4
Manufacturing Science
The momentum created during the COVID-19 pandemic has highlighted the importance of speedy, high-quality, scalable, reliable, and affordable vaccine manufacturing, as means to prepare and respond quickly to potential influenza pandemics (Aars et al., 2021). Adding to the current influenza vaccine manufacturing landscape described in Chapter 1, understanding pandemic vaccine manufacturing preparedness and new vaccine technologies that have been rapidly developed and achieved first authorization during the COVID-19 pandemic offers the potential to be used for influenza pandemic preparedness (CEA, 2019). This chapter considers how to build on the momentum of the COVID-19 vaccines through continued progress on the strengthening of influenza vaccine manufacturing science and preparedness. The audacious goal would be to ensure that there is active global manufacturing capacity to produce up to 20 billion doses in 1 year to cover at least 80 percent of the world’s population in the event of an influenza pandemic (Hoyt and Johnston, 2021).
This chapter addresses the third of the four dimensions of vaccine research, development, and manufacturing examined by the committee. It begins by describing the role of manufacturing science in the development of COVID-19 vaccines. The chapter then considers how lessons learned from the production of COVID-19 vaccines (shown in Box 4-1) can be applied to both seasonal and pandemic influenza vaccines in the dimension of manufacturing science. The final section presents recommendations drawn from these findings.
MANUFACTURING SCIENCE AND THE DEVELOPMENT OF COVID-19 VACCINES
Global collaboration is essential during pre-pandemic and pandemic times. An increase in the number of vaccine manufacturers establishing new partnerships, collaborations, and agreements to adopt or strengthen manufacturing scale-up and scale-out capabilities has helped accelerate the fast rollout of the COVID-19 vaccines. In early 2020, manufacturers, as well as governments, began to invest in ramping up manufacturing capacity (scale-up) and establishing partnerships with other manufacturers (scale-out) to produce the needed supplies of COVID-19 vaccines (Hoyt and Johnston, 2021). Governments pledged billions of dollars to companies with promising candidates. For example, the U.S. government-initiated public–private partnership Operation Warp Speed invested more than $10 billion in the clinical development and manufacturing of COVID-19 vaccines (HHS, 2020b). There are limits to expanding capacity in the short term, and establishing new facilities is a time-consuming process. Still, the progress made in this regard during the COVID-19 pandemic points to the opportunity to improve global manufacturing capacity for influenza vaccines and set a course for sustainability and continued progress, including preparedness for pandemic influenza.
The development of COVID-19 vaccines has also highlighted the role of basic and translational research in accelerating vaccine development (see Chapter 2). The online publication of the viral sequence made a major difference in the development of vaccines against COVID-19. On January 10, 2020, the SARS-CoV-2 sequence was posted online (Paden et al., 2020), and laboratories worldwide used this sequence to design, synthesize, and develop COVID-19 vaccine candidates. The Global Initiative on Sharing All Influenza Data (GISAID) played an important role by providing a mechanism that both producers of sequence data and consumers of sequence data found mutually acceptable. It worked very well in this pandemic response, as it had worked during the 2013 pre-pandemic H7N9 influenza outbreak (Hay and McCauley, 2018). It took approximately 1 week to produce mRNA and DNA vaccine candidates from the published sequence, 3 weeks to produce viral vectors, and 2 months to produce protein-based candidates (Corbett et al., 2020; Rappuoli et al., 2021). Therefore, different platforms had very different development times during COVID-19, with the novel mRNA vaccine technology being the platform with the fastest development timelines to create a vaccine prototype.
Use of the various new production platforms used to manufacture COVID-19 vaccines also has illuminated the complexities of the vaccine development ecosystem and the many challenges that manufacturers face. For instance, supply shortages occurred during the COVID-19 response, in the manufacturing of not only the specific vaccines but also all laboratory reagents and materials (Rele, 2021). There were some specific, unanticipated challenges, such as an unanticipated pipette tip shortage (Woolston, 2021). Furthermore, there was a shortage in supply of specialty chemicals, such as some of the lipid components of lipid nanoparticles (Schmidt, 2021). Similar shortages have been observed in previous pandemic outbreaks of influenza, such as H1N1 in 2009, with respect to shortages of sterile eggs, cell lines, and other necessary tools.
International institutions and organizations played a critical role in the production of COVID-19 vaccines. The COVAX project was established between the World Health Organization (WHO); the Coalition for Epidemic Preparedness Innovations (CEPI); Gavi, the Vaccine Alliance; and the United Nations Children’s Fund (UNICEF) for the distribution and procurement of COVID-19 vaccines. COVAX is part of the Access to COVID-19 Tools Accelerator that aims to guarantee a fair and equitable global access to vaccines (Gavi, 2021a). These sorts of multilateral collaborations can guarantee procurement of vaccines to ensure a sustainable market for any manufactured product. Gavi, the Vaccine Alliance now procures vaccines for 17 diseases and negotiates pricing for various regions, having lower-income countries contribute a lower share of the costs (Gavi, 2021b).
Beyond actual production of the COVID-19 vaccines, manufacturers increased capacity at early clinical trial stages to be ready to start distribu-
tion immediately after authorization. Additionally, various scale-out initiatives were undertaken where manufacturers agreed to produce vaccines developed by other companies. For example, Merck agreed to manufacture the Johnson & Johnson vaccine, while Sanofi and Novartis manufactured the Pfizer-BioNTech vaccine (Merck, 2021; Novartis, 2021; Sanofi, 2021). Increasingly, the pharmaceutical industry has outsourced manufacturing to contract manufacturing organizations, which can aid in reducing costs and, as such, lower the financial risk at an early stage of development. Furthermore, it would be useful if vaccine producers shared more data during the preclinical and clinical development of pandemic vaccinations. GISAID proved to be an exceptionally effective mechanism for sharing data of viral isolate sequences during the COVID-19 pandemic, providing a model for similar efforts to be made for other data.
During the COVID-19 pandemic, many pharmaceutical and vaccine companies have been generous and altruistic in sharing knowledge and costs and optimizing collaboration, but such collaboration is exceptional and may not become common practice or applicable during interpandemic periods (Knobler et al., 2020). Renewed business and financial strategies (short- and long-term) will be needed and are critical to ensuring a global, sustainable vaccine manufacturing framework, particularly in low- and middle-income countries (LMICs).
Finally, many of the COVID-19 vaccine candidates have involved a university or college assisting in preclinical research or clinical trials. For example, the AstraZeneca–University of Oxford vaccine started as a collaboration between the Jenner Institute of the University of Oxford and a spinoff company, Vaccitech (AstraZeneca, 2020a). Financing of development came from various sources, including Oxford Sciences Innovation, Google Ventures, and Sequoia Capital (Baker, 2020). To produce doses for the initial clinical trials, a collaboration was set up with the Italian company Advent Srl (IRBM Group, 2020), after which an agreement was signed between AstraZeneca, CEPI, and Gavi that secured the production of 300 million doses. An additional agreement was signed with the Serum Institute of India for 1 billion doses to be allocated to LMICs (AstraZeneca, 2020b). Similar collaborations existed for other vaccines, such as when Pfizer and Moderna teamed up to create an mRNA influenza vaccine (BioNTech, 2018).
