6
Diagnostic Stewardship
LABORATORY CLINICIAN PERSPECTIVE
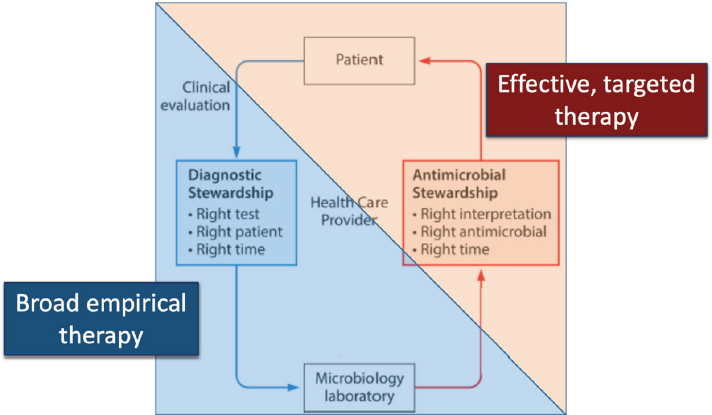
Antimicrobial stewardship and diagnostic stewardship are complementary practices that go hand in hand to guide therapeutic decisions (Figure 6-1), said Carey-Ann Burnham (Chief Clinical Officer, Pattern Bioscience). Diagnostic stewardship can be defined as:
Coordinated guidance and interventions to improve appropriate use of microbiological diagnostics to guide therapeutic decisions. It should promote appropriate, timely diagnostic testing, including specimen collection,
SOURCE: Presented by Carey-Ann Burnham, October 14, 2022; adapted from Messacar et al., 2017.
and pathogen identification and accurate, timely reporting of results to guide patient treatment. (WHO, 2016)
Or stated more simply as, “getting the right test to the right patient at the right time.”
Considerations for determining the right test include: the analytical performance characteristics (e.g., sensitivity/specificity), the diagnostic yield, and the context of local epidemiology. Choosing the right patient depends on appropriate use criteria, the patient population, and pre-test probability. The right time for a test, said Burnham, is a time during which the test results fit into the clinical workflow and will have an impact on patient care. Laboratory management and capacity should also be considered by assessing factors such as space, cost, hands-on time, and throughput.
Diagnostic stewardship occurs throughout the lifecycle of a test, although most decisions are made in the preanalytical phase. At this phase, decisions must be made about test ordering, specimen collection and transport, specimen acceptance and rejection criteria, and specimen processing. The next stage of testing is the analytical phase, said Burnham; at this point, diagnostic stewardship includes decisions about methodology, batch vs. on demand processing, and where a test will be processed (central laboratory vs. point-of-care). Finally, the postanalytical phase includes
considerations about how results are reported, what interpretation is provided, and whether notification of results is active or passive. Conversations about diagnostic stewardship often focus on reducing testing that is inappropriate or unnecessary, said Burnham. However, both overutilization and underutilization of testing are problematic. The failure to order a necessary diagnostic test at the right point in the workflow has been shown to be one of the most common medical errors in ambulatory practice settings (Gandhi et al., 2006).
Diagnostic stewardship—and its relationship with antimicrobial stewardship—will vary depending on the practice setting. Burnham considered practice settings across four categories, each of which has a different approach to diagnostic stewardship: outpatient, emergency department, inpatient, and critical care inpatient. In each of these settings, she said, a diagnostic stewardship program should consider:
- Where will testing take place (e.g., at the patient’s bedside, local laboratory, central laboratory, pharmacy, or patient’s home)?
- Who will collect the specimens and what guidance should they receive?
- How will test results be reported and communicated, and what guidance will be provided for interpretation of results?
- What, if any, action will be taken based on test results?
Burnham suggested that some laboratorians have concerns about testing that takes place in pharmacies or in the home. However, testing in these locations is already taking place; from a diagnostic stewardship standpoint, the best approach may be to provide guidance across settings to support accurate test results.
