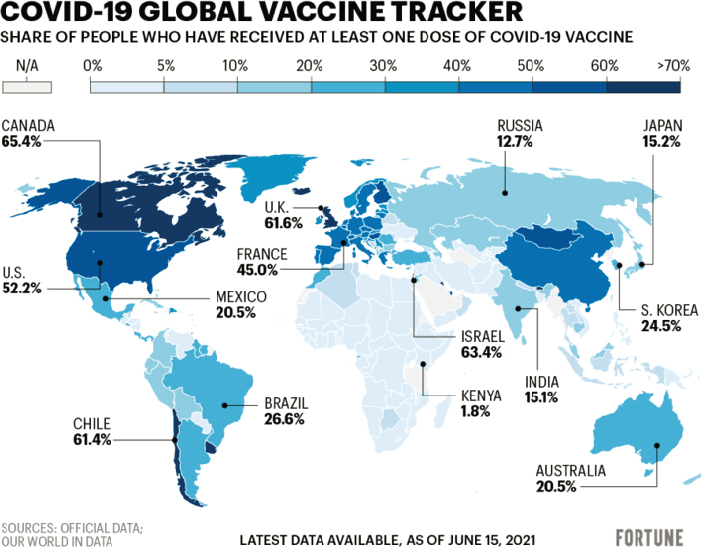
Ultimately, HICs mostly bypassed COVAX and pursued bilateral advance purchase; for instance, the United States and the European Union bought 3-fold more vaccines than they would need for their entire populations (Cohen and Kupferschmidt, 2021). Moreover, population coverage targets differed between self- and aid-financed countries. Figure 5-1 depicts the global share of individuals who received at least one dose as of mid-June 2021, starkly illustrating the extent to which the worldwide rollout has been inequitable for less wealthy nations.
Many of the challenges that fueled this disappointing COVAX performance operate out of its direct control. These include multiple forms of vaccine nationalism and associated lack of donor support and industry behavior. For instance, the time lost in coaxing HICs to join in 2020 contributed to delays in fundraising and resource mobilization, and heavy reliance on a few technologies and manufacturers (mostly based in the United States and the European Union) limited the speed and capacity of vaccine scale-up. It could be argued that the major “failure” of COVAX is naivety in anticipating these challenges. However, a more “anticipatory” design may not have made much difference in terms of access during a time of vaccine shortage and could have even had the unintended consequence of legitimizing the political economy of vaccine nationalism.
SOURCE: Our World in Data. https://bit.ly/3vdSoOv (accessed December 19, 2021).
The facility’s self-finance window was perhaps a more avoidable design flaw. Inadequately self-financed HICs joined COVAX, which undermined its buying power. Most donor funds supplied through the AMC, which was used to finance the facility, were not pledged until late in the first half of 2021. Limited buying power had serious ramifications for inter- and intra-country equity in vaccine allocation and further limited the ability to support supply chains and manufacturing required for effective vaccine scale-up.
In response to this vaccine shortage and the vastly inequitable distribution of the limited supplies available, the “Financing Pandemic Preparedness and Response” paper commissioned by the Independent Panel for Pandemic Preparedness and Response (IPPPR), called on wealthy countries to donate 1 billion doses to COVAX by September 2021 and another 1 billion by the middle of 2022 (Radin and Eleftheriades, 2021). At the WHA, Ghebreyesus also called on all manufacturers to either offer any new product to COVAX before it enters the market or commit 50 percent of the doses to COVAX (Cohen and Kupferschmidt, 2021).
APPROACHING EQUITY IN VACCINE ACCESS FOR INFLUENZA
The COVID-19 pandemic has provided an unprecedented opportunity to move the needle on how equity is addressed in pandemic preparedness and response (PPR). Moral appeals can help to deal with equity issues during times of acute scarcities, but the reality is that policy makers need to be incentivized to promote equity. It is important to consider three questions when approaching issues of equity in access to influenza vaccines: (1) which strategies can be employed to improve equitable access, (2) what is the extent to which those strategies need to be specific to influenza, and (3) how much focus should be placed on equitable access to vaccines versus other medical products that are critical during a pandemic.
The question of how to improve equitable access spawns a set of further considerations related to whether equity should be dealt with directly or indirectly. It can be addressed directly either by considering equity and advancing discrete solutions—for example, via mechanisms such as WHO’s Pandemic Influenza Preparedness (PIP) Framework—or through the physical location of production facilities (e.g., distributed manufacturing). Equity can be dealt with indirectly by increasing supplies to surge and building capacity to the degree that rationing becomes less biting. Michael Kremer of the University of Chicago has described the benefits equity principle, which holds that increasing supply naturally reduces the wait for everyone. Game theory shows that during a shortage, a gap emerges between social value and price; governments have powerful incentives to prioritize their domestic production, implement vaccine export bans, and hoard vaccines (Kremer, 2021). As a result, game theory predicts a lopsided vaccine allocation that prevents a socially (Pareto) optimal solution where global morbidity and mortality are reduced as much as possible. In practice, relying on principles of equity is not an effective tactic when scarcity is acute. Even when enshrined in law, equity access mechanisms are relatively ineffective during shortages. One strategy that can be useful during periods of acute scarcity is to ensure that capacity is in place with immediate inputs.
To the question of how specific to influenza the solutions to promote equitable access should be, Chapter 4 showed how the market for seasonal vaccination is limited to the extent that the basis may be insufficient to keep manufacturing warm for a pandemic. This is especially the case with the newfound potential transferability of platform technologies. If the focus is skewed toward the seasonal manufacturing base, the financing scale may be restricted to merely the tens of millions—not the hundreds of billions needed for a strong pandemic vaccine response.
How much focus should be placed on equitable access to vaccines during a pandemic versus other critical medical products is complicated
by the mandate of the committee’s Statement of Task (which is focused on vaccines and vaccination). However, vaccine financing solutions should not be siloed. When a new pandemic strikes, it is not possible to know a priori the optimal combination of containment measures, such as vaccines, diagnostics, and therapeutics. Ideally, coordination and financing mechanisms would have built-in flexibility to develop, manufacture, and deliver vaccines as well as other therapeutic tools.
LESSONS FROM THE COVID-19 VACCINE EXPERIENCE
At least three valuable lessons can be gleaned from COVID-19. First, paying up front to develop a portfolio of vaccine candidates and maintain sufficient surge manufacturing capacity can yield a huge return on investment. Second, having vaccine governance structures before a pandemic is essential. Third, such structures should specifically target how to achieve equitable access for low-income settings.
COVID-19 has demonstrated that it is critical to have vaccine governance structures and financing before a pandemic strikes, because it can alleviate the scarcity and perverse nationalism that underpin and exacerbate global inequities. Up-front financing is needed not only for manufacturing capacity but also to facilitate pre-procurement of vaccines and therapeutics prior to the lengthy process of obtaining full regulatory approval. The pandemic illustrated how paying for overcapacity to deliver vaccines during a pandemic is an inexpensive and cost-effective strategy. If there were a single decision-making entity for the world, it would likely maintain that spending several billion dollars per year on pandemic preparedness is highly rational. For example, Operation Warp Speed (OWS) paid for itself many times over: with a cost of $13 billion, it would have paid for itself in just 12 hours by virtue of accelerated vaccinations. Furthermore, the global benefits of the first 3 billion doses likely generated worldwide social benefits of $17.4 trillion, or $5,800 in benefits per course (Kremer, 2021).
Vaccine governance structures should specifically target low-income settings whose varied funding needs are often overlooked during a response, as seen during the COVID-19 pandemic. To work toward developing systems that promote access to vaccines against future pathogens with pandemic potential, the needs of low-income settings should be considered specifically, rather than looking at funding considerations from an exclusively global perspective. In this sense, reframing acceptable wait times for pandemic vaccines could be heralded as a major achievement of the COVID-19 pandemic response. The global dialogue now centers on whether the poorest countries can receive vaccines 2–3 months rather than 2–3 years after wealthier countries. This represents a seismic shift in what is regarded as acceptable access.
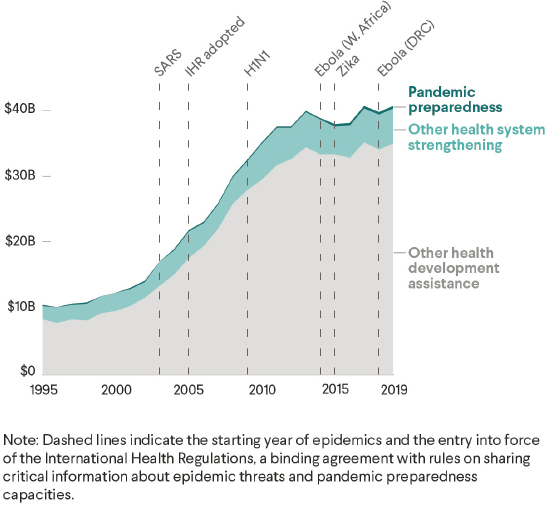
HOW HAS PANDEMIC FINANCING CHANGED DURING COVID-19?: FUNDING FOR COVID-19 PREPAREDNESS AND RESPONSE
In 2019, $374 million—representing less than 1 percent of overall official development assistance for health (DAH)—was spent on strengthening pandemic preparedness in low- and middle-income countries (LMICs) (Figure 5-2). Another $5.2 billion was spent on strengthening health systems, some of which could improve countries’ ability to deal with epidemics and pandemics. However, even this volume of spending has been shrinking. Since 2003, global aid for health system strengthening has fallen from 22 to 14 percent of overall annual DAH (Burwell et al., 2020). Major mechanisms used to finance PPR at WHO, the World Bank, and other multilaterals include emergency/contingency funds, pandemic insurance bonds, pooled/block funding, and liability insurance (NASEM, 2016). Table 5-1 provides an overview of each category of financing modalities. However, these modalities are largely aimed at “response” rather than “preparedness.” Preparedness spending is mainly a by-product of other health system spending. For example, Global Fund grants make significant contributions
SOURCE: https://www.cfr.org/report/pandemic-preparedness-lessons-COVID-19/pdf/TFR_Pandemic_Preparedness.pdf (accessed December 19, 2021).