MANUFACTURING SCIENCE: APPLYING LESSONS FROM THE PRODUCTION OF COVID-19 VACCINES TO VACCINES FOR INFLUENZA
Lessons learned from the production of COVID-19 vaccines highlight the critical importance of pandemic vaccine manufacturing preparedness, especially at the global level. Particular needs demonstrated by the COVID-19
experience include the establishment of (1) continuous, adaptive, flexible, and sustainable manufacturing infrastructure; (2) incentives to enable and facilitate strong, coordinated collaborations among manufacturers; and (3) establishment of global and local support manufacturing networks. In addition, given the vital role vaccine manufacturers play and their responsibilities and accountabilities for the products they develop, produce, and license, the committee identified specific actions that would strengthen seasonal and pandemic influenza vaccine manufacturing. The following sections detail lessons learned from the production of COVID-19 vaccines that can be applied to the manufacturing of influenza vaccines in the areas of global vaccine manufacturing preparedness, scientific and technical considerations, accelerating the timeline for vaccine production, the role of manufacturers and global manufacturing inequities, economic and market considerations, and global stakeholders and international collaborations.
Global Vaccine Manufacturing Preparedness
The 2019–2030 WHO Global Influenza Strategy, the 2020–2030 U.S. National Influenza Vaccine Modernization Strategy, and other documents stress the importance of optimizing global influenza vaccine manufacturing science and technology (HHS, 2020a; WHO, 2019). The COVID-19 pandemic has provided clarity on the need for a global effort to strengthen vaccine manufacturing capacity and response commensurate with the size of the global problem—a need highlighted above.
The speed at which a vaccine can be designed, developed, produced, tested, authorized, and widely and equitably distributed is the driving factor behind the response to a pandemic, significantly affecting the spread and burden of disease. A rapid response not only provides vital health benefits but also addresses important social and economic consequences (Shang et al., 2021). Therefore, considering the speed of developing a safe and effective vaccine is of the essence for pandemic influenza vaccine development and preparedness. Box 4-2 outlines transformations in vaccine research, development, and production applied to producing COVID-19 vaccines that helped accelerate manufacturing timelines.
Scientific and Technical Considerations
Pre-Pandemic Vaccines
For influenza, continuous monitoring systems for circulating influenza strains, such as the Tool for Influenza Pandemic Risk Assessment, developed by WHO in 2016, are very valuable for rapid identification of new virus strains that have the potential to cause a pandemic (Harrington et
al., 2021). These strains can therefore be used to develop, manufacture, and stockpile so-called pre-pandemic vaccines. These vaccines would be deployed and used early in a pandemic response, diminishing the disease burden and allowing for pandemic-specific vaccines to be advanced in parallel. Currently, there are few pre-pandemic vaccines licensed for influenza (Soema et al., 2015). However, AUDENZ, an influenza A H5N1 adjuvanted monovalent vaccine, was approved by the U.S. Food and Drug Administration (FDA) in February 2020 and is the first of its kind designed to be stockpiled to protect against an influenza A pandemic (Cision PR Newswire, 2020).
Vaccine Platform Technologies
The years of research and development (R&D) for SARS-CoV and MERS-CoV vaccines offered an advantage to speed up the process and to rapidly advance manufacturing science for COVID-19. For instance, the discovery of di-proline mutation stabilization of pre-fusion S from MERS-CoV and the subsequent demonstration that this mutation also stabilized pre-fusion S from seasonal coronaviruses and SARS-CoV enabled the rapid adoption for SARS-CoV-2 vaccines (Duan et al., 2020; Pallesen et al., 2017). Prior work also permitted an acceleration of the vaccine products into clinical phases
of development, with the time to start the first COVID-19 clinical trials approximately 66 days for mRNA vaccines, 3 months for viral vector vaccines, and 6 months for protein-based vaccines (Corbett et al., 2020; Rappuoli et al., 2021).
The seasonal influenza vaccine production process is a well-established machine that renews vaccines every 6 months. Every year, an estimated 1.48 billion doses of influenza vaccines can be produced and used for seasonal influenza programs worldwide. Despite the current infrastructure, however, an influenza pandemic may require rapid capability to manufacture 4.15–8.31 billion doses to produce and distribute a sufficient number of vaccine doses in a timely and equitable manner worldwide (Sparrow et al., 2021) (see Figure 4-1). The current vaccine production strategy for an influenza pandemic is to switch manufacturing capacity for seasonal vaccine into production of pandemic vaccines. Seasonal vaccines consist of multiple strains, so, for instance, the four-strain vaccine would translate into four doses of a single-strain pandemic vaccine, depending on the dose of the antigen or whether a dose-sparing adjuvant is included. In addition, the seasonal manufacturing is restricted to a time limit between the provision of seeds until a target number of doses is prepared for the season; a pandemic would not have such time restrictions. Approximately 90 percent of seasonal vaccines in use are inactivated virus vaccines (Matthews, 2006). Out of these, 90 percent are produced using the embryonated chicken egg-based platform (Bender, 2019).
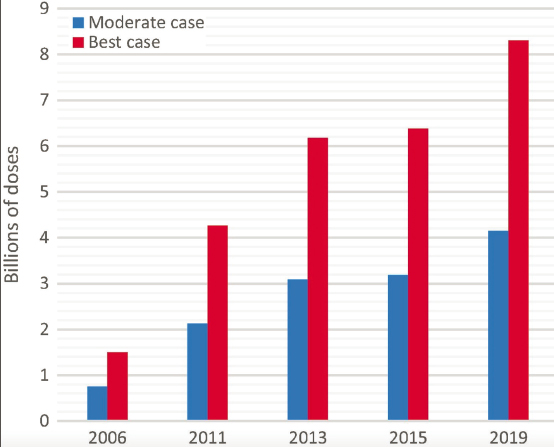
SOURCE: Sparrow et al., 2021.
Reliance on the egg-based platform to meet the demands for pandemic vaccine production is challenging. First, to produce the large number of vaccines necessary during a pandemic, all seasonal vaccine manufacturing capacity must be converted to pandemic vaccine production at maximum capacity. Second, the time between strain selection and administering vaccines is lengthy as egg-based vaccine production averages 6 months (Rajaram et al., 2020). Third, production strategy is dependent on supplies. As noted earlier, during the COVID-19 pandemic, supply chain issues became a rate-limiting step due to increases in demand and reduced production and delivery of raw materials (Hatchett et al., 2021; Paris, 2020; Rele, 2021). Finally, the bottleneck for producing these vaccines is a steady supply of embryonated chicken eggs (Buckland, 2015). Therefore, investment in alternative platforms is necessary to ensure readiness for the next influenza pandemic.
Conclusion 4-1: A pandemic would require an increase in manufacturing facilities that would be cost-effective only if they were used beyond pandemic vaccine production (Plotkin et al., 2017). Therefore, public–private partnerships are needed to invest in this technology to produce vaccines for seasonal influenza to ensure preparedness for a pandemic.
Cell-Based Technology
As discussed previously, the current principal alternative vaccine production platform uses cell-based technology (Rajaram et al., 2020). These vaccines are currently more expensive than vaccines produced using the egg-based system (Ng, 2012).
mRNA-Based Vaccines
The speedy development and high efficacy rate of mRNA-based vaccines for COVID-19 have fueled the attention and faith in the mRNA vaccine platform technology (Tenforde et al., 2021). This creates an opportunity to employ mRNA technology to develop and test vaccines for other viruses, such as influenza, and their use in acute outbreaks. Vaccines relying on mRNA technology have reached prominent levels during the COVID-19 pandemic, showing that this platform can be feasible and safe for the use in pandemics (Anand and Stahel, 2021). During a pandemic response, those who would benefit from technology transfer are the same groups who would have to find means of using existing facilities to produce the vaccine. Therefore, such capacity building and technology transfer is best done during interpandemic periods and has been proposed by the G20 as a way to close the global gaps in pandemic preparedness (G20, 2021).
