5
A General Framework for Respiratory Protection Oversight and Guidance
Prior chapters have established the context for developing a framework in response to the committee’s charge to
make recommendations for a framework of responsibilities and authorities, including those of federal agencies, to provide a unified and authoritative source of information and effective oversight in the development, approval, and use of respirators, including conformity assessment processes to meet the needs of these populations (see Box 1-1 in Chapter 1).
Together, Chapters 2–4 provide this context by reviewing the regulatory, legal, and other measures currently in place for respiratory protection for workers not covered by a respiratory protection program (RPP) and for the general population. Many of the challenges highlighted in these chapters relate to the fragmented regulatory landscape and the lack of a cohesive national strategy for addressing the respiratory protection needs of these two populations. In this chapter, the committee sets out a general framework for meeting these challenges. Chapters 6 and 7 build on this general framing. Responding to its charge and building pragmatically on structures in place, the committee proposes two separate frameworks—one for uncovered workers and the other for the general population. We note that the underlying concepts related to these frameworks are comparable, with a systems approach being embraced as the path to risk reduction through assurance of respiratory protection. Perhaps a unified framework will be feasible in the future, but the committee did not perceive that approach as practicable at present.
A SYSTEMS APPROACH TO REDUCING RISKS FROM INHALATION HAZARDS
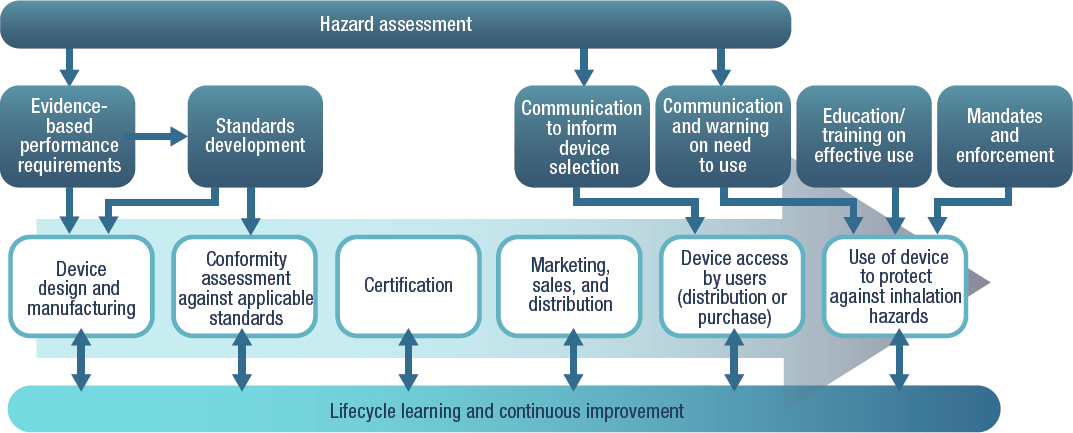
The committee’s frameworks have the purpose of ensuring effective respiratory protection for those needing it when exposure poses a risk deemed to be unacceptable. The committee took a systems approach to addressing how that purpose can be achieved, detailing the many steps and their components that are required to make respiratory protective devices available when needed and ensure their effective use. An overview of those diverse processes is depicted in Figure 5-1, which is a simplification of a complex system, as each step has its own components and operates in a societal context not easily distilled into graphical form. While the component processes depicted in Figure 5-1 are the same for workers and the public, differences in how they are implemented, as described below, signify the need for separate frameworks for the two populations.
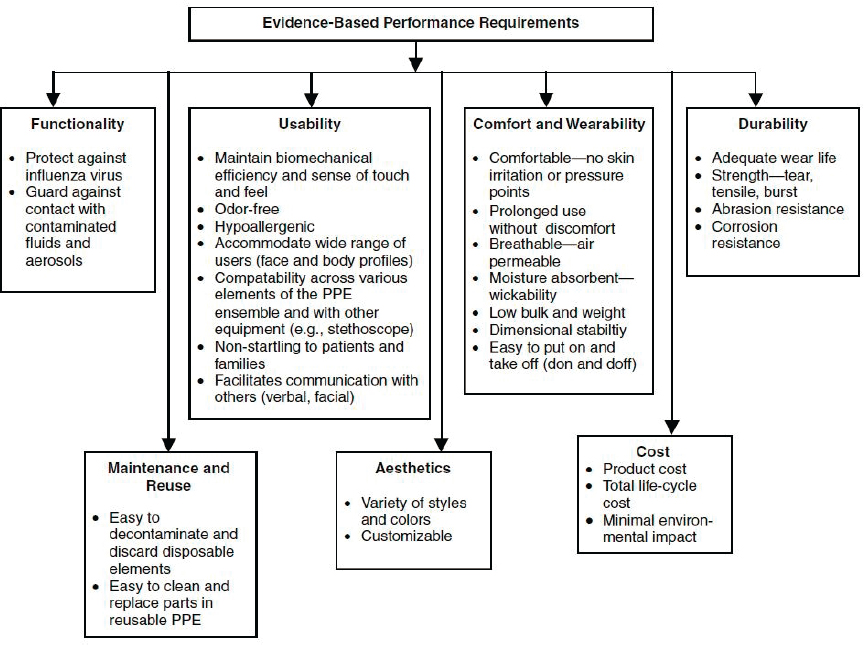
The committee’s approach to understanding respiratory protection as a system builds on a prior Institute of Medicine report, Preparing for an Influenza Pandemic: Personal Protective Equipment for Health Care Workers (IOM, 2008). That report describes the first step in the sequence depicted in Figure 5-1—the design of respiratory protective devices—as part of a lifecycle from evidence-based performance requirements to field testing. Figure 5-2, first published in that report, outlines a set of overarching performance requirements to guide the design and development of PPE in a manner that prioritizes functional characteristics, such as filtration efficiency, and user-centric needs. From this broad set of design requirements, more specific requirements can be identified for specific use scenarios, considering the hazard and the intended users, including challenges faced by vulnerable groups in terms of gaining access to respiratory protective devices.