TABLE 5-1 Types of Financing Modalities Used for Pandemics
| Type of Financing Modality | Examples | Year of Inception | Overview | Level of Funding |
|---|---|---|---|---|
| Emergency/contingency funds | WHO Contingency Fund for Emergencies (CFE) | 2016 | A rapidly available (24 hours or less), post-Ebola source of funding established by the UN General Assembly to respond to disease outbreaks and health emergencies (including natural disasters and armed conflict) but not earmarked for other health activities. | US$77 million (2018–2019) |
| The UN Central Emergency Response Fund (CERF) | 2006 | US$35.8 million (2017) for the Democratic Republic of the Congo (DRC) during its Ebola outbreak | ||
| Pandemic insurance bonds for complex, contested risks | The World Bank Pandemic Emergency Financing (PEF) Facility | 2017 | A “catastrophe-bond” issuing system that offered additional opportunity for financial markets to diversify from traditional forms of investment (with a prospect of a return) to help the world’s poorest countries respond to cross-border, large-scale outbreaks. This hit major hurdles, particularly its tight requirement for specific trigger criteria to be met, which is limited to seven viruses (including influenza) and requires cases to be confirmed in more than one country. | US$61.4 million for DRC to fight Ebola (including $50 million for the 10th outbreak) |
| The African Risk Capacity (ARC) Outbreak and Epidemic Insurance Programme | Intended first issue of Cat bond or reinsurance cover in 2021 | With funding from The Rockefeller Foundation and Swiss Agency for Development and Cooperation and strategic and technical support from the Africa Centres for Disease Control and Prevention and the WHO Regional Office for Africa, the ARC developed a sovereign parametric insurance product to address the financing needs of member countries to contain public health emergencies arising out of common infectious diseases in Africa. | Unknown |
| Pooled/block funding | To be further developed: An objective outbreak-focused integrated framework for disbursements | — | An integrated system that directs funds to responders from a central, impartial, coordinating body with exclusive remit to protect/improve global public health—best to be WHO. Actors would need to prove that they are worthy of investment. Donor preferences to fund particular activities should be incorporated into a WHO-led financial redistribution system targeting each outbreak when it occurs to appeal to the donors. Accompanied by a transparent system of accountability, effective use of technology, and strong leadership. | — |
| Multilateral vaccine tiered-pricing system, the Global Alliance for Vaccines and Immunization (Gavi) | 2000 | A dynamic resource mobilization model to expand vaccines of hepatitis B (Hep B) monovalent, tetravalent (DTP3-Hep B), and pentavalent (DTP3-Hep B-Haemophilus influenzae type b); pneumococcal; and rotavirus. The model reaches out to current and new public and private partners with a diversified portfolio of instruments (direct contributions, innovative finance mechanisms and platforms), increases ownership of implementing countries through co-financing, influences market shaping through new market entrants and price reductions in vaccine manufacturing, and mobilizes advocacy of civil society networks while involving a wide range of partners. | US$4.2 billion (2000–2010) $7.4 billion (2011–2015) $9.3 billion (2016–2020) $16.0 billion (2021–2025) $1.5 billion (2026–2037) |
| Type of Financing Modality | Examples | Year of Inception | Overview | Level of Funding |
|---|---|---|---|---|
| Partner Contributions, Pandemic Influenza Preparedness (PIP) Framework | 2011 | Annual contributions from influenza vaccine, diagnostic, and pharmaceutical manufacturers that use the WHO Global Influenza Surveillance and Response System (GISRS) are pooled under a formula agreed upon by industry representatives. Partner contributions, which can fluctuate annually, are used to fund inter-pandemic preparedness efforts at the country and regional levels (70 percent) and response activities during a pandemic (30 percent). | US$227.7 billion (2012–June 2021), or approximately $28 million annually | |
| Liability insurance | COVAX Facility no-fault compensation program for COVID-19 vaccines | 2021 | WHO and Chubb Limited (NYSE: CB), through ESIS Inc., a Chubb company, signed an agreement on behalf of the COVAX Facility on February 17, 2021, for the administration of a no-fault compensation program for the 92 LMICs and economies eligible for support via the Gavi COVAX Advance Market Commitment of the COVAX Facility. This is the first and only vaccine injury compensation mechanism operating on an international scale. This offers eligible individuals in eligible countries and economies a process to receive compensation for rare but serious adverse events associated with COVAX-distributed vaccines until June 30, 2022. It aims to significantly reduce the need for recourse to courts, a potentially lengthy and costly process. | US$8.3 billion (AMC for 92 LMICs) |
to health security (Boyce et al., 2021), and the fund is the largest multilateral provider of grants for health system strengthening.
New and evolving financing arrangements during the COVID-19 pandemic illustrate what funding might be available, without significant changes in governance and financing instruments, for an influenza pandemic. COVID-19 funding can be loosely broken apart into investment to support preparedness or response, although those categories overlap somewhat.
COVID-19: Funding for Preparedness
One month after WHO formally declared COVID-19 a Public Health Emergency of International Concern (PHEIC), its Contingency Fund for Emergencies and the United Nations (UN) Central Emergency Response Fund allocated just $23.9 million; 3 months later, the UN Global Humanitarian Response Plan remained only 5 percent financed. The World Bank’s Pandemic Emergency Financing Facility’s Cash Window did not trigger until 3 months after the PHEIC was declared and was inadequate for the COVID-19 response. The full $196 million insurance payout was released in late April 2020, but it was shared among 64 countries; 59 already had management programs. Other international facilities later came into play, such as the International Monetary Fund’s (IMF’s) Rapid Financing Instrument and Rapid Credit Facility, which provide emergency lending to eligible countries, and, according to a paper commissioned by the IPPPR, largely fulfilled their stated objectives to contain an outbreak with pandemic potential. However, the major barrier was the lack of facilities that were ready to fill the financing gap during the earliest days and weeks of a pandemic (Radin and Eleftheriades, 2021).
COVID-19: Funding for Response
Adequate response to a pandemic requires mobilizing billions of dollars to execute well-prepared response plans at short notice. Within 6 months of the COVID-19 PHEIC declaration, more than $70 billion were committed to LMICs through multilateral agencies, with an additional $50 billion disbursed from multilateral agencies to partners. But because greater than 90 percent of these funds were in the form of debt, wealthier middle-income countries—which have greater borrowing capacities—tended to receive more funds from multilateral agencies than poorer countries (Radin and Eleftheriades, 2021). Moreover, most of this funding was directed at mitigating the broader socioeconomic consequences of the health crisis rather than at addressing the crisis itself.
In terms of vaccine development and procurement specifically, an estimated $13 billion has been invested by governments and phil-
anthropic institutions to develop, produce, and purchase COVID-19 vaccines other than China’s. The Biomedical Advanced Research and Development Authority (BARDA) grants and advance market commitments alone exceeded $10 billion (Chagar et al., 2021; Sabin-Aspen Vaccine Science and Policy Group, 2021). As of May 2021, total funding for COVID-19 vaccines and treatments had reached $119.1 billion across 277 initiatives—51.6 percent of which is country specific, and 48.4 percent multi-region.
OWS represents the largest global effort for COVID-19 vaccines. As of June 2021, OWS had invested an estimated $18 billion, much of which was channeled into late-stage clinical development and early manufacturing. Agreements were put in place to buy 455 million doses to distribute in the United States. During the same period, CEPI invested just $1.4 billion to support COVID-19 vaccines. In contrast to recipients of OWS funding, who are committed only to the United States, CEPI’s funding is associated with recipients’ commitments to promote global access and ensure affordable costs. Manufacturers are directly approaching countries and organizations, creating a “complicated ecosystem for COVID-19 vaccines that is comprised of a patchwork of countries that have and do not have vaccines” (Kim et al., 2021, p. e1017). In the United Kingdom and the European Union, the Oxford-AstraZeneca vaccine development was funded almost exclusively by governments or foundations. Similarly, the German government contributed $445 million to Pfizer-BioNTech’s vaccine (Ramachandran et al., 2021).
However, as discussed at the beginning of this chapter, this financing has been inadequate to ensure timely global access to vaccines. According to the Rockefeller Action Plan for Financing Global Vaccination and Sustainable Growth (published in April 2021), ACT-A and COVAX will deliver 2 billion doses by 2021, but this rate (20 percent) is insufficient, and these initiatives face a shortfall of $22.1 billion. Even with a $12 billion infusion from the World Bank, The Rockefeller Foundation has argued that “the world needs to aim higher” (The Rockefeller Foundation, 2021). In spite of investments in developing COVID-19 vaccines and broadening access to them, the demand for multilateral development bank financing during the pandemic has generally been slow (Glassman, 2021). The Rockefeller Foundation has called for the use of instruments such as IMF’s special drawing rights to fill financing gaps in terms of long-term pandemic response. Similarly, the Center for Global Development has proposed creating a multilateral vaccine purchase window within IMF’s rapid financing instrument/concessional equivalent (rapid credit facility) (Hicklin and Brown, 2021).
New regional partnerships have recently been forged with the aim of filling the void left by COVAX’s budgetary shortfalls. For instance, the
COVID-19 African Vaccine Acquisition Task Team, the Africa Medical Supplies Platform, and the Africa Centres for Disease Control and Prevention (CDC) began a COVID-19 vaccines preorder program in 2021 for all African Union (AU) member states. Afreximbank will facilitate payments by providing advance procurement commitment guarantees of up to US$2 billion to the manufacturers on behalf of the AU member states.
FINANCING PPR: ECONOMIC PRINCIPLES
Our recommendations for expanding and coordinating investment in PPR do not require arcane economic reasoning but are based on broad lessons from the COVID-19 pandemic and basic economic principles.
The key economic lessons are that pandemics can still happen in modern times despite medical and communications advances, they can explode rather than sputter out, and exploding can lead to almost incalculable social costs. Added to the enormous toll due to death and illness are the output losses as economies are shut down to mitigate the health losses. The U.S. Congressional Research Service (CRS, 2021) estimated that COVID-19 reduced global economic growth in 2020 to an annualized rate of -3.4 to -7.6 percent, with global trade falling by 5.3 percent. Major advanced economies may operate below their potential output level through at least 2024, with the local economic fallout leading to the worst unemployment levels since the Great Depression.1
Basic economics propounds the principle of return on investment. According to this principle, projects are not worth undertaking if they do not generate sufficient benefits in excess of expenditures. On the other hand, investments that generate large benefits at moderate expense are particularly attractive. The principle holds true for consumers deciding whether to purchase a new appliance and firms deciding whether to enter a new line of business and also for societies—individual countries or international groups—deciding whether to undertake a new program. Prior to COVID-19, HICs may have considered the small benefit of quelling an exotic epidemic disease in the developing world only worth a fraction of a small aid budget. COVID-19 now shows that the losses are more in line with those that defense and health programs are designed to avoid, justifying much higher spending levels. The principles do not imply that countries should spend the whole of their defense or health budgets on PPR—this would only be justified if pandemics were certain to happen every year. However, the possibility that pandemics may arise every one or two decades may justify spending some fraction of these budgets.
___________________
1 This section is meant to be grounded in explaining the econometrics point of view, which the committee is not necessarily endorsing.