Conclusion 4-2: The capacity to manufacture RNA vaccines needs to be expanded with a blueprint for the establishment of continuous, adaptive, flexible, and sustainable manufacturing infrastructure. Improvements also need to be made to vaccine formulations to improve aspects such as the cold chain requirements for mRNA vaccines (Crommelin et al., 2021).
A 2021 analysis by Public Citizen estimates that to produce 8 billion doses of the Moderna COVID-19 mRNA vaccine in 1 year would require at least 55 production lines set in 14 different facilities and with a total investment of at least $23 billion (Kis and Rizvi, 2021). This highlights the need for distributed manufacturing. Indeed, WHO, with the goal to expand the capacity of LMICs to produce COVID-19 vaccines, launched a request for expressions of interest to advance a vaccine transfer hub for the technology transfer of mRNA vaccines immediately (WHO, 2021a). A total of 19 manufacturers from more than a dozen countries in Africa, Asia, and Latin America responded. However, proprietary issues have not been resolved at this moment and may be a limiting factor in achieving these goals to increase global manufacturing capacity. Box 4-3 summarizes gaps and challenges for the use of mRNA-based vaccines as a tool for influenza preparedness.
Conclusion 4-3: In addition to the insufficient manufacturing capacity for mRNA vaccines, clinical data on the efficacy of mRNA vaccines for various types of viruses, including influenza, are very limited. Therefore, to make decisions on whether mRNA technology should be developed for influenza vaccines, clinical data need to be expanded.
Recombinant Protein Vaccines
Recombinant protein vaccines are another platform of interest for the development of influenza vaccines. Various platforms can be used for the production of recombinant proteins. Using mammalian and insect cell culture systems results in high-quality proteins with the correct posttranslational modifications. However, because of proprietary obstacles, use of these cell lines is relatively costly (Barr et al., 2018). Bacterial systems, such as the use of Escherichia coli are less costly but result in incorrect folding of the full-length HA domain (Aguilar-Yáñez et al., 2010; DuBois et al., 2011). Yeast systems are also less costly than mammalian cell lines, but yeast require culture in low pH conditions, which may result in hyperglycosylation of the protein (Murugan et al., 2013; Saelens et al., 1999). A diverse portfolio of vaccine platforms is essential to achieve the manufacturing capacity that will be required during a pandemic, and protein-based vaccines are an essential tool to be used in a pandemic when there is an enormous need for vaccines. Furthermore, the development and use of appropriately selected adjuvants could reduce barriers and timelines by decreasing the amount of vaccine antigen required per dose or by broadening, or lengthening, the conferred protection, possibly allowing protection even with single-dose vaccination (Lemoine et al., 2021).
Accelerating the Timeline for Influenza Vaccine Production
Leveraging Experience with Seasonal Influenza Vaccines to Enhance Pandemic Preparedness
In an influenza pandemic, the vaccine manufacturing capacity will be based on the seasonal influenza vaccine production ecosystem and will be better prepared compared to COVID-19. The existing conventional production capacity for seasonal influenza vaccines is well poised to have a rapid changeover and can be converted to pandemic vaccine production relatively rapidly (Rockman et al., 2020). This is an advantage in preparing for a pandemic influenza scenario, contrary to what occurred with the COVID-19 pandemic, where the world was not vaccine ready even after having suffered from two prior zoonotic coronavirus epidemics.
Conclusion 4-4: Historical knowledge of manufacturing seasonal and pandemic influenza vaccines can be used to further strengthen pandemic influenza vaccine R&D and ensure a path toward accelerated production and testing timelines. Manufacturing steps, such as the validation of bulk manufacturing and scale-up production, should be able to occur more rapidly compared to the production of COVID-19 vaccines.
Ensuring Fill and Finish Capacity and the Availability of Raw Materials
The manufacturing of pandemic vaccines can be planned and performed accurately only after the bulk vaccine is produced, as this is largely based on the yield and growth of the virus in egg-based or cell-based systems. Filling of vials can start only after the yield is established, and the yield will depend on the viral strain and vaccine platform used. As dosing and final filling requirements remain unknown until clinical trial data are available (see below), clinical trials are the rate-limiting step in this process. At present, moreover, the United States has the fill and finish capacity for approximately 150 million vaccines, whereas in the event of a pandemic, an estimated 600 million would be needed (NASEM, 2019).
Conclusion 4-5: Capacity for fill and finish needs to be expanded and strengthened, alongside the availability of other consumables, such as needles and syringes. Also crucial is the availability of the raw materials, such as chicken eggs, needed to manufacture the vaccines. To enable speed and avoid limitations in bulk manufacturing, these raw materials need to be made available rapidly and in sufficient quantities.
Advancing Vaccine Candidates Through Clinical Development
Other factors that can improve and accelerate the development time of pandemic vaccines include changes to the framework used for advancing vaccine candidates through clinical development. For influenza, when a novel virus is identified, bulk vaccine is produced and the United States creates a stockpile. However, the system of surveillance and development differs from that of seasonal vaccines. Unlike seasonal vaccines, pre-pandemic vaccines first have to be shown to induce immunogenicity in relevant populations, after which the vaccines need to undergo clinical trials to assess efficacy and safety before they are authorized for use in a pandemic. Such trials can, however, only be conducted once a pandemic is in progress. Traditionally, and before the COVID-19 pandemic, clinical phases I, II, and III of development generally occurred sequentially. During the COVID-19
pandemic, it became clear that these processes can be shortened by ensuring a seamless transition between clinical phases (GAO, 2021).
Investing in the Development of Platforms for Seasonal Influenza Vaccines
Continuous investments by the public sector are needed to develop a variety of vaccine platforms for seasonal influenza, including novel platforms such as mRNA and recombinant protein vaccines. Expansion of vaccine platforms will be gradual as the private sector develops the means to build capacity for novel vaccine development. During this period of capacity building, existing vaccine platforms will be important to meet the demand.
Conclusion 4-6: As digital technologies, the science of vaccine formulation, and adjuvant technologies expand, new platforms for the development of seasonal influenza vaccines will be needed to complement rather than replace conventional vaccine platforms.
The Role of Manufacturers and Global Manufacturing Inequities
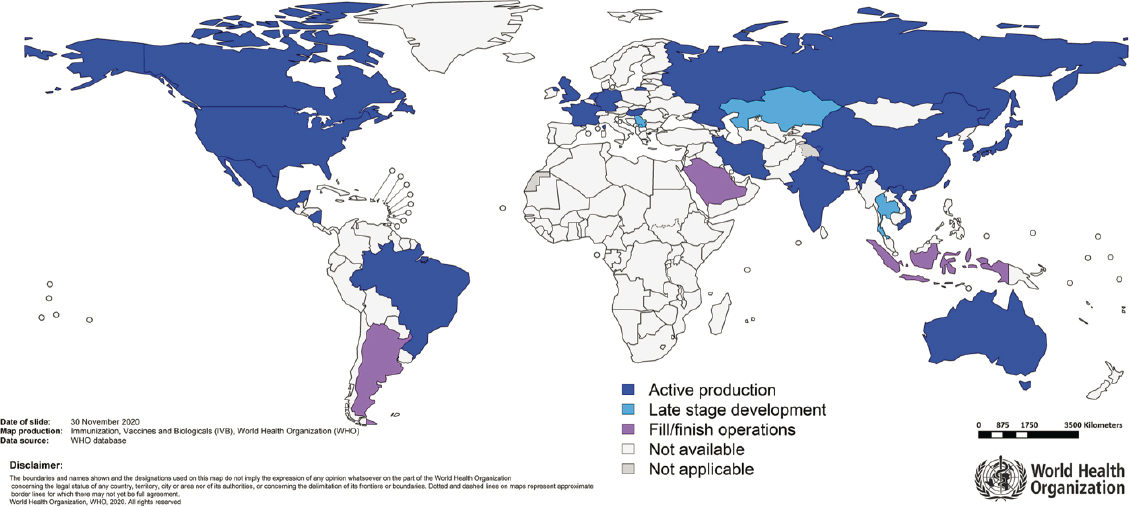
As discussed earlier, vaccine manufacturers have undergone significant transformation during the COVID-19 pandemic, and acceleration in technology transfer has been evident. However, global inequity in vaccine manufacturing and distribution continues to be a hurdle. As of 2019, there were 40 active influenza vaccine production facilities worldwide. Twenty of these were located in high-income countries (HICs), with 15 in upper-middle- and five in lower-middle-income countries (Sparrow et al., 2021). Box 4-4 lists the essential responsibilities of vaccine manufacturers, while Box 4-5 outlines the attributes of effective vaccine manufacturing technology transfer during pandemics.
