Proceedings of a Workshop
| IN BRIEF | |
|
April 2022 |
Lessons Learned from the COVID-19 Pandemic to Improve Diagnosis
Proceedings of a Workshop—in Brief
On January 14, 2022, the Board on Health Care Services of the National Academies of Sciences, Engineering, and Medicine hosted a virtual workshop focused on examining changes to diagnostic paradigms in response to the COVID-19 pandemic and considering the lessons learned and opportunities for improving diagnosis within the U.S. health care system.1 The workshop highlighted the pandemic’s impact on diagnostic pathways, the exacerbation of inequities, diagnostic responsibilities for public health, and the novel diagnostic strategies and tools that have been developed since COVID-19 emerged. This workshop was the fourth in a series on diagnostic excellence funded by the Gordon and Betty Moore Foundation.2 This Proceedings of a Workshop—in Brief highlights the presentations and discussions that occurred at the workshop.3
BACKGROUND
Daniel Yang of the Gordon and Betty Moore Foundation described the foundation’s framework of diagnostic excellence, which incorporates the six domains of health care quality from the Institute of Medicine report Crossing the Quality Chasm: A New Health System for the 21st Century (IOM, 2001). “We believe that diagnosis must be safe, timely, effective, efficient, patient-centered, and equitable across populations,” he said. He added that while all six domains are important, they are frequently in tension with one another and often require difficult trade-offs to achieve diagnostic excellence. For instance, while home-based tests may improve patient access and reduce the cost of a COVID-19 diagnosis, they are also less accurate compared to the gold standard polymerase chain reaction (PCR) tests, he said. Yang noted that “the COVID-19 pandemic has underlined the critical importance of timely, accurate, and equitable diagnosis,” and he urged clinicians to reconsider fundamental features of diagnosis, such as location and timing.
CHANGING DIAGNOSTIC PARADIGMS DUE TO THE COVID-19 PANDEMIC
Eric Rubin, editor-in-chief of the New England Journal of Medicine (NEJM), commented that infectious disease outbreaks present communications challenges as well as technical challenges. He highlighted the role of medical journals in sharing accurate information and added that there is a “tremendous tension between timeliness and quality” of information with coverage of COVID-19. The role of medical journals is to provide important guidance for clinical practice, while the main-
__________________
1 The workshop agenda and presentations are available at https://www.nationalacademies.org/event/01-14-2022/lessons-learned-from-the-covid-19-pandemic-to-improve-diagnosis (accessed February 24, 2022).
2 Past workshops in this series have explored improving diagnosis for sepsis, acute cardiovascular events, and cancer. More information about the workshop series is available at https://www.nationalacademies.org/our-work/advancing-diagnostic-excellence-a-workshop-series (accessed February 24, 2022).
3 This Proceedings of a Workshop—in Brief is not intended to provide a comprehensive summary of information shared during the workshop. The information summarized here reflects the knowledge and opinions of individual workshop participants and should not be seen as a consensus of the workshop participants, the planning committee, or the National Academies of Sciences, Engineering, and Medicine.
![]()
stream media has a different role, he said. He explained that the pace of diagnoses indicates if there is an outbreak, while highlighting various outbreaks around the world in recent decades and the sometimes lengthy process of detecting them. But persistent diagnostic challenges remain, he said, including diagnostic availability, particularly in countries with poor infrastructure; diagnostic performance characteristics (i.e., sensitivity and specificity); equitable distribution of tests; and the implications of incorporating home testing into the diagnostic process. He added that NEJM is interested in publishing articles on diagnostics that have the ability to change the paradigms of diagnosis, and it has instituted a new policy that requires transparency about how well studies represent the population that is primarily affected by the disease.
IMPACT OF THE COVID-19 PANDEMIC ON DIAGNOSTIC PATHWAYS, PERCEPTIONS OF DIAGNOSIS, AND SETTINGS OF CARE
Michael Osterholm, professor at the University of Minnesota, described the diagnostic challenges and opportunities of rapidly characterizing a new disease. Because of his previous involvement in other coronavirus outbreaks—severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS)—he thought the emergence of SARS-CoV-2 would not result in widespread transmission, as the vast majority of previous coronavirus transmissions occurred well into the clinical illness. It soon became clear that the asymptomatic transmission of this new virus was spreading far more quickly, similar to an influenza, said Osterholm, and countries began ramping up test capacity. However, for many low-resource settings without access to PCR machines, specialized equipment, or trained technicians, this was not possible. Additionally, the global manufacturing and supply chains could not ramp up fast enough to meet demand. Specifically highlighting U.S. challenges, Osterholm noted the decision by the Centers for Disease Control and Prevention (CDC) to develop its own PCR assay instead of using the one created by the World Health Organization (WHO). Unfortunately, this process took additional time, and shortly after the test kits were sent to the state and local level, the CDC announced that the tests had too many false positives and should no longer be used (Lee et al., 2021). This contributed to a testing backlog, with tests only being processed in the CDC lab in Atlanta, Georgia, which limited test eligibility to only those who had returned from China or to those who had been in contact with an infected individual. In the first 2 months of the pandemic, just 500 tests had been processed in the United States, he said. Even once this capacity increased, with Emergency Use Authorizations for additional tests, there were still numerous shortages reported, with 86 percent of responding labs reporting shortages of needed items even in September 2020 (ASM, 2021). Determining “how diagnostic testing is used in our communities, how it’s available, how is it in fact available throughout the whole world, is a much greater challenge” than rapidly characterizing a new disease, Osterholm concluded.