Return on investment is a rational way to increase the urgency of investment. Yet, governments, industry, and international finance institutions do not operate on purely rational terms, particularly in the realms of global public goods and preparedness—both of which are difficult to measure and quantify. Norms can be a powerful force to support international coordination and take advantage of positive externalities, as evidenced by efforts in other areas, such as the International Whaling Commission moratorium on commercial whaling in 1982. The world needs to advance its norms related to the scale of investment for PPR. Such norms may support PPR, even if they may not be relied on in the “crisis mode” of an ongoing pandemic. As described in Chapter 1, the window of political will for rethinking global investment strategies and establishing new norms concerning global public health may close quickly after the world’s recovery from the COVID-19 pandemic.
Sands and others (2016b) note that infectious disease crises can cause immense economic disruption, yet mainstream macroeconomic forecasting consistently underestimates the risk. This results in underinvestment in PPR. The authors attributed this to the absence of readily available input data for such analyses and suggested an approach by which the global health community could help develop such inputs and a framework to use them in assessing economic vulnerability of individual countries and regions to infectious disease crises. They argued that incorporating these risks in influential macroeconomic analyses, such as the reports from IMF’s Article IV consultations, rating agencies, and risk consultancies, would improve economic risk forecasting and reinforce government and donor incentives to mitigate infectious disease risks.
Another reason for historically low spending may be that investment in pandemic preparedness has aspects of a global public good. Research and development (R&D) grants by one country produce knowledge and new products that benefit all. Other countries have an incentive to save on grant spending by free riding on the investment of others. The epidemiology of disease spread and mutation means that disease may not be quelled anywhere unless quelled everywhere. The interconnectedness of the global economy may mean that even a country with low infection rates may continue to experience a recession if its trading partners are suffering from an outbreak. A country with adequate vaccine capacity may not be able to run it—or factories for any other products—if global supply chains are knotted up by a pandemic. The World Bank Group (2019) dubs pandemic preparedness the “ultimate” public good: it is non-excludable, is non-rival, and provides benefits to countries across the world. Economic logic says that countries only considering their individual interests will underinvest in public goods unless active measures are taken to agree on a better solution. For pandemic
response, this may call for an international agreement, asking participating countries to boost spending—for example, through public policy legislation and taxation.
Even if the whole world were coordinated in a single country, pharmaceutical manufacturers and other firms in a free market could not be counted on to provide an adequate level of investment for an output, such as pandemic preparedness, due to the large gap between social and commercial incentives. We mentioned global social benefits from of a course of COVID-19 capacity estimated to be more than $5,000 (Kremer, 2021). However, the private value and return on investment for the companies actually installing this capacity does not match the social value. As noted by Michael Kremer, firms earned $6–$40 for a course of COVID-19 vaccine, orders of magnitude less than the social benefit. Competition and private information about benefits restrain the prices firms can charge. High prices are repugnant to consumers, and may spur regulation in ordinary times, but that is especially acute during a crisis. These forces are understandable, but reduced return on investment leads, by the aforementioned principle, to less investment and production. That repugnance during a pandemic may lead to acute undersupply. With firms rarely able to charge anything close to the social value, pharmaceutical companies should be offered substantial government incentives that are creatively structured to avoid getting the most value for spending—for example, through AMCs (Kremer, 2021).
Another relevant economic principle is the value of early investment. Due to compound interest, saving $100 per year for 10 years leads to a larger account than $1,000 saved in year 10. Investing before a pandemic has similar compound returns. Expanding vaccine capacity slowly during times of no disease pandemics can be much cheaper, avoiding price spikes, supply chain disruptions, and out-and-out stockouts of needed inputs. Forces for nationalizing resources present during a crisis are absent during times of no disease pandemics and may accumulate to an adequate level that can then serve the globe during crises.
Behavioral economics emphasizes the importance of salience to decision making. Rational computations based on deaths and other adverse events do not necessarily drive investments or shape decision making. (See the discussion of antimicrobial resistance and the O’Neill Report in Chapter 1.) Vocal supporters are a crucial catalyst in spurring investment, as evident in the relative wealth of funding for HIV and dearth of funding for efforts to combat influenza, noncommunicable diseases, and antimicrobial resistance. Momentum may also be an issue. As described in Chapter 1, the window of political will for rethinking global investment strategies may close quickly after the world’s recovery from the COVID-19 pandemic.
IPPPR’S ARGUMENT ABOUT PPR FINANCING
IPPPR’s Financing Pandemic Preparedness and Response report made a compelling case for financing PPR outside of the traditional voluntary and overseas development assistance channels (Radin and Eleftheriades, 2021). The logic is consistent with many of the economic principles laid out in the previous section. The report notes that PPR benefits are experienced globally, so it is insufficient to manage a global public good exclusively at a national level.
However, the international system lacks the financing architecture to adequately coordinate and accelerate investment in the global public goods required to contain outbreaks. HICs have largely deprioritized PPR investments and relegated these to discretionary aid, even though these countries have the most to lose economically. HICs’ higher funding capacities and higher economic benefits from ending a pandemic can justify asking them to contribute more, even a higher share of their incomes, to finance this type of public good. It should not be financed through aid budgets. These investments should be a policy priority because they generate such large positive externalities for the entire world. Effective coordination of these investments would be a helpful “nudge” to HICs and generate large benefits, in contrast to other investments that they would make anyway.
The IPPPR report authors argue that during the COVID-19 pandemic, the main non-vaccine-specific financing gaps were in the areas of preparedness and early response, during the first 100 days from transition from outbreak to pandemic. They also argue that numerous global institutions are capable of supporting—or upscaling support for—PPR. On the vaccine front, these include CEPI and its capacity for R&D, ACT-A, including COVAX for pooled procurement, WHO’s Emergency Program for technical support and operational capacity, and the Bill & Melinda Gates Foundation (BMGF), Gavi, and regional organizations such as the Africa CDC for coordinating delivery. Thus, there is no need to establish new institutions; what is needed is improved coordination and scale of financing.
SCALE OF INVESTMENT NEEDED TO IMPROVE GLOBAL PPR
Before and after the COVID-19 pandemic, studies developed estimates about the scale of investments needed to demonstrably improve PPR response worldwide. All of those studies underscore the importance of billions of dollars more in annual investments and improved coordination for the allocation and monitoring of funding; Table 5-2 provides an overview of the proposed quantities of funding. However, none of those studies looked specifically at influenza.
TABLE 5-2 Proposed Quantities of Funding Needed for Pandemic Preparedness
| Proposed Plan | Funding Estimates |
|---|---|
| Rockefeller Plan (2021) | $44 billion through IMF Special Drawing Rights (SDRs) for COVID-19 to achieve a 70 percent vaccination rate in LMICs by the end of 2022 $650 billion in SDRs to catalyze an inclusive, sustainable global recovery in LMICs at no additional cost to wealthy nations |
| Craven et al. (2020) |
$357 billion over 10 years estimated to build five epidemic preparedness pillars $85–$130 billion over 2 years for ramp-up phase to close epidemic preparedness gaps, followed by steady-state funding of $20–$50 billion annually
|
| Independent Panel for Pandemic Preparedness and Response (IPPPR, 2021) | $5–$10 billion/year for preparedness $50–$100 billion in a crisis distributed by an international pandemic-financing facility |
| High-Level Independent Panel on Financing the Global Commons for Pandemic Preparedness and Response (HLIP, 2021) | $15 billion/year increase in international financing (public funding) for pandemic preparedness and response 1 percent GDP increase for LMICs for public health spending over the next 5 years |
| Global Health Risk Framework, National Academies of Sciences, Engineering, and Medicine (2016) | $4.5 billion/year committed by G7, G20, and UN to mobilize incremental financial resources Independent assessment in 2017 and every 3 years thereafter to monitor progress of implementation for upgrading public health systems, the cost of enhancing WHO’s PPR capabilities, and proposed incremental investment in research and development |
| World Bank (2012) | $1.9–$3.4 billion/year for preparedness in One Health systems to bring the global zoonotic disease prevention and control system up to World Organisation for Animal Health and WHO standards, depending on disease prevalence |
Preparedness-Focused Investments
In 2016, the Global Health Risk Framework made a case for investing in pandemic preparedness, which recommended that the G7, G20, and UN should commit to mobilizing incremental financial resources required—estimated to be at least US$4.5 billion per year—and monitoring the progress of implementation by an independent assessment in 2017 and every 3 years thereafter (NASEM, 2016; Sands et al., 2016a).
In May 2021, McKinsey estimated that five epidemic preparedness pillars could be built over 10 years at a cost of $357 billion (Craven et al., 2020). The report calls for a ramp-up phase to close gaps—at an estimated $85–$130 billion over 2 years—followed by steady-state funding of $20–$50 billion annually. Financing estimates for five pillars include the following:
- “Always on” systems, including border health, supply chain preparation and global stockpile, emergency operations, communication and messaging, regular simulations and cross-sector exercises: $56 billion total
- Disease surveillance, including pathogen surveillance and sequencing ($48 billion), U.S. national public health institutes ($3 billion), specialized surveillance programs ($4 billion), notifiable-disease and Integrated Disease Surveillance and Response-like surveillance ($19 billion), population-representative surveillance foundation ($19 billion), and data integration ($4 billion): $96 billion total
- Prevention agenda, including global immunization, limited human-wildlife interactions, mapped global virome, and containing antimicrobial resistance: $88 billion total
- Health care capacity, including assessing gaps in health care systems and closing pandemic-specific gaps: $54 billion total
- R&D, including new antiviral, antibody, and vaccine platforms ($6 billion); scaled vaccine manufacturing capacity ($42 billion); closing known vaccine and therapeutic gaps ($14 billion): $62 billion total
The IPPPR report proposed an International PPR Financing Facility to raise additional reliable financing for pandemic preparedness and rapid surge financing for response in a pandemic (Radin and Eleftheriades, 2021). Over the long term (10–15 years), this would require $5–$10 billion per year for ongoing preparedness, providing the ability to disburse up to $50–$100 billion at short notice. This single instrument would effectively finance two windows: (1) stable long-term cash flows for capacity and (2) front-loading financing that could kick in within 2 weeks to contain an outbreak through prearranged operational response plans.
In July 2021, the G20 High-Level Independent Panel on Financing the Global Commons for Pandemic Preparedness and Response’s (HLIP’s) report, A Global Deal for Our Pandemic Age¸ called for $75 billion over the next 5 years in international financing for PPR, to plug major existing gaps. This would represent at least doubling current spending levels and be targeted to four gaps: infectious disease surveillance, resilience of national health systems, global capacity to supply and deliver vaccines and other medical countermeasures, and global governance. The High Level Independent Panel underscores that these costs are “negligible” compared to those of a major pandemic.