The general system sequence outlined in Figure 5-1 begins with product design and manufacturing. The second major process is conformity assessment, which focuses on “product effectiveness—verifying and ensuring that a product meets specific criteria for use in workplaces and other locations” (IOM, 2011, p. 19). To reinforce the systems paradigm, conformity assessment has its own set of subprocesses or subsystems and distinct regulatory structures for workers and the public, as discussed in Chapter 2. Certification of the respiratory protective device (e.g., by the National Institute for Occupational Safety and Health [NIOSH]/National Personal Protective Technology Laboratory [NPPTL], or a third party) is next, providing a mark of assurance as to effectiveness and safety for users of the product.
As shown in Figure 5-1, the process steps associated with the manufacturer culminate in product marketing, sales, and distribution (e.g., to retailers or directly to purchasers). The next step is access to the product by users who need a respiratory protective device for protection against
NOTE: PPE = personal protective equipment.
SOURCE: IOM, 2008.
a specific inhalation hazard. Access can occur in two ways: respiratory protective devices may be provided to users, or users may purchase them from a retailer. For example, an employer that has instituted an RPP may purchase respiratory protective devices and provide them to employees to protect them against inhalation hazards in the workplace. Or a family might choose to purchase and store such devices because of potential risks from wildfire smoke or airborne infectious agents. Thus, the workers not covered by an RPP and members of the public could procure the product directly in the marketplace for personal use, if affordable. For both employers and individuals purchasing a device, communication of hazard-specific specifications for the device need to be provided to ensure selection of an appropriate product.
The next critical process step is effective use of the respiratory protective device, triggered by a specific inhalation hazard for which it has been determined that other controls cannot adequately prevent exposure. Communication regarding the need to use the device and education and training in its use (e.g., donning, doffing, cleaning, and storage) are critical inputs to this step. In some cases, as in workplaces where workers are exposed to inhalation hazards that cannot otherwise be controlled, use of respiratory protection may be mandated and compliance monitored.
Finally, underlying these six process steps is the paradigm of ongoing learning and continuous improvement that is critical to catalyzing advances in each step and ensuring the robustness of the system in responding to changes. Examples of such processes include postmarket audits and ongoing usability evaluations.
It should be noted that, although depicted in Figure 5-1 as sequential, these process steps could occur simultaneously and iteratively as processes are modified based on experience. The dynamic nature of the continuously evolving system is indicated by the bidirectional arrows connecting each process step to the ongoing process of lifecycle learning and continuous improvement at the foundation of the system.
Across the general sequence of process steps depicted in Figure 5-1, the components are comparable for the two targets of this report—workers not covered by RPPs and the public. This chapter acknowledges these parallels, while Chapters 6 and 7 describe the committee’s specific frameworks for these two groups, respectively. As described in Chapters 2 through 4, some of the processes are carried out by existing entities to varying degrees and are highly fragmented. For instance, the identification of evidence-based requirements to inform product design is carried out by manufacturers, occupational safety and health professionals, and researchers for the worker group. In the case of the public, roles are not as well established, although researchers are engaged in doing some groundwork for the identification of such requirements. Similarly, those employers with Occupational Safety and
Health Administration (OSHA)-required RPPs are responsible for training employees in the use of respirators, while there is no such designated authority for the public.
In the sections below, the committee introduces its approach to addressing the element of its charge concerned with making recommendations for the requested framework. The committee’s proposed framework extends from the design, manufacturing, and testing of respiratory protective devices to their delivery to users who need to be prepared to use them in a way that will provide the desired level of protection, as depicted in Figure 5-1. The frameworks for workers without RPPs and the public (Chapters 6 and 7, respectively) address each process in this sequence and reflect the diversity of the populations that are potential users of respiratory protective devices.
A GENERAL FRAMEWORK FOR OVERSIGHT AND GUIDANCE TO MEET RESPIRATORY PROTECTION NEEDS
The framework described in this section is designed to ensure that the processes depicted in Figure 5-1 can be implemented, evaluated, and modified with experience over time. Key terminology used here is defined as follows:
- framework: the overall structure for reducing risk from inhalation hazards among workers not covered by an RPP and the public
- functions: what the elements of the framework need to accomplish
- subfunctions: the various activities needed to accomplish each of the functions
- coordinating entity: the unified and authoritative entity responsible for coordination, and decision making, and effective oversight with respect to the framework’s functions
The committee’s framework was guided by the six principles introduced in Chapter 1. In proposing the framework, the committee adopted a four-step process along the lines of the National Planning Frameworks within the National Preparedness System, proposed by the Department of Homeland Security11:
- describing the core functions (i.e., needed capabilities);
- aligning key roles and responsibilities for delivering those capabilities;
___________________
11 See https://www.fema.gov/emergency-managers/national-preparedness/frameworks (accessed August 20, 2021).
- describing coordinating structures that will enable all stakeholders to work together; and
- laying the foundation for further operational coordination and planning that will synchronize efforts of all stakeholders across the public and private sectors.
The subsections that follow describe the key functions that must be carried out within the framework. The chapter then goes on to consider the general needs related to aligning roles and responsibilities with the framework and potential models for coordinating structures. The mapping of stakeholder roles and responsibilities to functions and specific coordination structures are discussed for workers and the public in Chapters 6 and 7, respectively.