While there are several rapid diagnostics available and coming on the horizon, said Burnham, there are challenges in getting these tests into clinical practice. Clinical guidelines are important to encourage uptake and should include guidance on when and how to use a given test and information that is specific to users in working in different settings (e.g., critical care, surgery). Rapid diagnostics can be expensive but can also have high clinical value, so uptake may also require a shift in reimbursement policy. There is a need for more research on the impact of rapid diagnostics on multiple outcomes including clinical outcomes, workflow, and decision-making. In addition, said Burnham, there is a need for formal mechanisms for ongoing evaluation of the performance of rapid diagnostics; this will require strong partnerships between microbiology and antimicrobial stewardship programs.
ANTIBIOTIC STEWARDSHIP PROGRAM PERSPECTIVE
When a clinician is faced with a patient who has possible sepsis, the clinician will obtain cultures and then prescribe empiric antibiotics, said Ritu Banerjee (Medical Director, Pediatric Antimicrobial Stewardship Program, Vanderbilt University). Banerjee identified a couple of ways in which empiric antibiotic selection can go wrong. First, a clinician may prescribe an antibiotic that is ineffective against the infecting organism, resulting in poor outcomes including death. Second, a clinician may prescribe antibiotics that are unnecessarily broad-spectrum; the treatment may be effective but unnecessarily contribute to the emergence of resistance. The challenge, said Banerjee, is that conventional bacterial blood cultures and antibiotic susceptibility testing methods typically take days to yield actionable information. Rapid diagnostics can shorten this time, but the clinical impact must be demonstrated.
Banerjee shared evidence from observational studies (Table 6-1) and randomized controlled trials (RCTs) (Table 6-2) that looked at the outcomes associated with the use of rapid diagnostics. Banerjee observed that a variety of tests and methods are in use, but not all tests are implemented with antibiotic stewardship program oversight. Most observational studies demonstrated a decrease in time to optimal therapy for patients but results (e.g, mortality, length of stay, and cost savings) were mixed. The RCT studies also demonstrated a decrease in time to optimal therapy but only one demonstrated improvement in mortality.
Banerjee and colleagues conducted the Blood Culture Identification (BCID) trial, which was a single center, prospective RCT (Banerjee et al., 2015). All patients with positive blood culture were randomized in the laboratory to one of three arms: control (conventional culture and susceptibility testing), BCID (conventional culture and susceptibility testing plus BCID rapid test), or BCID plus stewardship (conventional culture and susceptibility testing, BCID rapid test, and treatment recommendations). In the two arms that included rapid testing, the organism was identified earlier and antibiotic escalation for patients on ineffective therapy happened more quickly (Figure 6-2). Importantly, said Banerjee, antibiotic de-escalation—narrowing a broad-spectrum therapy to target the cultured organism—was only faster in the arm that included the oversight of an antibiotic stewardship team. Banerjee said that this demonstrates the value of integrating a rapid test with stewardship. Clinical outcomes—including mortality, length of stay, adverse events, and cost—were not significantly different between the different arms (Table 6-3).
The Randomized Trial Evaluating Clinical Impact of Rapid Identification and Susceptibility Testing for Gram-negative Bacteremia (RAPIDS GN) was another prospective RCT carried out by Banerjee and colleagues. For
TABLE 6-1 Observational Studies: Rapid Blood Culture Diagnostic and Outcomes
| Test | ASP | Decrease TOT | Mortality benefit | ΔLOS (days) | Cost Savings | Study |
|---|---|---|---|---|---|---|
| PNA FISH | Y | Y | Y | N | - | Forrest, 2008 |
| Xpert MRSA | Y | Y | Y | -6.2 | 21K | Bauer, 2010 |
| MALDI-TOF | Y | Y | Y | -1.8 | 19K | Perez, 2013 |
| MALDI-TOF | Y | Y | Y | -2.8 | - | Huang, 2013 |
| Verigene GP | Y | Y | N | -21.7 | 60K | Sango, 2013 |
| mecA PCR | Y | Y | - | -3 | - | Nguyen, 2010 |
| PNA FISH | N | N | - | N | - | Holtzman, 2011 |
| Verigene | N | Y | Y | - | Y | Suzuki, 2015 |
| BCID | Y | Y | - | - | - | Messacar, 2016 |
| MALDI-TOF | Y | Y | N | N | - | Malcolmson, 2017 |
| BCID | Y | N | N | N | - | Tseng, 2018 |
| Accelerate Pheno | Y | Y | - | - | - | Robinson, 2021 |
| Metaanalysis (31 studies) | Y/N | Y | Y (ASP only) | Y | - | Timbrook, 2017 |
SOURCE: Presented by Ritu Banerjee, October 14, 2022.