Response-Focused Investment
Response-focused investment spans both short- and long-term response. Investment in short-term response includes both preparedness and the first few months of a response phase. For instance, the IPPPR facility is designed such that international finance institutions and multilateral and regional bank capital would kick in after 3–4 months, with the ability to cover months 3–18 of a pandemic (Radin and Eleftheriades, 2021). Proposed investments in a longer-term response include the Rockefeller Plan (2021), which called for $44 billion through IMF Special Drawing Rights for COVID-19; this plan could potentially be replicated in another pandemic scenario (The Rockefeller Foundation, 2021).
STRATEGIES TO DRIVE INVESTMENT IN INFLUENZA PREPAREDNESS AND RESPONSE: ENGAGING HIGH- AND MIDDLE-INCOME COUNTRIES
PPR are public goods that are not most effectively funded by development aid. Thus, it is critical to bring HICs and middle-income countries to the table to drive investments in both preparedness—such as surveillance, vaccine R&D, and keeping facilities warm—and response. The latter includes both early and longer-term response to finance vaccine production at the scale required and then to procure and deploy the vaccines. The entire world will not be able to invest in PPR—particularly in components such as R&D and surveillance (Stutzman et al., 2020)—because LICs have opportunity costs. Ideally, countries would contribute their own funding to support health systems strengthening and surveillance—for example, Ghana is seeking international awards and funding from U.S. CDC and PIP to bolster its surveillance capacities—but this is not always feasible. Therefore, in the context of PPR, HICs should make a “big bet” that is in their own self-interest, because HICs have the most to lose, economically, during a pandemic. This point was underscored by the G20 HLIP report.
Multiple studies have concluded that lack of coordination for global access to COVID-19 vaccines significantly threatens HICs’ economic recovery. For example, a study commissioned by the International Chamber of Commerce Research Foundation considered the global costs for advanced economies—after their populations are fully vaccinated—due to a continued uncoordinated approach to global vaccine distribution. It estimated that the world risks a loss of global GDP of up to US$9.2 trillion in 2021 alone; advanced economies would pay 49 percent of that regardless of their own vaccination rates (Çakmaklı et al., 2021; CARE Action + Booz Allen Hamilton, 2021).
To move these efforts forward, a key question is how to coordinate and structure middle- and high-income countries’ investments in this global public good, which promotes equity, in a way that makes it the most attractive to them. The approach for universal health coverage (e.g., meetings of ministers of health and finance) is unlikely to suffice, because national investments alone are not adequate for funding and coordinating global public goods. Instead, it may be best for HICs to adopt a system of assessed contributions to a major fund. The proposed International PPR Financing Facility would raise additional reliable financing for pandemic preparedness and rapid surge financing for response (Radin and Eleftheriades, 2021). Contributions to it should be based on an ability-to-pay formula, with larger and higher-income countries paying the most. These contributions would not be considered official development assistance and in addition to that type of aid. The proposed Global Health Threats Council could allocate and monitor funding from this investment. Factors for success would be a lean secretariat, coupled with a focus on working with and through existing organizations. The intent is to fill the gap in preparedness and early response for about 3–4 months, until international financial institutions and multilateral bank capital are triggered.
KEY CONSIDERATIONS IN DETERMINING SCALE AND INCENTIVES FOR FINANCING PPR
Drawing on the IPPPR and other recent reports, the committee identified five key categories to consider when determining the scale of financing and types of incentives to use for influenza and aligned pandemic vaccine financing. These categories, which span both the preparedness and response landscape, are (1) surveillance, (2) R&D, (3) manufacturing, (4) procurement, and (5) delivery and deployment. Within each category, it is important to consider what is known about influenza funding, which considerations should be taken into account when planning for future financing, and what financing structures can generate the incentives for influenza.
Surveillance
The PIP Framework’s financing for surveillance is inadequate, and WHO’s Global Influenza Surveillance and Response System (GISRS) has had issues securing sufficient funding. To address this shortfall, incentives should be put in place that balance local ownership with regional or global financing.
Current Funding for Influenza and Other Respiratory Pathogens
Overall, the state of funding for surveillance of influenza and other respiratory pathogens is insufficient. WHO’s funding for influenza surveillance is inadequate to cover scaling up GISRS surveillance to other respiratory pathogens, and it is supplemented by relatively small streams of financing from bilateral actors and partnerships. The committee did not systematically calculate the amounts spent on surveillance at the multilateral, regional, or national levels, as doing so would require a greater degree of transparency and longer study time frame. However, it did analyze examples of influenza-specific funding by several major players, including WHO, the World Bank, The Rockefeller Foundation, and several U.S. government agencies.
The PIP Framework has a yearly budget of approximately $28 million, of which about 90 percent goes into the Partner Contribution’s Preparedness and Response funds. This pool is further broken down into (1) response funds and (2) preparedness funds for use at the country and regional levels. The latter includes funds for building surveillance capacity in WHO member states (WHO, 2019). Partner Contributions are based on a formula established when the PIP Framework was launched and have remained static since. GISRS has had difficulty securing funding for its running budget of approximately $30 million per year; it has had to seek international awards through entities such as the U.S. CDC (Dauphin, 2015). OIE/FAO Joint Network of Expertise on Animal Influenza (OFFLU), the global network of expertise on animal influenzas, is a joint collaboration among the Food and Agriculture Organization of the United Nations (FAO), the World Organisation for Animal Health (OIE), and WHO. OFFLU is poorly funded, receiving support averaging around $250,000 annually between 2005 and 2015 (Dauphin, 2015).
The World Bank’s Regional Disease Surveillance Systems Enhancement (REDISSE) Programs I and II are financed by a combination of International Development Association credits and grants, International Bank for Reconstruction and Development loans, and single- and multi-donor trust funds. These programs have invested in building technical capacity
in LMICs (World Bank, 2020). Between 2007 and 2012, the Rockefeller Foundation’s Disease Surveillance Networks Initiative also provided grants, mostly targeted regionally. This funding led to a number of smaller regional networks, including the Mekong Basin Disease Surveillance network, East African Integrated Disease Surveillance network, and Southern Africa Centre for Infectious Disease Surveillance (The Rockefeller Foundation, 2013). REDISSE provides an example of how ministries of finance can be regionally linked for surveillance activities through regional grant funding or loan obligations. The East-Africa Public Health Laboratory Networking Project (EAPHLNP, 2021) and African Integrated Disease Surveillance network also show how national laboratories can function with regional interdependence. Each has received political buy-in and guidance from the African Union.
Additional surveillance strengthening programs include the PREDICT project, which was part of the U.S. Agency for International Development’s (USAID’s) Emerging Pandemic Threats program. It is led by the One Health Institute at the University of California, Davis, and has worked to improve both global surveillance for pathogens with spillover potential and workforce training. It received about $200 million over 10 years (Klein, 2020). The Preventing Emerging Pathogenic Threats program has also been allocated $9.97 million over 3.5 years by the U.S. Defense Advanced Research Projects Agency (DARPA) to focus on the Lassa and Ebola viruses (UC Davis News and Media Relations, 2019).
A major gap exists between these funding streams and the financing required to ramp up investments in surveillance for epidemic preparedness. As noted, McKinsey estimated that $25–$40 billion would be needed annually for 2 years, followed by $6–$10 billion per year for steady-state functions (Craven et al., 2020). In 2020, Dobson et al. called for an estimated $22–$31 billion per year on a global scale for preparedness against zoonotic disease outbreaks. This would support initiatives to monitor wildlife trade, programs to reduce spillovers, and programs for early detection and control, among other areas (Dobson et al., 2020). Morgan et al. (2021) estimated that supporting the implementation of integrated, sustainable, country-owned systems for national surveillance would require an investment of about $1–$4 per person each year.
Considerations for Future Financing
Ensuring future financing for surveillance warrants several key considerations. A crucial lesson learned from the COVID-19 pandemic is that collecting data daily—which is not currently done for influenza and many other diseases—can be a powerful, positive incentive for investment. Additionally, funding and governance should emphasize local ownership rather than a top-down approach. Domestic financing at the local level is critical,
because implementing and maintaining systems for disease surveillance cannot rely exclusively on global funds. The tendency is to focus on the genomic and centralized aspects of surveillance at the expense of on-the-ground molecular testing. Thus, financing needs to address broader steps crucial to effective surveillance systems, such as national planning for the use of those systems.
Financing Structures to Generate Incentives
Financing structures can be leveraged to generate incentives to fund surveillance systems. For example, external financing has significant positive externalities and a powerful argument for it, but this may require core funding and defined contributions for sustained investment. Attempting to fund surveillance through episodic sources, such as the World Bank’s International Development Association, is problematic. An alternative option is using funds earmarked for surveillance for governmental and nongovernmental recipients (e.g., REDISSE II, Africa CDC). The Global Fund and Gavi use co-financing arrangements as a model for incentivizing domestic investment alongside external financial support. An opportunity also exists in the investments in surveillance for COVID-19, and other diseases could be leveraged as a foundation for building broader national and/or regional capabilities, including for influenza.
R&D
Pandemic vaccines require the ability to undertake R&D for new vaccines and manufacture the vaccines at scale. Dedicated push funding is required because the pull from seasonal influenza vaccines is not sufficient to drive pandemic vaccine development.
Current Funding for Influenza
Funding for platform technologies and next-generation influenza vaccines is small relative to their potential to transform the vaccine landscape (see Chapter 4). Although the Sabin-Aspen Vaccine Science and Policy Group has called for a well-funded moonshot program for a universal vaccine, this has largely not been met with commensurate financing (Sabin-Aspen Vaccine Science and Policy Group, 2019). The most substantial investments are from HICs and high-income regions, such as the United States and the European Union. CEPI has made significant strides in forging alliances for financing and coordinating R&D for new vaccines to prevent and control infectious diseases during epidemics. However, CEPI’s mandate is mainly focused on developing vaccines in the early stages, through Phase
II clinical trials, and influenza is not in its portfolio. New solutions are needed to fund high-cost, late-stage development (Sabin-Aspen Vaccine Science and Policy Group, 2019).
On the positive side, some political buy-in exists for the universal influenza vaccine. The U.S. National Institutes of Health’s National Institute of Allergy and Infectious Diseases (NIAID) 2018 strategic plan highlights its commitment to support the research needed to advance the development of a vaccine that provides long-lasting protection against multiple strains for seasonal and potentially pandemic influenza (Erbelding et al., 2018). The strategy notes that “advances in influenza virology, immunology, and vaccinology make the development of a ‘universal’ influenza vaccine more feasible than a decade ago” (Erbelding et al., 2018, p. 347) due to efficiencies and insights in deep-gene sequencing and advances in structural biology, among other scientific innovations. In September 2019, the Trump administration issued an executive order directing the U.S. Department of Health and Human Services to modernize production by supporting the development of new manufacturing technologies for more robust and longer-lasting vaccines against a broader range of influenza viruses (Schnirring, 2019).
