2
Clinical Practice Guidelines and the U.S. Preventive Services Task Force
WHAT ARE CLINICAL PRACTICE GUIDELINES?
Since their proliferation in clinical settings in the United States in the late 1970s, clinical practice guidelines (CPGs) have evolved, and there is a preference for those that explicitly emphasize the use of the evidence base when possible (IOM, 2011; Weisz et al., 2007). In 2011, the Institute of Medicine defined CPGs as “statements that include recommendations intended to optimize patient care that are informed by a systematic review of evidence and an assessment of the benefits and harms of alternative care options” (IOM, 2011, p. 15). The systematic reviews that inform CPGs tend to reveal evidence gaps as well, though CPG developers have varying approaches to addressing them. Some CPG developers explicitly limit their statements to evidence-based recommendations, while others make recommendations in the absence of sufficient evidence using alternative approaches. The U.S. Preventive Services Task Force (USPSTF) and several other guideline developers prefer the former approach.
While the committee’s statement of task included developing a taxonomy of evidence gaps and suggesting approaches to closing evidence gaps in clinical prevention in general, it explicitly named the USPSTF several times. For this reason, the committee approached the taxonomy as a tool for use beyond the Agency for Healthcare Research and Quality (AHRQ), the National Institutes of Health’s (NIH’s) Office of Disease Prevention (ODP), and the USPSTF, but kept the USPSTF in the main point of focus.
Barriers to Implementing Clinical Practice Guidelines
The USPSTF’s scope is to “make recommendations on clinical preventive services to primary care clinicians,” including screening tests, behavioral counseling, and preventive medications. The USPSTF recommendations only address services offered in primary care settings or those to which primary care clinicians can refer (USPSTF, n.d.-d). However, several contexts pose a threat to the broad dissemination and implementation of these recommendations, particularly in underserved or diverse clinical settings. There are many types of contextual barriers to implementing clinical guidelines, including but not limited to the health organization system and health professional context; guidelines context; patient context; and legal, social, and political contexts (Correa et al., 2020; Hurwitz, 1999). These barriers can also influence the equity of access to, use of, and receipt of health care.
Guidelines Context
Specific barriers to implementation in the guideline context may be simpler to resolve, but they include both lack of access or awareness of the existence of guidelines and an awareness of too many different guidelines regarding the same subject (Rauh et al., 2018). They also include beliefs regarding the credibility of the evidence base (or lack thereof), or that the recommendation is impractical to implement (Correa et al., 2020). Another guideline-related barrier includes the belief that guidelines “restrict clinical judgment, challenge professional autonomy, and limit treatment options” (Correa et al., 2020, p. 6).
Health Professional and Health Systems Context(s)
Barriers in the health professional context center on clinician attitudes and behaviors having to do with their own autonomy, their practice, their patients, or all three. Clinicians may be unable to implement or may actively avoid implementing for a variety of reasons. These include variations in priorities between clinicians and the health systems in which they work; clinicians having more confidence in their own clinical experience than in guidelines; clinician resistance to change or doubts about the efficacy of an intervention, or perceived loss of autonomy in following guidelines; or a lack of communication between clinicians regarding best practices for implementation (Correa et al., 2020).
Health systems also face barriers to implementing CPGs. Health systems may struggle with a lack of clear protocols or processes to successfully implement new guidelines, and they may lack tools or resources to provide certain services. They may also lack services in their communi-
ties to which they would refer patients (e.g., psychiatric care to which a patient could be referred after being screened for depression) (Anthony et al., 2010). New guidelines can be challenging to implement when the recommendation reduces the frequency of a current standard practice, particularly when both clinicians and patients are accustomed to the current practice. Reductions in services in particular may concern health systems that implementation will result in reduced patient visits and thus revenue (Fischer et al., 2016; IOM, 2011). Health systems may also lack resources and equipment to implement certain recommendations.
Clinicians and health systems may also feel they are too busy or overwhelmed with their current caseloads and be disinclined to incorporate additional services into their work (Fischer et al., 2016). Priorities for specific health conditions or services may also vary by health system or provider, leading to different services being offered (Harding et al., 2014).
Patient Context
The patient context is another consideration when implementing CPGs. Patients may be reluctant to request or accept suggestions for a service for a variety of reasons, including limited language, literacy, or health literacy; limited motivation to follow a certain recommendation; or concerns about affordability. They may also turn down offers of preventive services based on fears of real or perceived harms (Jones et al., 2010). Conversely, they may request a preventive service more frequently than is recommended, particularly if the suggested frequency of a service is reduced in a new recommendation statement (Allen et al., 2013). Patients may be suspicious that a reduction in frequency is a “cost-savings measure by insurers and/or providers” (Allen et al., 2013, p. 1).
Legal and Sociopolitical Context(s)
Legal and sociopolitical contexts may relate specifically to a health system organization’s or other practice’s work culture and are described above. Additional legal and sociopolitical contexts include case law related to CPGs. Some courts have upheld CPGs as national legal standards for care, while others have disregarded them in favor of local standards for care (Merenstein, 2004). This uncertainty can lead to clinicians ordering more diagnostic tests for patients than is recommended, for fear of malpractice lawsuits (IOM, 2011).
Barriers to implementing CPGs can also reflect and reinforce health disparities (Welch et al., 2017). Guideline-issuing bodies do not always consider equity when developing recommendations, possibly contributing to additional difficulties implementing them in underserved or marginalized communities (Lang et al., 2021). As mentioned earlier, the Patient Protection
and Affordable Care Act of 2010 (ACA) requires private health insurers to provide certain preventive services at no cost to patients (KFF, 2015). However, despite this provision, patients with employer-covered insurance are still paying between $75 million and $219 million annually for preventive care, according to a recent estimate by Hoagland and Shafer (2021).