The Core Functions
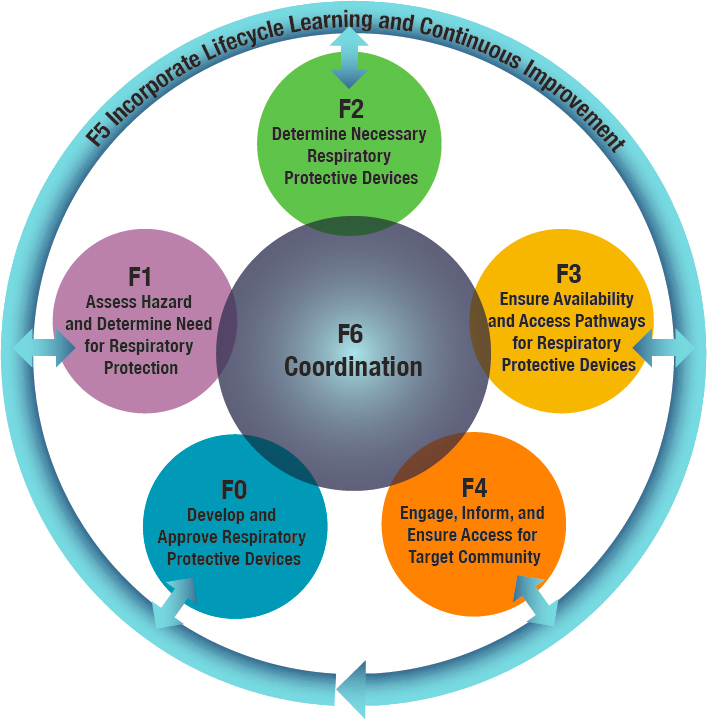
Figure 5-3 shows the core, high-level functions that must be carried out as part of the framework:
- Function F0: Develop and approve respiratory protective devices;
- Function F1: Assess hazard and determine need for respiratory protection;
- Function F2: Determine the necessary respiratory protective devices;
- Function F3: Ensure availability and access pathways for respiratory protective devices;
- Function F4: Engage, inform, and ensure access for the target community;
- Function F5: Incorporate lifecycle learning and continuous improvement; and
- Function F6: Coordination
These core functions make up a comprehensive framework that builds on the experience reviewed in Chapters 2 through 4 and addresses the identified gaps. For example, the eighth conclusion in Chapter 4 states that the public does not know where, which, and when respiratory protective devices should be used. Therefore, the core function “Determine the necessary respiratory protective device” is an essential element of the framework. Similarly, the other core functions are designed to fill gaps identified in the conclusions presented in Chapters 2–4. As discussed above for the process steps shown in Figure 5-1, while the functions are ordered numerically, this is not intended to imply a specific sequence in which they must occur; the functions are dynamic, and their sequencing will be determined by the specific situation, with some occurring simultaneously.
Each of the functions has component functions, or subfunctions, as depicted in Figures 5-4 through 5-7. These subfunctions may be ongoing processes or may be triggered by a specific hazard (see Table 5-1). In the context of respiratory protection needs for emergencies, the ongoing subfunctions represent critical aspects of national preparedness.
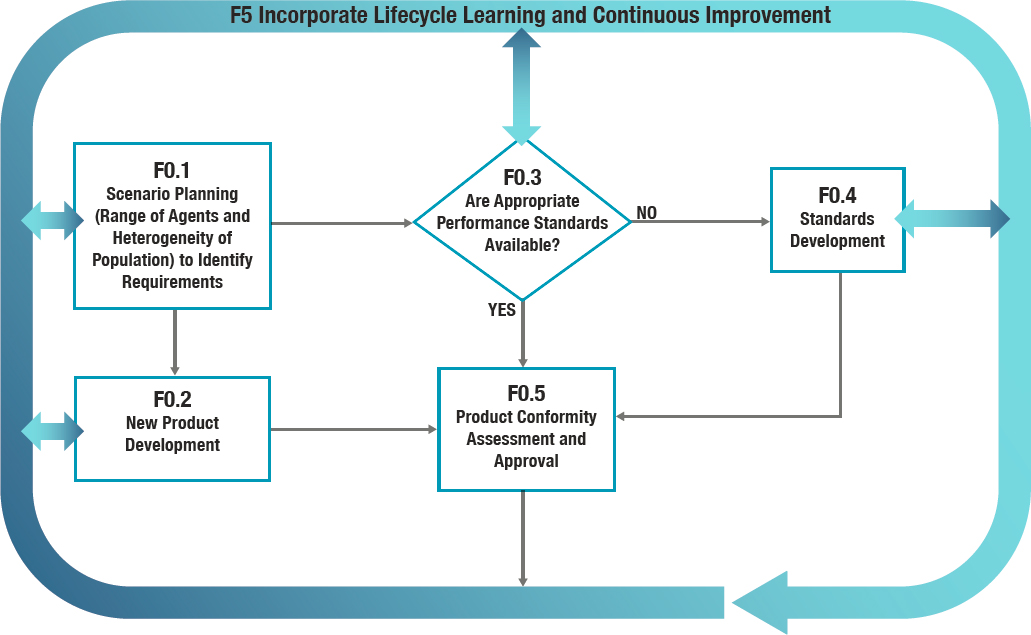
Function F0 (see Figure 5-4) encompasses the continuous development and approval of respiratory protective devices as technologies evolve and needs and standards change. Its subfunctions are linked directly to the first three processes depicted in Figure 5-1. Within this function, new product development is informed by ongoing scenario planning, which considers both the range of potential hazardous agents for which respiratory protection may be needed and the heterogeneity of the population that may require respiratory protection. Product performance standards may
TABLE 5-1 Ongoing and Event-Triggered Activities for Each of the Core Framework Functions
| Function | Preparedness (Ongoing) Activities | Postevent Activities | |
|---|---|---|---|
| F6. Coordination | F0. Develop and Approve Respiratory Protective Devices |
|
|
| F1. Assess Hazard and Determine Need for Respiratory Protection |
|
|
|
| F2. Determine the Necessary Respiratory Protective Devices |
|
|
|
| F3. Ensure Availability and Access Pathways for Respiratory Protective Devices |
|
|
|
| F4. Engage, Inform, and Ensure Access for the Target Community |
|
|
|
| F5. Incorporate Lifecycle Learning and Continuous Improvement |
|
|
|
be developed or refined in response to such scenario planning and form the basis for the conformity assessment process on which product approval depends. Postmarket surveillance is an important element of the conformity assessment process, facilitating continuous improvement in the development of respiratory protective devices.