Although the committee did not systematically compile financial data, it identified illustrative examples of the current scale of funding for influenza vaccine R&D. The U.S. government spends $250–$300 million annually on influenza research, in addition to further spending on related programs, such as biodefense and biotechnology (Sabin-Aspen Vaccine Science and Policy Group, 2019). The European Innovation Council Accelerator Pilot has invested €148 million in 36 companies, including for the multi-season universal influenza vaccine OXIVAX (European Commission, 2021). As of 2019, the European Union had also funded initiatives for novel influenza vaccine development through the seventh Framework Programme for Research and Technological Development (FP7) and Horizon 2020, its research and innovation program. FP7 funded 25 influenza vaccine projects for about €87 million; an additional €18 million was granted under Horizon 2020 (Navarro-Torné et al., 2019), which also established a new influenza research partnership with India that includes matched financing (Press Trust of India, 2018).
In 2018, BMGF launched a $12 million Universal Influenza Vaccine Development Grand Challenge with the goal of identifying
novel, transformative concepts that will lead to the development of universal influenza vaccines offering protection from morbidity and mortality caused by all subtypes of circulating and emerging (drifted and shifted) Influenza A subtype viruses and Influenza B lineage viruses for at least three to five years.
Pilot grants ranging from $250,000 to $2 million were awarded by BMGF and the Flu Lab—an influenza-focused charitable organization—with the aim of starting clinical trials by 2021 (Sabin-Aspen Vaccine Science and Policy Group, 2019). The Wellcome Trust supported the development of CIDRAP’s R&D roadmap to accelerate progress toward development of universal or broadly protective influenza vaccines (Sabin-Aspen Vaccine Science and Policy Group, 2019).
Considerations for Future Financing
Multiple considerations should be taken into account to secure future financing for influenza vaccine R&D. A 2016 WHO consultation on Global Action Plan on Influenza Vaccines (GAP I–II) identified a host of challenges related to current financing mechanisms, which underscore the need for the industry to create stronger incentives—and secure substantially more funding—to push the development of platform technologies and universal vaccines (Røttingen, 2016). Specifically, long-standing challenges for influenza vaccines include the lengthy and costly business of failure. Bringing new products to launch can take more than a decade and cost at least $1 billion—yet, at the outset, the chance of success is just 5 percent. U.S. public funding for rapid R&D response capabilities is insufficient and heavily reliant on BARDA. Furthermore, developing novel-antigen, novel-platform, broad-spectrum vaccines is hampered by many technical and regulatory challenges. Thus, a core challenge is to find ways to incentivize a smooth and sustainable shift of R&D efforts and know-how from a relatively stable market of current seasonal vaccines to a more uncertain market of pandemic and universal vaccines (Røttingen, 2016).
Funding structures for next-generation research targets are often not flexible enough to allow quick pivoting or redirection of resources. GloPID-R highlighted this barrier, which is a particular challenge for the rapid mobilization of research funds and resources, early engagements with ethics committees, and adaptive studies and trial design (Norton et al., 2020). In its 2020 background paper focused on financing pandemic preparedness R&D, the Global Preparedness Monitoring Board (GPMB)—a joint effort of the World Bank and WHO—similarly emphasized the need for major national research organizations and their governments to improve funding for collaborative R&D (World Bank Group, 2019).
Financing Structures to Generate Incentives
Financing structures that could generate incentives for R&D include push and pull funding mechanisms and structured funding. COVID-19 economic models have found that push incentives—such as direct-cost
reimbursement—were hindered by funders’ lack of visibility into firms’ private cost information (Snyder et al., 2020). However, push funding could be used for early influenza vaccine dosing trials. For example, during the 2018 yellow fever epidemic, Brazil reduced the vaccine dose to one-fifth—in accordance with WHO’s advice—which accelerated deployment. Evidence is promising that this approach might be effective for SARS-CoV-2, which could also be an analog for an influenza vaccine. For influenza, some evidence indicates that reducing the dose of an interim musculature shot or switching to intradermal could be effective strategies for vaccine dose-stretching (Kremer, 2021).
Economic modeling has demonstrated that pull incentives awarding advance purchase agreements to bidding firms were effective for late-stage vaccine development (i.e., Phase III clinical trials and manufacturing) during COVID-19 (Snyder et al., 2020). The optimal pull programs incentivized participation of nearly all the pharmaceutical firms involved in developing 10 COVID-19 viable candidates, nearly doubling the net benefits generated from the free market. AMCs can be an effective pull funding mechanism when the target is sufficiently defined. An AMC works by reducing risks—it defines the target vaccine minimum standards (efficacy, public health impact), total AMC market size, contract price, and predicted demand. Innovators and firms are assured of a subsidized price if they develop a product meeting standards and can agree to abide by affordable prices even after the AMC is depleted. Donors or country government funders are assured that funds will only be used if a highly valuable product is developed, that a competitive market is established for firms, and that their up-front investment results in sustainable supply after the AMC is depleted. Although it is not yet clear that the influenza target is well defined, working toward a universal vaccine should be a priority.
There is a strong economic case for middle-income countries, such as Russia and China, to invest in influenza vaccine R&D (Yang, 2021). The GPMB suggested several mechanisms for structured funding of vaccine development—albeit not influenza specific (World Bank Group, 2019). The first is to establish CEPI-like mechanisms for diagnostics and therapeutics to jump-start product development. This could be accomplished by new entities, an expanded CEPI structure, or an expansion of structures like GloPID-R. The second is to create research funders preposition funds—for example, through a multi-donor trust fund at the World Bank—that can be released to research organizations within days to kick-start essential research when an outbreak occurs. Additionally, the World Bank could add a senior health scientist to its own leadership to spearhead its initiatives to strengthen R&D preparedness during inter-pandemic periods. The third suggestion is that “no regret” funding should be always available to incentivize at-risk R&D before a PHEIC is declared. This amount should
be reasonable to spend or lose, even if an outbreak does not expand to a major epidemic or pandemic.
Manufacturing
The next pandemic will not necessarily be caused by an influenza virus—it might even be a pathogen currently unknown to cause human disease (i.e., “Disease X”). Thus, vaccine manufacturing networks should not focus exclusively on influenza. This will require garnering broader investment in pathogen R&D alongside a universal influenza vaccine and establishing incentives for rapid scale-up of production capacity. It could be possible to create a ranked system of regionalized manufacturing centers aligned with existing agreements for clinical trials.
Current Funding for Influenza
It is critical to explore strategies for keeping mRNA and other platform technology manufacturing processes warm between pandemics for both influenza and coronavirus vaccines. The Pandemic Preparedness Partnership, launched by the UK government, prepared a report for the 2021 G7, mapping a 100 Day Mission to respond to future threats by embedding pandemic-ready manufacturing processes and capacity into business as usual (PPP, 2021). The report identifies multiple opportunities to leverage expanded vaccine production capacity between pandemics to justify maintaining surge capacity—for instance, via mass adult vaccination campaigns for various diseases, including influenza and coronaviruses. However, as discussed in Chapter 4, mRNA/platform technology mapping for influenza demonstrates that the current system for keeping facilities warm is ill prepared for these rapidly evolving new technologies. Moreover, there remains no clear cost estimate for achieving an ever-warm state for influenza vaccine platform technology, which warrants urgent research.
Considerations for Future Financing
Considerations related to future financing to keep vaccine production systems warm between pandemics are influenced by the current impetus toward distributed manufacturing, for both influenza and other pathogens with pandemic potential. However, it is important to carefully consider the distributed manufacturing business model, rather than merely distributing for the sake of doing so. If the desired outcome is to administer the greatest number of vaccines to the most people as quickly as possible, the best strategy may not necessarily require excessive differentiation between consolidated and distributed manufacturing. For example, if the United States had the
entire global vaccine capacity and had immunized its population first, this might have only delayed global distribution by a few weeks. The more effective solution might involve early investment in critical technology that is generating returns, thus reducing scarcity of vaccines and associated products.
For suppliers of lifesaving materials, the general rule of thumb is at least five different manufacturers in at least five different locations. Regional and international collaborations can bolster efforts to create more diversified configurations of vaccine manufacturing, thereby promoting equity. For instance, the goals of the Africa Union Partnership for African Vaccine Manufacturing, launched in April 2021, include (1) developing a coordinated manufacturing agenda across the continent, (2) bolstering five regional partnership production sites over 10–15 years, (3) mobilizing financing, (4) strengthening regional regulation systems, (5) increasing technology transfers, and (6) developing African universities as R&D hubs (Jerving, 2021).
Most pressingly, in a distributed context, incentives need to be established to build the demand and expand the market for pandemic vaccines, because the private market alone will likely be insufficient to meet demand. Analyses of emerging infectious disease vaccines typically show that under realistic financing assumptions, expected returns are significantly negative (Vu et al., 2020).
Financing Structures to Generate Incentives
To keep vaccine manufacturing facilities warm between pandemics, several options for financing structures can generate market incentives. One strategy is to subsidize dormant capacity directly; however, maintaining facilities on standby is an expensive option. Another approach is to use manufacturing capacity for seasonal influenza to produce pandemic vaccines, although relying exclusively on this option is less viable as the system transitions away from egg-based technologies. A third option is to use the manufacturing capacity for other respiratory infections or diseases, such as adult vaccination for seasonal influenza or tuberculosis. This may be a more effective option in the shift to more versatile platform technologies.
Procurement and Access
For vaccine procurement and access, a future COVAX-like facility for influenza or other pandemic-potential pathogens should be designed with a focus on ensuring that it is resilient to deep political pressures.
Current Funding for Vaccine Procurement
Vaccine procurement methods fall into three main categories: self-procurement, donations through international organizations, and interstate
procurement. Within the self-procurement approach, a government purchases vaccines from manufacturers directly on the open market. This method typically allows states to independently select the manufacturer of their choice. Although the negotiation process can result in lower prices per unit, this approach requires specialized technical knowledge in medicine and law, the financial resources necessary, a certification process for licensing vaccine safety and efficacy, and the downstream-appropriate cold-chain infrastructure to store and deliver vaccines. Some self-procuring states use advance purchase agreements (APAs), which are contracts between a manufacturer and the government that lie dormant and unenforceable until triggered by a predetermined event, when they become legally binding. The second method is to procure through international organizations (sometimes called “pooled co-procurement”), in which nongovernmental and intergovernmental organizations, such as Gavi, UNICEF, and WHO, acquire vaccines, typically through tiered pricing, and countries pay a percentage of the cost through co-financing. This approach can support providing vaccines to LICs lacking the capacity to self-procure. In some cases, the organizations procure and administer the vaccines directly if the state lacks the infrastructure. AMCs can fall under and support this mechanism; for example, the G7 and BMGF committed to buy a vaccine against LMIC-specific strains of pneumococcal disease when this vaccine was developed.
