7
Revising ATSDR’s PFAS Clinical Guidance
Since the potential for harmful effects of per- and polyfluoroalkyl substances (PFAS) exposure were made known to the public in 2000, clinicians have increasingly needed guidance on advising their communities regarding sources, routes, and effects of exposure, even as significant uncertainty about the health effects of exposure remains (see Chapter 3). The C-8 Medical Panel was the first body to offer clinical guidance regarding PFAS exposure, but the guidance was limited to members of the class action lawsuit (C-8 Medical Panel, 2013).1 In response to this increasing need from clinicians for advice about responding to this environmental hazard, the Agency for Toxic Substances and Disease Registry (ATSDR) published clinical guidance regarding PFAS exposure in December 2019 (ATSDR, 2019).
RECOMMENDATIONS FOR CHANGES TO ATSDR’S CLINICAL GUIDANCE
In accordance with its Statement of Task (see Chapter 1), the committee in this chapter recommends several changes to ATSDR’s guidance to ensure consistency with the conclusions, findings, and recommendations in this report. The Statement of Task specifies three main considerations regarding changes to the guidance. The first two—decision making for PFAS testing and PFAS concentrations informing clinical care of exposed patients—are discussed in Chapter 5; the third—clinical follow-up care specific to PFAS exposure—is addressed in Chapter 6.
The following recommendations illustrate the potential use of the information presented in previous chapters of this report:
Recommendation 7-1: The Agency for Toxic Substances and Disease Registry (ATSDR) should update its PFAS clinical guidance to make it more succinct and accord with the review of PFAS-associated health effects, exposure reduction considerations, PFAS testing recommendations and interpretation, and recommendations for clinical follow-up presented in this report. When describing the health effects of PFAS, ATSDR should avoid using terms typically used to categorize toxicants, such as “endocrine disrupter” or “neurotoxin,” because they are vague and not necessarily clinically meaningful. When discussing the strength of the association between PFAS and a health outcome, ATSDR should use standard categories of association (such as sufficient evidence of an association, limited or suggestive evidence of an association, inadequate or insufficient evidence of an association, and limited or suggestive evidence of no association).
Recommendation 7-2: The Agency for Toxic Substances and Disease Registry (ATSDR) should incorporate a reader-centered approach when developing its guidance, with the knowledge that many different audiences will turn to its clinical guidance document to prepare for discussions with their clinicians. ATSDR should also solicit feedback on the guidance from a variety of stakeholders, such as
___________________
1 See Chapter 1 for a brief overview of the lawsuit, Jack W. Leach et al. v. E.I. du Pont de Nemours & Company (no. 01-C-608 W.Va., Wood County Circuit Court, filed April 10, 2002), and Chapter 6 for details regarding the clinical guidance for class members.
community groups, practicing clinicians, and medical associations. In addition, ATSDR should encourage clinicians to use evidence-based organizational health literacy strategies to support shared, informed decision making; patient-centered care; cultural humility; and accessible language when communicating with patients about potential health risks.
Recommendation 7-3: The Agency for Toxic Substances and Disease Registry (ATSDR) should develop a process for updating its PFAS guidance that adheres to criteria for making guidelines trustworthy, such as being based on a thorough, transparent, unbiased review of the evidence and being developed by a knowledgeable panel of experts free from strong biases and conflicts of interest. A review of the evidence on the health effects of PFAS should be completed by an authoritative neutral party every 2 years, and the clinical guidance should be updated every 5 years or sooner if warranted by the evidence on the health effects of PFAS. Clinicians and members of communities with elevated PFAS exposure should be engaged to inform the problem and review updated guidance.
In addition to these considerations, the committee believed it would be useful to address some more technical aspects of the ATSDR guidance document, including its writing and design and its dissemination and implementation. To this end, the committee referred to existing clinical guidance on PFAS exposure, as well as materials on health literacy and health communication, including the Agency for Healthcare Research and Quality’s (AHRQ’s) Health Literacy Universal Precautions Toolkit (Brega, 2015) and the Centers for Medicare & Medicaid Services’ (CMS’s) Toolkit for Making Written Material Clear and Effective (McGee, 2010). Given the Centers for Disease Control and Prevention’s (CDC’s) long-standing commitment to effective health communication (Donovan, 1995; Gagen and Kreps, 2019; Roper, 1993; Tinker and Silberberg, 1997), the committee decided not to focus on the finer details of these considerations but on their high-level aspects.
