Appendix D
Evidence Review: Methods and Approach
This appendix describes the approach and methods that the committee used to address the portions of the Statement of Task that asked for “an objective and authoritative review of current evidence regarding human health effects of those PFAS [per- and polyfluoroalkyl substances] being monitored in the CDC’s [Centers for Disease Control and Prevention’s] National Report on Human Exposure to Environmental Chemicals” (see Box 1-3 in Chapter 1). The Statement of Task specifically asked the committee to:
Assess the strength of evidence for the spectrum of putative health effects suggested by human studies (including immune response, lipid metabolism, kidney function, thyroid disease, liver disease, glycemic parameters and diabetes, cancer, and fetal and child development) to establish a basis for prioritized clinical surveillance or monitoring of PFAS health effects. This assessment should characterize the likelihood of those health effects occurring (qualitative probability) given real world human exposures and identify the human populations at most risk (consider life stage, health status, exposure level). Data/evidence gaps that contribute to uncertainty about health effects of most concern should be annotated.
The committee decided that this portion of the Statement of Task required three different determinations:
- qualitative categories that describe the strength of evidence of PFAS putative health effects that can be used to prioritize clinical surveillance or monitoring,
- identification of the human populations at most risk (considering life stage, health status, exposure level) from PFAS exposure, and
- a scoping review that maps the data or evidence gaps that contribute to uncertainty about health effects of most concern.
To produce these outputs, the committee developed a multistage process. The first stage was to catalog what is known about PFAS and their health effects. The committee identified all authoritative reviews of the PFAS identified by the CDC (see Table 1-1 in Chapter 1) and all human health outcomes. The second stage was to identify any recent, high-quality systematic reviews between PFAS and any human health outcome; as noted below, the results of this stage were uninformative for the committee’s goals. The third stage was to review the published research articles describing the association between exposure to PFAS and human health outcomes, based on the authoritative and systematic reviews. The committee’s review approach improved efficiency while minimizing the risk of excluding scientific findings that would inform the committee’s recommendations.
The committee was charged with assessing
the strength of evidence for the spectrum of putative health effects suggested by human studies (including immune response, lipid metabolism, kidney function, thyroid disease, liver disease, glycemic parameters and diabetes, cancer, and fetal and
child development) to establish a basis for prioritized clinical surveillance or monitoring of PFAS health effects.
However, the committee did not restrict its evaluation to only the listed putative health effects.
The next section of the appendix covers the committee’s analysis of the authoritative reviews; the following section covers its original literature review, and the final section covers the committee strength-of-evidence determination.
AUTHORITATIVE REVIEWS
The committee defined authoritative reviews to be reviews produced by government agencies or other bodies that publish strength-of-evidence determinations through a process that includes peer review. The committee focused on national or international organizations or agencies that influence other organizations. The following organizations met these criteria for authoritative reviews:
- C-8 Science Panel Reports
- European Food Safety Authority (EFSA)
- International Agency for Research on Cancer (IARC)
- Organisation for Economic Co-operation and Development (OECD)
- National Toxicology Program (NTP)
- Agency for Toxic Substances and Disease Registry (ATSDR)
- U.S. Environmental Protection Agency (EPA)
Table D-1 summarizes the authoritative reviews found by the committee. Among the authoritative reviews, the ATSDR’s Toxicological Profile for Perfluoroalkyls included the greatest number of PFAS included in the committee’s Statement of Task (MeFOSAA not included) and was the most recent (literature search conducted in September 2018). The other authoritative reviews were older and included chemicals that were also included in the ATSDR’s Toxicological Profile for Perfluoroalkyls. Therefore, the ATSDR’s Toxicological Profile for Perfluoroalkyls was used by the committee as the basis for the next stages of the review process.
TABLE D-1 Authoritative Reviews Found by the Committee
| Review | PFAS Chemicals Covered in Review | Health Endpoints Covered in Review | Date of Last Literature Search by the Organization |
|---|---|---|---|
| ATSDR Toxicological Profile for Perfluoroalkyls | PFBA, PFHxA, PFHpA, PFOA, PFNA, PFDA, PFUnA, PFDoDA, PFBS, PFHxS, PFOS, FOSA | Not limited | September 2018 |
| EFSA Risk to Human Health Related to the Presence of Perfluoroalkyl Substances in Food | PFOA, PFNA, PFHxS, PFOS | Fertility and pregnancy outcomes, development effects, neurotoxic outcomes, immune outcomes, endocrine effects, metabolic effects, kidney function, cardiovascular disease and mortality, bone mineral density | March 2013 |
| EPA Health Effects Document (PFOA) | PFOA | Serum lipids, cardiovascular disease, liver disease, kidney disease, diabetes, developmental toxicity, thyroid effects, immunotoxicity, cancer: kidney and testicular cancer, neurotoxicity, steroid hormones | 2015 |
| EPA Health Effects Document (PFOS) | PFOS | Serum lipids, cardiovascular disease, liver disease, kidney disease, diabetes, | 2015 |
| developmental toxicity, thyroid effects, immunotoxicity, cancer: testicular and kidney, neurotoxicity, steroid hormones | |||
| IARC Monograph | PFOA | Cancer | June 2014 |
| NTP Monograph | PFOA, PFOS | Immunotoxicity | May 2016 |
| OECD Synthesis Paper | PFOA, PFOS, PFHxS, PFNA, PFDA, PFBS, PFBA, PFHxA, PTFE, PVDF, PFBE | Developmental toxicity, hypocholesteremia, ulcerative colitis, thyroid diseases, testicular cancer, kidney cancer, preeclampsia | Not presented, published 2013 |
| C-8 Science Panel Probable Link Reportsa | PFOA | Heart disease, kidney disease, liver disease, osteoarthritis, Parkinson’s disease, autoimmune disease, infectious disease, neurodevelopmental disorders in children, respiratory disease, evaluation of stroke, thyroid disease, cancer, diabetes, birth defects, pregnancy-induced hypertension, miscarriage and stillbirth, preterm birth and low birthweight | Last report published in 2012 |
a See http://www.c8sciencepanel.org/prob_link.html (accessed July 1, 2022).
