5
Prioritized Research Agenda
This chapter addresses Charges 2 and 4 of the Statement of Task, which call for the identification of the current scientific challenges for understanding low-dose and low-dose-rate radiation-induced health effects and for recommending a long-term strategic and prioritized research agenda to address scientific research goals. The committee’s prioritization process is discussed in Section 5.1 and the research priorities in Sections 5.2–5.4. Section 5.5 provides the timeline and cost estimates for implementing the recommended research agenda, and Section 5.6 compares the committee’s recommended agenda to those of other entities.
5.1 LOW-DOSE RADIATION RESEARCH CHALLENGES AND OVERVIEW OF RESEARCH PRIORITIES
The committee addressed Charge 2 of its Statement of Task by listing challenges for epidemiological and biological research as well as some that are common to both research approaches (see Box 5.1). These challenges arise because the effects of low-dose and low-dose-rate radiation exposures are assumed to be subtle and difficult to distinguish from those caused by other stressors or “spontaneous” changes that adversely affect the normal functions of cells, tissues, and organs. Moreover, a full understanding of possible effects may be complicated by change in the magnitude of observed effect with dose, dose rate, type of radiation, and duration of exposure.
The committee addressed Charge 4 of its Statement of Task by proposing an agenda for a multidisciplinary research program intended to improve the evidence base used as the foundation for protection of the U.S. population
against the adverse health effects that result from exposures to low-dose and low-dose-rate radiation. Knowledge gaps of the current radiation protection system are summarized in Box 3.1 in Chapter 3. The proposed research agenda aims to fill these gaps by integrating information from epidemiological analyses of the adverse health effects of low-dose radiation with information on cell and molecular responses of humans and laboratory models to exposures to low-dose and low-dose-rate radiation revealed by new-generation analytical tools. Approaches for integrating information from radiation biology and epidemiology to enhance low-dose health risk assessment are described in detail elsewhere (NCRP, 2020a). The greatest benefits and most efficient and rapid progress in the program will be achieved through coordinated multidisciplinary research that harnesses new technologies, methodologies, and biomedical understanding that are being developed by the U.S. scientific enterprise. To achieve this, a sustained and coordinated low-dose and low-dose-rate research community will need to be created and nurtured.
The committee recommended research program leverages advances in modern science and sets ambitious goals for revitalized low-dose radiation research in the United States: to improve understanding of adverse human health effects from exposures at doses and dose rates experienced by the U.S. population, to identify mechanisms for induction of these health effects, to develop improved risk models for doses and dose rates at which direct measurement of risks is not possible or limited, and to develop more individualized risk estimates. As such, it is expected to require multiple decades of investment. These investments are expected to be at the level of $100 million per year over a period of 10–15 years, but periodic reassessments are required as suitable study cohorts and necessary infrastructures are established (see Section 5.4).
The criteria used by the committee to identify priorities for low-dose and low-dose-rate research included (1) existing human, laboratory model, and cellular evidence for adverse health effects resulting from radiation exposure; (2) limitations in the current radiation protection system in the United States; (3) feasibility of improving low-dose and low-dose-rate risk estimation models given newly available technologies and resources as well as increased understanding of human disease mechanisms; and (4) issues of concern for exposed populations and impacted communities. The committee’s list of 11 research priorities considered these four criteria, and the proposed approaches for addressing each are summarized in Table 5.1. These research priorities are broadly classified as epidemiological research, biological research, and research infrastructure. The committee strongly emphasizes the need for integration across the research lines and anticipates that the most impactful projects will include work in more than one research line and will be carried out by multidisciplinary teams and that some may require collaboration with international partners.
The epidemiological line of research focuses on directly quantifying the risks of adverse health outcomes following low-dose and low-dose-rate exposures to the types of radiation that are or may be experienced by the U.S. population. The biological line of research focuses on the mechanistic underpinnings of the effects of low-dose and low-dose-rate radiation on molecular pathways including influences from intra- and extracellular interactions and the identification of potential causal relationships to different health outcomes. The infrastructure line of research focuses on developing or deploying new observational and experimental systems, computational technologies, and shared access data systems.
The proposed research will address cancer and non-cancer health outcomes including cardiovascular disease, neurological disorders, immune dysfunction, cataracts, and heritable genetic effects for both internal and external exposures. It also encourages a focus on directly observing health effects from low-dose and low-dose-rate exposures, complemented with biological studies that emphasize exposures below 10 milligray (mGy) representative of the majority of exposures of interest for radiation protection, and/or exposures delivered at dose rates around 5 mGy/h.
The order of the research priorities in Table 5.1 does not imply an order of significance; instead, the priorities are considered to be equally important. Some of these activities can be initiated immediately, and others can only begin after a better foundation is built from current or new research or with additional input from the research and broader stakeholder community, including the impacted communities. The committee’s views on the timeline for research activities for the first 10–15 years are discussed in Section 5.5.
In the committee’s judgment, the 11 research priorities will enable more accurate estimation of adverse health effects that result from exposure to low-dose and low-dose-rate radiation and will dramatically improve knowledge of the complex cellular and molecular processes that are engaged during transduction of low-dose and low-dose-rate radiation damage into adverse health outcomes. The committee also noted that some of the research priorities can have additional benefits including capacity building, training of the next generation of radiation researchers, and development of tools that could be transferrable to other lines of research. However, the committee recognizes that these research priorities do not represent a complete list of important low-dose and low-dose-rate radiation research questions. For example, studies designed to confirm or strengthen the basis for existing scientific findings, particularly those that are controversial or lack a clear interpretation, are also important. In addition, research in radioecology, in the psychological effects following low-dose radiation exposures, and in radiation risk communication are not included in the recommended strategic agenda but are topics that are worth exploring by the low-dose radiation program in the future.
TABLE 5.1 Committee Recommended Research Priorities for Low-Dose and Low-Dose-Rate Radiation Research
| Priority Research Goal | Approach | Integration Across Research Lines |
|---|---|---|
| Epidemiological Research | ||
| E1 Develop and deploy analytical tools for radiation epidemiology. | Develop cohorts of sufficient size, with detailed health information and biosample collection and accurate dosimetry, to support epidemiological studies of radiation-induced health effects in medically, occupationally, and environmentally exposed U.S. populations. | B2–B4; I1–I3 |
| E2 Improve estimation of risks for cancer and non-cancer health outcomes from low-dose and low-dose-rate external and internal radiation exposures. | More precisely define health outcomes to enable exclusion of diseases caused by other effects, identifying easily measured signatures that can serve as disease surrogates by improving dosimetry and identifying and compensating for confounding and modifying factors. | B1–B4; I1–I3 |
| E3 Determine factors that modify the low-dose and low-dose-rate radiation-related adverse health effects. | Assess the impact of genetic makeup, epigenomic status, DNA repair efficacy, comorbidities, exposure history to radiation and other agents, lifestyle and psychosocial factors, and immune status on radiation-induced adverse health outcomes. | B1–B4; I1–I3 |
| Biological Research | ||
| B1 Develop appropriate model systems for study of low-dose and low-dose-rate radiation-induced health effects. | Identify laboratory model systems in which molecular, cellular, and pathological features of radiation-induced health effects are similar to humans. | E2–E3; I1–I4 |
| B2 Develop biomarkers for radiation-induced adverse health outcomes. | Identify radiation-induced changes in cellular and molecular features that causally link to adverse health effects in appropriate model systems. | E1–E3; I1–I4 |
| B3 Define health-effect dose-response relationships below 10 mGy and below 5 mGy/h. | Establish radiation dose-response curves for molecular and cellular endpoints and for associated early- and late-stage diseases at doses below 10 mGy and dose rates below 5 mGy/h. | E1–E3; I1–I4 |
| B4 Identify factors that modify or confound estimation of risks for radiation-induced adverse health outcomes. | Assess the impact of genetic makeup, epigenomic status, DNA repair efficacy, comorbidities, exposure history to radiation and other agents, lifestyle factors, and immune status on low-dose and low-dose-rate radiation-induced adverse health effects and associated cellular and molecular response endpoints. | E1–E3; I1–I4 |
| Research Infrastructure | ||
| I1 Tools for sensitive detection and precise characterization of aberrant cell and tissue states. | Identify, develop, and deploy bulk and single-cell -omicsa and image measurement and computational analysis workflows to quantify disease-linked cellular and molecular signatures that are sufficiently sensitive, reliable, and low cost for wide-scale application. | E1–E3; B1–B4 |
| I2 Harmonized databases to support biological and epidemiological studies. | Develop accessible databases that document exposure levels, rates, types, and durations as well as cell, molecular, and health outcomes for human populations and experimental models. | E1–E3; B1–B4 |
| I3 Dosimetry for low-dose and low-dose-rate exposures. | Elucidate biological localization of internalized radionuclides; directly measure radiation-induced damage and associated response mechanisms; develop high-fidelity anatomically and physiologically based dosimetry; develop and apply modern statistical and computational methods for dose reconstruction. | E1–E3; B1–B4 |
| I4 Facilities for low-dose and low-dose-rate exposures. | Ensure access to low-dose and low-dose-rate exposure facilities, including those allowing internal exposure in model systems by a variety of routes (e.g., inhalation, ingestion) or invest in new facilities. | B1–B4 |
a The broader field of “-omics” includes genomics, transcriptomics, proteomics, metabolomics, and radiomics.
NOTE: mGy = milligray.
The research priorities are discussed in Sections 5.2–5.4 with emphasis on significance, current status, and promising research directions. The committee expects that the specific tactics for addressing these priorities will be developed with input from the extended research community and other stakeholders, including the impacted communities. Importantly, the committee recognizes that the list of priorities will likely evolve as biological understanding and research tools advance and as the research community and other stakeholders are engaged with the program (see Chapter 6).
5.2 EPIDEMIOLOGICAL RESEARCH PRIORITIES
Epidemiological studies of radiation-exposed human populations have provided the information that is used worldwide to estimate risks of adverse health outcomes from these exposures and to guide regulatory decision-making. However, past studies have been limited in their ability to inform on risks from low-dose and low-dose-rate radiation exposures that are of most relevance to U.S. populations because of the challenges described in Box 5.1. The following sections suggest several ways to help overcome some of these challenges and therefore increase understanding of the adverse health effects that result from exposure to low-dose and low-dose-rate radiation. These include development and deployment of analytical tools for radiation epidemiology (see Section 5.2.1), improved estimation of risks for cancer and non-cancer health outcomes (see Section 5.2.2), and identification of factors that modify the low-dose and low-dose-rate radiation-related adverse health effects (see Section 5.2.3).
5.2.1 Develop and Deploy Analytical Tools for Radiation Epidemiology (Priority E1)
Scientific and Decision-Making Value
Powerful analytical tools are now available or are being developed that allow more accurate measurement of radiation exposures and exposure rates and more precise definition of the adverse health effects that may arise due to radiation exposures. Application of these tools in epidemiological studies of human populations will improve investigations of adverse health effects that may be caused by low-dose and low-dose-rate exposures. Selection of the most appropriate study populations and study designs can best be done by the low-dose and low-dose-rate research community with input from the broader research community and other stakeholders, including the impacted communities. These might include populations exposed during childhood, those exposed during medical procedures or occupationally, and those who are environmentally exposed, for example, those living near
nuclear waste sites who may ingest or inhale radioactive materials from contaminated environments, or those exposed to background radiation including residential radon (see Table 2.1).
Current Status and Promising Research Directions
Epidemiological studies aiming to directly quantify the adverse health effects that result from low-dose and low-dose-rate radiation exposures, either internal or external, will require careful selection and detailed characterization of study populations that allow examination of lifetime risks of radiation exposures. These study cohorts may be assembled in several ways, for example, by
- Building on existing historic cohorts. These cohorts may have been assembled to address a different scientific question but might be repurposed to assess risks at low doses of radiation. In this case, investigators would take advantage of data already collected and collect additional information through medical records, Medicare and Medicaid claims, death records, or other means to support studies of radiation risks. In some cases, this may require data harmonization in order to enable combination of multiple cohorts. An example of that approach is the Million Person Study that consists of more than 30 historic cohorts (Boice et al., 2022a). Historic cohorts for which biological samples exist may be of great value as they could allow more precise definition of disease phenotypes or assessment of molecular response endpoints—assuming that sample collection and preservation methods are compatible with the assays to be performed. In some cases, active follow-up of a subgroup of cohort members may be possible to collect additional information and biosamples or passive follow-up at regular intervals, to update the incidence or mortality data and to conduct new dose-response evaluations.
- Initiating new retrospective cohorts. These cohorts might be assembled by reconstructing exposures to the populations of interest over time and examining how these exposures affected health endpoints over the years by looking for disease occurrence or death from disease through cancer registries, medical records, Medicare and Medicaid claims, death records, or other means. The EPI-CT cohort of medically exposed individuals (see Section 2.1.2) is an example of an international retrospective cohort that was facilitated by electronic record linkage, and uses an improved and standardized dosimetric approach.
- Initiating studies that have both retrospective and prospective phases. These cohorts might be assembled using a retrospective
- Developing prospective cohorts. Assembly of the very large numbers of individuals (likely millions) and the long follow-up (several decades depending on the population) for disease occurrence needed for low-dose and low-dose-rate epidemiological studies requires significant effort and resources. However, assembly of informative, new cohorts may become feasible in the future by employing more efficient sampling methods and by taking advantage of more precise information about radiation exposures and disease phenotypes that inform on etiology that is expected to be captured in future, computationally accessible electronic medical records (EMRs). Work on development of a “Learning Health System” (Kuntz et al., 2019) and on sharing and aggregation of data for COVID-19-related research (Dron et al., 2021; Park et al., 2021) suggest strategies that might be adopted to move radiation epidemiology in this direction.
approach to recruit participants who already have substantial follow-up and obtaining more detailed information for a subsample of the cohort, for example, socioeconomic and lifestyle factors during lifetime via interviews or other means and biosamples for molecular and other analyses.
Irrespective of the study design, desired characteristics recorded for each cohort member include (1) information on radiation types and exposure routes and precise estimates of dose and dose rates; (2) detailed health status including incidence1 of precisely defined adverse health outcomes; and (3) information on physical, chemical, and social environmental factors that may confound or modify radiation dose-adverse health-effect associations. When such information is difficult or expensive to collect for all cohort members, principled study designs based on sampling (e.g., case-cohort design, nested case-control design, validation sampling design) can be used, together with modern statistical analysis methods (Kim, 2015; Langholz and Thomas, 1990). In addition, two-phase study designs offer
___________________
1 To understand the etiology of cause-and-effect associations following low-dose radiation exposures, there is a need to conduct incidence-based follow-up studies. It is broadly recognized that the absence of a nationwide cancer incidence registry in the United States has detrimental effects on epidemiological research. The Virtual Pooled Registry Cancer Linkage System, coordinated by the North American Association of Central Cancer Registries with funding from the National Cancer Institute, is an ongoing effort to connect researchers performing minimal risk linkage studies with multiple U.S. population-based cancer registries. Acquiring incidence information for endpoints other than cancer will require electronic medical record linkages because there are no registries for these other disease endpoints.
cost-effective sampling strategies for the collection of additional information (which may be expensive) on a subset of the overall cohort (Tao et al., 2020).