The committee reviewed several other materials published by other organizations to aid clinicians in addressing exposure to PFAS and determining options for clinical follow-up. Table 7-1 describes the clinical guidance documents published by the Pediatric Environmental Health Specialty Units (PEHSUs), and PFAS Research, Education, and Action for Community Health (PFAS-REACH) and identifies the strengths of each.
TABLE 7-1 Description of PFAS Clinical Guidance Documents
| Clinical Guidance Document | Description of Content | Strengths of Document |
|---|---|---|
| How to Conduct a Clinical Visit with Patients Concerned About PFAS (PEHSU, 2021) | Includes a numbered list of five strategies for clinicians to use in discussing PFAS exposure with concerned patients and provides some detail on how to apply them. Also provides some references to other materials with further detail. |
Focuses on empathy and shared decision making and the potential need for exposure reduction. Offers advice only for overall approaches to navigating discussions with patients, and directs clinicians to supplementary Pediatric Environmental Health Specialty Units (PEHSUs) and Agency for Toxic Substances and Disease Registry (ATSDR) resources. |
| PFAS Exposure: Information for Patients and Guidance for Clinicians to Inform Patient and Clinician Decision Making: For Clinicians (PFAS-REACH, 2021) | Summarizes clinical services recommended by other trustworthy groups for adults and children with above-average PFAS exposure. Categorizes information by type of service—laboratory tests, exams, or counseling topics |
Accessible summary of material produced by others. Does not claim to provide rigorous analysis of others’ findings. Does provide more information than the PEHSU document in an accessible format. |
WRITING AND DESIGN OF ATSDR’S CLINICAL GUIDANCE
The CMS Toolkit for Making Written Material Clear and Effective provides advice about paying attention to content, organization, writing style, engagement, and motivation (McGee, 2010):
- “Content” relates to what readers want and need to know and whether the information provided is accurate and up to date.
- The “organization” of the written material involves how well the material paces readers by grouping information in meaningful sections.
- “Writing style” refers to whether the text is conversational and uses the active voice, is specific and concrete, and uses familiar and culturally appropriate terms.
- “Engagement and motivation” refers to whether the material has a positive and friendly tone, offers trustworthy information sources, provides relatable statistics, or offers information about how readers can learn more.
The CMS Toolkit also includes “design” principles related to such matters as
- overall design and page layout;
- font, size of print, and contrast;
- headings, lists, and blocks of text;
- use of color;
- photographs and illustrations; and
- tables, charts, and diagrams (McGee, 2010).
As noted above, the committee limited its attention to design intricacies and overall layout, other than noting that the ATSDR guidance document is likely too long for the target audience of clinicians. Best practices for guidance (AAFP, 2021) include providing recommendations that are specific, offer clear direction, and are succinct—principles consistent with comments made to the committee by community members and clinicians (see Appendix B). Incorporating such best practices and using a reader-centered approach will allow ATSDR’s clinical guidance to reach and support as many clinicians (and other community members) as possible.
DISSEMINATING AND IMPLEMENTING ATSDR’S CLINICAL GUIDANCE
Process for Updating the Guidance
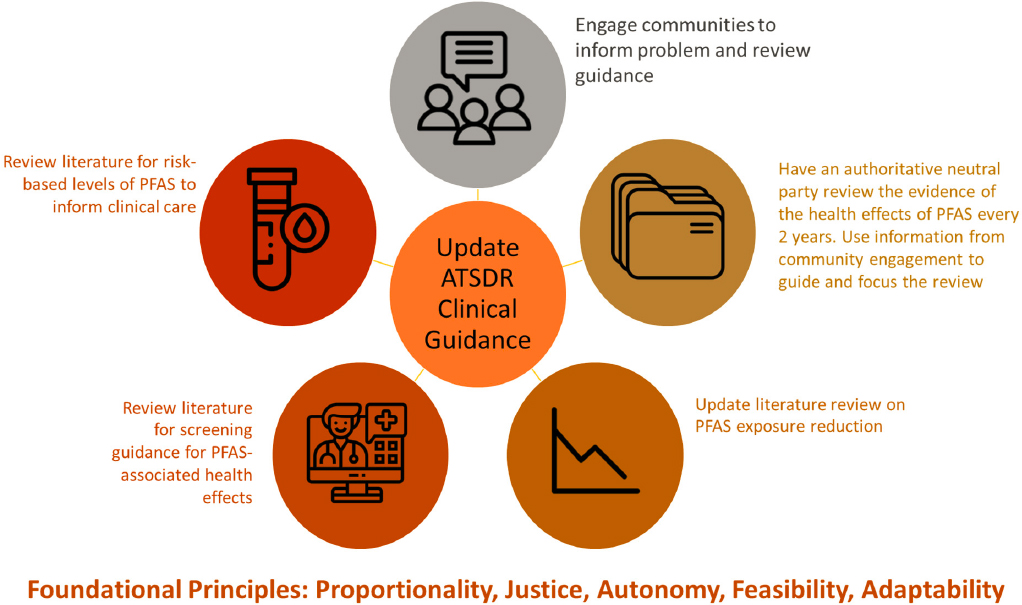
