The opioid overdose crisis worsened during the COVID-19 pandemic, especially among racially minoritized population groups, as noted by Magdalena Cerdá in Chapter 3. The presence in many communities of the powerful synthetic opioid fentanyl has driven much of this increase, said Rahul Gupta. Compounding this is the fact that people are not able to access the addiction treatment they need, he added. “In 2020, only 1 out of 10 people who needed treatment for illicit drug use received it at a specialized facility,” said Gupta (SAMHSA, 2021). There simply are not enough opioid treat-
ment programs (OPTs) available, he said, and many of the OTPs that exist are facing challenges in providing care. These challenges arise first from the fact that OTPs historically have been siloed from health care delivery systems. In addition, some do not offer all three forms of medication for opioid use disorder (OUD), not all of them accept Medicaid, and some take a punitive approach to treatment, said Gupta.
GOALS AND PRIORITIES OF THE CURRENT ADMINISTRATION
In March 2021, President Biden signed into law the American Rescue Plan Act,1 which invested nearly $4 billion in expanding access to vital mental health and substance use disorder services, said Gupta. First-year policy priorities included reviewing methadone treatment policies and modernizing the way methadone is used.
The President’s inaugural national drug strategy2 also prioritizes expanding access to quality treatment and reducing racial inequities related to addiction, especially in health care and the criminal justice system. “We need to build a system of care that proactively seeks to diagnose and treat individuals, rather than waiting until they come into the criminal justice system or present with an overdose,” said Gupta. He also advocated exploring what the federal government can do to help OTPs become more culturally and linguistically competent and more accessible to all, including by accepting insurance.
Gupta added that while evidence-based treatment can save lives, stigma often prevents people from getting the help they need. He compared attitudes about addiction today to attitudes about cancer in the past. With the formation of the American Cancer Society in 1913, he said, “Our country leaned into this. We made sure that we advocated for change, we improved treatments, and today cancer is a disease that receives more attention and funding than just about anything else.” Not only are more treatments now available, but a diagnosis of cancer engenders compassion rather than blame. “Yet, that is not the case for the disease of addiction,” he said.
___________________
1 For more information about the American Rescue Plan Act of 2021, go to https://www.congress.gov/bill/117th-congress/house-bill/1319 (accessed May 6, 2022).
2 The Biden Administration’s National Drug Control Strategy was released soon after this workshop was held and is available at https://www.whitehouse.gov/wp-content/uploads/2022/04/National-Drug-Control-2022Strategy.pdf (accessed May 2, 2022).
The U.S. Department of Health and Human Services Response to the Opioid Crisis
Of the three approved medications to treat OUD, methadone is the least easily prescribed, despite the fact that decades of research show that it effectively reduces opioid cravings, illicit opioid use, and risk of opioid overdose, and increases rates of treatment retention, said Rebecca Haffajee, Acting Assistant Secretary for Planning and Evaluation and Principal Deputy Assistant Secretary for Planning and Evaluation at the U.S. Department of Health and Human Services (HHS) (see Figure 4-1). Given the escalating opioid crisis, experts across HHS came together in 2021 to leverage the best evidence available and devise a new overdose prevention strategy,3 said Haffajee. This strategy identified four key target areas: (1) primary prevention, harm reduction, evidence-based treatment, and recovery support; (2) four crosscutting principles: equity, coordination, collaboration, and integration; (3) data and evidence; and (4) reducing stigma. Haffajee
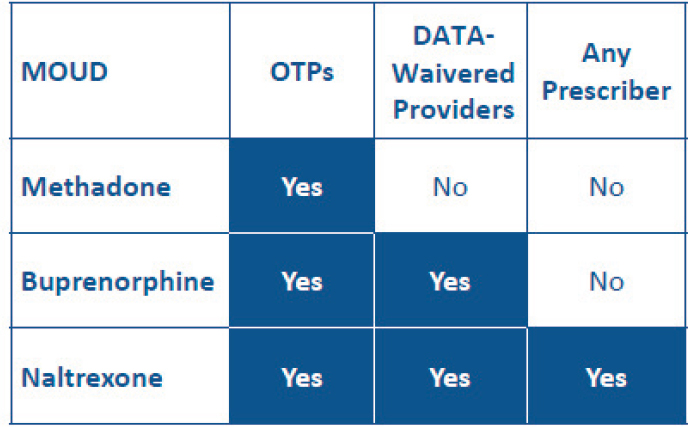
NOTE: DATA = Drug Addiction Treatment Act; MOUD = medications for opioid use disorder; OTPs = opioid treatment programs.