TABLE 6-2 Randomized Controlled Trials: Rapid Blood Culture Diagnostic and Outcomes
| Test | ASP | Decrease TOT | Mortality benefit | ΔLOS (days) | Cost Savings | Study |
|---|---|---|---|---|---|---|
| PNA FISH | Y | Y | Y | N | - | Forrest, 2008 |
| Xpert MRSA | Y | Y | Y | -6.2 | 21K | Bauer, 2010 |
| MALDI-TOF | Y | Y | Y | -1.8 | 19K | Perez, 2013 |
| MALDI-TOF | Y | Y | Y | -2.8 | - | Huang, 2013 |
| Verigene GP | Y | Y | N | -21.7 | 60K | Sango, 2013 |
| mecA PCR | Y | Y | - | -3 | - | Nguyen, 2010 |
| PNA FISH | N | N | - | N | - | Holtzman, 2011 |
| Verigene | N | Y | Y | - | Y | Suzuki, 2015 |
| BCID | Y | Y | - | - | - | Messacar, 2016 |
| MALDI-TOF | Y | Y | N | N | - | Malcolmson, 2017 |
| BCID | Y | N | N | N | - | Tseng, 2018 |
| Accelerate Pheno | Y | Y | - | - | - | Robinson, 2021 |
| Metaanalysis (31 studies) | Y/N | Y | Y (ASP only) | Y | - | Timbrook, 2017 |
NOTE: ASP = antibiotic stewardship program oversight; GN = gram-negative; LOS = length of stay; Org = organism; SS = subjects selected; TOT = time to optimal therapy
SOURCE: Presented by Ritu Banerjee, October 14, 2022.
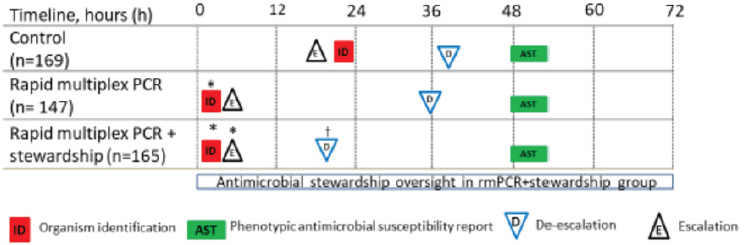
NOTE: PCR = polymerase chain reaction; * = significant vs. control; † = significant vs. control and Blood Culture Identification
SOURCE: Presented by Ritu Banerjee, October 14, 2022; Banerjee et al., 2015.
TABLE 6-3 Clinical Outcomes in BCID trial
| Outcome | Control (n=207) | BCID (n=198) | BCID + Stewardship (n=212) | P-value |
|---|---|---|---|---|
| Length of stay (days) | 8 (5,15) | 8 (5,15) | 8 (5,16) | 0.60 |
| ICU within 14 days | 16 (7.7%) | 5 (2.5%) | 10 (4.7%) | 0.06 |
| 30-day mortality | 22 (10.6%) | 20 (10.1%) | 18 (8.5%) | 0.74 |
| 30-day readmissiona | 6 (2.9%) | 6 (3%) | 8 (3.8%) | 0.88 |
| Toxicity/adverse drug rxn | 3 (1.4%) | 3 (1.5%) | 2 (0.9%) | 0.82 |
| Blood cx clearance in 3d | 147 (71%) | 131 (66%) | 146 (69%) | 0.79 |
| C. difficile/MDRO in 30db | 15 (7.2%) | 16 (8.1%) | 21 (9.9%) | 0.62 |
| Overall costsc | $65,450 ($27,192) |
$66,887 ($23,935) |
$68,729 ($29,064) |
0.78 |
NOTE: BCID = Blood Culture Identification; C. difficile = Clostridioides difficile; cx = culture; ICU = intensive care unit; MDRO = multidrug resistant organisms; rxn = reaction
a with same organism
b VRE, MRSA, ESBLs, Gram-negatives resistant to > 3 drug classes
c Mean (median) among inpatients with available data (n = 544)
SOURCE: Presented by Ritu Banerjee, October 14, 2022; adapted from Banerjee et al., 2015.
this study, patients were randomized to either a control arm (conventional culture and susceptibility testing) or a rapid testing arm (conventional culture and susceptibility testing plus rapid test); both arms included oversight by an antibiotic stewardship team. The results of the trial showed that patients who had resistant organisms detected in their blood and were on ineffective therapy initially were switched to effective therapy almost two days faster than patients in the control arm. Unfortunately, said Banerjee, the trial did not find significant differences in mortality, length of stay, readmissions, or other clinical outcomes (Banerjee et al., 2021).
