2
Background on the PCOR Data Infrastructure and Office of the Secretary PCOR Trust Fund
This chapter provides an overview of the patient-centered outcomes research (PCOR) data infrastructure and the PCOR Trust Fund. A brief historical overview of the program’s origins and relevant legislation is followed by a discussion of the current state and elements of the data infrastructure, as well as the framework used by the Assistant Secretary for Planning and Evaluation (ASPE) to organize its activities in this area. The chapter also describes the data infrastructure projects and products that have been funded from the Office of the Secretary PCOR Trust Fund (OS-PCORTF) over the years, including prior evaluations of these efforts.
HISTORY OF THE PCOR TRUST FUND
Between 2003 and 2010, three laws were enacted that facilitated the expansion of research on the outcomes and effectiveness of treatments and interventions used in health care in a broad sense. In 2003, the Medicare Prescription Drug, Improvement, and Modernization Act provided authorization for the Agency for Healthcare Research and Quality (AHRQ) to support research comparing the outcomes and effectiveness of treatments and clinical approaches and to disseminate the findings from this research. In 2009, the American Recovery and Reinvestment Act provided additional funding to AHRQ, the National Institutes of Health, and the U.S. Department of Health and Human Services (HHS) for research that compares the effectiveness of medical options. In 2010, the Patient Protection and Affordable Care Act
provided further authorization for research that assists patients, clinicians, purchasers, and policy makers in making informed health decisions.
The PCOR Trust Fund within the Department of the Treasury was established by Congress in 2010. The goals of the PCOR Trust Fund are to fund PCOR, disseminate research findings, and build data capacity for PCOR. The bulk of the PCOR Trust Fund funding (around 80 percent of it), which is focused on research, is made available through the Patient-Centered Outcomes Research Institute (PCORI). PCORI is a nongovernmental organization established by Congress with the mandate to improve the quality and relevance of evidence available to help patients, caregivers, clinicians, employers, insurers, and policy makers make better-informed health decisions.1 Approximately 16 percent of the PCOR Trust Fund is allocated for disseminating research findings, incorporating findings into clinical practice, and training researchers in PCOR. The agency coordinating this work is AHRQ.
The remaining funding, which constitutes four percent of the PCOR Trust Fund, is allocated for building data capacity for PCOR. ASPE, under delegation of authority by the Secretary of HHS, coordinates across relevant federal health programs to build data capacity for PCOR, including administering the OS-PCORTF. Specifically, Section 937(f) of the Public Health Service Act instructed the Secretary of HHS to
… provide for the coordination of relevant Federal health programs to build data capacity for comparative clinical effectiveness research, including the development and use of clinical registries and health outcomes research networks, in order to develop and maintain a comprehensive, interoperable data network to collect, link, and analyze data on outcomes and effectiveness from multiple sources including electronic health records.2
In 2020, the PCOR Trust Fund was reauthorized through 2029, as part of H.R.1865 of the Further Consolidated Appropriations Act. The most recent statute specified intellectual and developmental disabilities, as well as maternal mortality, as research priorities. The statute also called for PCOR studies to include consideration of the full range of outcomes data, including potential burdens and economic impacts. Specifically, the law states the following:
Research shall be designed, as appropriate, to take into account and capture the full range of clinical and patient-centered outcomes relevant to, and that meet the needs of, patients, clinicians, purchasers, and policy-
___________________
1https://www.pcori.org/about/about-pcori.
2https://aspe.hhs.gov/collaborations-committees-advisory-groups/os-pcortf/about-os-pcortf.
makers in making informed health decisions. In addition to the relative health outcomes and clinical effectiveness, clinical and patient-centered outcomes shall include the potential burdens and economic impacts of the utilization of medical treatments, items, and services on different stakeholders and decision-makers respectively. These potential burdens and economic impacts include medical out-of-pocket costs, including health plan benefit and formulary design, non-medical costs to the patient and family, including caregiving, effects on future costs of care, workplace productivity and absenteeism, and healthcare utilization.3
This National Academies of Sciences, Engineering, and Medicine study is focused on issues relevant to continued work on the PCOR data infrastructure. In other words, it is focused on priorities for the use of the OS-PCORTF, or the four percent of the funding that is allocated for HHS to conduct work related to the data infrastructure for PCOR.