NOTE: ATSDR = Agency for Toxic Substances and Disease Registry; EFSA = European Food Safety Authority; EPA = U.S. Environmental Protection Agency; IARC = International Agency for Research on Cancer; NTP = National Toxicology Program; OECD = Organisation for Economic Co-operation and Development.
The committee’s review did not assess the quality of the authoritative reviews, but it notes several areas where the ATSDR’s Toxicological Profile for Perfluoroalkyls could be strengthened. First, the toxicological profile does not provide a detailed description of the evidence identification methods and does not document decisions as to why specific studies may have been excluded. Second, the study quality assessment does not appear to follow a standard approach, and in some cases it is difficult to identify the study designs that were included in the review. Third, the process to assess the strength of the evidence is not always clear.
REVIEW OF SYSTEMATIC REVIEWS
The committee’s review of systematic reviews consisted of the following steps: literature search, screening of abstracts, full text review of studies identified in the abstract screening, evaluation of a final set of relevant studies, evidence assessment, and synthesis.
Literature Search
Systematic reviews were identified through searches of the medical and scientific literature on three databases: Embase Update, Medline, and Scopus. These three searchable databases index biological, chemical, medical, and toxicological publications. Search terms included full and abbreviated chemical names, common and manufacturer trade names, Chemical Abstracts Service (CAS) numbers, and MeSH1 descriptors for each of the PFAS species of interest. Systematic reviews were included regardless of when they were published or where they were conducted. Systematic reviews were considered if they reviewed human studies, were classified as review papers, and were published in English. The databases were searched on June 28, 2021.
___________________
1 MeSH descriptors are sets of terms naming descriptors in a hierarchical structure that permits searching at various levels of specificity.
Ovid Embase Update Search Terms
| 1 | (“335-76-2” or “335-67-1” or “375-92-8” or “375-95-1” or “355-46-4” or “1763-23-1” or “2058-94-8”).rn. | 3673 |
|---|---|---|
| 2 | Limit 1 to (human and English language and “review”) | 154 |
| 3 | exp perfluorooctanesulfonic acid/ or exp perfluorohexanesulfonic acid/ or exp perfluorononanoic acid/ or exp perfluorooctanesulfonic acid/ or exp perfluorodecanoic acid/ or exp perfluorooctanoic acid/ or exp perfluoroundecanoic acid/ or (“Methyl-perfluorooctane sulfonamide” or “Methylperfluorooctane sulfonamidoacetic acid” or “Perfluorodecanoic acid” or “Perfluoroheptanesulfonic acid” or “perfluorohexane sulfonic acid” or “Perfluorohexanesulfonic acid” or “Perfluorononanoic acid” or “Perfluorooctane sulfonic acid” or “Perfluorooctanesulfonic acid” or “Perfluorooctanoic acid” or “perfluoroundecanoic acid”).mp. or (“MeFOSAA” or “PFHxS” or “n-PFOA” or “Sb-PFOA” or “PFOA” or “PFDA” or “PFUnDA” or “n-PFOS” or “Sm-PFOS” or “PFOS” or “PFNA” or “Perfluorinated chemical” or “perfluorinated compound” or “perfluorinated chemicals” or “perfluorinated compounds”).mp. | 7814 |
| 4 | Limit 3 to (human and English language and “review”) | 352 |
MEDLINE Search Terms
| 1 | (“335-76-2” or “335-67-1” or “375-92-8” or “375-95-1” or “355-46-4” or “1763-23-1” or “2058-94-8”).rn. | 331 |
|---|---|---|
| 2 | Limit 1 to (English language and “review articles” and humans) | 1 |
| 3 | Limit 1 to (English language humans and “review” or “scientific integrity review” or “systematic review”) | 1 (same article) |