Nicole Franks, chief quality officer at Emory University Hospital Midtown, discussed the health systems perspective on changes to the diagnostic process. She highlighted the concept of triage, describing how patients accessed health care. Prior to the COVID-19 pandemic, the goal of triaging was to “get the patient to the right level of care where the highest quality of care could be delivered at the lowest cost in a timely fashion,” Franks said. But once the COVID-19 pandemic began, mindsets shifted to mass casualty triage principles, she said. While Emory initially used a strategy to enable patients with COVID-19 to access its health care system that was similar to the process used for its Ebola response in 2014–2015 (e.g., isolation of the health care team and optimization of personal protective equipment [PPE]), that quickly became too cumbersome owing to the huge volume of patients and lack of PPE.
Emory also pivoted to other ways of caring for patients without COVID-19, such as using telehealth. Franks outlined several challenges that health systems experienced in the diagnostic process. For example, once tested, would patients be sent to a cohorted area while awaiting results?4 Would they be discharged in a different way? The necessity to base diagnosis on symptoms alone without a test result also led to high variability in how and where care was delivered, and facilities varied in their instructions. Finally, she noted the challenges of changing information for the health care workforce, including staff having to relearn guidance week to week, and staff seeing unprecedented levels of illness and death day after day. She called for greater attention to support workforce well-being, as many continue to retire or change careers because of burnout.
Rainu Kaushal, professor at Weill Cornell Medicine, described initiatives using data science to understand the long-term effects of COVID-19, especially postacute sequelae of SARS-CoV-2 infection (PASC).5 Data on PASC are rapidly evolving, and there are numerous challenges: the diagnosis, expression, frequency, incidence, and prevalence seem to be very dependent on variants; it is a cluster of symptoms and conditions; and there is limited “research-ready data.” A new ICD-10 code was established for PASC in October 2021,6 but it is very generic, she said, and many diagnostic
__________________
4 Cohorting as defined by the CDC is the infection prevention and control practice “of grouping patients infected or colonized with the same infectious agent together to confine their care to one area and prevent contact with susceptible patients” (CDC, 2015).
5 Postacute sequelae of COVID-19 (PASC) is also known as “long COVID” and is a condition marked by the continuation of COVID-19 symptoms—or the emergence of new ones—after recovery from acute (or the initial phase of illness of) COVID-19. See https://www.yalemedicine.org/conditions/long-covid-post-acute-sequelae-of-sars-cov-2-infection-pasc (accessed March 4, 2022).
6 The ICD-10 (International Classification of Diseases, Tenth Revision) is a medical coding system used by health care providers to
questions remain. To address these questions, the National Institutes of Health (NIH) funded the RECOVER initiative,7 which is collaborating with 41 sites through the Patient-Centered Outcomes Research Network (PCORnet). These sites track and analyze large-scale health data to better understand, prevent, and treat PASC. As of November 2021, PCORnet had data on more than one million patients who tested positive for COVID-19.
As a specific example within this initiative, she said these 41 health systems have standardized their electronic health record data and real-world data (e.g., patient-reported outcomes and social determinants of health). Key data elements are extracted, transformed, curated, and then provided to the sites following quality assurance checks. With this centralized data repository, researchers and clinicians can create visualizations on incidence, clusters of conditions in rank order, and different time periods where prevalence might change, as well as addressing other queries. After incorporating a literature review and clinician input, Kaushal said it had developed a screening algorithm for the PASC phenotype consisting of 6,000 diagnostic clusters resulting in 10 key areas to focus on symptom presentation (e.g., neurological issues, mental health issues). Some example findings include that 57 percent of post-COVID adults screened had a new symptom or condition within PASC, and this increased to 73 percent for those who were hospitalized with COVID-19. There is much more work to be done to accurately and efficiently improve diagnosis for this condition, said Kaushal.
In a panel discussion, speakers commented on the challenges with testing, including delayed diagnosis, disruptions in the diagnostic process, and the impact on the operations of an organization, as well as supply chain challenges. Osterholm highlighted the investments needed to create more surge capacity in diagnostic testing for the next emergency. Shortages in supplies and workers—in countries around the world, not just the United States—demonstrate that “we are not prepared” for the next disaster, he said. Franks discussed the creation of the “presumed positive/may have had a negative test” designation in the initial days of the pandemic when it was challenging to make timely and accurate diagnoses, adding that while the presumed positive diagnosis helped with decision making, the uncertainty regarding accuracy caused disruptions and could have been improved by increased care coordination and communication. Franks and Osterholm also discussed the importance of public health measures, with Franks pointing out that the biggest missed opportunity was not using masking and appropriate distancing to mitigate transmission. Osterholm stated that screening is ineffective unless it is ongoing and frequent enough to detect a sufficient number of individuals to help reduce transmission. Finally, Kaushal highlighted the dramatic impact that patient input had during the first surge in New York City in 2020, saying that capturing the patient voice enabled researchers to put a finer lens on the role of social determinants of health and think about how to address those specifically.
