1
Introduction and Background1
Over the past decade, the pharmaceutical industry has decreased investment in neurological and psychiatric disorders despite broad recognition of the vast unmet need for treatment of these disorders, which affect huge numbers of people worldwide (Jarvis, 2012). The explosion of technology development in genetics and single-cell analysis over the past 5 years, however, may reverse this trend, said Steven Hyman, director of the Stanley Center for Psychiatric Research at the Broad Institute of Harvard and MIT. Significant progress has been seen recently in developing therapies for rare monogenetic disorders such as spinal muscular atrophy (FDA, 2020b) and Dravet syndrome (FDA, 2016, 2020a); however, progress continues to lag for more common neurological and psychiatric disorders that are more heterogeneous, such as Parkinson’s disease, Alzheimer’s disease, and schizophrenia. For these disorders, critical challenges include poor mechanistic insight and a lack of biomarkers that can be used to stratify individuals within broad symptom-based diagnostic categories, which hinder progress in developing novel therapeutics based on genetic findings. Recognizing these challenges, Hyman said industry has begun to coalesce around the idea that stratifying patients through the use of biomarkers may improve the likelihood of success in clinical trials.
___________________
1 The planning committee’s role was limited to planning the workshop, and the Proceedings of a Workshop was prepared by the workshop rapporteurs as a factual summary of what occurred at the workshop. Statements, recommendations, and opinions expressed are those of individual presenters and participants; have not been endorsed or verified by the Health and Medicine Division of the National Academies of Sciences, Engineering, and Medicine; and should not be construed as reflecting any group consensus.
Inspired by an imperative to exploit relevant technologies and explore how they can be used to elucidate disease mechanisms and advance the development of biomarkers, the National Academies of Sciences, Engineering, and Medicine’s Forum on Neuroscience and Nervous System Disorders (Neuroscience Forum) convened a workshop in October 2021. This is the latest in a series of workshops aimed at addressing the challenges of moving from unmet medical need to therapeutic development for neurological and psychiatric disorders, said Hyman. He co-chaired this workshop with Dimitri Krainc, the Aaron Montgomery Ward Professor and Chairman of the Department of Neurology at Northwestern University, Feinberg School of Medicine.
Earlier workshops in this series addressed a range of topics related to improving therapeutic development in this field. In 2012, the Neuroscience Forum hosted a workshop titled Improving the Utility and Translation of Animal Models for Nervous System Disorders that explored the limitations in the use of animal models, recognizing that even though they cannot model human brain disorders, they are critical as models of disease mechanisms (IOM, 2013). This was followed by a workshop in 2013 titled Improving and Accelerating Therapeutic Development for Nervous System Disorders, which explored the possibility of advancing human experimental biology, human observation, and phenotyping along with then-rapidly advancing genetic technologies (IOM, 2014). In 2016, a third workshop titled Therapeutic Development in the Absence of Predictive Animal Models of Nervous System Disorders (NASEM, 2017) considered innovative approaches to disease modeling, including human induced pluripotent stem cells (iPSCs) and three-dimensional organoids and spheroids derived from iPSCs; novel gene editing techniques; and computational modeling.
WORKSHOP OBJECTIVES AND APPROACH
The goal of this fourth workshop in the series was to bring together a diverse group of stakeholders to explore how new genetic and neuroscience technologies can be used to identify targets and biomarkers that enable patient stratification2 in clinical trials and advance therapeutic development (see Box 1-1). The workshop was predicated on the idea that a deep mechanistic understanding of disease pathogenesis for neurological and psychiatric disorders, applied to both basic and translational studies, can fuel the development of appropriate treatments for diverse populations,
___________________
2 Patient stratification is the “grouping [of] patients into disease sub-groups, where the specific pathological processes involved are better defined (e.g., clinical/molecular phenotypes).” Source: https://cordis.europa.eu/programme/id/H2020_SC1-PM-02-2017 (accessed February 2, 2022).
said Krainc. He noted that increasing ancestral diversity in both basic and translational studies is a critical first step in the process.