U.S. PREVENTIVE SERVICES TASK FORCE
History and Scope
In 1984, the U.S. Department of Health and Human Services (HHS) assistant secretary Edward N. Brandt commissioned the USPSTF to “improve the health of people nationwide by making evidence-based recommendations about clinical preventive services” (USPSTF, 2021a). The USPSTF was assembled as an effort to continue and expand the innovative work of the Canadian Task Force on the Periodic Health Examination (CTFPHE), which was founded in 1976 and began issuing preventive service recommendations in the late 1970s (CTFPHE, 1979; McGinnis and Woolf, 1990). The CTFPHE’s report laid out the basic elements of preventive evidence assessment as they lead to clinical recommendations. These included comprehensively reviewing the literature; prioritizing methods for assessing individual interventions, including determining the burden of suffering and ability to measure outcomes; understanding the risks and benefits of an intervention; rating the effectiveness of interventions; grading the level of evidence for a recommendation; and defining a research agenda to address unanswered questions (CTFPHE, 1979). As an independent, volunteer panel of 20 experts in clinical prevention and evidence-based medicine,1 the USPSTF sought to examine “more current issues in preventive medicine and more recent evidence not available to [the CTFPHE] and to move the debate forward into new areas not previously addressed” (McGinnis and Woolf, 1990, p. S12). Its objectives were to develop risk factor–specific recommendations for CPGs, address behavioral and structural barriers to prevention, and develop a prevention research agenda (McGinnis and Woolf, 1990).
Because the USPSTF adopted the methodology used by the CTFPHE, the two bodies were able to collaborate “in a binational effort to review evidence and develop recommendations on preventive services,” (USPSTF, 1989) sending and receiving members to each other’s meetings (McGinnis and Woolf, 1990). In 1989, the USPSTF published its first report on CPGs, Guide to Clinical Preventive Services: An Assessment of the Effectiveness of 169 Interventions, and was disbanded (USPSTF, 1989).
___________________
1 The USPSTF currently comprises 16 members each serving 4-year terms.
The second task force was assembled in September 1990 and met through April 1994 (USPSTF, 1996). The task force received support from HHS’s Office of Disease Prevention and Health Promotion and Agency for Healthcare Policy and Research (AHCPR) to “analyze systematically scientific evidence pertaining to clinical preventive services that had been published since the first edition of the Guide” (USPSTF, 1996, p. xxix). In 1999, the U.S. Congress reauthorized AHCPR and renamed it the Agency for Healthcare Research and Quality (AHRQ). The legislation also states that the USPSTF
shall review the scientific evidence related to the effectiveness, appropriateness, and cost-effectiveness of clinical preventive services for the purpose of developing recommendations for the health care community, and updating previous clinical preventive recommendations.2
However, despite having the authority to do so, the USPSTF does not evaluate evidence related to cost-effectiveness when issuing a grade for a topic:
[USPSTF] excludes costs from its determination of the benefits and harms of a clinical preventive service. This deliberate decision was made to maintain a clear focus on the science of clinical effectiveness (i.e., “what works”), and not consider cost, in part to avoid any misperception that the Task Force’s purpose is to limit health care based on cost…. The Task Force recognizes that cost is a consideration for insurers, health systems, purchasers, and consumers. The USPSTF maintains that regardless of how health care is financed, it is essential to understand what the science says about which preventive services work. (USPSTF, 2021c)
Congress also authorized the AHRQ director to convene and appoint members to the USPSTF, and authorized AHRQ to “provide ongoing administrative, research, and technical support for [USPSTF] operations, including coordinating and supporting the dissemination of [its] recommendations.”3 AHRQ published the third and final edition of the original Guide in 2002 (USPSTF, 2002). By 2005, AHRQ had been publishing the USPSTF’s recommendations as they were issued for each topic online, as well as “pocket guides” with abridged recommendations the
___________________
2 106th U.S. Congress, First Session. Public Law 106-129, Section 915. Healthcare Research and Quality Act of 1999. https://www.ahrq.gov/sites/default/files/wysiwyg/policymakers/hrqa99.pdf (accessed July 20, 2021).
3 106th U.S. Congress, First Session. Public Law 106-129, Section 915. Healthcare Research and Quality Act of 1999. https://www.ahrq.gov/sites/default/files/wysiwyg/policymakers/hrqa99.pdf (accessed July 20, 2021).
task force had issued individually in the preceding years (USPSTF, 2005, 2007a, 2009, 2010, 2012, 2014a).
U.S. Preventive Services Task Force Process
The process of selecting a topic for review through publishing the final recommendation statement takes 3 years on average (Borsky, 2021). Presently, the USPSTF process for reviewing and developing recommendations begins with soliciting topics for nomination (USPSTF, 2021d) (see Figure 2-1). Any individual or organization can nominate a topic at any time; the USPSTF identifies those that are within its scope and prioritizes each one with the input of its stakeholders and partner liaisons. The task force then works with one of the Evidence-based Practice Centers (EPCs) supported by AHRQ to develop a draft research plan, which is posted for public comment. After the USPSTF and the EPC review and
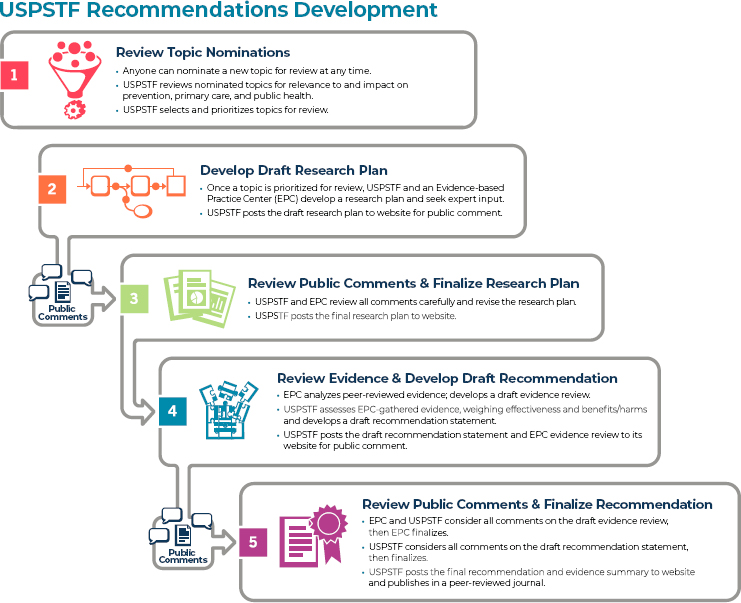
SOURCE: USPSTF, 2021d.