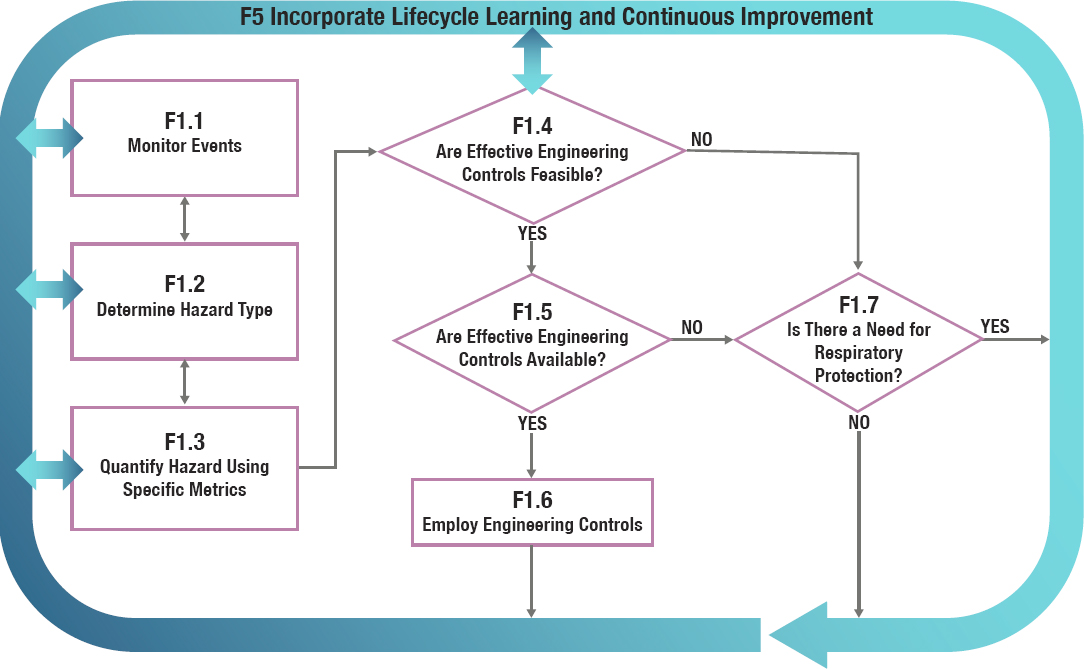
Function F1 (see Figure 5-5) encompasses the ongoing surveillance for and assessment of a hazard to determine whether there is a need for respiratory protection, considering first whether the hazard can be adequately mitigated through other measures higher in the hierarchy of controls, with consideration of the acceptability of the risk posed. In Function F2 (see Figure 5-6), the hazard assessment from Function F1 is used to determine the protective device that best meets the respiratory protection needs posed by the hazard, as determined by the type, risk, and duration of exposure; peak exposure concentration; degree of susceptibility; and feasibility of mitigating the exposure. This is a challenging task in the workplace, since “well-validated air sampling and analytic methods are not readily available” for infectious aerosols (Brosseau et al., 2021). The challenge is compounded in determining respiratory protection needs when the agent may be ubiquitous, such as the needs for the public exposed to infectious agents and workers engaged in fighting wildfires. Consequently, predicting the required characteristics of a respiratory protective device that needs to be effective in multiple environments posing difficult-to-characterize risks, such as SARS-CoV-2 or wildfire smoke, will necessitate effective tools, and such approaches as qualitative and computational methods (Kochenderfer, 2015) may be worth considering. Also to be considered are such user-centric factors as the usability of the device across different subgroups of the population.
Once the necessary level of protection (e.g., filtration efficiency) has been determined for the specific inhalation hazard of interest (e.g., SARSCoV-2), the next subfunction is to determine whether an approved respiratory protective device with the required specifications exists in the marketplace (see Figure 5-6). For respirators for workers, this step might be carried out by utilizing NIOSH/NPPTL’s Certified Equipment List (CEL)22—a tool designed “to help respirator users, safety managers, industrial hygienists, and others to determine if the respirator they are currently using or considering using is NIOSH-certified” (NIOSH, 2020). By enabling approved respiratory protective devices to be identified and ordered directly from their manufacturers, this function will also limit use of the counterfeits currently plaguing the supply chain and adversely affecting both manufacturers and users. As shown in Figure 5-6, once the availability of the
___________________
22 NIOSH’s CEL is available at https://wwwn.cdc.gov/niosh-cel (accessed September 28, 2021).

* Factors considered in determining the necessary protection include the type, risk, and duration of exposure; peak exposure concentration; degree of susceptibility; and feasibility of mitigating the exposure.
desired respiratory protective device(s) is confirmed, the next subfunction is to develop guidelines for its effective use to address the specific inhalation hazard. These guidelines will then be used to tailor guidance for last-mile communication with the target community with regard to effectively addressing the inhalation hazard (Function F4).
If a device with the needed specifications does not exist, the framework triggers the design and development of a new product with the engagement of manufacturers (Function F0). One major concern of manufacturers is the inconsistent demand and absence of volume guarantees for products designed to meet specific needs that require large infrastructure investments for design, development, and manufacturing (McCullough, 2021). The framework therefore needs to incorporate mechanisms for incentivizing manufacturers to produce new products when needed and in sufficient quantity, as well as for incentivizing the development of an adequate market for these products. Since developing a new product will take time, the framework initiates efforts to rapidly find and recommend a respiratory protective device for immediate and interim use. This immediate solution of using the best available device while waiting for manufacturers to design and develop the required respiratory protective device for the specific hazard at hand may raise liability concerns for manufacturers. This issue can be addressed by such measures as that adopted for some respiratory protective devices during the COVID-19 pandemic—extension of liability protection to manufacturers and distributors by invoking and including such devices as covered countermeasures under the 2005 Public Readiness and Emergency Preparedness (PREP) Act (CRS, 2021).