In addition to updates to the ATSDR guidance stemming from this report, the committee proposes the establishment of a process for regularly updating the guidance (see Figure 7-1). The first step in that process is engaging with impacted communities to inform understanding of the issue at hand. It will also be important for ATSDR to update its reviews regarding PFAS-associated health effects, as well as to catalyze future research by identifying gaps in the evidence. This process should include both a review of guidelines issued by other authoritative bodies reflecting decisions about the health effects of PFAS and a review of the epidemiologic literature to identify any new studies that may warrant updating or revising ATSDR’s own authoritative guidance. These reviews should be conducted by a neutral party every 2 years, or sooner if a watershed study, such as a large cohort or nested case-control study, on PFAS exposure and health effects is published before the next review is scheduled. AHRQ, the U.S. Preventive Services Task Force (USPSTF), and other organizations use similar processes that may be informative. One such process includes conducting “living” systematic reviews to continually update an existing review as new evidence becomes available (Elliott et al., 2017). It will be important in this process to incorporate reviews conducted by the U.S. Environmental Protection Agency, the National
Toxicology Program, and other authoritative bodies. The committee proposes that ATSDR revise its guidance within 5 years of its 2021 analysis, consistent with the timelines for updating of the National Guideline Clearinghouse (NGC) and the USPSTF recommendations. The process for updating the literature review should encompass studies on PFAS exposure reduction, including those evaluating behavior change, interventions, or clinical measures, as well as studies on risk-based levels of PFAS to inform clinical care. It will also be important to review recommendations on standard care, as clinical follow-up recommendations would change if, for example, a beneficial screening test existed for a PFAS-associated health outcome. Finally, as noted above, the updating process should incorporate approaches for assembling feedback from clinicians and community members prior to each review. It should also include processes for updated documents to be reviewed by clinicians, in consultation with community members impacted by PFAS (see Figure 7-1 for an overview of this proposed updating process).
Transparency and Trustworthiness
Transparency enhances the trust clinicians and others place in clinical guidance. To advise ATSDR in this regard, the committee turned to the Institute of Medicine’s (IOM’s) standards for developing trustworthy clinical practice guidelines (CPGs) (IOM, 2011). The 2011 IOM report Clinical Practice Guidelines We Can Trust defines CPGs as
statements that include recommendations intended to optimize patient care. They are informed by a systematic review of evidence and an assessment of the benefits and harms of alternative care options. (p. 15)
The definition also includes six attributes required to make CPGs trustworthy, including that they
- are based on a systematic review of the existing evidence;
- are developed by a knowledgeable, multidisciplinary panel of experts and representatives from key affected groups;
- consider important patient subgroups and patient preferences, as appropriate;
- are based on an explicit and transparent process that minimizes distortions, biases, and conflicts of interest;
- provide a clear explanation of the logical relationships between alternative care options and health outcomes, and provide ratings of both the quality of evidence and the strength of recommendations; and
- are reconsidered and revised as appropriate when important new evidence warrants modifications of recommendations (p. 26).
The above definition of and standards for trustworthiness are intended to distinguish CPGs as transparent and methodologically rigorous, evidence-based guidelines as compared with other important forms of clinical guidance, such as expert advice, consensus statements, and practice bulletins. CPGs provide recommendations based on the balance of benefits and harms of different interventions for preventing or treating disease. Although ATSDR may be unable to issue CPGs regarding general PFAS exposure, as the complexity of the data may make a formal systematic review to inform such CPGs difficult, it should still strive to ensure that its clinical guidance reflects these criteria when possible.