revise as needed, the final plan is posted online. The EPC conducts the systematic review according to the research plan, and the USPSTF begins assessing the evidence after the EPC sends its draft review. The USPSTF then drafts a recommendation statement and posts it online for public comment, and after a review of public comments the EPC finalizes its evidence review. The USPSTF then finalizes its recommendation statement, and both are posted online, and as of 2016, published in a journal of the JAMA Network (Bauchner et al., 2016). The USPSTF also considers previously graded topics for updates starting 2–3 years after they are first published, first soliciting stakeholder input and examining the likelihood that enough new evidence has been developed that the recommendation for practice may be updated.
The development of recommendations for preventive services “typically requires deliberation using comprehensive systematic reviews of the best available evidence and a series of complex judgments” (USPSTF, 2019). The USPSTF assessment of the overall evidence for a clinical preventive service requires two key determinations: “assessment of the degree of certainty about the net benefit (benefits minus harms) and assessment of the magnitude of net benefit for the population for whom the service is intended” (for a visual of this matrix, see Table 2-1) (USPSTF, 2019).
Grading Scheme
The USPSTF last updated its methods for understanding certainty and net benefit and linking intermediate outcomes and health outcomes in prevention in 2018 (USPSTF, 2018). The USPSTF updated its grading scheme most recently in 2012 and prior to that in 2007 (USPSTF, 2018). Currently, grades A and B indicate that the task force recommends an intervention, grade C indicates selectively offering a service, and grade D indicates that the task force recommends against implementing a pre-
| Certainty of Net Benefit | Magnitude of Net Benefit | |||
|---|---|---|---|---|
| Substantial | Moderate | Small | Zero/Negative | |
| High | A | B | C | D |
| Moderate | B | B | C | D |
| Low | Insufficient (I Statement) | |||
ventive service. The task force makes these assessments by evaluating the evidence using the following steps:
- Assessing the adequacy of evidence at the key question level.
- Assessing the adequacy of evidence at the linkage level.
- Estimating the magnitude of benefit and harm of the preventive service.
- Evaluating the certainty of the evidence for net benefit for the preventive service.
- Estimating the magnitude of the net benefit of the preventive service.
- Developing a recommendation grade for the preventive service in the relevant population, based on the above parameters (USPSTF, 2017a).
Evidence Gaps and I Statements
While the recommendations for or against a clinical service receive a grade, some topics are given I statements, indicating that the USPSTF has not found sufficient evidence to make a recommendation at all. In 2009, the task force announced that it would shift from using the phrase “I recommendation” to “I statement” to reduce confusion among clinicians about whether a topic with an I was recommended or not (USPSTF, 2017b). Topics can receive an I statement instead of a graded recommendation due to lack of evidence, a lack of good quality evidence, or an abundance of conflicting evidence. Without clear or good quality evidence, the USPSTF cannot determine the balance of benefits and harms and does not make a recommendation about the provision of the service. As of the publication of this report, 53 topics have I statements either for a specific subpopulation or for recommending the service at all (see Appendix A). On average, 8.4 years elapse before a preventive service with an I statement is issued a definitive grade recommendation by the USPSTF, if it is to receive one at all (Klabunde et al., 2021).
While I statement text is usually centered around identifying the missing evidence that must be developed in order to update a topic with a letter grade, recommendations with letter grades also identify evidence gaps and research needs either to reinforce or change recommendations or to generally advance prevention research on the topic. Since 1989, USPSTF recommendations have suggested evidence gaps or research needs for various topics in one way or another, but in 2007, the USPSTF introduced a new section of its recommendation statements called “Other Considerations.” This section could include cost and dissemination and
implementation considerations for clinicians and policymakers, as well as “key gaps in the evidence and … priorities for future research and research funding” (Barton et al., 2007, p. 125). In 2019, however, another methods update restructured the section to give “Research Needs and Gaps” its own section. The details of those sections vary widely in specificity and scope. See Appendix F.
A small number of the USPSTF’s 129 recommendations are currently marked as “archived,” “inactive,” or “referred” and refer users to other guideline developers. Two refer users to the Advisory Committee on Immunization Practice (for information on adult and child immunization practices) (USPSTF, n.d.-a, n.d.-b), and two refer users to the Discretionary Advisory Committee on Heritable Disorders in Newborns and Children (for information related to phenylketonuria and sickle cell disease screenings) (USPSTF, 2008, n.d.-c). The other two refer users to the Community Guide website (for information related to using seatbelts and preventing motor vehicle injury, and preventing youth violence) (USPSTF, 2007b, 2014b).
This committee was charged with the task of developing a taxonomy to characterize types of evidence gaps and research needs in clinical prevention research, particularly as they relate to topics considered by the USPSTF. When reviewing a preventive service, the USPSTF may find there is insufficient evidence to assess whether the service (or certain individuals potentially receiving a preventive service) should be recommended for a variety of reasons. For example, insufficient evidence may relate to limited effectiveness of interventions; questionable value of early intervention; conflicting evidence of existing studies; limited treatment evidence in lower-risk subgroups; subclinical vs clinical endpoints; and variable impact of screen-detected disease (Atkins, 2021). For examples of each, see Table 2-2.