EXACERBATION OF HEALTH INEQUITIES DURING THE COVID-19 PANDEMIC
Utibe Essien, assistant professor at University of Pittsburgh, discussed structural racism and the COVID-19 pandemic with three major foci: data related to COVID-19 inequities, drivers of those inequities, and strategies to address the inequities within the health care system. When data on U.S. COVID-19 rates began to emerge, racial and ethnic minorities showed higher rates of infection, mortality, and hospitalization, said Essien, adding that patients should have access to accurate testing and diagnosis in order to be included in these data. He explained the importance of considering risk factors, access to care, and missed care in examining the drivers of disparities (see Figure 1). For example, he said that the higher prevalence of chronic conditions among minority populations was a likely driver of severe COVID-19 infection. Another driver includes the overrepresentation of Black workers in essential industries, who were exposed to a higher risk of infection because they were unable to work from home and maintain safe distancing (McCormack et al., 2020).
Moving beyond the pandemic, Essien suggested focusing on structural factors, such as addressing pollution in neighborhoods, access to education and health care, and economic stability. He provided several strategies to address these factors, including enhancing the collection of race and ethnicity data in health care; expanding access to health care; providing equitable testing, contact tracing, and treatment; and elevating community and social service investment.
Chau Trinh-Shevrin, professor at NYU Langone Health, further emphasized the impact of the pandemic on health inequities with a zip code map of COVID-19 cases that clearly showed the stark disparities during the first wave of the pandemic in New York City in April 2020, noting
The neighborhoods hardest hit were socially disadvantaged and predominantly composed of lower-income racial and ethnic minority populations…with high population density and household size, limited English proficiency, and other social determinants of health disparities.
__________________
“catalog health conditions by categories of similar diseases under which more specific conditions are listed, thus mapping nuanced diseases to broader morbidities” (AAPC, 2021).
7 For more information, see: https://recovercovid.org (accessed March 3, 2022).
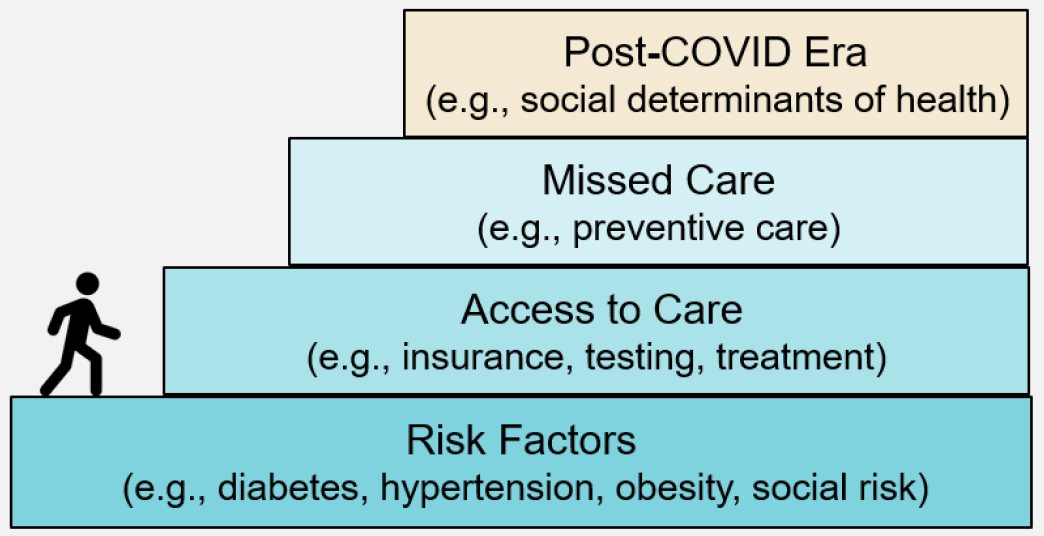
SOURCE: Utibe Essien presentation, January 14, 2022.
However, at the time, a large percentage of racial and ethnic data by zip code and by cases, hospitalizations, and deaths, was missing. Emphasizing the importance of data quality in reducing inequities, she said much of this information is still missing (CDC, 2022a).