SOURCE: Presented by Rebecca Haffajee, March 3, 2022.
___________________
3 To learn more about the HHS Overdose Prevention Strategy, go to https://www.hhs.gov/overdose-prevention (accessed April 5, 2022).
noted that this strategy differs from the approach taken by the previous administration by shifting the focus to overdose rather than opioids alone and by taking a life span and continuum-of-care approach, with integration across sectors and types of care.
To close the gap between research and care, HHS is also substantially increasing investments to address overdose above and beyond the increases in funding seen in response to the COVID-19 pandemic, said Haffajee. For example, she cited the National Institute on Drug Abuse’s (NIDA’s) medication development program, which has funded research on the long-acting formulation of methadone that has received fast-track Food and Drug Administration (FDA) designation. “This formulation has the potential to increase treatment, uptake, adherence, and retention,” said Haffajee. She added that NIDA’s Clinical Trials Network4 is also supporting development of innovations such as remote monitoring of methadone ingestion and incorporating pharmacists into collaborative care models.
As part of the National Institutes of Health (NIH) Helping to End Addiction Long-Term (HEAL) Initiative, NIH has also launched the HEALing Communities Study,5 which is testing the integration of prevention, overdose treatment, and medication-based treatment at an array of settings (e.g., primary care, emergency department, community settings) in 67 urban and rural communities across 4 states, said Haffajee. To address concerns about providing treatment for OUD within the criminal justice system, the NIH HEAL Initiative is also testing different interventions to enhance quality care through the Justice Community Opioid Innovation Network (JCOIN).6 Haffajee noted that as part of that project, a survey conducted in 21 states with the highest opioid overdose rates found that methadone was available in only 9 percent of the 583 prisons managed by prison systems surveyed (Scott et al., 2021). Even when it was available, it was often limited to subsets of patients such as pregnant women, she said. One of the problems they uncovered was that while establishing jail-based OTPs was onerous and costly, transporting patients to local OTPs was equally as onerous and cost prohibitive, said Haffajee.
Also under the umbrella of HHS, the Center for Medicare and Medicaid Innovation (CMMI) administers the Maternal Opioid Misuse (MOM) delivery model,7 a patient-centered service delivery model that is supporting
___________________
4 To learn more about NIDA’s Clinical Trials Network, go to https://nida.nih.gov/about-nida/organization/cctn/clinical-trials-network-ctn (accessed May 6, 2022).
5 To learn more about the HEALing Communities Study, go to https://heal.nih.gov/research/research-to-practice/healing-communities (accessed April 5, 2022).
6 To learn more about the Justice Community Opioid Network (JCOIN), go to https://heal.nih.gov/research/research-to-practice/jcoin (accessed March 5, 2022).
7 To learn more about the Maternal Opioid Misuse (MOM) model, go to https://innovation.cms.gov/innovation-models/maternal-opioid-misuse-model (accessed April 5, 2022).
3,300 to 5,000 pregnant and postpartum Medicaid beneficiaries and their infants in eight states (Esposito et al., 2021). Haffajee said the goal is to improve quality of care and reduce costs, as well as to reduce fragmentation of care and improve care coordination, including with OTPs and medications for opioid use disorder (MOUD) providers. She added that since the beginning of 2020, OTPs can receive bundled payments under Medicare Part B for services that include dispensing of medications, including methadone, for OUD. Haffajee noted that the Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment (SUPPORT) for Patients and Communities Act8 also requires coverage of all three FDA-approved medications for OUD, with implementation of this requirement to be rolled out between 2020 and 2025.