The third method involves the interstate sale of vaccines. This approach is relatively rare, but it was used during the 2009 H1N1 influenza pandemic (Turner, 2016). During that pandemic, HICs mostly used self-procurement, while LICs tended to depend on donations through international organizations. Of the more than 634 million doses obtained, almost 60 percent were via central government purchases, nearly 30 percent from other sources, and only about 12 percent deployed internationally through donations to WHO’s Vaccine Deployment Initiative, which was created specifically to manage the distribution of vaccines then (see Chapter 2). That 12 percent of donated vaccines accounted for about 94 and 91 percent of the African and Southeast Asian regions’ vaccines, respectively. Self-procurement was heavily associated with HICs—nearly 99 percent of vaccines in the European region were acquired through self-procurement, compared to just 6 percent in Africa. In a case study of vaccine procurement during the 2009 pandemic, Eccleston-Turner maintains that these methods were ineffective for developing states and predicts that during the next influenza pandemic, global demand for vaccines will exceed the worldwide supply, which is dominated by developed states with existing APAs (Turner, 2016).
Similar trends have emerged during the COVID-19 pandemic. HICs have cornered the vaccine market through APAs with prices that are heavily dependent on individual procurement contracts. Only a relatively small percentage of vaccines are donated or sold to international organizations for
allocation to LMICs through entities such as COVAX. Even before vaccines were authorized and approved, many countries entered into bilateral bulk purchasing agreements with individual manufacturers to secure doses at lower negotiated prices. For instance, through two separate agreements, Moderna secured a $3.2 billion contract to provide the United States with 200 million doses at roughly $16 per dose; the European Union secured doses from Moderna at $18 per dose. Pfizer also received two separate contracts for a total of 200 million doses at $19.50 per dose, while the European Union paid just under $15.15 (Ramachandran et al., 2021).
However, prices of vaccines may not reflect a fair return on public investments in vaccine R&D, and these types of “competitive pricing” could quickly make vaccines unaffordable for LMICs. At a 2020 hearing of a U.S. House Oversight and Investigations Subcommittee, leading manufacturers were asked whether they would supply COVID-19 vaccines at cost or no profit. Only two—AstraZeneca and Johnson & Johnson—stated that they would be willing to do so for a limited number of doses or during “the emergency pandemic period.” Both Pfizer and Moderna said that they would not. Further uncertainty is fueled by the lack of clarity around how to define the “pandemic period” during which companies have promised to supply their products at lower prices or to share technology with other manufacturers. Although AstraZeneca pledged not to profit from its vaccine during the pandemic period, the company also specified that it could declare the pandemic over by July 2021. Pharmaceutical executives have stated they anticipate returning to “commercial pricing” as early as late 2021, and Pfizer has already raised the prices of the European Union’s future orders by more than 60 percent (Ramachandran et al., 2021).
In early March 2021, COVAX declared its intention to make 1.8 billion doses available to countries with AMCs by the end of 2021, which would cover an estimated 28 percent of those countries’ populations (Hall et al., 2021). In the interim, some LMICs and regions have used self-procurement. However, this has exacerbated problems related to manufacturers charging customers different prices for the same product. For example, according to a senior health official in South Africa, the country purchased 1.5 million doses of the Oxford/Astra-Zeneca vaccine for health workers at $5.25 per dose, more than double the price ($2.15 per dose) for the European Union. This price disparity emerged only due to inadvertent disclosures, as the European Union had kept its prices confidential in exchange for vaccine discounts. The South African Deputy Director-General of Health said his government had been told that $5.25 was the set price for a country classified by the World Bank as upper middle income (Dyer, 2021).
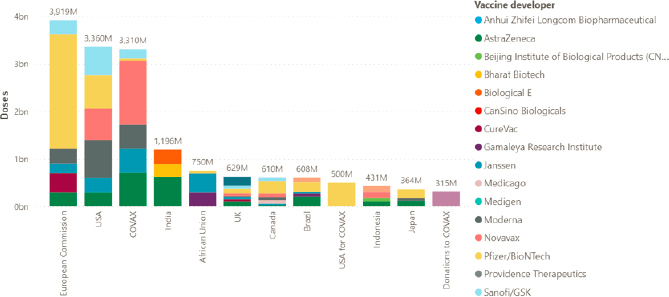
Figure 5-3 provides an overview of COVID-19 vaccine supply agreements by country and group, illustrating the extent to which APAs have dominated the market, often leaving behind LMICs. For instance, COVAX
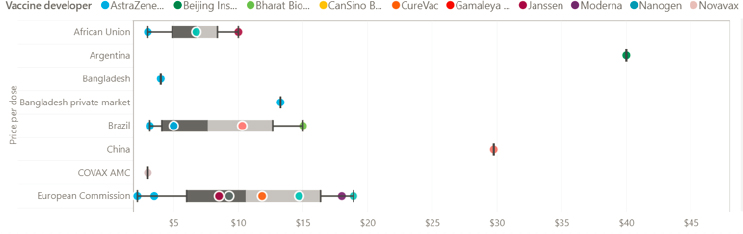
collectively procured approximately the same percentage of vaccines as the United States has individually, as of June 2021. UNICEF has tracked reported COVID-19 vaccine prices per dose, which demonstrates how the procurement system has led to wide variances in cost (for the same vaccines) for different customers (see Figure 5-4).
Considerations for Future Financing
The global distribution of seasonal influenza vaccines remains highly inequitable across regions. The PIP Framework’s procurement system for LMICs covers just a small percentage of the total vaccine capacity, so a new COVAX-like procurement and access facility will likely be required to ensure that countries in the WHO Eastern Mediterranean, South East Asia, and Africa Regional Offices have access. Between 2004 and 2017, the share
SOURCE: https://www.unicef.org/supply/covid-19-vaccine-market-dashboard (accessed December 19, 2021).
SOURCE: https://www.unicef.org/supply/covid-19-vaccine-market-dashboard (accessed December 19, 2021).
of seasonal doses increased in the Americas (41–55 percent) and declined in both Europe (34–22 percent) and the Western Pacific (24–17 percent). In Africa, the Eastern Mediterranean, and Southeast Asia, the combined share of doses increased from 5 to 6 percent. The latter is unacceptably low, given that these regions represent almost half of the world’s population (Sparrow et al., 2021). Furthermore, in determining the optimally equitable distribution, it is important to consider economic losses in addition to death rates.
The COVID-19 pandemic has demonstrated that theoretical models for effective and equitable delivery may not stand up to political pressure. Efforts by COVAX and other entities to improve global access were undermined by vaccine nationalism; amid the chaos of competing priorities, HICs tended to embrace bilateral deals that would allow for scale-up. Successful incentives to promote equitable distribution must be catalyzed by political commitments and supported by mechanisms and models of cooperation that can withstand political pressure.
Game theory analyses have identified two ways that mechanisms such as COVAX can shape incentives to promote scale-up and broaden global access to vaccines. The first is to leverage self-enforcing good behavior norms, global prestige, and social sanctioning. This approach is based on articulating principles of how countries ought to structure bilateral deals and transparency. The second strategy is to create extra benefits that only those making equitable or “good” deals can enjoy (McAdams et al., 2020). There have been calls to create separate versions of COVAX-like mechanisms for different diseases and expand existing facilities to vaccines against pandemic pathogens. However, it is difficult to predict the type of pandemic that is likely to strike next and important to design a platform specific to the immediate situation.
Financing Structures to Generate Incentives
Appropriate financing structures can generate procurement incentives and promote equitable access worldwide. For example, vaccine supplies for LICs should be included as components of grants and contracts with vaccine developers, as CEPI did with some manufacturers for COVID-19. This should be done before Phase III trials, when efficacy will be established and global demand will be high. Furthermore, providing up-front, at-risk manufacturing to global procurers, such as Gavi and the Global Fund, can enable large-volume pre-purchasing, as done by both the United States and the United Kingdom during COVID-19.
After its inception in 2016, CEPI developed an equitable access policy with input from key stakeholders and formed an Equitable Access Committee as a subcommittee of the CEPI Board. Early in the COVID-19 pandemic, CEPI included clauses in partnering agreements with vaccine
manufacturers that included a manufacturing component to secure doses for “a global procurement and allocation entity” that had not yet been developed. CEPI has sought to enable low pricing by requiring developers to enter into procurement deals with Gavi for COVAX. In such cases, the price is determined by negotiations between the developer and Gavi on behalf of the facility. In addition, CEPI has the right to redirect the supply to another public-sector procurer. All the agreements require compliance with CEPI’s equitable access policy and/or contain similar commitments in principle, including pricing at levels that are affordable to the people who need the vaccines (CEPI, 2021). This provides a model for how future equitable access clauses may be built into AMCs for influenza vaccines.
Delivery and Deployment
Although delivery and deployment of vaccines (“vaccination”) faces serious challenges—particularly in LICs—the financing for this is often overlooked.
Current Funding for Influenza
Delivery is often an underconsidered and underresourced cost, yet it can be more substantial than the cost of vaccine production. CARE estimates that countries or donors should invest $5 in delivery for every $1 invested in COVID-19 vaccine doses (Janoch et al., 2021). Although the delivery cost of influenza vaccines globally during a future pandemic has not been clearly quantified, estimates of the delivery component’s cost for COVID-19 may be instructive.
McKinsey has reported that during COVID-19, far less has been invested in planning for and implementing country vaccine rollout than for procurement. According to COVAX reports, in-country rollout costs about $1.50 per dose for delivery to the first 20 percent of the population, which does not include the health care workforce required. COVAX would only be able to provide a small percentage of this cost. An estimated $1 per dose—about $1.3 billion—would need to come from other domestic, bilateral, and/or multilateral sources. The World Bank’s $12 billion lending programs could theoretically help provide vaccine delivery support, but countries may not choose this borrowing mechanism (Hall et al., 2021).
A report from UNICEF, WHO, Gavi, BMGF, Harvard, ThinkWell, and the World Bank experts estimated that delivering COVID-19 vaccines to approximately 20 percent of the population in the 92 AMC countries would cost $2.018 billion—including country, regional, and global costs—about $1.66 per dose or $3.70 per person vaccinated with two doses (COVAX Working Group on Delivery Costs, 2021). Of this total cost, 57
percent is in-country outreach and fixed site delivery costs and 28 percent is country up-front fees (e.g., cold-chain installation, training). The report notes that delivery costs will vary substantially based on a range of factors, including specific target groups, population outreach strategies, geography, and local prices.
