1
Introduction: SMART Vaccines and Smart Priorities
Health is wealth. The public health benefits of vaccines extend over multiple generations—an effect that could be considered as powerful as the value of the spread of education in our society. These health gains continue over generations to have a direct bearing on workforce productivity gains and the economic progress of nations. Moreover, scaling up of vaccination has been acknowledged as one of the most important mechanisms to help reduce the health achievement gaps between the developed and developing countries by 2035—a goal referred to as the “grand convergence” (Jamison et al., 2013).
Previous reports have noted that the impact of infectious diseases has shrunk from accounting for one-third of all deaths in 1990 to just one-quarter of all deaths in 2010 (Lozano et al., 2012). One recent modeling estimate found that “among children born during 1994–2013, vaccination will prevent an estimated 322 million illnesses, 21 million hospitalizations, and 732,000 deaths over the course of their lifetimes, at a net savings of $295 billion in direct costs and $1.38 trillion in total societal costs” (Whitney et al., 2014, p. 352).
While these numbers and those reported by others (Rousch et al., 2007; Hinman et al., 2011; van Panhuis et al., 2013) do not account for other important benefits of vaccines and thus must certainly underestimate the broader value conferred by vaccines on society (Bloom et al., 2005), they do serve to point out an important aspect in improving the health and economic status of nations: the need to invest in the development of new and improved vaccines to tackle a range of unmet, neglected, or emerging needs in infectious and non-infectious disease prevention (Nabel, 2013; Dye, 2014; Greenwood, 2014; Rappuoli et al., 2014).
Progress in science, engineering, and policy systems has dramatically transformed the landscape of vaccine development (De Gregorio and Rappuoli, 2014). Since the development of the first vaccine by Edward Jenner in the late 1700s, vaccine development has been pursued “empirically” by the isolation and inactivation of disease-causing microorganisms. But after nearly two centuries, starting in the 1980s, more “modern approaches” to vaccine development began to emerge to tackle diseases that could not be addressed with the empirical approach. Many of those approaches have the same conceptual basis as the traditional approaches, but they have more often capitalized on technologic advancements such as genome-level targeting or have capitalized on the structural understanding of the strains. An illustrated history is shown in Figure 1-1.
Vaccine development has become progressively more expensive. Decision makers and investors often focus on understanding the monetary and other returns on the investments—when they are making decisions about which vaccines to use as well as about which vaccines to develop. Yet at the same time, there remains considerable doubt as to which vaccines are most desirable to produce, and there are no commonly used methods for reaching consensus on priorities.
The typical valuation measures have centered purely on health metrics or else on a combination of health and economic metrics such as cost-effectiveness or cost versus benefit when prioritizing new vaccine development. These approaches, while functional, are quite narrow and limited (Bloom et al., 2005). Given this situation, not having a comprehensive measure often leads to vaccines being undervalued against other investment priorities, such as therapeutics or the construction of infrastructure.
Against this background, the path that led to the current work began with the National Vaccine Plan (HHS, 2011) developed by the U.S. Department of Health and Human Services’ National Vaccine Program Office (NVPO). One of the top priorities set forth in that plan was the development of new and improved vaccines.