Data from the National Survey of Substance Abuse Treatment Services (N-SSATS) suggests that there have been some modest gains in the number of OTPs and the number of clients receiving methadone in OTPs over the past few years, said Haffajee. However, she said, “There is still a dearth of capacity and there are wait lists associated with many OTPs.” Moreover, there are additional barriers to accessing methadone in certain settings, such as in justice settings and long-term care facilities, she said. Lack of integration with other medical care, such as prenatal care, further hinders the provision of methadone to certain populations, said Haffajee. Helena Hansen added that adolescents are often overlooked for methadone treatment as well.
SAMHSA Efforts to Improve OUD Treatment
Beyond the HHS overdose prevention strategy, the Substance Abuse and Mental Health Services Administration (SAMHSA), an agency of HHS, leads public health efforts to reduce the impact of substance use disorder and mental illness. Within SAMHSA, the Center for Substance Abuse Treatment (CSAT) oversees key federal regulations governing OTPs, said Yngvild Olsen, director for CSAT. Waivers to these regulations, defined in Title 42 of the Code of Federal Regulations (CFR) Part 8,9 have occurred in response to the COVID pandemic, resulting in significant changes in the field over the past 2 years, said Olsen.
“Up until 2020, OTP regulations laid out a strict, and many have said too strict, process for OTP medical directors to approve take-home doses of methadone,” said Olsen. The basis for approval depended not only on the length of time a person had been enrolled in the OTP, but also considered
___________________
8 To learn more about the SUPPORT for Patients and Communities Act, go to https://www.congress.gov/bill/115th-congress/house-bill/6 (accessed May 6, 2022).
9 To learn more about Title 42 of the Code of Federal Regulations Part 8, go to https://www.ecfr.gov/current/title-42/chapter-I/subchapter-A/part-8?toc=1 (accessed May 2, 2022).
whether the therapeutic benefits outweigh the risks of providing the patient doses that could be taken outside the clinic. Many states and OTPs interpreted this risk to include the presence of any non-methadone-controlled substance or medication, based on drug testing. As was described by Walter Ginter and Abby Coulter in Chapter 2, this often resulted in patients either not being given take-home doses or having them discontinued, triggering the requirement that they come into an OTP almost daily to get their dose. Many OTPs also interpreted the regulations related to other services as requiring all patients to participate in counseling or face discharge from the program. Olsen said there have also been concerns about the use of technology in providing methadone, particularly in light of the 42 CFR Part 8 requirement for a comprehensive in-person physical examination prior to starting treatment with methadone for an OUD.
Following the World Health Organization’s declaration of a global COVID-19 pandemic on March 11, 2020, SAMHSA, the Drug Enforcement Administration (DEA), and other federal agencies issued a flurry of exceptions to regulations impacting OTPs, said Olsen. Most significant, she said, was the blanket exception10 that allows OTP medical directors to provide up to 28 days of methadone take-homes to stable patients, and up to 14 days for unstable patients, if the OTP believes they can safely manage these doses. Olsen said that over the past 2 years, 43 states and the District of Columbia have taken advantage of the blanket methadone take-home exception, although some OTPs and states reportedly have backed away from the exception. Olsen noted that SAMHSA also provides block grants and state opioid response program grants to ensure coverage for individuals who are uninsured or whose health insurance does not include the coverage benefit of methadone-related treatment services. Meanwhile, SAMHSA recently released guidance to extend the take-home medication exception for 1 year past the COVID-19 public health emergency to allow time for the agency to pursue more permanent rulemaking.
Another significant change brought by the COVID pandemic was the authorization made permissible by the Centers for Medicare & Medicaid Services (CMS) flexibilities on telehealth, and SAMHSA regulatory exceptions to allow OTPs to provide patients with counseling, peer support, and check-ins via telehealth. “Emerging data and my own experience demonstrate that the ability to connect with patients through different telehealth platforms have generally been positive, from OTP, clinician, and patient perspectives, although not a complete replacement for all services,” said Olsen.
Notably, she said, SAMHSA elected to exempt the requirement for an in-person physical exam only for patients getting buprenorphine, but not methadone, at an OTP.