She added that data aggregation can also result in misclassification. For example, available data within New York City suggested Asian Americans were least affected by the virus, but when looking more closely across tens of thousands of patients in the city, she explained that South Asians, who are often classified as “Other,” actually had high rates of positivity and hospitalization, and those of Chinese descent had the highest mortality rates—but these insights were missed when the data was aggregated (Marcello et al., 2021). Trinh-Shevrin underscored the need to share data disaggregation by ethnic group and to also use innovative approaches to reduce misclassification errors in order to really understand the nature and prevalence of COVID-19 disparities. In addition, she highlighted the importance of community engagement to reduce COVID-19 disparities through increased awareness and education; to increase trust in guidelines to control COVID-19 infection and transmission; for promotion of vaccine acceptance and uptake; and for inclusion of disproportionately affected racial and ethnic minority populations in clinical trials.
Adriann Begay, senior officer at HEAL,8 described efforts to address health inequities in Indigenous communities during the COVID-19 pandemic. To understand the root of the inequities that this community faces, Begay briefly touched upon the complex history of federal health policy including the legal basis for Indigenous communities’ right to health care based on multiple treaties, executive orders, court decisions, and numerous other laws and acts (American Indian Policy Review Commission, 1977; ANA, 2022; BIA, 2022).9 Despite the legal obligation of the United States to provide these services, Indigenous populations face some of the worst health disparities when compared to other racial and ethnic groups, and the pandemic has exacerbated these disparities.
Multigenerational homes, lack of running water, difficulty accessing health care services, the lack of resources, and mistrust in the health system contributed to the high rates of infections, hospitalizations, and deaths in this community, she said. There were also trust issues with recruitment for vaccine clinical trials, in part because of historical transgressions against their population, she recounted, such as the forced sterilization of Indigenous women in the 1970s. But despite these obstacles, through partnering with the community and increasing cultural competence of health care providers, the Navajo Nation led the country in highest vaccination rates once they became available, and corresponding hospitalizations and deaths declined (Powder, 2021; Villarreal, 2021). Begay emphasized the importance of culture, land, language, ceremonies, and song in Navajo culture, and the importance of respecting these
__________________
8 HEAL (Health, Equity, Action, and Leadership) “trains and transforms frontline health professionals through building a community dedicated to serving the under-served as their lifelong choice.” See https://healinitiative.org/how-were-different/mission-and-vision (accessed March 22, 2022).
9United States v. Mitchell, 463 U.S. 206, No. 81-1748 (1983).
elements when working with these populations. Begay concluded, “Hearing historical truths are difficult, but it is only through truth that we can move forward, and understanding and respecting these truths are crucial in elevating the health status of Indigenous people.”
Monica Webb Hooper, deputy director at the National Institute on Minority Health and Health Disparities, discussed strategies to mitigate disparities in COVID-19 diagnostics, focusing on the NIH Rapid Acceleration of Diagnostics-Underserved Populations Initiative (RADx-UP). She highlighted the interrelated factors associated with health care, socioeconomics, and social determinants of health, with the overarching context of structural and systemic racism and discrimination, which contributed to COVID-19 cases and outcomes for many racial and ethnic minority populations. She added that disparities in COVID-19 transmission, infection, and death are caused by “social inequities that have led to more preexisting conditions like heart disease, obesity, diabetes and other comorbidities, as well as social inequities that increase the likelihood of exposures.”
Webb Hooper explained that RADx-UP is focused on taking technologies that are already FDA authorized and applying community-engaged research methods to increase access and uptake of COVID-19 diagnostics in underserved and vulnerable populations. With an overarching focus on interventions to reduce COVID-19 health disparities, she shared the three main activities of RADx-UP: (1) increasing testing access and uptake of COVID-19 diagnostic testing, while understanding and addressing disparities associated with testing; (2) assessing ethical, structural, social, behavioral, environmental, and contextual factors around testing, and investigating multilevel testing barriers, cultural beliefs, expectations, mistrust, and communication preferences; and (3) implementing specific, targeted testing approaches in educational settings serving underserved and vulnerable children and their families, while identifying scalable and sustainable testing implementation strategies to maintain in-person learning.
Providing an example of its work thus far, she described a project in Baltimore that implemented free community testing through a partnership with religious leaders. RADx-UP found a positivity rate of 31.5 percent for Latino people in the community compared with just 3.4 percent of White people (Bigelow et al., 2021). In discussing lessons learned from the initiative, Webb Hooper discussed the importance of community engagement and trust by applying community-engaged research methods, creating partnerships with community health clinics to connect with underserved populations, and framing return-to-school testing as a school safety measure to increase participation.
During the discussion, speakers offered various tools and strategies to mitigate health disparities and improve diagnosis through community engagement. Trinh-Shevrin emphasized that the key to building trust is persistent community engagement with “repeated encounters using trusted messengers.” Essien suggested adapting technologies for different communities, noting an example of pulse oximetry monitors that report different oxygen level results because of varying skin colors despite other identical demographic variables. He called for more innovation to improve clinical devices, as well as access to these technologies, and also pointed to the lack of accuracy and precision in collecting data to understand disparities. Essien emphasized the importance of collecting data on social factors that drive health outcomes “to help ensure that communities are receiving the care that they need.” Webb Hooper added that using standardized measures to collect data is critical. Essien and Trinh-Shevrin also called for new paradigms in clinical trial recruitment to ensure equitable representation and to overcome financial barriers in enrolling. Trinh-Shevrin also noted that while telemedicine may be the future of health care, issues of inequitable access will need to be addressed so current disparities are not exacerbated.