Relevant to issue (3), above, is the issue of confounding. Without the benefit of random assignment, comparison groups in epidemiological studies may differ with respect to factors other than radiation exposure. If these factors are also related to the adverse health outcome of interest, then the observed effect of radiation on adverse health outcome risk may be indistinguishable from the effects of these other risk factors (Schubauer-Berigan et al., 2020), that is, be confounders. Inadequately accounting for confounding can lead to bias in an estimate of the association between radiation exposure and adverse health outcome.2
Strategies to increase the accuracy of radiation exposure estimates may include accessing records of medical exposure types and levels; using individual and in-home radiation monitors that report continuously on occupational and environmental exposures; generating accessible geospatial databases of environmental radiation and other contaminants; and utilizing computational algorithms that accurately estimate organ- and cell-specific dose and dose rates from external and/or internal sources and that account for source radiation type, internal versus external exposure, and body size and composition, anatomic location, and sex. These algorithms may be further improved by incorporating information from biological studies using new-generation nanoscale analysis tools that reveal how individual photons or ions alter DNA, individual proteins, and organelles in individual cells and how these alterations are subsequently processed biologically. Relevant analytical technologies are described in more detail in Section 5.4.1 (Priority I1). In addition, it is crucial to understand the random and systematic errors in this information, which could lead to dose estimation errors.
Resources that may be exploited to improve individual cohort members’ health status assessments may include
- EMRs made accessible for purposes authorized by law or by the patient while maintaining individual privacy. Concepts and tools developed to support research in COVID-19 testing, treatments, and vaccines and/or the development of a “Learning Health System” (Kuntz et al., 2019) may be particularly important guides.3 The utility of EMRs for low-dose and low-dose-rate radiation research can be increased by developing and promoting nationwide use of health outcomes data standards that will guide collection of
___________________
2 Not all risk factors are confounders. In addition, confounding is more likely to impact exposed-unexposed comparisons than dose-response analyses.
3 Mike Snyder, Stanford University, presentation to the committee on November 17, 2021.
- Information from cellular and molecular analyses of tissues including associated microbiomes in which low-dose and low-dose-rate radiation-induced diseases may originate and biofluids4 that interact with these tissues. Technologies for precise disease phenotyping are described in detail in Section 5.4.1 (Priority I1). These analyses may include assessment of the presence of signatures that have been associated with low-dose and low-dose-rate radiation-induced disease in laboratory model systems. They may quantify individual characteristics (e.g., genetic, epigenetic, and immune status) that may influence risk of developing adverse health outcomes from low-dose and low-dose-rate radiation exposures. Studies of cells and biomolecules in biofluids that interact with radiation-induced disease sites may be particularly important because not all anatomical sites are easily accessible (Bhawal et al., 2020; Hampel et al., 2021; C. Huang et al., 2021; Quigley et al., 2017). Indeed, recent genomic studies demonstrate the existence of clonal subpopulations within individuals including those irradiated in utero (Applegate et al., 2021) that originate and are propagated during development (Li et al., 2021; Moore et al., 2020). These clonal subpopulations may carry genomic or epigenomic changes that put the cells carrying them at increased risk of radiation-induced disease. New-generation single-cell analysis tools may identify and characterize these at-risk cells (Adhikari et al., 2020; HuBMAP Consortium, 2019; Rozenblatt-Rosen et al., 2020; Stuart et al., 2019) but biofluid sampling will likely be needed to enable their practical detection in humans.
- Information on health status from Internet of Medical Things (IoMT) devices (Popov et al., 2022). These devices may include wearable and in-home sensors and smart phones that can provide information on health endpoints such as weight, gait and balance, voice pathology, heart function, temperature, glucose and other aspects of blood chemistry, cognitive function, and eye movement, which may be adversely affected by exposure to low-dose and low-dose-rate radiation. These devices may also reveal information about food and drug consumption that may alter or mimic
the information needed for low-dose and low-dose-rate radiation epidemiology (e.g., precisely defined disease states and accurate, anatomically precise measures of radiation exposure). Information on individual medical exposures to radiation will be especially important.
___________________
4 Biofluids may include blood, oral swabs, urine, and fecal material and would be collected at appropriate time points based on the hypotheses to be tested.
- Information from geospatial databases that can be accessed to provide information about physical, chemical, and social environmental factors that may influence aspects of human physiology that may directly or indirectly influence or confound the identification of low-dose and low-dose-rate radiation-induced adverse health effects (Olney, 2021). Examples of geospatially defined features that might be associated with individuals in study cohorts include aspects of health5 climate,6 environmental pollutants,7 and levels and types of environmental radiation (Dindaroǧlu, 2014). The information from geospatial databases could complement the individual-level factors obtained by the IoMT devices or be the source of information when individual-level monitoring is not available. Harmonized databases to support biological and epidemiological studies are discussed in Section 5.4.2 (Priority I2).
radiation-induced health effects. This type of data can be collected on a sample of the cohort population over a long time span. Selected technologies available for characterization of human health endpoints are summarized in Section 5.4.1 (Priority I1).
5.2.2 Improve Estimation of Risks for Cancer and Non-Cancer Health Outcomes from Low-Dose External and Internal Radiation Exposures, Including Suitable Surrogate Biomarkers of Health Risk Where Appropriate (Priority E2)
Scientific and Decision-Making Value
The quantitative relationship between exposure to radiation and cancer risk at the low doses (<100 mGy) most commonly encountered by the U.S. population and low dose rates (below 5 mGy/h) is assumed to be linear, but linearity is not well established experimentally or epidemiologically at these low doses. Even less is known about the levels of risk for health outcomes other than cancer, including cardiovascular disease, neurological disorders, immune dysfunction, cataracts, and heritable genetic effects. However, if such risks exist at low doses and dose rates, they could lead to substantial changes in risk-benefit analyses for activities that involve low-dose radiation exposures. Also, the health impacts of internal exposures to low-dose and low-dose-rate radiation are not well studied, although both routes of exposure are relevant to the U.S. population. Advances in dosimetry, biology, geospatial epidemiology, and disease phenotyping are now sufficient
___________________
5 See https://www.cdc.gov/dhdsp/maps/gisx/resources/geo-spatial-data.html.
to encourage the design of epidemiological studies that can identify and quantify adverse health outcomes resulting from low-dose and low-dose-rate radiation exposures, while identifying and excluding factors other than radiation that may affect the same adverse health outcomes (i.e., confounding and modifying effects).
Current Status and Promising Research Directions
Cancer is the most well-established adverse health outcome resulting from radiation exposure (Berrington de González et al., 2017; Hauptmann et al., 2020; Kitahara et al., 2015; Little et al., 2022a; UNSCEAR, 2006a). Evidence from a range of exposure scenarios at higher doses consistently demonstrates elevated cancer risks for nearly all tissues based on data from the atomic bombing survivors (Grant et al., 2017) and other exposed populations (UNSCEAR, 2008), with highest risks for leukemia (excluding chronic lymphocytic leukemia) and tumors of the brain, bladder, skin, and thyroid. Evidence accumulated during the past decade has shown that these patterns persist even in populations exposed to low-dose and low-dose-rate radiation (mean doses <100 mGy; see, e.g., Berrington de Gonzalez et al., 2020; Kitahara et al., 2015), although direct evidence at doses around 10 mGy remains limited, and uncertainties remain as to how these endpoints are influenced by radiation type and route of exposure.
Even greater uncertainty exists regarding low-dose radiation-associated health effects for outcomes other than cancer (cardiovascular, neurological, heritable genetic, and other effects; see Section 2.2), but, generally, associations have been observed over the past decade at doses lower than previously considered important for these effects. For example, although the International Commission on Radiological Protection (ICRP) considers cardiovascular effects to fall under the category of tissue reactions and assumes a threshold for induction of 500 mGy, some epidemiological analyses and meta-analyses indicate an elevation of risk at doses less than 500 mGy (e.g., Little et al., 2012; Tran et al., 2017) and indicate that risk of circulatory disease may follow a linear dose-risk relationship at doses below 500 mGy (Little et al., 2012). Association of cardiovascular disease with low doses of radiation remains controversial due to the possibility of uncontrolled confounding by lifestyle factors.
The future availability of detailed and precise information on cancer and non-cancer health outcomes (see Section 5.3.2 for Priority B2), coupled with accurate information on levels and rates of radiation exposure (see Section 5.4.3 for Priority I3), offers the opportunity to explore dose-response relationships at doses relevant to exposed U.S. populations more precisely. This will require appropriate study designs and deployment of new association discovery methodologies to ensure that these associations
are statistically significant and false discovery is minimized in these high-dimensionality datasets. During study design, consideration needs to be given to focusing on health outcomes that are precisely defined (e.g., using molecular, cellular, and/or physiological assessment tools), ideally with respect to factors relevant to etiology and that are not influenced by treatment strategies that may change over time or with economic status. Quantitative health outcomes and molecular surrogates thereof that are suggested by epidemiological studies at higher doses or that have been shown in laboratory model studies to be related to low-dose and low-dose-rate radiation exposure might be given special attention. Association discovery may be based on traditional statistical methods or may take advantage of the recent development of computational methods (e.g., machine learning algorithms and artificial intelligence). The ultimate goal is to identify adverse health outcomes that are caused by exposure to low-dose and low-dose-rate radiation.
Statistical and computational methods for association discovery need to address statistical power and bias. Bias can be ameliorated by applying Bradford Hill’s considerations for assessing causality, together with modern statistical methods for making causal inference from epidemiological data (Marshall and Galea, 2015). In addition, statistical methods have recently been introduced to estimate causal dose-response curves, based on generalized propensity score models to account for confounding, together with parametric, semi-parametric, or non-parametric dose-response models.8 Non-parametric models do not rely on an assumed functional form for the association between radiation dose and outcomes (e.g., linear no-threshold). These methods have been applied to low-level air pollution research and could be applied to low-dose radiation epidemiology but require extension to accommodate measurement error in radiation doses. Additional methodological research is needed to establish and compare the statistical properties of new analysis methods, in order to understand their appropriate application in different scientific contexts.
Analyses of low-dose radiation effects do not need to be overly reliant on arbitrary levels of statistical significance (Wasserstein and Lazar, 2016). In fact, within individual studies, the type I error rate (i.e., the false-positive rate) can be inflated due to conducting multiple statistical hypothesis tests for several outcomes, both overall and among subgroups. Instead, inference can be focused on confidence intervals, which provide the range of (excess) risks over which the data are consistent. In addition, it is essential to recognize that a single study is unlikely to provide definitive evidence of radiation effects at low doses and whether those effects differ according to biological
___________________
8 Francesca Dominici, Harvard T.H. Chan School of Public Health, presentation to the committee on December 9, 2021.
factors, lifestyle factors, or other environmental exposures. Instead, evidence needs to be combined from multiple studies. Meta-analyses of study-level data can be useful to estimate average effects across studies but require that similar statistical models are fitted to the underlying individual-level data. Alternatively, pooled analyses of harmonized individual-level data can be used, if they are available, together with mixed-effects models that allow for unobserved heterogeneity across studies. To this end, Bayesian methods (full or empirical) need to be developed and applied (Smith et al., 1995). Dose-response shapes and patterns of effect modification can be informed by, but not explicitly depend on, a model for effects at higher doses. They may also be informed by the dose-response curve shape observed in exposed laboratory models as described in Section 5.3.3 for Priority B3.
Classical machine learning and newer artificial intelligence algorithms implemented on increasingly powerful computational platforms are now being used to integrate large-scale -omics, image, biological, and clinical datasets (Goecks et al., 2020) in ways that identify regulatory mechanisms that may control biological and clinical phenotypes and/or that predict biological or medical behavior. Artificial intelligence has an increasing impact in biological (Jumper et al., 2021) and medical areas (Topol, 2019). Application of classical machine learning and newer artificial intelligence algorithms to identify adverse health endpoints that are associated with low-dose and low-dose-rate exposures and the operative response mechanisms will become increasingly productive as robust and accessible datasets are developed for carefully designed study retrospective or prospective cohorts. These tools are summarized in Section 5.4.1 for Priority I1.
5.2.3 Determine Factors That Alter the Low-Dose and Low-Dose-Rate Radiation-Related Adverse Health Effects (Priority E3)
Scientific and Decision-Making Value
Several factors may influence an individual’s sensitivity to radiation and therefore need to be considered in risk assessment and risk management. Age at exposure has been shown to modify the radiation dose-response relationship for some cancer types. Higher risks have generally been reported for individuals exposed at younger ages (Grant et al., 2017), and for breast and uterine cancers, reported risks were higher for women exposed around the age of menarche (Brenner et al., 2018; Utada et al., 2018). Animal studies have provided some insights on the biological mechanisms underlying the influence of age at exposure to individual response to radiation (see Applegate et al., 2020, for review). Radiation dose-response relationships may also vary between sexes with some studies suggesting higher risks for women compared to men (ICRP, 2007). One notable example of radiation
sensitivity of women versus men comes from analyses of the Life Span Study cohort of atomic bombing survivors which showed a three times higher excess risk of incident lung cancer due to radiation exposure among women compared to men (Cahoon et al., 2017; Hu et al., 2021). However, recent analyses of occupational cohorts within the Million Person Study did not provide evidence of significant differences in the lung cancer risks between men and women (Boice et al., 2022c). The National Council on Radiation Protection and Measurements (NCRP) is currently evaluating sex-specific differences in lung cancer radiation risks and assesses their use in transfer models and lifetime risk projections. Little is known about the effects of other factors and on other health outcomes. Plausible modifiers may involve the host (e.g., inherited genetic susceptibility, existence of clonal subpopulations, immune constitution, comorbid medical conditions) and/or lifestyle (e.g., smoking, obesity, physiological stress, diet). Knowledge of the effects of these other factors on low-dose and low-dose-rate radiation-induced health outcomes may allow for more individualized risk assessments and risk management. Variation in individual response to radiation is a topic of growing importance for radiological protection (Rajaraman et al., 2018; Wojcik et al., 2018) and of an ongoing ICRP task group.9
Current Status and Promising Research Directions
Evidence for inherited genetic susceptibility to radiation response derives largely from rare genetic syndromes in which individuals demonstrate hypersensitivity to the killing effects of radiation and frequently have increased risk for developing cancer, such as ataxia-telangiectasia, Nijmegen breakage syndrome, and others (Pollard and Gatti, 2009). Much of the research on radiation-related health impacts in individuals who are autosomal recessive for these deleterious mutations in genes has been used to understand pathways related to high- and low-dose radiation responses, particularly DNA damage responses. Evidence from both in vitro and animal studies suggests that at least some radiation responses differ when comparing low- to high-dose exposures. Nevertheless, the research on genetic modifiers of high-dose radiation effects on health provides important proof of principle that such studies are important. To date, because of small sample sizes and lack of replication in independent populations, these studies have not robustly identified germline genetic variants that modify radiation-related health risks. Future studies that assess potential germline variation in radiation-related health risks need to follow best practices
___________________
9 See Task Group 111 Factors Governing the Individual Response of Humans to Ionising Radiation, https://www.icrp.org/icrp_group.asp?id=169.
established over the last decade, including sufficient sample size and replication of results in independent populations.