| 4 | (“Methyl-perfluorooctane sulfonamide” or “Methylperfluorooctane sulfonamidoacetic acid” or “Perfluorodecanoic acid” or “Perfluoroheptanesulfonic acid” or “perfluorohexane sulfonic acid” or “Perfluorohexanesulfonic acid” or “Perfluorononanoic acid” or “Perfluorooctane sulfonic acid” or “Perfluorooctanesulfonic acid” or “Perfluorooctanoic acid” or “perfluoroundecanoic acid”).mp. | 4159 |
| 5 | Limit 4 to (English language and “review articles” and humans) | 77 |
| 6 | (“MeFOSAA” or “PFHxS” or “n-PFOA” or “Sb-PFOA” or “PFOA” or “PFDA” or “PFUnDA” or “n-PFOS” or “Sm-PFOS” or “PFOS” or “PFNA” or “Perfluorinated chemical” or “perfluorinated compound” or “perfluorinated chemicals” or “perfluorinated compounds”).mp. | 5852 |
| 7 | Limit 6 to (English language and “review articles” and humans) | 192 |
| 8 | Limit 4 to (English language humans and “review” or “scientific integrity review” or “systematic review”) | 81 |
| 9 | Limit 6 to (English language humans and “review” or “scientific integrity review” or “systematic review”) | 203 |
| 10 | (“Methyl-perfluorooctane sulfonamide” or “Methylperfluorooctane sulfonamidoacetic acid” or “Perfluorodecanoic acid” or “Perfluoroheptanesulfonic acid” or “perfluorohexane sulfonic acid” or “Perfluorohexanesulfonic acid” or “Perfluorononanoic acid” or “Perfluorooctane sulfonic acid” or “Perfluorooctanesulfonic acid” or “Perfluorooctanoic acid” or “perfluoroundecanoic acid”).rn. | 2896 |
| 11 | Limit 10 to (English language and “review articles” and humans) | 49 |
| 12 | Limit 10 to (English language humans and “review” or “scientific integrity review” or “systematic review”) | 51 |
Scopus Search Terms
( ( ( TITLE-ABS-KEY ( ”MeFOSAA” OR “PFHxS” OR “n-PFOA” OR “Sb-PFOA” OR “PFOA” OR “PFDA” OR “PFUnDA” OR “n-PFOS” OR “Sm-PFOS” OR “PFOS” OR “PFNA” OR “Perfluorinated chemical*” OR “perfluorinated compound*” ) ) OR ( TITLE-ABS-KEY ( ”Methylperfluorooctane sulfonamide” OR “Methylperfluorooctane sulfonamidoacetic acid” OR “Perfluorodecanoic acid” OR “Perfluoroheptanesulfonic acid” OR “perfluorohexane sulfonic acid”
OR “Perfluorohexanesulfonic acid” OR “Perfluorononanoic acid” OR “Perfluorooctane sulfonic acid” OR “Perfluorooctanesulfonic acid” OR “Perfluorooctanoic acid” OR “perfluoroundecanoic acid” ) ) OR ( CHEMNAME ( ”Methyl-perfluorooctane sulfonamide” OR “Methylperfluorooctane sulfonamidoacetic acid” OR “Perfluorodecanoic acid” OR “Perfluoroheptanesulfonic acid” OR “perfluorohexane sulfonic acid” OR “Perfluorohexanesulfonic acid” OR “Perfluorononanoic acid” OR “Perfluorooctane sulfonic acid” OR “Perfluorooctanesulfonic acid” OR “Perfluorooctanoic acid” OR “perfluoroundecanoic acid” ) ) OR ( CASREGNUMBER ( ”335-76-2” OR “335-67-1” OR “375-92-8” OR “375-95-1” OR “355-46-4” OR “1763-23-1” OR “2058-94-8” ) ) ) AND ( INDEXTERMS ( human* ) OR TITLE-ABS-KEY ( human* ) ) ) AND NOT INDEX ( medline ) AND ( LIMIT-TO (“ar” ) ) AND ( LIMIT-TO ( LANGUAGE , “English” ) )
Screening of Abstracts
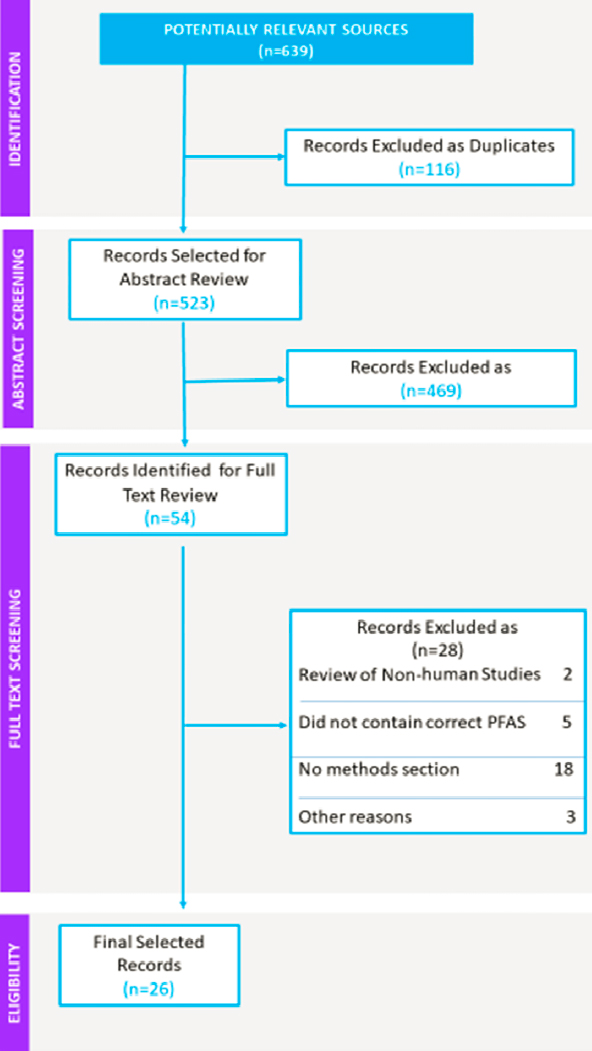
The literature search identified 639 potentially relevant systematic reviews. The publications were imported into PICO Portal, a web-based tool for collaborative citation screening for systematic reviews.2 After importing to PICO Portal, 119 articles were identified as duplicates, leaving 520 for title and abstract screening. The review used the following population exposure comparison and outcome (PECO) statement:
The inclusion and exclusion criteria related to the PECO statement were as follows:
Title and abstract screening was completed by two screeners. Disagreements were resolved by an adjudicator who helped facilitate a consensus decision. Fifty-four articles were included for full-text review.