___________________
10 For more information about this exemption, go to https://www.samhsa.gov/medicationassisted-treatment/statutes-regulations-guidelines/methadone-guidance (accessed June 7, 2022).
Since the early days of the pandemic, SAMHSA has made other changes as well, said Olsen. After DEA issued guidance on establishing mobile medication units, SAMHSA issued its own guidance on funding for such units and defining which services can be provided. With the new mobile medication allowance and federal funding to support them, some states have begun to work with accreditation bodies to establish these units, said Olsen. The goal of mobile medication units, she said, is to try to get methadone to people wherever they are, whether that is in jail or prison, rural areas, or residential settings where pregnant people might be receiving services, or through other substance use disorder services.
Even with these regulatory changes, other challenges and opportunities remain, said Olsen. The first of these is the stigma around methadone. “Despite 50 years of clinical and research experience with this medication, I can’t tell you how many times I’ve heard patients say they need to get off that stuff, as Walter [Ginter] mentioned, because they or their mother or their sister or significant other has told them it’s just replacing one drug for another,” said Olsen. She said she’s also had physician colleagues tell her they will not consider patients for a liver transplant or replace someone’s arthritic hip, or treat their severe bipolar disorder, unless methadone is permanently stopped. Patients have told her how badly they have been treated by primary care providers and asked to have the OTP be their primary care home. Behavioral health specialists working in addiction treatment have told her they do not believe in methadone, although they view buprenorphine as an opportunity to help people with recovery. She said she has also heard of OTP staff behaving as if they do not believe in the benefits of methadone treatment in helping people with OUD stay safe and reach their goals.
“So not only do we need to address stigma broadly across the board, but the health care profession at large needs significant training around methadone,” she said.
Another challenge that needs to be addressed, said Olsen, is how to optimize quality of care for people with moderate to severe OUD, who often present with “a complex mix of multiple substance use disorders, physical and mental health conditions, the effects of trauma and discrimination, and other factors related to social determinants of health.” In 2015, SAMHSA issued guidelines that describe a framework for what a high-quality specialty treatment system that includes methadone within the context of 42 CFR section 8 might look like (SAMHSA, 2015). She said the agency welcomes feedback on these guidelines as well as on how to promote the adoption of these strategies at federal, state, and clinic levels.
Drug Enforcement Administration’s Efforts to Expand Access to OUD Treatment
DEA is committed to expanding access to medications for OUD, according to Kristi O’Malley, senior advisor of the Diversion Control Division within DEA at the time of the workshop and now assistant administrator of DEA. As mentioned earlier, they have temporarily lifted the requirement for in-person evaluation so that patients can receive buprenorphine prescriptions “issued for legitimate medical purposes” through telemedicine, she said. In coordination with SAMHSA, they have also waived certain parts of regulations for take-home doses of methadone to ensure that stable patients “can receive medication even if they are unable to leave their homes,” said O’Malley, and they have promulgated regulations to implement a provision of the SUPPORT Act, which authorizes a pharmacy to deliver a controlled substance to an administering practitioner for the purposes of providing MOUD. She said “this step forward in patient care streamlines the process, because previously a patient needed to get the medication dispensed at the pharmacy, take it to the treatment provider, and then get it administered, and now that step has been removed.” In July 2021, DEA also passed a regulation that allows the expansion of mobile narcotic treatment programs by “allowing DEA registrants who are authorized to dispense methadone for OUD to implement a mobile component to their registration, eliminating the separate registration requirement.” She added that they have been working with SAMHSA and the Bureau of Prisons (BOP) to help provide people who are incarcerated with OUD with more access to methadone through a potential hub and spoke model making the narcotic treatment programs the hub and medication units with the prison the spoke.
O’Malley said that when DEA takes action against physicians illegally prescribing narcotics, patients can be left with nowhere to go for treatment. Thus, she said, DEA has partnered with HHS in the Opioid Rapid Response Program11 to make sure patients are able to get the services they need. She added, “We’ve done the same with pharmacies, targeting them in a positive way to make sure they get information and know DEA supports them supplying [MOUD] to those who need it.”