Immune constitution is thought to modify the effects of radiation either by altering the efficacy with which damaged cells are removed or by altering cytokine levels that alter cell phenotypes including differentiation state, proliferation, and motility. Advances in immunology over the past three decades led to the extension of the original self/non-self-theory, adding common alarm signals, so-called damage-associated molecular patterns, or danger signals as chief drivers for immune engagement (Matzinger, 1994). More recently, recognition systems for RNA and DNA have been added to the list of biomolecules that signal danger to the immune system. This is important because radiation exposures, like many stresses and injuries, get relayed in vivo through shared pathways, especially along the danger sensing and inflammatory/immune signaling cascades, that are open to amplification and exacerbation over time (McBride et al., 2004). Permanent damage may be localized, such as the stem cell compartments or their niches, whose function can be directly or indirectly affected along with their resilience to other stresses (Rodrigues-Moreira et al., 2017). Such permanent radiation damage has been shown largely in studies utilizing higher radiation doses and in infectious, inflammatory, and autoimmune diseases, for example through epigenetic modifications or through persistent senescence that disturbs tissue dynamics causing inflammation (Campisi et al., 2011).
Low-dose radiation scenarios are less studied, but one example that might be relevant is inflammatory recall where open chromatin memory domains become readily accessible to homeostatic transcription factors and in essence change the threshold for activation (Larsen et al., 2021). It is possible that low-dose and low-dose-rate radiation also alter the homeostatic intracellular redox rheostat to affect responses to other challenges, just as much as other challenges might affect the redox rheostat and therefore the response to radiation. It is currently also unclear if the immunological changes following low-dose radiation exposures actually link to the same long-term and late clinical disease outcomes as high-dose radiation exposures or if other outcomes are more relevant (Boerma et al., 2022). It is also still unclear whether there is memory of low-dose radiation exposures or what the importance in the context of other stressors such as obesity, infection, or trauma might be. Feasibility and cost have limited most of these studies primarily to detecting imbalances in peripheral immune cell subsets with immune function. However, this is changing as more powerful tools (see Section 5.4.1 for Priority I1) for assessment of immune phenotypes are developed. These can now be deployed to determine whether immunological changes seen post-exposure are associated with adverse health impacts and how these are modified
by other stressors. The latter is of interest because an expert who briefed the committee on aspects of brain function and human physiology10 noted that the brain operates as a strong signal amplifier to help the body respond to stress, infection, and other evolutionarily important survival endpoints. The brain, and particularly the hypothalamus, receives and sends chemical signals that modify cellular behavior throughout the body, especially aspects of immune function. Depending on context, these interactions can be either beneficial (e.g., by enhancing immune response to invading pathogens) or deleterious (e.g., by stimulating cancer cell growth or contributing to cachexia). Overall, these mechanisms raise the possibility that stress, immune function, and neural signaling interact to influence how cells, tissues, and organs respond to radiation.
Regarding lifestyle factors, perhaps the best studied factor to date is tobacco exposure and its impact on estimates of radiation-related lung cancer risks, with most evidence supporting a sub-multiplicative effect (Cahoon et al., 2017; Tomasek, 2013). Whether these findings are consistent for tobacco and radiation exposures on other outcomes also associated with tobacco exposure (e.g., bladder cancer and cardiovascular disease) or for combined effects of radiation and other lifestyle factors such as obesity and diet is unknown. Understanding these combined effects is important because both obesity and diet contribute to disparities in many diseases, including cancer and cardiovascular disease.
There is also limited evidence relating to the health risks associated with combined exposure to radiation and other agents to which people are exposed and which may modify the effect of radiation. However, this is of great importance in different occupational and environmental exposure settings where populations are exposed to radiation and chemicals or other harmful agents. The last comprehensive review of co-exposure effects (UNSCEAR, 2000) concluded that, in general, genotoxic agents with similar biological and mechanistic behavior when acting at the same time will interact in a concentration-additive manner. This conclusion was supported by more recent studies (Nuta et al., 2014), but other reports demonstrated greater-than-additive effects of radiation and arsenic compounds on some cellular endpoints (Hornhardt et al., 2006).
Epidemiological studies with high-quality data on host and lifestyle factors (e.g., from increasingly powerful geospatial databases) as well as biospecimens that can be used for germline genomics or other biomarker studies (e.g., epigenomic, protein, and transcriptional signatures that regulate cell and tissue behavior; see Section 5.3.2), together with statistical and
___________________
10 Daniel Marks, Oregon Health & Science University, presentation to the committee on November 16, 2021.
computational methods discussed in Section 5.2.1 (Priority E1), can provide evidence on how these factors modify low-dose and low-dose-rate radiation health effects. Major advances in genomic technologies that now allow for large-scale genotyping and sequencing to assess both common and rare genetic variants hold promise for the development of studies aimed toward understanding heritable genetic contributions to radiation-related health impacts (see Section 5.4.1 for Priority I1).
5.3 BIOLOGICAL RESEARCH PRIORITIES
Elucidation of the mechanisms that are involved in the conversion of low-dose and low-dose-rate radiation-induced damage into adverse health effects provides important support to epidemiological studies of radiation risk. Mechanistic understanding also may suggest strategies to mitigate adverse health effects that result from low-dose and low-dose-rate exposures. Mechanistic studies can focus on adverse effects in humans and/or on laboratory models thereof. Laboratory model-based studies tend to be better controlled, adequately statistically powered, and less prone to confounding, therefore substantially strengthening the evidence for disease causation and the underlying dose-response relationships, provided they accurately model the disease pathogenesis following irradiation. The following sections suggest several aspects of mechanism-based biological research that may increase understanding of how low-dose and low-dose-rate radiation exposures lead to adverse health effects, including development of improved laboratory models (see Section 5.3.1), identification of biomarkers for radiation-induced health effects (see Section 5.3.2), defining health effect-dose relationships below 10 mGy and below 5 mGy/h (see Section 5.3.3), and identification of factors that modulate or mimic radiation health effects (see Section 5.3.4).
5.3.1 Develop More Accurate Model Systems for Study of Low-Dose and Low-Dose-Rate Radiation-Induced Health Effects (Priority B1)
Scientific and Decision-Making Value
Biological studies of low-dose and low-dose-rate radiation health effects in humans will rely on the use of laboratory model systems. Informative models may be engineered cells and tissues grown in the laboratory (Suckert et al., 2021; Tognon et al., 2021) and/or engineered animal models (Collaborative Cross Consortium, 2012; Paunesku and Woloschak, 2018). The utility of information from the study of these models depends on the extent to which they accurately mirror the aspects of human biology targeted for study. The development of these models can be guided by the increasing information about the molecular, cellular, and physiological
characteristics of normal and aberrant human tissues and human diseases revealed using new-generation analytical tools. Multiple models may be used to cover different aspects of radiation-induced adverse health outcomes, and multiple models of the same outcome can be employed to guard against artifacts that may be specific to a single model. Extrapolation from experimental data to possible effects in humans is considered more reliable when similar molecular responses and/or outcomes are observed in a variety of model systems. Integration of the information gained from laboratory models and from epidemiological studies will improve understanding of the mechanisms underlying low-dose and low-dose-rate radiation-induced adverse health outcomes, improve risk estimates for the low-dose and low-dose-rate exposures experienced by the U.S. population, and suggest strategies to mitigate risks.
Current Status and Promising Research Directions
Laboratory models have strengths and weaknesses for low-dose radiation studies. In general, models comprising engineered cells, organoids (collections of cells grown in three-dimensional [3D] cultures), and tissues grown in the laboratory are more convenient and lower cost than engineered animal models but are limited to studies of specific cellular and molecular processes and mechanisms. However, animal models are better suited to studies of disease outcomes. Several characteristics and uses of engineered cells and tissues and animal models are discussed in the following sections.
For decades, studies of the biological effects of radiation have focused on cells isolated from humans and laboratory animals. These studies have revealed much of what is currently known about cellular mechanisms to respond to radiation-induced damage. In general, the cell types most studied have been those that can be easily grown in the laboratory. However, these cell types typically have been grown in two-dimensional cultures, and often they are not the cell types in which radiation-induced diseases arise. Moreover, they typically have not included the diversity of cells (e.g., epithelial, endothelial, and immune) that may interact in tissues to alter responses to radiation. These limitations can be reduced by employing new bioengineering technologies that generate 3D, multicellular biological systems, either organoids (Nagle and Coppes, 2020) or “organs-on-a-chip” (also known as tissue chips) (Low and Tagle, 2017). Tissue chips are now routinely used and have even been launched into space (Low and Giulianotti, 2019). Tissue chips coupled with microfluidics are becoming increasingly sophisticated to allow for interaction of tissue-specific cells with immune cell subsets and the microbiome, more closely modeling aspects of human and animal physiology. Artificially engineered scaffolding that supports
3D organ development in the laboratory environment is another example of designing advanced functional biomimetic structures (Nikolova and Chavali, 2019). Collectively, these systems provide exciting new tools for the investigation of molecular, cellular, and tissue responses to low-dose and low-dose-rate radiation. Processes that appear important can then be promoted for assessment in animal models and eventually for association with radiation exposure in epidemiological studies.
Mice are the animal species most commonly used for studies of physiology and disease formation, and several strains have been exceedingly well characterized biologically and genetically. Transgenic mouse strains are readily available for study of the influence of specific genes on radiation-induced adverse outcomes. Development of crosses between mouse strains that vary substantially in many aspects of disease formation also provides tools to discover the genes, molecular processes, and disease processes that may be influenced by exposure to low-dose and low-dose-rate radiation. The Collaborative Cross (Complex Trait Consortium, 2004), which combines the genomes of eight genetically and phenotypically diverse founder strains, has been created as a community resource to facilitate the genetic analysis of complex traits and may be particularly useful in dissecting the biology and genetics of low-dose and low-dose-rate radiation-induced adverse outcomes (Collaborative Cross Consortium, 2012).11 Genetic engineering tools such as CRISPR gene replacement (Doudna, 2020; see Section 5.4.1 for Priority I1) also will be useful in studies of the impact of defined human gene sequences. However, commonly used mouse strains are limited since they were derived from relatively few original sources, mostly at the beginning of the 20th century, so they do not represent the genetic diversity found in humans or even in wild-type mice. In addition, they may acquire new genetic variations over time that alter key aspects of radiation responses that are not typically found in mice or humans. A case in point is the C3H/HeJ mouse strain that derived from the C3H/HeN mice and developed a mutation in Toll-like receptor 4 (TLR-4); as a result, it only poorly responds to lipopolysaccharide challenge with suboptimal tumor necrosis factor-alpha production and as such might not be the preferred model to study radiation-induced danger signaling and inflammation. Similarly, using BALB/c mice for radiation mutagenesis might be misleading considering the DNA-dependent protein kinase catalytic subunit (DNA PKcs) deficiency and hence abnormal DNA repair efficacy of these mice. Comparisons of genotypes between mice and humans can reveal such phenomena.
The use of mice to study cancer endpoints is particularly well established. These studies can be conducted in the context of a well-developed
___________________
11 Andrew Wyrobek and Antoine Snijders, Lawrence Berkeley National Laboratory, presentation to the committee on January 24, 2022.
and expanding framework of the key biological processes that contribute to disease pathogenesis known as the Hallmarks of Cancer (Hanahan, 2022; Hanahan and Weinberg, 2011; Paunesku et al., 2021). This framework includes aspects of tumor cell survival and dissemination, vascularization, immune surveillance, and tumor-stromal interactions that are important in cancer genesis and progression. In addition, the National Cancer Institute’s (NCI’s) Mouse Models of Cancer Consortium and follow-on efforts have developed a wealth of genetically engineered mice for the study of processes leading to cancer. Many are available through the NCI Mouse Repository at the Frederick National Laboratory.12 NCI is currently developing a community resource of well-characterized patient-derived xenografts (Sun et al., 2021). Studies of low-dose and low-dose-rate radiation-induced cancers will benefit from these concepts and resources.
The above-mentioned and other developments in cell and animal models have led to a good but incomplete understanding of the mechanisms underlying radiation-induced cancer (see, e.g., UNSCEAR, 2021). In addition, most investigations so far have focused on the early stages of carcinogenesis, initiation in particular, and relatively little is known about the effects of radiation exposures on later stages of carcinogenesis. New experimental models including new model organisms apart from the mouse may be needed for these studies, possibly including pigs, dogs, and nonhuman primates—in compliance with legislation and guidelines governing biomedical research—because of the greater resemblance to human anatomy, immunology, and lifespan compared to small laboratory animals (Maynard et al., 2021). A full appreciation of radiation-induced disease formation, and therefore the experimental system to model that, will need to take into account both the genomic and epigenomic abnormality carried by the target cells (e.g., the breast epithelial cells that may develop to breast cancers) as well as the proximal and distal environments in which these cells exist.
Animal models also are being developed for the study of adverse health outcomes other than cancer that might be caused by low-dose and low-dose-rate radiation exposures. Specific disease models exist for cardiovascular disease progression (Jia et al., 2020; Liao et al., 2015) and neurocognitive deficits (Dawson et al., 2018) as examples. Humanized mouse models with bone marrow reconstitution using human hematopoietic stem cells (Ando et al., 2008) permit detailed investigation into lineage development in the context of external or internal exposures, but might not be an appropriate model for long-term studies. Finally, patient-derived single-cell models and construction of tissue array models can provide personalized determination of treatment options and responses to radiation, including risk assessment.
___________________
12 See https://frederick.cancer.gov/resources/repositories/nci-mouse-repository.
Mouse strains modeling these endpoints exist but have not been fully exploited. The adverse outcome pathway (AOP) framework (see, e.g., NCRP, 2020b; Svingen et al., 2021) is likely going to provide a means to integrate knowledge on the pathogenesis of radiation-associated non-cancer health effects. These and future models will facilitate studies of possible radiation-induced pathologies that so far have been only lightly explored.
It is increasingly recognized that cells function within a specific biological context and that they are strongly controlled by the signals they receive, as illustrated in Figure 5.1. The fact that these signals can come from cells that are in close proximity to the irradiated cell or from distal organs such as the brain only adds further complexity and underscores the need to study radiation responses in vivo as much as possible. Proximal interactions are often referred to as “bystander responses” and are well recognized by radiation biologists (Tomita and Maeda, 2015; UNSCEAR, 2021). Not only do these interactions determine how individual cells respond to radiation but they are also ultimately critical for the maintenance of tissue and organ viability. In other words, indirect effects of radiation on cells surrounding the cells of origin for radiation-associated cancers and non-cancerous pathologies could be as important as the direct radiation effects noted above in determining the outcomes of exposures and therefore need to be modeled accordingly. This is clearly demonstrated by pioneering studies that have shown that the incidence of cancers and by extension other diseases can be strongly influenced by the microenvironment(s) in which abnormal cells
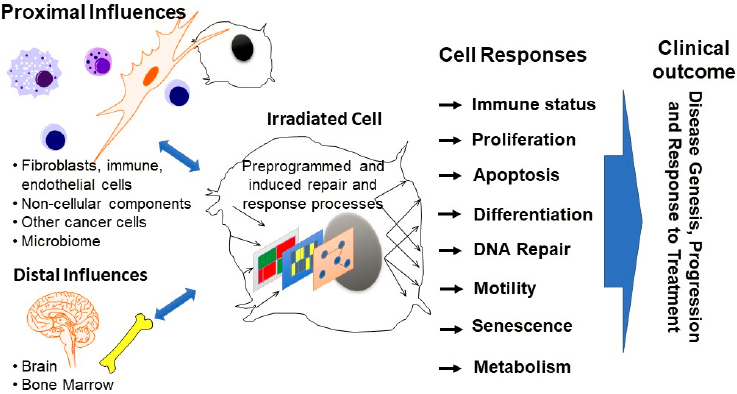
exist (see Bissell and Hines, 2011; Mintz and Illmensee, 1975; Nelson and Bissell, 2006; Radisky and Bissell, 2004). Indeed, cells that form cancers in one environment can behave normally in another (Mintz and Illmensee, 1975). Models genetically engineered to enable study of cellular interactions (e.g., by genetically labeling or functionally modifying cells of interest) will facilitate study of cellular interactions on low-dose and low-dose-rate radiation-induced adverse outcomes.