ENABLING DATA INFRASTRUCTURE FOR PCOR AND ASPE’S STRATEGIC FRAMEWORK
As the coordinator for the PCOR data infrastructure investment portfolio across HHS agencies, ASPE guides the strategic framework and vision for PCOR data infrastructure, sets funding priorities in collaboration with agencies and departmental leaders, and coordinates interagency workgroups. ASPE’s work is assisted by a Leadership Council for the OS-PCORTF, which includes representatives of several other HHS agencies, including the following:
- Administration for Children and Families;
- Administration for Community Living;
- Assistant Secretary for Preparedness and Response;
- Agency for Healthcare Research and Quality;
- Centers for Disease Control and Prevention;
- Centers for Medicare & Medicaid Services;
- Food and Drug Administration;
- Health Resources and Services Administration;
- National Institutes of Health;
- Office of the Chief Technology Officer;
- Office of the National Coordinator for Health Information Technology; and
- Substance Abuse and Mental Health Services Administration.
___________________
The agencies that are represented on the Leadership Council bring a variety of perspectives linked with their missions, which include (1) producing data, (2) conducting or funding research, or (3) making evidence-based decisions. The Leadership Council provides input on priorities for the portfolio, including projects to fund.
Using a strategic plan and agency leadership as guides, the OS-PCORTF approach to building data capacity has been characterized by incremental, modular investments. ASPE and its partners conduct an annual review of the progress that has been made and set priorities for the following year, based on statutory priorities and the need to build toward desired functionalities. The current framework for building data capacity is organized according to five data infrastructure goals. These five goals enable functionalities to collect, link, and analyze data for patient-centered research.
Figure 2-1 is a visual representation of the framework that has guided the PCOR data infrastructure portfolio in recent years (as discussed, the reauthorization presents an opportunity for a new strategic direction). The diagram was developed in an attempt to operationalize the mandate for the OS-PCORTF and guide decision making. Definitions for the key terminology used for the framework are provided on the right hand side of the figure. The bottom row shows potential sources of data for PCOR studies, feeding into the PCOR data infrastructure. Data collected as part of clinical care includes data collected for health care delivery and for billing purposes. Examples of primary data collected as part of research studies include data from clinical trials and national health surveys. Other examples of data sources include Medicare or Medicaid claims data, quality or outcomes data collected by health care providers for the purposes of improving health care value, Food and Drug Administration data on the safety of medications and medical devices, and Centers for Disease Control and Prevention data on births and deaths provided by state public health authorities.
The framework describes the relationship between the data sources and major functionalities (middle row), including the “pillars” or core research functions and focus areas that support the research. The key functionalities are described in further detail in Box 2-1. The major building blocks are the services, standards, policies, and governance that enable the use of the data for research, described in Box 2-2. The top row shows the key data users and contributors of data.
The work of HHS agencies toward building data capacity for PCOR is guided by strategic planning that has continuously evolved over the years. The work evolves based on changes in legislation, new priorities in public health and medical care, new HHS policies, strategic opportunities, and the evolving data infrastructure needs of stakeholders.
To gather input on challenges and priorities for the PCOR data infrastructure, in 2020 ASPE commissioned a study centered on an “online
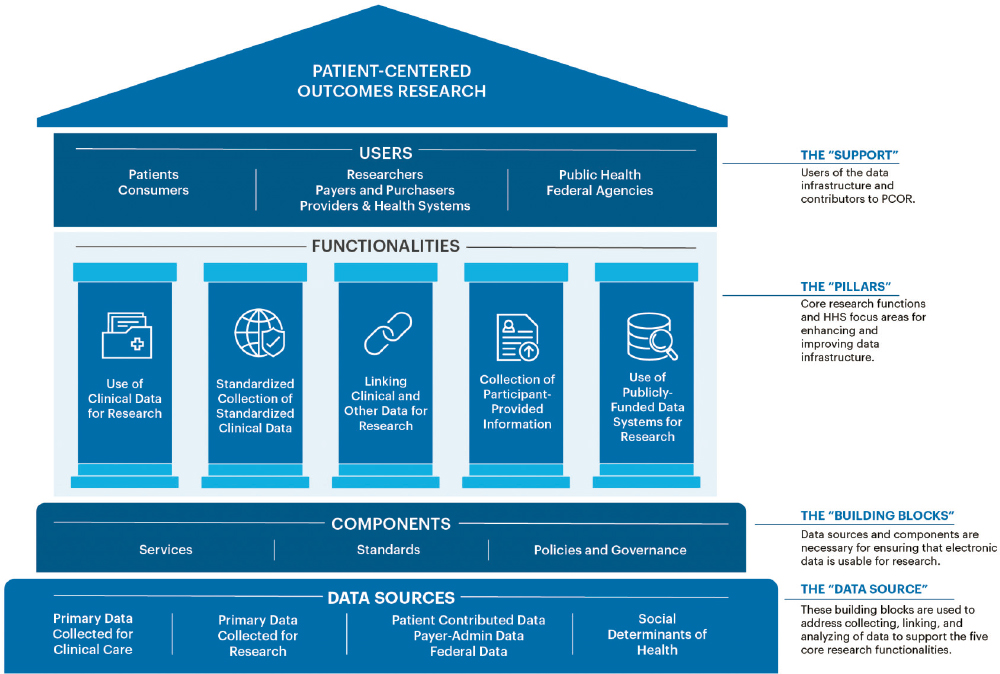
SOURCE: Workshop Presentation by ASPE, May 3, 2021.