___________________
11 To learn more about the Opioid Rapid Response Program, go to https://www.cdc.gov/opioids/opioid-rapid-response-program.html (accessed May 6, 2022).
REGULATORY CHANGE IN THE WAKE OF NECESSITY: LESSONS LEARNED FROM COVID-19
Decades of research, as well as the testimony of Walter Ginter, Brenda Davis, and Abby Coulter in Chapter 2, confirm that strict methadone regulations have caused tremendous burden, stigma, and burnout among people with OUD, said Noa Krawczyk, assistant professor at the Center for Opioid Epidemiology and Policy at the New York University Grossman School of Medicine. “It’s like liquid handcuffs,” according to a qualitative study by Frank and colleagues (Frank et al., 2021). Or as one patient in Frank’s study said, “When I was on methadone, I was like a double slave. Like you’re a slave to the heroin already, and you’re on methadone, you’re a slave to the methadone and to the clinic.”12
The COVID-19 pandemic brought about not only revised regulations around take-homes and telemedicine, said Krawczyk, but also an opportunity for her and other researchers to examine how these regulations affected outcomes among people with OUD. Among some 30 studies conducted across the United States, researchers have collected data from OTPs across more than 25 states, she said.
Implementation of Revised Take-Home Regulations
The first question Krawczyk and her colleagues asked was how new take-home methadone regulations have been implemented in practice. Although a survey of OTPs across the country, conducted by the Office of the Inspector General of HHS, found that nearly 90 percent of OTPs endorsed increased take-homes to some extent with patients, at least two studies indicate that most patients were not affected by the regulatory change, said Krawczyk. For example, Krawczyk and colleagues showed that 70 percent of the OTPs surveyed in Pennsylvania indicated that none of their patients were eligible for 28-day take-homes (Krawczyk et al., 2022). A study in Connecticut showed that while there was an increase in the number of people eligible for 14- and 28-day take-homes, still only a minority of patients were impacted (Brothers et al., 2021).
Studies also showed that OTP providers had mixed opinions and perceptions about methadone take-homes, said Krawczyk. These varied from
___________________
12 During the open discussion period, workshop attendees were provided an opportunity to provide comments via an online chat platform. In this regard, Susan Staats Combs, co-owner of the Shelby County Treatment Center and Chilton County Treatment Center in Alabama, cautioned against the use of terms like “liquid handcuffs,” which are hurtful and can increase stigma. “Patients need it to survive, and it does not need to be paired with handcuffs. Methadone treatment, as do other medications, helps keep people alive! ‘Liquid life’ is what it should be,” she said.
one provider who said that trusting patients to manage their methadone was beneficial to the patient–provider relationship to another who believed strict policies were warranted because of the risk of abuse and the “huge liability” for OTPs (Hunter et al., 2021; Suen et al., 2021).
Several challenges and facilitators related to implementing longer take-homes have also been identified, said Krawczyk. Challenges included a lack of consensus or guidance around defining stability for take-homes, concerns about exacerbating already existing disparities (e.g., some people not having access to safe storage due to lack of stable housing), and implicit biases and previous experiences with patients that could influence provider discretion around take-homes (Hatch-Maillette et al., 2021; Madden et al., 2021; Peavy et al., 2020). Telehealth-related challenges included questionable sustainability of reimbursement and how this might affect continuation of remote monitoring that began during the pandemic (Hunter et al., 2021; Joseph et al., 2021). Facilitators cited by OTPs included leveraging multidisciplinary treatment teams to make take-home decisions and using pillboxes and other strategies to reduce concerns around safety and diversion, said Krawczyk (Dunn et al., 2021; Kidorf et al., 2021; Krawczyk et al., 2022; Levander et al., 2021a).
Impact of Revised Take-Home Regulations on Overdose and Diversion
Researchers also explored how changes in take-home regulations affected the incidence of overdose and diversion, said Krawczyk. Opioid-related fatalities have increased dramatically over the past year, most driven by the synthetic opioid, fentanyl (CDC, 2022). However, methadone has historically and continues to represent a very small proportion of opioid-related fatalities (approximately 5 percent), said Krawczyk (see Figure 4-2). Moreover, she said, methadone-related poisonings, overdose deaths, and diversion also changed very little following COVID-19 regulatory changes.