5.3.2 Develop Biomarkers for Radiation-Induced Adverse Health Outcomes (Priority B2)
Scientific and Decision-Making Value
The Food and Drug Administration’s Biomarkers, Endpoints, and other Tools (BEST) Resource glossary defines a biomarker as a “characteristic that is measured as an indicator of normal biological processes, pathogenic processes, or responses to an exposure or intervention, including therapeutic interventions.”13 The BEST glossary further defines biomarkers for susceptibility or risk, diagnosis, monitoring, prognosis, prediction of behavior, response to perturbation, and safety. Biomarkers for risk, diagnosis, and response will be particularly important for low-dose and low-dose-rate radiation research. Biomarkers of risk may identify individuals who are susceptible to radiation-induced adverse outcomes. Diagnostic biomarkers may identify adverse outcomes that are preferentially induced by low-dose and low-dose-rate radiation such that these can be assessed in epidemiological studies or that are associated with other etiologies so that cases with these biomarkers can be excluded. Biomarkers of response may identify cellular and molecular features or biological processes that change in response to low-dose and low-dose-rate radiation exposures. Importantly, all biomarkers can be measured with increasing precision, sensitivity, and reproducibility using new-generation analytical tools. The integration of biomarkers of radiation-induced and non-radiation-induced risk and response discovered in biological studies into epidemiological studies promises to increase the power to identify subtle effects and enable the exploration of factors that influence individual susceptibility. Measurement of response biomarkers will provide important insights about the mechanisms that are engaged as humans respond to low-dose and low-dose-rate radiation exposures.
___________________
13 See https://www.fda.gov/drugs/biomarker-qualification-program/about-biomarkers-and-qualification.
Current Status and Promising Research Directions
New and powerful analytical tools (see Section 5.4.1 for Priority I1) now enable precise and sensitive measurement of the cellular and molecular components and the organizations thereof (i.e., biomarkers) for normal, irradiated, and diseased tissues. This section provides examples of how biomarker analyses are already being deployed by the radiobiology research community and suggests areas where additional research is needed to inform on low-dose and low-dose-rate radiation effects with emphasis on response and diagnostic biomarkers. Risk biomarkers are discussed in Section 5.3.4 (Priority B4).
Regarding response biomarkers, measurements of changes in large-scale gene expression biomarkers after many different perturbations of biological systems have resulted in the definition of thousands of regulatory and functional networks and signaling pathways that enable cell and tissue function. The National Institutes of Health’s (NIH’s) Library of Integrated Network-Based Cellular Signatures has made important recent contributions in this area (Keenan et al., 2018). Many pathways and networks are now curated and publicly available. For example, the Molecular Signatures Database14 includes 50 “Hallmark Gene Sets” that represent well-defined biological states or processes.
Recent work has identified some of the major signaling pathways that are activated by different, mostly high doses of radiation (Brackmann et al., 2020; Mukherjee et al., 2019; Paul et al., 2019) and low-dose radiation-induced persistent transcriptome changes in genes associated with immune function (Snijders et al., 2012), but expansion of such studies and replication of results is essential for establishing robust biomarkers. Indeed, it remains to be determined whether activation of each response biomarker is associated with an increase or decrease in adverse health effects. In addition, it is already known that circulating small molecules such as miRNA can act as radiation-damage signaling molecules and that they tie in with health outcomes (Chakraborty et al., 2020; Soares et al., 2021). These and follow-on studies to elucidate the mechanisms engaged as cells and tissues respond to low-dose and low-dose-rate radiation are promising but will benefit from cohesive approaches similar to those deployed by The Cancer Genome Atlas (TCGA; Collins and Barker, 2007), Human BioMolecular Atlas Program (HuBMAP Consortium, 2019), and Human Tumor Atlas Network (HTAN; Rozenblatt-Rosen et al., 2020). The multidisciplinary low-dose and low-dose-rate radiation research program envisioned by this committee might directly engage with these programs. Importantly, these
___________________
studies will require increased access to low-dose and low-dose-rate exposure facilities as described in Section 5.4.4 (Priority I4).
In the context of cancer or non-cancer health outcomes, advanced -omics technologies include those with single-cell resolution capabilities, promise to reveal the relevant biomolecules, the pathways involved (including both DNA damage signaling and other signaling pathways), and the dose dependence of low-dose effects. Analytical technologies that efficiently assay multiple endpoints in single cells (see Section 5.4.1 for Priority I1) allow simultaneous assessment of genomic, epigenomic, and transcriptomic events in individual cells. This is especially useful at the low-dose radiation range because these technologies can identify the response of the small number of cells that stochastically incur damage as a result of exposures to low doses of radiation. These studies on individual cells could lead to development of an “atlas” of mechanistic changes that may enable more precise definition of disease subtypes that are more strongly associated with low-dose radiation exposure. Such approaches will also aid the identification of biomarkers that could be applied in population studies and for the development of AOPs.
The development of low-dose and low-dose-rate response biomarkers will benefit from analysis of biospecimens collected longitudinally. It is expected that the majority of studies that allow for longitudinal sample collection will be carried out using laboratory models (see Section 5.3.1 for Priority B1) exposed in facilities designed for low-dose and low-dose-rate exposures (see Section 5.4.4 for Priority I4); however, longitudinal studies in humans are also possible (Johnson et al., 2022). Both engineered tissues and animal models will be informative; however, animal models will enable studies of irradiated tissues as well as biosamples including blood (whole blood, serum, and plasma), urine, saliva, buccal cells, skin swabs, hair follicles, fecal material, and cerebrospinal fluid. All biomaterials can be preserved in appropriate materials (e.g., for stabilization of RNA or phosphoproteins) and can be analyzed for biomarker identification of disease progression in a retroactive manner or with targeted approaches for quantitation of biomarkers and changes associated with known disease progression. As an example, changes in circulating lipid composition are often associated with cardiovascular disease, and lipidomics could offer an informative collective profile of lipid classes in addition to particular species with high correlation to this outcome.
Diagnostic biomarkers are increasingly being identified and can subdivide anatomically defined diseases into subgroups that differ according to prognosis, etiology, anatomic origin, pathway usage, and response to therapy. Breast cancer, for example, can now be subdivided into at least five subgroups depending on the diagnostic biomarkers used (Ellis and Perou, 2013). The TCGA PanCancer effort has identified biomarkers that define
four cell-of-origin biomarker patterns that persist across 33 different types of cancer (Hoadley et al., 2018). Finally, genome sequencing studies have identified genomic signatures in cancers that suggest specific, non-radiation causative agents for individual tumors (Alexandrov et al., 2020). The observed differences in etiology between the subtypes raise the possibility that some cancer subtypes (and by extension other disease subtypes) could be more susceptible to induction by low-dose and low-dose-rate radiation than others. The observation by Ahadi et al. (2020) of cell-of-origin subtypes suggests that some will be more affected by internal radiation than others. Biomarkers are also being developed for other health endpoints that may be important in low-dose and low-dose-rate radiation research including cardiovascular diseases (Dhingra and Vasan, 2017), neurodegenerative diseases (Hansson, 2021), and aging (Ahadi et al., 2020).
The concept that disease subtypes are differentially susceptible to induction by low-dose and low-dose-rate radiation and mode of delivery can be tested for biological plausibility in laboratory models that accurately represent specific disease subtypes. Subtypes that appear most strongly influenced by aspects of low-dose and low-dose-rate radiation exposure can then be tested in humans in epidemiological studies (see Section 5.2.2 for Priority E2).
5.3.3 Define Health-Effect Dose-Response Relationships Around 10 mGy or 5 mGy/h (Priority B3)
Scientific and Decision-Making Value
Experimental studies at the molecular, cellular, tissue, and whole-organism levels can be used to examine the shape of the dose-response relationships for each of the relevant endpoints and inform the biological and physical factors that influence the response at doses nearing 10 mGy and 5 mGy/h (i.e., at the range where epidemiological studies may not be able to provide firm conclusions). Studies that integrate this information with that from epidemiological studies will increase understanding of the mechanisms involved and reduce the uncertainties of current risk estimates for low-dose and low-dose-rate exposures experienced by the U.S. population.
Current Status and Promising Research Directions
Radiation exposures around 10 mGy lead to molecular, cellular, and health outcomes that are not as well defined as those at higher doses, in particular cytotoxic doses. Advancements in assay technologies and in the ability to accurately measure these outcomes increase the ability to establish radiation dose-response curves for molecular and cellular endpoints and
for associated early- and late-stage diseases at different dose rates. Because these endpoints may differ from those related to radiation cytotoxicity, new models will have to be developed rather than relying for guidance on cytotoxic outcomes (see Section 5.3.1 for Priority B1) and will require increased access to low-dose and low-dose-rate exposure facilities (see Section 5.4.4 for Priority I4).
Direct induction of DNA damage and subsequent mutations by radiation are well studied, particularly in the case of DNA double strand breakage and the cellular response to it, sensing and repairing that damage. However, double strand breakage is rare at low doses and at low-dose rates for low linear energy transfer (LET) radiation15 but the repair may be not as efficient as at higher doses, and the responses in most cells will ultimately be determined by other forms of damage. While the importance of DNA damage and misrepair resulting in mutations in driving carcinogenesis is recognized, there remains little consensus on the existence of a distinct mutational signature of radiation exposure that would allow more confident attribution of an individual case to the exposure. However, there is growing evidence that genetic signatures can be defined that identify risk factors other than radiation as causative for individual cancers (Alexandrov et al., 2020), so these can be excluded from radiation epidemiological studies.
Similarly, there is an incomplete catalogue of the target genes for radiation carcinogenesis, the target cell populations for specific cancer types, and their radiation dose dependence. The impact of low-dose radiation exposures, and exposures to differing radiation qualities, on mutational loads in individuals is unknown but can now be assessed by modern high-throughput sequencing methods in human and animal model studies.16
Also relevant to low-dose radiation exposures is the role of transmissible genomic instability (i.e., the phenomenon of persistent elevation of mutation frequency in the descendants of irradiated cells). However, there are some indications that a threshold of exposure, around 100 mGy low-LET radiation, needs to be exceeded to trigger such instability (see UNSCEAR, 2021). The relevance and importance of “adaptive response,” whereby a low “priming” dose of radiation can induce DNA repair or immune response mechanisms that reduce quantitatively the outcome of a second higher-dose exposure, are equally undefined (see UNSCEAR, 2021). New tools for comprehensive analysis of DNA, modified DNA, RNA, and proteins in single-cell analysis tools (see Section 5.4.1 for Priority I1) will provide information about the mechanisms that operate following low-dose and low-dose-rate radiation exposures. Importantly, the damage caused by
___________________
15 Damage produced by high-LET radiation including from alpha particles from ingested radionuclides typically does cause double strand breaks (Stap et al., 2008).
16 Phil Jones, Sanger Institute, presentation to the committee on November 17, 2021.
radiation or other exposures is not necessarily immediately proximate in time to the disease-causing alteration, as illustrated by the human genetics of chromosomal or gene-specific diseases. Often the molecular incident cause is present long before the chromosomal damage, which poses an immense challenge for studies of disease etiology and pathogenesis, one that AOP approaches maybe be helpful in addressing.
The role of DNA damage and repair in the context of non-cancer health outcomes and the radiation doses at which this damage occurs is little understood. In cataract formation, for example, genes such as ATM, RAD9, and PTCH1 are known to modify the induction of lens opacities following radiation exposures, but these genes are generally not considered to be significant contributors to atherosclerotic disease.
There are alternatives to direct mutation as causative mechanisms for health effects, namely epigenetic modifications, which include DNA methylation which is generally associated with gene silencing; post-translational modifications (PTMs) of chromatin, and more than 200 known covalent modifications of the histone octamer by methylation, acetylation, phosphorylation, or ubiquitination, all associated with gene activation or silencing; chromatin remodeling that affects transcription factor accessibility and thereby gene expression; and structural changes including nucleosome replacement or higher-order nuclear topology controlling tissue-specific and tissue-appropriate gene expression (Feinberg et al., 2016; Jenuwein and Allis, 2001).
Epigenomic changes can be induced at a distance from radiation-damaged cells via chemical signaling from the damaged cells (Sprung et al., 2015). Epigenetic changes occur in many epithelial tissues including skin after aging and sun exposure in precisely those genomic regions with mutational errors in invasive squamous cell carcinoma (Vandiver et al., 2015), a concept likely extending to ionizing radiation exposure. Epigenetic changes also cause various other human diseases in addition to cancer such as protein aggregation diseases, metabolic diseases, neurological and psychiatric diseases, and imprinting disorders (Kungulovski and Jeltsch, 2016). Several recent studies support a direct link between low-dose radiation and epigenetic changes (reviewed in Lei et al., 2020; Leung et al., 2021; Miousse et al., 2019; Tharmalingam et al., 2017), and a comprehensive analysis in terms of the full range of epigenetic modifications and alterations to chromatin structure after low-dose exposures is clearly warranted. Understanding these genomic dynamics and their low-dose-response relationships as they relate to cancer and non-cancer disease pathologies will have a bearing on risk assessment. How much of low-dose effects are in fact mediated through reactive oxygen damage forms an intricate part of this equation and needs to be explored in detail. The modulation of reactive oxygen species is likely directly or indirectly related to changes in mitochondrial function that have
been observed after low-dose exposures (Shimura et al., 2016). Some studies suggest that reactive oxygen damage from low-dose and repeated low-dose exposures may even contribute to the proliferation of pre-cancerous cells in tissues (Fernandez-Antoran et al., 2019). Furthermore, the role of radiation damage to other organelles (excluding the cell nucleus) has received little attention, but this too may have an impact on health outcomes and follow different dose-response relationships (Paunesku et al., 2021). These biological endpoints are increasingly accessible for study using the analytical tools described in Section 5.4.1 (Priority I1).
5.3.4 Identify Factors That Modify or Confound Estimation of Risks for Radiation-Induced Adverse Health Outcomes (Priority B4)
Scientific and Decision-Making Value
Estimates of the risks of adverse health outcomes from low-dose and low-dose-rate radiation exposures may be modulated by events unique to an individual or confounded by exposure to factors other than low-dose and low-dose-rate radiation that produce the same adverse health outcomes. Modifiers that are identified in studies of laboratory model systems can be tested in epidemiological studies for their impact on risk estimation in human populations.
Current Status and Promising Research Directions
Studies of the responses of genetically and biologically diverse laboratory model systems (see Section 5.3.1 for Priority B1) to low-dose and low-dose-rate radiation and/or to chemical and environmental perturbagens will facilitate identification of risk modifiers and confounders. Numerous factors might be considered as risk modifiers by altering aspects of the processes by which radiation-induced DNA damage is translated into adverse health outcomes (e.g., DNA damage repair, damage surveillance, immune competence, stress response). These may vary between individuals or populations and may include functional genetic polymorphisms; epigenomic modifications that vary with age, sex, dietary intake, and environmental exposure and lifestyle; immune function including past “education”; and overall health status. Risk biomarkers for these events can be defined in laboratory model systems (see Section 5.3.1 for Priority B1) and then evaluated in epidemiological studies of U.S. populations (see Section 5.2.1 for Priority E1).