Another analysis estimated that about $74 billion would be required to reach COVID-19 vaccine herd immunity in LMICs—67 percent of this ($50 billion) for procurement and 33 percent ($24 billion) for delivery. This delivery cost is prohibitive for many countries; an estimated 20 percent of LMICs would have a total vaccine cost that is at least 10 times higher than their baseline annual immunization spending (Mustafa Diab et al., 2021).
Considerations for Future Financing
In considering financing strategies for vaccine delivery and deployment, a key lesson from the COVID-19 pandemic and other recent outbreaks is that most countries are far from adequately prepared to deploy critical medical supplies to their populations at the breadth and speed required. Health systems in LMICs typically lack capacity to deal with infectious diseases—particularly providing adult vaccines. In HICs, infectious disease control also tends to be a weak point in otherwise strong health systems. Successful responses to outbreaks tend to occur more often in countries with relatively strong existing health systems and recent infectious disease experience, such as Vietnam.
Furthermore, it is not efficient to create exclusively influenza-specific systems (e.g., cold chains, workforce). Thus, efforts to strengthen delivery and deployment capacity should build on and expand existing influenza frameworks such that they can be leveraged for a range of respiratory pathogens. Vaccine hesitancy continues to hinder influenza vaccine uptake, but local manufacturing capacity and stronger regulatory structures could increase local trust.
COVID-19 revealed the absence of an entity responsible for ensuring that liability policies and vaccine injury compensation programs were in place before the vaccine. However, COVAX did create a new program to compensate eligible individuals in 92 LMICs without requiring legal processes. The program, funded by a small levy on each dose, is the first and only vaccine injury compensation mechanism established on a global scale. Only 25 countries, including the United States and wealthier regions within Asia and Europe, have mechanisms to compensate individuals who receive serious vaccine-related injuries for routinely recommended vaccines (Sabin-Aspen Vaccine Science and Policy Group, 2021).
Financing Structures to Generate Incentives
Linking preparedness for influenza and other pathogens of pandemic potential to endemic diseases could help to increase national funding, including for deployment. Ideally, deployment strategies would not be specific to influenza. For example, they could cover other adult vaccinations, including COVID-19 boosters, in line with the WHO Immunization Agenda 2030, which emphasizes the importance of vaccination across the lifespan. External deployment financing should be channeled to support national vaccination and pandemic plans—particularly with the support of large nongovernmental organizations, such as the Bangladesh Rural Advancement Committee in Bangladesh, and through existing international organizations with experience in vaccine deployment–related planning, implementation, supply chains, and monitoring, such as UNICEF and Gavi.
KEY FINDINGS AND CONCLUSIONS
Surveillance Financing
- Surveillance system gaps are related both to financing and to barriers for more closely aligning or integrating these systems. They are far from being an LMIC problem; many HICs have moved away from financing surveillance systems efficiently, and many financed systems underperform in terms of accuracy, scope, and transparency. This is at least in part due to lack of political buy-in and country leadership.
- Domestic and external funding, accompanied by synchronous laws and legal instruments, may enable national surveillance systems to be more regionally operational and interdependent (e.g., sharing technical expertise, capital goods, operating expenditures, and information).
- The amounts and allocation of funding for surveillance should be sufficient to develop integrated surveillance systems that include all forms of influenza (human and animal) at a minimum and, ideally, include other respiratory pathogens with pandemic potential.
- Surveillance is primarily a global risk mitigation—not a development—issue. The economic theory argument is very compelling for surveillance of influenza and other pathogens with pandemic potential.
- While surveillance is a “best buy” in terms of cost-effectiveness, strengthening surveillance systems requires more than incentives. Each country’s surveillance system generates strong positive externalities
- that benefit all countries. This makes surveillance an area where there is a strong logic for sustained domestic and external financing. It should therefore not compete with other development priorities.
R&D, Manufacturing, and Procurement Financing
- For influenza and other pathogens with pandemic potential, industry is often insufficiently certain of the government interests and regulatory pathway to bring a profitable product to the market.
- A concerted effort is needed to demonstrate the direction that pharmaceutical companies should be moving in within the influenza space, toward transformation next-generation technologies and universal influenza vaccine targets.
- A moonshot for universal influenza vaccines should be initiated to support a portfolio of next-generation/platform vaccines, which would offer higher efficacy and the potential for a more sustainable business model for scale-up. Such an effort should benefit from and include a similar quest for more broadly protective (“variant-proof”) vaccines against coronaviruses with pandemic potential.
- The scale of investment in universal influenza vaccines to date (millions) is nowhere near the scale required (billions) to incentivize the risky undertaking of such R&D.
- Both push and pull incentives will be critical to demonstrate a proof of concept for the market of influenza vaccines. A massive push will be required to elicit pre-competitive scientific and biological analyses for influenza vaccine targets. AMCs (pulls) have most commonly been used by HIC donors to incentivize vaccine production and capacity for proven vaccine approaches, rather than to drive new science. AMCs can be usefully deployed as an incentive to solve hard biology problems such as those implicit in a universal influenza vaccine.
- Universal influenza and platform technology push and pull instruments should include equitable access provisions, in line with CEPI’s model during the COVID-19 pandemic.
Markets and Deployment
- The scalability of influenza vaccines is limited by the number of companies and production processes and by limited markets where the burden of this illness is uncertain.
- A vaccine market supported by an immunization program able to deliver vaccines, coupled with a population demand for these vaccines, are essential elements to ensure a sustainable vaccine manufacturing base.
- Stronger national plans for deploying seasonal influenza vaccines (using next-generation/platform technologies) and recognizing that vaccines will increasingly be available across the lifespan (in line with the WHO Immunization Agenda 2030) are essential for vaccine markets and have a strong public health rationale.
- Deployment activities require proper technical guidance and designated financial resources, but care should be taken to not frame deployment as a positive externality. Vaccine deployment requires designated financing from high- and middle-income countries according to a Global Public Good framework and also national accountability.
- UNICEF and Gavi have decades-long experience in deployment planning, implementation, supply chains, monitoring, and policies. Both are well positioned to take the lead on global coordination for vaccine deployment, particularly in LMICs.
REFERENCES
Boyce, M. R., A. Attal-Juncqua, J. Lin, S. McKay, and R. Katz. 2021. Global Fund contributions to health security in 10 countries, 2014–20: Mapping synergies between vertical disease programmes and capacities for preventing, detecting, and responding to public health emergencies. Lancet Global Health 9:e181–e188. https://www.thelancet.com/journals/langlo/article/PIIS2214-109X(20)30420-4/fulltext (accessed August 22, 2021).
Burwell, S., F. Townsend, T. Bollyky, and S. Patrick. 2020. Improving pandemic preparedness: Lessons from COVID-19. Council on Foreign Relations Independent Task Force Report Number 78. https://www.cfr.org/report/pandemic-preparedness-lessons-COVID-19/pdf/TFR_Pandemic_Preparedness.pdf (accessed October 25, 2021).
Çakmaklı, C., S. Demiralp, S. Kalemli-Özcan, S. Yeşiltaş, and M. A. Yıldırım. 2021. The economic case for global vaccinations: An epidemiological model with international production networks. Cambridge, MA: National Bureau of Economic Research. https://www.nber.org/system/files/working_papers/w28395/w28395.pdf (accessed October 25, 2021).
CARE Action + Booz Allen Hamilton. 2021. The case for U.S. investment in global COVID-19 vaccinations. https://www.care.org/wp-content/uploads/2021/04/The-Case-for-U.S.-Investment-in-Global-COVID-19-Vaccinations-Policy-Paper.pdf (accessed December 19, 2021).
CEPI (Coalition for Epidemic Preparedness Innovations). 2021. Enabling equitable access to COVID-19 vaccines: Summary of equitable access provisions in CEPI’s COVID-19 vaccine development agreements. https://www.readkong.com/page/enabling-equitable-access-to-covid-19-vaccines-summary-of-4837506 (accessed October 25, 2021).
Chagar, A., M. Thomas, L. Zuo, and M. Watson. 2021. The R&D response to COVID-19: What can we learn for the vaccine ecosystem? Sabin-Aspen Vaccine Science and Policy Group. https://www.kearney.com/documents/20152/134482507/THE+R%26D+RESPONSE+TO+COVID-19.pdf/4ff5d86b-fa90-c542-33ba-1fa2deb9f46e?t=1621954683000 (accessed October 25, 2021).
Cohen, J., and K. Kupferschmidt. 2021. Fairer shares. Science 372(6545):903.
COVAX (COVID-19 Vaccines Global Access) Working Group on Delivery Costs. 2021. Costs of delivering COVID-19 vaccine in 92 AMC countries. https://www.who.int/publications/m/item/costs-of-delivering-covid-19-vaccine-in-92-amc-countries (accessed December 19, 2021).
Craven, M., A. Sabow, L. Van der Veken, and M. Wilson. 2020. Not the last pandemic: Investing now to reimagine public health systems. McKinsey Report. https://www.mckinsey.com/industries/public-and-social-sector/our-insights/not-the-last-pandemic-investing-now-to-reimagine-public-health-systems (accessed October 24, 2021).
CRS (U.S. Congressional Research Service). 2021. Global economic effects of COVID-19. Report R46270. Updated July 9, 2021. https://crsreports.congress.gov/product/pdf/R/R46270 (accessed July 16, 2021).
Dauphin, G. 2015. Coordination tripartite OMS/FAO/OIE pour le contrôle et la prévention des virus influenza zoonotiques. Illustration avec OFFLU, réseau mondial d’expertise vétérinaire. Bulletin de l’Académie Vétérinaire de France 224.
Dobson, A. P., S. L. Pimm, L. Hannah, L. Kaufman, J. A. Ahumada, A. W. Ando, A. Bernstein, J. Busch, P. Daszak, and J. Engelmann. 2020. Ecology and economics for pandemic prevention. Science 369(6502):379–381.
Dyer, O. 2021. COVID-19: Countries are learning what others paid for vaccines. BMJ 372:n281.
Erbelding, E. J., D. J. Post, E. J. Stemmy, P. C. Roberts, A. D. Augustine, S. Ferguson, C. I. Paules, B. S. Graham, and A. S. Fauci. 2018. A universal influenza vaccine: The strategic plan for the National Institute of Allergy and Infectious Diseases. The Journal of Infectious Diseases 218(3):347–354.
European Commission. 2021. EU research and innovation supporting vaccine development for COVID-19. https://op.europa.eu/en/publication-detail/-/publication/69212491-6f3d-11eb-aeb5-01aa75ed71a1 (accessed December 19, 2021).
Glassman, A. 2021. Money in the bank: What’s behind slow demand for MDB financing for vaccines? https://www.cgdev.org/blog/money-bank-whats-behind-slow-demand-mdb-financing-vaccines (accessed June 16, 2021).