Generally, vaccines are manufactured in HICs, and this affects distribution as these countries are also the first to receive vaccines and provide them to their population, while LMICs have minimal influenza vaccine manufacturing capacity (Palache et al., 2017). As of June 2021, one-third of HIC populations had received at least one dose of COVID-19 vaccine, compared to just 1 percent in LMICs (Berkley, 2021). As of September 2021, that percentage had only risen to just over 2 percent (Our World in Data, 2021) (see also Chapter 3). Many of these countries additionally have limited use of the seasonal influenza vaccines, as programs needed for that purpose are not in place. In some cases, this is due to the high costs of a vaccination program, while in other cases, it is caused by a need for surveillance to evaluate the need for a yearly program, competing demands, or the need
to strengthen health systems (Sparrow et al., 2021). Consequently, it is important to overcome the obstacle of convincing certain regions of the world about the importance of investing in the seasonal influenza landscape, and even more so investing in the new vaccine platforms for influenza preparedness. This situation is complicated by the fact that the seasonal influenza vaccine has limited and variable efficacy and the fact that many LMICs lack the resources to make diagnoses and to identify new, rare adverse event patterns. Quality control, quality management, and pharmacovigilance are key factors contributing to past successes of technology transfer efforts and are essential for technology transfer to LMICs. Figures 4-2 and 4-3 show the prevalence of adult immunization programs in the world and reflect that many countries have influenza programs in place, particularly in Europe and the Americas. However, there appears to be a significant disparity in the existence of these programs between HICs and LMICs.
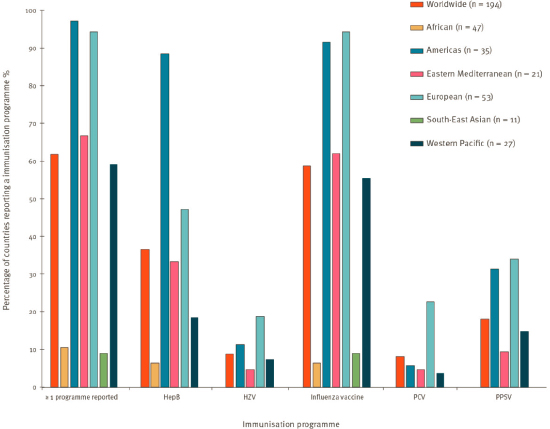
NOTE: Vaccines include Hepatitis B (HepB); Herpes Zoster (HZV), also known as Shingles; Influenza; Pneumococcal Conjugate (PCV); and Pneumococcal polysaccharide (PPSV).
SOURCE: Williams et al., 2021.
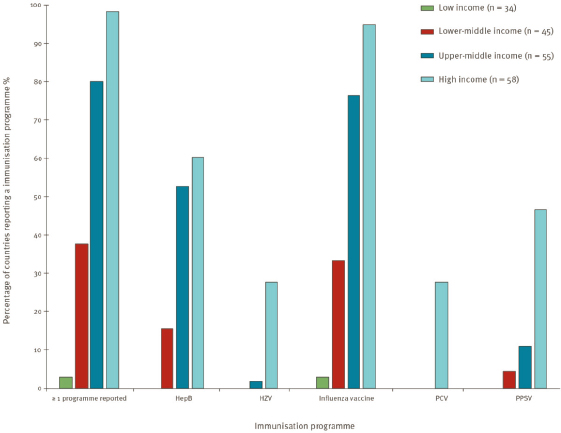
NOTE: Vaccines include Hepatitis B (HepB); Herpes Zoster (HZV), also known as Shingles; Influenza; Pneumococcal Conjugate (PCV); and Pneumococcal polysaccharide (PPSV).
SOURCE: Williams et al., 2021.
While LMICs currently lack the infrastructure for adequate vaccine manufacturing capacity (Figueroa et al., 2021), some initiatives to fill this gap have been conducted. In 2006, WHO launched the Global Action Plan (GAP) for influenza vaccines, intended to address predicted shortages of global vaccine supply when there is an influenza pandemic (Palkonyay and Fatima, 2016). Between 2009 and 2019, PATH supported vaccine manufacturers in Brazil, China, India, and other countries to develop, manufacture, and supply seasonal and pandemic influenza vaccines. The focus of these initiatives was on increasing capacity for both seasonal and pandemic vaccines, and thereby access to the vaccines in LMICs (PATH, 2019).
As noted earlier, during the COVID-19 pandemic, there were unprecedented partnerships between vaccine developers, which allowed the scale-up and scale-out of vaccine manufacturing in parallel with clinical development. Previous work was valuable in developing a model for accelerated vaccine development for COVID-19. Manufacturers have predicted the ability to produce 12 billion COVID-19 vaccines in 2021, which would be enough to vaccinate 70 percent of the world’s population, assuming the majority would require two doses of vaccine (Irwin, 2021). However, since the moment the vaccines received Emergency Use Authorization in various countries, purchases by local governments have determined where the vaccines were distributed, resulting in vaccine inequity globally (Hyder et al., 2021). There is considerable variation in production capacity (see Figure 4-4) for various vaccine platforms, and not all vaccine producers can expand capacity evenly (Grand View Research, 2020). This makes it difficult for policy and decision makers to understand the supply chains involved and take urgent action where it is most required.
Additionally, global needs change continuously during a pandemic. This has also become evident during the COVID-19 pandemic. For instance, the emergence of virus variants of concern may require new vaccines or boosters, the length of immunity and need for boosters to extend immunity remains unknown, and it remains unclear whether and when the vaccines can be used for young children (WHO, 2021b). Additionally, some countries may want to purchase and store a surplus of vaccines for potential future use. These factors affect the requirements and the supply of vaccines to various regions of the world.
