2
The Process to Create the Dietary Guidelines for Americans Is Both Complicated and Complex: Background and Context for Task 3
The Dietary Guidelines for Americans (DGA) are the foundation for all federal food assistance programs, military food programs, and dietary guidance provided to the public by the federal government, with the goal of reaching all Americans (USDA/HHS, 2020) (see Chapter 1). Thus, it is hard to overstate the importance of the DGA as a cornerstone of federal nutrition policy. This chapter provides an overview of some key elements of the process by which the DGA are developed and discusses the inherent challenges in this process, as identified by the Committee on Redesigning the Process for Establishing the Dietary Guidelines for Americans (the 2017 report committee) in its report, Redesigning the Process for Establishing the Dietary Guidelines for Americans (NASEM, 2017a). Thus, this chapter provides the context for the current report and background information for Task 3 from the Statement of Task (see Chapter 1, Box 1-2).
This chapter begins with a discussion of factors that influence public trust in nutrition science (and by extension, in the DGA)—transparency, integrity, and trustworthiness—along with a discussion of the way that conflict of interest (COI) is assessed and mitigated in the process of developing the DGA. The second section of the chapter highlights the administrative complications and complexities associated with the process to develop the DGA. The complexity of the processes to develop the DGA have implications for the analytic power and sophistication that are required for using multiple types of data simultaneously to derive public health recommendations. Therefore, the committee concludes the chapter by highlighting the potential of systems science methods to address
these complexities. Overall, this chapter provides context for Task 3 as well as issues that were addressed in the 2017 National Academies report (NASEM, 2017a), which was designed to ensure that the DGA process delivers guidelines that are scientifically robust and perceived as trustworthy by the scientific community and the public.
COMPLEXITIES RELATED TO TRUST IN NUTRITION SCIENCE
The DGA have previously been met with a degree of controversy by some in the nutrition research community, the food industry, and public health advocacy groups (Nestle, 2018). The integrity and trustworthiness of the DGA were brought into stark relief by controversy surrounding the release of the Scientific Report of the 2015 Dietary Guidelines Advisory Committee (2015 DGAC Scientific Report) and the subsequent 2015–2020 DGA (Nestle, 2018). In particular, recommendations in the 2015-2020 DGA about intake of red meats and cholesterol generated public concern about the influence of the meat and egg industries on the DGA (Nestle, 2018; Goldman, 2016). Discrepancies between the language used to describe recommendations in the 2015 DGAC Scientific Report and language used in the 2015–2020 DGA, especially recommendations for intake of red and processed meats, have been highlighted as contributing to public confusion and erosion of public trust in the DGA (Goldman, 2016). The 2015 DGAC Scientific Report also included discussions around environmental sustainability and dietary patterns, which were widely criticized by a number of industry lobbying groups (Goldman, 2016). In its hearing with the secretaries of the U.S. Department of Health and Human Services (HHS) and the U.S. Department of Agriculture (USDA), the House Committee on Agriculture also raised the point that inclusion of sustainability issues was outside the scope of the DGA (NASEM, 2017a). Nevertheless, discussions around sustainability would further inform the controversies inherent in the DGA. The 2017 National Academies report also noted that “Congress … raised questions about the process to develop the DGA, asking whether the process could be trusted by the American people” (NASEM, 2017a). In addition, discrepancies in language used to describe recommendations were also raised relative to the 2020 DGAC Scientific Report and 2020–2025 DGA, specifically for intakes of added sugars and alcohol (Mayer-Davis et al., 2021; Lindquist et al., 2021). In this instance, the 2020 DGAC Scientific Report recommended limiting added sugars to 6% of daily calories but the 2020–2025 DGA retained the recommendation in the 2015-2020 DGA of 10% of daily calories. Similarly, the 2020 DGAC Scientific Report recommended reducing the recommend consumption of alcohol to one drink per day or less for males but the 2020–2025 DGA retained the recommendation in the 2015-2020 DGA of two drinks or less per day for males. In summary,
these differences between the DGAC Scientific Report and DGA, as well as continued concern around possible COI of Dietary Guidelines Advisory Committee members and the DGA process itself (Achterberg et al., 2022), were highlighted by the popular press (Jacobs, 2020; Washington Post, 2021) as well as interest groups and individual web-based publications (e.g., blog posts) (Harvard, 2021; Sullivan, 2021). This media coverage could affected public trust in the DGA.
Public Trust in Nutrition Science
Although public trust in scientists has remained relatively constant over the past half-century, there has been “a steady erosion of public trust in expertise in general, rather than in science specifically” (Garza et al., 2019). Loss of public trust in expertise may be related to the inherent complexity of science and its iterative nature, as well as growing political and ideological polarization around policies that rely heavily on science, such as vaccine safety and the use of genetically modified organisms (Garza et al., 2019). Public trust may be challenged when experts voice opposing viewpoints, change recommendations based on updated evidence, or update recommendations in ways that conflict with previously welcomed or long-held beliefs (e.g. the perceived health effects of a particular food or nutrient). It has been suggested that “nutrition science may be the area that provides the most ammunition for distrust, given the combination of uncertainty, public interest, and powerful preferences” (Rosenbaum, 2017). The 2017 report committee noted that the public does not fully use the DGA as, at the time, “less than 10 percent of the Americans actually follow the guidelines” (NASEM, 2017a). Congress suggested that this could result from “a lack of confidence, in part because of how the DGA have been developed and hence a low level of trust in the ultimate recommendations” (NASEM, 2017a). Even when guidelines are considered trustworthy, numerous other factors affect the degree to which an individual’s food choices align with such guidelines (Jahns et al., 2015; Nicklas et al., 2013).
Issues related to public trust in clinical and public health guidelines are not unique to the DGA. Enhancing public trust in clinical practice guidelines was emphasized by the Institute of Medicine (IOM) in a prior report, Clinical Practice Guidelines We Can Trust (IOM, 2011). The report highlighted the need to distinguish “methodologically rigorous, transparent, evidence-based guidelines” that patients and the public can trust and that could potentially “enhance translation of research into practice” and “enhance adoption of guidelines by all stakeholders” (IOM, 2011). The importance of creating trustworthy guidelines and the need for increased
transparency and rigorous, evidence-based practices were highlighted by both reports produced by the 2017 report committee (NASEM 2017a,b).
The field of nutrition—and by extension, the DGA —is particularly challenged to maintain public trust (Garza et al., 2019; Ioannidis and Trepanowski, 2018). First, food is intimately connected to daily life for both experts and the general population; second, diverse social, cultural, and behavioral influences affect beliefs about nutrition; third, the food industry plays a sizable role in funding, conducting, and disseminating nutrition research (Rudd, n.d.; IOM, 2006); and fourth, many nutrition messages (e.g., from food marketers, bloggers, and social media influencers) may be disconnected from nutrition science. A 2019 Pew Research Center survey demonstrated that, although trust in scientists was relatively strong between 2016 and 2019, less than 30 percent of respondents felt nutrition researchers had the “best interest of the public in mind” or provided fair and accurate information “most of the time” (Funk et. al, 2019). Although many of the issues described above, and the Pew survey itself, do not specifically address public trust in the DGA, they highlight the challenges the DGA face in gaining public trust regardless of their specific content.
Conflicts of Interest
The topic of adequate disclosure of nutrition researchers’ potential COI has received increasing attention during the past several years (Ioannidis and Trepanowski, 2018). For example, only 12 percent of respondents in the 2019 Pew survey thought that nutrition researchers were transparent about reporting COI “most of the time” (Funk et al., 2019). The scientific community has traditionally focused on the disclosure of financial COI, but nutrition researchers may also have several types of non-financial conflicts, such as conflict of commitment, allegiance bias, advocacy or activism, and their own regular dietary choices (Ioannidis and Trepanowski, 2018). Importantly, the potential effects of nutrition research–associated COI are heightened because of the close relationship between nutrition research and public health policy—an issue that is compounded by increasing support of nutrition research by funders that seek to affect public policy (Garza et al., 2019). Several authoritative bodies (related to nutrition specifically and medicine or public health more generally) have recently highlighted the need for increased objectivity and transparency in the successful assessment of COI and management of its potential effect (Garza et al., 2019).
The first product of the 2017 report committee outlined several recommendations for the management of COI of members of the DGAC (NASEM 2017a). The USDA and the HHS implemented many changes in
response to these recommendations (Stoody, 2019; Snetselaar et. al., 2021). Issues pertaining to COI of individual members of the DGAC are outside the scope of the current committee’s Statement of Task.
USDA and HHS have policies for COI that apply to individual employees. USDA, specifically the Nutrition Evidence Systematic Reviews (NESR), has well-defined COI policies governing individuals involved in systematic review projects. As mentioned, the two Departments also have well-defined COI policies for members of the DGAC (USDA, n.d.a.). COI policies for the “writing team” (the employees from several federal agencies who are responsible for drafting the DGA) stipulate that “members are asked to recuse themselves from participating in activities that could be or could be perceived to be a conflict of interest” (NASEM, 2017a). A unique challenge for USDA may be that agricultural output is a key determinant of diet quality, and agriculture is also intimately connected to economic interests given that “the production of agricultural commodities plays a significant role in the economy of the United States.”1 These conflicting loyalties could be construed as creating an inherent institutional COI for the DGA process that may contribute to a loss of public trust in the DGA (Garza et al., 2019). Management of COI by the federal agencies is likely to remain important given the current context surrounding public trust in nutrition science and the DGA.