Full Text Review
For the full text review, articles were excluded because they did not include human studies (n = 2), did not include relevant PFAS (n = 5), did not have a methods section (n = 18), or did not evaluate the association of PFAS exposure with a human health outcome (n = 3) (see Figure D-1). Thus, the full text review covered 26 articles.
___________________
2 See https://picoportal.net (accessed July 1, 2022).
Evaluation and Evidence Assessment
The quality of the 26 included systematic reviews was evaluated using the AMSTAR-2 (A MeaSurement Tool to Assess systematic Reviews) (Shea et al., 2017), which has been used by several other committees of the National Academies of Sciences, Engineering, and Medicine (NASEM, 2019, 2021). AMSTAR-2 includes several critical appraisal domains (see Box D-1). The quality assessment was conducted by a staff member and was confirmed by a committee member. The committee conducted a critical appraisal of the systematic reviews because systematic reviews can be subject to a range of biases.
Synthesis: Results
Of the 26 systematic reviews identified by the committee, 9 included studies published after the ATSDR’s Toxicological Profile for Perfluoroalkyls (Bartell and Vieira, 2021; Boesen et al., 2020; Deji et al., 2021; Dzierlenga et al., 2020; Ferrari et al., 2019; Luo et al., 2020; Petersen et al., 2020; Steenland and Winquist, 2020; Xie et al., 2020; Zare Jeddi et al., 2021). Although all were of moderate quality, some reviews covered the same original data and papers, and the committee found it challenging to synthesize across them. As a result, the systematic reviews were used as sources for reference in the committee’s determination of the biologic plausibility between PFAS and a health effect, but they were not formally included as part of the final strength-of-evidence determination.
ORIGINAL LITERATURE REVIEW
The original literature review consisted of the following steps: literature search, screening of abstracts, full text review of studies identified in the abstract screening, evidence mapping and evaluation, and data abstraction.
Literature Search
As was done for the review of reviews, the literature for the original literature review was identified through searches of the medical and scientific literature on three databases: Ovid Embase, Ovid Medline, and Scopus. These three searchable databases index biological, chemical, medical, and toxicological publications. Search terms included full and abbreviated chemical names, common and
manufacturer trade names, the CAS numbers, and MeSH3 descriptors for each of the PFAS species of interest. There were no time constraints or geographic constraints included in the search. The only constraints were human studies and English language only. The databases were searched on March 30–31, 2021.
Ovid Embase Search Terms4
| 1 | (“335-76-2” or “335-67-1” or “375-92-8” or “375-95-1” or “355-46-4” or “1763-23-1” or “2058-94-8”).rn. | 3547 |
|---|---|---|
| 2 | exp perfluorooctanesulfonic acid/ | 3114 |
| 3 | exp perfluorohexanesulfonic acid/ | 625 |
| 4 | exp perfluorononanoic acid/ | 890 |
| 5 | exp perfluorooctanesulfonic acid/ | 3114 |
| 6 | exp perfluorodecanoic acid/ | 713 |
| 7 | exp perfluorooctanoic acid/ | 3493 |
| 8 | exp perfluoroundecanoic acid/ | 386 |
| 9 | (“Methyl-perfluorooctane sulfonamide” or “Methylperfluorooctane sulfonamidoacetic acid” or “Perfluorodecanoic acid” or “Perfluoroheptanesulfonic acid” or “perfluorohexane sulfonic acid” or “Perfluorohexanesulfonic acid” or “Perfluorononanoic acid” or “Perfluorooctane sulfonic acid” or “Perfluorooctanesulfonic acid” or “Perfluorooctanoic acid” or “perfluoroundecanoic acid”).mp. | 5503 |
| 10 | (“MeFOSAA” or “PFHxS” or “n-PFOA” or “Sb-PFOA” or “PFOA” or “PFDA” or “PFUnDA” or “n-PFOS” or “Sm-PFOS” or “PFOS” or “PFNA” or “Perfluorinated chemical” or “perfluorinated compound” or “perfluorinated chemicals” or “perfluorinated compounds”).mp. | 6579 |
| 11 | 1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 or 10 | 7610 |
| 12 | (exp animal/ or nonhuman/) not exp human/ | 6205240 |
| 13 | 11 not 12 | 5435 |
| 14 | Limit 13 to english language | 5232 |
| 15 | Limit 14 to “pubmed/medline” | 540 |
| 16 | 14 not 15 | 4692 |
| 17 | Limit 16 to article | 3517 |
Scopus Search Terms5
( ( ( TITLE-ABS-KEY ( ”MEFOSAA” OR “PFHXS” OR “N-PFOA” OR “SB-PFOA” OR “PFOA” OR “PFDA” OR “PFUNDA” OR “N-PFOS” OR “SM-PFOS” OR “PFOS” OR “PFNA” OR “PERFLUORINATED CHEMICAL*” OR “PERFLUORINATED COMPOUND*” ) ) OR ( TITLE-ABS-KEY ( ”METHYL-PERFLUOROOCTANE SULFONAMIDE” OR “METHYLPERFLUOROOCTANE SULFONAMIDOACETIC ACID” OR “PERFLUORODECANOIC ACID” OR “PERFLUOROHEPTANESULFONIC ACID” OR “PERFLUOROHEXANE SULFONIC ACID” OR “PERFLUOROHEXANESULFONIC ACID” OR “PERFLUORONONANOIC ACID” OR “PERFLUOROOCTANE SULFONIC ACID” OR “PERFLUOROOCTANESULFONIC ACID” OR “PERFLUOROOCTANOIC ACID” OR “PERFLUOROUNDECANOIC ACID” ) ) OR ( CHEMNAME ( ”METHYL-PERFLUOROOCTANE SULFONAMIDE” OR “METHYLPERFLUOROOCTANE SULFONAMIDOACETIC ACID” OR “PERFLUORODECANOIC ACID” OR “PERFLUOROHEPTANESULFONIC ACID” OR “PERFLUOROHEXANE SULFONIC ACID” OR “PERFLUOROHEXANESULFONIC ACID” OR “PERFLUORONONANOIC ACID” OR
___________________
3 MeSH descriptors are sets of terms naming descriptors in a hierarchical structure that permits searching at various levels of specificity.