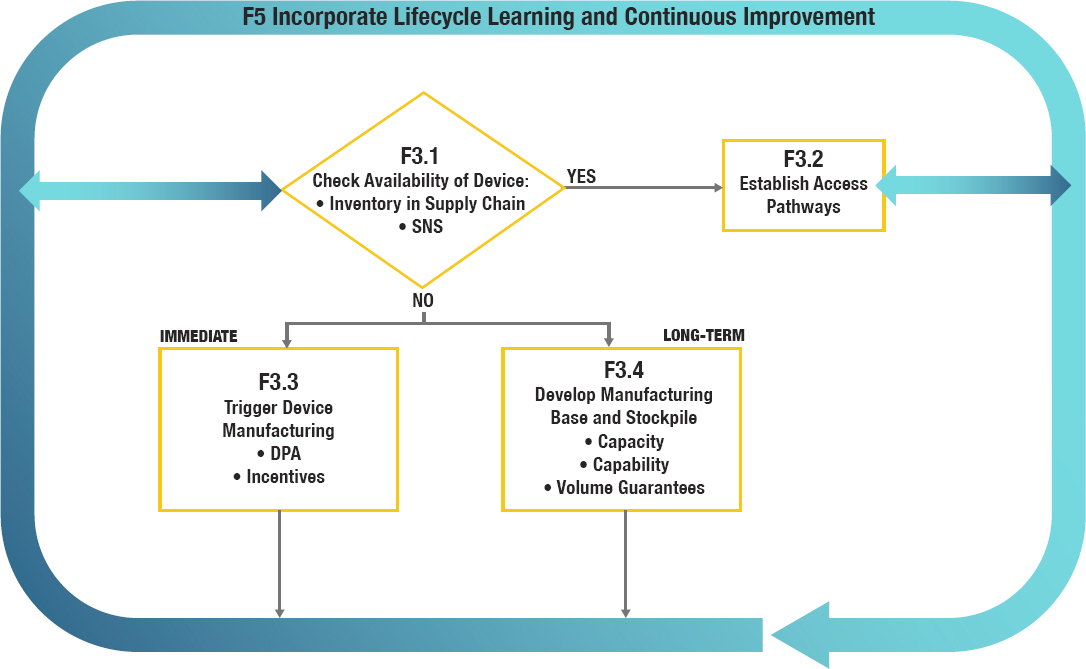
If an approved device that meets the needs for the specific hazard at hand exists, availability needs to be ensured in terms of both supply and pathways by which end users can access the devices (see Function F3, Figure 5-7). Evaluation of supply can be accomplished by consulting with the devices’ manufacturers and by checking the Strategic National Stockpile (SNS). Stockpiling is a critical ongoing subfunction within Function F3 for ensuring adequate supplies of respiratory protective devices during a large-scale emergency. While manufacturers may not share their inventory levels publicly for competitive reasons, the framework needs to include a mechanism for facilitating the confidential sharing of that information for purposes of further action.
If adequate inventory of the desired device is not available, the framework triggers two actions as shown in Figure 5-7. The first is short-term, and could include invoking the Defense Production Act (DPA) and providing incentives for manufacturers of the needed products, such as financial and volume guarantees. In the long term, a mechanism for developing a sustainable manufacturing base with surge capability is necessary to ensure adequate capacity and capability to produce and supply the necessary
NOTE: DPA = Defense Production Act; SNS = Strategic National Stockpile.
devices. Public–private partnerships with delineated responsibilities and accountabilities will be needed to meet this need.
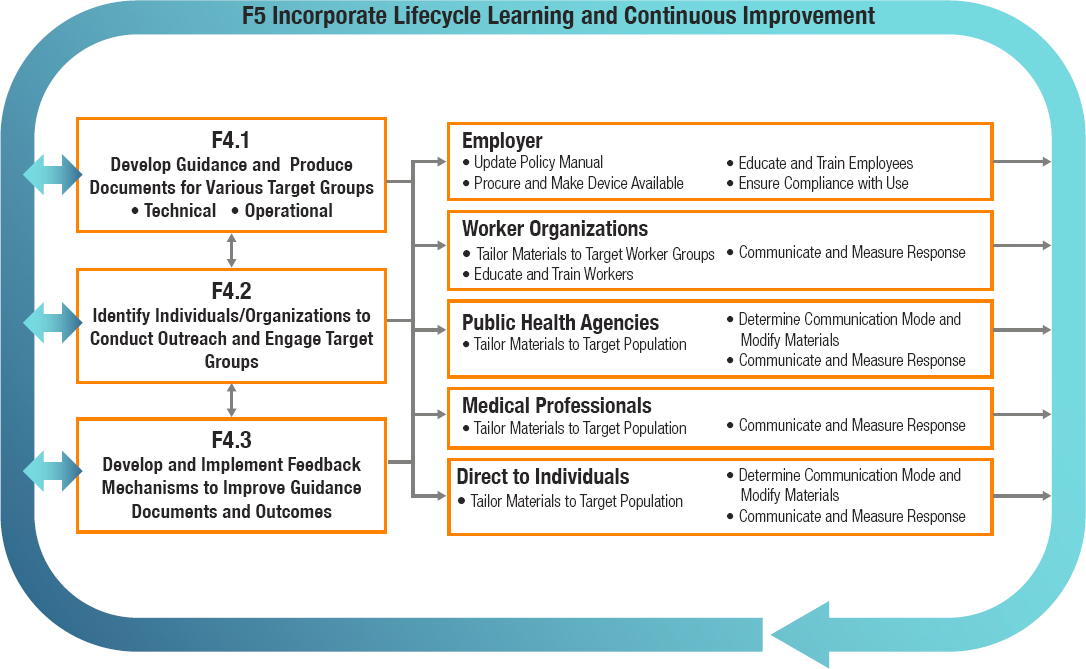
The next major function (F4) is to engage, inform, and ensure access for the target community (see Figure 5-8). The first step here is to develop guidance, which must be uniform, consistent, timely, and continuously updated as new facts emerge, especially in the event of an ongoing public health emergency. The guidelines developed under Function F2 with hazard-specific recommendations for a respiratory protective device serve as the foundation for the development of guidance for target audiences. The approach taken to this step needs to address the technical aspects of the recommended protection (e.g., which device, when and why to use it); how the target populations may react to the recommendations given local, cultural, and socioeconomic factors; and how the target populations will best be reached with accessible information. Therefore, as shown in Figure 5-8, the next step is to identify individuals and organizations that will be responsible for conducting the outreach and engaging the target population to communicate guidance on effective use of respiratory protection against the inhalation hazard in question. Engagement is not limited to the one-way dissemination of information; it is important that mechanisms for engaging with target audiences be bidirectional. Feedback mechanisms need to be developed and implemented to enhance the guidance documents and outcomes. Furthermore, guidance needs to be targeted specifically to the distinct audiences responsible for last-mile communication with users, such as employers, worker organizations, and medical and public health professionals. In turn, these stakeholders can use this guidance in tailoring their communications to reflect the characteristics of their target communities. Additionally, the approach taken in this step will need to facilitate direct communication with users. An important aspect of this function is continuously monitoring for and countering the spread of misinformation.
The next major function (F5) covers the ongoing learning and continuous improvement that is critical to catalyzing advances in each of the core functions and reflects the preparedness–response cycle. The dynamic nature of the continuously evolving framework is denoted by the bidirectional arrows connecting the other core functions to F5 in Figures 5-3 through 5-8. Periodic reporting on progress toward established goals and objectives serves as a mechanism for ensuring ongoing accountability and national preparedness for respiratory protection.