prioritization activity.”4 The findings from that study were among the materials reviewed by the committee as background for its work. The prioritization activity involved stakeholders with a variety of backgrounds, such as health care providers, researchers, and health policy experts, who were asked to generate lists of challenges and needed improvements for the five functionalities included in ASPE’s framework for the PCOR data infrastructure. Participants were then asked to vote on the challenges and
___________________
4https://aspe.hhs.gov/sites/default/files/migrated_legacy_files//197426/PCOR-Data-Infrastructure.pdf.
improvements in order to generate prioritized lists. The activity resulted in 87 items described as challenges and 76 items described as needed improvements for the data infrastructure. Box 2-3 summarizes the needs that emerged as priorities from this activity.
OVERVIEW OF THE DATA INFRASTRUCTURE PROJECTS AND PRODUCTS
This section describes the data infrastructure project and products funded through the OS-PCORTF, in order to provide some additional context about the ongoing work. As of 2021, the OS-PCORTF had funded 61 data infrastructure projects, which included 33 active and 28 completed projects. Appendix E shows the full list of projects, with additional details about each. The 53 projects funded during the first 10 years (from 2010 to 2019) translated to 76 agency awards, totaling approximately $131 million. Many of the projects are conducted as collaborations that involve two or more HHS agencies or with the involvement of additional agencies in an advisory capacity.
As discussed earlier, each project addresses one or more of the five functionalities in the data infrastructure framework. Table 2-1 shows a count of the projects by the infrastructure functionalities they address. Table 2-2 summarizes the number of products that resulted from OS-PCORTF data infrastructure projects by product category, and Table 2-3 provides examples of the types of products that are produced.
“Use cases” based on the priorities of the HHS Secretary play an important role in the way priorities are set for the OS-PCORTF. Table 2-4 shows examples of HHS priorities and PCOR projects. ASPE evaluates these projects in the context of their fit with the current strategic plan and the projects’ potential contributions to building a data infrastructure that will support PCOR. Figure 2-2 shows how some of the thematic areas that have been funded over the years map onto the five functionalities in the current conceptual framework.
| Data Infrastructure Functionality Addressed | Number of Projects |
|---|---|
| Standardized collection of standardized clinical data | 27 |
| Collection of participant-provided information | 12 |
| Linking of clinical and other data for research | 38 |
| Use of clinical data for research | 35 |
| Use of enhanced publicly funded data systems for research | 19 |
NOTE: Some projects address multiple functionalities.
SOURCE: https://aspe.hhs.gov/collaborations-committees-advisory-groups/os-pcortf/explore-portfolio.
TABLE 2-2 Products Produced by Office of the Secretary PCOR Trust Fund Projects
| Product Category | Number of Products |
|---|---|
| Datasets, databases, and linked data | 5 |
| Data elements and information models | 3 |
| Health information technology standards | 6 |
| Tools and guides | 8 |
| Software and analytic services | 8 |
| Data governance products | 3 |
| Publications | 8 |
| Project reports and briefs | 20 |
| Vignettes, project spotlights, and portfolio reports | 15 |
| Annual reports | 6 |
| Evaluation reports | 2 |
| Total | 84 |
SOURCE: https://aspe.hhs.gov/collaborations-committees-advisory-groups/os-pcortf/os-pcortf-product-library.