Hall, S., L. Kaplow, Y. S. Sun, and T. Z. Holt. 2021. “None are safe until all are safe”: COVID-19 vaccine rollout in low- and middle-income countries. McKinsey and Co. https://www.mckinsey.com/industries/healthcare-systems-and-services/our-insights/none-are-safe-until-all-are-safe-covid-19-vaccine-rollout-in-low-and-middle-income-countries (accessed October 24, 2021).
Hicklin, J., and H. Brown. 2021. Vaccine financing: How a redesigned IMF instrument can provide a shot in the arm for the global pandemic response. Washington, DC: Center for Global Development.
HLIP (High-Level Independent Panel). 2021. A global deal for our pandemic age: Report of the G20 High-Level Independent Panel on Financing the Global Commons for Pandemic Preparedness and Response. https://pandemic-financing.org/report/foreword (accessed October 24, 2021).
IPPPR (Independent Panel for Pandemic Preparedness and Response). 2021. COVID-19: Make it the last pandemic. https://theindependentpanel.org/wp-content/uploads/2021/05/COVID-19-Make-it-the-Last-Pandemic_final.pdf (accessed December 19, 2021).
Janoch, E., M. Rodriguez, and B. Turner. 2021. Our best shot: Women frontline health workers around the world are keeping you safe from COVID-19. https://www.care.org/news-and-stories/health/our-best-shot-women-frontline-health-workers-around-the-world-are-keeping-you-safe-from-covid-19 (accessed October 24, 2021).
Jerving, S. 2021. AU launches partnership for African vaccine manufacturing. https://www.devex.com/news/au-launches-partnership-for-african-vaccine-manufacturing-99654 (accessed April 14, 2021).
Kim, J. H., P. Hotez, C. Batista, O. Ergonul, J. P. Figueroa, S. Gilbert, M. Gursel, M. Hassanain, G. Kang, B. Lall, H. Larson, D. Naniche, T. Sheahan, S. Shoham, A. Wilder-Smith, N. Strub-Wourgaft, P. Yadav, and M. E. Bottazzi. 2021. Operation Warp Speed: Implications for global vaccine security. The Lancet Global Health 9(7):E1017–E1021. https://www.thelancet.com/journals/langlo/article/PIIS2214-109X(21)00140-6/fulltext (accessed December 19, 2021).
Klein, A. 2020. This won’t be the last pandemic. Where will the next one come from? New Scientist. https://www.newscientist.com/article/2246259-this-wont-be-the-last-pandemic-where-will-the-next-one-come-from (accessed October 24, 2021).
Kremer, M. 2021. Presentation at National Academy of Medicine international workshop on COVID-19 lessons to inform pandemic influenza response. https://nam.edu/event/international-workshop-on-covid-19-lessons-to-inform-pandemic-influenza-response-day1 (accessed October 24, 2021).
McAdams, D., K. K. McDade, O. Ogbuoji, M. Johnson, S. Dixit, and G. Yamey. 2020. Incentivising wealthy nations to participate in the COVID-19 vaccine global access facility (CooV): A game theory perspective. BMJ Global Health 5(11):e003627.
Morgan, O. W., X. Aguilera, A. Ammon, J. Amuasi, I. S. Fall, T. Frieden, D. Heymann, C. Ihekweazu, E.-k. Jeong, G. M. Leung, B. Mahon, J. Nkengasong, F. N. Qamar, A. Schuchat, L. H. Wieler, and S. F. Dowell. 2021. Disease surveillance for the COVID-19 era: Time for bold changes. The Lancet 397(10292):2317–2319.
Mustafa Diab, M., A. Zimmerman, S. Dixit, W. Mao, I. Bharali, A. Kristoffersen, G. Yamey, and O. Ogbuoji. 2021. The cost of procuring and delivering COVID-19 vaccines in low- and middle-income countries: A model of projected resource needs. http://dx.doi.org/10.2139/ssrn.3824690 (accessed October 25, 2021).
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016. Global health risk framework: Pandemic financing: Workshop summary. Washington, DC: The National Academies Press.
Navarro-Torné, A., F. Hanrahan, B. Kerstiëns, P. Aguar, and L. Matthiessen. 2019. Public health–driven research and innovation for next-generation influenza vaccines, European Union. Emerging Infectious Diseases 25(2):e180359. https://wwwnc.cdc.gov/eid/article/25/2/18-0359_article#suggestedcitation (accessed October 25, 2021).
Norton, A., L. Sigfrid, A. Aderoba, N. Nasir, P. G. Bannister, S. Collinson, J. Lee, G. Boily-Larouche, J. P. Golding, E. Depoortere, G. Carson, B. Kerstiëns, and Y. Yazdanpanah. 2020. Preparing for a pandemic: Highlighting themes for research funding and practice—perspectives from the Global Research Collaboration for Infectious Disease Preparedness (GloPID-R). BMC Medicine 18(1):273.
PPP (Pandemic Preparedness Partnership). 2021. 100 days mission to respond to future pandemic threats. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/992762/100_Days_Mission_to_respond_to_future_pandemic_threats__3_.pdf (accessed December 19, 2021).
Press Trust of India. 2018. India, EU to develop new influenza research programme named Horizon 2020; pay €15 million each to fund project. https://www.firstpost.com/india/india-eu-to-develop-new-influenza-research-programme-named-horizon-2020-pay-e15-million-each-to-fund-project-5063951.html (accessed October 25, 2021).
Radin, E., and C. Eleftheriades. 2021. Financing pandemic preparedness and response. Independent Panel for Pandemic Preparedness and Response, Background Paper 14. https://theindependentpanel.org/wp-content/uploads/2021/05/Background-Paper-14Financing-Pandemic-Preparedness-and-Response.pdf (accessed October 25, 2021).
Ramachandran, R., M. Dhodapkar, J. S. Ross, and J. L. Schwartz. 2021. Future of COVID-19 vaccine pricing: Lessons from influenza. BMJ 373:n1467.
The Rockefeller Foundation. 2021. One for all: An action plan for financing global vaccination and sustainable growth. New York: The Rockefeller Foundation.
Røttingen, J.-A. 2016. Alternative models for influenza vaccine R&D financing. Presentation by CEPI at the third WHO consultation on Global Action Plan on influenza vaccines (GAP III). https://www.who.int/influenza_vaccines_plan/GAPIII_Session4_Rottingen.pdf (accessed October 25, 2021).
Sabin-Aspen Vaccine Science and Policy Group. 2019. Accelerating the development of a universal influenza vaccine. https://www.influenzer.org/app/uploads/2020/01/sabin-aspen-report-digital.pdf (accessed October 25, 2021).
Sabin-Aspen Vaccine Science and Policy Group. 2021. Powering vaccine R&D: Opportunities for transformation. https://www.aspeninstitute.org/wp-content/uploads/2021/05/Sabin-Aspen-Report-2021.pdf (accessed October 25, 2021).
Sands, P., C. Mundaca-Shah, and V. J. Dzau. 2016a. The neglected dimension of global security—a framework for countering infectious-disease crises. New England Journal of Medicine 374(13):1281–1287.
Sands, P., A. El Turabi, P. A. Saynisch, and V. J. Dzau. 2016b. Assessment of economic vulnerability to infectious disease crises. The Lancet 388:2443–2488.
Schnirring, L. 2019. Trump signs executive order to improve flu vaccines. https://www.cidrap.umn.edu/news-perspective/2019/09/trump-signs-executive-order-improve-flu-vaccines (accessed October 25, 2021).
Snyder, C. M., K. Hoyt, D. Gouglas, T. Johnston, and J. Robinson. 2020. Designing pull funding for a COVID-19 vaccine. Health Affairs 39(9):1633–1642.
Sparrow, E., J. G. Wood, C. Chadwick, A. T. Newall, S. Torvaldsen, A. Moen, and G. Torelli. 2021. Global production capacity of seasonal and pandemic influenza vaccines in 2019. Vaccine 39(3):512–520.
Stutzman, H., A. Micah, and J. Dieleman. 2020. Funding pandemic preparedness: A global public good. https://www.thinkglobalhealth.org/article/funding-pandemic-preparedness-global-public-good (accessed October 25, 2021).
Turner, M. 2016. Vaccine procurement during an influenza pandemic and the role of advance purchase agreements: Lessons from 2009-H1N1. Global Public Health 11(3):322–335.
UC (University of California) Davis News and Media Relations. 2019. $9m to preempt zoonotic spillover threats, protect military and local communities. https://www.ucdavis.edu/news/9m-preempt-zoonotic-spillover-threats-protect-military-and-local-communities (accessed October 25, 2021).
Vu, J. T., B. K. Kaplan, S. Chaudhuri, M. K. Mansoura, and A. W. Lo. 2020. Financing vaccines for global health security. National Bureau of Economic Research, working paper 27212. https://www.nber.org/papers/w27212 (accessed October 25, 2021).
WHO (World Health Organization). 2019. Partnership Contribution (PC) Preparedness High-Level Implementation Plan II 2018–2023, revised version. Geneva, Switzerland: World Health Organization.
WHO. 2021a. WHO chief warns against “catastrophic moral failure” in COVID-19 vaccine access. UN News January 18, 2021. https://news.un.org/en/story/2021/01/1082362 (accessed July 22, 2021).
WHO. 2021b. Director-General’s opening remarks at the World Health Assembly—24 May 2021. Geneva, Switzerland: World Health Organization https://www.who.int/director-general/speeches/detail/director-general-s-opening-remarks-at-the-world-health-assembly---24-may-2021 (accessed July 22, 2021).
WHO. 2021c. Joint COVAX statement on supply forecast for 2021 and early 2022. Geneva, Switzerland: World Health Organization. https://www.who.int/news/item/08-09-2021-joint-covax-statement-on-supply-forecast-for-2021-and-early-2022 (accessed October 25, 2021).
World Bank. 2020. Epidemic preparedness and response. https://www.worldbank.org/en/results/2020/10/12/epidemic-preparedness-and-response (accessed October 25, 2021).
World Bank Group. 2019. Pandemic preparedness financing: Status update. https://www.worldbank.org/en/search?q=Pandemic+preparedness+financing%3A+Status+update (accessed October 25, 2021).
Yang, S. 2021. Rising-power competition: The COVID-19 vaccine diplomacy of China and India. Emerging Voices on the New Normal in Asia. National Bureau of Asian Research. https://www.nbr.org/publication/rising-power-competition-the-covid-19-vaccine-diplomacy-of-china-and-india (accessed October 25, 2021).








