While the BCID and RAPID GN trials did not find significant differences in clinical outcomes between traditional culture testing and rapid testing, Banerjee said that there were important lessons learned from both trials. First, rapid blood culture diagnostics, when implemented in collaboration with antibiotic stewardship teams, can improve treatment optimization for patients with bloodstream infections. Optimized treatment means faster antibiotic modifications, more judicious antibiotic use, and more timely antibiotic administration. Second, Banerjee said that understanding the local context is important for study design and interpretation. The impact of a rapid diagnostic will vary depending on local resistance rates, antibiotic prescribing practices, patient populations, the availability of stewardship programs, and baseline laboratory practices. Another lesson learned from these trials is that clinical outcomes (e.g., mortality or length of stay) may not be appropriate endpoints. These outcomes are dependent on many factors, Banerjee said, and de-escalation or targeted antibiotic treatment “should be sufficient justification for implementing a novel diagnostic.” Other downstream impacts of improved medical decision-making should also be selected as endpoints for clinical utility studies, including laboratory workflow efficiencies and infection control activities such as time to isolation.
Banerjee closed by saying that she has “a lot of hope” that there are novel and innovative diagnostics on the horizon. Her “wish list” for diagnostics would include direct-from-specimen testing, point-of-care testing, tests that can rule bacterial infection in or out, and tests that could distinguish bacterial colonization from bacterial infection.
INDUSTRY/DEVELOPER PERSPECTIVE
Diane Flayhart (Global Program Leader, Becton, Dickinson and Company) focused her presentation on four main questions:
- How effectively do current diagnostics inform antibiotic stewardship?
- Is there enough data to understand how drug-resistant infections affect specific patient populations?
- Are new metrics needed to drive diagnostic use and antibiotic stewardship efforts?
- Are more advocates needed to drive awareness for Diagnostic and Antibiotic Stewardship?
To address the first question about how effectively current diagnostics inform antibiotic stewardship, Flayhart’s company and its partners conducted a study which included a literature review followed by interviews with frontline clinicians and a survey of an additional 50 clinicians (BD, 2022). The study was designed to examine how diagnostics are currently used to combat AMR, capture lessons learned on better use of current diagnostics, estimate the value of diagnostics, describe how the use of diagnostics can be improved, highlight priority unmet needs, and inform guidelines (Figure 6-3).
Flayhart shared a selection of preliminary results from the first two phases of the study. They found that the guidelines on antimicrobial stewardship programs recommend the use of rapid diagnostics in combination with traditional methods and that most guidelines for individual practice areas recommend traditional methods. Sporadic recommendations for point-of-care or rapid testing were not specific about how and when to use these diagnostics. Another key finding, said Flayhart, was that minimal data were being gathered about how surveillance and diagnostics have impacted hospital formulary access policy. Flayhart explained that to tie the impact of a diagnostic to antibiotic stewardship, it is necessary to have these data. Interviews with clinicians revealed several issues, including heterogeneity of diagnostic availability, the cost of diagnostics, and a lack of awareness of AMR and the utility of rapid tests. Flayhart said that this study was motivated in part because her company spends a lot of time and money developing diagnostic tests, so it is critical to understand how and why they are or are not being used by frontline clinicians.
Next, Flayhart addressed the issue of whether there are enough to data to understand how drug resistant infections affect specific patient populations. She shared data about age and geographic distribution of antibiotic-resistant bacterial infections. Further, she noted that the CDC is undertaking efforts to understand the links between health inequities and antibiotic resistance. A 2022 CDC presentation described a few associations:1
- Community-associated ESBL-Enterobacterales: Higher incidence rates in areas with lower median incomes, lower high school
___________________
1 Information from Dawn Siewert, Antimicrobial Resistance Coordination and Strategy Unit, National Center for Emerging and Zoonotic Infectious Diseases, U.S. Centers for Disease Control and Prevention; presented at World AMR Congress in September 2022.