New evidence reviewed to update a topic with an I statement may also not be enough to upgrade it to receive a letter grade recommendation. The ACA requires the USPSTF and the Community Preventive Services Task Force (CPSTF) to provide annual reports to Congress, outlining major evidence gaps in clinical prevention research and highlighting high-priority areas or topics that “deserve further examination” (USPSTF, 2020). Appendix E contains a list of all reports to Congress. Some research priority topics in recent years identified by the USPSTF have included adolescent health and health inequities, substance use, violence prevention, cancer prevention, and cardiovascular health. Since the USPSTF submitted its first annual report to Congress, some topics that originally had I statements no longer do, while others have had two or three updates with no change. For example, new evidence upgraded a recommendation
TABLE 2-2 Types of Evidence Gaps That May Lead to an I Statement for a Topic
| Type of Gap | Example |
|---|---|
| Limited effectiveness of interventions | Tobacco cessation in teens Cognitive impairment in older adults (or age bracket) Suicide screening |
| Questionable value of earlier intervention | Vision screening in older adults |
| Conflicting evidence of existing studies | Calcium/vitamin D for fracture prevention |
| Limited treatment evidence in lower risk subgroups | AAA screening in women Osteoporosis in men |
| Subclinical versus clinical endpoints | Glaucoma treatment Osteoporosis treatment in men |
| Variable impact of screen-detected disease | Thyroid dysfunction Iron-deficiency |
NOTE: AAA = abdominal aortic aneurysm.
SOURCES: Adapted from a presentation by David Atkins on March 16, 2021; Atkins, 2021.
around hearing loss in newborns from an I in 2001 to a B in 2008.4 Additionally, the recommendation that clinicians screen children for obesity and offer or refer them to comprehensive, intensive behavioral intervention to promote improvement in weight status received an I statement in 2004 and was upgraded to a B in 2010, due to a “number of new studies” (USPSTF, 2011). In contrast, some I statements no longer exist because new evidence has changed the key questions about the preventive service itself. For example, in 2008, computed tomographic colonography and fecal DNA testing were a type of screening for colorectal cancer given an I statement by the USPSTF. When the USPSTF updated the colorectal cancer screening in 2016, new evidence about the harms and benefits of those screenings had already emerged, and the USPSTF updated its recommendation so that it no longer included an I statement or separate grade for those specific screenings. Computed tomographic colonography and fecal DNA testing were included in a list of other screenings that were recommended for certain age groups (regarding screening for colorectal cancer, adults aged 50–75 years received an A grade recommendation; adults aged 45–49 years received a B grade recommendation; and adults aged 76–86 years received a C grade recommendation).
Revisiting I statements, however, can engender substantial costs in time and fiscal resources, often without important yields in improved
___________________
4 This recommendation has since been archived.
guidelines that will ultimately elevate community health status levels. There is also the problem of opportunity costs for selected USPSTF activities, as there may be nascent but promising interventions appearing that require attention. In addition, evidence gaps in I statements that are not well described can result in resources (time and money) squandered on research that will not help turn the I statement into a letter grade recommendation.
U.S. Preventive Services Task Force’s Relationship with the Community Preventive Services Task Force
HHS established the CPSTF in 1996 to provide findings and recommendations about community preventive services, programs, and other interventions aimed at improving population health, and to complement the work of the USPSTF (CPSTF, n.d.-a). The collection of recommendations and findings made by the CPSTF are referred to as the Community Guide. As AHRQ supports the USPSTF, the Centers for Disease Control and Prevention (CDC) provides scientific and administrative support to the CPSTF. AHRQ and CDC staff members who support their respective task forces typically serve as partner liaisons for the other task force, attending the other’s meetings regularly and occasionally presenting their relevant work. Many of the CPSTF organizational and methodological characteristics derive directly from those of the USPSTF (Briss et al., 2000; Truman et al., 2000). The CPSTF is also guided by the Healthy People Initiative, which identifies population targets for improved public health outcomes in advance of each decade (NASEM, 2020).
The USPSTF and the CPSTF both collaborate with ODP to communicate with NIH’s institutes and centers (ICs) regarding evidence gaps and research needs identified in their evidence assessments. ODP disseminates newer I statements or insufficient evidence statements that have been issued by the task forces and collects information from the ICs about the relevant research they have funded (ODP, 2021). The CPSTF methodology contrasts with that of the USPSTF by focusing on interventions at the population level as opposed to the individual level, and thus more frequently evaluating a body of evidence in which “non-randomized studies commonly have behavioral outcomes as endpoints” (Grossman and Elder, 2015, p. 3). However, the more interactive complementary nature of the two task forces allows for the interventions recommended by the CPSTF to support the implementation and delivery of interventions recommended by the USPSTF. For example, the USPSTF recommends screening for colon and rectal cancer among adults aged 45–75 years, and the CPSTF recommends a multitude of different interventions to increase certain types of colon and rectal cancer screenings, including
using reminder and recall systems, engaging community health workers, and reducing structural barriers for clients (CPSTF, 2005, 2006, 2009, 2010a,b,c, 2016, 2019; USPSTF, 2021b). The USPSTF’s and the CPSTF’s complementary work is visualized in Figure 2-2.