DIAGNOSIS AND PUBLIC HEALTH RESPONSIBILITY
Raina Merchant, professor and director of the Center for Digital Health at the University of Pennsylvania, described the role of social media and digital data in diagnosis, using the term infodemic to describe the volume and reach of data during the pandemic.10 She works across different platforms to mine data and use insights to assess digital engagement and inform digital equity and access. She highlighted examples of ways digital technologies, including smart-phones, mobile applications, wearable sensors, and social media, have been used to inform how the public thinks about diagnoses in public health emergencies:
- Public sentiment: A Twitter map pulled millions of tweets each day to track mental health and expressions of loneliness and depression (Guntuku et al., 2020), and showed that people began posting about symptoms they attributed to COVID-19 before they were officially diagnosed.
__________________
10 The World Health Organization defines infodemic as “too much information including false or misleading information in digital and physical environments during a disease outbreak. It causes confusion and risk-taking behaviours that can harm health. It also leads to mistrust in health authorities and undermines the public health response” (WHO, 2022).
- Tracking disease: Using metadata from cellphones, Instagram locations (The Economist, 2020), and smart thermometers to track movement of individuals, researchers have demonstrated the potential roles of such data for disease surveillance and information spread, and to help understand risk.
- Tracking misinformation: Misinformation is a public health crisis; for example, researchers found that one-third of information about COVID-19 on the app TikTok was incorrect, misleading, or incomplete (Southwick et al., 2021). Merchant noted a need to evaluate the effectiveness of various approaches to address misinformation.
- Tracking risk: Digital apps are emerging to assist in contact tracing.11 Contacts are traced using GPS colocalizations with others on the app, supplemented by scanning QR-codes on high-traffic public areas.
Merchant concluded by discussing the need for using targeted approaches to public health messaging that considers rapidly changing information, is responsive to the needs of the public, and uses an equity lens.
J. Marc Overhage, chief health information officer at Anthem, discussed the role of public health infrastructure in managing diagnosis and care at the population level. For many years, people have been working to share data among participants in the health care ecosystem, and while there has been progress, the ability to aggregate data is still limited. Nationally, he said, we struggled to bring together data from health care situations throughout the system during the pandemic. He called for a knowledge-based reporting process—an agile way to identify a concern and quickly deploy relevant information to thousands of systems across the country. He outlined four key opportunities to improve the identification of novel diseases: (1) use the technologies that already exist to increase data visibility, (2) move from a reporting model to an interoperability model for public health, (3) sustain efforts over time through established and tested systems, and (4) develop an agile mechanism for adapting to new situations, including policy and technological components.
Martin McKee, professor at the London School of Hygiene & Tropical Medicine, discussed lessons learned from bringing the public health model to diagnosis in the United Kingdom. He highlighted the success of mass testing, vaccine rollout, and high rates of genomic sequencing. Reviewing the UK genomic surveillance program, he said the UK sequences 20 percent of all samples, which is one of the highest rates in the world. McKee discussed the four different pillars of testing: (1) PCR tests done in public health and National Health Service labs and hospitals for health care workers and seriously ill patients, (2) PCR and lateral flow tests done for the wider population by commercial partners, (3) antibody testing, and (4) antibody and diagnostic testing for national surveillance and research (DHSC, 2020). McKee shared the “expensive failure” of contact tracing, partly caused by not relying on established public health infrastructure and knowledge, and the lack of support for citizens who needed to isolate if they tested positive. He shared the UK’s key lessons, saying that testing should be as easy as possible for people and have no costs attached. In the UK, up to April 1, 2022, anyone could order a pack of seven tests each day for free, either to be delivered or picked up at a local pharmacy, which differs significantly from the experience of obtaining rapid tests in the United States.
The panel discussion highlighted important policy levers that can be implemented to improve public health infrastructure for diagnosis and surveillance. Overhage said one of the biggest challenges in the United States is the fractured responsibility and decision making at the local, state, and national levels, with varying policies at every step. McKee highlighted the importance of cocreation of policies and explained that many of the mistakes in the UK were because decision makers were not soliciting and considering input from frontline workers and those on the ground, so policies did not reflect the lived reality for most people. Merchant emphasized the importance of identifying policy solutions now to enable better positions for the next emergency, including policy preparedness itself and how to establish allocation of resources so the public health infrastructure is more robust and resilient. McKee added that investing in public health and pandemic preparedness is critical.