Summary of Complexities Related to Trust in Nutrition
The Committee on Redesigning the Process for Establishing the DGA noted that “Another potential reason for lack of adherence to dietary intakes recommended in the DGA is that the public has ‘lost faith’ in the DGA” (NASEM 2017a). The controversies that arose following the release of the 2015–2020 DGA highlight issues surrounding public trust in nutrition science and continue to challenge the nutrition research community and the trustworthiness of the DGA. Given the erosion of trust in expertise in general and the public attention on COI in nutrition research, these issues are likely to remain pertinent to future DGA cycles. Therefore, assessment and transparent management of both individual and institutional COI will remain important for maintaining and increasing public trust in the DGA.
___________________
1 Federal Agriculture Improvement and Reform Act of 1996, Public Law 104-127. (April 4, 1996) 7 U.S.C. 7401, 110 Stat. 1032. Section 512. https://www.congress.gov/104/plaws/publ127/PLAW-104publ127.pdf.
COMPLEXITIES RELATED TO THE PROCESS OF CREATING THE DIETARY GUIDELINES FOR AMERICANS
The process to develop the DGA involves complexities that are temporal, administrative, and methodological. The committee reviewed these complexities, which have implications for how the DGA process is operationalized within and across DGA cycles.
The Dietary Guidelines Process Is Time-Restricted
The National Nutrition Monitoring and Related Research Act requires that the DGA be published at least every 5 years (see Chapter 1).2 The process used to create each edition of the DGA primarily occurs during the last 2–3 years of each 5-year cycle, as described in the committee’s report Evaluating the Process to Develop the Dietary Guidelines for Americans, 2020–2025: A Midcourse Report (NASEM, 2022) and detailed in the timeline in Appendix B. The 2017 report committee recommended a redesign of the DGA process that uses the full 5-year period to provide “continuity across 5-year cycles, with some activities spanning across DGA cycles,” which it said would also assist with strategic planning, particularly for the systematic reviews to be conducted or updated (NASEM, 2017a). The recommended redesign also advised USDA and HHS to redistribute the current functions of the Dietary Guidelines Advisory Committee to three separate groups (see Box 1-1).
The process used to create the 2020–2025 DGA was time-constrained and consisted of three distinct periods, each with key participants, processes, and data. The distinct periods can be described as occurring before the appointment of the DGAC, during the DGAC’s review of the evidence, and during the development of the DGA by USDA and HHS (Appendix B). The elements of this process are detailed in the 2017 National Academies report (NASEM, 2017a).
The Dietary Guidelines Process Is Complicated to Administer
USDA and HHS alternate providing administrative leadership for developing each edition of the DGA. The two Departments sign a memorandum of understanding to cover this work early in each cycle (Appendix B). They appoint an external Federal Advisory Committee—the DGAC, composed of experts in topics relevant to the guidelines—to
___________________
2 National Nutrition Monitoring and Related Research Act of 1990, Public Law 101-445, 101st Cong. (October 22, 1990). https://www.govinfo.gov/content/pkg/STATUTE-104/pdf/STATUTE-104-Pg1034.pdf.
review current nutrition evidence (DGA, n.d.). Federal staff support the DGAC’s work and provide access to much of the data used by the DGAC to develop its report. For example, NESR (formerly known as Nutrition Evidence Library [NEL]) in USDA supports the DGAC in conducting systematic reviews, regardless of which agency takes the lead in the given cycle. A Dietary Guidelines writing team of federal staff from USDA and HHS consider the DGAC Scientific Report, and other inputs (including prior DGAC Scientific Reports and guidance statements from other authoritative bodies), when writing the DGA. The DGA is then peer-reviewed by experts outside of the federal government, as well as by federal agencies that operate nutrition programs and/or administer nutrition policies3 before they undergo departmental clearance and are publicly released (DGA, n.d).
The recommendations from the 2017 National Academies report to redesign the DGA process would not reduce the administrative complications of the DGA process. As mentioned previously in this chapter, these recommendations include creation of an additional committee that would be subject to the process required by the Federal Advisory Committee Act,4 as well as the likely need to revise the charter of the DGAC (NASEM 2017a). Nonetheless, if implemented, the 2017 report’s recommendations would provide tools for developing additional continuity between DGA cycles as well as additional strategic planning for systematic reviews and data needs. Implementing these recommendations were intended to result in a DGA process that would “become more agile, flexible, and effective—and address more topics of interest to the general public” (NASEM, 2017a).
The Dietary Guidelines Process Relies on Multiple Types of Complex Data
Multiple types of data are gathered, assessed, and interpreted to create the DGAC Scientific Report (which informs the DGA) (Appendix C). These data include—but are not limited to—measures of health status and dietary intake of foods and nutrients among various U.S. population groups. These data are derived from numerous national and other data collection efforts that differ in scope, collection frequency, and data
___________________
3 For example, Agriculture Research Service, Food and Nutrition Service, Food Safety and Inspection Service, National Institute, Food and Drug, Centers for Disease Control and Prevention, etc.
4 For more information, see the Federal Advisory Committee Act (FACA) brochure https://www.gsa.gov/policy-regulations/policy/federal-advisory-committee-management/advice-and-guidance/the-federal-advisory-committee-act-faca-brochure.
format. Additional types of data that are used in the process include information about how changes in dietary intake may affect the nutrient needs of individuals, available NESR systematic reviews on the relationships between dietary intake and outcomes of interest, and the literature used to conduct new systematic reviews on relevant topics.
This section of the chapter provides a description of how these different types of data are used together. It also includes a discussion of the challenges of using dietary data given their particular relevance to the creation of dietary guidance. The process of evaluating and integrating these data is complex and may require more powerful analytic approaches and tools than currently used (NASEM, 2017a). This complexity also adds to the challenges of developing DGA that are cohesive and readily accepted.
Non-Dietary Data Used to Create the DGAC Scientific Report
Data on the health status of the U.S. population are collected via federal monitoring and surveillance systems, such as continuous or recurring population surveys. The DGAC uses data from these surveys to obtain estimates on the prevalence of health conditions that are possibly related to dietary intake. The National Health and Nutrition Examination Survey (NHANES) (see Box 2-1 and Appendix C) is a major data source for this purpose, as it collects several health-related variables in continuous 2-year cycles.
Dietary Intake Data Used to Create the DGAC Scientific Report
Dietary intake is a complex phenomenon because humans consume a range of foods and beverages, and potentially supplements, in varying combinations and amounts. Adding to this multidimensionality, dietary intake is dynamic over the short and the long term (Reedy et al., 2017, 2018). This is because dietary intake may vary not only by day and season, but also by stage of the life course (e.g., adolescence, pregnancy, or lactation). Dietary patterns may also vary by cultural background and in relation to other factors (e.g. food security, use of food banks, home gardens, etc.). Consequently, it is challenging to develop guidance for dietary intake in a diverse population.
Dietary data are fundamental
Data on dietary intake are used to assess current intakes and sources of guidance-based food groups and nutrients (e.g., sodium, added sugars), identify nutrients of concern, characterize dietary exposures in the studies evaluated in systematic reviews, and model patterns of food intake that meet nutrient needs and reduce chronic disease risk. This widespread use of data on dietary intake by the
DGAC, in systematic reviews, food pattern modeling, and descriptive data analysis, makes the development of the DGA uniquely sensitive to the challenges with these data, which are discussed below.
Dietary data are complex to collect and use
“Usual” dietary intake (as opposed to intake on a given day) (Beaton et al., 1997; NRC, 1986) is the phenomenon of most salience to the DGA but cannot be directly observed in most contexts. Consequently, self-report tools are used, typically with the aim of making inferences about usual intake (Thompson and Subar,
2017). WWEIA collects 24-hour dietary recalls. These provide a key source of information about current food and nutrient intakes, which is used to assess nutrients of concern and model food intake patterns (Box 2-1). Recalls measure dietary intake with less systematic error than long-term tools, such as food frequency questionnaires (FFQs), but they are subject to substantial random measurement error, including day-to-day variation in intake (Freedman et al., 2014, 2015). Progress has been made in analytic methods to account for within-person variation appropriately in modeling usual intake (Dodd et al., 2006; IOM, 2000; Kirkpatrick et al., 2022), and these methods have been incorporated into the DGA process (e.g., in the process of identifying nutrients of concern [Bailey et al., 2021]). However, attention is needed to determine if appropriate analytic methods have been applied in the studies using short-term dietary assessment methods that are included in the systematic reviews. Further, in making inferences from WWEIA and other studies using short-term data, the potential implications of systematic error should be considered.
The dietary data included in systematic reviews are primarily from cohort studies, which often use FFQs or brief screeners that aim to directly assess usual intake by asking about intake over long periods of time (e.g., past year or past 30 days) (Thompson and Subar, 2017). Data from FFQs and brief screeners have more systematic measurement error than those from 24-hour recalls (Freedman et al., 2014, 2015). This error can attenuate observed diet–health relationships, and this must be considered when conducting systematic reviews, particularly if the available statistical strategies to mitigate the effect of systematic error have not been applied (Freedman et al., 2011). Because FFQs and brief screeners ask about a finite list of foods, their appropriateness with use for different population subgroups and cultures must also be considered (Frongillo et al., 2019; Kirkpatrick et al., 2019; Obbagy et al., 2022).