The immune system is the conduit where intracellular and intercellular responses merge and where local responses have systemic reach. Immune system alterations following exposure to high doses of radiation are well
established in that radiation damage gets sensed and interpreted through common pattern recognition receptors that signal danger from damaged “self” (Harding et al., 2017; Härtlova et al., 2015; Mackenzie et al., 2016). This response feeds into evolutionary conserved innate immune cell activation/inflammatory pathways (e.g., NFκB and type-I interferons) that potentially bridge to the adaptive arm of the immune system through dendritic cell cross-priming and T-cell activation. Redox reactions play a significant role in this, derived in part from these inflammatory cascades involving reactive oxygen species (ROS)-generating reactions (e.g., inducible nitric oxide synthase or nicotinamide adenine dinucleotide phosphate oxidase) which are very similar to the initial radiation-induced ROS. Traditionally, this relationship has been thought to be dose dependent with low doses being considered anti-inflammatory in certain disease states but not others. This concept needs to be reexamined under well-defined conditions and over a wide range of doses using modern technologies.
Because defects in danger signaling are known to contribute to a host of human diseases including cardiovascular, autoimmune, and cancerous, it is conceivable that low-dose radiation-induced danger signals could affect disease initiation and/or progression and therefore health outcomes, especially if poorly controlled. In fact, there is already evidence from high-dose exposures linking persistent and/or unrepaired DNA damage to tissue senescence and sustained inflammatory infiltration in vivo (Rodier et al., 2009). Focused studies could determine the low-dose radiation response to danger signaling and the sensitivity of the different mechanisms for immune sensing of danger and tissue damage and how these affect immune balance and outcome in the short and long terms. For instance, redox status sensing through Nrf-2 reportedly acts at doses as low as 20 mGy while micronuclei (i.e., DNA damage) remain unrepaired, highlighting the possibility of different dose responses for different mechanisms (Rodrigues-Moreira et al., 2017; Rothkamm and Löbrich, 2003). Similarly, the engagement of central-systemic feedback loops, especially hematopoietic imbalances or amplification of signals in the hypothalamus, which are known in the context of higher radiation doses, can be evaluated; this will be highly relevant for organ interaction, amplification, morbidity, and mortality. Evaluating the effect of low-dose radiation on tumor incidence and the immune reactions that are generated in susceptible and resistant animal models will be important as will be the role of immunogenetics, including major histocompatibility pathways, and how they factor into radiation sensitivity and immune activation post-exposure.
There is still much to understand about the mechanistic basis for the differences in radiosensitivity in various tissues, though differences in DNA damage response are likely to contribute given their importance to cancer induction. Differences between tissues are primarily due to epigenomic
changes that occur during development. However, they also may be influenced by lifestyle and other factors. Additionally, the number of cells at risk for transformation into cancer cells is likely to be important. The target cells and tissues for radiation-associated non-cancer outcomes are not well characterized, and information on these and their responses to radiation insult will be important in making judgments on low-dose risk extrapolation. For circulatory diseases and cognitive dysfunction, inflammation is likely going to play a key role. Hence, identifying and characterizing the target cells and tissues involved will be crucial in developing appropriate risk models. Overall, a better understanding of the pathogenesis of radiation diseases, especially non-cancer outcomes, at all organizational levels is required.
These processes can now be studied in laboratory models using the suite of -omics and multiscale molecular imaging tools that are described in Section 5.4.1 (Priority I1). Moreover, these tools can be integrated with similar data on normal and diseased tissues that are available in public databases emerging from NIH and international programs such as TCGA, the International Cancer Genome Consortium, HuBMAP, and NCI’s HTAN to facilitate identification of precise disease signatures that can be tested for association with low-dose radiation exposure. The application of these and other methods to further characterize mechanisms of low-dose and low-dose-rate health effects combined with the AOP approach to integrate knowledge provides a route to develop a framework on the critical steps in the pathogenesis of radiation health outcomes that can inform risk assessment for all health outcomes. Even for radiation carcinogenesis where the mechanisms are relatively well studied, many questions remain as to the modifiers of risk such as comorbidities, underlying genetic variants, and exposures to other agents. Collectively, these efforts can be geared toward assessing the impact of genetic makeup, epigenomic status, DNA repair efficacy, comorbidities, history of exposure to radiation and other agents, lifestyle factors, and immune status on radiation response outcomes and biomarkers for ensuing health effects.
If unique biomarkers for low-dose and low-dose-rate radiation-induced adverse health effects are defined, they can be compared to unique biomarkers for adverse health effects produced by causes other than radiation. These can be identified by treating laboratory models with agents known from the literature or public-domain genetic or chemical perturbation databases to produce adverse health outcomes or biomarker responses similar to those produced by low-dose and low-dose-rate radiation. If a biomarker is not unique to radiation, then agents that induce the same diagnostic biomarkers as low-dose and low-dose-rate radiation are potential confounders of the radiation-biomarker association in observational epidemiological studies (see Section 5.2.2 for Priority E2). However, potential confounders may be
eliminated if they manifest cellular or molecular biomarkers that identify adverse health effects that are associated with etiologies other than radiation (e.g., genomic signatures in cancers that suggest tobacco use, defective DNA mismatch repair, ultraviolet light exposure, or aflatoxin exposure as causative events; see Alexandrov et al., 2020).
5.4 RESEARCH INFRASTRUCTURE PRIORITIES
The low-dose radiation research program can take advantage of the remarkable advances that have been made in the past two decades in understanding the behavior of complex biological systems and disease phenotyping that may improve the sensitivity of future epidemiological and experimental studies. These advances have been powered by the health research programs supported by NIH and by advances in measurement, data science, and computer technology supported by the Department of Energy (DOE), NIH, and the National Science Foundation.
5.4.1 Tools for Sensitive Detection and Precise Characterization of Aberrant Cell and Tissue States (Priority I1)
Scientific and Decision-Making Value
Major advances in measurement, data science, and computer technology over the past 20 years have resulted in a wealth of tools that can be deployed to report on the molecular compositions and multiscale molecular and physical structures that comprise normal and diseased cells and tissues. These tools can be readily applied to reveal the responses to perturbations including those induced by low-dose and low-dose-rate radiation. These revolutionary tools, in aggregate, allow quantification of subtle cellular and molecular processes important in radiation research that have previously not been possible, including changes in single cells as a result of the interactions of those cells with radiation.
Current Status and Promising Research Directions
-Omics analysis tools enable assessment of the molecular components that comprise cells and tissues. Work in this area was initiated by the development of robust, fast, and low-cost nucleic acid sequencing tools to support the Human Genome Program co-led by DOE and NIH. Development of these tools was further stimulated by TCGA (Collins and Barker, 2007) in the United States and by the International Cancer Genome Consortium (ICGC, 2010) worldwide. As a result, robust tools are available in many research and clinical laboratories in the United States that
can quickly generate sequences for entire genomes and transcriptomes for less than $1,000.17 Work in this area continues today, driven by NIH and DOE programs and substantial U.S. industry investments. Long-read-length sequencing technologies are emerging that may enable identification of genomic abnormalities in otherwise difficult-to-sequence regions of the genome (Nurk et al., 2022) and make it possible to assess alternative splicing solutions. These tools have been enhanced by the development of experimental and computational workflows that allow analysis of DNA and RNA in archived tissues and by the development of easily accessible reference databases, against which test sequences can be compared in efforts to identify differences between normal and diseased cell populations and between individuals and to identify contaminating microbial species and/or changes that result from external perturbations including exposure to ionizing radiation.
Nucleic acid sequencing tools have been further enhanced by the development of experimental and computational workflows that enable assessment of aspects of the spatial organization of chromatin within cells and/or epigenomic DNA modifications that influence gene expression and cellular function. The epigenome comprises several types of chemical or conformational modifications to DNA and the associated proteins. These include DNA methylation (i.e., the addition of a methyl group to cytosine, generally at CpG dinucleotides); PTM of nucleosomes, including methylation, acetylation, phosphorylation, and ubiquitinylation; chromatin compaction; and higher-order chromosomal folding. In addition, long-read sequencing can allow indirect measurement of chromatin compaction and even higher-order folding.18 The NIH Roadmap Epigenomics Mapping Consortium has been instrumental in advancing this area (Bernstein et al., 2010).
The successful development of genome analysis tools inspired the development of tools to study other cellular components including new and more efficient mass spectrometry–based approaches for analysis of proteins, metabolites, cellular carbohydrates, and lipids (Aardema and MacGregor, 2002). Protein analysis has progressed to include protein modifications beyond phosphorylation, with fine mapping of glycans (Y. Huang et al., 2021). Metabolites are now considered an integral part of human physiological status and progression of disease, with many acting as signaling
___________________
17 See https://www.genome.gov/about-genomics/fact-sheets/Sequencing-Human-Genomecost.
18 A recent multi-investigator study examined the three most popular protocols for whole-genome methylation sequencing analysis, all of which involve bisulfite conversion, which is selective for unmethylated cytosine, as well as enzymatic deamination, targeted methylation capture, long-read sequencing, and array-based methods. This study showed high concordance between assays and specific advantages and disadvantages of the individual methods that can be used to select assay designs for a given study (Foox et al., 2021).
molecules and contributing to a pro-inflammatory status. Lipids, an integral part of metabolism, are shown to have a high degree of structural complexity, particularly in the context of increased oxidative stress, with contribution to progression of adverse health effects (Natesan and Kim, 2021). Many aspects of mass spectrometry–based analysis are being accelerated by NIH’s Clinical Proteomic Tumor Analysis Consortium (Ellis et al., 2013).
Early-generation -omics analysis tools enabled analysis of materials isolated from collections of cells or tissues. However, tools for analyses of single cells or parts thereof are also increasingly available. These began with the development of flow cytometry and sorting for high-speed cell analysis and purification (Herzenberg et al., 1976), including at Los Alamos National Laboratory (Fulwyler, 1965; Van Dilla et al., 1969) and Lawrence Livermore National Laboratory (Gray et al., 1987). However, recent developments based on nucleic acid barcoding allow efficient analysis of DNA, DNA modifications, and RNA in isolated single cells (Quake, 2022) and in situ (Moses and Pachter, 2022). Others enable single-cell mass spectrometry (Lanekoff et al., 2022).
These techniques, when applied to tissues exposed to low-dose radiation or to diseased tissues from individuals in exposed populations, will provide fine phenotyping of cell populations (e.g., immune system); tissue-specific responses and microenvironment interactions including with microbiomes; and identification of small molecules (e.g., metabolites, lipids) that have the potential to act as signaling molecules and transcription factors or induce epigenetic changes. New methods such as Repair-seq can map the genetic dependencies of DNA repair outcomes (Hussmann et al., 2021).19
Applications of -omics technologies are already being employed in space biology research, cancer research, and neuroscience, to name a few. Given that perturbations with low-dose exposures may not be quantifiable at a whole-tissue level with multiple cell populations due to a low signal, current knowledge is lacking in understanding how single changes may lead to long-term health effects. Such technological advances can provide important information on initiation and advancement in a range of low-dose radiation-associated health effects other than cancer.
Imaging analyses complement -omics analyses by providing information about the hierarchical organizations of the molecules, organelles, cells, and tissues; the functional consequences of interactions between these entities; and how these organized entities respond to perturbations including radiation. Work in imaging and -omics is currently being accelerated by the HuBMAP Consortium (2019) and HTAN (Rozenblatt-Rosen et al., 2020), which aim to develop comprehensive -omics and image-based atlases of
___________________
19 Dale Ramsden, University of North Carolina at Chapel Hill, presentation to the committee on November 16, 2021.
normal and cancerous tissues. Similarly, NIH’s Brain Research Through Advancing Innovative Neurotechnologies® (BRAIN)20 initiative, which aims to revolutionize understanding of the human brain, also contributes to accelerating work in imaging and -omics. In medicine, radiomics use data-characterization algorithms to extract features from medical images beyond what can be observed by radiologists and can improve disease classification and predict clinical outcome among other benefits (Mukherjee et al., 2020).
Recent progress in noninvasive, anatomic imaging now enables more precise definition of the extent of disease as well as assessment of the molecular state of disease and quantification of variations therein. This capability comes from advances in imaging technology, reporter chemistries, and image visualization and analysis tools. Areas of advance include (a) easy-to-use 3D ultrasound imaging for quantification of flow, fibrosis/stiffness, and other molecular features revealed by injected microbubbles coupled to affinity ligands (Ajmal, 2021); (b) improved computed tomography (CT) and micro CT imaging at lower radiation dose based on photon counting and spectral analysis for assessment of tissue composition, and precise mapping of contrast reagents (Garnett, 2020); (c) faster magnetic resonance imaging (MRI) resulting from high-density receivers, highly undersampled acquisitions and faster scan times, and improved image reconstruction and visualization (Kataoka et al., 2022); (d) faster positron emission tomography (PET) imaging informed by radiotracers engineered to report on molecular or cellular status (e.g., metabolic activity, fibrosis, DNA synthesis, apoptosis, drug concentration, and inflammation; see Choudhury and Gupta, 2017); and (e) optical coherence tomography and diffuse optical spectroscopy for imaging of eyes, skin, bone, brain, breast, and other accessible tissues (Wilson et al., 2016). Integration of images acquired from different imaging modalities (e.g., ultrasound/CT, PET/CT, and PET/MRI) further increases the information that can be obtained. Reconstruction and characterization of image features from all modalities have benefited substantially from recent developments in artificial intelligence and machine learning that are made possible by new-generation computational technologies.
More precise disease phenotypes (e.g., in cancer, cardiovascular disease, cataracts, and dementias) revealed by these technologies may increase the precision of epidemiological studies of the effects the low-dose and low-dose-rate radiation. Their increasing availability in medical centers throughout the United States, increasing safety, and decreasing cost make it reasonable to consider deploying them in future radiation health effects studies. These technologies are being applied to improve disease detection and treatment so that overall mortality resulting from radiation exposures may be decreased.
___________________
Multi-omics image analyses of disease biopsies (and normal tissue) can provide information on cellular and subcellular proteins, cell distributions, and interactions between them (Lun and Bodenmiller, 2020; Schapiro et al., 2022). Multi-round immunostaining and imaging technologies allow expression of many different proteins to be measured in the same tissue section, thereby allowing assessment of cellular composition, interactions between cells, assessment of signal transduction cascades, and detailed immunophenotyping (Black et al., 2021; Lin et al., 2015). Similar multi-protein analysis can be accomplished using multiplexed ion beam imaging (Coskun et al., 2021) and spatially defined mass spectrometry–based techniques (Gessel et al., 2014), with applications in spatial distribution of lipids and metabolites. Metabolic imaging can also be achieved with methods such as fluorescence lifetime imaging microscopy (Datta et al., 2020) to characterize metabolic states and involvement of specific processes such as in mitochondria (glycolysis or oxidative phosphorylation). Spatial genomics and transcriptomics technologies and interactions are particularly well suited to study clonal heterogeneity in tissues (Marx, 2021; Zhao et al., 2022).