CONCLUSION
The need for clear clinical guidance regarding PFAS exposure has only increased, despite the remaining uncertainties regarding the specific health risks of exposure. Yet it is difficult to distill the necessary information for effective clinical practice with respect to PFAS exposure, its possible health effects, and options for shared decision making once exposure has been confirmed. In addition to the distillation of the information itself, moreover, the writing, design, and general implementation of the clinical guidance are as important and as complex as the distillation of the information itself. This chapter offers recommendations that, if implemented, may assist ATSDR in its efforts to offer the guidance needed by clinicians. The committee believes ATSDR can continue to offer up-to-date, useful, and trustworthy clinical guidance by incorporating the updated data on the health effects of PFAS exposure presented in earlier chapters of this report, as well as adhering to the important risk communication and health literacy principles discussed above.
REFERENCES
AAFP (American Academy of Family Physicians). 2021. Clinical practice guidelines. https://www.aafp.org/family-physician/patient-care/clinical-recommendations/definitions/cpg.html (accessed December 28, 2021).
ATSDR (Agency for Toxic Substances and Disease Registry). 2019. PFAS: An overview of the science and guidance for clinicians on per- and polyfluoroalkyl substances (PFAS). https://www.atsdr.cdc.gov/pfas/docs/ATSDR_PFAS_ClinicalGuidance_12202019.pdf (accessed March 25, 2020).
Brega, A. G., J. Barnard, N. M. Mabachi, B. D. Weiss, D. A. DeWalt, C. Brach, M. Cifuentes, K. Albright, and D. R. West. 2015. AHRQ health literacy universal precautions toolkit, second edition. Rockville, MD. AHRQ Publication No. 15-0023-EF. https://www.ahrq.gov/health-literacy/improve/precautions/toolkit.html (acessed July 1, 2022).
C-8 Medical Panel. 2013. Information on the C-8 (PFOA) Medical Monitoring Program screening tests prepared by the medical panel for the C-8 class members. http://www.c-8medicalmonitoringprogram.com/docs/med_panel_education_doc.pdf (accessed January 28, 2022).
Donovan, R. J. 1995. Steps in planning and developing health communication campaigns: A comment on CDC’s framework for health communication. Public Health Reports 110(2):215–217. https://stacks.cdc.gov/view/cdc/64183 (accessed July 1, 2022).
Elliott, J. H., A. Synnot, T. Turner, M. Simmond, E. Akl, S. McDonald, G. Salanti, J, Meerpohl, H. MacLehose, J. Hilton, D. Tovey, I, Shemilt, and J. Thomas, on behalf of the Living Systematic Review Network. 2017. Living systematic review: 1. Introduction—the why, what, when, and how. Journal of Clinical Epidemiology 91:23–30. https://doi.org/10.1016/j.jclinepi.2017.08.010.
Gagen, D. M., and G. L. Kreps. 2019. An examination of the clarity of government health websites using the Centers for Disease Control and Prevention’s Clear Communication Index. Frontiers in Communication 4. https://doi.org/10.3389/fcomm.2019.00060.
IOM (Institute of Medicine). 2011. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press. https://doi.org/10.17226/13058.
McGee, J. 2010. Toolkit for making written material clear and effective. Baltimore, MD: Centers for Medicare & Medicaid Services. https://www.cms.gov/Outreach-and-Education/Outreach/WrittenMaterialsToolkit (accessed July 1, 2022).
PEHSU (Pediatric Environmental Health Specialty Unit). 2021. How to conduct a clinical visit with patients concerned about PFAS exposure. https://deohs.washington.edu/pehsu/sites/deohs.washington.edu.pehsu/files/PFAS%20Clinical%20Visit%20tips%20for%20clinicians_AK_Jan%202021.pdf (accessed June 30, 2022).
PFAS-REACH (PFAS Research, Education, and Action for Community Health). 2021. PFAS exposure: Information for patients and guidance for clinicians to inform patient and clinician decision making—For people in PFAS-impacted communities. https://wordpress.silentspring.org/wp-content/uploads/2021/06/PFAS-REACH-Medical-screening-guidance_PFAS-impacted-communities.pdf (accessed June 30, 2022).
Roper, W. L. 1993. Health communication takes on new dimensions at CDC. Public Health Reports 108(2):179. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1403358.
Tinker, T. L., and P. G. Silberberg. 1997. An evaluation primer on health risk communication programs and outcomes. https://www.atsdr.cdc.gov/risk/evalprimer/index.html (accessed June 28, 2022).