More broadly, the validity and reliability of dietary assessment tools for estimating particular dietary components require careful consideration, especially for some population subgroups (e.g., children versus adults) (Frongillo et al., 2019; Kirkpatrick et al., 2019). Biomarker-based validation studies show that energy intake based on any self-report tool is highly biased, while estimates of other dietary components, such as protein and potassium, are less biased (Freedman et al., 2014, 2015). Furthermore, high-quality validation studies are challenging to conduct (Frongillo et al., 2019; Kirkpatrick et al., 2019), and data on the performance of dietary assessment tools in diverse populations are lacking. The absence of reference measures for true intake is a limiting factor in validation (Frongillo et al., 2019; Kirkpatrick et al., 2019). Although there is attention to the discovery of new recovery (Kaaks et al., 2002), predictive (Tasevska et al., 2005), and concentration biomarkers, with the potential
to improve the estimation of dietary intake and associations with health and health outcomes, biomarkers also have measurement error and do not provide contextual information about meal patterning, the food source, or foods consumed concomitantly (Subar et al., 2015).
Additional considerations in the interpretation of dietary intake data relate to the underlying databases (Obbagy et al., 2022). The Food and Nutrient Database for Dietary Studies and the Food Patterns Equivalents Database (Ahuja et al., 2013; USDA, 2021a,b) are updated biennially but are not able to keep pace with rapid changes in the food supply. Thus, they may not represent foods consumed, including by individuals from different cultures (Ahuja et al., 2013). Moreover, sampling foods for nutrient analyses is costly (Ahuja et al., 2013), posing a challenge to comprehensive analyses. Further, as noted by the 2017 report committee, these databases use average composition values and do not sufficiently account for variability in composition (e.g., variability in the sodium content of different brands of a given food) (NASEM, 2017a). These limitations contribute to measurement error when estimating dietary intake using self-report tools and pose a barrier to accounting for heterogeneity in food composition (e.g., in food pattern modeling).
Dietary intake data summary
Dietary intake data are critical inputs in the development of the DGA, although they are challenging to collect and interpret. Their limitations can be mitigated by applying appropriate expertise in their collection, analysis, and interpretation (see NASEM, 2017a, Box 4-4). In addition to biomarker discovery, technology-enabled dietary assessment tools are an active area of research (Boushey et al., 2017; Eldridge et al., 2019), highlighting the need for the DGA to be agile enough to be open to the possibility of incorporating additional sources of dietary intake data in the future.
Studies of Diet and Health Are Often Observational
The work of NESR, and much of the work of the DGAC, involves synthesizing existing evidence about the associations of dietary components, behaviors, and patterns with health outcomes. Ultimately, the strength of the DGAC’s conclusions depends on the strength of the available evidence base.
Evidence hierarchies generally rank randomized controlled trials (RCTs) above observational study designs in terms of study strength (Siegfried, 2017). Confounding is a substantial concern in observational studies, given that diet quality tracks with many other strong risk factors for diseases (including sociodemographic characteristics, physical activity level, body weight, smoking, and sleep). The 2017 report committee
similarly recognized the strengths of RCTs in minimizing confounding and providing strong evidence for causality (NASEM, 2017a). However, the committee also recognized that:
Given that many nutrition studies use observational designs and the populations and settings included more closely reflect the real world, these observational studies can provide other important insights that are complementary to the results of randomized trials. In addition, observational designs are employed when randomized trials cannot be conducted for reasons such as ethical concerns or logistical challenges (NASEM, 2017a).
Thus, the 2017 report committee supported NESR’s practice of including evidence from RCTs and nonrandomized trials as well as from observational studies (including prospective and retrospective cohort studies, controlled before/after studies, and nested case-control studies) (NASEM, 2017a).
Beyond the concerns about feasibility and generalizability noted by that committee, there are other reasons why RCTs may fall short of the ideal (Vorland et al., 2021). RCTs may be subject to bias, especially when blinding of exposure status is difficult. RCTs can be used to study only a single or small number of exposures at a specified dose, duration, and timing; often have a study population that does not represent the population at large; and may not capture the exposure ranges of interest.
Additionally, analytic approaches exist that can minimize or overcome some of the major issues with observational studies. Mendelian randomization uses genetic variants as proxies for environmental exposures of interest, including diet. Given that associations derived from Mendelian randomization analysis are less likely to be affected by confounding and reverse causation, several studies use this approach to examine the causal effects of diet on a variety of diseases (Larsson, 2021). Sibling studies compare outcomes between siblings with differences in the exposure of interest and are likely to reduce confounding because other genetic, environmental, and behavioral variables are likely more similar within than between families. For example, sibling studies have been used to compare early life exposures such as breastfeeding (Adair, 2009). The 2020 DGAC Scientific Report includes both sibling studies and Mendelian randomization studies for some systematic reviews (DGAC, 2020; NASEM, 2022), but there might be other instances where the inclusion of these designs may be appropriate. Other rigorous approaches include quasi-experimental designs that take advantage of natural experiments (such as the implementation of a new policy) and the application of modern statistical analytic methods that allow demonstration of causal inference using observational data, including clear specification of hypothesized
relationships (e.g., using a directed acyclic graph) (Davey Smith, 2019; Glass et al., 2013; Visontay et al., 2021).
Previous high-impact, public-health initiatives have not needed to rely on evidence from RCTs. Multiple examples of this scenario exist outside of nutrition, including tobacco control and seatbelt use. Inevitably, at least some, and most likely the majority, of evidence contributing to the DGA for the foreseeable future will be derived from observational studies. Therefore, it is important for those developing the DGA to continue to seek, include, and promote rigorous observational designs and analytic approaches.
How Different Types of Data Are Used Together in the Dietary Guidelines
In this portion of the chapter, the committee briefly considers how the data were used together to create the 2020 DGAC Scientific Report (DGAC, 2020). The four ways that data are used in the DGA process were described in Chapter 6 of the 2017 National Academies report (NASEM, 2017a) and are shown in Box 2-2.
To answer the key questions posed in each chapter of the 2020 DGAC Scientific Report, the DGAC used various data sources and cross-referenced other chapters as necessary to arrive at its conclusions. The 2020–2025 DGA provides a process model that describes the three approaches—data analysis, food pattern modeling, and NESR systematic reviews—that were used to examine the evidence assembled by the DGAC.5 According to the Departments, “Each of these approaches has its own rigorous, protocol-driven methodology, and plays a unique, complementary role in examining the science” (USDA/HHS, 2020). Data analyses employed national data sets to describe the current health status and dietary intakes of Americans. Food pattern modeling estimated how changes in the amounts or types of foods and beverages in a food pattern would affect the ability of various population subgroups to meet their nutrient needs. NESR systematic reviews were used to answer specific questions about how diet and health are related (USDA/HHS, 2020).
To illustrate this process, the committee considered the example of added sugars. Guidance related to added sugars has changed across the last four DGAC Scientific Reports as the DGACs considered how to handle non-nutritive sources of energy—from “discretionary calorie allowance” (2005), to “solid fats and added sugars” (2010), and finally to a specified
___________________
5 Process models are graphical representations of existing or planned processes and are excellent resources for understanding what functions are performed and how those functions interact. https://irp.fas.org/doddir/army/strap/strpsec3.htm (accessed June 5, 2022).
limit on the amount of energy specifically from added sugars—less than 10 percent in 2015 and less than 6 percent in 2020.
To inform its conclusion related to added sugars, the 2020 DGAC considered three types of data (Figure 2-1), including:
- analyses of dietary intake data from NHANES WWEIA to determine the estimated proportion of energy from the top sources of added sugars;
- systematic reviews of the relationship between added sugars consumption and growth, body size, weight, cardiovascular disease, and type 2 diabetes; and
- food pattern modeling to estimate the “amount of added sugars that can be accommodated in a healthy diet.” Food pattern modeling for individuals aged 2 years and older was conducted across 12 calorie levels, accounting for needs associated with pregnancy and lactation. Separate food pattern modeling was conducted for children younger than 24 months of age (DGAC, 2020).