The information these tools provide on the cellular compositions of normal and diseased tissues, on functional states, and on the functional consequences of interactions between cells may be used to define more precise disease phenotypes in epidemiological studies and to elucidate the cellular and molecular mechanisms that are influenced by low-dose and low-dose-rate radiation. The single-cell capabilities seem particularly well suited to study the stochastic interactions of low-dose radiation with the cells and components that comprise tissues.
Advances in fluorescence, X-ray, and electron microscopy, chemistry, and computation have been combined to allow imaging of the organization and dynamics of single molecules, protein complexes, and organelles (Liu et al., 2015). Innovations in fluorescence microscopy and fluorescence labeling chemistries include super-resolution fluorescence microscopy for single-molecule localization and tracking and fluorescence correlation spectroscopy and single-molecule fluorescence resonance energy transfer for assessment of molecular movements and interactions (Nickerson et al., 2014). In addition, novel sample preparation techniques, such as tissue clearing, and preparation techniques that expand the biospecimen itself coupled with large-format imaging tools, such as light sheet microscopies, facilitate rapid 3D imaging of extended cellular volumes (Zhang et al., 2017). New X-ray microscopes allow 3D imaging of structures as small as 20 nanometers (nm), while scanning electron microscopes coupled with serial block-face sectioning or focused ion beam sectioning allow 3D imaging of structures as small as 4 nm (Riesterer et al., 2020). The development of cryoelectron tomography offers the possibility of subnanometer imaging of molecular complexes in single cells (Chua et al., 2022). Additional innovations in microscopy, chemistry, and image integration allow specimens to be moved
between imaging platforms, thereby enabling multimodal imaging (e.g., correlative light and electron microscopy; López et al., 2017).
These tools, when applied in radiation biology, will allow direct study of the structures and organizations of the proteins, protein complexes, and organelles that are directly and indirectly affected by ionizing radiation. These include DNA repair complexes in nuclei and nucleoli; organelles such as mitochondria, macropinosomes, and lamellipodia that may be involved in cell death responses; and filopodia-like protrusions that mediate intercellular interactions and motility (Johnson et al., 2022).
Revolutionary advances in single-particle cryogenic electron microscopy (cryo-EM) are now being made through NIH-supported cryo-EM centers that enable atomic resolution measurements of single-molecule and protein-complex structures that do not readily form crystals.21 In addition, DOE supports next-generation synchrotrons (e.g., bright, storage ring, and X-ray free-electron lasers),22 which can enhance studies of the dynamics of protein interaction and responses to perturbations as well as intracellular structures. Development of these technologies will support fundamental studies of the molecular responses to radiation. These technologies could be deployed in future research into the biophysical effects of cell interactions with single photons or ions and direct measurements of the damage to specific cellular components including DNA, associated repair complexes, mitochondria, membranes, and other features of cells that may be influenced by the passage of ionizing radiation. Such information may be useful in the development of more precise multiscale dosimetry models.
The Internet of Things refers to devices equipped with sensors that connect and exchange data over the internet or other communications networks. IoMT refers to devices that inform all aspects of human health or performance or exposure (Dwivedi et al., 2022). IoMT devices may include smart phones, in-home monitors, implantable devices, and wearable devices that allow continuous assessment and reporting of a wide range of physiological and psychological endpoints or exposures in humans (Greco et al., 2020). The growing list of accessible endpoints includes weight, gait and balance, voice pathology, heart function, temperature, glucose and other aspects of blood chemistry, cognitive function, eye movement, food and drug consumption, disease detection, geographic location, and exposure to environmental agents.
Deployment of IoMT devices for low-dose and low-dose-rate radiation studies offers the possibility of accurate assessments of individual exposures to radiation and physiological changes that may be associated with such exposures. Wearable dosimeters may continuously report exposures of radiation from environmental or medical procedures. In-home and geographically
___________________
21 See https://www.cryoemcenters.org/cryoem-centers.
22 See https://science.osti.gov/User-Facilities/User-Facilities-at-a-Glance/BES/X-Ray-Light-Sources.
dispersed environmental sensors may also improve estimates of exposures to radioactive materials. Wearable monitors of aspects of human physiology and cognitive function that report continuously may allow more accurate assessment of changes in health status that may be associated with radiation exposures. Deployment of IoMT devices already exists in some occupational settings (where personal electronic dosimeters are used) and can be considered for future-generation epidemiological studies aimed at improving estimates for risk of exposure to low-dose and low-dose-rate radiation.
Advances in -omics, image analysis, and distributed sensor technologies can now provide measurements of the molecular and cellular compositions, organizations, and static and dynamic interactions between them in normal and diseased tissues both prior to and as they respond to more precisely known levels of radiation and other perturbagens. Computational tools are now being developed with support from NIH and DOE that interpret these data to reveal how biological systems function and make the data and analytical tools available to the scientific community. Machine learning tools reveal networks that regulate behavior and discover differences between normal and diseased tissues that may be causally related to disease genesis and progression. Dynamic modeling tools describe how complex systems respond in the short and long terms to perturbations and, more importantly, predict how they will respond to perturbations including radiation.
Several computational concepts implemented on increasingly powerful computational platforms are used to integrate -omics, images, and biological phenotypes and clinical data (Goecks et al., 2020) in ways that identify regulatory mechanisms that may control biological and clinical phenotypes and/or identify molecular signatures and phenotypes that are associated with perturbations. These concepts include (1) dimensionality reduction to select the most important features in integrated datasets, (2) Bayesian learning methods that include prior knowledge during learning, (3) supervised and unsupervised classification and regression methods that organize health effects into discrete categories (e.g., disease subtypes, genomic or epigenomic23 states, and/or functional molecular networks) and that identify associations
___________________
23 One of the most exciting developments in both epigenome analysis methods and their computational interpretation is the development of single-cell -omics methods that can measure, most typically, pairwise combinations of DNA methylation, chromatin modifications, and gene expression but have been extended to chromatin structure as well. These measurements are more limited in the genome-scale comprehensiveness of a given analysis, compared to bulk (non-single-cell measures), but they open the door to advanced methods from statistical mechanics, including entropy, stochastic processes, and critical phenomena, which allow for time-dependent modeling, epigenomic landscape analysis, and, most importantly, bottom-up modeling—namely, the ability to predict and model the underlying molecular network state using measurements taken only from within that cell (Teschendorff and Feinberg, 2021). Such approaches are particularly important in the study of low-dose radiation effects since their cellular effects are comparatively low frequency and stochastic but will impact multiple molecules within the radiation-impacted cells.
of these with response to perturbations, (4) deep learning methods that use multilayer neural networks to identify complex relationships in complex datasets (typically without providing information about the mechanisms that drive the relationships), and (5) ensemble learning wherein many models of associations are developed and averaged to produce predictions. These machine learning tools applied to measurements of cells and tissues following exposure to low-dose and low-dose-rate radiation will allow identification of the features and regulatory mechanisms that are influenced by radiation as well as precise health effects that result from exposures. Tools that link features and mechanisms to health effect phenotypes will suggest potentially causal relationships that can be tested in experimental systems.
Dynamic models are central to understanding the behavior of complex biological systems including responses to perturbation. Elucidation of the emergent properties of the system is an important goal of many such models. Models have been developed that describe chemical reactions, protein folding, cell phenotypes, immune function, interactions of cells that comprise complex tissues, and interactions between organs and between individual organisms and their environments. The model used depends on scale and complexity of the system and on the level of understanding of the processes that influence the behavior of the system. Commonly used model types include agent-based models (ABMs; Metzcar et al., 2019), differential equations (Chaves and de Jong, 2021), and molecular dynamic (MD) models (Ingólfsson et al., 2022).
ABMs typically consist of computational objects situated in space and time, called agents, that represent biological objects of interest (e.g., the diverse organelles, cell types, mechanical structures, and/or organs that comprise irradiated systems). Rules are defined that describe how each agent sends and receives signals and how it acts based on received signals (e.g., change in state, movement, growth, death). These rules are based on biological understanding or hypotheses, can be as simple or complex as biological understanding allows, and can be different in scale and type (e.g., cell phenotype, molecular activity, and organs) as long as rules can be defined that link them. Such models are well suited for the exploration of how cells and tissues respond to radiation-induced changes in the signals encoded in the rules. The number of agents and complexity of rules can be as large as computational capability allows. ABMs are already used to describe the responses of complex biological systems to ionizing radiation.24
Partial differential equation (PDE) models calculate the concentrations and/or the velocities of the components comprising a system. In general, the components in PDE models are similarly scaled (e.g., describing interacting
___________________
24 Sylvain Costes, National Aeronautics and Space Administration, presentation to the committee on September 24, 2021.
chemicals or components of the immune system). The number of components of PDE models can become large, but modern computers make managing millions of equations tractable. However, development of models that integrate across biological and molecular systems is difficult. PDE models may be useful in understanding the behavior of regulatory networks that are influenced by radiation.
MD models predict how every atom in a protein or other molecular system will move over time, based on physical laws governing inter-atomic interactions. The utility of MD simulations is increasing as better structures of proteins and protein complexes are determined (e.g., using single-particle cryo-EM or next-generation synchrotrons) and as computing power increases to allow simulation of biological processes at relevant timescales. MD models may be particularly useful in exploring radiation–matter interactions but are computationally demanding. The high-performance computers available in DOE’s national laboratories are well suited to MD simulations; the exoscale systems now being deployed at Argonne National Laboratory and Oak Ridge National Laboratory and the powerful cloud computing platforms that are now available from public domain providers are particularly noteworthy.
Major advances in computing technologies and architectures have powered many advances in biomedical analysis technologies—for example, by providing the computational power for massively parallel sequencing, multiplex imaging, MD simulations, artificial intelligence, cryo-EM, secure federated computing, and more.
5.4.2 Harmonized Databases to Support Biological and Epidemiological Studies (Priority I2)
Scientific or Decision-Making Value
Funding agencies and publishers of scientific articles increasingly require plans for data management and data sharing for research they support or publish. Several frameworks for data management and stewardship have been published; for health sciences, the most cited are the FAIR (findable, accessible, interoperable, and reusable) principles (Wilkinson et al., 2016). Despite major advances in open and free exchange of data in many fields of science, transparency and application of the FAIR principles in human studies of environmental health issues remain highly controversial.
Current Status and Promising Research Directions
The Human Genome Project and follow-on cancer analysis programs stimulated the development of databases of healthy individuals against
which new populations could be conveniently compared. A key feature of these reference databases is their public accessibility and convenience of use. Examples of commonly used resources include GenBank, an NIH genetic sequence database of annotated collections of publicly available DNA sequences;25 NCI’s Genomic Data Commons, a unified repository and cancer knowledge base that enables data sharing across cancer genomic studies in support of precision medicine;26 the University of California, Santa Cruz’s Genome Browser, which describes the sequences, genes, and other components of the normal genome;27 CBioPortal for Cancer Genomics, which provides visualization, analysis, and download of large-scale cancer genomic datasets;28 the ENCODE portal, which informs on functional elements in the human genomes;29 the Genome Aggregation Database (gnomAD), which aggregates exome and genome sequencing data from various large-scale sequencing projects;30 the Catalog of Somatic Mutations in Cancer, which helps explore the impact of somatic mutations in human cancer;31 and the Trans-Omics for Precision Medicine (TOPMed) program, which provides disease treatments tailored to an individual’s unique genes and environment.32 Recently, work during the COVID-19 pandemic demonstrated the feasibility of making EMRs computationally accessible to identify higher-risk populations and for other purposes, while maintaining the required confidentiality. In addition, a growing number of geospatial databases are becoming available that inform on aspects of the environment, health care, economic status, social status, transportation, and other factors, which may reveal confounding events when included in next-generation epidemiological studies. Importantly, NIH, the National Aeronautics and Space Administration, and other federal agencies are increasingly committed to the development of databases in which information is available under FAIR principles (Wilkinson et al., 2016). The availability of data under FAIR principles already has stimulated the development of thousands of computational tools that access these reference databases. These tools can be aggregated, as in done in Galaxy, to support accessible, reproducible, and transparent computational research.33 These and similar reference databases will be valuable in low-dose radiation research—for example, by serving as references against which radiation-perturbed normal
___________________
25 See https://www.ncbi.nlm.nih.gov/genbank.
26 See https://gdc.cancer.gov.
27 See https://genome.ucsc.edu.
28 See https://www.cbioportal.org.
29 See https://www.encodeproject.org.
30 See https://gnomad.broadinstitute.org.
31 See https://cancer.sanger.ac.uk/cosmic.
and diseased tissues can be compared in order to more accurately define aspects of biology that are influenced by exposure to radiation, documenting the levels of environmental exposures experienced by individuals in U.S. populations, and improving understanding of the phenotypes of diseases that may be caused by radiation exposures.
5.4.3 Dosimetry for Low-Dose and Low-Dose-Rate Exposures (Priority I3)
Scientific or Decision-Making Value
Understanding the effects of exposures to low-dose and low-dose-rate radiation depends on accurate information about the levels of such exposures and consideration of radiation type, mode of exposure, and individual characteristics. Recent developments in several aspects of radiation dosimetry suggest that future low-dose and low-dose-rate radiation studies can benefit from improved personal dosimeters, computational phantoms, biokinetic and source-term models, and tools for environmental radiation exposure. In parallel with these developments for radiation dosimetry, modern statistical and computational methods for dose reconstruction are needed to fully integrate detailed dosimetry data into modern analyses of epidemiological studies.
Current Status and Promising Research Directions
Accurate measurements of cell-, tissue-, and organ-specific radiation exposures experienced by medically and occupationally exposed individuals will improve understanding of the health effects that result from those exposures. Recent work in harmonization efforts demonstrates high variability in reported monitoring results (Fantuzzi et al., 2014; Mayer et al., 2021; Stadtmann et al., 2018), thereby necessitating investigation of the effects of calibration bias in dosimetry monitoring systems and periodic harmonization intercomparison studies. Further harmonization regarding reconstruction from reported exposure quantities spanning generational application of ICRP Publications 26, 60, and 103 in reconstruction of fundamental organ dosimetry further mobilizes existing data sources.34 Use of IoMT wearable dosimeters may facilitate harmonization for occupationally exposed individuals. Inclusion of detailed information in EMRs of lifetime organ-specific radiation doses from medical diagnostic procedures will enable more accurate assessment of such exposures and support epidemiological studies of the adverse health effects that result from such exposures.
___________________
This will be an important undertaking given that exposure from medical diagnostic procedures is the biggest human-made radiation source but will require substantial changes in medical practice and recordkeeping and good records of the doses delivered and the reasons for exposure.
In the case of neutron dosimetry, recent international harmonization intercomparisons demonstrated greater variability compared to photon-based systems (Fantuzzi et al., 2014; Mayer et al., 2021; Stadtmann et al., 2018). This variability was attributed to the breadth of energies from source neutron fields and the high variability in the neutron dose coefficients at low energies (10 kilo-electronvolts [keV] to 100 keV). Neutron dosimeters (e.g., thermoluminescence albedo dosimeters, etched-track detectors, superheated emulsions, and direct ion storage chambers) have inherent limitations, with ongoing challenges in determination of directional response, making dose estimation and reconstruction a continuing challenge (Stricklin et al., 2021). Improving characterization of source radiation fields with directional dependence will improve occupational neutron dosimetry and enable more accurate assessment of organ-specific doses. Furthermore, knowledge of worker activities (source exposure based on radiation work permit, posture, and duration) informed by IoMT wearable detectors coupled with innovation in rapid Monte Carlo radiation transport modeling can provide near-real-time organ dose estimation to accompany typical reported values of effective dose (lacking organ-specific information). The quantitative information from these health and exposure monitoring devices, coupled with health information from EMRs, can provide increasingly accurate health and exposure metrics for next-generation studies of the health effects of low-dose and low-dose-rate radiation, assuming that personal privacy issues can be properly managed.