4 Not all chemical names mapped to a heading.
5 The search was limited to articles and English language.
“PERFLUOROOCTANE SULFONIC ACID” OR “PERFLUOROOCTANESULFONIC ACID” OR “PERFLUOROOCTANOIC ACID” OR “PERFLUOROUNDECANOIC ACID” ) ) OR ( CASREGNUMBER ( ”335-76-2” OR “335-67-1” OR “375-92-8” OR “375-95-1” OR “355-46-4” OR “1763-23-1” OR “2058-94-8” ) ) ) AND ( INDEXTERMS ( HUMAN* ) OR TITLE-ABS-KEY ( HUMAN* ) ) ) AND NOT INDEX ( MEDLINE ) AND ( LIMIT-TO (“AR” ) ) AND ( LIMIT-TO ( LANGUAGE , “ENGLISH” ) )
Ovid Medline Search Terms
| 1 | (“335-76-2” or “335-67-1” or “375-92-8” or “375-95-1” or “355-46-4” or “1763-23-1” or “2058-94-8”).rn. | 330 |
|---|---|---|
| 2 | (“Methyl-perfluorooctane sulfonamide” or “Methylperfluorooctane sulfonamidoacetic acid” or “Perfluorodecanoic acid” or “Perfluoroheptanesulfonic acid” or “perfluorohexane sulfonic acid” or “Perfluorohexanesulfonic acid” or “Perfluorononanoic acid” or “Perfluorooctane sulfonic acid” or “Perfluorooctanesulfonic acid” or “Perfluorooctanoic acid” or “perfluoroundecanoic acid”).mp. | 4050 |
| 3 | (“MeFOSAA” or “PFHxS” or “n-PFOA” or “Sb-PFOA” or “PFOA” or “PFDA” or “PFUnDA” or “n-PFOS” or “Sm-PFOS” or “PFOS” or “PFNA” or “Perfluorinated chemical” or “perfluorinated compound” or “perfluorinated chemicals” or “perfluorinated compounds”).mp. | 5700 |
| 4 | (“Methyl-perfluorooctane sulfonamide” or “Methylperfluorooctane sulfonamidoacetic acid” or “Perfluorodecanoic acid” or “Perfluoroheptanesulfonic acid” or “perfluorohexane sulfonic acid” or “Perfluorohexanesulfonic acid” or “Perfluorononanoic acid” or “Perfluorooctane sulfonic acid” or “Perfluorooctanesulfonic acid” or “Perfluorooctanoic acid” or “perfluoroundecanoic acid”).rn. | 2837 |
| 5 | 1 or 2 or 3 or 4 | 6194 |
| 6 | Animals/ not (Animals/ and Humans/) | 4772259 |
| 7 | 5 not 6 | 4664 |
| 8 | Journal Article/ | 30061095 |
| 9 | 7 and 8 | 4548 |
| 10 | Limit 9 to English language | 4379 |
Abstract Screening
The literature search identified 5,172 potentially relevant studies. The studies were imported into PICO Portal, a web-based tool for collaborative citation screening for systematic reviews.6 After importing the studies to PICO Portal, 112 articles were identified as duplicates, leaving 5,060 articles to be screened. The titles and abstracts were screened for relevance to the research questions for the review. The review used the following PECO statement:
The inclusion and exclusion criteria related to the PECO statement were as follows:
Inclusion Criteria: Is an epidemiologic or human study; includes a quantitative measure of the PFAS species measured in the CDC’s National Report on Human Exposure to Environmental Chemicals; assesses the evidence for an association of PFAS and a health outcome in humans; and English language only