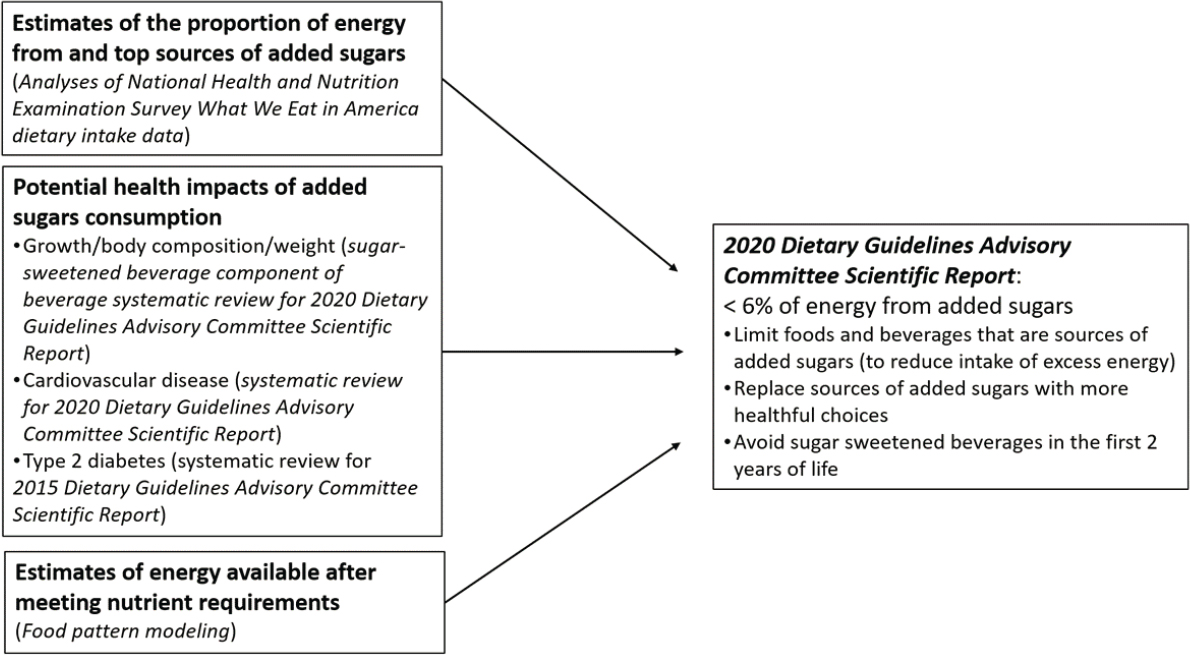
SOURCE: DGAC, 2020.
The 2020 DGAC Scientific Report noted that the totality of evidence across systematic reviews “generally support conclusions that favor limiting the consumption of added sugars to, at most, very low amounts” (DGAC, 2020). This was viewed as particularly important given the high proportion of energy contributed by added sugars to the diets of children and adults based on the NHANES WWEIA analyses (DGAC, 2020). Based on a slight decrease in the consumption of added sugars over the past decade, the 2020 DGAC Scientific Report concluded that a reduction is feasible (DGAC, 2020). The food pattern modeling suggested that after meeting food group and nutrient needs, 6 percent or less of energy intake was available for consumption of added sugars at most energy levels. The 2020 DGAC Scientific Report thus suggested “that for adults and children ages 2 years and older, a recommendation of less than 6 percent of energy from added sugars is more consistent with a dietary pattern that is nutritionally adequate while avoiding excess energy intake than is a pattern with less than 10 percent energy from added sugars” (DGAC, 2020). Avoiding added sugars during the first 2 years of life was recommended based on separate food pattern modeling.
The example in Figure 2-1 is relatively simple, but it still illustrates the complexity inherent to the process of integrating and interpreting multiple types of data to develop dietary guidance. The 2017 National Academies report notes:
Properly evaluating and calibrating results from a variety of data sources and methodological approaches is critical to understanding and interpreting the body of evidence to arrive at appropriate conclusions, as all study designs have innate limitations and can be susceptible to different types of bias.…Taking the limitations of evidence sources into account is crucial for building guidelines that are based on the totality of scientific evidence. (NASEM, 2017a)
The 2020 DGAC Scientific Report contains a chapter titled “Integrating the Evidence” that notes that the “Dietary patterns provide a framework for the dietary guidelines recommendations within and across life stages” (DGAC, 2020). That chapter describes the types of data that were considered but not how they were integrated. Further, while analytic frameworks were developed for each methodology (e.g., for each systematic review) and a process model is available in the 2020–2025 DGA, an overarching analytic framework to guide how different types of evidence are integrated through the process is lacking. The 2017 National Academies report noted that although recent editions of the DGAC Scientific Report have used conceptual frameworks, an analytic framework would be useful “to structure topic selection, synthesis, and interpretation of the evidence” (NASEM, 2017a). The report further noted that “Such an analytic
framework would help clarify the purpose of each question, the relationship between questions, what questions are answerable, and what topics need evidence” (NASEM, 2017a). An overarching analytic framework could also clarify how the DGAC weighs different types of evidence—including additional inputs, such as prior DGAC Scientific Reports, prior editions of the DGA, and public comments submitted during the DGAC review period—to arrive at its interpretations and conclusions (DGAC, 2020).
Summary of Complexities Related to the Process of Creating the Dietary Guidelines for Americans
The DGA process is time-constrained, complicated to administer, and requires evaluating and interpreting complex data. These data come from diverse sources collected using various methods at varying times for different purposes. Although some of these data were collected at least in part to support development of the DGA, the systematic reviews that support the DGA also consist of studies that were conducted for the authors’ purposes but were then selected and reviewed to support the needs of the DGA process. The challenges of using all these forms of data simultaneously underscore the need for an overarching analytic framework with integrative approaches to make the most of the data that are or could be used for the development of the DGA.
SUPPORTING THE RIGOR AND INTEGRITY OF THE DIETARY GUIDELINES BY ADDRESSING THE COMPLEXITIES THROUGH SYSTEMS SCIENCE
The report Redesigning the Process for Establishing the Dietary Guidelines for Americans proposed that the federal agencies invest in and employ newer scientific approaches that can help describe and address the complex systems involved in nutrition and health, namely, systems science approaches (NASEM, 2017a). In this section of this chapter, the committee explores such systems science approaches with an eye toward opportunities that have arisen since publication of the 2017 National Academies report.
Rationale for Using Systems Science Approaches
Without systems science approaches (Box 2-3), it can be difficult to disentangle the complex multifactorial relationships between dietary intake and health that occur throughout a person’s lifetime and account for important population-level health characteristics, including health
disparities (Dangour et al., 2017). These relationships involve many complex systems that cross various scales, ranging from physiologic systems in an individual to environmental systems, including policies that affect populations (Langellier et al., 2019; Friel et al., 2017). This complexity was reflected in the 2017 National Academies report, which underscored “the multifactorial and interconnected nature of diet, health, and a host of other non-dietary factors” (NASEM, 2017a). The DGA have increasingly acknowledged the complexity of dietary intake and its relation to health. Examples of such acknowledgments include the focus on dietary patterns by the 2010 DGAC and the inclusion of conceptual maps to consider the broad array of possible determinants of diet, physical activity, and health and related outcomes in the 2010–2015 DGA (USDA/HHS, 2010) and 2015–2020 DGA (HHS/USDA, 2015). The 2020–2025 DGA included specific recommendations for all life stages for the first time, including infants and toddlers and pregnant and lactating individuals (USDA/HHS, 2020), which recognizes that dietary intake is dynamic across the life cycle.
Nonetheless, the 2017 National Academies report noted that methods such as the current approach to food pattern modeling are not capable of addressing the full extent of complexity of dietary intake and health or the variability in dietary patterns in the population (NASEM, 2017a). Further, the process of developing guidance for dietary intake in a diverse population is complex and involves many stakeholders and the integration of various sources of data, all with different strengths and limitations. There is thus a growing need for analytic frameworks and tools that can appropriately manage the complexity of both dietary intake as an exposure and its relationships with health, as well as the process of developing guidance for dietary intake in a diverse population.
Accordingly, the application of systems science to the DGA, as recommended in the 2017 National Academies report, has become more urgent. Indeed, the approaches used to date “are not designed to understand how strings of actions, reactions, and new actions among multiple health-relevant sectors and diet may affect an overall system outcome such as weight and chronic disease occurrence” (NASEM, 2017a). The 2017 National Academies report encouraged use of systems thinking, mapping, and modeling to expand “knowledge of the diet-health relationships of interest, inform the translation of the guidelines to maximize impact, and identify relevant connections across stakeholders,” as well as to prioritize research and data collection needs (NASEM, 2017a). Figure 2-2 demonstrates that:
implementation of systems approaches for the DGA involves an organized, iterative process in which an initial systems map is generated, which in turn serves as a blueprint for the systems models. It then
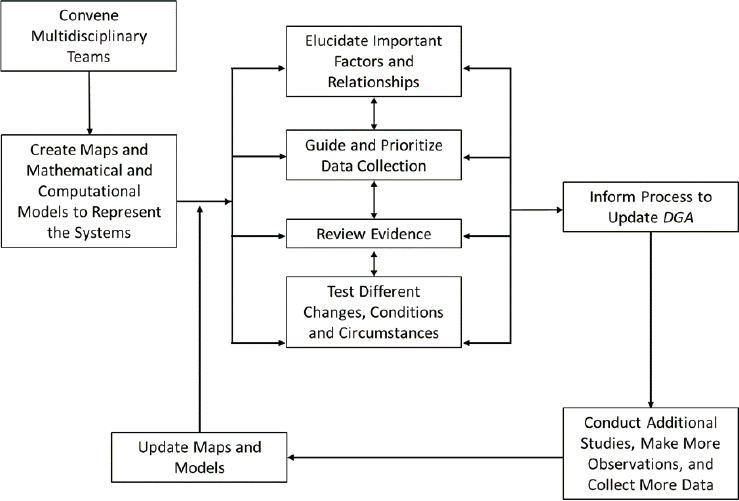
SOURCE: Adapted from NASEM, 2017a.
guides data collection and study design and implementation, generating more data to further augment and refine the systems maps and models. (NASEM, 2017a)
The 2017 National Academies report also indicated that:
because the systems involved are complex, fully comprehending these systems will take time over multiple cycles of the DGA. An initial representation or model of a system will help guide subsequent scientific exploration and data collection, which in turn can further develop the model. (NASEM, 2017a)
This suggests that instead of waiting for further information and the development of additional capabilities, integration of systems science can occur immediately.