TABLE 2-3 Examples of Products Produced by Office of the Secretary PCOR Trust Fund Projects
| TECHNICAL PRODUCTS | |||
|---|---|---|---|
Dataset
|
Data Element & Information Model
|
HIT Standards
|
Tools and Guides
|
Software
|
Software and Analytic Services
|
Data Governance
|
Stakeholder Engagement
|
| COMMUNICATION AND DISSEMINATION PRODUCTS | ADMINISTRATIVE DELIVERABLES | ||
Publications
Web & Social Media
Webinar
|
Presentations
Project Reports and Briefs
Portfolio-related reports
|
Project implementation plans
|
Contract/grant deliverables
|
NOTE: HIT = health information technology; PII = personally identifiable information; RFI = request for information; RFP = request for proposals; TEP = technical expert panel.
SOURCE: Presentation by ASPE, January 29, 2021, public meeting.
| HHS Secretarial Priority | Example Projects | Lead Agency |
|---|---|---|
| Opioids and mental health | An Addiction Medicine Network to Address the United States’ Opioid Crisis | NIH |
| Value-based care | Validating and Expanding Claims-based Algorithms of Frailty and Functional Disability for Value-based Care and Payment | ASPE |
| Maternal mortality | Surveillance Network: Maternal, Infant, and Child Health Outcomes Associated with Treatment of Opioid Use Disorder during Pregnancy | CDC |
| Social determinants of health | Creating a National Small-Area Social Determinants of Health Data Platform | AHRQ |
| Emergency preparedness | Assessing and Predicting Medical Needs in a Disaster | ASPR |
| Patient empowerment and interoperability | Technologies for Donating Medicare Beneficiary Claims Data to Research Studies | CMS/NIH |
| Data and innovation | Training Data for Machine Learning to Enhance Patient-Centered Outcomes Research Data Infrastructure | ONC |
NOTE: NIH = National Institutes of Health; CDC = Centers for Disease Control and Prevention; AHRQ = Agency for Healthcare Research and Quality; ASPR = Office of the Assistant Secretary for Preparedness and Response; CMS = Centers for Medicare & Medicaid Services; ONS = Office of the National Coordinator for Health Information Technology.
SOURCE: Presentation by ASPE, January 29, 2021, public meeting.
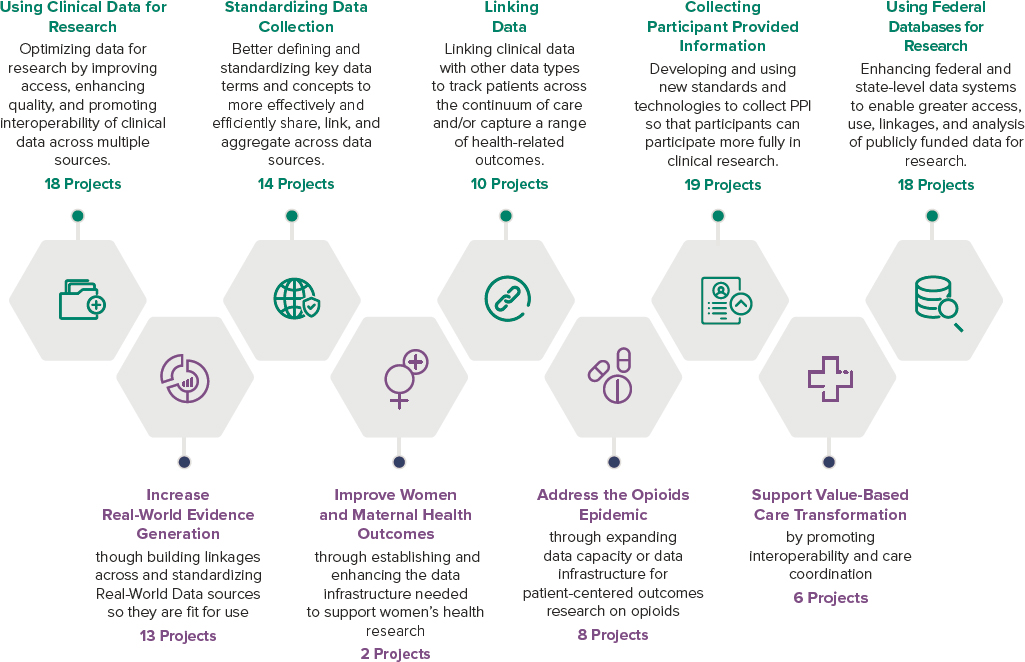
SOURCE: Presentation by ASPE, January 29, 2021, public meeting.