NOVEL DIAGNOSTIC STRATEGIES AND TOOLS THAT EMERGED DURING THE COVID-19 PANDEMIC
Rick Bright, senior vice president of pandemic prevention and response at the Rockefeller Foundation, discussed the development and implementation of diagnostic tools outside of the clinical setting. Centralized testing can lead to delays in diagnoses and treatment and an increased risk of transmission as people travel to different sites to get tested, he said. He believed the paradigm is moving in the right direction, bringing diagnostics closer to patients and into the home. Bright shared some examples of personalized and proactive technologies, such as devices that
__________________
11 Contact tracing is defined by CDC as a “working with a patient (symptomatic and asymptomatic) who has been diagnosed with an infectious disease to identify and provide support to people (contacts) who may have been infected through exposure to the patient” (CDC, 2022b).
can scan a person’s eyes to detect early signs of cancer or illness. There are also disposable contact lenses that can monitor blood pressure, and color changing tattoos that can detect biochemistry changes and help identify infections in the body. He described the future potential of multiplex platforms, in which collected samples can be tested for influenza, respiratory syncytial virus, SARS-CoV-2, or other pathogens simultaneously. But several challenges remain, including appropriate data collection, storage, and management; data privacy and security; delivery of appropriate and timely treatment by linking early diagnostics to treatments; regulatory pathways for diagnostics; and reimbursement and financial incentives. As a vision for the future, Bright believes the paradigm shifts from the COVID-19 pandemic have empowered people with the tools to play a larger role in their own health care and emergency response.
Kathleen Weber, president of molecular solutions at OraSure Technologies, discussed lessons learned from home diagnostics. She highlighted the experience with her company’s first in-home test—the OraQuick In-Home human immunodeficiency virus (HIV) test—that launched in the United States in 2012, including the benefit of a noninvasive sampling method (e.g., oral fluid) in expanding access, the ability of consumers to test and manage their results, and the benefits of broadening the risk–benefit discussion from the individual to population level. Focusing on the introduction of self-testing for COVID-19, she noted the following additional insights:
- The diagnostic regulatory ecosystem is not built for speed. Many challenges delayed the ability to bring products to market quickly.
- Leveraging existing platforms accelerated development.
- The pandemic highlighted telehealth as a tool to manage diagnosis and treatment.
- Public–private partnerships are critical for deployment and procurement of tests, including their role in expanding global production capacity.
- There is a critical need to consider global supply chain vulnerabilities and how they relate to diagnostic testing.
In conclusion, Weber offered considerations for the future, such as the ease of methods like oral swabs or urine samples, and continuing investment in noninvasive sample types and platform technology. But to challenge the existing paradigms, mindsets need to continue evolving regardless of whether there is a pandemic, she said.
William Rodriguez, chief executive officer at FIND,12 focused his comments on the implications for innovative diagnosis in global health emergencies, particularly diagnostic innovations in low- and middle-income countries. He emphasized an equity framework focused on vaccine and testing equity and mitigation of COVID-19’s impact, and he noted that the most successful message thus far has been realizing that “no one is safe anywhere until everyone is safe everywhere.” While there has been almost 10 years of diagnostic technology innovation and progress compressed into just 2 years, with “unprecedented acceleration in development and availability” of molecular tests, surveillance-based tools, genomics and sequencing tools, and antigen rapid tests in low- and middle-income counties with limited resources, there are still large inequities in global testing, and the world is still slow in detecting the emergence of new variants, he said.
Today, Rodriguez said, high-income countries are testing at 80-fold higher rates than low-income countries and submitting nearly 1,000 times more sequences to GISAID.13 Inequity issues have worsened, for both COVID-19 testing and diagnosis, as well as for other diseases. Drawing on lessons from the experience in low- and middle-income countries rolling out self-testing for hepatitis and HIV, he recommended community engagement (e.g., digital connectivity and open access digital modules), connecting communities to health systems (e.g., linking communities with their lab results), and strengthening health systems, including support for disease surveillance (e.g., interoperability across health information systems, evidence on use and impact of digital tools, and strategic implementation of tools to enable effective diagnosis).
In the panel discussion, workshop participants discussed innovations to help shorten the timeline of diagnostics approval and use. Weber noted that the development of OraSure’s Ebola test was accelerated by the use of sample panels provided by CDC; however, the lack of such panels has been a challenge for COVID-19 diagnostics. Bright agreed but argued that this responsibility cannot sit solely within government control and management; instead, he called for an independent, unbiased entity that can collect samples, standardize assays, and compare different tests
__________________
12 FIND is a global alliance for diagnostics. For more information, see https://www.finddx.org/about (accessed March 17, 2022).
13 GISAID is a global science initiative established in 2008 that provides open-access rapid sharing of genomic data of emerging viruses as it becomes reported and available.
instead of individual companies duplicating the process. Rodriguez called for a global governance system for pandemics that considers the lessons COVID-19 has taught about nationalism and implications for obtaining supplies. Bright added that if there were a global standardized data system that allowed for sharing of different types of data, it might be possible to develop a standardized baseline about what is happening in the ecosystem, and an early warning system that alerts at the early stages of an outbreak.