During a pandemic, manufacturers are generally more willing to collaborate than between pandemics. An example of a collaboration between manufacturers that has been initiated during the COVID-19 pandemic to scale-up and scale-out is the collaboration between AstraZeneca and the University of Oxford, each of which also has many partners for fill and finish (AstraZeneca, 2020a). Pfizer and BioNTech are an example of a successful alliance that allows novel technology developments from biotechnology companies to be developed at a large scale (Pfizer, 2020). Further collabora-
tion and technology transfer might enhance the global supply and lift the burden of the pandemic. However, technology transfer is not a matter of just handing over protocols. It is a complex process; training people and setting up a new facility with all the critical quality attributes, critical process parameters, and comparable protocols is a time-consuming and costly process. New facilities will have to set up supply chains, train people, and provide registration documentation to regulatory agencies. As an example, the Pfizer BioNTech collaboration started in August 2018 when the two companies began collaborating on an mRNA influenza vaccine (BioNTech, 2018). This allowed the companies to work together for a substantial period of time that was essential for the effective collaboration during the pandemic. Given the complexity and challenges of carrying out technology transfer with limited resources, an initiative was undertaken by WHO in 2007 in collaboration with the Netherlands Vaccine Institute to increase technology transfer of seasonal influenza vaccine manufacturing using the egg-based platform. The initiative included setting up a facility training hub built at an industrial scale for LMICs; 15 countries participated and received training and technical assistance (Hendriks et al., 2011). The program resulted in an expansion of the global manufacturing capacity for seasonal and pandemic influenza vaccines, but it also showed that long-term investments and local, national, and international regulatory agility, alignment, and consistency are critical, with seasonal influenza vaccine uptake locally as an essential factor.
Currently, then, there is a need to increase vaccine manufacturing capacity to strengthen global health security, as this capacity is the bottleneck in the current rollout of COVID-19 vaccines. To this end, a global manufacturing network is needed to build up regional infrastructure and expertise. In particular, the manufacturing structure requires collaborations for technical support or knowledge transfer; savvy management of intellectual property; methods for handling complexities in supply chains infrastructure; regulatory science (see Chapter 5); and robust financing to support the costs associated with preparedness and pandemic response (Aars et al., 2021; Hotez et al., 2021).
Conclusion 4-7: Global manufacturing networks can aid in establishing public–private partnerships to allow surge capacity in a pandemic. They can also help build up regional infrastructure and expertise in LMICs to expand manufacturing capacity in these regions and improve health security for the countries. There is a need for sustainable and transparent partnerships to make technology transfer successful and increase existing vaccine manufacturing capacity.
Chapter 2 emphasizes the importance of standardizing assays, reagents, and animal models for each vaccine platform. Technology transfer needs to
include these standardized methods and high-quality data. Sustained funding for these developments from the public sector, in close collaboration with the private sector, is warranted, with the requirement that the private sector support the technology transfer.
Conclusion 4-8: To increase vaccine manufacturing globally, manufacturing processes need to be developed and facilities operationalized in LMICs. Vaccine manufacturing capacity in LMICs could be improved by enhancing technical competencies and quality management systems, providing technical support for the development of seasonal and pandemic influenza vaccines, and developing platforms for local technical training. These measures would help LMICs acquire the manufacturing knowledge needed to expand global production capacity.
Conclusion 4-9: LMICs also need support in the clinical development of newly manufactured vaccines to aid in coordinating and evaluating results of clinical trials. Additionally, given that manufacturers are responsible for safety, effectiveness, and product quality, their regulatory and pharmacovigilance capacity needs to be strengthened.
Conclusion 4-10: Considerable and continuous investment is necessary to establish manufacturing capacity in LMICs to prepare for a pandemic. Early involvement of key stakeholders, such as ministries of health, in the development of guidelines for the use of influenza vaccines is crucial. The socioeconomic costs of pandemics make these types of investments worthwhile. It will be essential to form regional manufacturing hubs of sufficient scale that are sustainable to meet the demands of a pandemic response. Local facilities focused on flexible fill and finish could also expand availability while reducing long-term costs and risks. Incentives will need to be provided regionally to ensure that there is a sustainable market for these vaccines.
Economic and Market Considerations
As discussed earlier, rapid advancement of COVID-19 vaccines, especially for mRNA-based platforms, was enabled by investments from the public sector into basic research contributing to establishing the mRNA vaccine technology and investments by the private sector, where shareholders took a financial risk of pandemic mRNA vaccine development and manufacturing. At the same time, the public sector provided the market for sales of the vaccine. Substantial and immediate investment in advanced vaccine manufacturing in parallel with investment in vaccine R&D is crucial to accelerating the manufacturing process. Distinguishing between highly ef-
fective forms of public expenditure—supporting the basic scientific research enterprise, maintaining intellectual property mechanisms that encourage private investment, and creating markets for pandemic vaccines—and forms of public expenditure with more mixed track records can help ensure the most effective future expenditures of public funds for pandemic preparedness. There is a particular need for public funds to address market failures, such as the timely supply of pandemic vaccine to LMICs. Additionally, the economic viability of manufacturing facilities, especially in LMICs, during interpandemic periods needs to be addressed; otherwise the investments in these facilities could fail.
For influenza vaccines, however, there would be a significant cost barrier to developing new and novel vaccines, considering egg-based vaccines are currently a far less expensive option (Chen et al., 2020). Moreover, barriers to entry, such as FDA licensing and regulatory requirements for vaccine manufacturers, make it difficult for new actors to enter the market (Honeycutt et al., 2005). To overcome these challenges, financial investments from the public and private sectors need to be ramped up and consolidated. For instance, there is interest among private companies, such as Pfizer and Moderna, in developing mRNA vaccines directed at influenza, which was in development pre-COVID-19, and the influenza vaccine research facilitated the COVID-19 vaccine response (Moderna, 2021; Pfizer, 2021).
A conflict that remains for these new vaccine manufacturing platforms is that the question of waiving intellectual property (IP) rights has not been settled (CGD, 2021). Several potential solutions have been proposed, including patent pools (Billette de Villemeur et al., 2021; Hotez et al., 2021). There are major cost differences between the various vaccine platforms, which are defined by the material IP, as some of these vaccine platforms are relatively new and there are some proprietary issues (Krishtel and Malpani, 2021; Martin and Lowery, 2020). There are also the costs of the infrastructure needed to create the ability to manufacture these types of vaccines (Excler et al., 2021; Rosa et al., 2021). However, IP protections were key factors that supported that development of the technologies and created the willingness of the private sector to invest in COVID-19 vaccines, including mRNA-based vaccines. There is a risk of counterproductive changes to the IP system when it comes to pandemic preparedness.
Volatile political climates have led to changes in the quality of R&D, manufacturing, and investments in vaccines (Califf et al., 2020). These changes in political environments and support can disrupt the global governance of the vaccine ecosystem, leading to changes in vaccine quality requirements, varied investment commitments, and inequality in the access and distribution of vaccines and the use and acceptance of vaccines. It is essential that appropriate value propositions be designed to justify invest-
ments in vaccination research, development, and manufacturing capacity extending beyond public health benefits to include the pandemic’s major social and economic costs. The International Monetary Fund estimated the global economy lost $12 trillion in 2020–2021 as a result of the COVID-19 pandemic, a cost that could have been significantly offset with better vaccine preparedness (Academy Health, 2020).
Conclusion 4-11: Financial institutions, in conjunction with multilateral donors and private philanthropic organizations, need to recognize the potential public health and economic impacts of an influenza pandemic, hence incentivizing them to create sustainable financial vehicles and encouraging the financing of manufacturing organizations in LMICs. These investments will need to be sustained both for seasonal and pandemic influenza vaccine R&D activities and for the harmonization and mutual recognition of data and standards, equivalence agreements and quality control measures, and strengthening of regulatory and pharmacovigilance capacity.
Global Stakeholders, International Collaborations, and Networks
As recently as 2005, the influenza vaccine market consisted of only two major manufacturers and was of little interest to new actors. It took the H5N1 outbreak of the same year for the United States and other stakeholders to expand the market, largely through public–private partnerships. These partnerships allowed new products to be developed, new manufacturers to enter the market, and production capacity to expand (Neal et al., 2020). Experience with the H5N1 influenza outbreak of 2005 and the COVID-19 pandemic can be used to guide preparation for the next influenza pandemic.