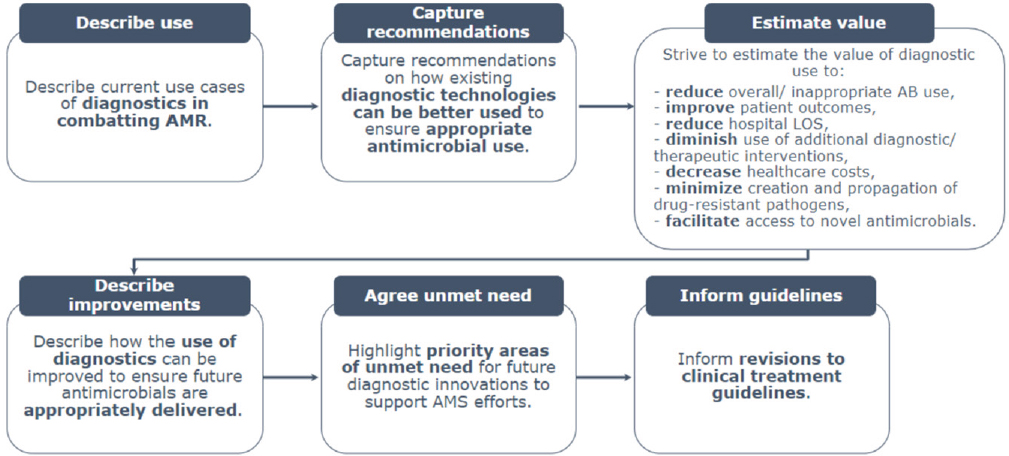
NOTE: AB = antibiotic; AMS = antimicrobial stewardship; AMR = antimicrobial resistance; LOS = length of stay
SOURCE: Presented by Diane Flayhart, October 14, 2022; Survey Study Design, BD, Wellcome, Pfizer.
- Community-associated C. difficile infections: Higher incidence rates in communities with low-incomes, foreign-born populations, non-English speaking households, and crowding in the home.
- Candidemia and MRSA: Higher rates in Black individuals, possibly reflecting differences in medical conditions, healthcare access, socioeconomic status, and access to housing.
- Antibiotic prescribing and/or inappropriate prescriptions: Higher volumes/rates in rural areas, non-Hispanic White individuals, pediatric populations, and older persons with viral respiratory diseases.
- Antibiotic prescribing or diagnosis with bacterial pathogen: Lower volumes in Black individuals.
education rates, higher percentages of persons without health insurance, and those with limited English proficiency.
Flayhart considered whether new metrics are needed to drive diagnostic use in antibiotic stewardship or to understand how antibiotics are being used within healthcare systems. Flayhart suggested there is a need for national metrics for antibiotic use at the hospital level. There are complexities and challenges with accomplishing this, but it would allow antibiotic stewardship teams to measure how diagnostics are impacting antibiotic use. Antibiotic use increases during cold and flu season, which could offer an opportunity for stewardship programs to track and evaluate appropriate antibiotic use and the role of diagnostics (Yu et al., 2022).
Finally, Flayhart turned to the role of advocacy in improving diagnostic and antibiotic stewardship. She emphasized the importance of engaging outside of the “AMR bubble.” For example, when new rapid diagnostics for sepsis are brought to market, there is a need to engage and involve organizations such as the Sepsis Alliance,2 ENDSEPSIS,3 and the Global Sepsis Alliance.4 In addition, there may be creative ways, such as the use of film or other media, to better engage the lay public.
DISCUSSION
Role of policy
Banerjee suggested that policies in the form of clinical guidelines “would go a long way” towards integrating diagnostics into care. When clinicians, administrators, and payers can refer to one set of guidelines that recommend when and how to use diagnostics, those guidelines determine the new standard of care. Another effective approach would be a policy that
___________________
2 See https://www.sepsis.org/ (accessed January 23, 2023).
3 See https://www.endsepsis.org/ (accessed January 23, 2023).
4 See https://www.global-sepsis-alliance.org/ (accessed January 23, 2023).
mandates diagnostic stewardship programs that are conducted in partnership with microbiology laboratories. These programs would help support uptake of diagnostics and appropriate use of diagnostics.