ENVISIONING THE FUTURE OF DIAGNOSTIC EXCELLENCE
In a final session, workshop panelists reflected on their visions for diagnostic excellence using lessons learned from the COVID-19 pandemic. Preeti Malani, chief health officer at the University of Michigan, highlighted the challenge of those who self-test at home but do not have a primary care provider and may not receive proper follow-up and treatment. She added that testing is being used by “people who are vaccinated, boosted, and have higher income,” and she emphasized the need to consider people without easy access to testing who may be more vulnerable to COVID-19. Building on that, Nicole Alexander-Scott, past director of the Rhode Island Department of Health, emphasized the importance of prioritizing communities that were hit hardest by the pandemic, considering the limitations and barriers of many social determinants of health, and developing proactive and aggressive strategies to address the disparities. Nicole Lurie, director at the Coalition for Epidemic Preparedness Innovations, emphasized,
We need an end-to-end system that makes it clear who’s responsible for what. That will help us get diagnostics developed faster, more efficiently, and better so they can be made available more equitably.
In addition, Lurie commented on the importance of codesigning systems and tools with communities to help make it easier for patients to take control of their health care. She added that the health care system remains fairly paternalistic and is “having an incredibly painful prolonged shift to what patient centered really means,” which has implications for the future of diagnostic excellence.
Steven Schachter, professor at Harvard Medical School, added that as diagnostic testing and chronic disease monitoring moves closer to the patient, support should be available for those individuals to help understand the results. He also highlighted lessons in accelerating the RADx Tech production of at-home COVID-19 tests:14 the need to understand the perspectives and education level of the public early on and who the user community would be, and interacting with policy makers at an earlier stage. He suggested a multistakeholder consortium with a shared vision of the future that can work collaboratively to define the regulatory pathway and what information is needed at all levels. Alexander-Scott concluded by emphasizing the importance of including the community voice as an equal partner going forward.
Suggestions from workshop speakers to improve diagnostic excellence are outlined in Box 1.
__________________
14 The RADx Tech initiative aims to speed the development, validation, and commercialization of innovative point-of-care and home-based tests, as well as improve clinical laboratory tests, that can directly detect the virus. See https://www.nih.gov/research-training/medical-research-initiatives/radx/radx-programs (accessed March 11, 2022).
REFERENCES
AAPC (American Academy of Professional Coders). 2021. What is ICD-10? https://www.aapc.com/icd-10 (accessed March 3, 2022).
American Indian Policy Review Commission. 1977. American Indian Policy Review Commission final report submitted to Congress May 17, 1977. Vol. 1. Washington, DC: U.S. Congress.
ANA (Administration for Native Americans). 2022. American Indians and Alaska Natives—The trust responsibility: Fact sheet. https://www.acf.hhs.gov/ana/fact-sheet/american-indians-and-alaska-natives-trust-responsibility (accessed March 3, 2022).
ASM (American Society for Microbiology). 2021. Supply shortages impacting COVID-19 and non-COVID testing. https://asm.org/Articles/2020/September/Clinical-Microbiology-Supply-Shortage-Collecti-1 (accessed March 17, 2022).
BIA (Bureau of Indian Affairs). 2022. Frequently asked questions. https://www.bia.gov/frequently-asked-questions (accessed March 3, 2022).
Bigelow, B. F., R. E. Saxton, A. Flores-Miller, H. H. Mostafa, M. J. Loss, K. H. Phillips, A. M. Moore, W. D. Hale, T. M. Tolson, N. S. McCann, C. L. Catlett, S. H. Golden, J. M. Clark, and K. R. Page. 2021. Community testing and SARS-CoV-2 rates for Latinxs in Baltimore. American Journal of Preventive Medicine 60(6):e281-e286.
CDC (Centers for Disease Control and Prevention). 2015. Multidrug-resistant organisms (MDRO) management glossary. https://www.cdc.gov/infectioncontrol/guidelines/mdro/glossary.html (accessed January 7, 2022).
CDC. 2022a. COVID data tracker: Demographic trends of COVID-19 cases and deaths in the US reported to CDC. https://covid.cdc.gov/covid-data-tracker/#demographics (accessed March 3, 2022).
CDC. 2022b. Interim guidance on developing a COVID-19 case investigation & contact tracing plan: Overview. https://www.cdc.gov/coronavirus/2019-ncov/php/contact-tracing/contact-tracing-plan/overview.html (accessed March 4, 2022).
CMS (Centers for Medicare and Medicaid Services). 2022. ICD-10-CM official guidelines for coding and reporting FY22. https://www.cms.gov/files/document/fy-2022-icd-10-cm-coding-guidelines-updated-02012022.pdf (accessed March 3, 2022).
DHSC (Department of Health and Social Care, United Kingdom). 2020. Guidance: COVID-19 testing data: Methodology note. https://www.gov.uk/government/publications/coronavirus-covid-19-testing-data-methodology/covid-19-testing-data-methodology-note (accessed March 3, 2022).
Guntuku, S. C., G. Sherman, D. C. Stokes, A. K. Agarwal, E. Seltzer, R. M. Merchant, and L. H. Ungar. 2020. Tracking mental health and symptom mentions on Twitter during COVID-19. Journal of General Internal Medicine 35(9):2798-2800.