___________________
6 See https://picoportal.net (accessed July 1, 2022).
Exclusion Criteria: Did not review health effects of PFAS; reviewed only animal or mechanistic studies; was about chemicals other than the PFAS included in the Statement of Task; did not include a quantitative measure; or was on the ecological effects of PFAS or PFAS exposure pathways
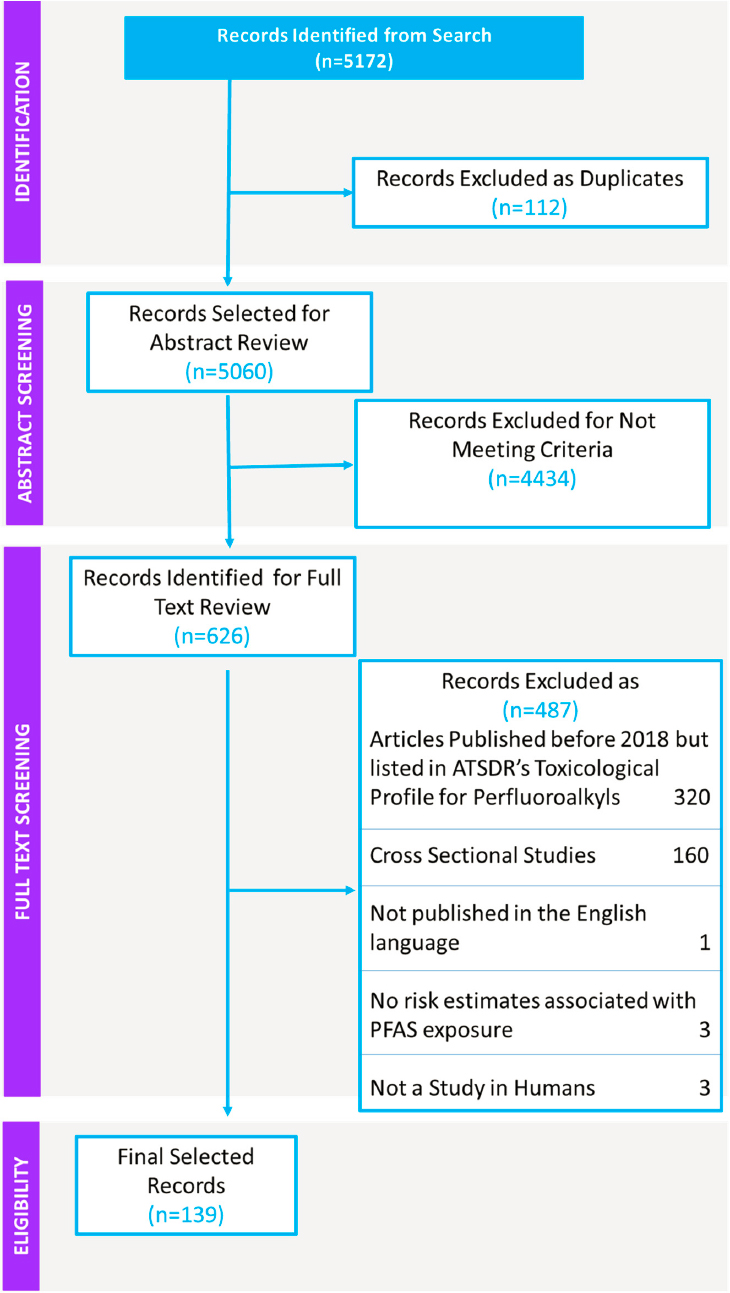
The literature search identified 5,172 potentially relevant studies. After removal of duplicates (112 articles), 5,060 articles were subject to title and abstract screening by two independent reviewers.
Full Text Review
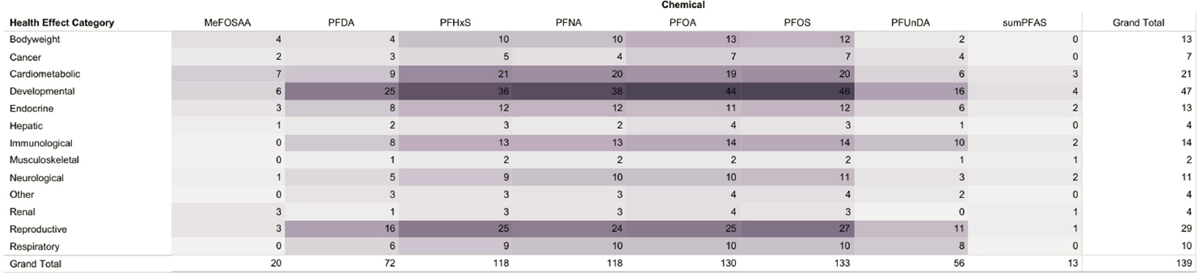
For the full text review, 4,434 of the articles identified in the literature search were excluded because the titles and abstracts did not meet the inclusion criteria, so 626 articles were subject to full text review. During that review, additional articles were excluded if they were published before 2018 or listed in the references to the ATSDR’s Toxicological Profile for Perfluoroalkyls (n = 320); were cross-sectional in design (n = 160); were not published in English (n = 1); did not provide risk estimates associated with PFAS exposure (n = 3); or were not studies in humans (n = 3) (see Figure D-2). Cross-sectional studies were largely excluded because this study design measures exposure and disease at the same time so cannot determine cause and effect. Thus, the full text review covered 139 articles.
Evidence Mapping and Evaluation
The committee then categorized the 139 articles according to the human health outcomes studied. The committee mapped the evidence with the goal of determining evidence gaps and to inform strategies for the evidence evaluation and evidence synthesis (see Figure D-3).
The committee focused on those endpoints for which additional review might change the committee’s understanding of the association between PFAS exposure and health outcomes.
The committee conducted a narrative evaluation of the study quality and considered factors that may contribute to the study’s risk of bias (see Box D-2) using a tool adapted from the Navigation Guide (Woodruff and Sutton, 2014). Bias is a systematic error that leads to study results that differ from the actual results. Bias can lead to an observed effect when one does not exist or to no observed effect when there is a true effect. Risk of bias is the appropriate term, as a study may be unbiased despite a methodological flaw (Higgins et al., 2019). The risk-of-bias assessment in a systematic review is based on the quality of the individual component studies (Eick et al., 2020).