The COVID-19 vaccine development experience has highlighted shifts in the roles of different actors that engage during the vaccine development process. Vaccine partnerships between developers and manufacturers to perform scale-up and scale-out in parallel with clinical development are essential. The scientific community has called for a primer for change whereby, in addition to multi-national corporations, the academic sector, small biotech companies, and the entrance of developing country manufacturers would play more prominent roles in developing, testing, and supplying vaccines for the global markets (Aars et al., 2021).
Conclusion 4-12: International collaborations and global stakeholders are essential components of ensuring support for manufacturing science. An important consideration is to ensure that global and local
manufacturing support networks exist and that the private sector is encouraged to participate in the development of manufacturing technologies. This requires providing incentives to enable and facilitate stronger, coordinated collaborations among manufacturers, primarily through contracts and commitments.
RECOMMENDATIONS
Recommendation 4-1: The U.S. Department of Health and Human Services and the World Health Organization should develop a plan for a sufficient and self-sustainable global supply of influenza vaccines for pandemics. This includes
- Convening, supporting, and encouraging multi-national, public, and private vaccine manufacturers to benchmark, prioritize, and harmonize influenza vaccine manufacturing; and
- Enhancing and expanding support of the global influenza vaccine manufacturing network, creating manufacturing hubs for greater collaboration, and building capacity to address challenges in manufacturing in low- and middle-income countries.
Recommendation 4-2: Vaccine manufacturers should take a risk-based approach to pandemic influenza preparedness. This approach would be most effective if incentivized, and could include
- Participating during research and development, data sharing, technology adoption, and training activities with international partners;
- Expanding internal capacity to assess the production needs and their risks;
- Using scientific evidence to design strategies to reduce risks (e.g., World Health Organization prequalification, licensing, and marketing); and
- Formalizing technology transfer (scale-up and scale-out) activities, taking into consideration timelines and the outcomes for equitable costs, access, and distribution.
REFERENCES
Aars, O. K., M. Clark, and N. Schwalbe. 2021. Increasing efficiency in vaccine production: A primer for change. Vaccine: X 8:100104.
AcademyHealth. 2020. Research insights: The economics of vaccine development and supply: Responding to COVID-19 and preparing for the next threat. https://academyhealth.org/sites/default/files/ah_ri_economics_of_vaccines_brief_final.pdf (accessed October 19, 2021).
Aguilar-Yáñez, J. M., R. Portillo-Lara, G. I. Mendoza-Ochoa, S. A. García-Echauri, F. LópezPacheco, D. Bulnes-Abundis, J. Salgado-Gallegos, I. M. Lara-Mayorga, Y. Webb-Vargas, F. O. León-Angel, R. E. Rivero-Aranda, Y. Oropeza-Almazán, G. M. Ruiz-Palacios, M. I. Zertuche-Guerra, R. M. DuBois, S. W. White, S. Schultz-Cherry, C. J. Russell, and M. M. Alvarez. 2010. An influenza A/H1N1/2009 hemagglutinin vaccine produced in Escherichia coli. PLOS ONE 5(7):e11694.
Anand, P., and V. P. Stahel. 2021. The safety of COVID-19 mRNA vaccines: A review. Patient Safety in Surgery 15(1):20.
AstraZeneca. 2020a. AstraZeneca and Oxford University announce landmark agreement for COVID-19 vaccine. https://www.astrazeneca.com/media-centre/press-releases/2020/astrazeneca-and-oxford-university-announce-landmark-agreement-for-covid-19-vaccine.html (accessed October 19, 2021).
AstraZeneca. 2020b. AstraZeneca takes next steps towards broad and equitable access to Oxford University’s potential COVID-19 vaccine. Cambridge, UK: AstraZeneca.
Baker, S. 2020. COVID vaccine front-runner is months ahead of her competition. Bloomberg Businessweek. July 14.
Barr, I. G., R. O. Donis, J. M. Katz, J. W. McCauley, T. Odagiri, H. Trusheim, T. F. Tsai, and D. E. Wentworth. 2018. Cell culture-derived influenza vaccines in the severe 2017–2018 epidemic season: A step towards improved influenza vaccine effectiveness. NPJ Vaccines 3:44.
Bender, E. 2019. Accelerating flu protection. Nature 573(7774):S60–S61.
Berkley, S. 2021. Why we must improve vaccine manufacturing before the next pandemic. TIME. June 9.
Billette de Villemeur, E., V. Dequiedt, and B. Versaevel. 2021. Pool patents to get COVID vaccines and drugs to all. Nature 591(7851):529.
BioNTech. 2018. BioNTech signs collaboration agreement with Pfizer to develop mRNA-based vaccines for prevention of influenza. Mainz, Germany: BioNTech. August 16, 2018.
Buckland, B. C. 2015. The development and manufacture of influenza vaccines. Human Vaccines & Immunotherapeutics 11(6):1357–1360.
Califf, R., S. Gottlieb, M. Hamburg, J. Henney, D. Kessler, M. McClellan, and A. von Eshenbach. 2020. 7 former FDA commissioners: The Trump administration is undermining the credibility of the FDA. The Washington Post. October 2.
CEA (Council of Economic Advisers). 2019. Mitigating the impact of pandemic influenza through vaccine innovation. Washington, DC: White House.
CGD (Center for Global Development). 2021. Would exempting COVID-19 vaccines from intellectual property rights improve global access and equity? https://www.cgdev.org/debate/would-exempting-covid-19-vaccines-intellectual-property-rights-improve-global-access (accessed October 19, 2021).
Chen, J.-R., Y.-M. Liu, Y.-C. Tseng, and C. Ma. 2020. Better influenza vaccines: An industry perspective. Journal of Biomedical Science 27(1):33.
Cision PR Newswire. 2020. Seqirus announces U.S. FDA approval of its first-ever adjuvanted, cell-based pandemic influenza a (h5n1) vaccine. https://www.prnewswire.com/news-releases/seqirus-announces-us-fda-approval-of-its-first-ever-adjuvanted-cell-based-pandemic-influenza-a-h5n1-vaccine-300997595.html?tc=eml_cleartime (accessed October 19, 2021).
Corbett, K. S., D. Edwards, S. R. Leist, O. M. Abiona, S. Boyoglu-Barnum, R. A. Gillespie, S. Himansu, A. Schäfer, C. T. Ziwawo, A. T. DiPiazza, K. H. Dinnon, S. M. Elbashir, C. A. Shaw, A. Woods, E. J. Fritch, D. R. Martinez, K. W. Bock, M. Minai, B. M. Nagata, G. B. Hutchinson, K. Bahl, D. Garcia-Dominguez, L. Ma, I. Renzi, W.-P. Kong, S. D. Schmidt, L. Wang, Y. Zhang, L. J. Stevens, E. Phung, L. A. Chang, R. J. Loomis, N. E. Altaras, E. Narayanan, M. Metkar, V. Presnyak, C. Liu, M. K. Louder, W. Shi, K. Leung, E. S. Yang,
A. West, K. L. Gully, N. Wang, D. Wrapp, N. A. Doria-Rose, G. Stewart-Jones, H. Bennett, M. C. Nason, T. J. Ruckwardt, J. S. McLellan, M. R. Denison, J. D. Chappell, I. N. Moore, K. M. Morabito, J. R. Mascola, R. S. Baric, A. Carfi, and B. S. Graham. 2020. SARS-CoV-2 mRNA vaccine development enabled by prototype pathogen preparedness. bioRxiv 2020.2006.2011.145920.