IOM (Institute of Medicine). 2001. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
Lee, J. S., J. M. Goldstein, J. L. Moon, O. Herzegh, D. A. Bagarozzi, Jr., M. S. Oberste, H. Hughes, K. Bedi, D. Gerard, B. Cameron, C. Benton, A. Chida, A. Ahmad, D. J. Petway, Jr., X. Tang, N. Sulaiman, D. Teklu, D. Batra, D. Howard, M. Sheth, W. Kuhnert, S. R. Bialek, C. L. Hutson, J. Pohl, and D. S. Carroll. 2021. Analysis of the initial lot of the CDC 2019-novel coronavirus (2019-NCOV) real-time RT-PCR diagnostic panel. PLOS ONE 16(12):e0260487.
Marcello, R. K., J. Dolle, A. Tariq, S. Kaur, L. Wong, J. Curcio, R. Thachil, S. S. Yi, and N. Islam. 2022. Disaggregating Asian race reveals COVID-19 disparities among Asian American patients at New York City’s public hospital system. Public Health Reports 137(2):317-325.
McCormack, G., C. Avery, A. K.-L. Spitzer, and A. Chandra. 2020. Economic vulnerability of households with essential workers. JAMA 324(4):388-390.
Powder, J. 2021. Keys to the Navajo Nation’s COVID-19 vaccination success. https://publichealth.jhu.edu/2021/keys-to-the-navajo-nations-covid-19-vaccination-success (accessed March 3, 2022).
Southwick, L., S. C. Guntuku, E. V. Klinger, E. Seltzer, H. J. McCalpin, and R. M. Merchant. 2021. Characterizing COVID-19 content posted to TikTok: Public sentiment and response during the first phase of the COVID-19 pandemic. Journal of Adolescent Health 69(2):234-241.
The Economist. 2020. Instagram may offer clues about the spread of the new coronavirus. https://www.economist.com/graphic-detail/2020/03/17/instagram-may-offer-clues-about-the-spread-of-the-new-coronavirus (accessed March 3, 2022).
Villarreal, M. 2021. Navajo Nation battles back from COVID-19, leads country in vaccinations. https://www.krqe.com/news/new-mexico/navajo-nation-battles-back-from-covid-19-leads-country-in-vaccinations (accessed March 3, 2022).
WHO (World Health Organization). 2022. Infodemic. https://www.who.int/health-topics/infodemic#tab=tab_1 (accessed March 17, 2022).
DISCLAIMER: This Proceedings of a Workshop—in Brief has been prepared by Jennifer Lalitha Flaubert, Ruth Cooper, and Megan Snair as a factual summary of what occurred at the meeting. The statements made are those of the rapporteurs or individual workshop participants and do not necessarily represent the views of all workshop participants, the planning committee, or the National Academies of Sciences, Engineering, and Medicine.
*The National Academies of Sciences, Engineering, and Medicine’s planning committees are solely responsible for organizing the workshop, identifying topics, and choosing speakers. The responsibility for the published Proceedings of a Workshop—in Brief rests with the workshop rapporteurs and the institution. The planning committee comprises Andrew Bindman (Co-Chair), Kaiser Permanente; Derek Angus (Co-Chair), University of Pittsburgh; Amesh Adalja, The Johns Hopkins Center for Health Security; Suzanne Bakken, Columbia University; Marie-Carmelle Elie, University of Alabama School of Medicine; Nicole Lurie, Coalition for Epidemic Preparedness Innovations; Kathryn McDonald, The Johns Hopkins University; Kosali Simon, Indiana University; and Saul Weingart, Rhode Island Hospital and Brown University.
REVIEWERS: To ensure that it meets institutional standards for quality and objectivity, this Proceedings of a Workshop—in Brief was reviewed by Chau Trinh-Shevrin, NYU Langone Health, and Eric J. Rubin, Harvard T. H. Chan School of Public Health and New England Journal of Medicine. Leslie J. Sim, National Academies of Sciences, Engineering, and Medicine, served as the review coordinator.
STAFF: Jennifer Lalitha Flaubert, Ruth Cooper, Anesia Wilks, Nikita Varman, and Sharyl Nass, Board on Health Care Services, Health and Medicine Division, National Academies of Sciences, Engineering, and Medicine
SPONSORS: This workshop was supported by the Gordon and Betty Moore Foundation.
For additional information regarding the workshop, visit https://www.nationalacademies.org/event/01-14-2022/lessons-learned-from-the-covid-19-pandemic-to-improve-diagnosis.
Suggested citation: National Academies of Sciences, Engineering, and Medicine. 2022. Lessons learned from the COVID-19 pandemic to improve diagnosis: Proceedings of a workshop—in brief. Washington, DC: The National Academies Press. https://doi.org/10.17226/26567.
Health and Medicine Division

Copyright 2022 by the National Academy of Sciences. All rights reserved.