Computational methods and capabilities have continued to evolve since release of the 2017 National Academies report, enabling a greater range of approaches to data collection and study design that can better characterize the complex systems involved. At the same time, there has been exponential growth in the amount of data available on the systems
of factors that affect diet and health, including so-called Big Data—data that are not collected to address a specific hypothesis but rather as part of daily activities and processes (Lee et al., 2021). Moreover, there has been increasing acknowledgment of the diversity that exists in our society, how this diversity may affect and be affected by nutrition, and stark inequities among various population subgroups, all of which further contribute to the complexity of the systems involved (Zorbas et al., 2021).
Recently, there has been a movement toward precision medicine and precision health (Collins and Varmus, 2015), which involves finding ways to better tailor health-related recommendations and treatments to the specific characteristics and circumstances of different people. The National Institutes of Health (NIH) indicated that precision nutrition was a priority when it convened a Precision Nutrition: Research Gaps and Opportunities Workshop in January 2021 (NIDDK, 2021). Importantly, better tailoring of nutrition-related recommendations and dietary guidelines to individuals (personalization) and life stages requires a better understanding of the biological, behavioral, social environmental, and other complex systems interactions that affect dietary intake and the relationships between nutrition and various health outcomes (O’Sullivan et al., 2018). Without better understanding and addressing these systems via systems science approaches and methods, such tailoring attempts may potentially fail, leading to inappropriate conclusions that may introduce bias and worsen inequities.
Utility of Systems Science Approaches
Incorporating systems science approaches and methods can support the rigor and integrity of the DGA in numerous ways and lead to “making dietary recommendations as transparent, applicable, and robust as possible” (NASEM, 2017a). First, systems science calls for and utilizes these broader, more diverse, and more systems-oriented data and study designs that better represent the complex systems of factors that may affect diet, nutrition, and health (e.g., Lee et al., 2021), which can help the DGA process. Second, systems science can provide an array of new systems methods and techniques (such as agent-based modeling, system dynamics modeling, social network analysis) to elucidate the complex relationships between diet, nutrition, and health more effectively and account for these relationships in the DGA (Ananiadou, et al., 2009; Kiekens, et al., 2022; Olsina, et al., 2019). Third, systems science can help show the impact of changing circumstances (e.g., changes in the global food system, or changes in the local built environment) on short-term and longer-term health outcomes (Popkin, 2017; Wang, et al., 2014). Fourth, as shown in the iterative approach in Figure 2-2, systems science approaches can help to identify and prioritize questions of interest and data gaps.
Fifth, systems science can support precision nutrition and thus dietary recommendations that are more relevant to diverse populations. Finally, because the process to create the DGA is a system in and of itself, it can help better integrate the entire DGA process, showing how all the parts and processes fit together through an overarching analytic framework.
All of this means that it is essential to expand the use of various systems approaches and methods beyond their prior uses and what may be perceived as their primary uses. For example, the utility of systems modeling for the DGAC goes far beyond simple forecasting of intervention outcomes on health (Epstein, 2008). Systems modeling can be used to answer questions that cannot be addressed by clinical trials and observational studies (Luke and Stamatakis, 2012). For example, systems modeling can help reveal the underlying causal mechanisms driving health outcomes (Hedström and Ylikoski, 2010). This is particularly important in the complex areas of diet, nutrition, and health (Galea et al., 2010). Systems modeling helps identify important data gaps that can then be filled by traditional nutrition, clinical, and epidemiologic studies (Mabry et al., 2022). Systems approaches are also useful for exploring heterogeneity and identifying inter-individual variability and the ranges of potential outcomes. For example, systems approaches allow for a better connection of dietary intake patterns with more distal, future health outcomes. Systems approaches also hold promise for understanding how physical, social, historical, and political contexts shape individual behavior and health. This could lead to better tailoring of dietary recommendations for different people and populations, eventually leading to greater health equity (Kasman et al., 2021). Finally, using systems mapping can support a more iterative approach to the development of the DGA, so each new edition of the DGA could build progressively and efficiently on prior versions (Lee et al., 2017).
Recent Applications of Systems Science to Nutrition
The 2017 National Academies report noted that the application of systems science was still in its infancy within the field of nutrition (NASEM, 2017a). There had been several calls to increase the use of systems approaches within public health (Green, 2006; Mabry et al., 2010) as well as nutrition (Bennett et al., 2015; Hammond and Dubé, 2012; Lee et al., 2017). Such calls may have prompted further applications of systems mapping and modeling methods to nutrition-related topics since the 2017 National Academies report was published. Table 2-1 provides a short list of recent applications of systems approaches to nutrition, equity, food security, body weight, and the food system. These, and other similar examples provide a foundation on which the DGA process can build.
TABLE 2-1 Example of Applications of Systems Mapping and Modeling Relevant to the Dietary Guidelines for Americans
| Nutrition Topic | Title and Reference |
|---|---|
| Diet and nutrition |
Complex systems approaches to diet: A systematic review (Langellier et al., 2019)
Can following formula-feeding recommendations still result in infants who are overweight or have obesity? (Ferguson et al., 2020) |
| Body weight |
Integrating complex systems methods to advance obesity prevention intervention research (Hennessy et al., 2020)
The spread of obesity in a large social network over 32 years (Christakis and Fowler, 2007) Applications of social network analysis to obesity: A systematic review (Zhang et al., 2018) |
| Food security |
A systems science perspective and transdisciplinary models for food and nutrition security (Hammond and Dubé, 2012)
Using systems science to gain insight into childhood food security in the United States: Report of an expert mapping workshop (Fleischer et al., 2018) |
| Equity |
An agent-based model of income inequalities in diet in the context of residential segregation (Auchincloss et al., 2011)
Food system dynamics structuring nutrition equity in racialized urban neighborhoods (Freedman et al., 2022) Dynamics of the complex food environment underlying dietary intake in low-income groups: A systems map of associations extracted from a systematic umbrella literature review (Sawyer et al., 2021) |
| Global food systems |
Use of the delta model to understand the food system and global nutrition (Smith et al., 2021)
Relationship between shifts in food system dynamics and acceleration of the global nutrition transition (Popkin, 2017) The value of a food system approach. In the book: Food security and global environmental change (Ericksen et al., 2010) |
In addition to the nutrition-specific academic work highlighted in Table 2-1, there is a much larger body of research using systems approaches in public health and the clinical sciences that can inform USDA and HHS as they adopt and integrate systems mapping and modeling. This body includes important applications in infectious disease (particularly HIV/AIDS and more recently COVID-19), physical activity, chronic disease prevention, health care delivery, disparities, equity, and inclusion.
Further, the Departments can draw on expertise and experience from other government agencies and scientific institutions that have funded, directed, or participated in systems science initiatives. These include numerous panels and reports from the National Academies (IOM, 2015; NASEM, 2021; NRC, 2008) and the NIH (NCI, 2019; NIDDK, 2021; NIH, 2018; PLOS, 2022).
Summary of Supporting the Dietary Guidelines for Americans Through Systems Science
Developing dietary guidance for a diverse population is a complex process, with numerous components that may interact in unexpected ways. Systems approaches can support the DGA to reflect the complexity inherent in dietary intake and its relationships with various health outcomes over time, including heterogeneity across the population and changes over the life cycle. Application of systems approaches to the process itself can facilitate efficiencies to create a continuous learning process whereby each version of the DGA can build more seamlessly on the prior version and create transparency in how different types of data are integrated and interpreted. Several recent applications related to nutrition illustrate how the DGA process can begin to leverage systems science approaches and methods (Table 2-1).
CONCLUSIONS
In this chapter, the committee discussed the context for key issues that were addressed by the 2017 National Academies Report (NASEM, 2017a). The 2017 report emphasized the low adherence by the public to the DGA and the relationship between low adherence and a lack of trust in the recommendations (NASEM 2017a). Although there were certain controversial topics in the 2015–2020 DGA, issues surrounding trust in nutrition science continue to challenge the nutrition research community and the trustworthiness of the DGA. Given the erosion of trust in expertise in general and public attention on COI in nutrition research, these issues are likely to persist in future DGA cycles. Therefore, transparent management of both individual and institutional COI will remain important for maintaining and increasing public trust in the DGA.
The DGA process is time-restricted, complicated to administer, and requires the evaluation, synthesis, integration, and interpretation of complex data. The data used for systematic reviews, food pattern modeling, and descriptive analysis come from a variety of sources and are collected at varying times and for different purposes. Many of these data are collected at least in part to support development of the DGA. The
challenges of using all these forms of data simultaneously underscore the need for both an overarching analytic framework and integrative approaches.