The final function (F6)—coordination—is at the heart of the framework as shown in Figure 5-3. It is the “glue” that binds the core functions and is critical to realizing the framework’s ultimate goal. To carry out this essential function, a coordinating entity is needed, on an ongoing basis, to harness the domain-specific expertise of all stakeholders in respiratory protection in enhancing the nation’s preparedness and providing an actionable
response for workers not covered by RPPs and members of the public facing inhalation hazards.
Scope of the Coordination Function (F6): Current Stakeholder Authorities
The findings presented in Chapters 2 through 4 demonstrate that the existing system for the provision and oversight of respiratory protection for workers not covered by RPPs and the public is highly fragmented, and for the public, there are gaps in critical authorities, related in particular to conformity assessment and approval of respiratory protective devices. As depicted in Table 5-2, current stakeholders have jurisdictions that are tied
| Workplace Exposures | Nonoccupational Exposures | |
|---|---|---|
| Requirements for Respiratory Protective Devices |
Respirators
Surgical/medical masks and some surgical respirators used in health care
Respirators used in mining
|
Respiratory protective devices used for medical purposes, including source control
Flammability requirements for face coverings
|
| Requirements and Guidance on Use of Respiratory Protective Devices |
Mining
Pesticide handling
Radiological inhalation hazards
|
Guidance for communicable and environmental hazards and requirements in limited situations
Guidance for environmental hazards
Guidance for emergency preparedness
Guidance for Americans abroad
Guidance and requirements
|
to the characteristics of the hazard at hand, the locus of exposure (i.e., whether the exposure occurs in an occupational or nonoccupational setting, in the United States or abroad), and the population exposed. This fragmentation and the lack of a cohesive national strategy for informing, educating, and providing guidance for workers without RPPs and the public as to how they can protect themselves from inhalation hazards highlight the need for a central, accountable entity to perform a coordinating function across the framework. Absent such an entity, the goal of providing respiratory protection for workers without RPPs and the public may not be met satisfactorily.
Potential Organizational Configurations of the Coordinating Entity (F6)
This section describes several potential models for a unified and authoritative coordinating entity, drawing on the reviews in Chapters 2 through 4 and other findings from the committee’s information-gathering process. Such an entity or entities will be needed for both targets of this report, workers without RPPs and the public; the general considerations that follow are potentially applicable to both of these target groups. Note that the committee is not proposing here that a single entity should cover both groups, but offering a general discussion of options to set the stage for Chapters 6 and 7, wherein the specific considerations informing the preferred configuration for the coordinating entity for the worker and public frameworks, respectively, are discussed. The following discussion is thus conceptual and is not intended to designate roles for existing agencies or to describe the development of specific new entities.
The committee considered a range of configurations for a coordinating entity, from a single stand-alone entity—the centralized single-agency model—to a coalition of federal or possibly other agencies charged with accomplishing a specific operational goal—the interagency task force model. Between these two extremes of centralization, the committee considered a third configuration—the hub-and-spoke model—in which the coordinating entity would be empowered to harness and build on the expertise and experience of current stakeholders engaged in respiratory protection while having some degree of authority and the ability to remedy gaps. These three models, which are further described in the subsections below, are not necessarily mutually exclusive. It is possible that one model could be implemented initially and the architecture could later transition to another form as needs and conditions changed or new actions were taken, such as by Congress.