Crommelin, D. J. A., T. J. Anchordoquy, D. B. Volkin, W. Jiskoot, and E. Mastrobattista. 2021. Addressing the cold reality of mRNA vaccine stability. Journal of Pharmaceutical Sciences 110(3):997–1001.
Duan, L., Q. Zheng, H. Zhang, Y. Niu, Y. Lou, and H. Wang. 2020. The SARS-CoV-2 spike glycoprotein biosynthesis, structure, function, and antigenicity: Implications for the design of spike-based vaccine immunogens. Frontiers in Immunology 11(2593).
DuBois, R. M., J. M. Aguilar-Yañez, G. I. Mendoza-Ochoa, Y. Oropeza-Almazán, S. Schultz-Cherry, M. M. Alvarez, S. W. White, and C. J. Russell. 2011. The receptor-binding domain of influenza virus hemagglutinin produced in escherichia coli folds into its native, immunogenic structure. Journal of Virology 85(2):865–872.
Excler, J.-L., M. Saville, S. Berkley, and J. H. Kim. 2021. Vaccine development for emerging infectious diseases. Nature Medicine 27(4):591–600.
Figueroa, J. P., M. E. Bottazzi, P. Hotez, C. Batista, O. Ergonul, S. Gilbert, M. Gursel, M. Hassanain, J. H. Kim, B. Lall, H. Larson, D. Naniche, T. Sheahan, S. Shoham, A. Wilder-Smith, N. Strub-Wourgaft, P. Yadav, and G. Kang. 2021. Urgent needs of low-income and middle-income countries for COVID-19 vaccines and therapeutics. Lancet 397(10274):562–564.
G20 (G20 High-Level Independent Panel). 2021. A global deal for our pandemic age. https://pandemic-financing.org/wp-content/uploads/2021/07/Four-Major-Gaps.pdf (accessed October 19, 2021).
GAO (U.S. Government Accountability Office). 2021. Operation Warp Speed: Accelerated COVID-19 vaccine development status and efforts to address manufacturing challenges. https://www.gao.gov/products/gao-21-319 (accessed October 19, 2021).
Gavi (The Vaccine Alliance). 2021a. COVAX. https://www.gavi.org/covax-facility (accessed October 19, 2021).
Gavi. 2021b. Facts and figures. https://www.gavi.org/programmes-impact/our-impact/facts-and-figures (accessed October 19, 2021).
Grand View Research. 2020. COVID-19 vaccines market production capacity & development timeline: (by type: Virus-like particle (VLP), whole virus, protein, antibodies, viral vector, mRNA/DNA). https://www.grandviewresearch.com/industry-analysis/covid-19-vaccines-market (accessed October 19, 2021).
Harrington, W. N., C. M. Kackos, and R. J. Webby. 2021. The evolution and future of influenza pandemic preparedness. Experimental & Molecular Medicine 53(5):737–749.
Hatchett, R., M. Saville, M. Downham, T. Cueni, L. Bigger, P. Arthur, R. Suri, S. D. Prasad, and R. Bech Hansen. 2021. Towards vaccinating the world: Landscape of current COVID-19 supply chain and manufacturing capacity, potential challenges, initial responses, and possible “solution space”—a discussion document. https://www.ifpma.org/wp-content/uploads/2021/03/Summit_Landscape_Discussion_Document.pdf (accessed October 19, 2021).
Hay, A. J., and J. W. McCauley. 2018. The WHO Global Influenza Surveillance and Response System (GISRS)—a future perspective. Influenza and Other Respiratory Viruses 12(5):551–557.
Hendriks, J., M. Holleman, O. de Boer, P. de Jong, and W. Luytjes. 2011. An international technology platform for influenza vaccines. Vaccine 29:A8–A11.
HHS (U.S. Department of Health and Human Services). 2020a. National Influenza Vaccine Modernization Strategy 2020–2030. https://www.phe.gov/Preparedness/planning/nivms/Documents/nivms-2020-2030.pdf (accessed October 19, 2021).
HHS. 2020b. Explaining Operation Warp Speed. https://www.nihb.org/covid-19/wp-content/uploads/2020/08/Fact-sheet-operation-warp-speed.pdf (accessed October 19, 2021).
Honeycutt, A., T. Robinson, and C. Layton. 2005. Influenza vaccine economics. Triangle Park, NC: Research Triangle Institute.
Hotez, P. J., M. E. Bottazzi, and P. Yadav. 2021. Producing a vaccine requires more than a patent. Foreign Affairs. May 10.
Hoyt, K., and T. Johnston. 2021. A plan to break the vaccine manufacturing bottleneck. https://www.barrons.com/amp/articles/a-plan-to-break-the-vaccine-manufacturing-bottleneck-51621952245 (accessed October 19, 2021).
Hyder, A. A., M. A. Hyder, K. Nasir, and P. Ndebele. 2021. Inequitable COVID-19 vaccine distribution and its effects. Bulletin of the World Health Organization 99(6):406.
IRBM Group. 2020. The Jenner Institute signs an agreement with Advent to develop a novel coronavirus vaccine. https://www.irbm.com/news/the-jenner-institute-signs-an-agreement-with-advent-to-develop-a-novel-coronavirus-vaccine (accessed October 19, 2021).
Irwin, A. 2021. What it will take to vaccinate the world against COVID-19? Nature 592(7853):176–178.
Kis, Z., and Z. Rizvi. 2021. How to make enough vaccine for the world in one year. https://www.citizen.org/article/how-to-make-enough-vaccine-for-the-world-in-one-year (accessed October 19, 2021)
Knobler, S., C. Howell, M. Malchione, B. McCormick, and A. Mack. 2020. Lessons from COVID-19: Advancing development of universal influenza vaccines. Washington, DC: Sabin Vaccine Institute.
Krishtel, P., and R. Malpani. 2021. Suspend intellectual property rights for COVID-19 vaccines. BMJ 373:n1344.
Lemoine, C. H., R. V. Nidom, R. Ventura, S. Indrasari, I. Normalina, K. P. Santoso, F. Derouet, C. Barnier-Quer, G. Borchard, N. Collin, and C. A. Nidom. 2021. Better pandemic influenza preparedness through adjuvant technology transfer: Challenges and lessons learned. Vaccines 9(5):461.
Martin, C., and D. Lowery. 2020. mRNA vaccines: Intellectual property landscape. Nature Reviews Drug Discoveries 19(9):578.
Matthews, J. T. 2006. Egg-based production of influenza vaccine: 30 years of commercial experience. The Bridge 36(3).
Merck. 2021. Merck to help produce Johnson & Johnson’s COVID-19 vaccine; BARDA to provide Merck with funding to expand Merck’s manufacturing capacity for COVID-19 vaccines and medicines. Kenilworth, NJ: Merck. March 2.
Moderna. 2021. Moderna provides business update and announces three new development programs in infectious disease vaccines. https://investors.modernatx.com/news-releases/news-release-details/moderna-provides-business-update-and-announces-three-new (accessed October 19, 2021).
Murugan, S., S. Ponsekaran, L. Kannivel, L. N. Mangamoori, D. Chandran, S. Villuppanoor Alwar, C. Chakravarty, and S. K. Lal. 2013. Recombinant haemagglutinin protein of highly pathogenic avian influenza A(H5N1) virus expressed in Pichia pastoris elicits a neutralizing antibody response in mice. Journal of Virological Methods 187(1):20–25.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2019. Exploring lessons learned from a century of outbreaks: Readiness for 2030: Proceedings of a workshop. Washington, DC: The National Academies Press.