Impact of Revised Take-Home Regulations on Patient Experience and Quality of Life
In several qualitative studies, patients were interviewed about the impact of revised regulations on their lives, said Krawczyk (Harris et al., 2021; Levander et al., 2021b; Suen et al., 2021). She noted several themes identified in these studies: Longer take-home doses provided patients with greater autonomy and normalcy; however, individualized care was key because some patients believed more frequent contact was beneficial in certain circumstances. Some patients also reported that take-home doses supported patient treatment goals by increasing flexibility and independence.
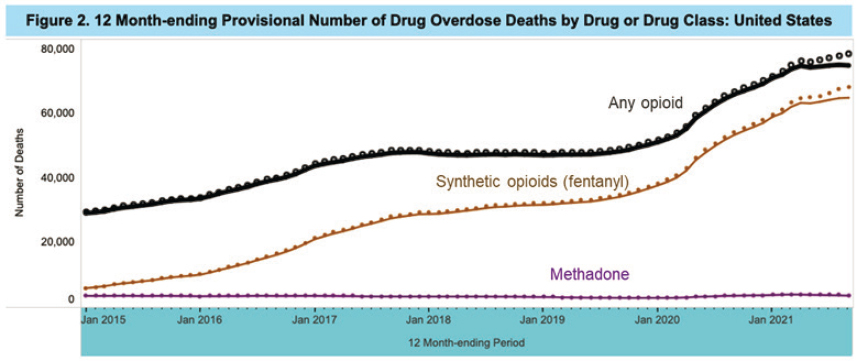
SOURCES: Presented by Noa Krawczyk, March 3, 2022; CDC, 2022.
Krawczyk added that there is little empirical evidence regarding the effects of regulatory changes on methadone retention. However, a study13 conducted through the NIDA clinical trials network is under way to investigate this question, she said.
Policy and Practice Implications of COVID-19-Related Regulatory Changes
Given that recent research indicates that the new take-home regulations are safe and effective, Krawczyk asserted that the regulations should be sustained and expanded beyond the COVID-19 pandemic. “This type of individual flexibility should really be extended to all aspects of methadone treatment, including counseling and drug testing,” she said. In addition, Krawczyk advocated for combining these reforms with other strategies, including expanding access to methadone via pharmacies, primary care, telehealth, and eliminating waiver requirements for buprenorphine.14
The majority of people in treatment for OUD, even in the United States today, still do not receive medication, said Krawczyk. “Increased access to medication in the criminal legal system is key for reaching populations that were historically neglected,” she said. Moreover, she advocated ensuring that all licensed treatment facilities in the United States offer and encourage the use of evidence-based medications for OUD.
UNDERSTANDING THE DISCONNECT BETWEEN STATE AND FEDERAL REGULATIONS
As Krawczyk noted, federal regulations may fail to achieve their intentions because of how they are implemented across states and OTPs. Haffajee agreed that synergizing federal and state policies with regard to COVID-19 flexibilities is challenging, in part because SAMHSA provided the flexibilities but states had to opt in and provide the services. She added that while the federal government has multiple ways of declaring a public health emergency, all states have public health emergency powers as well. “It’s a complicated rubric of different laws interplaying,” said Haffajee.
___________________
13 To learn more about the Optimal Policies to Improve Methadone Maintenance Adherence Long-Term (OPTIMMAL) study, go to https://nida.nih.gov/about-nida/organization/cctn/ctn/research-studies/optimal-policies-to-improve-methadone-maintenance-adherence-longterm-optimmal (accessed May 6, 2022).
14 During the open discussion period, workshop attendee Ruth Potee added that while she believes methadone should be prescribed by primary care doctors, just changing the regulation may not be sufficient, because less than 10 percent of primary care physicians have obtained the waivers necessary to prescribe buprenorphine, and far fewer actually prescribe it.