A trained reviewer from ICF International, the EPA, the National Academies, or Johns Hopkins University abstracted the critical domain information from each study that the committee used to support its judgment determinations regarding a study’s risk of bias, and the committee made the final risk of bias judgments for each study.7 Each paper was given an overall assessment of its risk of bias (low, probably low, probably high, or high risk of bias).
___________________
7 For some studies included in the committee’s review, the data had been previously abstracted by ICF or the EPA to support the EPA’s ongoing assessments of PFAS; newer evidence was abstracted by ICF or consultants at Johns Hopkins.
Data Abstraction
Data abstraction was completed by a trained scientist from ICF, the EPA, the National Academies, or Johns Hopkins.8 The data abstraction form included the following:
___________________
8 For some studies included in the committee’s review, the data had been previously abstracted by ICF or the EPA to support the EPA’s ongoing assessments of PFAS; newer evidence was abstracted by ICF or consultants at Johns Hopkins University.
Effect estimates from the individual studies included in the review were extracted into a database and uploaded to a public website (Tableau Public) to allow for visualizations, such as evidence maps and forest plots.9 The effect estimates in the Tableau represent those from the model most adjusted for confounders.
STRENGTH-OF-EVIDENCE DETERMINATION
To assess the strength of evidence regarding the potential for PFAS to cause a particular health effect, the committee then integrated the evidence reviewed in the ATSDR’s Toxicological Profile for Perfluoroalkyls and other authoritative reviews with the evidence from the original literature review from the epidemiologic studies.
The synthesis of available data was guided by a framework based on the Hill considerations (Hill, 1965), which help to determine whether associations are causal (see Box D-3). The committee did not consider the Hill considerations to be a heuristic for assessing causation in isolation, that is, as a checklist where each item must be met to establish causality. Rather, the committee considered them as a list of possible considerations meant to generate thoughtful discourse by the committee to help inform its determinations for the strength of evidence (Fedak, 2015; NASEM, 2018).
___________________
9 The committee’s public Tableau is available at https://public.tableau.com/app/profile/nationalacademies/viz/NASEMPFASEvidenceMaps/PFASEvidenceMap (accessed July 1, 2022). The information may be viewed as an evidence map or as a forest plot. Within forest plots, filters can be accessed using the “toggle filters” function in order to restrict the view to data on specific health effect categories and other factors (such as reference, chemical, study design, study population).
Animal and Mechanistic Studies
The Statement of Task called for the committee to “assess the strength of evidence for the spectrum of putative health effects suggested by human studies.” The committee focused on more recent epidemiological literature in line with its Statement of Task. However, the committee recognizes the importance of toxicologic evidence in making strength-of-evidence conclusions and relied on the toxicological data included in the ATSDR’s Toxicological Profile for Perfluoroalkyls, other authoritative reviews, and systematic reviews that provide integrative conclusions based on multiple lines of evidence. In synthesizing evidence in this manner, the committee acknowledges that animal evidence greatly improves the interpretation of the human studies.
An observed association between PFAS exposure and a health effect does not necessarily mean that the exposure is the cause of that outcome. Toxicologic evidence, whether it supports or conflicts with evidence from epidemiologic studies, provides insights about biologic processes and informs how an observed association might be interpreted. The degree of biologic plausibility itself influences whether the committee perceives positive findings to be indicative of a pattern or the product of statistical fluctuations. Ultimately, the results of the toxicology studies should be consistent with what is known about the human disease process if they are to support a conclusion that the development of the disease was influenced by an exposure (NASEM, 2018).
Categories of Association
Sufficient Evidence of an Association
For effects in this category, a positive association between PFAS and the outcome must be observed in studies in which chance, bias, and confounding can be ruled out with reasonable confidence. For example, the committee might regard as sufficient evidence of an association evidence from several small studies that is unlikely to be due to confounding or to otherwise be biased and that shows an association that is consistent in magnitude and direction. Experimental data supporting biologic plausibility strengthen the evidence of an association but are not a prerequisite, nor are they sufficient to establish an association without corresponding epidemiologic findings.
Limited or Suggestive Evidence of an Association
In this category, the evidence must suggest an association between exposure to PFAS and the outcome in studies of humans, but the evidence can be limited by an inability to rule out chance, bias, or confounding with confidence. One high-quality study may indicate a positive association, but the results of other studies of lower quality may be inconsistent.
Inadequate or Insufficient Evidence to Determine an Association
If there was not enough reliable scientific data to categorize the potential association with a health effect as “sufficient evidence of an association,” “limited or suggestive evidence of an association,” or on the other end of the spectrum, “limited or suggestive evidence of no association,” the health outcome was placed in the category of “inadequate or insufficient evidence to determine an association” by default. In this category, the available human studies may have inconsistent findings or be of insufficient quality, validity, consistency, or statistical power to support a conclusion regarding the presence of an association. Such studies may have failed to control for confounding factors or may have had inadequate assessment of exposure.
Limited or Suggestive Evidence of No Association
A conclusion of “no association” is inevitably limited to the conditions, exposures, and observation periods covered by the available studies, and the possibility of a small increase in risk related to the magnitude of exposure studied can never be excluded. However, a change in classification from inadequate or insufficient evidence of an association to limited or suggestive evidence of no association would require new studies that corrected for the methodologic problems of previous studies and that had samples large enough to limit the possible study results attributable to chance.