Neal, J. C., K. Cappio, G. Woollett, R. Hughes IV, C. Chepigin, and Z. Klein. 2020. Insights from the influenza vaccine market for COVID-19 vaccines. https://avalere.com/insights/insights-from-the-influenza-vaccine-market-for-covid-19-vaccines (accessed October 19, 2021).
Ng, S. K. 2012. Current cell-based influenza vaccine production technology as pandemic contingency. Human Vaccines & Immunotherapeutics 8(2):267–271.
Novartis. 2021. Novartis signs initial agreement to provide manufacturing capacity for Pfizer-BioNTech COVID-19 vaccine. Basel, Switzerland: Novartis. January 29.
Our World in Data. 2021. Coronavirus (COVID-19) vaccinations. https://ourworldindata.org/covid-vaccinations#citation (accessed October 19, 2021).
Paden, C. R., Y. Tao, K. Queen, J. Zhang, Y. Li, A. Uehara, and S. Tong. 2020. Rapid, sensitive, full-genome sequencing of severe acute respiratory syndrome coronavirus 2. Emerging Infectious Diseases 26(10):2401–2405. https://doi.org/10.3201/eid2610.201800.
Palache, A., A. Abelin, R. Hollingsworth, W. Cracknell, C. Jacobs, T. Tsai, and P. Barbosa. 2017. Survey of distribution of seasonal influenza vaccine doses in 201 countries (2004–2015): The 2003 World Health Assembly resolution on seasonal influenza vaccination coverage and the 2009 influenza pandemic have had very little impact on improving influenza control and pandemic preparedness. Vaccine 35(36):4681–4686.
Palkonyay, L., and H. Fatima. 2016. A decade of adaptation: Regulatory contributions of the World Health Organization to the global action plan for influenza vaccines (2006–2016). Vaccine 34(45):5414–5419.
Paris, C. 2020. Supply-chain obstacles led to last month’s cut to Pfizer’s COVID-19 vaccine-rollout target. The Wall Street Journal. December 3.
PATH. 2019. Supporting vaccine manufacturers to supply critical vaccines. https://www.path.org/articles/supporting-vaccine-manufacturers-supply-critical-vaccines (accessed October 19, 2021).
Pfizer. 2020. Pfizer and BioNTech to co-develop potential COVID-19 vaccine. March 17. https://investors.pfizer.com/investor-news/press-release-details/2020/Pfizer-and-BioNTech-to-Co-Develop-Potential-COVID-19-Vaccine/default.aspx (accessed October 19, 2021).
Pfizer. 2021. New RNA technology could get the flu vaccine right, every year. https://www.pfizer.com/news/featured_stories/featured_stories_detail/new_rna_technology_could_get_the_flu_vaccine_right_every_year (accessed October 19, 2021).
Plotkin, S., J. M. Robinson, G. Cunningham, R. Iqbal, and S. Larsen. 2017. The complexity and cost of vaccine manufacturing - an overview. Vaccine 35(33):4064–4071.
Rajaram, S., C. Boikos, D. K. Gelone, and A. Gandhi. 2020. Influenza vaccines: The potential benefits of cell-culture isolation and manufacturing. Therapeutic Advances in Vaccines and Immunotherapy 8:2515135520908121.
Rappuoli, R., E. De Gregorio, G. Del Giudice, S. Phogat, S. Pecetta, M. Pizza, and E. Hanon. 2021. Vaccinology in the post−COVID-19 era. Proceedings of the National Academy of Sciences 118(3):e2020368118.
Rele, S. 2021. COVID-19 vaccine development during pandemic: Gap analysis, opportunities, and impact on future emerging infectious disease development strategies. Human Vaccines & Immunotherapeutics 17(4):1122–1127.
Rockman, S., K. Laurie, and I. Barr. 2020. Pandemic influenza vaccines: What did we learn from the 2009 pandemic and are we better prepared now? Vaccines 8(2):211.
Rosa, S. S., D. M. F. Prazeres, A. M. Azevedo, and M. P. C. Marques. 2021. mRNA vaccines manufacturing: Challenges and bottlenecks. Vaccine 39(16):2190–2200.
Saelens, X., P. Vanlandschoot, W. Martinet, M. Maras, S. Neirynck, R. Contreras, W. Fiers, and W. M. Jou. 1999. Protection of mice against a lethal influenza virus challenge after immunization with yeast-derived secreted influenza virus hemagglutinin. European Journal of Biochemistry 260(1):166–175.
Sanofi. 2021. Sanofi to provide support to BioNTech in manufacturing their COVID-19 vaccine to help address public health needs. Paris, France: Sanofi. January 27.
Schmidt, C. 2021. New COVID vaccines need absurd amounts of material and labor. Scientific American. January 4.
Shang, Y., H. Li, and R. Zhang. 2021. Effects of pandemic outbreak on economies: Evidence from business history context. Frontiers in Public Health 9(146).
Soema, P. C., R. Kompier, J.-P. Amorij, and G. F. A. Kersten. 2015. Current and next generation influenza vaccines: Formulation and production strategies. European Journal of Pharmaceutics and Biopharmaceutics 94:251–263.
Sparrow, E., J. G. Wood, C. Chadwick, A. T. Newall, S. Torvaldsen, A. Moen, and G. Torelli. 2021. Global production capacity of seasonal and pandemic influenza vaccines in 2019. Vaccine 39(3):512–520.
Tenforde, M. W., S. M. Olson, W. H. Self, H. K. Talbot, C. J. Lindsell, J. S. Steingrub, N. I. Shapiro, A. A. Ginde, D. J. Douin, M. E. Prekker, S. M. Brown, I. D. Peltan, M. N. Gong, A. Mohamed, A. Khan, M. C. Exline, D. C. Files, K. W. Gibbs, W. B. Stubblefield, J. D. Casey, T. W. Rice, C. G. Grijalva, D. N. Hager, A. Shehu, N. Qadir, S. Y. Chang, J. G. Wilson, M. Gaglani, K. Murthy, N. Calhoun, A. S. Monto, E. T. Martin, A. Malani, R. K. Zimmerman, F. P. Silveira, D. B. Middleton, Y. Zhu, D. Wyatt, M. Stephenson, A. Baughman, K. N. Womack, K. W. Hart, M. Kobayashi, J. R. Verani, and M. M. Patel. 2021. Effectiveness of Pfizer-BioNTech and Moderna vaccines against COVID-19 among hospitalized adults aged ≥65 years—United States, January–March 2021. Morbidity and Mortality Weekly Report 70(18):674–679.
WHO (World Health Organization). 2019. Global influenza strategy 2019–2030. Geneva, Switzerland: World Health Organization.
WHO. 2021a. Establishment of a COVID-19 mRNA vaccine technology transfer hub to scale up global manufacturing. https://www.who.int/news-room/articles-detail/establishment-of-a-covid-19-mrnavaccine-technology-transfer-hub-to-scale-up-global-manufacturing (accessed October 19, 2021).
WHO. 2021b. The effects of virus variants on COVID-19 vaccines. https://www.who.int/news-room/feature-stories/detail/the-effects-of-virus-variants-on-covid-19-vaccines (accessed October 19, 2021).
Williams, S. R., A. J. Driscoll, H. M. LeBuhn, W. H. Chen, K. M. Neuzil, and J. R. Ortiz. 2021. National routine adult immunisation programmes among World Health Organization member states: An assessment of health systems to deploy COVID-19 vaccines. Eurosurveillance 26(17):2001195.
Woolston, C. 2021. “Does anyone have any of these?”: Lab-supply shortages strike amid global pandemic. Nature. https://pubmed.ncbi.nlm.nih.gov/33750928 (accessed October 19, 2021).