At the request of NVPO, the Institute of Medicine (IOM) launched a Phase I effort in late 2010 to help create an empirical foundation for a vaccine prioritization model based on multi-attribute utility theory (IOM, 2012). Multi-attribute utility theory offers a strong axiomatic base for evaluating vaccines beyond a single criterion such as the standard infant mortality equivalents (or in modern terms, life-years saved) or cost-effectiveness—both of which were previously used by the IOM to produce
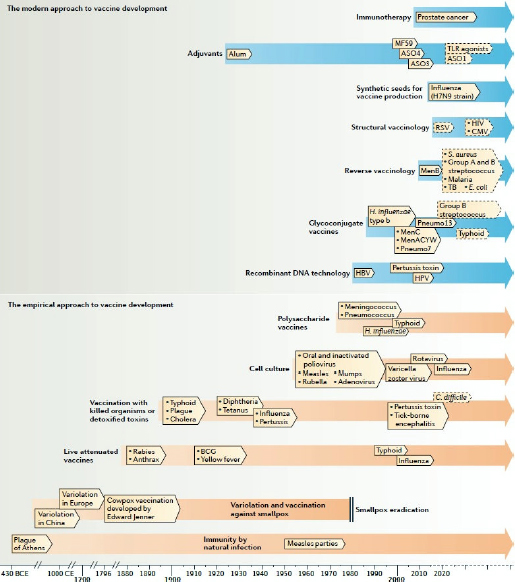
FIGURE 1-1
A timeline of the history of vaccines showing the technologies that have enabled their development. Vaccine research can be divided into two main periods, with the first being the empirical approach, which was based on isolating, inactivating, and injecting the microorganisms that cause disease. The second, modern approach began in the 1980s, when new technologies enabled advances in vaccine development that would not have been possible using the empirical approach.
NOTE: Closed boxes indicate licensed vaccines or vaccination practices that are already used. Boxes with a dashed border indicate vaccines that are still in development. BCG = Bacille Calmette–Guérin; C. difficile = Clostridium difficile; CMV = cytomegalovirus; E. coli = Escherichia coli; H. influenza = Haemophilus influenzae; HBV = hepatitis B virus; HPV= human papilloma virus; MenACYW = meningococcus serogroups A, C, Y, and W; Pneumo7 = 7valent pneumococcus vaccine; Pneumo13 = 13valent pneumococcus vaccine; RSV = respiratory syncytial virus; S. aureus = Staphylococcus aureus; TB = tuberculosis; TLR = Toll-like receptor.
SOURCE: De Gregorio and Rappuoli, 2014. Reprinted with permission from Macmillan Publishers Ltd.
rank-ordered priority lists (IOM, 1985, 1986, 2000). The Phase I report, Ranking Vaccines: A Prioritization Framework (IOM, 2012), that resulted from the IOM’s more recent work on vaccine prioritization introduced and validated a multi-attribute approach using 29 attributes, which was thought to offer a significant advance to the thought process of stakeholders. The Phase I committee tested the model1 using three hypothetical vaccine candidates for three diseases—influenza, tuberculosis, and group B streptococcus—for use in both the United States and in South Africa. The Phase I deliverable also included a blueprint of a software instantiation of the model, which was named Strategic Multi-Attribute Ranking Tool for Vaccines—or SMART Vaccines Beta.
A Phase II committee moved the development of this work forward by updating the model and the attributes in the model—28 of them plus another 7 user-defined entries (see Table S-1). In its report Ranking Vaccines: A Prioritization Software Tool (IOM, 2013; also see commentary in Phelps et al., 2014), the Phase II committee described the extensive testing it performed on the model using three additional vaccine candidates—for human papillomavirus, rotavirus, and pneumococcal infections—as it pursued the development of a functional prototype SMART Vaccines 1.0 in a Matlab environment that is executable in a Windows environment.
Project Process and Stakeholders’ Feedback
In fall 2013, following the release of the Phase II products, a nine-member committee was appointed by the Institute of Medicine and the National Academy of Engineering to carry out the Phase III project. Some of members had served previously on Phase I and Phase II committees, and some were new. Appendix D contains the biographical information of the members.
In addition to two committee meetings, the Phase III committee members met as subgroups in numerous teleconferences. Three technical consultants offered assistance to the committee on software enhancements and data synthesis. An additional consultant helped conduct usability studies with user groups in collaboration with the committee.
____________
1 The multi-attribute utility model embedded in SMART Vaccines consists of a computational submodel and a value submodel. Background information on these submodels, the overall modeling strategy, mathematical functions, and the associated assumptions (e.g., with costs and time horizon) are explained in Ranking Vaccines: A Prioritization Framework (IOM, 2012). Information about the model refinements and testing are included in Ranking Vaccines: A Prioritization Software Tool (IOM, 2013). This information is not repeated in this report.