The Pew Charitable Trusts recently analyzed OPT regulations as of June 1, 2021, in all 50 states and the District of Columbia. Early findings from this review were presented by Frances McGaffey, an officer with Pew’s substance use prevention and treatment initiative. To conduct this review, the research team coded state regulations and laws, compiled their results and sent them to state officials for verification. The work will be published as an interactive tool on the Pew website in Summer 2022. One state, Wyoming, provided no data as the state has no OTPs or related regulations, said McGaffey.
McGaffey highlighted some of the key findings:
- Twenty states restrict providers from opening new OTPs in some ways, either by requiring a certificate of need, placing a cap on the number of facilities that can open (one state, Indiana), or placing a moratorium on new OTPs (one state, West Virginia). McGaffey noted that restrictions on new OTPs contribute to long drive times to clinics, particularly in rural areas where the drive time is estimated to be six times longer than for people living in urban areas (Joudrey et al., 2019a).
- Although federal guidelines recommend avoiding administrative discharge of patients for violating program rules, such as for continued drug use, this practice is explicitly permitted in 27 states and prohibited in only 2 states, Massachusetts and South Carolina. McGaffey commented that research demonstrates it is safer to continue medication than to suddenly stop it (SAMHSA, 2020; White et al., 2005).
- While most states allow take-home medications during the first month of treatment, Pew found 10 states that prohibit this practice. Moreover, as McGaffey noted, “It’s important to note that clinic policy matters here. Just because it’s allowed by the state, that doesn’t mean the provider allows it.” Indeed, in a study conducted in collaboration with the Urban Survivors Union, Figgatt and colleagues reported wide variation in how clinics adapted to COVID-19–related flexibilities (Figgatt et al., 2021).
- Ten states also have take-home stability criteria that are more stringent than the eight requirements laid out in the federal rules, said McGaffey. For example, California requires that patients are employed, in school, a homemaker, retired, or medically disabled. “This limits the ability of people who might be earlier in their recovery, unstably housed, or just not in a position to be in school or employed from receiving take-home medications,” said McGaffey.
- Federal guidelines require adequate substance use counseling to each patient as clinically necessary, but do not include a set counseling schedule. Nonetheless, McGaffey said 23 states impose counseling requirements and often tie them to the ability to receive take-home medications, despite research showing that strict counseling requirements can reduce retention in treatment and that medication without counseling can be effective (Hochheimer and Unick, 2022).
- Twenty-six states require more than the eight urine drug screens mandated under federal rules; 10 of these states require observation of sample collection, a practice that McGaffey notes can be degrading to patients. Moreover, she said urine drug screenings may not be necessary for patient safety, citing a Bronx OTP that suspended urine drug screenings because of COVID-19 and reported no fatal overdoses over the next 3 months (Joseph et al., 2021).
McGaffey concluded that in multiple areas, state rules do not align with federal requirements. One implication of this is that states could take steps to improve access to care even without waiting for federal action. For example, she said, Colorado recently changed regulations to remove non-evidence-based rules around urine drug screening, staffing ratios, and take-home medication.
McGaffey suggested that a more permanent solution might be legislation at the federal level, but acknowledged the challenge of issuing mandates versus providing flexibilities to different states. Moreover, she noted that changing federal rules around methadone will not be sufficient, said McGaffey. “Federal policy makers will have to work very closely with their partners in the states to make sure that changes are implemented on the ground and that patients actually receive methadone that meets their needs.” She added that state policy makers are often unaware of the areas of misalignment with federal regulations. One of the goals of Pew’s research, she said, is to raise awareness of the areas in which state regulations are not based on evidence.15
Individual workshop participants discussing current federal methadone regulations and regulatory flexibilities introduced in response to the COVID-19 pandemic offered potential ways forward to improve methadone access within the context of the existing regulatory landscape (see Box 4-1).
___________________
15 For more information about the Pew Charitable Trusts’ Substance Use Prevention and Treatment Initiative, go to https://www.pewtrusts.org/en/projects/substance-use-prevention-and-treatment-initiative (accessed June 10, 2022).
