Improved computational tools and Monte Carlo radiation transport modeling capabilities are now emerging that can be leveraged and further developed to allow integrated, multiscale dosimetry from the track structure to the organ and organ-system levels. Anthropomorphic phantom models were originally rooted in the “Reference Man” paradigm, which evolved to an age-specific stylized (mathematical) model employed in the MIRD Schema35 and CT-based voxel phantoms adopted by ICRP (2009, 2016a,b), which corrects for sex- and age-specific (newborn, 1-year-old, 5-year-old, 10-year-old, 15-year-old, and 20- to 25-year-old) differences. Currently, ICRP has improved upon limitations of organ contouring and has improved resolution (e.g., thin-walled organs, expanded definition of organ targets) of adult male and female hybrid mesh-type phantoms (ICRP, 2020). The mesh phantoms have the added functionality of being deformable, permitting
___________________
35 Developed by the Medical Internal Radiation Dose (MIRD) Committee of the Society of Nuclear Medicine.
the creation of expanded morphometry (beyond reference), in addition to articulated postures (ICRP, 2020; Yeom et al., 2019, 2021).
These phantoms can be further developed for use in estimation of organ doses for prospective and retrospective studies of low-dose and low-dose-rate radiation effects beyond reference models based on 50th percentile Western European Caucasian anatomical parameters (ICRP, 1975). This includes development of phantoms representative of non-reference ages beyond the six reference ages described above, as well as representative of cohort demographics, as has been conducted with the Japanese phantom-based models of the J45 phantoms (Sato et al., 2020). Underrepresented cohorts currently not adopted by ICRP models, notably pregnant women of various gestational periods, need to be adopted to accurately assess organ doses for specific populations (Makkia et al., 2019; Maynard et al., 2011, 2014, 2015a,b; Petoussi-Henss, 2021). Improved computational capabilities permit the implementation of these enhanced mesh-type phantom models. Although the mesh-type phantoms represent anatomic fidelity in reference models, expansion to include realistic distribution of anatomical parameters, for example, adipose tissue in addition to subcutaneous, will more accurately model individuals in non-reference populations.
Phantom models can be further improved by taking advantage of innovations in targeted radionuclide therapies in nuclear medicine such as targeted alpha therapies. These include development of 3D models of tissue microstructure (e.g., definition of kidney model at the nephron level) to model internal radionuclide deposition and archived samples to determine 3D spatial distribution of deposition.36 At the whole-organ level, models of both intraorgan and interorgan blood vasculature used to differentiate radionuclide decays in organ parenchyma from radionuclide decays in organ blood content can further inform the dose distribution in the human body using phantom models to estimate radiation dose, bridging organ-level with organ microstructure dosimetry for low-dose radiation exposures and uptakes.
Computational dosimetry based on anatomically enhanced phantom models to permit individual or subcohort dose assessment now appears feasible. However, widespread use will require enhancements in commonly used Monte Carlo radiation transport codes to accommodate mesh-type phantoms and proposed multiscale anatomical enhancements without degraded computational performance. Radiation transport, especially at low energies, must be enhanced through appropriate variance reduction techniques or establishment of justified low-energy cutoffs for particle transport to maintain reasonable computational processing times, with challenges
___________________
mostly lying with radiation transport of changed particles, whether secondary from indirectly ionizing radiation or primary.
While dosimetric models for internal emitters depend primarily on the emission properties of radionuclides, biokinetic models depend on radionuclide inventory (and progeny), chemical form (e.g., solubility, transformed states in the body), physical form (e.g., particle size), route of intake, and age of intake. Existing models are well described in national and international consensus-based reports (ICRP, 1995, 2016b; NCRP, 2009b). However, current biokinetic models of occupational cohorts have largely reflected healthy Caucasian working adult males, resulting in deterministic (i.e., “reference”) biokinetic models (NCRP, 2009b, 2018b) with few recent exceptions (Martinez et al., 2022). Expansion of biokinetic models representative of the broader public or cohort (age- and sex-specific) can lead to internal dose assessment models that include expanded morphometry and other associated factors (diet, smoker dosimetry, radon background). Current innovations in nuclear medicine have adopted patient-specific radionuclide biodistribution (Fisher, 2021), whose principles can be expanded to cohort-specific (e.g., age, sex, pregnant women) factors of radionuclide metabolism for both prospective epidemiological study design and retrospective dose reconstruction to improve identification of dose-response relationships for internalized radionuclides.
Applications of variability of internal dose coefficients have been preliminarily explored in the field of consequence management in the establishment of dose-based public protection–derived response levels (Cochran et al., 2020). Current tools permit consideration of population-specific cohorts37 whose dose estimation must be expanded to reflect impacts from internalized radionuclides. Where dose reconstruction for internal radiation uptakes is often limited by available data, integration of high-fidelity source-term modeling, such as from reactor models (Bixler and Nosek 2021; Wieselquist et al. 2020) or consequence management or fallout tools (Auxier et al., 2017), may supplement both prospective and reconstructive efforts correlating internal dosimetry quantities with high-fidelity source-term modeling of radionuclide inventory and validating with in-field experimental data (environmental monitors, personnel monitors, nuclear forensics tools).
Despite advances in dosimetric modeling, errors in estimated radiation doses can arise from uncertainty in dosimetry parameters, as well as from measurement error in the underlying radiation exposure data (see Section 5.2.1) that are supplied to dosimetry systems. These dose errors can introduce bias in estimation of the dose-response shape and reduce statistical power. Modern statistical and computational methods for dose
___________________
reconstruction and risk estimation can be applied to account for dose errors that are classical or Berkson in form and that are independent or shared across individuals. These include flexible (i.e., semiparametric or nonparametric) statistical models and simulation-based approaches. In particular, recent research has focused on two-dimensional Monte Carlo approaches that simulate alternative sets of doses for an entire cohort rather than a single set that emerges when each individual’s dose is estimated independently from other individuals; these sets of doses are then integrated into dose-response analyses (Kwon et al., 2016; Simon et al., 2015; Stram et al., 2015, 2021). At the same time, a growing body of statistical research has developed methods for measurement error in epidemiological data, particularly in nutritional epidemiology, including methods that correct for correlated errors in exposures (e.g., dose error) and outcomes (e.g., errors to outcome misclassification; Bennett et al., 2017; Keogh et al., 2020; Shaw et al., 2020, 2021; Wu et al., 2019).38 Failure to further develop and apply these methods in epidemiological studies, particularly the large studies that will be required to detect radiation health effects at low doses with sufficient statistical power, will continue to limit the statistical analysis and interpretation of large and complex dose reconstructions for quantifying radiation risk.
5.4.4 Facilities for Low-Dose and Low-Dose-Rate Exposures (Priority I4)
Scientific and Decision-Making Value
The potential gains from a new low-dose radiation research program are highly dependent on available facilities that are tailored to the specific needs of this line of research. These facilities significantly differ from facilities that are used primarily for higher-dose experiments or applications that tend to involve a high-dose-rate delivery, making it very difficult to “turn the source down” to deliver low-dose and low-dose-rate exposures. In other words, a higher-dose activity facility cannot be adjusted easily for use in low-dose radiation research.
Current Status and Promising Research Directions
The committee reached out to several radiation facilities that have capabilities to support low-dose research to understand and document those capabilities. The committee selected “larger” facilities, but it recognizes that other smaller facilities including several cesium-137 irradiators or X-ray sources are routinely used for low-dose research in universities, hospitals,
___________________
38 See Supplementary Appendix B of Gilbert et al., 2020, for a full discussion on dose-error methodology for radiation epidemiology studies.
and other settings. Responses from the facilities, except the response from the Argonne Tandem Linac Accelerator System (ATLAS) facility at Argonne National Laboratory, are shown (unedited) in Appendix E. These responses contain information useful to the research community other than what is summarized in Table 5.2 for the purposes of this committee’s task. In addition to the facilities listed in the table, the committee also reached out to the National Institute of Standards and Technology (NIST) and the University of North Carolina (UNC) at Chapel Hill. NIST did not respond to the committee’s request and a UNC representative told the committee that facilities that house nanobeams for irradiation of cells and animals at UNC are still in the research stage.
An assessment of multiple large radiation facilities that have capabilities to carry out low-dose research revealed that diminished support for radiation research in the United States has left the radiation research community with inadequate exposure facilities to support low-dose and low-dose-rate radiation research and address high-priority issues:
- There are no facilities available to facilitate inhalation studies to understand the effects of internal emitters on the lungs; in the past, the United States had a facility at the Inhalation Toxicology Research Institute in New Mexico, but this facility was terminated. The construction of instrumentation that delivers doses to animals via inhalation and dosimetry support for inhalation studies is complex.
- Facilities for low-dose-rate exposures (similar to those at the Institute for Environmental Sciences in Japan and several in Europe including FIGARO in Norway, and others in the United Kingdom, France, and Italy)39 are not available in the United States. These facilities permit animal exposures and can tune the dose down to be delivered over long exposure times, unlike facilities in the United States.
- Facilities similar to the deep underground facilities in Europe and Canada designed to eliminate the effects of background radiation are of restricted access or of limited capabilities. The committee is aware of the underground radiation biology laboratory at the radioactive waste disposal site Waste Isolation Pilot Plant in New Mexico (Castillo et al., 2021; Van Voorhies et al., 2020).
- High-LET sources such as carbon ion and others can be found in Japan and Germany; several are under construction in Russia, but none are currently available in the United States.
___________________
39 See https://www.concert-h2020.eu/en/Concert_info/Access_Infrastructures/Bulletins.
TABLE 5.2 Available Facilities for Low-Dose and Low-Dose-Rate Research in the United States
| Facility | Radiation Type or Description of Facility | Location | Start; End Year | Main Purpose | Dose Range (max; min) |
|---|---|---|---|---|---|
| AFRRI | Triga reactor (mixed field gamma/neutron) | AFRRI complex | 1969; no plan | Materials, cells, animals; equipment | Information not provided |
| AFRRI | High-level cobalt (mono-energetic) | AFRRI complex | 1968; no plan | Materials, cells, animals | Information not provided |
| AFRRI | Low-level cobalt (mono-energetic) | AFRRI complex | 1974; no plan | Materials, cells, animals | Information not provided |
| AFRRI | Linac (mono-energetic, 4, 10, 15 MeV) | AFRRI complex | 2013; no plan | Materials, cells, animals | Information not provided |
| ATLAS | Ion beams | Argonne National Laboratory, Physics Division | Not provided | Basic research in nuclear physics; production of radioisotopes; currently no radiobiology | Information not provided |
| CSU | Low-dose-rate γ ray (137Cs) tissue culture facility | Main CSU campus (MRB 08) | Not provided; not planned | Radiobiology experiments | Information not provided |
| CSU | Low-dose-rate γ ray (12 individual 137Cs) tissue culture facility | Main CSU campus (MRB 12) | 1997; no plan | Radiobiology experiments | Information not provided |
| CSU | Low-dose-rate neutron tissue culture facility (14.1 MeV) | Main CSU campus (MRB 02) | Expected to be operational in 2022; no plan | Engineered human tissues (“tissues on a chip”) | Information not provided |
| Dose Rate (standard setup) | Inhalation Experiments | Available to Outside Users; Available Adjacent Infrastructure | References Demonstrating Capabilities |
|---|---|---|---|
| From less than 10 R/h to more than 100 kR/h | No | Yes; Yes | Bene et al., 2021 |
| From less than 1 R/h to more than 100 kR/h | No | Yes; Yes | Bene et al., 2021 |
| From less than 1 R/h to more than 100 kR/h | No | Yes; Yes | Bene et al., 2021 |
| From 0.05 to 6 Gy/min | No | Yes; Yes | Bene et al., 2021 |
| Low dose rate not standard but possible | No | Yes but not for radiobiology; Not available | Information not provided |
| Currently 9.3 mGy/h but can increase to 500 mGy/h or decrease to background | No | Yes; Yes | Kato et al., 2006 |
| 1 mGy/h to 100 mGy/h | No | Yes; Yes | Amdur and Bedford, 1994; Bedford, 2001; Huang et al., 2011; Kato et al., 2006, 2007; Ochola et al., 2019; Peng et al., 2012; Ulsh et al., 2001; Wilson et al., 2008 |
| 3.6 mGy/h to a factor of 20 lower | No | Intended; Yes | Information not provided |
| Facility | Radiation Type or Description of Facility | Location | Start; End Year | Main Purpose | Dose Range (max; min) |
|---|---|---|---|---|---|
| CSU | Low-dose-rate γ ray (137Cs) vivarium | Main CSU campus (MRB 06) | —; No plan | Mice and medaka fish | Information not provided |
| CSU | Low-dose-rate neutron (252Cf) vivarium | CSU Foothills Campus | 2017; not planned (new source planned for 2022) | Small animals | Information not provided |
| LLUMC | Synchrotron-based proton facility (50–250 MeV) | G2 | 1990; not planned | Patient treatment; clinical research; radiobiology (cells and tissues) | Information not provided |
| LLUMC | Synchrotron-based proton facility (50–250 MeV) | HBL | 1990; not planned | Physics and radiobiology (cells, tissues, and animals) | Information not provided |
| LLUMC | Linear accelerator (2–22 MeV) | TrueBeam | 2019; not planned | Patient treatment; clinical research; radiobiology (cells and tissues) | Information not provided |
| NSRL | Protons (linac or tandem Van de Graaff) | Brookhaven National Laboratory | ~2003; >2030 | Radiobiology research (cells, tissues, or animals) and electronics testing | >>1 Gy; as low as 0.1 to 0.2 mGy |
| NSRL | Ions (electron-beam ion source, tandem Van de Graaf) | Brookhaven National Laboratory | ~2003; >2030 | Radiobiology research (cells, tissues, or animals) and electronics testing | >>1 Gy; as low as 0.1 to 0.2 mGy |
| RARAF | Neutrons | RARAF, Irvington, NY | 2015; not planned | Radiobiology (mice, tissue, and cells) | Up to 10 Gy; no lower limit |
| Dose Rate (standard setup) | Inhalation Experiments | Available to Outside Users; Available Adjacent Infrastructure | References Demonstrating Capabilities |
|---|---|---|---|
| 10 cGy/day to 0.41 cGy/min | No | Yes; Yes | Ochola et al., 2019; Shakhov et al., 2012 |
| 1 mGy/day | No | Yes; Yes | Acharya et al., 2019; Borak et al., 2021; Perez et al., 2020 |
| Standard setup is 100–200 cGy/min but can range from 1 to 300 cGy/min | No | Yes but limited; Yes | Unternaehrer-Hamm et al., 2020 |
| Standard setup is 100–200 cGy/min but can range from 1 to 300 cGy/min | No | Yes but limited to outside clinical care hours; Yes | Information not provided |
| 5–600 cGy/min | No | Yes but limited; Yes | Information not provided |
| Between 0.01 and 1 Gy/min | No | Yes; Yes | La Tessa et al., 2016; Simonsen et al., 2020 |
| Between 0.01 and 1 Gy/min | No | Yes; Yes | La Tessa et al., 2016; Simonsen et al., 2020 |
| Up to 3 Gy/h | No | Yes; Yes | Marino, 2017; Xu et al., 2015 |
| Facility | Radiation Type or Description of Facility | Location | Start; End Year | Main Purpose | Dose Range (max; min) |
|---|---|---|---|---|---|
| RARAF | Ion beams (LET = 8–1,000 keV/μm) | RARAF, Irvington, NY | 1980; not planned | Radiobiology (cells and thin tissues; microbeam available) | Up to 100 Gy; single particle |
| RARAF | FLASH (6 or 9 MeV electrons) | RARAF, Irvington, NY | 2020; not planned | Radiobiology (mice, tissues, and cells) | Up to 100 Gy; 0.1 Gy |
| RARAF | FLASH (4.5 MeV protons) | RARAF, Irvington, NY | 2018; not planned | Radiobiology (cells and thin tissues) | Up to 100 Gy; 1 msec at selected dose rate |
| RARAF | VADER 137Cs gamma | CUIMC, New York | 2017; not planned | Radiobiology (mice and cells) | 1 month; 5 min at selected dose rate |
NOTE: AFRRI = Armed Forces Radiobiology Research Institute; ATLAS = Argonne Tandem Linac Accelerator System; CSU = Colorado State University; CUIMC = Columbia University Irving Medical Center; HBL = horizontal beam lateral; LET = linear energy transfer; LLUMC = Loma Linda University Medical Center; NSRL = NASA Space Radiation Laboratory; RARAF = Radiological Research Accelerator Facility.