REFERENCES
Bartell, S. M., and V. M. Vieira. 2021. Critical review on PFOA, kidney cancer, and testicular cancer. Journal of the Air and Waste Management Association 71(6):663–679. https://doi.org/10.1080/10962247.2021.1909668.
Boesen, S. A. H., M. Long, M. Wielsoe, V. Mustieles, M. F. Fernandez, and E. C. Bonefeld-Jorgensen. 2020. Exposure to perflouroalkyl acids and foetal and maternal thyroid status: A review. Environmental Health 19(1):107. https://doi.org/10.1186/s12940-020-00647-1.
Deji, Z., P. Liu, X. Wang, X. Zhang, Y. Luo, and Z. Huang. 2021. Association between maternal exposure to perfluoroalkyl and polyfluoroalkyl substances and risks of adverse pregnancy outcomes: A systematic review and meta-analysis. Science of the Total Environment 783:146984. https://doi.org/10.1016/j.scitotenv.2021.146984.
Dzierlenga, M. W., L. Crawford, and M. P. Longnecker. 2020. Birth weight and perfluorooctane sulfonic acid: A random-effects meta-regression analysis. Environmental Epidemiology 4(3):e095. https://doi.org/10.1097/EE9.0000000000000095.
Eick, S. M., D. E. Goin, N. Chartres, J. Lam, and T. J. Woodruff. 2020. Assessing risk of bias in human environmental epidemiology studies using three tools: Different conclusions from different tools. Systematic Reviews 9(1):249. https://doi.org/10.1186/s13643-020-01490-8.
Fedak, K. M., A. Bernal, Z. A. Capshaw, and S. Gross. 2015. Applying the Bradford Hill criteria in the 21st century: How data integration has changed causal inference in molecular epidemiology. Emerging Themes in Epidemiology 12:14. https://doi.org/10.1186/s12982-015-0037-4.
Ferrari, F., A. Orlando, Z. Ricci, and C. Ronco. 2019. Persistent pollutants: Focus on perfluorinated compounds and kidney. Current Opinion Critical Care 25(6):539–549. https://doi.org/10.1097/MCC.0000000000000658.
Higgins, J. P. T., J. Thomas, J. Chandler, M. Cumpston, T. Li, M. J. Page, and V. A. Welch. 2019. Cochrane Handbook for Systematic Reviews of Interventions. Hoboken, NJ: John Wiley & Sons.
Hill, A. B. 1965. The environment and disease: Association or causation? Proceedings of the Royal Society of Medicine 58(5):295–300.
Luo, Y., Z. Deji, and Z. Huang. 2020. Exposure to perfluoroalkyl substances and allergic outcomes in children: A systematic review and meta-analysis. Environmental Research 191:110145. https://doi.org/10.1016/j.envres.2020.110145.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2018. Advances in Causal Understanding for Human Health Risk-Based Decision-Making: Proceedings of a Workshop—in Brief. Washington, DC: The National Academies Press.
NASEM. 2019. Review of DOD’s Approach to Deriving an Occupational Exposure Level for Trichloroethylene. Washington, DC: The National Academies Press.
NASEM. 2021. The Use of Systematic Review in EPA’s Toxic Substances Control Act Risk Evaluations. Washington, DC: The National Academies Press.
Petersen, K. U., J. R. Larsen, L. Deen, E. M. Flachs, K. K. Haervig, S. D. Hull, J. P. E. Bonde, and S. S. Tottenborg. 2020. Per- and polyfluoroalkyl substances and male reproductive health: A systematic review of the epidemiological evidence. Journal of Toxicology Environmental Health B Critical Reviews 23(6):276–291. https://doi.org/10.1080/10937404.2020.1798315.
Shea, B. J., B. C. Reeves, G. Wells, M. Thuku, C. Hamel, J. Moran, D. Moher, P. Tugwell, V. Welch, E. Kristjansson, and D. A. Henry. 2017. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. British Medical Journal 358:j4008. https://doi.org/10.1136/bmj.j4008.
Steenland, K., and A. Winquist. 2021. PFAS and cancer, a scoping review of the epidemiologic evidence. Environmental Research 194:110690. https://doi.org/10.1016/j.envres.2020.110690.
Woodruff, T. J., and P. Sutton. 2014. The Navigation Guide systematic review methodology: A rigorous and transparent method for translating environmental health science into better health outcomes. Environmental Health Perspectives 122(10):1007–1014. https://doi.org/10.1289/ehp.1307175.
Xie, W., W. Zhong, B. M. R. Appenzeller, J. Zhang, M. Junaid, and N. Xu. 2020. Nexus between perfluoroalkyl compounds (PFCs) and human thyroid dysfunction: A systematic review evidenced from laboratory investigations and epidemiological studies. Critical Reviews in Environmental Science and Technology 1–46. doi: 10.1080/10643389.2020.1795052.
Zare Jeddi, M., R. Soltanmohammadi, G. Barbieri, A. S. C. Fabricio, G. Pitter, T. Dalla Zuanna, and C. Canova. 2021. To which extent are per- and poly-fluorinated substances associated to metabolic syndrome? Reviews in Environmental Health 37(2):211–228. https://doi.org/10.1515/reveh-2020-0144.