BOX 1-1
Framing Questions for Stakeholders’ Feedback
Uses and Applications: Please describe your experience in using SMART Vaccines 1.0. What aspects of the software are beneficial? What functionalities or features can be enhanced or modified? Where does the software fit within your decision-making process?
Data Needs and Structure: What advice can you provide in terms of using available resources and databases toward creating a data framework for SMART Vaccines? What type of data estimation strategies and standardization tools would be useful for your decision making with the use of SMART Vaccines?
Host: What would be your expectations from the potential host of SMART Vaccines?
As part of their sustained outreach efforts, the committee members demonstrated SMART Vaccines 1.0 (the Phase II product) to a wide range of stakeholders at various meetings of major professional societies, federal advisory committees, and vaccine manufacturers and their consortiums. Additionally, the committee organized a public meeting to gather feedback from other stakeholder leaders for use in refining SMART Vaccines. The meeting’s speakers are listed in Appendix C, and the questions posed to them are given in Box 1-1.
Use Case Scenarios and Data Synthesis
To evaluate the utility of and support for SMART Vaccines in the stakeholder community (see Box S-1), the committee, in collaboration with the project sponsors—the U.S. Department of Health and Human Services’ National Vaccine Program Office and the Fogarty International Center of the National Institutes of Health—decided on three user groups to serve as early evaluators of SMART Vaccines.
The three user groups were the Public Health Agency of Canada, the New York State Department of Health, and the Serum Institute of India (see Box 1-2). Each user group had different interests in employing SMART
BOX 1-2
Profiles of User Groups
The Public Health Agency of Canada (PHAC) is the main agency responsible for public health in Canada. PHAC is one of seven departments and agencies that make up the Canadian government’s health portfolio, and it reports to parliament through the minister of health. PHAC works in close collaboration with all levels of government (provincial, territorial, and municipal) and also with nongovernment organizations, including civil society and business organizations, and with other countries and international organizations, such as the World Health Organization, to share knowledge, expertise, and experiences.
The New York State Department of Health (NYSDOH) bureau of immunization is responsible for the control and prevention of vaccine-preventable diseases. NYSDOH educates providers and the public about vaccines and vaccine-preventable diseases, conducts surveillance and outbreak control activities, and distributes vaccines to ensure their availability to vulnerable populations and populations with limited ability to pay for vaccines. NYSDOH bases routine immunization activities on the recommended immunization schedules, updated annually by the Advisory Committee for Immunization Practices. NYSDOH provides outreach to ensure optimal compliance with those vaccine recommendations.
The Serum Institute of India Limited manufactures life-saving immunobiologicals at affordable prices for India and other countries. It is the world’s largest producer of measles and diphtheria-tetanus-pertussis (DTP) group of vaccines and many of its products. In addition to its product portfolio, the Serum Institute of India also works toward bringing down the prices of newer vaccines, including the hepatitis-B vaccine, the rabies vaccine, and other combination vaccines besides DTP.
Vaccines to inform their respective vaccine development or policy planning efforts. Their backgrounds, test cases, and suggestions for enhancing SMART Vaccines are discussed in Chapter 3. A consultant to the committee from Microsoft Corporation led the usability studies with these three user groups and prepared an independent report containing suggestions for improving SMART Vaccines (see Appendix A).
A fourth use case scenario was carried out by the committee as a simulation to demonstrate a different application for SMART Vaccines: to show how the software might be used to reverse engineer vaccine product characteristics. In addition to these four formal use case scenarios, the committee also worked with the Mexican Ministry of Health to gain additional input for enhancing the software.
Each user group worked closely with the committee, which provided various data needed for the software evaluation. The interactions with the user groups informed the committee’s thinking on the data framework development described in Chapter 2.
This page intentionally left blank.