Overall, a substantial investment in facilities specifically designed for internal and external exposures to low-dose and low-dose-rate radiation of types relevant to exposed or potentially exposed U.S. populations will be needed to support a revitalized low-dose radiation research program in the United States.
5.5 ESTIMATED TIMELINE AND COSTS
The development of a robust research program that can provide information about the risks to humans that may result from exposures to low-dose and low-dose-rate radiation and about the involved mechanisms will require significant investments in biological research, dosimetry, epidemiology, facilities, data curation and coordination, education and outreach, and communication. This might be accomplished by establishing and nurturing an interactive multidisciplinary program as illustrated conceptually in Figure 5.2 comprising interacting hubs focusing on basic and translational biology, analytical and computational technologies, and epidemiology. The
| Dose Rate (standard setup) | Inhalation Experiments | Available to Outside Users; Available Adjacent Infrastructure | References Demonstrating Capabilities |
|---|---|---|---|
| Adjustable | No | Yes; Yes | Miller et al., 1999; Randers-Pehrson et al., 2009 |
| 16 mGy/sec to 600 Gy/sec | No | Yes; Yes | Garty et al., 2022 |
| Up to 4 kGy/sec | No | Yes; Yes | Grilj et al., 2020 |
| 0.1–1 Gy/day (time-variable) | No | Yes; Yes | Garty et al., 2020 |
analytical and computational technology hub would provide tools for model system characterization to the biology hub and for assessment of adverse health effects to the epidemiology hub. The biology hub would elucidate mechanisms of response and provide radiation response signatures for assessment in the epidemiology hub. The epidemiology hub would explore associations between radiation and adverse health events and refer strong associations to the biology hub for assessment of mechanism and causality. Program output might include data and methods, radiation response mechanisms, risk estimates, and improved dosimetry
The committee’s research agenda extends for 15 years, to 2037. By that time, several of the biological research priorities (e.g., development of model systems [Priority B1] and development of biomarkers for radiation-induced adverse health outcomes [Priority B2]) and research infrastructure priorities (e.g., tools for detection and precise characterization of aberrant cell and tissue states [Priority I1] and dosimetry for low-dose and low-dose-rate exposures [Priority I3]) are expected to be completed or to be approaching completion and providing critical information. However, it is likely that the epidemiological research priorities will extend further into the future based on progress with improving dosimetry for epidemiological studies (Priority I3), further establishment of database infrastructure (Priority I2), and advances on the most biologically important components of low-dose and low-dose-rate radiation (e.g., through research on Priorities B2 and B4). That said, certain aspects of the epidemiological research priorities can begin immediately to complement planning for future epidemiological studies. Notably, these efforts should include engagement with the research and other stakeholder communities to examine to what extent some of the committee-recommended research can be conducted by adding to existing population studies and to identify the appropriate study populations for next-generation radiation epidemiology studies. Certain epidemiological studies could require substantial time for completion, possibly three decades or more. This means that long-term commitment for the program (see Section 6.1) is essential for its success.
The committee estimates that funding needed to set up the program is on par with the congressionally authorized funds for 2023 and 2024 (see Appendix A), that is, at the level of $30 million and $40 million annually, respectively, but needs to rise to the level of $100 million annually thereafter and remain at that level through about 2037. Periodic reassessments are required as large epidemiological studies and necessary research infrastructures are established. Although the committee recognizes that the exact form of the program will be determined by the funding agency after consultation with stakeholders, the following prototypical program is intended to justify the $100 million annual funding level. Specifically the program might include
- A basic and translational biology hub comprising approximately 20 individual laboratories, 6 multidisciplinary centers, 3 technology development and deployment centers, and a basic science data coordinating center (cost ~$50 million annually). This effort would be similar in scale and composition to other targeted basic and translational biomedical research programs now operated by NIH (e.g., NIH HuBMAP; the NCI HTAN program, the NCI Cancer Systems Biology program).
- An epidemiology hub comprising three epidemiology centers, an epidemiology data coordinating center, and a dosimetry center (cost ~$30 million annually) dedicated to quantification of risks to U.S. populations exposed to low-dose and low-dose-rate radiation. This program is similar in size to the one carried out at the Radiation Effects Research Foundation (see Section 4.5.6).
- An education, outreach, and policy hub comprising an education center that develops training curricula and administers a training grant program, a science policy center, and an outreach and communication center (cost ~$10 million annually).
- A facilities development program for low-dose and low-dose-rate exposures (cost ~$5 million annually).
- Program administration, internal and external coordination, grants management, and operation of an external advisory committee (cost ~$5 million annually). A key aspect of program administration will be to ensure coordination between low-dose basic research, epidemiological research, technological research, education, and outreach efforts within the U.S. program and with non-U.S. low-dose radiation research programs.
Congressionally authorized funds for the program were at the level of $20 million in 2021 but much lower amounts ($5 million) were appropriated. Congressional staff who briefed the committee noted that additional funds could become available, with the appropriate justification and with evidence of research progress.40 The committee recognizes that the research priorities it identified need to be achievable within reasonable budgets. Although it did not make a detailed assessment of the costs associated with the proposed research agenda, the committee used its informed collective judgment to provide a rough estimate of the research costs. Overall, it found that for the period 2023–2024, the currently authorized funds (if fully appropriated, which is rarely the case) may be sufficient to initiate
___________________
40 Alyse Huffman and Adam Rosenberg, Committee on Science, Space, and Technology, Energy Subcommittee, U.S. House of Representatives, presentations to the committee on August 26, 2021.
the low-dose radiation program, but significant investments, at the level of $100 million annually, will be required for the period 2025–2037.
The estimate of $100 million annually provided by the committee is larger than the amount authorized so far for the low-dose radiation program. However, the large number of individuals within the U.S. population that are exposed to low-dose and low-dose-rate radiation annually as a result of medical, occupational, and environmental exposures (see Table 2.1 for examples) and the substantial costs for complying with radiation protection standards and guidelines make it critical that the risk management and mitigation efforts for the U.S. population be guided by complete information about possible risks for cancer, cardiovascular disease, neurological disease, and other disease risks that may exist. This will require the $100 million annual program scale recommended by the committee. Appropriations at the level of $5 million per year are not adequate to even initiate a meaningful low-dose radiation research program—as seen in 2021 when funds for the program were at that level and the program was not initiated. The committee cautions that inadequate funding for the program will lead to continued scientific and policy debates about risks of low doses of radiation and the possible inadequate protection of patients, workers, and members of the public from the adverse effects of radiation.
5.6 COMPARISON OF THE COMMITTEE-RECOMMENDED RESEARCH AGENDA TO THOSE OF OTHER ENTITIES
The committee reviewed the research agendas proposed by other entities, in particular, the Multidisciplinary European Low-dose Initiative (MELODI; see Section 4.5.1), ICRP (see Section 4.5.2), and the Canadian Organization on Health Effects from Radiation Exposure (COHERE; see Section 4.5.5), and compared those with its recommended agenda. Overall, it found consistency in the main scientific issues recognized to be of highest priority by the different entities. These include improving cancer risk assessments for different radiation types and modes of exposure, understanding risks of health outcomes other than cancer, and improving dosimetry. Similar to this committee’s research agenda, ICRP and MELODI prioritize the need for a more individualized risk assessment. ICRP is also concerned with the effects of radiation on non-human biota, a topic not addressed by the research agendas proposed by MELODI, COHERE, and this committee. The overall concordance in these research agendas indicates that collaboration and cooperation at the international level will be important to avoid unnecessary duplication of efforts, to share costs for projects, and to identify the most promising synergies among the different research programs.
In addition to the general consistency in the main scientific topics recognized to be of highest priority by the different entities, there is also
consistency in the approach of phrasing these priorities. Specifically, this committee, similar to MELODI, ICRP, and COHERE, is neutral regarding the possible outcomes of the research and does not favor or appear to favor a certain possible outcome. A notable exception to this approach is that of the National Science and Technology Council (see Section 4.1.6), which phrased its priority as “defining the threshold of impact for low-dose and low-dose rate,” implying that such a threshold does exist.
Despite the general similarities in the research topics and approach, this committee’s approach to defining the research agenda differs in four important ways:
- It emphasizes the need for integration between the epidemiological and biological research lines it identified and true partnerships between the two research lines to achieve the program’s goals.
- It provides equal emphasis on understanding the risks of cancer and non-cancer health outcomes.
- It proposes the establishment of new epidemiological studies that can address questions about risks at low doses and dose rates and proposes that the appropriate populations are selected with input from the research community and other stakeholders, including the impacted communities.
- It is based on leveraging advances in biotechnology and research infrastructure to help address the research priorities.
5.7 CHAPTER SUMMARY, FINDINGS, AND RECOMMENDATION
Epidemiological and biological research on low-dose and low-dose-rate radiation faces several challenges (see Box 5.1). These challenges are most often ascribed to the fact that effects of low-dose and low-dose-rate radiation exposures are assumed to be subtle and difficult to distinguish from those caused by other stressors and/or “spontaneous” changes that adversely affect the normal functions of cells, tissues, and organs. Moreover, the magnitude of the effects may change with dose, dose rate, type of radiation, and mode of exposure.
The committee identified 11 research priorities broadly classified in three research lines: epidemiological research, biological research, and research infrastructure (see Table 5.1). These research priorities can help overcome the existing research challenges, will enable more accurate estimation of adverse health effects that result from exposure to low-dose and low-dose-rate radiation, and will improve knowledge of the complex cellular and molecular processes that are engaged during transduction of low-dose and low-dose-rate radiation damage into adverse health outcomes. The committee emphasizes the need for integration across the research
lines and anticipates that the most impactful research projects will include work in more than one research line and will be carried out by multidisciplinary teams. These research priorities do not represent a complete list of important low-dose and low-dose-rate radiation research questions. For example, studies designed to confirm or strengthen the basis for existing scientific findings, particularly those that are controversial or lack a clear interpretation, are also important. Some of the research priorities can have additional benefits including capacity building, training of the next generation of radiation researchers, and development of tools that could be transferrable to other lines of research.
The order of the research priorities does not imply an order of significance; instead, the priorities are considered by the committee to be equally important. Some of these activities can be initiated immediately, and others can only begin after a better foundation is built from current or new research or with additional input from the research and broader stakeholder community, including the impacted communities.
The committee expects that the specific tactics for addressing the research priorities and integrating epidemiological and biological components will be developed with input from the extended research community and other stakeholders, including the impacted communities. Importantly, the committee recognizes that the list of priorities will likely evolve as biological understanding and research tools advance and as the research community and other stakeholders are engaged with the program.
The committee estimates that funding needed to set up the program is on par with the congressionally authorized funds for 2023 and 2024, that is, at the level of $30 million and $40 million annually, respectively, but need to rise to the level of $100 million annually thereafter and remain at that level through about 2037. Although the committee recognizes that the exact form of the program will be determined by the funding agency after consultation with stakeholders, it provided a prototypical program comprising interacting hubs focusing on basic and translational biology, analytical and computational technologies, and epidemiology, intended to justify the $100 million annual funding level. The committee also notes that appropriations at the level of $5 million per year are not adequate to even initiate a meaningful low-dose radiation research program—as seen in 2021 when funds for the program were at that level and the program was not initiated. The committee cautions that inadequate funding for the program will lead to continued scientific and policy debates about risks of low doses of radiation and the possible inadequate protection of patients, workers, and members of the public from the adverse effects of radiation.
Finding 4: Epidemiological studies have played a crucial role in identifying risks (primarily for cancer) from medical, occupational, and
environmental radiation exposures at low doses. Existing epidemiological studies are unable to address a number of outstanding questions of low-dose and low-dose-rate exposures of concern to the U.S. population including the full range of potential adverse health effects, risks associated with doses around 10 milligray, and the potential impacts of genetic, lifestyle, environmental, and other factors that may also affect radiation-related risk estimates. Epidemiological studies designed to overcome these limitations can better elucidate adverse health effects of radiation exposure at low doses and low dose rates relevant to the U.S. population today.
Finding 5: Radiation biology studies have contributed to the mechanistic understanding of the effects of radiation on molecular pathways and intra- and extracellular processes. The application of novel and developing technologies will enable more precise definition of the cellular and molecular processes that are affected by low-dose and low-dose-rate exposures. Integration of this information with that from epidemiological studies will enable better quantification of the adverse health effects from low-dose and low-dose-rate exposures relevant to the U.S. population, increase understanding of the involved mechanisms, and inform on the most appropriate risk assessment models to be used.
Finding 6: Advances in biotechnology and research infrastructure have been driven by the vast research and development enterprise in the United States. These include new observational and experimental systems, tools for measurement and genetic manipulation, increased computational power, improved interpretative algorithms, and shared data access systems. These advances have enabled innovation and breakthroughs in many scientific areas including cancer research and treatment, environmental health effects research, and vaccine production. A revitalized low-dose radiation research program can likewise leverage and further develop these capabilities to enable scientific innovation and breakthroughs in radiation biology and epidemiology.
Recommendation A: Agencies responsible for the management of the multidisciplinary low-dose radiation program should fund low-dose and low-dose-rate radiation research in the 11 high-priority research topics identified by the committee and can address the scopes outlined in Finding 1. (See Table 5.1 for listing and approach for addressing the recommended priorities.) These research priorities are broadly classified as epidemiological research, biological research, and research infrastructure and are of equal importance.
Finding 7: Significant investments over a sustained period spanning several decades are necessary to develop a multidisciplinary low-dose radiation research program in the United States that leverages existing and developing research infrastructure that will achieve the goals outlined in Finding 1. The committee’s best estimate is that the investments required during the first 10–15 years of the program are at the level of $100 million annually and periodic reassessments are required as large epidemiological studies and necessary research infrastructures are established.






























