2020 Annual Report
The most recent (2020) annual report issued by ASPE for the PCOR data infrastructure work provides a synopsis of the 31 projects that were active in Fiscal Year 2020.5 As discussed, HHS priorities play an important role in the priorities for the PCOR data infrastructure projects. As such, along with addressing stakeholder priorities for the data infrastructure, the annual report places special emphasis on work that supports women’s health and improving maternal health outcomes, as well as COVID-19 pandemic response. Box 2-4 lists the active projects in these key areas. More detailed information about the projects is available on ASPE’s website.6
The annual report also highlighted three completed projects that enhanced functionalities likely to be useful to researchers:
- Adding Cause-Specific Mortality to National Center for Health Statistics’ National Hospital Care Survey by Linking to the National Death Index and CMS Master Beneficiary Summary File: These linkages allow researchers to conduct more robust studies of cause-specific mortality.
- Assessing and Predicting Medical Needs in a Disaster: Increased access to local, state, and federal datasets can improve data availability in a disaster, help analyze and improve response strategies, identify needs and trends for long-term recovery, and track the long-term health outcomes and consequences of a disaster.
- Emergency Medicine Opioid Data Infrastructure: Key Venue to Address Opioid Morbidity and Mortality (Project CODE PRO – Capturing Opioid Use Disorder Electronically and Patient Reported Outcomes): Use of standardized common data elements and patient-reported outcome measures captured in emergency department electronic health records can improve the opioid-use disorder data available in clinical registries for PCOR.
___________________
5https://aspe.hhs.gov/sites/default/files/private/pdf/259016/2020-os-pcortf-portfolio-report.pdf.
6https://aspe.hhs.gov/index.php/collaborations-committees-advisory-groups/os-pcortf/explore-portfolio.
2016–2019 Portfolio Review
ASPE periodically commissions in-depth reviews of its portfolio of data infrastructure projects. The most recent review focused on 43 projects that were funded between 2016 and 2019 in the form of individual agency awards (the review did not include multiagency awards).7 It looked at the extent to which the portfolio of projects advanced the strategic framework, and concluded that approximately three out of four awards included a focus on optimizing the use of clinical data for research. About half of the awards fell into one or more of these categories: enhancing the use of federal datasets for research, standardizing data collections, and (or) developing tools to collect participant-provided information. One out of four projects focused on linking data.
The evaluation also assessed the extent to which the portfolio addressed gaps that had been identified by the previous evaluation, conducted in 2017.8 The 2019 evaluation concluded that the gap most frequently addressed by the awards was to disseminate research findings, and it noted progress in the areas of implementing standards and improving data quality. Concerning whether the portfolio changed in response to changes in the health policy landscape, the recent evaluation noted that the portfolio expanded to include projects on enhancing the interoperability of health information, improving patient access to health information, and supporting advanced data science methods.
___________________
7https://aspe.hhs.gov/sites/default/files/migrated_legacy_files//194781/OS-PCORTFImpactReport508.pdf.
8https://aspe.hhs.gov/sites/default/files/migrated_legacy_files//180436/ASPEPCORTFEvaluation.pdf.
The 2019 assessment also included input from an expert panel on the future direction of the data infrastructure portfolio in several areas. In the area of refinements to the strategic framework, the panel identified the following opportunities for additional progress:9
- Integrating external factors such as financial and policy drivers that influence provider documentation and the social determinants that impact health outcomes;
- Identifying and incorporating cross-cutting barriers;
- Highlighting the role of data provenance in the usability of data for research; and
- Forming working groups that can provide input on each of the five functionalities.
The 2019 expert panel further identified the following as priorities for the data infrastructure:
- Addressing nontechnical challenges to the use and sharing of data, including challenges associated with data governance, data privacy, and data security;
- Providing targeted support to products that may be of broad interest and ready for adoption; and
- Engaging end users during the planning phases of project awards.
Finally, the 2019 expert panel identified the need for better metrics to assess the portfolio’s impact, including the following:
- Award-specific metrics to assess whether each individual project achieved its objectives;
- Metrics that track how products from projects are being used by other projects within the portfolio;
- A more prescriptive strategic roadmap and metrics that assess progress along the roadmap; and
- Use of website analytics and dissemination and translation metrics that track the number and type of dissemination products, and the ways in which other research initiatives leverage award outputs.
___________________
9https://aspe.hhs.gov/sites/default/files/migrated_legacy_files//194781/OS-PCORTFImpactReport508.pdf.
This page intentionally left blank.


