Systems science approaches and methods can support the DGA to reflect the complexity inherent in dietary intake, including heterogeneity across the population and changes over a person’s life cycle, and its relationships with various health outcomes over time. Application of systems science approaches and methods to the DGA process itself can facilitate efficiencies to create a continuous learning process whereby each version of the DGA can build more seamlessly on the prior version and create transparency in how different types of data are integrated and interpreted. Systems science approaches and methods would help to make the most of the data that are or could be used for the development of the DGA as well as to support transparency in the DGAC’s interpretation of the evidence (NASEM, 2017a).
REFERENCES
Achterberg, C., A. Astrup, D. M. Bier, J. C. King, R. M. Krauss, N. Teicholz, and J. S. Volek. 2022. An analysis of the recent US dietary guidelines process in light of its federal mandate and a National Academies report. PNAS Nexus 1(3).
Adair, L. S. 2009. Methods appropriate for studying the relationship of breast-feeding to obesity. Journal of Nutrition 139(2):408S-411S.
Ahmed, E., A. S. Elgazzar, and A. S. Hegazi. 2005. An overview of complex adaptive systems. https://arxiv.org/pdf/nlin/0506059.pdf (accessed September 22, 2022).
Ahuja, J. K. C., A. J. Moshfegh, J. M. Holden, and E. Harris. 2013. USDA food and nutrient databases provide the infrastructure for food and nutrition research, policy, and practice. Journal of Nutrition 143(2):241s-249s.
Ananiadou, S., B. Rea, N. Okazaki, R. Procter, and J. Thomas. 2009. Supporting systematic reviews using text mining. Social Science Computer Review 27(4):509-523.
Auchincloss, A. H., R. L. Riolo, D. G. Brown, J. Cook, and A. V. Diez Roux. 2011. An agent-based model of income inequalities in diet in the context of residential segregation. American Journal of Preventative Medicine 40(3):303-311.
Bailey, R. L., J. D. Ard, T. A. Davis, T. S. Naimi, B. O. Schneeman, J. S. Stang, K. G. Dewey, S. M. Donovan, R. Novotny, L. G. Snetselaar, J. de Jesus, K. O. Casavale, T. Pannucci, and E. E. Stoody. 2021. A proposed framework for identifying nutrients and food components of public health relevance in the Dietary Guidelines for Americans. Journal of Nutrition 151(5):1197-1204.
Beaton, G. H., J. Burema, and C. Ritenbaugh. 1997. Errors in the interpretation of dietary assessments. American Journal of Clinical Nutrition 65(4 Suppl):1100s-1107s.
Bennett, B. J., K. D. Hall, F. B. Hu, A. L. McCartney, and C. Roberto. 2015. Nutrition and the science of disease prevention: A systems approach to support metabolic health. Annals of the New York Academy of Sciences 1352:1-12.
Boushey, C. J., M. Spoden, F. M. Zhu, E. J. Delp, and D. A. Kerr. 2017. New mobile methods for dietary assessment: Review of image-assisted and image-based dietary assessment methods. Proceedings of the Nutrition Society 76(3):283-294.
CDC (Centers for Disease Control and Prevention). 2017. About the National Health and Nutrition Examination Survey. https://www.cdc.gov/nchs/nhanes/about_nhanes.htm (accessed September 22, 2022).
CDC. 2021. National Health and Nutrition Examination Survey. 2017-March 2020 data documentation, codebook, and frequencies. https://wwwn.cdc.gov/Nchs/Nhanes/2017-2018/P_DEMO.htm#RIDRETH1 (accessed September 22, 2022).
Christakis, N. A., and J. H. Fowler. 2007. The spread of obesity in a large social network over 32 years. New England Journal of Medicine 357(4):370-379.
Collins, F. S., and H. Varmus. 2015. A new initiative on precision medicine. New England Journal of Medicine 372(9):793-795.
Dangour, A. D., G. Mace, and B. Shankar. 2017. Food systems, nutrition, health and the environment. Lancet Planet Health 1(1):e8-e9.
Davey Smith, G. 2019. Post-modern epidemiology: When methods meet matter. American Journal of Epidemiology 188(8):1410-1419.
DGA (Dietary Guidelines for Americans). n.d. Who’s involved in updating the Dietary Guidelines. https://www.dietaryguidelines.gov/about-dietary-guidelines/process#:~:text=Developing%20the%20Dietary%20Guidelines%20involves%20a%20step-by-step%20process,team%20of%20Federal%20staff%20from%20USDA%20and%20HHS (accessed September 22, 2022).
DGAC (Dietary Guidelines Advisory Committee). 2010. Scientific report of the 2010 Dietary Guidelines Advisory Committee: Advisory report to the Secretary of Health and Human Services and the Secretary of Agriculture. Washington, DC: U.S. Department of Agriculture, Agricultural Research Service.
DGAC. 2015. Scientific report of the 2015 Dietary Guidelines Advisory Committee: Advisory report to the Secretary of Health and Human Services and the Secretary of Agriculture. Washington, DC: U.S. Department of Agriculture, Agricultural Research Service. https://health.gov/sites/default/files/2019-09/Scientific-Report-of-the-2015-Dietary-Guidelines-Advisory-Committee.pdf (accessed September 22, 2022).
DGAC. 2020. Scientific report of the 2020 Dietary Guidelines Advisory Committee: Advisory report to the Secretary of Health and Human Services and the Secretary of Agriculture. Washington, DC: U.S. Department of Agriculture, Agricultural Research Service. https://www.dietaryguidelines.gov/2020-advisory-committee-report (accessed September 22, 2022).
Dodd, K. W., P. M. Guenther, L. S. Freedman, A. F. Subar, V. Kipnis, D. Midthune, J. A. Tooze, and S. M. Krebs-Smith. 2006. Statistical methods for estimating usual intake of nutrients and foods: A review of the theory. Journal of American Dietetic Association 106(10):1640-1650.
Eldridge, A. L., C. Piernas, A. Illner, M. J. Gibney, M. A. Gurinovi´c, J. H. M. de Vries, and J. E. Cade. 2019. Evaluation of new technology-based tools for dietary intake assessment—An ILSI Europe Dietary Intake and Exposure Task Force evaluation. Nutrients 11(1).
Epstein, J. M. 2008. Why model? Journal of Artificial Societies and Social Simulation 11(412).
Ericksen, P., B. Stewart, J. Dixon, D. Barling, P. Loring, M. I. Anderson, and J. Ingram. 2010. The value of a food system approach. In Food security and global environmental change. (pp. 45-65). Oxfordshire, UK: Routledge.
Ferguson, M. C., K. J. O’Shea, L. D. Hammer, D. L. Hertenstein, R. M. Syed, S. Nyathi, M. S. Gonzales, M. Domino, S. Siegmund, S. Randall, P. Wedlock, A. Adam, and B. Y. Lee. 2020. Can following formula-feeding recommendations still result in infants who are overweight or have obesity? Pediatric Research 88(4):661-667.
Fleischer, N. L., A. D. Liese, R. Hammond, A. Coleman-Jensen, C. Gundersen, J. Hirschman, E. A. Frongillo, X. Ma, N. Mehta, and S. J. Jones. 2018. Using systems science to gain insight into childhood food security in the United States: Report of an expert mapping workshop. Journal of Hunger & Environmental Nutrition 13(3):362-384.
Freedman, D. A., J. K. Clark, D. W. Lounsbury, L. Boswell, M. Burns, M. B. Jackson, K. Mikelbank, G. Donley, Q. Worley-Bell, J. Mitchell, T. H. Ciesielski, M. Embaye, E. K. Lee, A. Roche, I. Gill, and O. Yamoah. 2022. Food system dynamics structuring nutrition equity in racialized urban neighborhoods. American Journal of Clinical Nutrition 115(4):1027-1038.
Freedman, L. S., A. Schatzkin, D. Midthune, and V. Kipnis. 2011. Dealing with dietary measurement error in nutritional cohort studies. Journal of National Cancer Institute 103(14):1086-1092.
Freedman, L. S., J. M. Commins, J. E. Moler, L. Arab, D. J. Baer, V. Kipnis, D. Midthune, A. J. Moshfegh, M. L. Neuhouser, R. L. Prentice, A. Schatzkin, D. Spiegelman, A. F. Subar, L. F. Tinker, and W. Willett. 2014. Pooled results from 5 validation studies of dietary self-report instruments using recovery biomarkers for energy and protein intake. American Journal of Epidemiology 180(2):172-188.
Freedman, L. S., J. M. Commins, J. E. Moler, W. Willett, L. F. Tinker, A. F. Subar, D. Spiegelman, D. Rhodes, N. Potischman, M. L. Neuhouser, A. J. Moshfegh, V. Kipnis, L. Arab, and R. L. Prentice. 2015. Pooled results from 5 validation studies of dietary self-report instruments using recovery biomarkers for potassium and sodium intake. American Journal of Epidemiology 181(7):473-487.
Friel, S., M. Pescud, E. Malbon, A. Lee, R. Carter, J. Greenfield, M. Cobcroft, J. Potter, L. Rychetnik, and B. Meertens. 2017. Using systems science to understand the determinants of inequities in healthy eating. PLoS One 12(11):e0188872.