Krainc described a stepwise strategy for developing biomarkers, and then using them to design adaptive clinical trials capable of demonstrating target engagement in patients (see Figure 1-1). The first step, he said, is to create biobanks from diverse populations of patients with neurological and psychiatric disease. In step 2, these samples will be analyzed using new technologies that facilitate identification of causal and risk variants, and genes linked to these variants. Next, it will be important to understand the functional effects of these variants in relevant cell types and tissues (step 3), followed by functional studies in human models of disease, such as pluripotent stem cell-derived neurons and glia (step 4), said Krainc. He noted that these studies will generate huge amounts of data, the integration of which will require sophisticated analytical approaches, including artificial intelligence and machine learning (step 5). These analyses will be designed to identify validated targets and biomarkers that can improve stratification of patients (step 6), which can then be used in adaptive clinical trials (step 7) to demonstrate target engagement. Krainc noted that while target engagement has been somewhat easier to demonstrate in single gene disorders, it will be more challenging in complex disorders. The workshop, he said, was designed around these steps to stimulate discussions about how to achieve these goals.
ORGANIZATION OF THE PROCEEDINGS
The workshop covered disorders considered both neurological and psychiatric. Chapter 2 gives an overview of the challenges that will be important to address in order to advance therapeutic development with new innovations in genetic and neuroscience technologies. The problem of insufficient genetic diversity in genetic research and efforts underway to build more ancestrally diverse genomic datasets are discussed in Chapter 3. Novel approaches aimed at linking genes to causality in complex diseases are discussed in Chapter 4. Chapter 5 describes several new technologies that enable identification and validation of molecular pathways relevant to neurological and psychiatric disorders. Biomarkers are discussed in Chapter 6: their importance in drug development, novel approaches being pursued, and a regulatory perspective on their use. Chapter 7 summarizes strategies and next steps proposed by individual workshop participants to advance development of novel treatments for neurological and psychiatric disorders. References cited throughout these proceedings are listed in Appendix A, and the workshop agenda is in Appendix B.
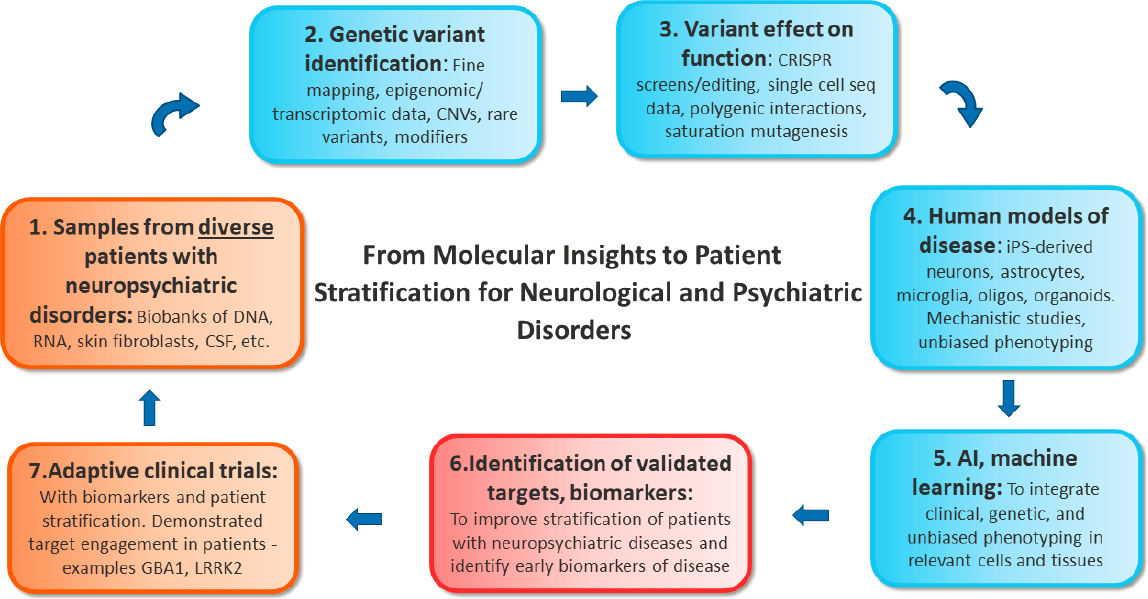
NOTE: CNV = copy number variation; CSF = cerebral spinal fluid; DNA = deoxyribonucleic acid; iPS = induced pluripotent stem cells; RNA = ribonucleic acid
SOURCE: Presented by Dimitri Krainc, October 5, 2021.
This page intentionally left blank.