Like its methodology, the CPSTF’s grades for findings differ from those of the USPSTF. The CPSTF will recommend for or against an intervention, and the strength of evidence is rated as “strong” or “sufficient.” A crucial difference is that the USPSTF grades connote varying degrees of the magnitude of net benefit and the degree of certainty about it, while the CPSTF findings category of “strong” or “sufficient” is a direct evaluation of the quality of the evidence base and not a reflection of the expected magnitude of benefits (CPSTF, n.d.-c). The CPSTF may also find that an intervention has IE. An intervention may be given an IE grade if there is a lack of research available or inconsistent results that prevent the CPSTF from determining if an intervention should be recommended at all. The CPSTF has turned each IE finding into factsheets that are publicly available on its website for use by public health practitioners, researchers, and funders (CPSTF, n.d.-b). When describing evidence gaps in recommended topics, the CPSTF refers users specifically to the systematic review that
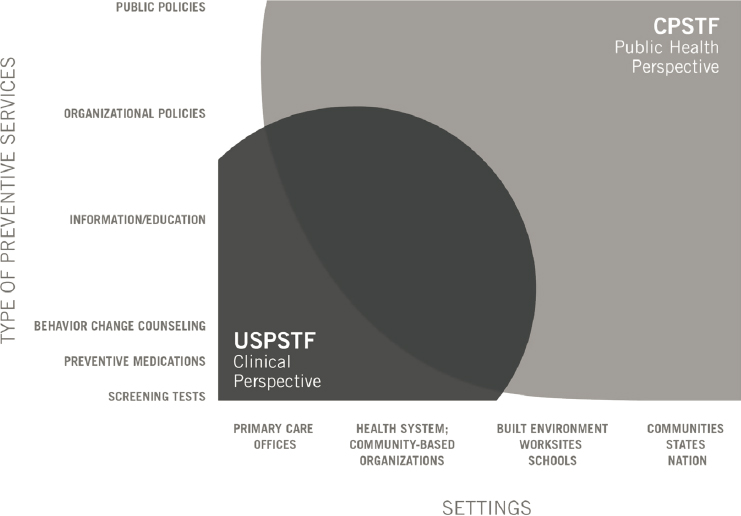
SOURCE: Grossman and Elder, 2015.
informed its assessment, and often uses the subcategories of “effectiveness,” “applicability,” and “implementation” to characterize evidence gaps.
HOW OTHER CLINICAL PRACTICE GUIDELINE DEVELOPERS ADDRESS EVIDENCE GAPS
The committee looked for possible helpful taxonomies from other CPG developers. While no such taxonomy was discovered, the search provided some insights into how other panels address insufficient evidence. For example, the American College of Cardiology and the American Heart Association and the United Kingdom National Screening Committee describe evidence gaps in a manner similar to those of the USPSTF’s I statements or the CPSTSF’s IEs, though nomenclature varies. If there is insufficient evidence regarding a service, they so state and do not recommend for or against the preventive service (Duff et al., 2019; Hamid et al., 2021).
Other bodies treat insufficient evidence differently. Instead of declining to recommend for or against a preventive service, some bodies use expert opinion to take the place of evidence when sufficient evidence is not available. With breast cancer screening, for example, the USPSTF issued a C grade for biennial mammograms in women aged 40–49 years, with moderate certainty that the net benefit of screening with mammography in women ages 40 through 49 is small (USPSTF, 2016). The American Cancer Society and the American College of Obstetricians and Gynecologists recommend breast cancer screening even for ages where it notes there are gaps in the evidence, based “primarily on consensus and expert opinion” (ACOG CPBG, 2017; Oeffinger et al., 2015).
Some groups produce disease-based guidelines (i.e., guidelines that discuss both prevention and treatment). As such, the noted research gaps span the prevention and treatment domains. Of note, some disease-based organizations assume the need to screen, so the research gaps do not always address the lack of demonstrated screening efficacy. For example, the Society for Vascular Surgery (SVS) provides a lengthy list of research gaps (“Areas in Need of Further Research”) for abdominal aortic aneurysm (AAA), but these dealt more with improving processes of care than whether the screening for AAA should be done (Chaikof et al., 2009). Because practicing clinicians often need guidance, especially when there is insufficient evidence, the purpose of the following examples is to show the variety of responses to insufficient evidence, rather than to criticize particular guidelines for differing in methodology from the USPSTF.
REFERENCES
ACOG CPBG (American College of Obstetricians and Gynecologists Committee on Practice Bulletins—Gynecology). 2017. Breast cancer risk assessment and screening in average-risk women. ACOG Practice Bulletin 179. https://www.acog.org/clinical/clinical-guidance/practice-bulletin/articles/2017/07/breast-cancer-risk-assessment-and-screening-in-average-risk-women (accessed October 19, 2021).
Allen, J. D., S. M. Bluethmann, M. Sheets, K. M. Opdyke, K. Gates-Ferris, M. Hurlbert, and E. Harden. 2013. Women’s responses to changes in U.S. Preventive Task Force’s mammography screening guidelines: Results of focus groups with ethnically diverse women. BMC Public Health 13:1169. https://doi.org/10.1186/1471-2458-13-1169 (accessed October 19, 2021).
Anthony, J. S., S.-Y. Baik, B. J. Bowers, B. Tidjani, C. J. Jacobson, and J. Susman. 2010. Conditions that influence a primary care clinician’s decision to refer patients for depression care. Rehabilitation Nursing 35(3):113–122. https://doi.org/10.1002/j.2048-7940.2010.tb00286.x (accessed November 2, 2021).
Atkins, D. 2021. Funder perspectives on addressing evidence gaps. Presented on March 16, 2021, at Meeting 4 of the Committee on Addressing Evidence Gaps in Clinical Prevention. https://www.nationalacademies.org/event/03-16-2021/addressing-evidence-gaps-in-clinical-prevention-committee-meeting-session-2 (accessed October 19, 2021).