Frongillo, E. A., T. Baranowski, A. F. Subar, J. A. Tooze, and S. I. Kirkpatrick. 2019. Establishing validity and cross-context equivalence of measures and indicators. Journal of the Academy of Nutrition and Dietetics 119(11):1817-1830.
Funk, C., M. Hefferon, B. Kennedy, and C. Johnson. 2019. Trust and mistrust in Americans’ views of scientific experts. Pew Research Center. https://www.pewresearch.org/science/2019/08/02/trust-and-mistrust-in-americans-views-of-scientific-experts/ (accessed June 6, 2022).
Galea, S., M. Riddle, and G. A. Kaplan. 2010. Causal thinking and complex system approaches in epidemiology. International Journal of Epidemiology 39(1):97-106.
Garza, C., P. J. Stover, S. D. Ohlhorst, M. S. Field, R. Steinbrook, S. Rowe, C. Woteki, and E. Campbell. 2019. Best practices in nutrition science to earn and keep the public’s trust. American Journal of Clinical Nutrition 109(1):225-243.
Glass, T. A., S. N. Goodman, M. A. Hernán, and J. M. Samet. 2013. Causal inference in public health. Annual Review of Public Health 34:61-75.
Goldman, T. R. 2016. Final 2015-20 Dietary Guidelines for Americans. Health Affairs. https://www.healthaffairs.org/do/10.1377/hpb20160331.683121/full/ (accessed June 6, 2022).
Green, L. W. 2006. Public health asks of systems science: To advance our evidence-based practice, can you help us get more practice-based evidence? American Journal of Public Health 96(3):406-409.
Hammond, R. A., and L. Dubé. 2012. A systems science perspective and transdisciplinary models for food and nutrition security. Proceedings of the National Academy of Sciences 109(31):12356-12363.
Harvard T. H. Chan School of Public Health. 2021. Dietary Guidelines for Americans 2020 released. Harvard: The Nutrition Source https://www.hsph.harvard.edu/nutritionsource/2021/01/12/2020-dietary-guidelines/ (accessed September 22, 2022).
Hedström, P., and P. Ylikoski. 2010. Causal mechanisms in the social sciences. Annual Review of Sociology 36(1):49-67.
Hennessy, E., C. D. Economos, and R. A. Hammond. 2020. Integrating complex systems methods to advance obesity prevention intervention research. Health Education and Behavior 47(2):213-223.
HHS/USDA (U.S. Department of Health and Human Services/U.S. Department of Agriculture). 2015. 2015–2020 Dietary Guidelines for Americans, 8th ed. https://health.gov/ourwork/food-nutrition/previous-dietary-guidelines/2015 (accessed September 22, 2022).
Ioannidis, J. P. A., and J. F. Trepanowski. 2018. Disclosures in nutrition research: Why it is different. Journal of the American Medical Association 319(6):547-548.
IOM (Institute of Medicine). 2000. Dietary Reference Intakes: Applications in dietary assessment. Washington, DC: The National Academies Press. https://doi.org/10.17226/9956 (accessed September 22, 2022).
IOM. 2006. Food marketing to children and youth: Threat or opportunity? Washington, DC: The National Academies Press. https://doi.org/10.17226/11514 (accessed September 22, 2022).
IOM. 2011. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press. https://doi.org/10.17226/13058 (accessed September 22, 2022).
IOM. 2015. Assessing the Use of Agent-Based Models for Tobacco Regulation. Washington, DC: The National Academies Press. https://doi.org/10.17226/19018.
Jacobs, A. 2020. Scientific panel on new Dietary Guidelines draws criticism from health advocates. New York Times. https://www.nytimes.com/2020/06/17/health/dietnutrition-guidelines.html (accessed September 22, 2022).
Jahns, L., L. McDonald, A. Wadsworth, C. Morin, Y. Liu, and T. Nicklas. 2015. Barriers and facilitators to following the Dietary Guidelines for Americans reported by rural, northern plains American-Indian children. Public Health Nutrition 18(3):482-489.
Kaaks, R., P. Ferrari, A. Ciampi, M. Plummer, and E. Riboli. 2002. Uses and limitations of statistical accounting for random error correlations, in the validation of dietary questionnaire assessments. Public Health Nutrition 5(6a):969-976.
Kasman, M., N. Breen, and R. A. Hammond. 2021. Complex systems science. In The science of health disparities research. Hoboken, NJ: Wiley-Blackwell. Pp. 243-256.
Kiekens, A., B. Dierckx de Casterlé, and A. Vandamme. 2022. Qualitative systems mapping for complex public health problems: A practical guide. PLoS One 17(2):e0264463.
Kirkpatrick, S. I., T. Baranowski, A. F. Subar, J. A. Tooze, and E. A. Frongillo. 2019. Best practices for conducting and interpreting studies to validate self-report dietary assessment methods. Journal of the Academy of Nutrition and Dietetics 119(11):1801-1816.
Kirkpatrick, S. I., P. M. Guenther, A. F. Subar, S. M. Krebs-Smith, K. A. Herrick, L. S. Freedman, and K. W. Dodd. 2022. Using short-term dietary intake data to address research questions related to usual dietary intake among populations and subpopulations: Assumptions, statistical techniques, and considerations. Journal of Academy of Nutrition and Dietetics 122(7):1246-1262.
Langellier, B. A., U. Bilal, F. Montes, J. D. Meisel, L. Cardoso, and R. A. Hammond. 2019. Complex systems approaches to diet: A systematic review. American Journal of Preventative Medicine 57(2):273-281.
Larsson, S. C. 2021. Mendelian randomization as a tool for causal inference in human nutrition and metabolism. Current Opinion in Lipidology 32(1):1-8.
Lee, B. Y., S. M. Bartsch, Y. Mui, L. A. Haidari, M. L. Spiker, and J. Gittelsohn. 2017. A systems approach to obesity. Nutrition Reviews 75(suppl 1):94-106.
Lee, B. Y., M. C. Ferguson, S. N. Cox, and P. H. Phan. 2021. Big data and systems methods: The next frontier to tackling the global obesity epidemic. Obesity (Silver Spring) 29(2):263-264.
Leischow, S. J., and B. Milstein. 2006. Systems thinking and modeling for public health practice. American Journal of Public Health 96(3):403-405.
Lindquist, J., D. M. Thomas, D. Turner, J. Blankenship, and T. K. Kyle. 2021. Food for thought: A natural language processing analysis of the 2020 dietary guidelines public comments. American Journal of Clinical Nutrition 114(2):713-720.
Luke, D. A., and K. A. Stamatakis. 2012. Systems science methods in public health: Dynamics, networks, and agents. Annual Review of Public Health 33:357-376.
Mabry, P. L., S. E. Marcus, P. I. Clark, S. J. Leischow, and D. Méndez. 2010. Systems science: A revolution in public health policy research. American Journal of Public Health 100(7):1161-1163.
Mabry, P. L., N. P. Pronk, C. I. Amos, J. S. Witte, P. T. Wedlock, S. M. Bartsch, and B. Y. Lee. 2022. Cancer systems epidemiology: Overcoming misconceptions and integrating systems approaches into cancer research. PLoS Medicine 19(6):e1004027.
Mayer-Davis, E. J., T. S. Naimi, and R. D. Mattes. 2021. Proposed reductions in limits on added sugar and alcohol for the new dietary guidelines: Our perspective. American Journal of Clinical Nutrition 114(2):405-406.
Milstein, B., J. B. Homer, and Homer Consulting. 2006. Background on system dynamics simulation modeling with a summary of major public health studies. Political Science 96(3): 452-458.
Mobus, G. E., and M. C. Kalton. 2015. Principles of systems science. New York: Springer-Verlag.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2017a. Redesigning the process for establishing the Dietary Guidelines for Americans. Washington, DC: The National Academies Press. https://doi.org/10.17226/24883 (accessed September 22, 2022).
NASEM. 2017b. Optimizing the process for establishing the Dietary Guidelines for Americans: The selection process. Washington, DC: The National Academies Press. https://doi.org/10.17226/24637 (accessed September 22, 2022).
NASEM. 2021. Using systems applications to inform obesity solutions: Proceedings of a workshop. Washington, DC: The National Academies Press. https://doi.org/10.17226/25900 (accessed September 22, 2022).
NASEM. 2022. Evaluating the process to develop the Dietary Guidelines for Americans, 2020–2025: A midcourse report. Washington, DC: The National Academies Press. https://doi.org/10.17226/26406 (accessed September 22, 2022)
NCI (National Cancer Institute). 2019. Workshop to facilitate cancer systems epidemiology research. National Institutes of Health. Bethesda, MD. https://epi.grants.cancer.gov/events/systems-epidemiology/ (accessed September 22, 2022).
Nestle, M. 2018. Perspective: Challenges and controversial issues in the Dietary Guidelines for Americans, 1980-2015. Advances in Nutrition 9(2):148-150.
Nicklas, T. A., L. Jahns, M. L. Bogle, D. N. Chester, M. Giovanni, D. M. Klurfeld, K. Laugero, Y. Liu, S. Lopez, and K. L. Tucker. 2013. Barriers and facilitators for consumer adherence to the Dietary Guidelines for Americans: The health study. Journal of the Academy Nutrition and Dietetics 113(10):1317-1331.