The Centralized Single-Agency Model
In the centralized single-agency model, a single entity or agency would be tasked with carrying out all the activities included within Functions F0 through F5. To an extent, the combination of OSHA and NIOSH/NPPTL reflects this model for those workers currently covered by RPPs, although OSHA and NIOSH have separate designated roles and do not function as a single entity. Implementing such a model de novo would necessarily entail reassignment of some authorities away from entities currently holding them. As discussed earlier in this chapter and conveyed in Table 5-2, there are current stakeholders throughout the federal government and at the state and local levels associated with some of the framework’s functions; these stakeholders harness the foundational knowledge and core competencies built over the years to carry out those activities. Building such core competencies in a new centralized agency would require significant investments in capacity building and entail a potentially lengthy timeframe. Moreover, reassigning responsibilities of current stakeholders or expanding their responsibilities and authorities could pose significant organizational, operational, and regulatory challenges and could impede successful realization of the centralized single-agency model. For example, the Environmental Protection Agency (EPA) holds responsibility for monitoring ambient air quality in partnership with states and municipalities. It fulfills this role adequately, and assigning it to a new centralized agency would not be useful. Furthermore, the establishment of a new federal agency or department would pose additional legislative challenges, such as requiring new legislation and/or appropriations from Congress, which could be time-consuming and politically fraught (Rowell, 2021).
In presenting the centralized single-agency model, the committee considers it a useful starting point for discussion while recognizing that it could not readily be imposed on existing structures, such as those currently overseeing the protection of workers from inhalation hazards. For the public, it is a potential approach.
The Interagency Task Force Model
At the other end of the continuum of centralization, a coalition of stakeholders could be charged with working collaboratively to serve as the coordinating entity. This same model could be used for carrying out specific functions on an ongoing or ad hoc basis. One advantage of the interagency task force model is the relative ease with which it could be established. The temporal/ad hoc nature of this model makes it suitable for narrowly scoped initiatives, such as the COVID-19 Health Equity Task Force, whose charge is “to identify and eliminate health and social disparities that result
in disproportionately higher rates of exposure, illness, hospitalization and death related to COVID-19” (HHS, 2021a).
On the other hand, task forces that bring together multiple agencies face challenges that could impede their performance. According to the Government Accountability Office (GAO) (2010), these challenges include the following:
- Developing and implementing overarching strategies can be hindered by the lack of information on roles and responsibilities of organizations involved or coordination mechanisms. Additionally, incorporating information drawn from multiple sources poses challenges with respect to managing and integrating that information.
- Organizational differences, including differences in agencies’ structures, planning processes, and funding sources, can hinder interagency collaboration. Agencies lack adequate coordination mechanisms to facilitate this collaboration during the planning and execution of programs and activities.
- Agencies involved in such a task force do not always have the right people with the right skills in the right jobs at the right time to meet the challenges they face, including having a workforce able to rapidly address crises. Moreover, agency performance management systems often do not recognize or reward interagency collaboration, and training is needed to understand other agencies’ processes or cultures.
Additionally, in contrast with the mission of a transient interagency task force charged with carrying out an assigned task for a defined period of time and then disbanding, the activities associated with Functions F0 to F5 in the committee’s framework must be carried out on a continuous basis by the stakeholders involved. This important requirement featured in the committee’s deliberations on the interagency task force model as a potential model for the proposed coordinating entity. Additionally, empowerment and accountability within the task force model could be problematic.
The Hub-and-Spoke Model
This model moves away from the interagency task force model toward an empowered coordinating entity. In the hub-and-spoke model, the coordinating entity serves as the hub that is empowered to harness and build on the expertise and experience of current stakeholders engaged in respiratory protection for carrying out all the activities within Functions F0 through F5 (the spokes). However, issues could still arise in joint decision making
under this model, such as when consensus among stakeholders cannot be achieved.
The committee found a precedent for such an architecture in the Public Health Emergency Medical Countermeasures Enterprise (PHEMCE), which “coordinates Federal efforts to enhance chemical, biological, radiological and nuclear threats (CBRN) and emerging infectious diseases (EID) preparedness from a medical countermeasure (MCM) perspective” (HHS, 2021b). The central hub in the PHEMCE is the Department of Health and Human Services’ (HHS’s) Office of the Assistant Secretary for Preparedness and Response (ASPR). Partner agencies include three HHS internal operating divisions—the Centers for Disease Control and Prevention (CDC), the Food and Drug Administration (FDA), and the National Institutes of Health (NIH)—as well as the Department of Defense (DoD), the Department of Veterans Affairs (VA), the Department of Homeland Security (DHS), and the U.S. Department of Agriculture (USDA). The PHEMCE also works closely with a variety of nonfederal partners, including state, local, tribal, and territorial governments; public health systems; academia; and private industry (ASPR, 2015). It focuses on the entire continuum from preparedness against potential threats to effective deployment of medical countermeasures should such threats eventuate. In adopting this broad and overarching concept of an “enterprise” and constituting a “core” group of agencies, the PHEMCE has a coordinating role, led by ASPR, with the authority and responsibility for achieving the overall mission while harnessing the strengths and capabilities of current organizations for the various activities necessary to realize its mission. For each mission component under the PHEMCE, a lead HHS agency with the necessary domain expertise and regulatory authority has been identified and assigned responsibility for that activity. For instance, ASPR is responsible for the “requirements setting” component, while FDA is responsible for “regulatory science management” (ASPR, 2015).
Thus, the PHEMCE provides a real-world example of a coordinating structure that could meet the need to address gaps in oversight related to respiratory protection. A coordinating structure with an architecture similar to that of the PHEMCE model could be well suited to drawing on the domain expertise, experience, and regulatory authority of the stakeholders enumerated in Table 5-2.
Successful Realization and Operation of the Coordinating Entity: The Essential Attributes
In considering the above three examples of potential architectures for a coordinating entity, the committee sought to identify essential attributes for the successful realization and operationalization of any such structure.