Barton, M. B., T. Miller, T. Wolff, D. Petitti, M. LeFevre, G. Sawaya, B. Yawn, J. Guirguis-Blake, N. Calonge, R. Harris, and U.S. Preventive Services Task Force. 2007. How to read the new recommendation statement: Methods update from the U.S. Preventive Services Task Force. Annals of Internal Medicine 147(2):123–127. https://doi.org/10.7326/0003-4819-147-2-200707170-00171 (accessed October 19, 2021).
Bauchner, H., P. B. Fontanarosa, and R. M. Golub. 2016. JAMA welcomes the U.S. Preventive Services Task Force. JAMA 315(4):351–352. https://doi.org/10.1001/jama.2015.18448 (accessed October 19, 2021).
Borsky, A. 2021. Personal correspondence with Amanda Borsky, AHRQ staff member, July 29, 2021. Available by request from the project Public Access File at https://www8.nationalacademies.org/pa/ManageRequest.aspx?key=51932 (accessed November 3, 2021).
Briss, P. A., S. Zaza, M. Pappaioanou, J. Fielding, L. Wright-De Agüero, B. I. Truman, D. P. Hopkins, P. D. Mullen, R. S. Thompson, S. H. Woolf, V. G. Carande-Kulis, L. Anderson, A. R. Hinman, D. V. McQueen, S. M. Teutsch, and J. R. Harris. 2000. Developing an evidence-based guide to community preventive services—methods. The Task Force on Community Preventive Services. American Journal of Preventive Medicine 18(1 Suppl):35–43. https://doi.org/10.1016/S0749-3797(99)00119-1 (accessed October 19, 2021).
Chaikof, E. L., D. C. Brewster, R. L. Dalman, M. S. Makaroun, K. A. Illig, G. A. Sicard, C. H. Timaran, G. R. Upchurch, Jr., and F. J. Veith. 2009. The care of patients with an abdominal aortic aneurysm: The Society for Vascular Surgery practice guidelines. Journal of Vascular Surgery 50(4 Suppl):S2–S49. https://doi.org/10.1016/j.jvs.2017.10.044 (accessed October 19, 2021).
Correa, V. C., L. H. Lugo-Agudelo, D. C. Aguirre-Acevedo, J. A. P. Contreras, A. M. P. Borrero, D. F. Patino-Lugo, and D. A. C. Valencia. 2020. Individual, health system, and contextual barriers and facilitators for the implementation of clinical practice guidelines: A systematic metareview. Health Research Policy and Systems 18(1):74. https://health-policy-systems.biomedcentral.com/articles/10.1186/s12961-020-00588-8 (accessed October 19, 2021).
CPSTF (Community Preventive Services Task Force). 2005. Cancer screening: Small media targeting clients—colorectal cancer. https://www.thecommunityguide.org/findings/cancer-screening-small-media-targeting-clients-colorectal-cancer (accessed July 19, 2021).
CPSTF. 2006. Cancer screening: Provider reminder and recall systems—colorectal cancer. https://www.thecommunityguide.org/findings/cancer-screening-provider-reminder-and-recall-systems-colorectal-cancer (accessed July 19, 2021).
CPSTF. 2009. Cancer screening: Provider assessment and feedback—colorectal cancer. https://www.thecommunityguide.org/findings/cancer-screening-provider-assessment-and-feedback-colorectal-cancer (accessed July 19, 2021).
CPSTF. 2010a. Cancer screening: Client reminders—colorectal cancer. https://www.thecommunityguide.org/findings/cancer-screening-client-reminders-colorectal-cancer (accessed July 19, 2021).
CPSTF. 2010b. Cancer screening: One-on-one education for clients—colorectal cancer. https://www.thecommunityguide.org/findings/cancer-screening-one-one-education-clients-colorectal-cancer (accessed July 19, 2021).
CPSTF. 2010c. Cancer screening: Reducing structural barriers for clients—colorectal cancer. https://www.thecommunityguide.org/findings/cancer-screening-reducing-structural-barriers-clients-colorectal-cancer (accessed July 19, 2021).
CPSTF. 2016. Cancer screening: Multicomponent interventions—colorectal cancer. https://www.thecommunityguide.org/findings/cancer-screening-multicomponent-interventions-colorectal-cancer (accessed July 19, 2021).
CPSTF. 2019. Cancer screening: Interventions engaging community health workers—colorectal cancer. https://www.thecommunityguide.org/findings/cancer-screening-interventions-engaging-community-health-workers-colorectal-cancer (accessed July 19, 2021).
CPSTF. n.d.-a. About the Community Preventive Services Task Force. https://www.thecommunityguide.org/task-force/about-community-preventive-services-task-force (accessed July 19, 2021).
CPSTF. n.d.-b. New resource outlines ways to use insufficient evidence findings. https://www.thecommunityguide.org/content/new-resource-outlines-ways-use-insufficient-evidence-findings (accessed July 19, 2021).
CPSTF. n.d.-c. Community Guide methodology. https://www.thecommunityguide.org/about/community-guide-methodology (accessed July 19, 2021).
CTFPHE (Canadian Task Force on the Public Health Examination). 1979. The periodic health examination. Canadian Medical Association Journal 121(9):1193–1254. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1801737/ (accessed November 2, 2021).
Duff, J. P., A. A. Topjian, M. D. Berg, M. Chan, S. E. Haskell, B. L. Joyner, Jr., J. J. Lasa, S. J. Ley, T. T. Raymond, R. M. Sutton, M. F. Hazinski, and D. L. Atkins. 2019. 2019 American Heart Association focused update on pediatric advanced life support: An update to the American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation 140:e904–e914. https://www.ahajournals.org/doi/10.1161/CIR.0000000000000731 (accessed October 19, 2021).
Fischer, F., K. Lange, K. Klose, W. Greiner, and A. Kraemer. 2016. Barriers and strategies in guideline implementation—a scoping review. Healthcare (Basel) 4(3):36.