NIDDK (National Institute of Diabetes and Digestive and Kidney Diseases). 2021. Precision nutrition: Research gaps and opportunities workshop. National Institutes of Health. https://www.niddk.nih.gov/news/meetings-workshops/2021/precision-nutrition-workshop (accessed September 22, 2022).
NIH (National Institutes of Health). 2018. BD2K behavioral and social sciences (BSS) and big data workshop. https://commonfund.nih.gov/bssbigdata (accessed September 22, 2022).
NRC (National Research Council). 1986. Nutrient adequacy: Assessment using food consumption surveys. Washington, DC: The National Academies Press. https://doi.org/10.17226/618 (accessed September 22, 2022).
NRC. 2008. Behavioral modeling and simulation: From individuals to societies. Washington, DC: The National Academies Press. https://doi.org/10.17226/12169 (accessed September 22, 2022).
Obbagy, J., R. Raghavan, L. K. English, M. K. Spill, C. L. Bahnfleth, M. Bates, E. Callahan, N. C. Cole, D. Güngör, J. H. Kim, B. J. Kingshipp, J. E. H. Nevins, S. R. Scinto-Madonich, J. M. Spahn, S. Venkatramanan, and E. Stoody. 2022. Strengthening research that answers nutrition questions of public health importance: Leveraging the experience of the USDA nutrition evidence systematic review team. Journal of Nutrition 152(8):1823-1830.
Olsina, L., P. Becker, D. Peppino, and G. Tebes. 2019. Specifying the process model for systematic reviews: An augmented proposal. Journal of Software Engineering Research and Development 7. https://doi.org/10.5753/jserd.2019.460 (accessed September 22, 2022).
O’Sullivan, A., B. Henrick, B. Dixon, D. Barile, A. Zivkovic, J. Smilowitz, D. Lemay, W. Martin, J. B. German, and S. E. Schaefer. 2018. 21st century toolkit for optimizing population health through precision nutrition. Critical Reviews in Food Science and Nutrition 58(17):3004-3015.
PLOS. 2022. NIH systems epidemiology. https://collections.plos.org/collection/nih/ (accessed September 22, 2022).
Popkin, B. M. 2017. Relationship between shifts in food system dynamics and acceleration of the global nutrition transition. Nutrition Reviews 75(2):73-82.
Reedy, J., S. M. Krebs-Smith, R. A. Hammond, and E. Hennessy. 2017. Advancing the science of dietary patterns research to leverage a complex systems approach. Journal of the Academy of Nutrition and Dietetics 117(7):1019-1022.
Reedy, J., A. F. Subar, S. M. George, and S. M. Krebs-Smith. 2018. Extending methods in dietary patterns research. Nutrients 10(5).
Rosenbaum, L. 2017. The march of science—the true story. New England Journal of Medicine 377(2):188-191.
Rudd, n.d. Food marketing. University of Connecticut Rudd Center for Food Policy and Health. https://uconnruddcenter.org/research/food-marketing/ (accessed September 22, 2022).
Sawyer, A. D. M., F. van Lenthe, C. B. M. Kamphuis, L. Terragni, G. Roos, M. P. Poelman, M. Nicolaou, W. Waterlander, S. K. Djojosoeparto, M. Scheidmeir, A. Neumann-Podczaska, and K. Stronks. 2021. Dynamics of the complex food environment underlying dietary intake in low-income groups: A systems map of associations extracted from a systematic umbrella literature review. International Journal of Behavioral Nutrition and Physical Activity 18(1):96.
Siegfried, T. 2017. Philosophical critique exposes flaws in medical evidence hierarchies. ScienceNews. https://www.sciencenews.org/blog/context/critique-medical-evidence-hierarchies (accessed September 22, 2022).
Smith, N. W., A. J. Fletcher, L. A. Dave, J. P. Hill, and W. C. McNabb. 2021. Use of the delta model to understand the food system and global nutrition. Journal of Nutrition 151(10):3253-3261.
Snetselaar, L. G., J. M. de Jesus, D. M. DeSilva, and E. E. Stoody. 2021. Dietary Guidelines for Americans, 2020–2025: Understanding the scientific process, guidelines, and key recommendations. Nutrition Today 56(6):287-295.
Sterman, J. D. 2006. Learning from evidence in a complex world. American Journal of Public Health 96(3):505-514.
Sterman, J. 2010. Business dynamics. New York: Tata McGraw-Hill.
Stoody, E. 2019. Responding to the National Academies of Sciences, Engineering, and Medicine study on the process to update the Dietary Guidelines for Americans. Washington, DC. https://www.dietaryguidelines.gov/sites/default/files/2019-05/Day%201%20Response%20to%20NASEM%20Study.pdf (accessed September 22, 2022).
Subar, A. F., L. S. Freedman, J. A. Tooze, S. I. Kirkpatrick, C. Boushey, M. L. Neuhouser, F. E. Thompson, N. Potischman, P. M. Guenther, V. Tarasuk, J. Reedy, and S. M. Krebs-Smith. 2015. Addressing current criticism regarding the value of self-report dietary data. Journal of Nutrition 145(12):2639-2645.
Sullivan, C. 2021. A look at the 2020–2025 Dietary Guidelines for Americans. Harvard Health Blog. https://www.health.harvard.edu/blog/a-look-at-the-2020-2025-dietary-guidelines-for-americans-2021031022098 (accessed September 22, 2022).
Tasevska, N., S. A. Runswick, A. McTaggart, and S. A. Bingham. 2005. Urinary sucrose and fructose as biomarkers for sugar consumption. Cancer Epidemiology Biomarkers and Prevention 14(5):1287-1294.
Thompson, F. E., and A. F. Subar. 2017. Dietary assessment methodology. In Nutrition in the prevention and treatment of disease, 4th ed. Edited by A. Coulston, C. Boushey, M. Ferruzzi and L. Delahanty. Amsterdam, The Netherlands: Elsevier Inc.
USDA (U.S. Department of Agriculture). 2021a. Food and nutrient database for dietary studies. https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/food-surveys-research-group/docs/fndds/ (accessed September 22, 2021).
USDA. 2021b. Food patterns equivalents database. https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/food-surveys-research-group/docs/fped-overview/ (accessed September 22, 2021).
USDA. n.d.a. Nutrition evidence systematic review roles and responsibilities. https://nesr.usda.gov/roles-and-responsibilities (accessed June 6, 2022).
USDA, n.d.b. Food and nutrient database for dietary studies (FNDDS). https://data.nal.usda.gov/dataset/food-and-nutrient-database-dietary-studies-fndds#:~:text=USDA's%20Food%20and%20Nutrient%20Database,to%20determine%20their%20nutrient%20values (accessed September 22, 2022).
USDA, n.d.c. FPED databases and SAS data sets. https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/food-surveys-research-group/docs/fped-databases/ (accessed September 22, 2022).
USDA/HHS (U.S. Department of Agriculture/U.S. Department of Health and Human Services). 2010. Dietary guidelines for Americans, 2010–2015, 7th ed. https://www.dietaryguidelines.gov/sites/default/files/2019-05/DietaryGuidelines2010.pdf (accessed September 22, 2022).
USDA/HHS. 2020. Dietary guidelines for Americans, 2020–2025, 9th ed. https://dietaryguidelines.gov/ (accessed September 22, 2022).
Visontay, R., M. Sunderland, T. Slade, J. Wilson, and L. Mewton. 2021. Are there non-linear relationships between alcohol consumption and long-term health? Protocol for a systematic review of observational studies employing approaches to improve causal inference. BMJ Open 11(3):e043985.
Vorland, C. J., A. W. Brown, J. A. Dawson, S. L. Dickinson, L. Golzarri-Arroyo, B. A. Hannon, M. Heo, S. B. Heymsfield, W. P. Jayawardene, C. N. Kahathuduwa, S. W. Keith, J. M. Oakes, C. D. Tekwe, L. Thabane, and D. B. Allison. 2021. Errors in the implementation, analysis, and reporting of randomization within obesity and nutrition research: A guide to their avoidance. International Journal of Obesity 45(11):2335-2346.
Wang, Y., H. Xue, L. Esposito, M. J. Joyner, Y. Bar-Yam, and T. T. K. Huang. 2014. Applications of complex systems science in obesity and noncommunicable chronic disease research. Advances in Nutrition 5(5):574-577.
Washington Post. 2021. Opinion: In issuing new dietary guidelines, Trump once again spurns science. Washington Post. https://www.washingtonpost.com/opinions/in-issuing-new-dietary-guidelines-trump-once-again-spurns-science/2021/01/01/d1c81310-4b8c-11eb-a9f4-0e668b9772ba_story.html (accessed September 22, 2022).
Zhang, S., K. de la Haye, M. Ji, and R. An. 2018. Applications of social network analysis to obesity: A systematic review. Obesity Review 19(7):976-988.
Zorbas, C., J. Browne, A. Chung, P. Baker, C. Palermo, E. Reeve, A. Peeters, and K. Backholer. 2021. National nutrition policy in high-income countries: Is health equity on the agenda? Nutrition Reviews 79(10):1100-1113.
This page intentionally left blank.
