Role of guidelines
Burnham pointed out that guidelines are typically developed by volunteers who have many other duties, so it would be useful to have financial resources for professional help with tasks such as literature reviews to help speed up the process, particularly given that this field evolves quickly over time. Additionally, guideline development should be useful for the end users, so it is important to incorporate the input of people who might rely on diagnostic tests, including laboratory directors, medical specialists and infectious disease experts. When considering policy levers for supporting the development guidelines (e.g., CDC or specialty societies), Flayhart and R. Patel agreed that more work is needed.
Role of reimbursement and incentives
Flayhart stated that it can be challenging to assess and demonstrate the value of diagnostics because patient health and outcomes are complex and multi-factorial. She emphasized the importance of collaborating with laboratories and stewardship teams to design clinical trials in a way that captures meaningful measurements. If a trial shows how a diagnostic can plug into the clinical pathway for patient care and improves outcomes, it is more likely to be deployed and reimbursed.
Opportunities for public awareness
Flayhart said that AMR should be viewed as a global phenomenon that impacts humans, animals, and the environment. There are public conversations about issues, such as antibiotic use in animal food products and environmental contamination of public waterways, she said, but what “we haven’t cracked yet” is getting people to ask questions about their own use of antibiotics. According to Flayhart, people should be having conversations with their doctors about why an antibiotic is being prescribed and if there is a diagnostic available to help make more informed treatment decisions. Bausch said that stakeholders should be more forthcoming about the potential harms of antibiotics so patients can weigh the pros and cons of taking a drug. The message for patients, said Flayhart, is that unlike other medications, taking antibiotics does not just impact the patient but everyone in their ecosystem.
Buy-in from hospital leadership
Diagnostic stewardship programs seem to be beneficial for many reasons, said Van Meter. However, how can hospital leadership be convinced
to invest in them? Burnham said that there is a delicate balance between a desire for physician autonomy and a desire to deploy resources appropriately. Like many things, she said, it starts with a “gentle approach.” For example, an alert could be built into the electronic health record that would simply ask the physician to confirm a prescribing choice. Eventually this could progress to a hard stop where the physician cannot take a certain step (e.g., prescribing or ordering a test) without following a certain pathway. Along the way, said Burnham, it is critical to include stakeholders in the process and focus on adding value whenever possible. Often, the lab might have to take on the initial burden of choosing a use case and a clinical partner to deploy the tool; once value is demonstrated, they can “take that on the roadshow” and champion it.
Following up on this, a virtual participant asked Banerjee to discuss how hospital leadership can be convinced of the value of diagnostics if they do not improve the metrics that are most important to hospitals (e.g., length of stay, mortality). Banerjee said that the value can be demonstrated through other metrics, for example, savings on the cost for antibiotics, or long-term savings due to faster infection control. There is value in reducing societal levels of AMR, and this should be reflected in accreditation or joint commission surveys. For example, if a survey asks hospitals if they monitor certain metrics related to AMR, hospital leadership is likely to adopt new practices to maintain accreditation and ratings.
Mechanism to measure performance
As new diagnostics are developed and deployed, said a participant, it will be critical to have an ongoing mechanism for ensuring performance. She asked panelists to comment on how this could be accomplished. Burnham said that there will need to be different approaches for ongoing evaluation of diagnostics. Putting this burden on device manufacturers is not realistic, said Burnham; it will need to be a coordinated effort among hospitals, laboratories, device manufacturers and government. If a shift in performance occurs or a new mechanism of resistance is detected, these results will need to be reported so that appropriate steps can be taken.
Clinical outcome studies
A participant commented that conducting clinical outcome studies poses a considerable challenge for test developers; studies may need to be conducted in multiple centers and in areas with high resistance. Banerjee responded that it can be difficult for companies, particularly smaller companies, to conduct studies that can adequately show the value of a new diagnostic. Partnering with larger organizations can be one approach to support these types of studies. However, at the end of the day, clinical outcome studies should be “diagnostic agnostic.” In other words, if it can
be demonstrated that a diagnostic provides actionable results within a certain timeframe and has downstream clinical benefit, then those benefits should be applied to equivalent tests as they move through clinical outcome studies.