Grossman, D. C., and R. W. Elder. 2015. Aligning the work of two U.S. task forces on behavioral counseling recommendations. American Journal of Preventive Medicine 49(3 Suppl 2):S174–S183.
Hamid, S., P. Desai, P. Hysi, J. M. Burr, and A. P. Khawaja. 2021. Population screening for glaucoma in UK: Current recommendations and future directions. Eye. https://doi.org/10.1038/s41433-021-01687-8 (accessed October 19, 2021).
Harding, K., J. Porter, A. Horne-Thompson, E. Donley, and N. F. Taylor. 2014. Not enough time or a low priority? Barriers to evidence-based practice for allied health clinicians. Journal of Continuing Education in the Health Professions 34(4):224–231.
Hoagland, A., and P. Shafer. 2021. Out-of-pocket costs for preventive care persist almost a decade after the Affordable Care Act. Preventive Medicine 150:106690.
Hurwitz, B. 1999. Legal and political considerations of clinical practice guidelines. BMJ 318(7184):661–664.
IOM (Institute of Medicine). 2011. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press. https://doi.org/10.17226/13058.
Jones, R. M., K. J. Devers, A. J. Kuzel, and S. H. Woolf. 2010. Patient-reported barriers to colorectal cancer screening: A mixed-methods analysis. American Journal of Preventive Medicine 38(5):508–516.
KFF (Kaiser Family Foundation). 2015. Preventive services covered by private health plans under the Affordable Care Act. https://www.kff.org/health-reform/fact-sheet/preventive-services-covered-by-private-health-plans (accessed July 19, 2021).
Klabunde, C. N., E. M. Ellis, J. Villani, E. Neilson, K. Schwartz, E. A. Vogt, and Q. Ngo-Metzger. 2021. Characteristics of scientific evidence informing changed U.S. Preventive Services Task Force insufficient evidence statements. American Journal of Preventive Medicine. https://www.sciencedirect.com/science/article/abs/pii/S0749379721004578 (accessed November 29, 2021).
Lang, E., S. Alves da Silva, and N. Persaud. 2021. Are guidelines fueling inequity? A call to action for guideline developers and their panelists. Chest 159(2):465–466.
McGinnis, J. M., and S. H. Woolf. 1990. Background and objectives of the U.S. Preventive Services Task Force. Journal of General Internal Medicine 5(5 Suppl):S11–S13.
Merenstein, D. 2004. A piece of my mind. Winners and losers. JAMA 291(1):15–16.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2020. Leading Health Indicators 2030: Advancing health, equity, and well-being. Washington, DC: The National Academies Press. https://doi.org/10.17226/25682.
ODP (Office of Disease Prevention). 2021. About the ODP. https://prevention.nih.gov/about-odp/strategic-plan/communicate-efforts-findings (accessed October 22, 2021).
Oeffinger, K. C., E. T. Fontham, R. Etzioni, A. Herzig, J. S. Michaelson, Y. C. Shih, L. C. Walter, T. R. Church, C. R. Flowers, S. J. LaMonte, A. M. Wolf, C. DeSantis, J. Lortet-Tieulent, K. Andrews, D. Manassaram-Baptiste, D. Saslow, R. A. Smith, O. W. Brawley, R. Wender, and American Cancer Society. 2015. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA 314(15):1599–1614.
Rauh, S., D. Arnold, S. Braga, R. Curca, R. Eckert, A. Frobe, M. Karamouzis, G. Lakatos, and J. L. Molitor. 2018. Challenge of implementing clinical practice guidelines. Getting ESMO’s guidelines even closer to the bedside: Introducing the ESMO practising oncologists’ checklists and knowledge and practice questions. ESMO Open 3(5):e000385.
Truman, B. I., C. K. Smith-Akin, A. R. Hinman, K. M. Gebbie, R. Brownson, L. F. Novick, R. S. Lawrence, M. Pappaioanou, J. Fielding, C. A. Evans, Jr., F. A. Guerra, M. Vogel-Taylor, C. S. Mahan, M. Fullilove, and S. Zaza. 2000. Developing the Guide to Community Preventive Services—overview and rationale: The Task Force on Community Preventive Services. American Journal of Preventive Medicine 18(1 Suppl):18–26.
USPSTF (U.S. Preventive Services Task Force). 1989. Guide to clinical preventive services: An assessment of the effectiveness of 169 interventions: Report of the U.S. Preventive Services Task Force. Baltimore, MD: Williams & Wilkins. https://wonder.cdc.gov/wonder/prevguid/p0000109/p0000109.asp (accessed November 2, 2021).
USPSTF. 1996. Guide to clinical preventive services: Report of the U.S. Preventive Services Task Force. 2nd ed. Washington, DC: U.S. Government Printing Office. https://www.ncbi.nlm.nih.gov/books/NBK15435 (accessed November 2, 2021).
USPSTF. 2002. Guide to clinical preventive services. 3rd ed. Rockville, MD: Agency for Healthcare Research and Quality. https://www.ncbi.nlm.nih.gov/books/NBK15199 (accessed November 2, 2021).
USPSTF. 2005. Guide to clinical preventive services 2005: Recommendations from the U.S. Preventive Services Task Force. Bethesda, MD: Agency for Healthcare Research and Quality. http://www.lumen.luc.edu/lumen/meded/hmps/pocketgd%5B1%5D.pdf (accessed November 2, 2021).
USPSTF. 2007a. Guide to clinical preventive services 2007: Recommendations of the U.S. Preventive Services Task Force. Rockville, MD: Agency for Healthcare Research and Quality. https://www.ncbi.nlm.nih.gov/books/NBK16363 (accessed November 2, 2021).