Box 5-1 summarizes the key takeaway messages gathered from current and former federal officials during a public meeting held by the committee (see Appendix A), which capture factors critical to the success of the coordinating entity whether it be focused on workers or the public. First, it is essential to articulate clearly the policy goal or vision for the entity, which will need to be supported at the highest levels, potentially including the White House and the Congress, to empower and guide the coordinating entity in carrying out its activities. The success of the leadership for the coordinating entity would be defined by its ability to establish a strong culture of collaboration and partnerships with other stakeholders in the respiratory protection enterprise to harness their domain-specific expertise. To that end, it will be essential for the entity to have the necessary level of authority and leadership commitment. Resources—financial, human, and infrastructure—will also be critical to ensuring that the coordinating entity can carry out its functions. The coordinating entity will need as well to ensure that decision making is based on a strong science, engineering, and technology foundation that guides its operations; therefore, the entity will
need to support evidence-based activities focused on preparedness, innovation, and advances for the future.
It will be important for the coordinating entity’s organizational structure to bring together different perspectives—e.g., product users and manufacturers, regulators, research institutions—in developing and implementing an overall strategy that encompasses the product lifecycle (see Figure 5-1) while embracing the guiding principles listed in Chapter 1. The entity will need to establish and sustain relationships at the federal, state, and local government levels so the developed policies, practices, and guidance can be seamlessly implemented to reach workers and the public. Well-defined channels of communication will be essential to complement these relationships so that the messaging is consistent, coordinated, current, and responsive at all stages of the information and implementation chain. These essential factors, along with regulatory, operational, and organizational considerations related to forming such an entity, need to be taken into account in considering different models for a coordinating entity.
Framework Requirements Concerning Diversity and Equity
As stated in Chapter 1, the committee believes strongly that its framework must incorporate equity as an overarching principle. This principle needs to be considered and addressed by the coordinating entity and other participants in the framework enterprise across each of the functions, as follows:
- Function F0: Develop and approve respiratory protective devices. Devices need to reflect the age range, gender, facial profiles, racial and ethnic diversity, and cognitive and physical abilities of potential users—both workers and the public.
- Function F1: Assess hazard and determine need for respiratory protection. Hazards need to be monitored with consideration of the range of exposures and the increased likelihood of exposures among certain groups.
- Function F2: Determine the necessary respiratory protective devices. Devices need to be available that match both the hazard and the populations that will use them, including their needs and resources.
- Function F3: Ensure availability and access pathways for respiratory protective devices. Access to devices needs to be ensured for all populations, addressing the challenges posed by some locations (e.g., rural areas subject to wildfire smoke, dense urban areas) and the socioeconomic status of subpopulations.
- Function F4: Engage, inform, and ensure access for the target community. Strategies for reaching target communities and ensuring access to respiratory protective devices need to address diversity and local context in all dimensions of communication: modality, language, cultural appropriateness, and socioeconomic and educational background, for example. The challenge of communication needs to be considered prospectively to ensure preparedness.
- Function F5: Incorporate lifecycle learning and continuous improvement. This critical aspect of ensuring effective functioning of the framework needs to be sufficiently broad in its reach to provide information on major and high-risk subgroups.
Consistent with its overarching nature, the principle of equity is woven throughout the discussion of the frameworks for workers not covered by RPPs and the public in Chapters 6 and 7, respectively.
REFERENCES
ASPR (Office of the Assistant Secretary for Preparedness and Response). 2015. PHEMCE mission components. https://www.phe.gov/Preparedness/mcm/phemce/Pages/mission.aspx (accessed September 21, 2021).
Brosseau, L. M., J. Rosen, and R. Harrison. 2020. Selecting controls for minimizing SARSCoV-2 aerosol transmission in workplaces and conserving respiratory protective equipment supplies. Annals of Work Exposures and Health 65(1):53-62.
CRS (Congressional Research Service). 2021. The PREP Act and COVID-19: Limiting liability for medical countermeasures. Updated September 23, 2021. https://crsreports.congress.gov/product/pdf/LSB/LSB10443 (accessed September 29, 2021).
GAO (Government Accountability Office). 2010. Key challenges and solutions to strengthen interagency collaboration. https://www.gao.gov/assets/gao-10-822t.pdf (accessed September 29, 2021).
HHS (Department of Health and Human Services). 2021a. About COVID-19 Health Equity Task Force. https://www.minorityhealth.hhs.gov/omh/browse.aspx?lvl=2&lvlid=100 (accessed September 28, 2021).
HHS. 2021b. Public health emergency medical countermeasures enterprise. https://www.phe.gov/Preparedness/mcm/phemce/Pages/default.aspx (accessed September 28, 2021).
IOM (Institute of Medicine). 2008. Preparing for an influenza pandemic: Personal protective equipment for healthcare workers. Washington, DC: The National Academies Press.
IOM. 2011. Certifying personal protective technologies: Improving worker safety. Washington, DC: The National Academies Press.
Kochenderfer, M.J. 2015. Decision making under uncertainty: Theory and application. Cambridge, MA: The MIT Press.
McCullough, N.V. 2021. Presentation at the January 26, 2021, meeting of the National Academies of Sciences, Engineering, and Medicine Committee on Respiratory Protection for the Public and Workers Without Respiratory Protection Programs at Their Workplaces, virtual. https://www.nationalacademies.org/event/01-25-2021/respiratory-protection-for-the-public-and-workers-without-respiratory-protection-programs-at-their-workplaces-committee-meeting-3 (accessed Septmeber 28, 2021).
NIOSH (National Institute for Occupational Safety and Health). 2020. Certified equipment list. https://www.cdc.gov/niosh/npptl/topics/respirators/cel/default.html (accessed September 28, 2021).
Rowell, A. 2021. The regulatory landscape regarding respiratory protection. Paper commissioned by the Committee on the Respiratory Protection for the Public and Workers Without Respiratory Protection Programs at Their Workplaces (see Appendix C).