USPSTF. 2007b. Motor vehicle occupant restraints: Counseling. https://www.uspreventiveservices-taskforce.org/uspstf/index.php/recommendation/motor-vehicle-occupant-restraints-counseling (accessed July 19, 2021).
USPSTF. 2008. Phenylketonuria in newborns: Screening. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/phenylketonuria-in-newborns-screening (accessed July 19, 2021).
USPSTF. 2009. Guide to clinical preventive services 2009: Recommendations of the U.S. Preventive Services Task Force. Rockville, MD: Agency for Healthcare Research and Quality. https://www.ncbi.nlm.nih.gov/books/NBK37637 (accessed November 2, 2021).
USPSTF. 2010. Guide to clinical preventive services 2010–2011: Recommendations of the U.S. Preventive Services Task Force. Rockville, MD: Agency for Healthcare Research and Quality. https://www.ncbi.nlm.nih.gov/books/NBK56707 (accessed November 2, 2021).
USPSTF. 2011. First annual report to congress on high-priority evidence gaps for clinical preventive services. https://www.uspreventiveservicestaskforce.org/uspstf/about-uspstf/reports-congress/first-annual-report-congress-high-priority-evidence-gaps-clinical-preventive-services (accessed August 27, 2021).
USPSTF. 2012. Guide to clinical preventive services 2012: Recommendations of the U.S. Preventive Services Task Force. Rockville, MD: Agency for Healthcare Research and Quality. https://www.ncbi.nlm.nih.gov/books/NBK115115 (accessed November 2, 2021).
USPSTF. 2014a. Guide to clinical preventive services 2014: Recommendations of the U.S. Preventive Services Task Force. Rockville, MD: Agency for Healthcare Research and Quality. https://www.ncbi.nlm.nih.gov/books/NBK235846 (accessed November 2, 2021).
USPSTF. 2014b. Youth violence: Counseling. https://www.uspreventiveservicestaskforce.org/uspstf/index.php/recommendation/youth-violence-counseling (accessed July 19, 2021).
USPSTF. 2016. Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement. Annals of Internal Medicine 164(4):279–296.
USPSTF. 2017a. Procedure manual section 6: Methods for arriving at a recommendation. https://www.uspreventiveservicestaskforce.org/uspstf/about-uspstf/methods-and-processes/procedure-manual/procedure-manual-section-6-methods-arriving-recommendation (accessed July 19, 2021).
USPSTF. 2017b. Update on methods: Insufficient evidence. https://www.uspreventiveservicestaskforce.org/uspstf/about-uspstf/methods-and-processes/update-methods-insufficient-evidence (accessed July 19, 2021).
USPSTF. 2018. Grade definitions. https://www.uspreventiveservicestaskforce.org/uspstf/about-uspstf/methods-and-processes/grade-definitions (accessed July 19, 2021).
USPSTF. 2019. Use of decision models in the development of evidence-based clinical preventive services recommendations. https://www.uspreventiveservicestaskforce.org/uspstf/about-uspstf/methods-and-processes/use-decision-models-development-evidence-based-clinical-preventive-services-recommendations (accessed July 19, 2021).
USPSTF. 2020. Reports to Congress. https://www.uspreventiveservicestaskforce.org/uspstf/about-uspstf/reports-congress (accessed July 19, 2021).
USPSTF. 2021a. About the USPSTF. https://www.uspreventiveservicestaskforce.org/uspstf/index.php/about-uspstf (accessed July 19, 2021).
USPSTF. 2021b. Colorectal cancer: Screening. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/colorectal-cancer-screening (accessed July 19, 2021).
USPSTF. 2021c. USPSTF and cost considerations. https://www.uspreventiveservicestaskforce.org/uspstf/about-uspstf/task-force-resources/uspstf-and-cost-considerations (accessed July 19, 2021).
USPSTF. 2021d. USPSTFrecommendations development process: Agraphic overview. https://www.uspreventiveservicestaskforce.org/uspstf/about-uspstf/methods-and-processes/uspstf-recommendations-development-process-graphic-overview (accessed July 19, 2021).
USPSTF. n.d.-a. Immunizations for adults. https://www.uspreventiveservicestaskforce.org/uspstf/index.php/recommendation/immunizations-for-adults (accessed July 19, 2021).
USPSTF. n.d.-b. Immunizations for children. https://www.uspreventiveservicestaskforce.org/uspstf/immunizations-children (accessed July 19, 2021).
USPSTF. n.d.-c. Sickle cell disease (hemoglobinopathies) in newborns: Screening. https://www.uspreventiveservicestaskforce.org/uspstf/index.php/recommendation/sickle-cell-disease-hemoglobinopathies-in-newborns-screening (accessed July 19, 2021).
USPSTF. n.d.-d. Understanding how the U.S. Preventive Services Task Force works: USPSTF 101. https://www.uspreventiveservicestaskforce.org/Home/GetFile/6/91/uspstf101_slides/pdf (accessed July 19, 2021).
Weisz, G., A. Cambrosio, P. Keating, L. Knaapen, T. Schlich, and V. J. Tournay. 2007. The emergence of clinical practice guidelines. Milbank Quarterly 85(4):691–727.
Welch, V. A., E. A. Akl, G. Guyatt, K. Pottie, J. Eslava-Schmalbach, M. T. Ansari, H. de Beer, M. Briel, T. Dans, I. Dans, M. Hultcrantz, J. Jull, S. V. Katikireddi, J. Meerpohl, R. Morton, A. Mosdol, J. Petkovic, H. J. Schunemann, R. N. Sharaf, J. A. Singh, R. Stanev, T. Tonia, M. Tristan, S. Vitols, J. Watine, and P. Tugwell. 2017. GRADE equity guidelines 1: Considering health equity in GRADE guideline development: Introduction and rationale. Journal of Clinical Epidemiology 90:59–67.


















